Abstract
Cancer is a significant cause of death worldwide. It has been suggested that the consumption of flavonoids decreases the risk for cancer by increasing phase II enzymes, such as Nicotinamide Adenine Dinucleotide Phosphate Hydrogen (NAD(P)H) quinone oxidoreductase 1 (NQO1), glutathione S-transferases, and Uridine 5'-diphospho- (UDP)-glucuronosyltransferases that assist in removing carcinogens from the human body. Flavonoids are bioactive compounds found in a variety of dietary sources, including fruits, vegetables, legumes, nuts, and teas. As such, it is important to investigate which flavonoids are involved in the metabolism of carcinogens to help reduce the risk of cancer. Therefore, the objective of this narrative review was to investigate the effects of commonly consumed flavonoids on NQO1 mRNA expression, protein, and activity in human cell and murine models. PubMed was used to search for peer-reviewed journal articles, which demonstrated that selected flavonoids (e.g., quercetin, apigenin, luteolin, genistein, and daidzein) increase NQO1, and therefore, increase the excretion of carcinogens. However, more research is needed regarding the mechanisms by which flavonoids induce NQO1. Furthermore, it is suggested that future efforts focus on providing precise flavonoid recommendations to decrease the risk factors for chronic diseases.
INTRODUCTION
Cancer is the second leading cause of death in the United States, contributing to 608,371 deaths in 2022. 1 The prevalence of cancer is also among the leading causes of death worldwide, and the number of cases per year is expected to rise to 29.5 million by 2040. 2 The National Cancer Institute 2 has emphasized that the incidence of cancer has become a burden to the American society, as it was estimated that in 2020 a total of 1,806,590 new cases were diagnosed. Prostate, lung, and colorectal cancers are the most diagnosed in men, whereas breast, lung, and colorectal cancers account for the most diagnosed in females. About 39.5% of individuals will have a cancer diagnosis during their lifetime. The estimated national expenditure for cancer care has dramatically increased over the years; for example, in 2018 in the United States, $150.8 billion was spent. 2
Epidemiological studies have suggested that diets rich in fruits, vegetables, and whole grains may protect humans from developing chronic diseases such as cancer, cardiovascular disease (CVD), and diabetes. 3 –5 Studies have demonstrated that certain phytochemicals, which are abundant in plants, can induce the expression of phase II enzymes (e.g., NAD(P)H quinone oxidoreductase 1 (NQO1), glutathione S-transferases (GST), UDP-glucuronosyltransferases (UGT), sulfotransferases (SULT), and methyltransferases). These phase II enzymes are present in the liver and other tissues (e.g., intestine), which are responsible for xenobiotic metabolism. The process of xenobiotic metabolism consists of enzymatic reactions that transform chemicals such as carcinogens and drugs into forms that are available for excretion from the body. 6 –9 It is important to understand the pharmacological roles of phytochemicals in xenobiotic metabolism, as humans today are exposed to a variety of carcinogens. 10,11
Electrophiles and oxidants, such as quinone and carbonyl metabolites, are ubiquitous in aerobic organisms as a result from metabolic processes and xenobiotic metabolism (e.g., phase I cytochrome P450 enzyme activities). 12 –14 These reactive intermediates (quinone and carbonyl metabolites) may be produced in the body from exogenous sources including burning of fossil fuels and cigarette smoke (polycyclic aromatic hydrocarbons), urban air and automobile exhaust (halogenated polycyclic aromatic hydrocarbons), high-temperature cooking of meat, fish, and poultry (heterocyclic aromatic amines), the intake of drugs, exposure to pesticides, and other similar activities commonly seen in industrialized nations. 14 –21 For instance, α- and β-unsaturated aldehyde by-products from lipid peroxidation and industrial processes are harmful electrophiles that have the capacity to promote cellular oxidative stress, thereby disrupting DNA, proteins, and plasma membranes. 22 Evidence indicates that frequent cellular exposure to free radicals promotes the synthesis of reactive oxygen species that facilitate the development of chronic diseases such as different types of cancers, CVD, atherosclerosis, inflammation, aging, and neurodegenerative conditions. 13,23 –25 As such, it is beneficial to excrete these reactive intermediates from the body, and therefore, reduce the risk for cancer and other chronic diseases (Fig. 1).
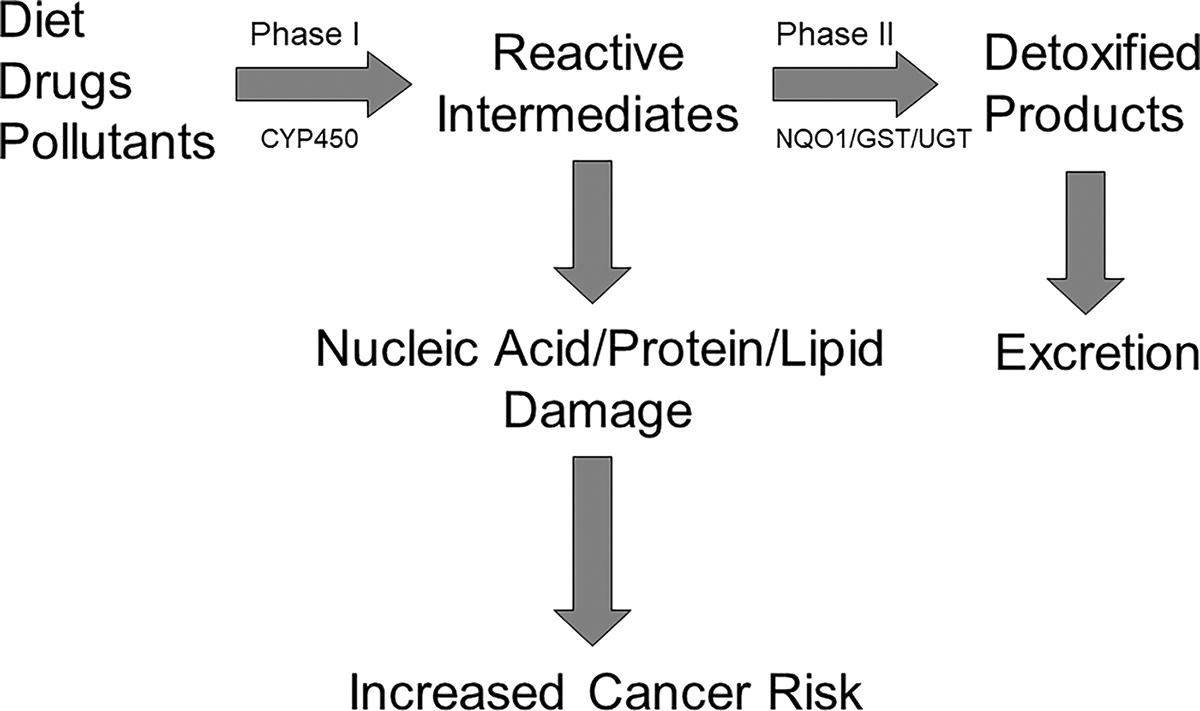
The process by which phase I and phase II biotransformation enzymes contribute to the generation of reactive intermediates and detoxified products. CYP450, cytochrome P450; GST, glutathione S-transferase; NQO1, NAD(P)H quinone oxidoreductase 1; UGT, UDP-glucuronosyltransferase.
Studies have demonstrated that a diversity of compounds such as phytochemicals, endogenous chemicals, therapeutics, environmental agents, and exogenous chemicals modulate the antioxidant or electrophile-response element (ARE/EpRE) gene involved in the transcription of phase II enzymes. 6 For example, some of the flavonoids ubiquitously found in plants have been noted to modulate cell signaling cascades. However, among the 12 subclasses of flavonoids, only 6 have been marked with dietary significance which are anthocyanidins, flavan-3-ols, flavonols, flavones, flavanones, and isoflavones (Fig. 2). 26 Flavonoids are primarily found in fruits, vegetables, legumes, nuts, and teas, as shown in Table 1. 28 –30 Proanthocyanidins, which are dimers, trimers, and oligomers of flavan-3-ols (or flavanols), are found in certain food items, such as apples, berries, red grapes, and cocoa. 29,31
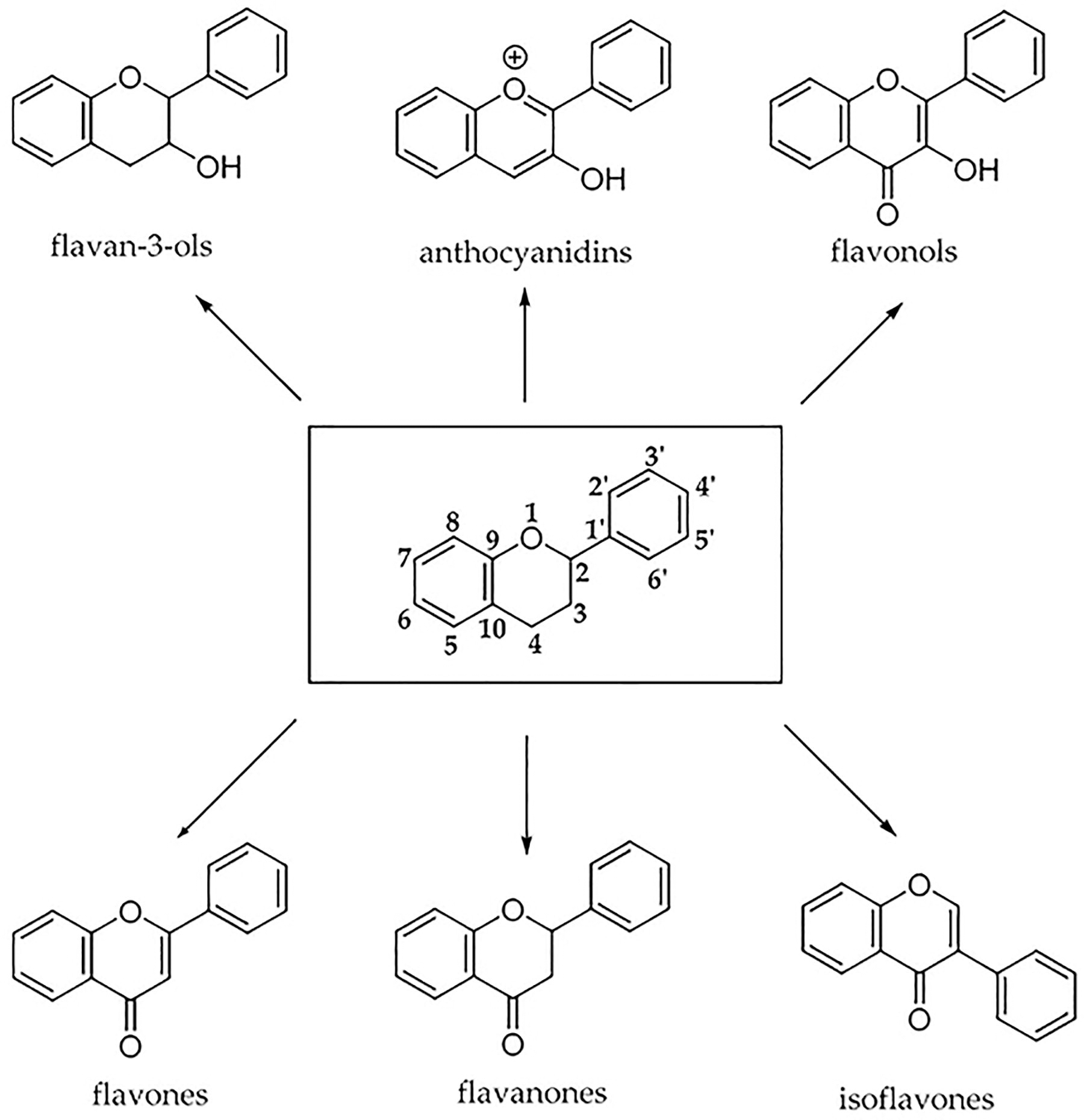
The chemical structures of the six major subclasses of flavonoids. 26
The Content of Flavonoids in Selected Dietary Sources
Studies have shown that the activation of the ARE/EpRE may occur via electrophiles and other compounds (e.g., flavonoids). 32,33 The activation of the ARE/EpRE occurs via nuclear factor erythroid 2-related factor 2 (Nrf2) binding to this response element. The transcription factor Nrf2 is a member of the cap “n” collar subfamily of basic region leucine zipper transcription factors, which is present in the cytoplasm under resting conditions. 33,34 When Nrf2 is found in the cytoplasm, it is normally bound to the cytoskeleton-bound Kelch-like erythroid cell-derived protein with cap “n” collar homology-associated protein 1 (Keap1). 35 The entry of environmental carcinogens or phytochemicals into the cytoplasm promotes the dissociation between Nrf2 and Keap1, thereby allowing Nrf2 to translocate into the nucleus to increase the transcription of phase II enzymes, 25,36 –38 such as NQO1, GST, UGT, and SULT. 6 –8,25,39 –41 Remarkably, Nrf2 has been suggested to increase longevity and healthspan, in part, by increasing NQO1. 42
NQO1, also known as quinone reductase, is a flavin adenine dinucleotide-dependent phase II enzyme that utilizes nicotinamide-adenine dinucleotide phosphate as an electron donor to reduce quinones to hydroquinones, followed by conjugation (e.g., glucuronidation) and/or excretion. This enzyme increases the polarity of a carcinogen and thus facilitates its excretion from the body (Fig. 3). 25,43 –48 Herein, this enzyme supplies the necessary tools for cells to reduce potentially detrimental compounds to less reactive metabolites that may be eliminated via the liver, intestine, kidneys, or lungs. 49 Interestingly, individuals with polymorphisms that reduce NQO1 expression display increased risk for cancer. 45
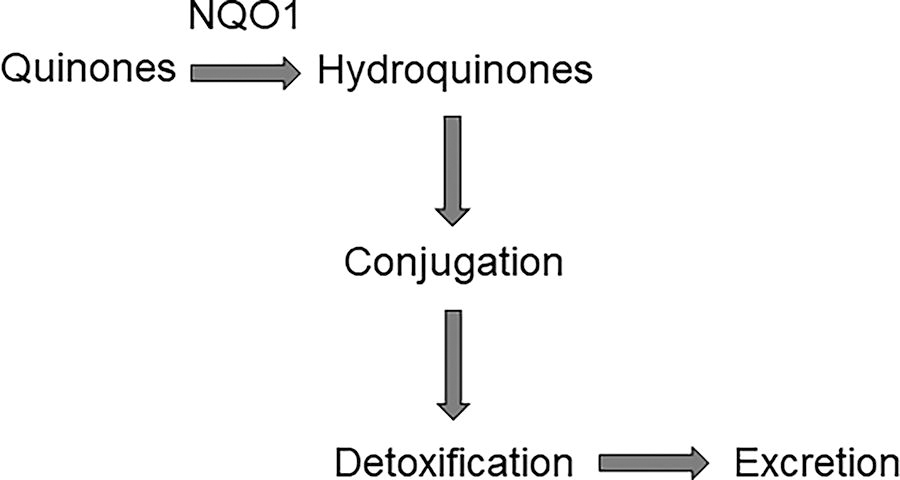
The mechanism by which NQO1 contributes to the excretion of carcinogens. NQO1, NAD
To our knowledge, there has not been a comprehensive review on the effects of flavonoids on NQO1 gene expression and activity in cell and animal studies. Therefore, the objective of this narrative review is to examine the effects of commonly consumed flavonoids on NQO1 gene expression and activity in human cell and murine models. It will be beneficial to discover the flavonoids that increase NQO1, which will contribute to decreasing the risk factors for chronic diseases such as cancer. This review will also provide insights regarding recommended intakes of flavonoids and future perspectives.
MATERIALS AND METHODS
PubMed was used to search for peer-reviewed journal articles that investigated the effects of specific concentrations (e.g., nM, µM, and mg/kg) of commonly consumed individual flavonoids 27 –29 on NQO1 gene expression and activity. The following key words were used: ‘nqo1’ OR ‘nadph quinone oxidoreductase 1’ OR ‘qr’ OR ‘quinone reductase’ AND the following flavonoids: ‘cyanidin’ OR ‘delphinidin’ OR ‘malvidin’ OR ‘pelargonidin’ OR ‘peonidin’ OR ‘petunidin’ OR ‘catechin’ OR ‘theaflavin’ OR ‘thearubigin’ OR ‘isorhamnetin’ OR ‘kaempferol’ OR ‘myricetin’ OR ‘quercetin’ OR ‘apigenin’ OR ‘luteolin’ OR ‘baicalein’ OR ‘chrysin’ OR ‘eriodictyol’ OR ‘hesperetin’ OR ‘naringenin’ OR ‘genistein’ OR ‘daidzein’. Articles that utilized isolated flavonoids in human cell and murine models, with clear descriptions of the methods and results were included in this narrative review. Articles were excluded if the studies used mixtures of various phytochemicals such as extracts, herbs, or concentrates. Studies were also excluded if there were concomitant treatments of drugs, compounds, toxicants, carcinogens, and/or other perturbations (e.g., induction of oxidation, diseases, and injuries). Lastly, articles were excluded if statistical significance was not indicated.
RESULTS
The results of flavonoids on NQO1 expression and activity in human cell and murine studies will be presented in this section and summarized in Tables 2 and 3.
Summary of the Effects of Flavonoids on NQO1 Gene Expression and Activity in Various Cell Types
↑, increase; ↓, decrease; ↔, no change; d, days; h, hours.
Summary of the Effects of Flavonoids on NQO1 Gene Expression and Activity from Various Animal Studies
↑, increase; ↓, decrease; ↔, no change; bw, body weight; d, days; g, grams; h, hours; i.p., intraperitoneal; kg, kilogram; mg, milligram; wk, weeks.
Modulation of NQO1 via anthocyanidins in cell studies
Cyanidin (1, 10, and 100 µM) did not affect NQO1 protein levels in human keratinocytes. 50 In contrast, Li et al. 53 reported an increase of NQO1 protein and mRNA expression by pelargonidin (30 and 50 µM) in mouse skin epidermal cells, while pelargonidin chloride (100 µM) increased NQO1 activity and mRNA in HepG2 cells. 51 Interestingly, however, pelargonidin did not alter NQO1 mRNA and protein levels in human ovarian adenocarcinoma cells at physiological concentrations (up to 2 µM). 52
Modulation of NQO1 via flavan-3-ols in cell and animal studies
Primary human skin fibroblast cells exposed to catechin (20 µM) induced NQO1 mRNA and protein, 56 and epigallocatechin-gallate (10 µM) increased activity in MCF7 cells. 54 In contrast, epigallocatechin-gallate (up to 2 µM) did not affect NQO1 mRNA and protein in primary normal human bronchial epithelial cells 55 and human ovarian adenocarcinoma cells. 52 Hepatic protein levels and mRNA expression of NQO1 were increased after treatment with epigallocatechin-3-gallate [75 mg/kg intraperitoneally (i.p.) injected] in Kunming mice. 78 However, epigallocatechin-3-gallate at 200 mg/kg (i.p. injected) decreased hepatic NQO1 mRNA and protein, 78 and catechin (2 g/kg diet) decreased hepatic activity in rats. 76 There were no changes in lung NQO1 activity in rats administered 20 mg/kg (i.p. injected) of epigallocatechin-3-gallate 77 and in mice given 40 mg/kg body weight (bw) (oral gavage) of catechin. 21
Modulation of NQO1 via flavonols in cell and animal studies
Quercetin (5–40 µM) increased mRNA expression, 58,60 protein, 60 and activity 60 of NQO1 in HepG2 cells. Likewise, NQO1 mRNA was increased by quercetin (10 µM) in human colon carcinoma (Caco-2) cells. 59 Furthermore, NQO1 mRNA and protein increased in human aortic endothelial cells (20 µM quercetin) 62 and in adult mouse microglia cells (up to 10 µM quercetin). 65 Quercetin (15 and 20 µM, respectively) increased NQO1 protein levels in MCF7 57 and human adult retinal pigment epithelial cells (ARPE-19). 64 In agreement with these results, NQO1 activity increased in Hepa-1c1c7 cells (1.25–15 µM quercetin) 61,63 and in MCF7 cells (15 µM quercetin). 57 However, no changes in mRNA and protein were reported in normal primary human skin fibroblast cells treated with up to 20 µM quercetin. 56 Coinciding with these outcomes, no changes in NQO1 mRNA or activity by quercetin (2 g/kg diet) were observed in rats. 76 Lastly, treatment with 10 µM kaempferol increased mRNA expression of NQO1 in Caco-2 cells. 59
Modulation of NQO1 via flavones in cell and animal studies
Apigenin (1.56 and 6.25 µM) induced NQO1 mRNA expression and protein levels in mouse skin epidermal cells (JB6 P+). 66 Likewise, baicalein (10, 20, and 40 µM) increased NQO1 mRNA, 67 whereas protein levels of NQO1 were increased by luteolin (10−2, 10−1, 1, 10, 102, and 103 nM) in HepG2 cells. 68 Luteolin (1 mg/kg bw) increased both liver NQO1 mRNA and protein in ICR mice. 86 Contrary to these results, luteolin (40 mg/kg intragastric gavage) decreased NQO1 protein in the small intestine 82 and liver 83 of C57BL/6 mice. Interestingly, luteolin did not affect liver NQO1 mRNA and protein in C57BL/6 mice (50 mg/kg bw gavage), 88 liver NQO1 protein in Kunming mice (100 mg/kg intragastrically), 84 and kidney NQO1 protein in Wister rats (80 mg/kg intragastrically). 85 Khan et al. 79 reported no significant change in hepatic activity of NQO1 by apigenin (5 mg/kg) in Swiss albino mice. In agreement with this study, chrysin (50 mg/kg bw orally) did not affect jejunum 80 and colon 81 NQO1 activity in rats. In addition, there were no changes in NQO1 mRNA in the cerebral cortex following chrysin treatment (50 mg/kg bw gastric gavage) in rats. 87
Modulation of NQO1 via flavanones in cell and animal studies
Eriodicytol (10, 20, 30, 50, and 100 µM) increased NQO1 protein in ARPE-19 cells. 70 However, naringenin (0.1–10 µM) did not affect NQO1 activity in Hepa-1c1c7 cells 69 and in the brain tissue of rats (50 mg/kg oral gavage). 89
Modulation of NQO1 via isoflavones in cell and animal studies
Genistein was reported to increase NQO1 mRNA expression and activity in human colon cancer cells (0.001–10 µM), 71 as well as mRNA and protein in breast cancer cells (10 µM). 72 Furthermore, genistein increased NQO1 mRNA (25 µM), protein (25 µM), and activity (5 and 25 µM) in Hepa-1c1c7 cells 73 and mRNA (25 µM) in Lymph Node Carcinoma of the Prostate (LNCaP) (human prostate cancer) cells. 74 In agreement with the cell culture studies, genistein (2 g/kg diet) increased NQO1 mRNA and activity in liver tissues of rats. 90 However, genistein (1500 mg/kg diet) did not alter hepatic NQO1 activity in male and female mice, but did increase NQO1 activity in the heart. 91 In contrast, genistein (5 mg/kg bw i.p.) did not impact heart NQO1 protein level in mice. 92 NQO1 mRNA (1, 5, and 25 µM), protein (25 µM), and activity (1, 5, 25 µM) were elevated by daidzein in Hepa-1c1c7 cells. 73 Daidzein (100 nM) was also demonstrated to increase protein levels in human umbilical vein endothelial cells. 75 On the other hand, daidzein (0.001–10 µM) did not alter NQO1 activity and mRNA in human colon cancer cells. 71 Contrary to these results, daidzein (1500 mg/kg diet) increased hepatic and kidney NQO1 activities in male and female mice, but did not affect heart NQO1 activity. 91
DISCUSSION
The effects of flavonoids on NQO1 expression and activity
The results from the presented studies indicate that multiple flavonoids modulate the phase II detoxification enzyme NQO1, which suggests that some flavonoids may decrease the risks for chronic diseases that are perpetuated by oxidative stress and toxicity. However, many studies utilized high concentrations of flavonoids, and therefore, these results will likely not apply to typical human consumption of these flavonoids. The physiological concentrations of flavonoids in the blood are typically <1 µM and up to 5 µM. 93 –96 Certain studies in this review successfully demonstrated increasing NQO1 with concentrations of ≤1 µM and up to 5 µM. For example, quercetin, 58,61 –63,65 apigenin, 66 luteolin, 68 genistein, 71,73 and daidzein 73,75 increased NQO1 at physiological concentrations in human cells (HepG2 cells, aortic endothelial cells, colon cancer cells, and umbilical vein endothelial cells) and in murine cells (adult mouse microglia cells, Heap-1c1c7 cells, and skin epidermal cells). In contrast, certain flavonoids did not modulate NQO1 at physiological concentrations in other studies, such as cyanidin, 50 pelargonidin, 52 epigallocatechin-gallate, 52,55 catechin, 56 quercetin, 56 naringenin, 69 and daidzein 71 in human cells (keratinocytes, ovarian adenocarcinoma cells, primary normal bronchial epithelial cells, normal primary skin fibroblast cells, and colon cancer cells), and in murine cells (Hepa-1c1c7 cells).
Previous publications suggest that flavonoid treatments should be below 2 g/kg diet or 200 mg/kg body weight to reflect typical concentrations in the blood by consuming a flavonoid-rich diet. 90,97 –100 Consumption of luteolin, genistein, and daidzein increased NQO1 in murine models (ICR and Swiss Webster mice) at physiological concentrations. 86,91 On the other hand, certain flavonoids did not affect NQO1 following consumption, such as catechin, 21 apigenin, 79 chrysin, 80,81,87 luteolin, 84,85,88 and naringenin 89 in Swiss albino, C57BL/6, and Kunming mice, as well as in Albino Wistar, Sprague Dawley, Wistar, and Swiss albino rats. Interestingly, luteolin decreased NQO1 in other studies involving C57BL/6 mice. 82,83
Digestion, absorption, and metabolism of flavonoids
Generally, flavonoids are found in dietary sources as glycosides and thus are attached to one or more monosaccharides. 101,102 In addition, proanthocyanidins (flavanol polymers) exist as dimers, trimers, and oligomers. 95 Unfortunately, the bioavailability of flavonoids is typically low and varies with the respective flavonoid subclasses. 95,103 –105 Interestingly, isoflavones display the highest bioavailability, whereas anthocyanins show the lowest bioavailability among the flavonoid subclasses. 93 Cells in the small intestine via enzymes (e.g., lactase phlorizin hydrolase) can hydrolyze these glycosides into their corresponding aglycones. Similarly, the intestinal microbiota can also hydrolyze the sugar moiety of glycosides and yield aglycone forms that may be readily absorbed by the colon. Additionally, intestinal microbiota can metabolize these aglycones into low molecular weight catabolites such as phenolic or aromatic acids which may become bioavailable. 102,106
Once the aglycones enter intestinal epithelial cells, they undergo phase I enzyme metabolism (e.g., cytochrome P450 enzymes) and, subsequently, phase II enzyme metabolism (e.g., NQO1, GST, UGT, SULT, and methyltransferases), which generates conjugated metabolites (e.g., glucuronides and sulfates). After intestinal conjugation occurs, these molecules reach the liver whereby phase II enzymes may continue biotransformation (e.g., methylation, sulfation, and glucuronidation) to yield multiple conjugates or metabolites; hence, biotransformation results in more polar compounds that can perhaps target tissues or be excreted by the body via urine or bile. 95,102 Thus, blood systemic circulation and urine show low concentrations of aglycones—ranging from absence to >70% (for epigallocatechin gallate). 107,108 Instead, flavonoids are primarily found as metabolites, such as methyl, sulfate, and glucuronide forms, with the majority, in general, existing as glucuronide conjugates. 102,109 The chemical structure of flavonoids is important since the clinical relevance of these compounds will be determined by their capacity to be converted into metabolites (e.g., daidzein → equol), as it has been shown that the biological activities of certain metabolites differ from their aglycone forms. Moreover, phase II metabolism differs between rodents and humans. 95,102,110 –114 As such, these characteristics should be considered when interpreting data from human, animal, and cell studies.
The mechanisms by which flavonoids increase NQO1 expression
During normal cellular conditions or periods of oxidative stress, the gene expression of NQO1 is regulated by the antioxidant response element (ARE) also known as the electrophile response element (EpRE). 25 Although multiple transcription factors (e.g., Jun, Fos, Fra, Maf, and Raf) may interact with the ARE promoter region, the Nrf2 transcription factor is believed to be the major regulator of cytoprotective responses to oxidative stress. 41,115,116 Under homeostatic conditions, Keap1 regulates Nrf2 in the cytoplasm by binding two molecules of Keap1 to Nrf2. The Nrf2-Keap1 complex is connected to an E3 ubiquitin ligase complex via an adapter protein called Cullin 3. 33,41,117,118 Under conditions of oxidative stress or the presence of electrophiles, the reactive cysteine residues of Keap1 are oxidized, thereby resulting in the dissociation of the Nrf2-Keap1 complex. Subsequently, Nrf2 is released in the cytoplasm and translocates to the nucleus to form a heterodimer with members of the small musculoaponeurotic fibrosarcoma (sMaf) protein family (MafF, MafG, and MafK) that bind in a sequence-specific manner to the ARE core promoter region, whereby this action results in the gene transcription of phase II enzymes (e.g., NQO1). 25,33,118
In addition, there are endogenous activators of the Nrf2/ARE pathway that stimulate phosphorylation of Nrf2 and therefore release Nrf2 from Keap1. 119 Most of these identified activators are protein kinases, such as mitogen-activated protein kinases (MAPK), c-Jun N-terminal kinase (JNK), extracellular signal-regulated protein kinase (ERK), p38, protein kinase-like endoplasmic reticulum-resident kinase (PERK), AMP-activated protein kinase (AMPK), protein kinase C (PKC), phosphatidylinositol 3-kinase (PI3K), and Akt. 33,120 These kinases are regulators of downstream transcription factors involved in carcinogenesis mechanisms such as differentiation, proliferation, apoptosis, and tumor induction. 121 Moreover, they are involved in the activation of ARE genes that mediate the transcription of phase II enzymes. 122
It has been demonstrated that flavonoids (and other phytochemicals) can modulate the induction of phase II enzymes. Evidence shows that flavonoids increase the expression and activity of NQO1 via Nrf2 binding to the ARE. However, the mechanisms by which flavonoids disrupt the Nrf2-Keap1 complex and promote the translocation of Nrf2 into the nucleus are not yet fully understood. 33 A recent study demonstrated that anthocyanins interact with the BTB domains in Keap1 and thus may “modify the interaction of this protein with Nrf2.” 123 In addition, certain flavonoids (e.g., rutin) can modify Keap1 cysteine residues via oxidation and alkylation, thereby stimulating the Nrf2/ARE pathway. 123,124 Other studies have shown that flavonoids (e.g., apigenin) regulate Nrf2 through epigenetic mechanisms. 66 Interestingly, certain flavonoids (e.g., quercetin, kaempferol, and apigenin) at high concentrations (0.5–60 µM) can induce EpRE-mediated gene transcription in Hepa-1c1c7 cells. Therefore, it seems that the pro-oxidant activities of selected flavonoids (e.g., after donating electrons via antioxidant processes) can induce the EpRE and thereby increase NQO1. 125 However, other flavonoids may display beneficial effects at low concentrations, whereas they exhibit harmful effects at high concentrations via various mechanisms. 33 Some of the endogenous activators may also be activated by certain flavonoids. Furthermore, various flavonoids have been demonstrated to increase Nrf2 mRNA and protein levels and Nrf2 nuclear translocation—as well as decrease Nrf2 ubiquitination and degradation. 33,67,68 However, additional research is needed to elucidate the precise molecular mechanisms by which flavonoids (and their concentrations) activate, stabilize, and translocate Nrf2 and thus increase phase II enzymes (e.g., NQO1) (Fig. 4). 33
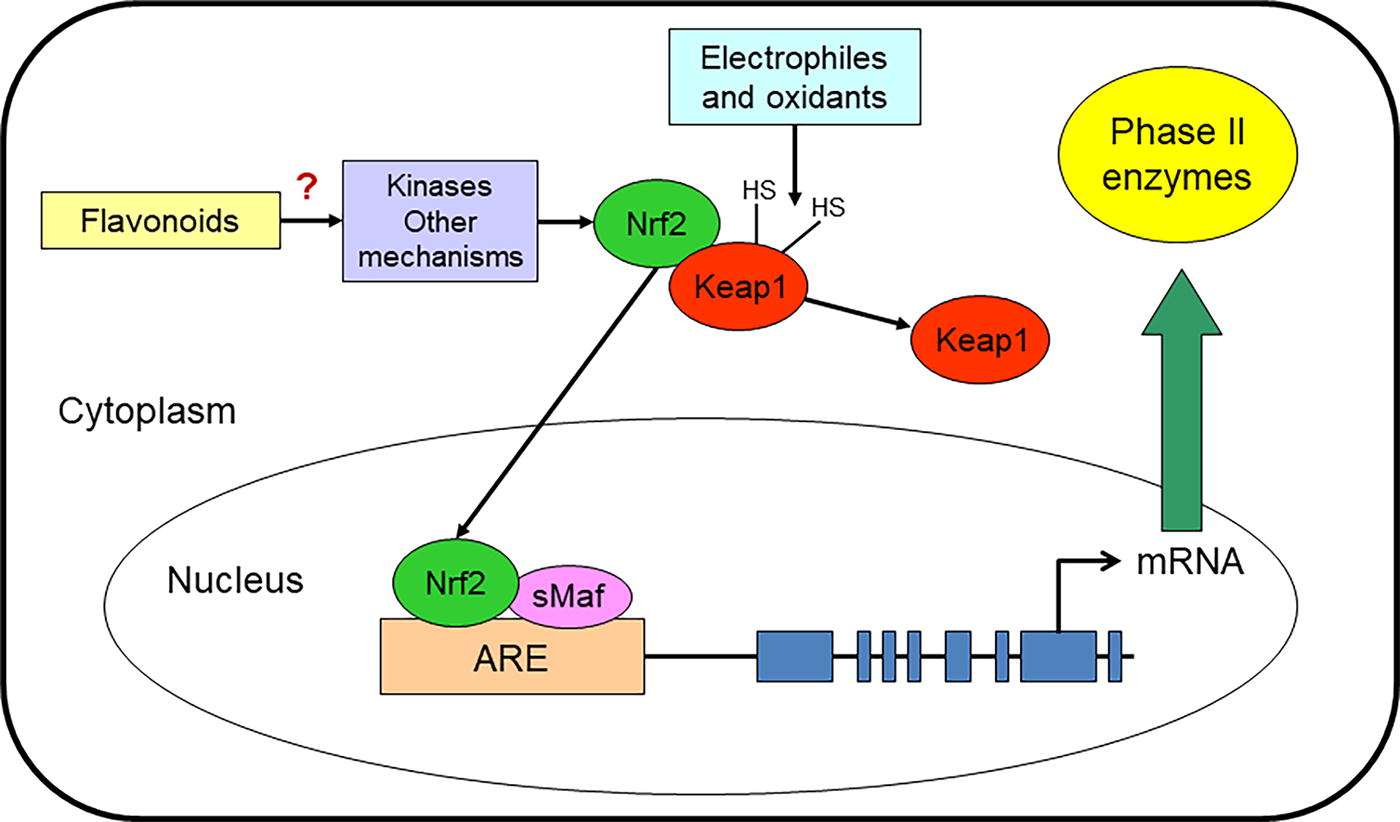
The proposed mechanisms by which Nrf2 increases the gene expression of phase II enzymes. It is thought that electrophiles and oxidative stress oxidize the cysteine residues of Keap1, which causes Keap1 to dissociate from Nrf2. Additionally, protein kinases (and other possible mechanisms) phosphorylate Nrf2, which allows Nrf2 to translocate to the nucleus. Subsequently, Nrf2 binds to sMaf, thereby forming a heterodimer that binds to the ARE. This binding, in part, increases gene expression, and therefore, protein translation of phase II enzymes. ARE, antioxidant response element; Keap1, Kelch-like ECH-associated protein 1; Nrf2, nuclear factor erythroid 2-related factor 2; sMaf, small musculoaponeurotic fibrosarcoma.
Recommendations for flavonoid consumption
It has been estimated that Americans consume a daily mean intake of ∼200 to 400 mg/day of flavonoids, whereas worldwide mean consumption of flavonoids is around 400 mg/day. 126 –131 It was reported that intake of flavonoids (500 mg/day) was inversely associated with cancer mortality. 132 Additionally, the COSMOS trial demonstrated that consumption of a cocoa extract supplement (500 mg/day of flavanols) decreased CVD death by 27%. 133 Interestingly, there are lower incidences of breast, prostate, and other cancers in Asian countries in which the daily intake of isoflavones is ∼25–50 mg, whereas in Western countries, the daily intake of isoflavones is <3 mg. 95,134 –140
Chun et al. 126 reported the major dietary sources of flavonoids in the United States come from tea, citrus fruit juices, wine, and citrus fruits. It was noted that teas are the most important source of flavonoids among Americans, especially for flavan-3-ols and flavonols which provide 167 mg of daily flavonoid intake. Common sources of quercetin (a flavonol) in a Western diet include teas, red wine, fruits, and vegetables and is available as dietary supplements in doses ranging from 200 to 1200 mg. 141
Currently in the United States, there are no dietary reference intakes available for flavonoids or other bioactive food components, as these molecules have not been established as essential nutrients. However, it is of importance to be able to establish recommended intakes for flavonoids, given that the scientific literature has demonstrated that these bioactive compounds can help prevent the onset of chronic diseases. It is recommended to engage in more research on this topic through more randomized controlled trials and observational, animal, and in vitro studies. It is thought that by establishing recommended intakes for bioactive food components, individuals will possess increased motivation to consume more flavonoid-rich fruits and vegetables. 142,143
It has been a challenge, however, to provide specific flavonoid recommendations to decrease the risk factors for chronic diseases due to different study designs and dietary assessment methods, limited data, metabolic differences, flavonoid bioavailability differences, toxicity issues, and formation of flavonoid metabolites. 94,143 –145 Interestingly, however, a recent review of randomized controlled trials and cohort studies suggested that flavanol consumption of 400–600 mg/day provided “moderate evidence supporting cardiometabolic protection.” Furthermore, it was emphasized that this guideline is based on foods rather than flavanol supplements. 145 Additional research on the health-promoting effects of phytochemicals (e.g., flavonoids) is necessary to understand the specific benefits of phytochemicals and to provide access of this information to the public.
Future studies and perspectives
Future studies need to be conducted to provide the appropriate dietary recommended intakes of flavonoids in humans. More clinical trials will help to understand how the health-promoting effects of flavonoids may differ between individuals due to the following characteristics: population differences, age, sex, dietary patterns, drug interactions, genotypes, and the intestinal microbiome. 94,102,143,145 It is worth noting that future in vitro and in vivo studies work with flavonoid concentrations of physiological relevance to be able to better understand the mechanisms by which flavonoids affect NQO1 gene expression and activity. In addition, it is important to understand how other nutrients, food components, the microbiome, and genetics may interact with the bioavailability of flavonoids. Furthermore, researchers should investigate the effects of flavonoid metabolites to determine if they affect NQO1 gene expression and activity and cardiometabolic health—as well as to identify appropriate biomarkers for flavonoid consumption. 94,102,114,143,145 Lastly, it is also important to understand the levels of toxicity for these compounds, 143 as some companies distribute these flavonoids as dietary supplements.
CONCLUSIONS
This narrative review demonstrates that certain flavonoids modulate NQO1 gene expression and activity. Flavonoids that increased NQO1 at physiological concentrations include quercetin, apigenin, luteolin, genistein, and daidzein. It is important to consider the relevance of using concentrations of flavonoids within a physiological range, while contemplating the adverse effects of high concentrations. To emphasize, future animal and cell studies should consider using doses that are applicable to a human diet. Additionally, more randomized controlled trials need to be done, which will help to provide specific dietary flavonoid recommendations for humans. However, it is evident that specific flavonoids can induce NQO1 gene expression and activity by promoting the translocation of Nrf2 into the nucleus and, subsequently, to the ARE promoter region. The reviewed studies suggest that selected flavonoids increase NQO1 and therefore may decrease the development and progression of chronic diseases such as cancer and CVD.
Footnotes
AUTHORS’ CONTRIBUTIONS
E.B.F. supervised the research and writing processes. G.P.B. and E.B.F. conceptualized and visualized the article. G.P.B. performed the initial investigation and methodology. G.P.B. wrote the original draft. E.B.F. reviewed and edited the original draft. E.B.F. created Table 1. G.P.B. and E.B.F. created Tables 2 and 3. E.B.F. created Figures 1, 3, and ![]() .
.
AUTHOR DISCLOSURE STATEMENT
The authors declare no conflict of interest.
FUNDING INFORMATION
The authors used no funding for this research.
