Abstract
Tumor necrosis factor-α (TNF-α) plays an important role in inflammatory processes. This study examined the effects of natural eggshell membrane (NEM®) (ESM Technologies, LLC, Carthage, MO, USA) on interleukin (IL)-2, IL-4, IL-6, IL-10, interferon-γ (IFN-γ), and TNF-α cytokine production by 4-day peripheral blood mononuclear cell (PBMC) cultures exposed to serial dilutions of either an aqueous extract of natural eggshell membrane (NEM-AQ) or NEM subjected to in vitro digestion (NEM-IVD). The effects on cytokine production were also assessed in the presence of phytohemagglutinin (PHA) and pokeweed mitogen (PWM) where exposure to NEM-AQ resulted in reduced levels of proliferation and statistically significant effects on IL-6, IL-10, IFN-γ, and TNF-α cytokine production. NEM-AQ reduced levels of IL-6, IL-10, IFN-γ, and TNF-α in cultures exposed to PHA. In cultures containing PWM, NEM-AQ reduced production of IL-10 and at the highest dose tested increased IL-6 and decreased TNF-α cytokine levels. NEM-IVD, at the two lowest concentrations of product, significantly reduced TNF-α production by PBMC cultures exposed to PWM compared with the in vitro digest control or native NEM. Taken together, these results suggest that NEM-AQ can influence signaling events in response to the T cell-specific mitogen PHA as well as to the mitogen PWM that require cellular cross-talk and that these effects may be partially mediated through a reduction in level of the pro-inflammatory cytokine TNF-α. The suppression of TNF-α production in the presence of NEM-IVD is promising for the use of NEM as a consumable anti-inflammatory product.
Introduction
T
In contrast, nutraceutical products widely used for joint health include glucosamine, chondroitin, and hyaluronic acid, thus ignoring a multifaceted action of complex natural products. Even the spotlight on hyaluronic acid seems to limit its focus on replenishing the synovial fluid and on stimulating chondrocytes to produce more hyaluronic acid, thus ignoring the many complex ways that hyaluronic acid can modulate cells and their behavior.
Natural eggshell membrane (NEM®) (ESM Technologies, LLC, Carthage, MO, USA) is a novel dietary supplement that has been shown in several human trials to be a clinically effective treatment for pain and stiffness associated with joint and connective tissue disorders, particularly osteoarthritis. 5,6 Eggshell membrane is primarily composed of fibrous proteins such as collagen type I. 7 However, eggshell membranes have also been shown to contain glycosaminoglycans, such as dermatan sulfate, chondroitin sulfate, 8 and hyaluronic acid, 9 and sulfated glycoproteins, including hexosamines such as glucosamine. 10 NEM contains up to about 5% of these various components, the unique combination of which may explain its biological activity. To further this understanding, a cytokine profile was determined from NEM-treated lymphocytes in cell culture.
The purpose of this study is to evaluate anti-inflammatory and immunomodulatory effects of NEM as well as its in vitro digest in a select series of human cell-based in vitro assays, in preparation for more comprehensive evaluations in vitro and in vivo.
Materials and Methods
Reagents
Phosphate-buffered saline (PBS) (pH 7.4), RPMI-1640 culture medium, fetal calf serum,
Preparation of NEM for in vitro bioassays
The NEM powder was reconstituted in physiological saline and allowed to rehydrate for 1 hour at room temperature. Solids included insoluble calcium carbonate from eggshell and were removed by centrifugation at 900 g for 10 minutes. The liquid was filtered through a sterile cellulose acetate syringe filter (pore size, 0.22 μm). This filtrate corresponded to a stock solution of 100 g/L product (same concentration as the in vitro–digested NEM [NEM-IVD], described below). This aqueous preparation is designated NEM-AQ.
Preparation of an in vitro digest of NEM
The in vitro digestion of NEM was performed according to methods published in the literature 11 –13 (see also Fig. 4). In brief, 3.75 g of NEM powder was added to 30 mL of PBS and shaken at room temperature for 1 hour. Following the 1-hour incubation, the sample was spun at 900 g for 10 minutes, and the aqueous solution was removed from the solids and sterile-filtered with a cellulose acetate filter (pore size, 0.22 μm). HCl (1 M) was then added until the solution reached pH 2.0. Porcine pepsin was then added at a concentration of 1.3 mg/mL, and the sample was left at 37°C for 60 minutes with shaking (to simulate digestion in the stomach). Next, sodium bicarbonate (NaHCO3) was used to increase the pH of the solution to 5.8 (irreversibly inactivating pepsin), and pancreatin (0.175 mg/mL) and porcine bile salts (1.1 mg/mL) were added to simulate intestinal digestion. The pH was then adjusted to 6.5, and the mixture was left at 37°C for 1 hour. The final volume of the digest was adjusted with saline to give a final concentration of NEM of 100 g/L. Following this in vitro digestion, the sample was centrifuged through a 10-kDa cutoff filtration spin column to remove the enzymes from the digested product. This filtration step was necessary to avoid the presence of digestive enzymes in the downstream treatment of cells with product. This step also avoided the use of enzyme inhibitors that potentially could have direct effects on cell signaling in downstream cell-based assays. The liquid after in vitro digestion and size-exclusion filtration is designated NEM-IVD.
As the control, saline alone (negative control) was simultaneously subjected to the in vitro digestion protocol as described above. This was an important control to determine whether any bile salts or breakdown products from the enzymes themselves have biological activity. This PBS control is designated as PBS-IVD.
SDS-PAGE
SDS-PAGE was performed to compare crude NEM-AQ, NEM-IVD, and the PBS-IVD control. Samples were denatured by boiling for 3 minutes in 1×Laemmli buffer and separated by gel electrophoresis through a 4–15% polyacrylamide Tris-HCl gel using Protein Plus dual color molecular weight standards for reference. Silver staining was performed in order to visualize proteins, and an image of the stained gel was captured with a Canon (Lake Success, NY, USA) PowerShot SD430 digital camera.
Purification of peripheral blood mononuclear cells
Healthy human volunteers between the ages of 20 and 50 years served as blood donors after written informed consent was obtained, as approved by the Sky Lakes Medical (Klamath Falls, OR, USA) Center Institutional Review Board. Isolation of peripheral blood mononuclear cells (PBMCs) was performed as previously described. 14 PBMCs were used to establish lymphocyte cultures for the measurement of cytokine production.
Cytokine production by 4-day PBMC cultures
Freshly purified PBMCs were resuspended in RPMI 1640 medium supplemented with 10% fetal bovine serum,
Th1/Th2 cytokine profile
The cytokines interleukin (IL)-2, IL-4, IL-6, IL-10, tumor necrosis factor-α (TNF-α), and interferon-γ (IFN-γ) were quantified in the 4-day PBMC culture supernatants using a flow cytometry cytometric bead array (CBA) kit (CBA human Th1/Th2 cytokine kit II, BD Biosciences) that allowed the determination of the levels of all six cytokines simultaneously. Samples were tested in duplicate following the manufacturer's instructions, data were acquired with a FACSCalibur™ flow cytometer (Becton-Dickinson, San Jose), and the analysis was performed with FlowJo software (TreeStar Inc., Ashland, OR, USA).
Statistical analysis
Statistical significance was tested using Student's t test performed with the program Microsoft (Redmond, WA, USA) Excel. All P values were two-sided and were considered significant when P<.05. Only statistically significant P values are reported.
Results
Lymphocyte proliferation assay
The lymphocyte proliferation assay evaluates whether a test product alters lymphocyte responsiveness to known signals such as mitogens. If any change in the proliferative response to known mitogens is seen in cells pretreated with test product, this is a good indication that the product has immunomodulatory effects and justifies further in-depth work on T and B lymphocyte signaling and activation.
Freshly purified human PBMCs were cultured for 4 days in the absence versus presence of serial dilutions of test products. Three parallel sets of cultures were established, where one tested the direct effect of test product on lymphocyte proliferation, and the two others examined the possible interference with response to the mitogen PHA or PWM. PHA produces a cleaner signal as it strictly induces proliferation of T lymphocytes, but PWM represents a more physiological signal mimicking the cellular interactions between monocytes/macrophages and T and B lymphocytes that occur in lymphoid tissue; therefore it is beneficial to test both in parallel. Positive controls included cells treated only with a mitogen in the absence of test product. No direct effects of product on lymphocyte proliferation were observed (data not shown). In the presence of PHA (Fig. 1A) and PWM (Fig. 1B), statistically significant decreases in proliferation of 30% and 15%, respectively, were seen with all three dilutions of NEM-AQ, indicating that pretreatment of PBMCs with NEM-AQ altered their response to subsequent signals.
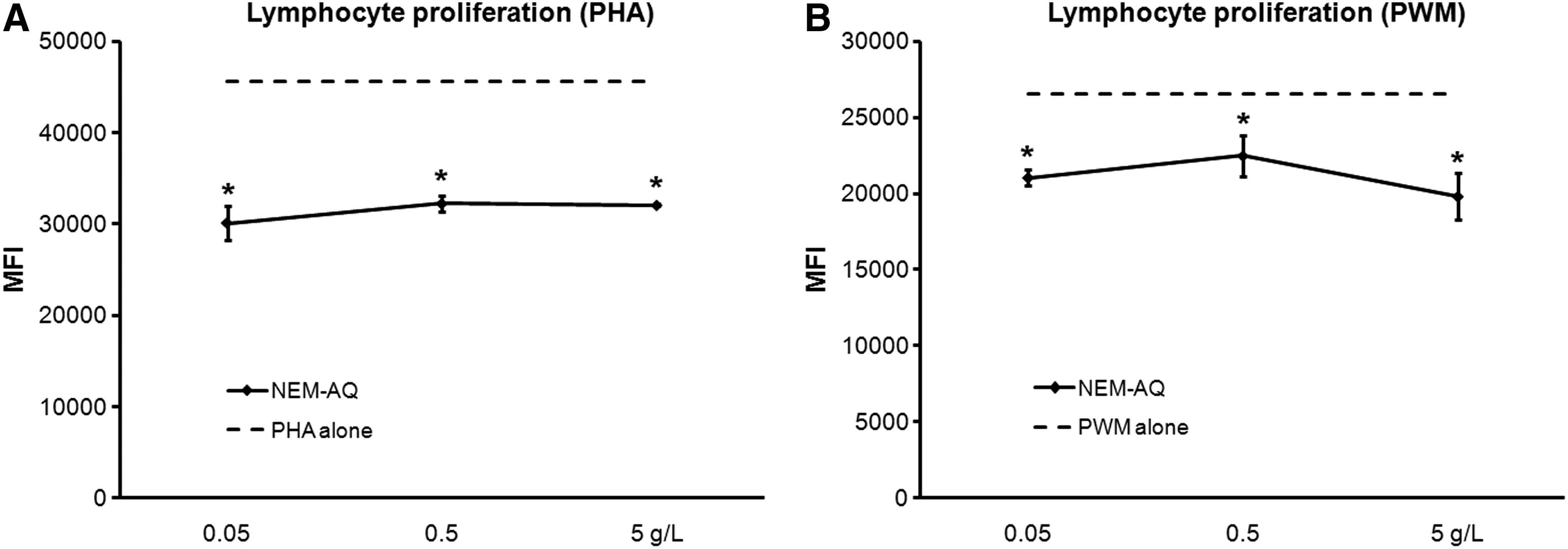
Effect of aqueous extract of Natural Eggshell Membrane (NEM) (NEM-AQ) on lymphocyte proliferation in the presence of
Changes in Th1/Th2 cytokine levels
Supernatants were collected from 4-day cultures where PBMCs were exposed to test products in the absence versus presence of the mitogens PHA and PWM and analyzed for the panel of Th1/Th2 cytokines IL-2, IL-4, Il-6, IL-10, IFN-γ, and TNF-α, using a CBA for flow cytometry. No statistically significant changes in cytokine production occurred in unstimulated cultures, whereas statistically significant changes in the cytokines IL-6, IL-10, IFN-γ, and TNF-α occurred in cultures that contained PHA (Fig. 2) or PWM (Fig. 3).
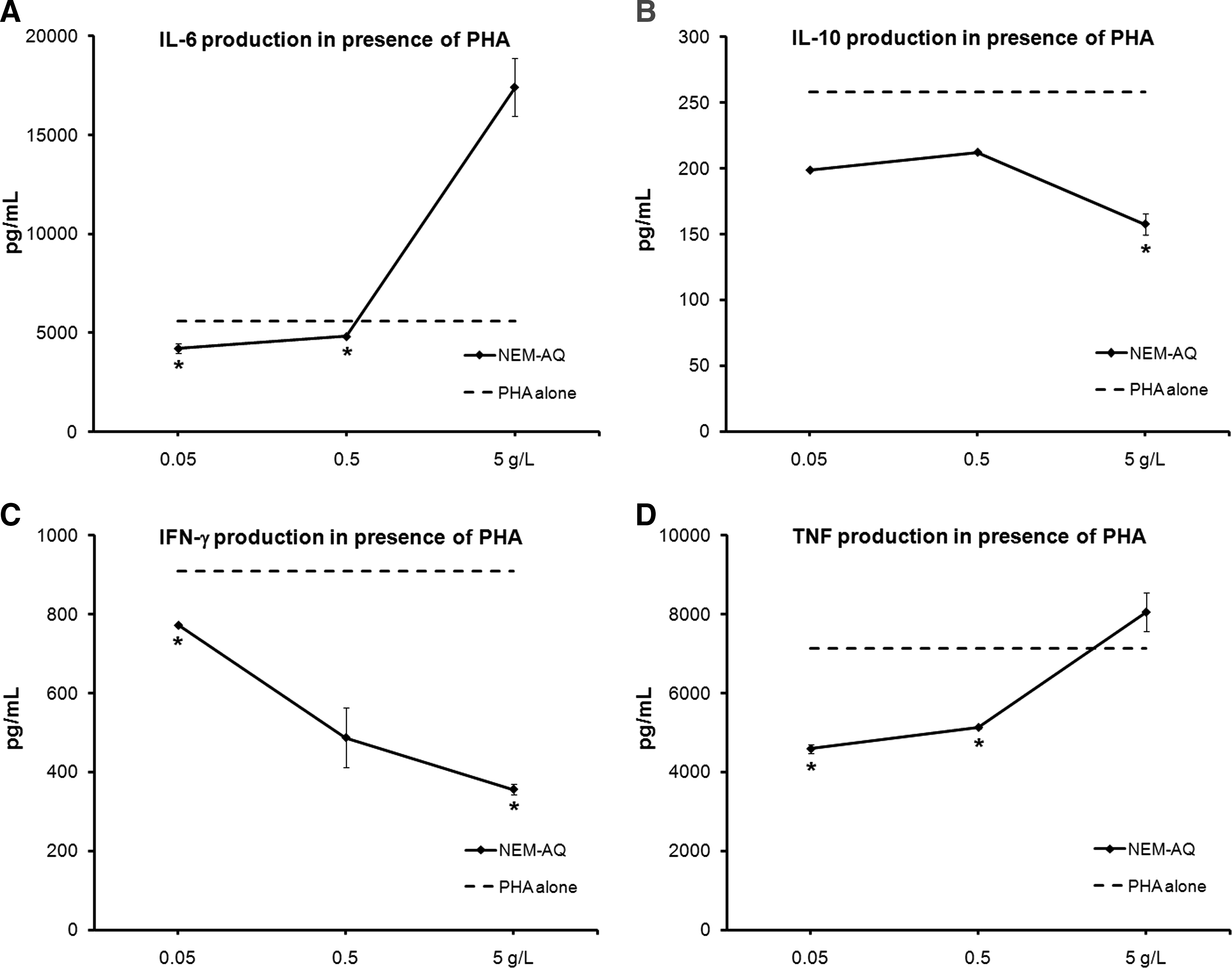
Effects of NEM-AQ on production of the cytokines
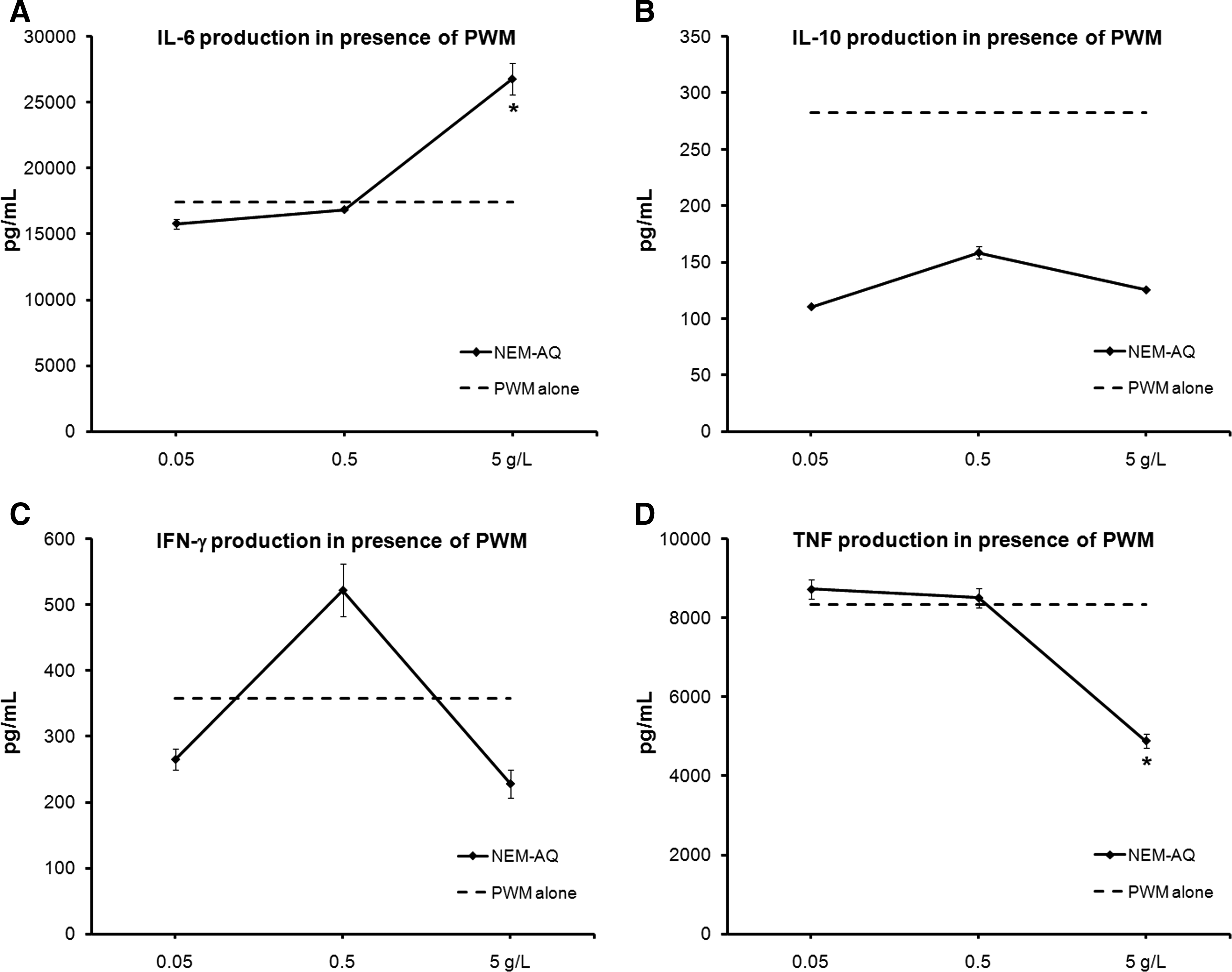
Effects of NEM-AQ on production of the cytokines
Cultures exposed to serial dilutions of NEM-AQ showed a biphasic response, including a strong increase of IL-6 at the highest dose tested (5 g/L) in the presence of both PHA and PWM, a decrease at the lower concentrations for PHA, and no effect at lower doses for PWM. Levels of IL-10, IFN-γ, and TNF-α decreased in the presence of all three doses of NEM-AQ in the presence of PHA. Effects in the presence of PWM were only seen at higher doses. The reduction in IFN-γ production in the presence of PHA was over 60% at the highest concentration of NEM-AQ tested. At the 0.05 g/L dose, a 35% reduction in TNF-α levels was seen, indicating a strong anti-inflammatory effect of NEM-AQ on TNF-α production at lower concentrations in the presence of the T-cell mitogen PHA.
In vitro digest
An in vitro digest as outlined in Figure 4 was performed on NEM-AQ as well as a saline control (PBS-IVD), and the resulting material was subjected to SDS-PAGE and silver stain detection in parallel with NEM-AQ. This analysis showed a reduction of high-molecular-weight material in the NEM-IVD sample (Fig. 5) compared with the NEM-AQ sample, whereas the PBS-IVD sample did not show any detectable protein.
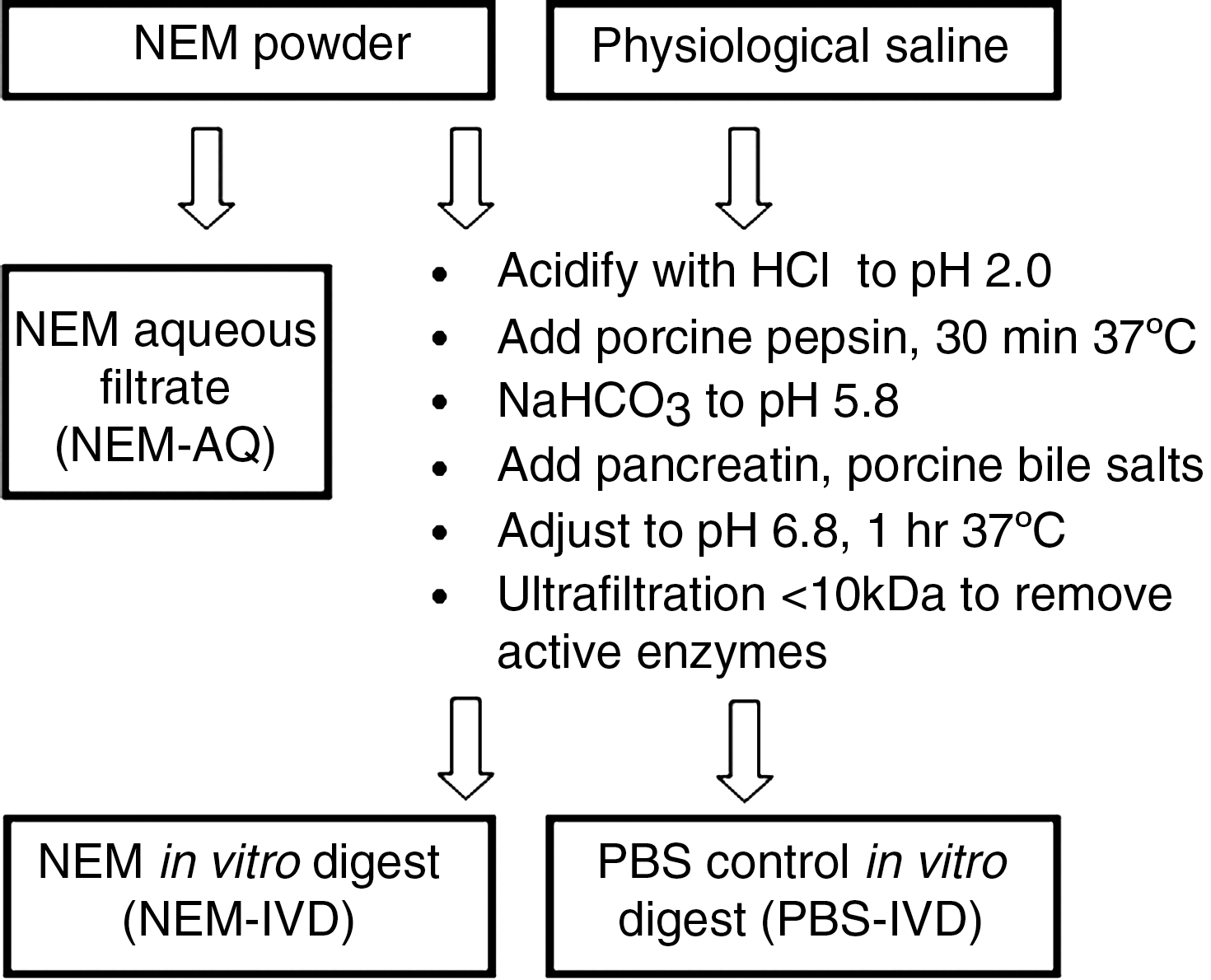
Diagram outlining the in vitro digestion procedure. Based on methods published in the literature, 11 –13 a stepwise process was performed that incorporated digestive enzymes derived from pig (porcine) and pH adjustments in order to mimic the digestive processes occurring in the stomach and small intestine. The final digested product was returned to physiological pH and subjected to size-exclusion centrifugation using a 10-kDa filtration column in order to remove the porcine enzymes. This process was performed with NEM-AQ, resulting in the product referred to as in vitro–digested NEM (NEM-IVD), as well as with phosphate-buffered saline (PBS), resulting in the product referred to as PBS-IVD.
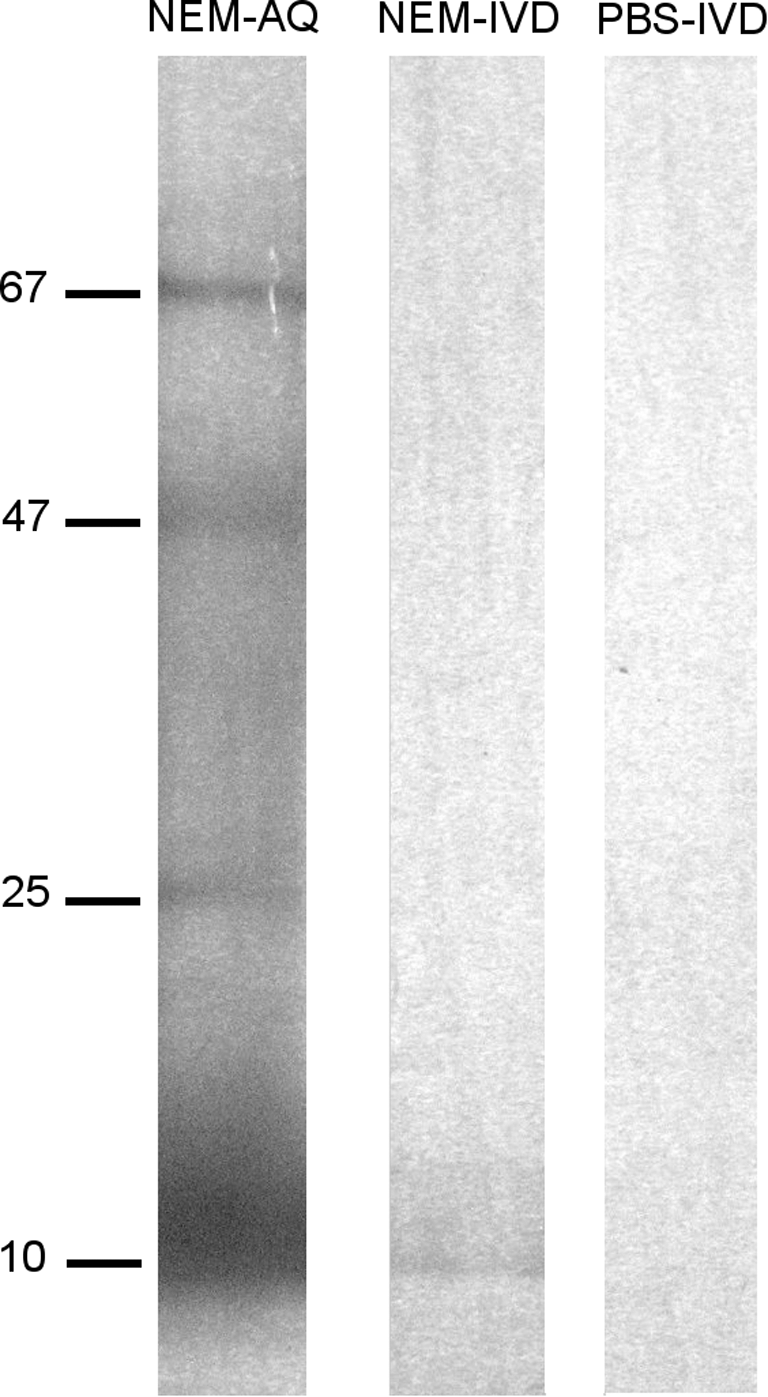
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis separation of NEM-AQ, NEM-IVD and PBS-IVD. Samples were separated on a 4–15% polyacrylamide gel using denaturing conditions, and bands were visualized by silver stain. Molecular sizes of major bands in the NEM-AQ lane are indicated in kDa on the left-hand side. This presentation of the data is used to show the effects of the in vitro digestion of NEM on the molecular weights of the resulting digested protein products. This comparison shows a loss of higher-molecular-weight products when comparing NEM-IVD with the undigested NEM-AQ. Within the sensitivity of sodium dodecyl sulfate–polyacrylamide gel electrophoresis and silver stain methods, no residual enzymes or breakdown products from the in vitro digestion process were detectable in the PBS-IVD sample.
Suppression of TNF-α production resulting from treatment of PBMCs with NEM-IVD
In the context of the mitogen PWM, reflecting an in vitro model of the cellular collaborations in lymphoid tissue, the native NEM-AQ showed an anti-inflammatory effect only at the highest dose used, and the effect returned to baseline at the lower doses. In contrast, NEM-IVD showed significant anti-inflammatory properties across a wide dose range with respect to TNF-α production (Fig. 6). The in vitro–digested saline control (PBS-IVD) had some effects on PBMC culture proliferation and cytokine production that were different from those of saline alone, suggesting the presence of residual material derived from the in vitro digestion process. The effects seen at the two lower doses may be most relevant for predicting in vivo outcomes. The data showed that in the presence of PWM, NEM-AQ had only a minor effect on TNF-α production, whereas NEM-IVD showed 45–55% suppression of TNF-α production (P<.04).
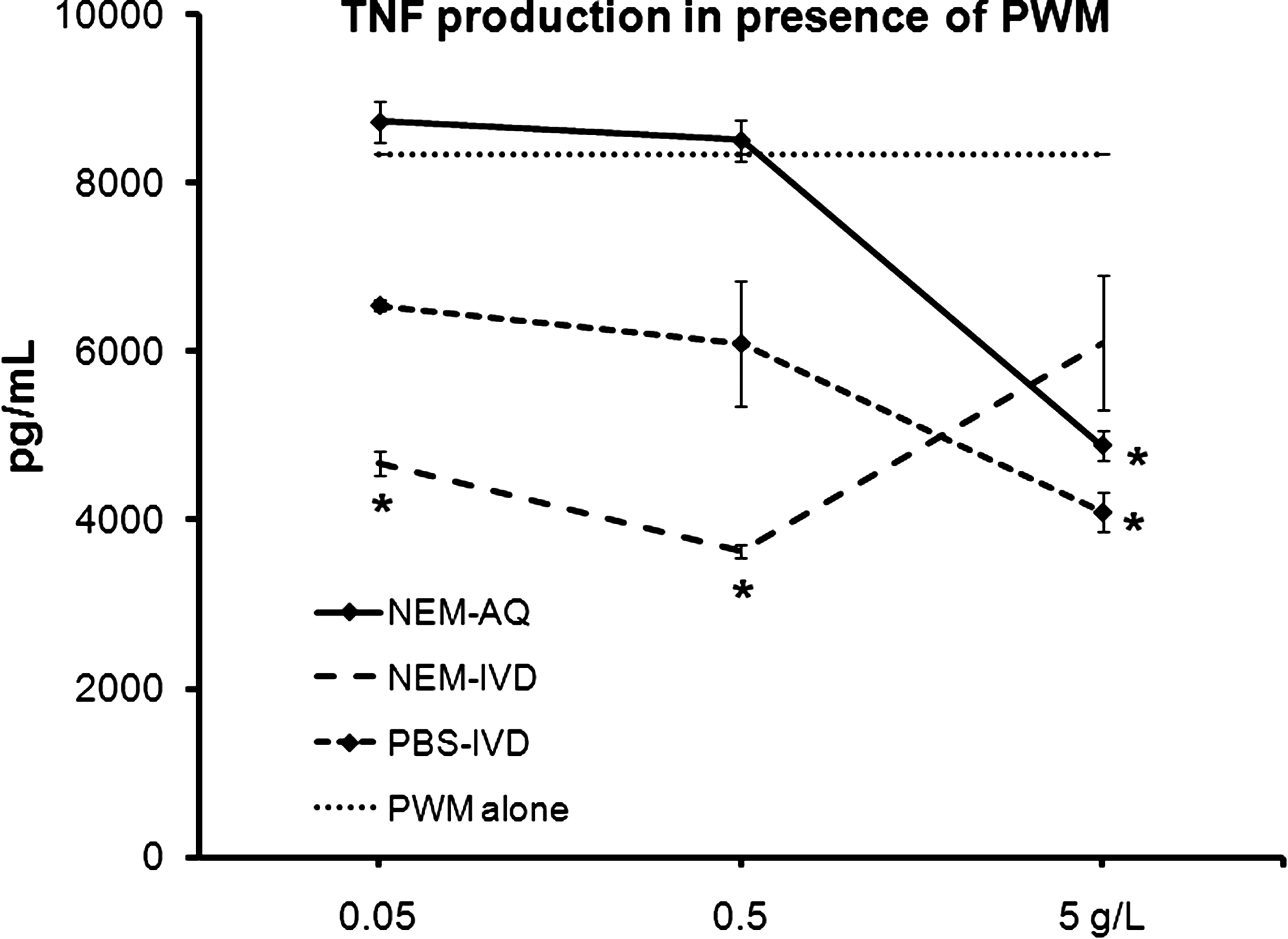
Comparison of the effect of NEM-AQ, NEM-IVD, and PBS-IVD on production of the cytokine TNF-α by 4-day peripheral blood mononuclear cell cultures simultaneously exposed to PWM. TNF-α production by cultures exposed to PWM was most affected by the NEM-IVD extract. At the highest dose of extracts tested (5 g/L), all three products produced a similar reduction in TNF-α production of 30–40%. However, it can be argued that this high dose is beyond a physiological relevant dose after consumption. Therefore, the data for the two lower doses may be more relevant for predicting in vivo outcomes. At the two lowest doses of extracts tested, a different picture emerged. Here NEM-AQ did not affect TNF-α production, showing cytokine levels similar to baseline (cultures exposed to PWM alone). The PBS-IVD extract showed some activity, lowering TNF-α production. However, NEM-IVD had the greatest effect in lowering TNF-α production. At the 0.5 and 0.05 g/L concentrations, NEM-IVD lowered TNF-α production by 55% and 45%, respectively. Statistically significant differences are indicated (*P<.05). The results shown are mean±SD values from a representative of three separate lymphocyte proliferation cultures using cells from three different donors.
Discussion
Osteoarthritis is often considered a local problem centered on the specific target area where bone and joint degradation is seen, such as, for example, a knee. However, osteoarthritis is a systemic disease, involving immune dysregulation and altered cytokine profile (Fig. 7). In particular, T cells likely play an important role in the pathogenesis and progression of osteoarthritis. Osteoarthritis involves infiltrating monocytes producing TNF-α. 15 It is also believed that peripheral blood leukocytes, which travel through the tissues of inflamed joints, are activated through exposure to locally produced mediators of inflammation (i.e., IL-1β, TNF-α, etc.). 16 We were therefore interested in studying cytokine production in peripheral blood mixed cultures including monocytes. This was accomplished through an extended proliferation/cytokine assay, where both digested and undigested NEM preparations with appropriate controls were tested in serial dilutions in the presence and absence of mitogens. Two mitogens were tested in parallel: PHA, which is a T-cell mitogen that will induce T-cell proliferation, and PWM, which is a mitogen that requires the collaboration of T cells, B cells, and monocytes in the culture.
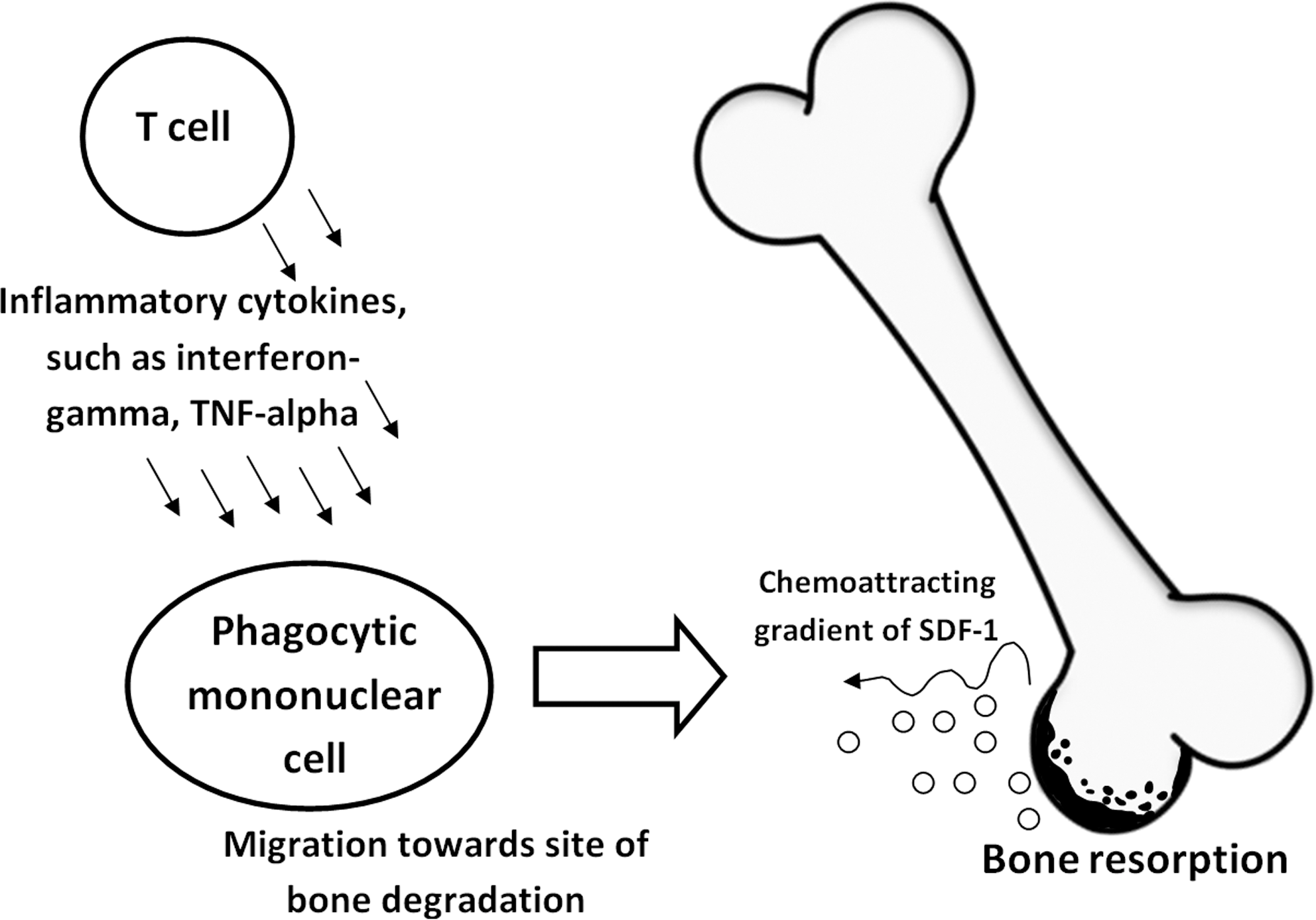
Diagram showing arthritic mechanisms of action involving T-cell–derived inflammatory cytokines in activation and migration of phagocytic mononuclear cells into areas of bone destruction. SDF-1, stromal cell–derived factor-1.
The significance of the data must be interpreted in light of the specificity of the culture conditions in the presence of the two separate mitogens, PHA and PWM, as well as the importance of the sequence in which stimulating agents were added. PWM is an aqueous extract from Phytolacca americana (pokeweed) that has mitogenic properties that involve mechanisms closely mimicking events in lymph nodes and other immune tissue where antigen presentation leads to co-stimulation and collaboration of multiple cell subsets. The mechanisms involve leukocyte aggregation. 17 RNA synthesis precedes DNA synthesis by 24 hours, after which cell division begins, involving up to 60% of the peripheral blood lymphocyte fraction. 18 The activation process involves T lymphocytes, B lymphocytes, and phagocytic mononuclear cells 19 in tandem and generates both T cell– and B cell–derived cytokines, 20 leading to generation of immunoglobulin-secreting plasma cells 21 and a shift in CD45 isoform expression indicative of plasma cell differentiation. 22 In contrast, an extract from Phaseolus vulgaris (red kidney bean) called PHA predominantly activates T lymphocytes, even though some B-cell activation can be seen as a result of the activated T cells triggering some B lymphocytes into proliferation. 23 Therefore, these two mitogens were used as a method to shed light on events that are strictly T cell mediated versus events that require complex cellular collaboration (B cells and T cells).
The reduction of proliferation in NEM-treated cultures should not be seen as a suppression of a mitogenic response, but rather as evidence that NEM has leukocyte signaling properties of its own. The sequential addition of NEM first, followed by mitogens after 5 minutes, allowed compounds in NEM to engage signaling in target cells so when the mitogens were subsequently added the resultant signal was diminished.
Because NEM affected both PHA and PWM mitogenicity, but in different ways, this finding suggests specific mechanisms, including that NEM contains compounds directly able to modulate T-cell activation, and that NEM also has immune-modulating properties in the context of a more physiological activation process, such as in the PWM model of lymphocyte activation.
Cytokine production was affected in the cultures, with significant changes in three inflammatory cytokines: IL-6, IFN-γ, and TNF-α. It is interesting that this did not change when NEM was passed through the in vitro digestion protocol, except for TNF-α. The reduction in TNF-α production with NEM-IVD was seen at 100-fold lower doses than with undigested NEM. In the case of PWM stimulation, NEM-IVD showed anti-inflammatory properties by drastically reducing the production of TNF-α, in contrast to the mild increase in TNF-α production when cells were pretreated with NEM-AQ. Thus, the in vitro digestion potentiated the anti-inflammatory action of NEM, so that much lower doses of NEM-IVD were seen to produce similar effects as 25-fold higher doses of NEM-AQ. This is relevant for suggesting anti-inflammatory mechanisms in vivo after consumption of NEM and subsequent digestion in the stomach.
The dose–responses seen in the different assays were in several cases nonlinear. This may be attributed to several confounding factors associated with the highest dose, suggesting that the biological effects observed at lower doses should receive the most attention. It may be argued that the highest dose we used (5 g/L) exceeds a likely physiologically relevant dose. However, it may also be argued that this dose may be reached locally along the intestinal mucosa after consumption. We suggest that the highest dose (5 g/L) is quite high and that the biological responses seen at the two lower doses may be more relevant for predicting in vivo outcomes. There is also the possibility that calcium may have been an issue at the highest dose. NEM contains some calcium from unseparated eggshell, so for the most part this will be insoluble calcium carbonate. At the 5 g/L dose of NEM, the amount of calcium present may still be sufficient to interfere with cellular signaling. Further dilution of NEM may dilute calcium to insignificant levels. As it is therefore unlikely that calcium contributed to the different responses seen at the lowest dose, it cannot be completely ruled out as a potential mechanism of disturbing or abrogating cellular signaling at the highest dose used.
In the case of the TNF-α cytokine testing, the most interesting difference between the effects of NEM-AQ and NEM-IVD was the reduction in TNF-α production in the presence of PWM that occurred with all three concentrations of NEM-IVD. These reductions in TNF-α production were strongest with the lowest doses of NEM-IVD, and this effect was opposite to that seen for NEM-AQ. Although the in vitro digestion procedure introduced compounds that were not completely removed by the size exclusion centrifugation step and that possessed bioactivity, the digestion process nevertheless increased the ability of NEM-AQ to reduce TNF-α production in 4-day PBMC cultures in the presence of PWM. The effect of NEM-IVD on TNF-α production in the presence of PWM was also different from the effect resulting from treatment of PBMC cultures with PBS-IVD and points to activities unique to the NEM-IVD product.
In particular, the result of NEM-AQ and NEM-IVD reducing TNF-α production is of interest in terms of identifying mechanisms of action pertaining to arthritis conditions because this cytokine is known to attract cell infiltration into arthritic joints and contribute to the inflammation within the joint.
Comparison of the effects of NEM-AQ and NEM-IVD on cytokine production by PBMC cultures revealed some differences that could not be entirely attributed to activities derived from the in vitro digestion process itself (such as enzyme breakdown products or residual bile salts that were not removed by the size exclusion centrifugation step). This unique effect of NEM-IVD with respect to TNF-α clearly warrants efforts to further investigate the effects of in vitro digestion on NEM. This is of particular importance as several biological TNF-α–inhibiting drugs have proven quite effective in treating arthritis but have been shown to have infrequent but often severe side effects. 24 A treatment, such as NEM, that has immunomodulatory properties that are likely more diffuse could potentially avoid the unfortunate side effects of the currently available biological drugs.
Footnotes
Acknowledgments
This study was performed at NIS Labs, an independent research lab specializing in natural products research, and was sponsored by ESM Technologies, LLC.
Author Disclosure Statement
K.F.B. and G.S.J. are employed by NIS Labs, an independent contract research laboratory. K.J.R. is employed by ESM Technologies, LLC in the function of Director of Scientific and Regulatory Affairs. The authors have no other financial interest in the subject matter.
