Abstract
Sex hormone replacement therapy helps improve quality of life in climacteric women. However, estrogen-induced cell proliferation in the uterus and mammary gland increases the risk for cancer in these organs. The lower incidence of mammary cancer in Asian women than in western women has been attributed to high intake of soy isoflavones, including genistein. Our previous work in the prepubertal rat uterus model showed that genistein (0.5 mg/kg body weight subcutaneously) caused an estradiol-like hypertrophy in myometrial and uterine luminal epithelial cells and an increase in RNA content in luminal epithelium; however, it did not induce cell proliferation, uterine eosinophilia, or endometrial edema. The present study investigated, in the same animal model, the effect of genistein administration (0.5 mg/kg body weight subcutaneously) before treatment with estradiol-17β (0.33 mg/kg body weight subcutaneously) on uterine responses that were not induced by genistein. Pretreatment with this phytoestrogen completely inhibited estradiol-induced mitoses in uterine luminal epithelium, endometrial stroma, and myometrium and partially inhibited estradiol-induced uterine eosinophilia and endometrial edema. These findings indicate that genistein protects against estrogen-induced cell proliferation in the uterus and suggest that future studies should investigate the possibility of using this agent to decrease the risk for uterine cancer after hormone replacement therapy in climacteric women.
Introduction
W
Although HRT in postmenopausal women alleviates climacteric symptoms and protects against associated diseases, it also increases the risk for breast or endometrial cancer, at least after a prolonged duration. 1 This effect seems to be caused by stimulation of target-organ cell proliferation by the estrogens used in HRT. Therefore, identification of a compound that mimics the beneficial responses to estrogen but does not induce cell proliferation is desirable.
If all responses to estrogen were mediated by the same mechanism and all estrogen receptors (ERs) were identical (as was first proposed for the cytosol-nuclear receptor–mediated genomic responses 2 ), there would be few possibilities to selectively induce therapeutically convenient responses to estrogen but not to cells at risk (cell proliferation).
In addition to the classic “cytosol-nuclear” ERs 2 (now α-ERs 3 –5 ), the existence of additional ERs has been proposed: eosinophil leukocyte ERs, 6 –8 membrane ERs, 9,10 cytoplasmic and nuclear type II ERs, 11 and β-ERs. 3 –5 The finding of different kinds of estrogen-binding proteins—presumably ERs—in the uterus opens the possibility of a dissociation of responses by agents displaying selective agonist or antagonist action on some but not all receptors. This dissociation was reported under several experimental conditions in the rat uterus. 7,8,12,13
The lower incidence of breast cancer in Asian women than in western women, 14 which parallels the higher amount of phytoestrogens (including soybean products) consumed by Asian women, 14 was attributed to lifestyle but not to genetic factors. 15,16 In fact, in East and Southeast Asia, the average intake of phytoestrogens is estimated to be more than 10 times higher than in the United States or Europe, 17 and plasma isoflavone concentrations are higher in Japanese women than in Europeans. This finding suggests that phytoestrogens and isoflavones may have a role in hormone-dependent breast cancer prevention. 18 This finding also led us to consider whether soy isoflavone genistein may dissociate responses to estrogen in the uterus.
We confirmed that hypothesis in a recent study. 19 We reported that in the uterus, genistein induced estrogen-like hypertrophy in myometrial and luminal epithelial cells and increased RNA content in luminal epithelium; however, it did not cause proliferation of various uterine cell types, uterine eosinophilia, or endometrial edema.
As part of a search for an agent that inhibits estrogen-induced uterine cell proliferation, the present study investigated whether genistein antagonizes uterine estrogenic responses that were not induced by genistein alone. Our findings may help identify a new therapeutic approach for climacteric women receiving HRT that decreases risk for tumor development, at least in the uterus. It might be possible to use this agent at earlier ages for prevention of hormone-dependent tumor development.
Materials and Methods
Experimental animal procedure
The experimental protocol was approved by the local ethical committee. Sprague–Dawley rats were bred and maintained in the vivarium of the University of Chile Medical School. The prepubertal rat model was chosen because the very low endogenous estrogen levels in control animals ensure the absence of estrogenic responses; at this age, all ERs and mechanisms were reported to fully respond to hormone stimulation. 20 Ten rats were used for each experimental condition. Female prepubertal (21-day-old) animals were treated subcutaneously with genistein of high-performance liquid chromatography–standard quality (Sigma; 0.5 mg/kg body weight) or vehicle (controls) (0.3 mL, 1:9 ethanol:saline). One hour later, the animals were treated with estradiol-17β (E2) (Merck; 0.33 mg/kg body weight) or vehicle (control). The dose of E2 was chosen according to former studies in the same prepubertal rat model; this dose ensured the maximal responses to hormone stimulation for all analyzed measures of estrogen action in the uterus. 21 –23 The dose of genistein was chosen to ensure an approximation to E2 molar concentrations that induce the maximal response to hormone stimulation (0.01–0.3 mg/kg). Uteri were excised under ether anesthesia 6 or 24 hours after treatment and fixed in neutral formalin for further histologic process.
Histologic, histochemical, and morphometric procedure
Each uterine formalin-fixed and dehydrated horn was cut into 3 pieces (superior, medium, and caudal). The pieces were paraffin-embedded in a single paraffin block so that 5-μm-thick uterine cross-sections from the 3 pieces could be evaluated together on the same histologic slide. For each animal, 1 group of hydrated uterine cross-sections was stained for 1–3 minutes in hematoxylin, washed in several changes of tap water, transferred to a saturated lithium carbonate solution, and kept for 1 minute. Subsequently, they were stained in 1% eosin Y aqueous solution, washed quickly in distilled water, and dehydrated in a graded series of ethanols and xylene. 24 The stained slides were evaluated microscopically for eosinophil quantification, 25,26 counting of mitoses in the various uterine histologic layers, 22,23 and morphometric estimation of endometrial edema. 12,24
Quantification of estrogenic responses
The following estrogenic responses were quantified: uterine eosinophilia; percentage of eosinophils according to the distribution in different uterine histologic layers and to their degree of degranulation; edema in deep endometrial stroma; and number of mitotic figures in uterine luminal epithelium, endometrial stroma, and myometrium. The variables for estrogen stimulation were evaluated at the times during which their maximal response to E2 occurs. 12,24,25 Uterine eosinophilia reaches maximal response at 6 or 24 hours after hormone treatment; endometrial edema reaches maximal response at 6 hours and declines thereafter; and uterine luminal epithelial, stromal, and myometrial cell proliferation reach maximal responses at 24 hours. To allow a comparison between the effects of genistein among the different measures of E stimulation, all responses were expressed as percentage of response to E2 (i.e., the value of vehicle-treated animals was considered a 0% response and the value of response to E2 was a 100% response).
For each animal, uterine eosinophilia 25 was assessed in 30 uterine sections, distributed along the uterus (superior, medium, and caudal); eosinophils were classified according to their location within the different uterine histologic layers and to their degree of degranulation. 22,26 Edema in deep endometrial stroma 24 was evaluated by counting the number of nuclei in thirty-six 1054-μm2 areas delimited by a standard circle located in the ocular piece of the microscope. As shown elsewhere, 24 an increase in the reciprocal value of cell density (a decrease in cell counts within a standard area) in a location that contains few cells and primarily consists of extracellular space reflects edema because an increase in its volume, but not in the cellular volume, leads to an increase in the reciprocal value of cell density. Estrogen-induced mitotic response was evaluated as an increase in the number of mitotic figures in luminal epithelium, endometrial stroma, and myometrium and was quantified for each animal in 18 uterine cross-sections considering all 3 uterine pieces. 22,23
Statistical analysis
According to previous studies 22 using the Tukey test of additivity, 27 for some measures of estrogen stimulation data must be submitted to logarithmic or square-root transformation to normalize distribution. Accordingly, uterine eosinophilia numbers were log-transformed. Transformed (uterine eosinophilia) and nontransformed (the remaining measures of E stimulation in the uterus) data were subjected to further statistical analysis. Because multiple comparisons were performed between the different experimental conditions, transformed and nontransformed data were subjected to the least-significant-difference a posteriori test. Two-tailed P values less than .05 were considered to indicate statistical significance. The common variance needed for this test was estimated from a 1-way unbalanced analysis of variance; for all reported results, the P value was less than. 05. In studies of uterine eosinophil degranulation and distribution proportion, the χ2 statistic was chosen to evaluate differences between the proportions. Stata 10.0 was used for statistical analysis. Results on studies of eosinophil degranulation and distribution within the various uterine histologic locations were not considered in control animals without E2 treatment because of the extremely low number of eosinophils observed in this experimental condition.
Results
Endometrial edema and uterine eosinophilia
Figure 1A shows edema in deep endometrial stroma induced by E2. Edema was significantly reduced in animals pretreated with genistein. Figure 1B demonstrates that estrogen-induced uterine eosinophilia measured in endometrium with myometrium was reduced by more than half after genistein pretreatment. As compared with E2-treated animals, in which most uterine eosinophils were located in endometrium with myometrium, genistein-pretreated animals had more eosinophils located in the mesometrium than did E2-treated animals without genistein pretreatment (Fig. 2A). Results reveal that the proportion of degranulated uterine eosinophils observed 6 and 24 hours after treatment with E2 was drastically reduced in animals pretreated with genistein (Fig. 2B). Eosinophil degranulation and distribution within the uterine histologic layers data are not reported for control animals because of the extremely low eosinophil counts.
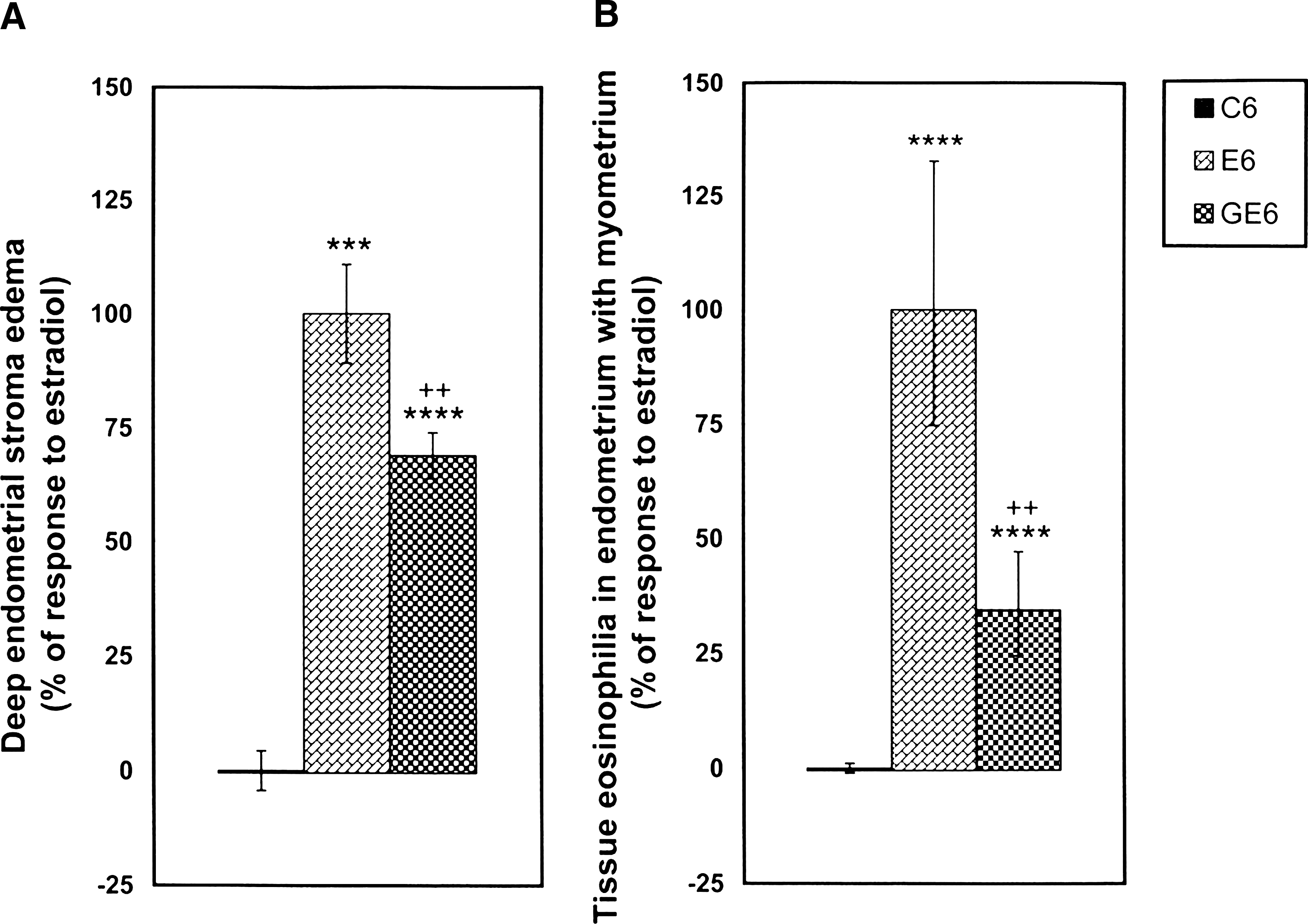
Effect of genistein pretreatment on estradiol-17β (E2)–induced edema in deep endometrial stroma (
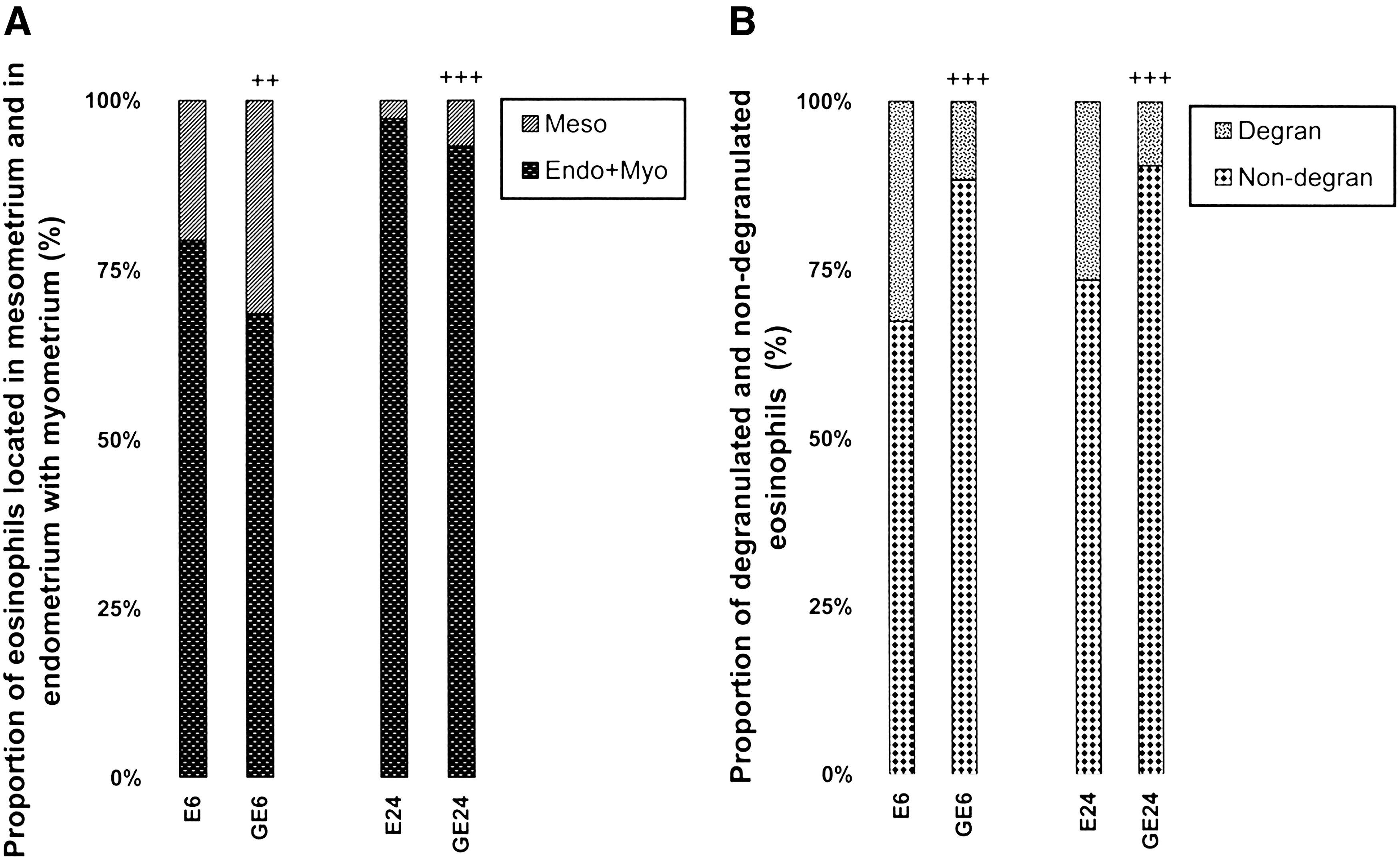
Effect of genistein pretreatment on estradiol-17β (E2)–induced change in proportions of uterine eosinophils located in the mesometrium and those in endometrium with myometrium (
Uterine cell proliferation
Figure 3 shows estrogen-induced cell proliferation in uterine luminal epithelium (Fig. 3A), endometrial stroma (Fig. 3B), and myometrium (Fig. 3C) 24 hours after hormone treatment. Genistein pretreatment almost abolished this response to E2 in these cell types.
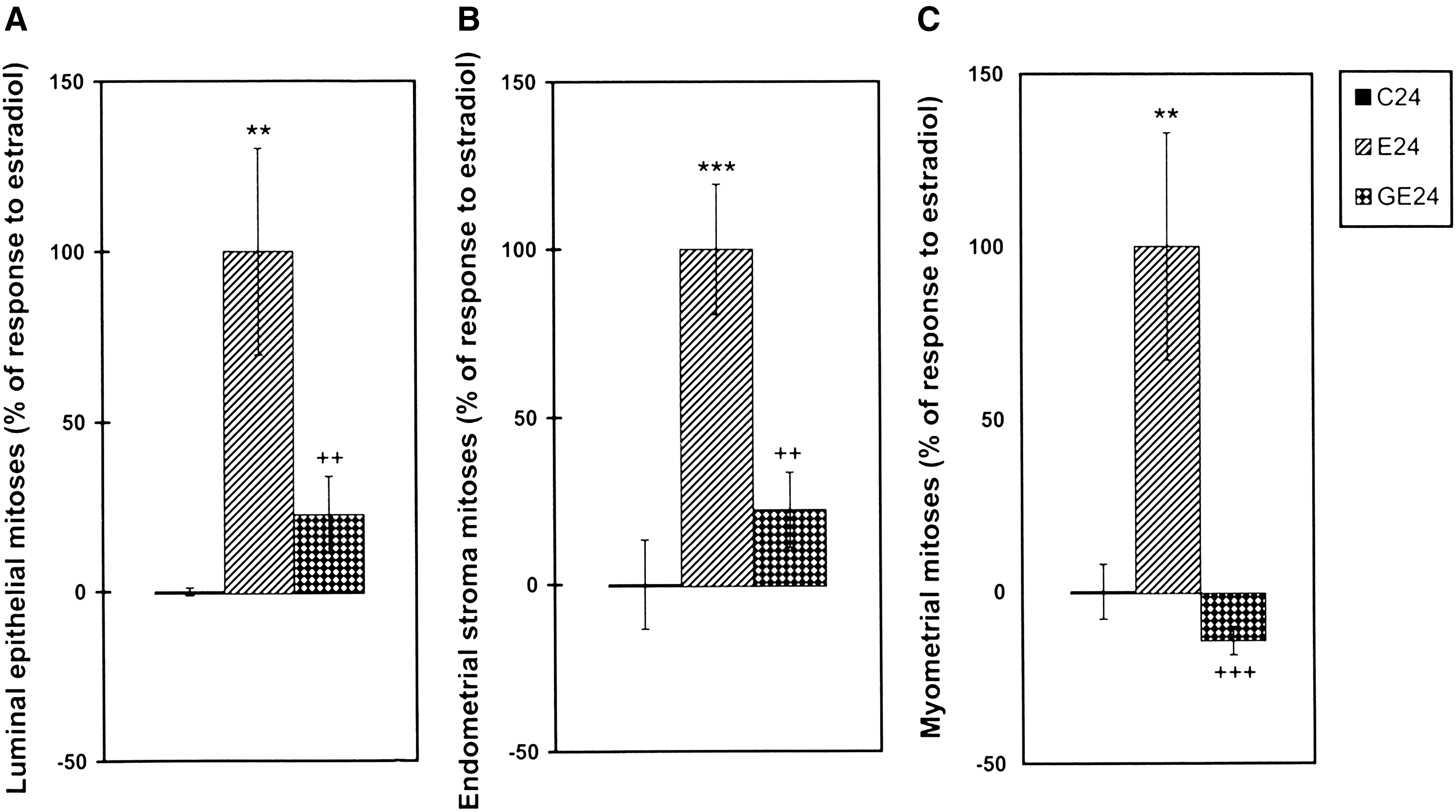
Effects of genistein pretreatment on estradiol-17β (E2)–induced increase in the number of mitotic figures in uterine luminal epithelium (
Discussion
We previously reported that genistein treatment increased uterine luminal epithelial cell RNA content and induced cell hypertrophy in uterine luminal epithelium and in myometrium, similar to responses induced by treatment with E2. 19 Genistein, however, did not induce uterine eosinophilia, endometrial edema, or uterine cell proliferation, responses that also were re induced by E2. 19 The selective induction of some estrogen-like genomic responses but not others by genistein may be explained by several factors. One possibility is that genistein-induced uterine cell hypertrophy and increase in RNA content 19 are not responses mediated by ERs. Second, genistein may be displaying a strong agonist interaction only with ERs involved in uterine cell hypertrophy and RNA increase but not with ERs involved in uterine cell proliferation, uterine eosinophilia, or endometrial edema. Third, genistein may display a strong agonist interaction with ERs involved in uterine cell hypertrophy and RNA increase and an antagonistic interaction with ERs involved in uterine cell proliferation, endometrial edema, and uterine eosinophilia. The second and third possibilities consider the existence of a different kind of ER that mediates different responses to estrogen stimulation. 7,8,22,28 –31
Many other explanations may be proposed to explain the selective inhibition of only some responses to estrogen: for instance, subtle differences in ER conformations caused by interaction with different ligands interacting with different co-activators or co-repressors within the cell, 32 and presence of different molecular chaperones (eg FKBP51, FKBP52, and Cyp40), which may be incorporated in the inactivated ER complexes from the different cell types. 33,34
According to our hypothesis, the finding that genistein almost completely inhibited E2-induced uterine cell proliferation strongly suggests that genistein is a strong competitive antagonist for ERs involved in these responses. The partial inhibition of estrogen-induced uterine eosinophilia and endometrial edema by genistein suggests a weaker competitive antagonism. Previously reported induction of uterine cell hypertrophy and increase in RNA content by genistein 19 suggests selective agonistic interaction of genistein with ERs involved in these responses.
It was previously proposed that surface ER receptors in the eosinophils are involved in estrogen-induced migration of the eosinophils to the uterus 7 and that a second kind of ER in the eosinophils 7,8,26 could be involved in eosinophil degranulation. Other researchers suggested that eosinophil degranulation and release of hydrolytic enzymes from the eosinophils facilitate migration of the eosinophils from mesometrium to other uterine cell layers. 7,8,35,36 The decrease in the proportion of degranulated eosinophils in the uterus under the effect of genistein pretreatment and the decrease in the proportion of eosinophils located in endometrium with myometrium agree with this hypothesis and suggest a possible competitive inhibition of E2-induced eosinophil degranulation by genistein.
The most relevant finding from this report is the strong inhibition of E2-induced uterine cell proliferation by genistein at the doses used in the present study. This response is considered to be at risk for endometrial cancer and myomas development. Further studies are necessary to observe the effects of genistein for long periods in the uterus and in other organs, such as the mammary gland, before the initiation of studies in humans. In human studies, it will be necessary to demonstrate whether genistein also antagonizes estrogen-induced uterine cell proliferation and whether this inhibition also occurs for other estrogen target tissues.
The interaction of genistein with estrogen receptors is not the only mechanism by which soy isoflavones may have bioactivity. They inhibit tyrosine kinases, have antioxidant activity, bind to and activate peroxisome proliferator regulators α and γ, inhibit enzymes in steroid biosynthesis, strongly influence natural killer cell function and the activation of specific T-cell subsets, and inhibit metastasis. These various properties may explain the much lower incidence of hormonally dependent breast cancer in Asian populations compared with that in Americans and Europeans. 37 Recently, it was proposed that genistein is a chemopreventive agent targeting the epigenome, which inhibits DNA methylation, thus antagonizing a potentially initiating event in cancer development. 38,39
The beneficial effect of high intake of soybean isoflavones is controversial. Although several authors 18,40,41 suggest that it reduces cancer risk, others consider it harmful. For instance, researchers reported that these isoflavones alter reproductive hormones, spermatogenesis, sperm capacitation, and fertility. 42
The effects of high intake of soybean isoflavones, including genistein, differ at various ages of exposure. It is now accepted that prenatal or neonatal exposure to hormones or hormone action–displaying xenobiotics during critical periods of development induces persistent changes in the action of related hormones. 43 This result is due to a modification in the activity of receptors and in the intensity of responses mediated by them. 44 This process, named “imprinting” by Csaba, 43,44 involves several biochemical, morphologic, and functional alterations in these cell types that persist through life and cause several diseases later in life. 45,46 In this context, prenatal or neonatal exposure to a high amount of soybean isoflavones, among them genistein, may present persistent beneficial or harmful effects.
It was reported that early exposure to genistein can protect against mammary cancer in rodents; on the basis of the critical role of timing of exposure, it was suggested that the protection was mediated through regulation of mammary growth and differentiation pathways. 47 In fact, genistein early in life causes enhanced mammary gland differentiation, alterations in cell proliferation, apoptosis, and upregulation of tumor-suppressor genes. 47 Genistein significantly increased the lordosis quotient of females and also enhanced the sexual activity of males. 48 Prenatal or neonatal treatment with genistein at environmentally relevant doses caused adverse consequences on female development, which was manifested in adulthood as altered ovarian differentiation leading to multioocyte follicles, estrous cyclicity disruption, infertility, mammary gland changes, and behavioral changes. 49 Rats with prenatal and early postnatal exposure to maternal dietary genistein (300 ppm) through gestation and lactation exhibited a decrease in anogenital distance, testis size, and serum testosterone levels, revealing male demasculinization. 50
In this context, we agree with the concerns that have been raised by others with regard to the high isoflavone content of soybean infant food and the possible adverse effects. 42 Human infants exclusively fed soybean infant food have blood levels of isoflavones at least 5-fold higher than do adult humans who eat a soy-rich diet; 42 therefore, they may develop alterations or diseases later in life that need to be carefully investigated in epidemiologic studies.
Finally, the effects of prenatal or neonatal exposure to genistein or other soy isoflavones may be affected by a simultaneous exposure to other environmental imprinters, such as polycyclic aromatic hydrocarbons present in urban milieu, dioxins, pesticides, food additives, and other polluting agents, thus changing the effects of soy isoflavones. For instance, it was reported that the polycyclic aromatic hydrocarbon benzopyrene modifies the imprinting caused by genistein alone. 51 Therefore, epidemiologic data from urban milieu or areas with high environmental contamination may not be comparable to those from other places.
We conclude that genistein protects against estrogen-induced cell proliferation, at least in the uterus, and suggest that further studies explore the possible use of this agent to decrease the risk for uterine tumor development after HRT in climacteric women. Additional studies should also investigate the possibility that genistein could be used at earlier ages for prevention of hormone-dependent tumor development. Further research is needed to evaluate the positive or negative effect of high genistein intake from soybean food at different ages; special caution is recommended with the use of soybean baby food because of the reported persistent effects caused by the mechanism of imprinting.
Footnotes
Acknowledgments
Financed by Research Team Grant in Science and Technology ACT07, Bicentennial Program in Science and Technology, CONICYT, Chile. We thank Ms. Iris Rodríguez for technical help.
Author Disclosure Statement
No competing financial interests exist.
