Abstract
Respecting the significant role of inflammation in acute ischemic stroke (AIS) development, we explored the correlations of inflammatory cytokines and lipoprotein-associated phospholipase-A2 (Lp-PLA2) with AIS and their early prognostic value. The retrospectively enrolled subjects [Study (AIS patients) and Control (healthy volunteers) groups] were determined for serum index levels. Neurological impairment and early prognosis of AIS patients were assessed. The relationship of National Institutes of Health Stroke Scale (NIHSS) and the indexes and the risk factors and predictive value of peripheral blood inflammatory cytokines combined with Lp-PLA2 for poor early prognosis were analyzed. Interleukin (IL)-1β, IL-6, tumor necrosis factor-α (TNF-α), and Lp-PLA2 levels rose in AIS patients, while IL-10 dropped. The NIHSS score positively correlated with IL-1β, TNF-α, and Lp-PLA2 and negatively correlated with IL-10. As AIS aggravated, IL-1β, IL-6, TNF-α, and Lp-PLA2 rose dependently, but IL-10 dropped. Patients in the poor early prognosis group had higher diabetes proportions, NIHSS scores and serum IL-1β, IL-6, TNF-α, and Lp-PLA2 levels at admission, as well as lower IL-10 levels than those in the good early prognosis group. IL-6, IL-1β, TNF-α, and Lp-PLA2 were risk factors, but IL-10 was a protective factor against poor early prognosis in AIS patients, with their combined detection showing high predictive value. Collectively, highly expressed IL-6, TNF-α, IL-1β, and Lp-PLA2 and lowly expressed IL-10 in patients with AIS closely related to AIS development and early prognosis, and their combination could increase the predictive value of NIHSS for AIS poor early prognosis.
Introduction
Cerebrovascular disease (CBVD) is a significant contributor to mortality in contemporary society, with ischemic stroke (IS) being the predominant form of CBVD, accounting for approximately 70% of all stroke cases (Herpich and Rincon, 2020). Acute ischemic stroke (AIS) is a type of stroke condition resulting from cerebral thrombosis or the local ischemia and hypoxia following cerebral vascular occlusion, which triggers disruption of brain cell energy metabolism, excessive calcium accumulation, oxidative stress, and inflammatory responses, thus leading to significant cellular death in the affected regions of the brain (Walter, 2022). Mounting evidence has demonstrated that China is the hardest-hit area of AIS. In a substantial, nationally representative cohort of individuals aged 40 years or older, the expected prevalence, incidence, and death rates of stroke in China in 2020 were 2.6%, 505.2 per 100,000 person-years, and 343.4 per 100,000 person-years, respectively (Tu et al., 2023b). With the rapid aging of the population, the rising incidence of unhealthy lifestyles among individuals, and the extensive exposure to stroke risk factors, the burden of stroke disease in China is surging. The incidence of CBVD events in China is projected to rise by approximately 50% by 2030 compared with 2010 (Tu et al., 2023a). The prevention and management of AIS remain a challenging endeavor in the coming years (Green et al., 2021; Rabinstein, 2020).
In stroke, the number of candidate biomarkers is increasing. These biomarkers include proteins, ribonucleic acid, lipids, or metabolites. In terms of stroke prognosis, although biomarkers may improve the diagnosis and management of stroke patients, there are currently few markers showing sufficient sensitivity, specificity, speed, accuracy, and cost-effectiveness; meanwhile, the value of most identified biomarkers as parameters that can support clinical decision-making to improve patient outcomes is still uncertain, emphasizing the need for additional work (Kamtchum-Tatuene and Jickling, 2019; Montellano et al., 2021). The inflammatory environment of the nervous system caused by stroke is currently recognized as one of the main determinants of brain parenchymal damage; in this case, inflammatory mediators may be an important target for ameliorating the management of stroke patients (Tirandi et al., 2023). Inflammation plays a significant role as a pathogenic mechanism in the context of AIS, contributing to the progression of stroke in individuals with cerebral circulation disorders (Pawluk et al., 2022). In the event of AIS, the immune system is influenced by interleukin (IL)-1β, IL-6, IL-10, and tumor necrosis factor-α (TNF-α) present in the peripheral blood, and these inflammatory cytokines exert their effects via the autonomic nervous system and the hypothalamus–pituitary–adrenocortical axis, ultimately causing the immunosuppression of stroke (Endres et al., 2022; Wytrykowska et al., 2016). Clinical research reveals that the partial improvement of brain injury can be achieved by the inhibition of inflammatory reactions; yet, it is important to note that inhibiting proinflammatory reactions may impede the healing of brain tissue damage and have an impact on the long-term recovery of cranial nerve function (Amantea et al., 2014; Anrather and Iadecola, 2016; Przykaza, 2021). Therefore, the monitoring of inflammatory cytokine expression levels in the peripheral blood of patients with AIS can serve as a valuable reference for determining the appropriate treatment approach for AIS.
Microcirculation disturbance functions as the pathophysiological foundation for the initial manifestation of each category of stroke (Guo et al., 2022; Sun et al., 2015). As one of the subtypes of the PLA2 superfamily and a proinflammatory enzyme that serves as an indicator of vascular inflammation, lipoprotein-associated phospholipase-A2 (Lp-PLA2) is linked to the likelihood of experiencing IS (Dua et al., 2022; Romero et al., 2012). Besides, Lp-PLA2 is predominantly bound to low-density lipoprotein (LDL), and it produces inflammatory molecules including oxidized fatty acids and lysophosphatidylcholine by hydrolyzing oxidized LDL, thus provoking inflammatory responses (Huang et al., 2017; Wang and Connolly, 2010). Accumulating studies imply that the risk of cardiovascular disease and recurrence increases as the level of Lp-PLA2 rises (Bonnefont-Rousselot, 2016; Epps and Wilensky, 2011). Nowadays, it has been demonstrated that Lp-PLA2 can serve as a predictor of IS (Zhang et al., 2021a). At present, the clinical research on AIS is more extensive, but there is little comprehensive analysis regarding the relationship of peripheral blood inflammatory cytokines and serum Lp-PLA2 with the development and prognosis of AIS patients. To expand the clinical diagnosis, treatment, and evaluation protocols for AIS patients, this study was to investigate the expression levels of peripheral blood inflammatory cytokines and serum Lp-PLA2 in AS, as well as their correlations with the extent of disease and predictive value for AIS poor early prognosis.
Materials and Methods
Study subjects
The patients with AIS (n = 240) who visited Chengdu Seventh People's Hospital from June 2021 to June 2023 were analyzed retrospectively. Among them, 22 cases did not meet the inclusion criteria (including 15 patients who were admitted to the hospital after 24 h of onset, and 7 patients with non-first-ever onset of the disease), 58 cases were excluded by the exclusion criteria (including 10 patients with a history of hemorrhagic disease in the past 6 months, 13 patients with severe consciousness disorders, 25 patients with complications of infections, tumors, or immune diseases, 8 patients with severe complications, and 2 patients with unstable vital signs), and 160 patients with AIS were eventually included in the Study group (Fig. 1). Among the health check-up volunteers (n = 160) admitted to Chengdu Seventh People's Hospital at the same term, 21 cases did not conform to the inclusion criteria, and 45 cases were ruled out by the exclusion criteria. At last, 94 healthy volunteers were enrolled in the Control group.
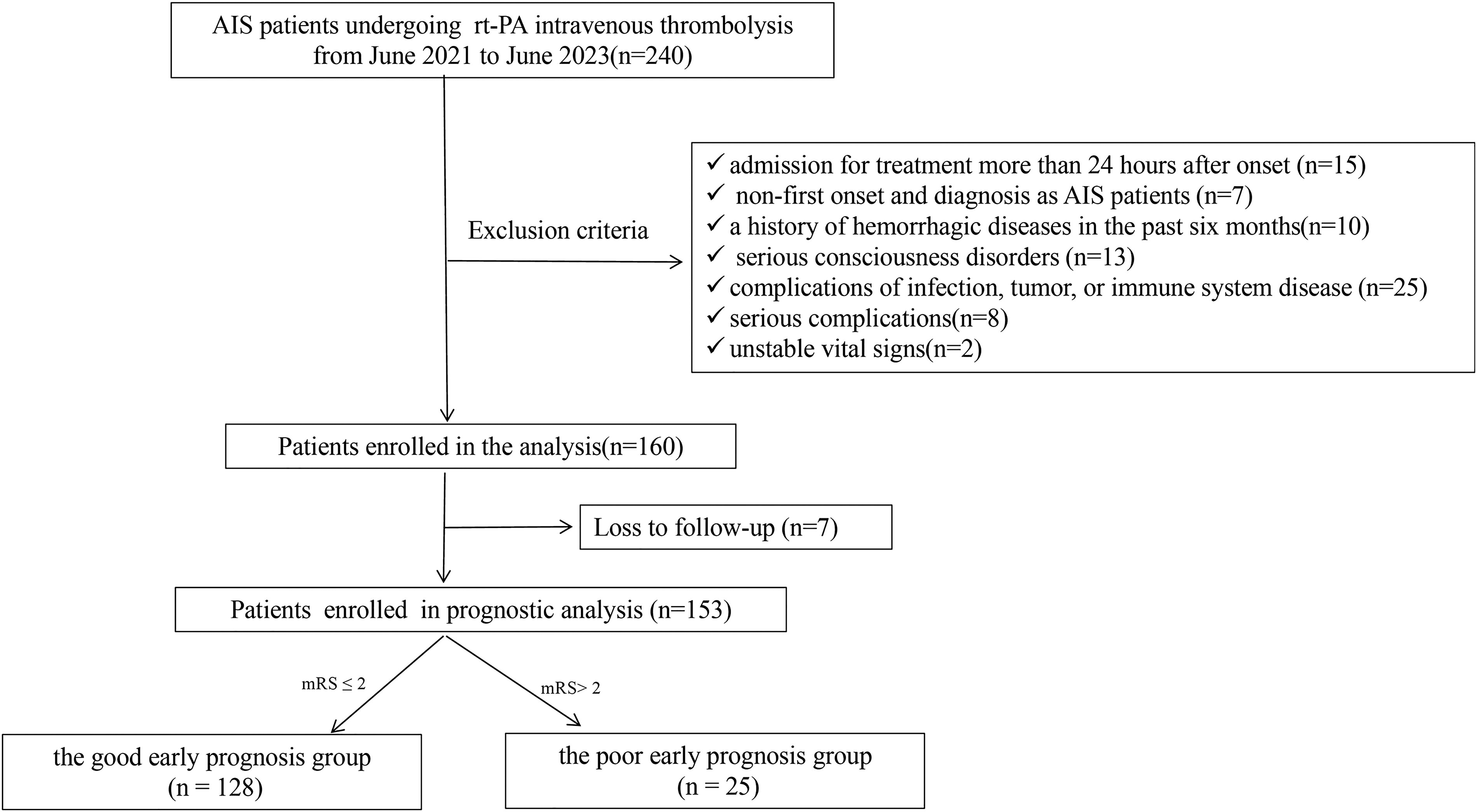
Patient flow chart of the cohort.
Inclusion and exclusion criteria
For the Study group, the inclusion criteria were as follows: (1) in line with the diagnostic criteria for AIS in the Chinese Guidelines for Diagnosis and Treatment of Acute Ischemic Stroke developed by the Cerebrovascular Disease Group of the Neurology Section of the Chinese Medical Association; (2) admission for treatment within 24 h of onset; (3) first onset and diagnosis as patients with AIS. Exclusion criteria were as below: (1) confirmed by imaging as cerebral hemorrhage or other pathological brain tissue diseases, such as cerebrovascular malformations and intracranial masses; (2) a history of hemorrhagic diseases in the past 6 months; (3) serious consciousness disorders; (4) complications of severe mental illness; (5) complications of infection, tumor, or immune system disease; (6) severe organ dysfunction; (7) concurrent with brain trauma; (8) serious complications; (9) pregnant or lactating women; (10) unstable vital signs.
Diagnostic criteria for AIS included acute onset, comprehensive or focal neurological deficits, symptoms and signs that persisted for several hours or more, the presence of responsibility infarct lesions confirmed by head computed tomography (CT) or magnetic resonance imaging (MRI) examination, and exclusion of cerebral hemorrhage and other brain lesions by transcranial CT or MRI.
As for the Control group, inclusion criteria comprised normal results for all indicators and a previously healthy body with no history of stroke, while exclusion criteria consisted of a history of severe organ dysfunction in the past, a history of infection, tumor, or immune system disease, a history of neurodegenerative diseases, intracranial infectious diseases, and intestinal inflammation, and pregnant or lactating women.
Sample and data collection
On the day of admission, baseline data on age, sex, body mass index (BMI), hypertension, diabetes, smoking, alcohol consumption, National Institutes of Health Stroke Scale (NIHSS) score, blood pressure, medication administration before disease onset (antiplatelet drugs, statins, anticoagulants, and hypoglycemic drug), stroke subtypes (large artery atherosclerosis, cardioembolism, small-artery occlusion, stroke of other determined etiology, and stroke of undetermined etiology), and location of lesion (anterior circulation and posterior circulation) were documented. Systolic blood pressure and diastolic blood pressure of patients were measured and recorded immediately after admission. The lesion location and stroke subtypes were evaluated by computed tomographic scan, magnetic resonance, electrocardiogram, echocardiography, carotid ultrasound, and transcranial Doppler. Stroke subtypes were classified according to the Trial of Org 10172 in Acute Stroke Treatment criteria (Adams et al., 1993). Subsequently, 5 mL fasting venous blood was gathered in ethylene diamine tetraacetic acid K2 anticoagulation vacuum tubes (GMS13017.2, Jiemei, Shanghai, China) and allowed to stand for 30 min, followed by 10-min centrifugation at 1,500 g. Later, the supernatant was collected and stored in a refrigerator at −80°C for enzyme-linked immunosorbent assay (ELISA).
ELISA
The levels of serum total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), uric acid (UA), serum creatinine (Scr), homocysteine (Hcy), C-reactive protein (CRP), Lp-PLA2, and peripheral blood inflammatory cytokines (IL-1β, IL-6, IL-10, and TNF-α) were tested by ELISA. The experiment was performed in strict accordance with the operating instructions of the TC kit (EH4163-CM, FineTest, Wuhan, Hubei, China), TG kit (LM-EL-1501, LMAI Bio, Shanghai, China), HDL-C kit (BLL-hlk1894, Baillie, Shanghai, China), LDL-C kit (BLL-hlk1892, Baillie), UA kit (HBP32112R, Shanghai Huabang Biotechnology Co., Ltd., Shanghai, China), Scr kit (GOY-H10488, Guyan, Shanghai, China), Hcy kit (FN-EH4011, FineTest), CRP kit (S0038-150 Beyotime, Shanghai, China), Lp-PLA2 kit (JLC22456, gelatins, Fuzhou, Jiangxi, China), IL-1β kit (CSB-E08053h-IS, CUSABIO, Wuhan, Hubei, China), IL-6 kit (LM-EL-1598, LMAI Bio), IL-10 kit (LM-EL-1310, LMAI Bio), and TNF-α kit (70-EK182-24, MultiSciences, Hangzhou, Zhejiang, China).
NIHSS score
The NIHSS was utilized to evaluate the neurological impairment of AIS patients upon admission. The scale consisted of the following 13 parts: consciousness level test (questions and commands), gaze and visual field, facial paralysis degree, upper and lower limb movement examination, ataxia examination (if ataxia existed, it was necessary to distinguish which limb was involved), existence or nonexistence of sensory disorder, language expression, normal or abnormal articulation, and existence or nonexistence of symptoms of neglect, with the total score ranging from 0 to 42 points. A higher score was indicative of a worse neurological deficit. According to the NIHSS score, the patients were allocated into the following two groups (Ma et al., 2023; Tsivgoulis et al., 2023): the mild group (NIHSS ≤ 5, n = 58); the moderate-to-severe group (NIHSS > 5, n = 102).
Follow-up and prognostic evaluation
After 6 months of admission, the early prognosis of AIS patients was assessed according to the modified Rankin Scale (mRS). The degree of disability of the patients was evaluated according to this scale (completely asymptomatic = 0 points; symptomatic but without significant functional impairment = 1 point; mild disabled = 2 points; moderate disabled = 3 points; moderately-to-severe disabled = 4 points; severe disabled = 5 points), which resulted in the classification of the good early prognosis group (mRS ≤ 2) and the poor early prognosis group (mRS > 2). At the same time, patients presenting with death were also categorized in the poor early prognosis group (mRS set at 6 points).
Statistical analysis
SPSS statistical software (21.0, IBM Corp., Armonk, NY, USA), MedCalc software (20.0, MedCalc software Ltd., Ostend, Belgium), and GraphPad Prism software (8.0.1, GraphPad Software Inc., San Diego, CA, USA) were employed for statistical analysis and graphing of data. The Kolmogorov–Smirnov test was utilized to examine the normal distribution. The measurement data of normal distribution were expressed as the mean ± standard deviation (SD), with intergroup comparisons implemented using the independent sample t-test, and comparisons among multiple groups implemented using one-way analysis of variance, followed by Tukey’s multiple comparison test. The measurement data of nonnormal distribution were expressed by quartile [median value (minimum, maximum)], with the Mann–Whitney U test applied for intergroup comparisons, Kruskal–Wallis rank sum test used among multiple groups, and Tukey’s multiple comparison test conducted afterward. Counting data were represented by the number of cases, and the chi-square test was used for intergroup comparisons of the binary variables. The correlation analysis was performed using Spearman. Logistic regression analysis was used to determine the influencing factors for poor early prognosis in AIS. The predictive value of peripheral blood inflammatory cytokines and serum Lp-PLA2 for poor early prognosis in patients with AIS was analyzed using receiver operating characteristic (ROC) curves. A significance level of α = 0.05 was used, and all P-values were derived from two-sided tests, and P < 0.05 was considered a statistically significant difference.
Results
Baseline data characteristics of the enrolled population
The general data of the Study and Control groups were analyzed. The results (Table 1) showed that there was no significant difference in terms of age, sex, drinking, TC, TG, HDL-C, LDL-C, UA, or Scr between the two groups (all P > 0.05). The percentages of the subjects with high blood pressure, diabetes, and smoking history, and BMI and expression levels of Hcy and CRP in the Study group, were all higher than those in the Control group (all P < 0.05). In addition, we collected clinical assessment and medical history of patients with AIS, as well as stroke subtypes and lesion location, as shown in Table 1.
General Data of the Enrolled Population
The Kolmogorov–Smirnov test was used to test for normal distribution. Normally distributed measures were presented as mean ± SD, and intergroup comparisons were made using the independent sample t-test. Nonnormally distributed measurements were expressed as quartiles, i.e., the median (minimum, maximum) and intergroup comparisons were made using the Mann–Whitney U test. Counting data were expressed as the number of cases, and the chi-square test was utilized for comparing intergroup dichotomous variables.
BMI, body mass index; CE, cardioembolism; CRP, C-reactive protein; DBP, diastolic blood pressure; Hcy, Homocysteine; HDL-C, high-density lipoprotein cholesterol; LAA, large artery atherosclerosis; LDL-C, low-density lipoprotein cholesterol; NIHSS, National Institutes of Health Stroke Scale; SAO, small-artery occlusion; SBP, systolic blood pressure; Scr, serum creatinine; SOE, stroke of other determined etiology; SUE, stroke of undetermined etiology; TC, total cholesterol; TG, triglyceride; UA, uric acid.
Patients with AIS exhibited highly expressed IL-1β, IL-6, TNF-α, and Lp-PLA2 and lowly expressed IL-10
We further determined the expression levels of peripheral blood serum inflammatory cytokines (IL-1β, IL-10, TNF-α, and IL-6) and Lp-PLA2 using ELISA. According to the results (Fig. 2A–E), the expression levels of serum IL-1β, IL-6, IL-10, TNF-α, and Lp-PLA2 in the Study group were (12.23 ± 2.77), [1.56 (0.65, 4.03)], [3.53 (0.22, 6.43)], (119.34 ± 20.49), and (154.29 ± 45.75), respectively, while those of the Control group were (7.18 ± 1.45), [0.68 (0.34, 1.21)], [7.83 (1.35, 10.44)], (61.85 ± 5.93), and (88.15 ± 21.33). Serum IL-1β, Lp-PLA2, IL-6, and TNF-α levels were higher, whereas serum IL-10 level was lower in the Study group than in the Control group (all P < 0.001).
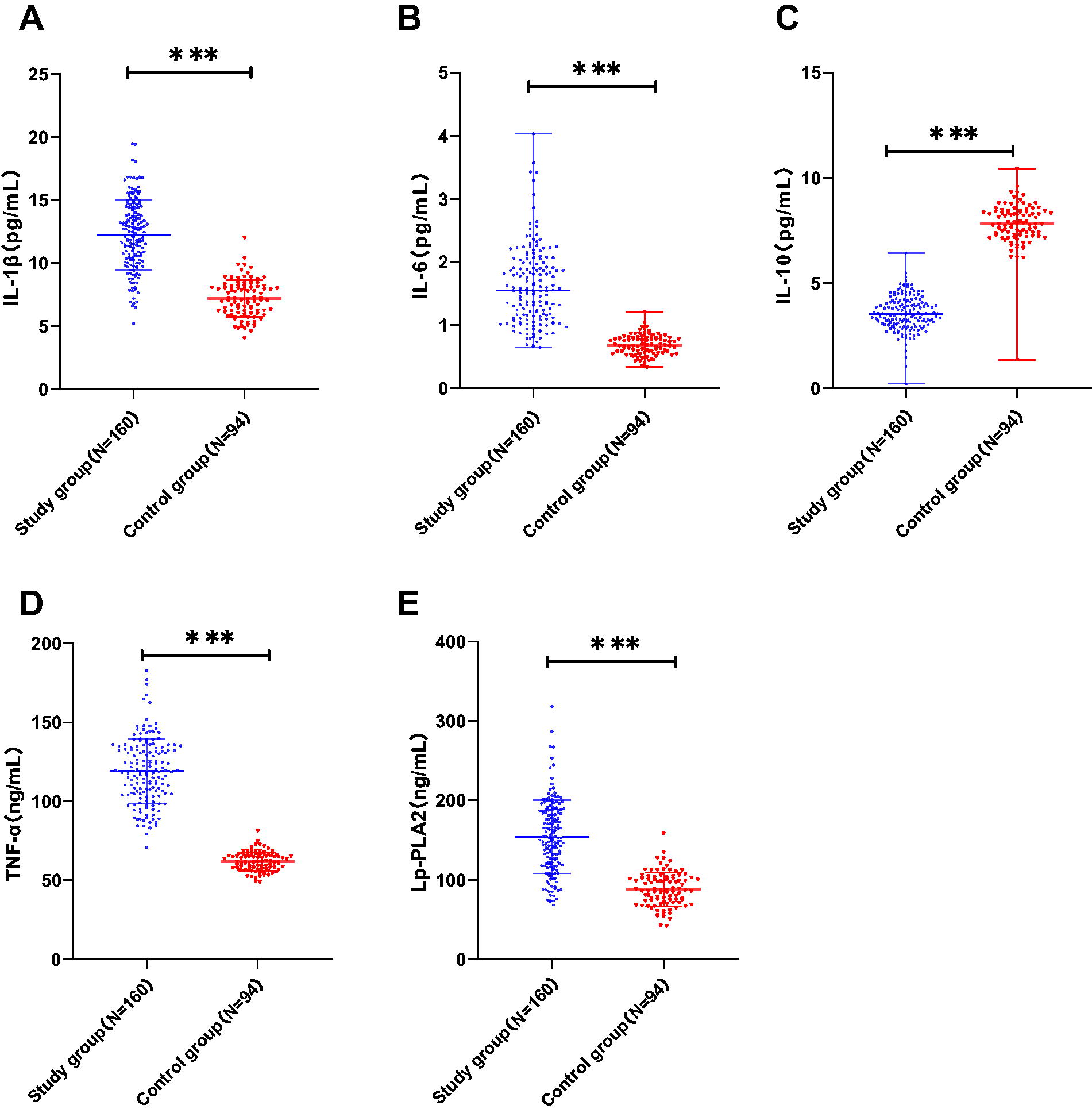
Patients with exhibited high expression levels of Lp-PLA2, IL-1β, IL-6, and TNF-α and low expression of IL-10. ELISA detection of peripheral blood serum IL-1β
Peripheral blood inflammatory cytokines and serum Lp-PLA2 altered in a dependent manner as the aggravation of AIS in patients
Subsequently, we assessed the degree of neurological impairment in patients with AIS using the NIHSS scale. Based on Spearman analysis (Fig. 3A–E), the NIHSS score of AIS patients was positively correlated with serum IL-1β (r = 0.556, P < 0.001, 95% CI = 0.435–0.657), IL-6 (r = 0.616, P < 0.001, 95% CI = 0.506–0.706), TNF-α (r = 0.532, P < 0.001, 95% CI = 0.407–0.637), and Lp-PLA2 (r = 0.526, P < 0.001, 95% CI = 0.400–0.633) expression levels, and negatively correlated with serum IL-10 expression [r = −0.273, P < 0.001, 95% CI = (−0.415)–(0.118)]. Thereafter, we assigned the patients with AIS to the mild group (n = 58) and the moderate-to-severe group (n = 102) and compared the inflammatory cytokine levels between the two groups. The results (Fig. 3F–J) elicited that serum IL-6, IL-1β, Lp-PLA2, and TNF-α levels in the moderate-to-severe group were higher than those of the mild group, and serum IL-10 exhibited an opposite trend (all P < 0.01). These results hinted that with the rise in neurological impairment severity in patients with AIS, serum Lp-PLA2, IL-1β, IL-6, and TNF-α levels rose, while serum IL-10 dropped in a dependent manner.
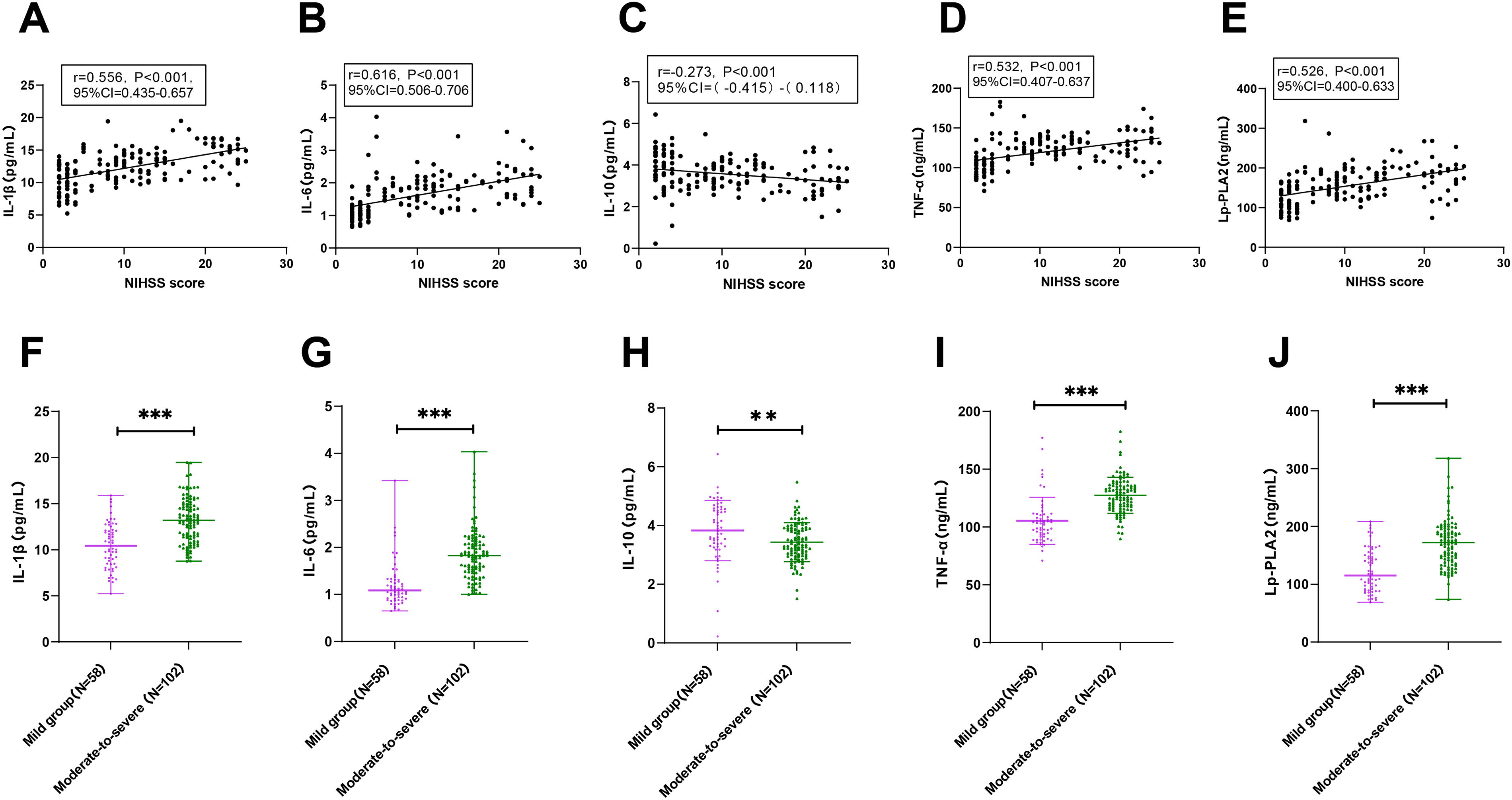
The expression levels of IL-1β, IL-6, IL-10, TNF-α, and Lp-PLA2 in patients with AIS with distinct degrees of neurological impairment. The relationship of NIHSS score with serum IL-1β
Risk analysis on peripheral blood inflammatory cytokines and Lp-PLA2 for the occurrence of poor early prognosis of patients with AIS
We regularly reviewed the health condition of patients with AIS 6 months after treatment. During the follow-up period, 7 patients were lost, and 153 patients were finally included for analysis. These patients were classified into the good early prognosis group (n = 128) and the poor early prognosis group (n = 25) according to the results of the mRS score. We compared the clinical baseline data of patients with AIS in the two groups. As shown in Table 2, compared with the good early prognosis group, patients in the poor early prognosis group had an increased proportion of diabetes, raised NIHSS score and serum IL-1β, IL-6, TNF-α, and Lp-PLA2 levels at admission, and reduced IL-10 levels (all P < 0.05); yet, there was no significant difference in age, sex, BMI, or other clinical baseline data between the two groups (all P > 0.05). Moreover, we included diabetes, NIHSS, IL-1β, IL-6, IL-10, TNF-α, and Lp-PLA2 in Table 2, with P < 0.05 as independent variables into the logistic multivariate regression analysis, in a bid to explore the independent risk factors for the occurrence of poor early prognosis in patients with AIS. As reflected by the results (Table 3), after adjusting diabetes and NIHSS, IL-1β [odds ratio (OR) = 6.836, P = 0.017, 95% CI = 1.413–33.068], IL-6 (OR = 85.108, P = 0.017, 95% CI = 2.217–3267.151), TNF-α (OR = 1.150, P = 0.039, 95% CI = 1.007–1.313), and Lp-PLA2 (OR = 1.044, P = 0.037, 95% CI = 1.003–1.087) were the risk factors for the poor early prognosis of patients with AIS, and IL-10 (OR = 0.001, P = 0.015, 95% CI = 0.000–0.252) was a protective factor for poor early prognosis in patients with AIS.
General Clinical Data of Acute Ischemic Stroke Patients with Different Prognoses
The Kolmogorov–Smirnov test was used to test for normal distribution. Normally distributed measures were presented as mean ± SD, and intergroup comparisons were made using the independent sample t-test. Nonnormally distributed measures were expressed as quartiles, i.e., the median (minimum, maximum).
BMI, body mass index; CE, cardioembolism; CRP, C-reactive protein; DBP, diastolic blood pressure; Hcy, Homocysteine; HDL-C, high-density lipoprotein cholesterol; IL, interleukin; LAA, large artery atherosclerosis; LDL-C, low-density lipoprotein cholesterol; Lp-PLA2, lipoprotein-associated phospholipase-A2; NIHSS, National Institutes of Health Stroke Scale; SAO, small-artery occlusion; SBP, systolic blood pressure; Scr, serum creatinine; SOE, stroke of other determined etiology; SUE, stroke of undetermined etiology; TC, total cholesterol; TG, triglyceride; TNF-α, tumor necrosis factor-α; UA, uric acid.
Logistic Multivariate Regression Analysis of the Factors Influencing Poor Early Prognosis in Patients with Acute Ischemic Stroke
CI, confidence interval; IL, interleukin; Lp-PLA2, lipoprotein-associated phospholipase-A2; NIHSS, National Institutes of Health Stroke Scale; OR, odds ratio; TNF-α, tumor necrosis factor-α.
Predictive value of peripheral blood inflammatory cytokines combined with serum Lp-PLA2 for adverse early prognosis in patients with AIS
Afterward, we compared whether peripheral blood inflammatory cytokines combined with serum Lp-PLA2 could improve the ability of NIHSS score to predict poor early prognosis in AIS patients. We plotted ROC curves to analyze the predictive value of peripheral blood inflammatory cytokines (IL-6, IL-1β, TNF-α, and IL-10), serum Lp-PLA2, NIHSS, and their combined detection for unfavorable early prognosis in patients with AIS. Based on the ROC curve analysis (Fig. 4), serum IL-1β [area under the curve (AUC) = 0.892], IL-6 (AUC = 0.910), IL-10 (AUC = 0.773), TNF-α (AUC = 0.679), Lp-PLA2 (AUC = 0.865), and NIHSS score (AUC = 0.918) all had predictive value for unfavorable early prognosis in patients with AIS. Furthermore, as illustrated by the MEDCALC analysis (Table 4), the combined detection of TNF-α, IL-1β, IL-10, IL-6, Lp-PLA2, and NIHSS demonstrated considerably higher predictive value for the adverse early prognosis in patients with AIS than the single detection of these indicators (all P < 0.05). The results indicated that the combined detection of peripheral blood inflammatory cytokines and serum Lp-PLA2 could elevate the predictive value of NIHSS for poor early prognosis in patients with AIS.
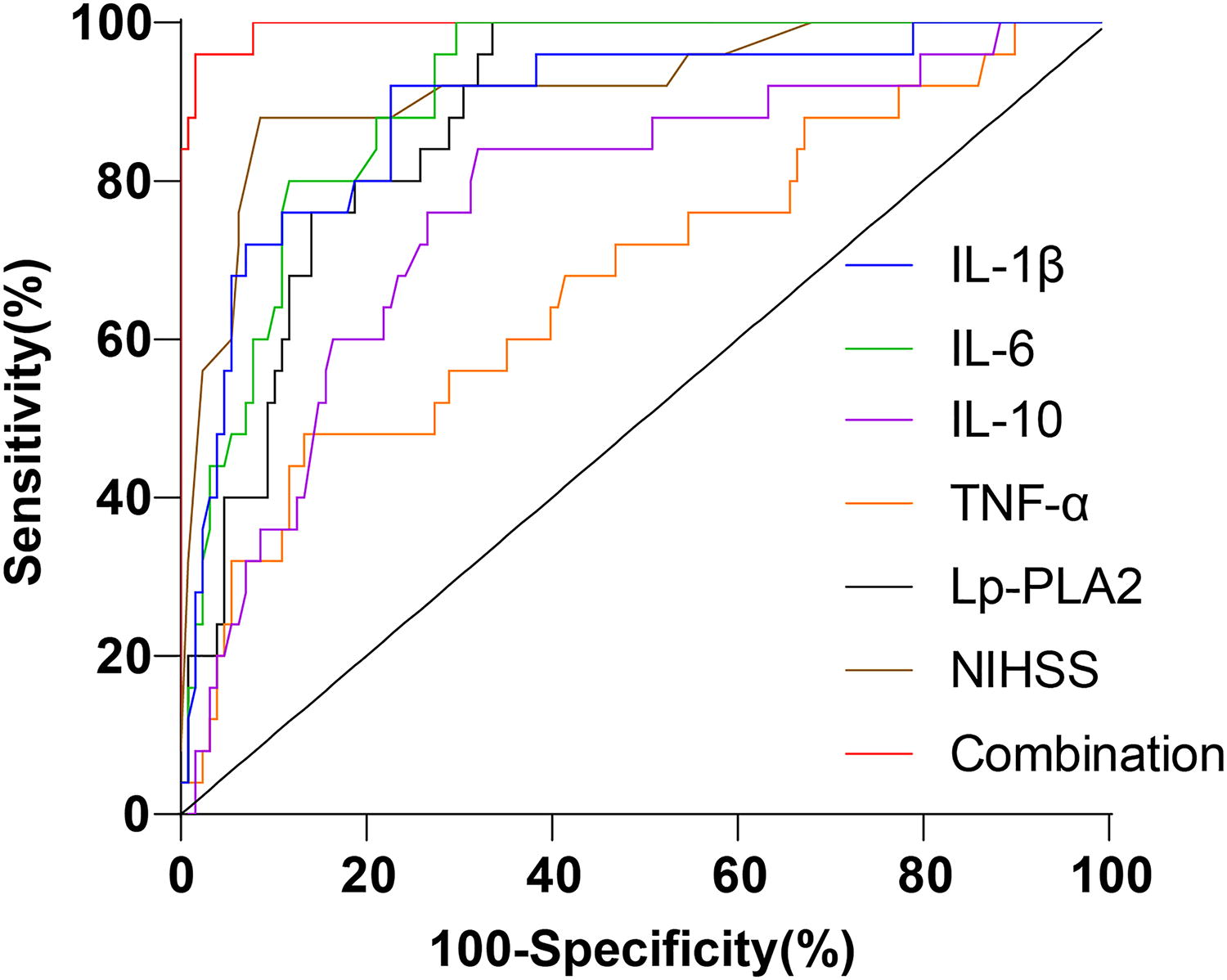
The predictive value of peripheral blood inflammatory cytokines paired with serum Lp-PLA2 for the unfavorable early prognosis of patients with AIS. The ROC curves were plotted to analyze the predictive value of peripheral blood inflammatory cytokines (IL-1β, TNF-α, IL-6, and IL-10), serum Lp-PLA2, NIHSS, and their combined detection for the adverse early prognosis of patients with AIS. AIS, acute ischemic stroke; IL, interleukin; Lp-PLA2, lipoprotein-associated phospholipase-A2; NIHSS, National Institutes of Health Stroke Scale; ROC, receiver operating characteristic; TNF-α, tumor necrosis factor-α.
The Predictive Value of Peripheral Blood Inflammatory Cytokines Combined with Serum Lp-PLA2 Detection for Early Poor Prognosis in Patients with Acute Ischemic Stroke
Multiple ROC AUC comparisons were performed using the Delong test from MEDCALC software (20.0, MedCalc software Ltd., Ostend, Belgium).
AUC, area under the curve; CI, confidence interval; IL, interleukin; Lp-PLA2, lipoprotein-associated phospholipase-A2; NIHSS, National Institutes of Health Stroke Scale; TNF-α, tumor necrosis factor-α.
Discussion
The inflammatory environment of the nervous system caused by stroke is currently established as one of the main determinants of brain parenchymal damage. The expression of inflammatory parameters may signify the importance of the inflammatory process in the early phase of IS for the poststroke neurological deficit (Lasek-Bal et al., 2019). In this regard, as a possible prognostic marker and therapeutic target for stroke patients, inflammatory mediators have been studied for a long time, which may be a vital target for improving the management of stroke patients (Tirandi et al., 2023; Tuttolomondo et al., 2008). Prior research suggests that increased IL-6 and reduced IL-10 levels in the early stage of AIS are linked with the extent of neurological deficit and/or stroke prognosis (Basic Kes et al., 2008). Also, high serum Lp-PLA2 levels are thought to correlate with AIS onset, disease severity, and relapse and can be used to guide clinical practice (Li et al., 2021). Meanwhile, increased Lp-PLA2 levels have been demonstrated to be independent of other risk variables and correlate with all-cause mortality within 1 year following AIS (Han et al., 2017). In our study, we investigated whether peripheral blood inflammatory cytokines (IL-1β, IL-6, IL-10, and TNF-α) in combination with Lp-PLA2 could increase the ability to predict clinical outcomes in AIS. The study has supplemented recent evidence from June 2021 to June 2023, suggesting that combining peripheral blood levels of inflammatory cytokines (IL-1β, IL-6, IL-10, and TNF-α) and Lp-PLA2 may help to improve risk stratification in patients with AIS who receive intravenous thrombolysis. Moreover, our findings showed that the level of Lp-PLA2, which mediates the inflammatory response, may play an important role in the pathophysiology of AIS, highlighting its potential as a diagnostic and therapeutic target for patients with AIS after intravenous thrombolysis.
Patients diagnosed with AIS frequently exhibit clinical manifestations such as hemiplegia and aphasia, which profoundly diminish their quality of life and pose a threat to their overall well-being (Zhu et al., 2020). Several studies have shown that the inflammatory response and certain inflammatory variables are important contributors to the progression of AIS (Bonaventura et al., 2016; Li et al., 2019). Therefore, enhancing the comprehension of the relationship between inflammatory markers and stroke is crucial for developing more efficient preventive, prognostic, and therapeutic strategies (Rezaeitalab et al., 2020). The current study mainly described that AIS patients exhibited dependent increases in serum TNF-α, IL-6, Lp-PLA2, and IL-1β levels, while dependent decreases in serum IL-10 as the disease progressed. The combined use of peripheral blood inflammatory cytokines and Lp-PLA2 could increase the predictive value for adverse early AIS prognosis compared with when used alone.
Several inflammatory cytokines, including TNF-α, IL-6, IL-10, and IL-1β, have been associated with AIS and studied as potential therapeutic targets and prognostic biomarkers (Doll et al., 2014). Besides, a recent investigation has validated that Lp-PLA2 is a novel inflammatory factor specific to blood vessels, which can serve as an early biomarker for predicting and diagnosing AIS (Cao et al., 2021). In this study, we analyzed the expression patterns of IL-6, IL-10, IL-1β, TNF-α, and Lp-PLA2 in patients with AIS and their relationship with AIS severity. In line with the findings of our study, elevated expression levels of Lp-PLA2, TNF-α, IL-1β, and IL-6 are observed in individuals with AIS relative to healthy controls, and these cytokines are associated with aggravated disease severity and poor outcomes (Lin et al., 2018; Liu et al., 2023; Wytrykowska et al., 2016). As stated previously, the anti-inflammatory cytokine IL-10 has the potential to suppress the production of proinflammatory cytokines, making them a potential therapy option for stroke (Sun et al., 2021). Other researchers have consistently observed a reduction in IL-10 levels following the onset of AIS, compared with the controls, and have linked the decreased level to neurological impairment and an unfavorable IS prognosis (Chi et al., 2021; Xie et al., 2013). Another cross-sectional investigation also found a strong correlation between a higher risk of IS and lower IL-10 concentration and selected genetic variants (Xie et al., 2013).
Furthermore, the application of the NIHSS also validates the connection between these cytokines and the extent of neurological damage in patients with AIS. The NIHSS is the most dependable grading system for predicting the outcome of a stroke, with higher scores indicating more severe neurological impairments (Khan et al., 2022; Nedeltchev et al., 2010). Notably, we found that the NIHSS score was positively related to TNF-α, Lp-PLA2, IL-1β, and IL-6 and negatively related to IL-10 in patients with AIS, which is mostly in line with what various existing studies have found (Bayat et al., 2023; Cao et al., 2021, 2023; Pawluk et al., 2020; Xin et al., 2022). Moreover, Lp-PLA2 is identified as a risk factor for early neurological impairment in patients with AIS (Wang et al., 2019). There is convincing proof that IL-6, IL-1β, and TNF-α are significant risk factors for recurrent IS and are associated with a worse prognosis in stroke patients (Licata et al., 2009; Smith et al., 2018; Welsh et al., 2008). Furthermore, a mouse stroke model has confirmed the neuroprotective function of IL-10 (Liesz et al., 2014). The lower level of IL-10 has been linked to both the intensity of symptoms and a negative possibility for recovery in patients with IS, underscoring that IL-10 is an essential factor in predicting patient outcomes (Sun et al., 2021). Altogether, it is reasonable that TNF-α, Lp-PLA2, IL-1β, and IL-6 are risk factors for poor early prognosis in patients with AIS, and IL-10 is a protective factor against poor early prognosis.
Importantly, as reflected by ROC curve and MEDCALC analysis, the combined use of IL-1β, IL-10, TNF-α, NIHSS, Lp-PLA2, and IL-6 had notably elevated predictive value for undesirable early prognosis in patients with AIS versus when used alone. Evidence is accumulating regarding the optimal prediction of the early prognosis of AIS by a combination of markers that have already been reported. For instance, Chang et al. have confirmed that the combination of the ankle-brachial index, miR-103, and Lp-PLA2 exhibits a superior predictive capacity for the unfavorable prognosis of AIS compared with every single indicator (Chang et al., 2024). In addition, the combination of IL-6, IL-8, and modified early warning score can serve as a momentous indicator for assessing the severity of early AIS, and it shows greater accuracy in evaluating the early prognosis of patients with AIS than the detection of a single indicator (Zhang et al., 2021b). To the best of our knowledge, this study has demonstrated for the first time that the combination of peripheral blood inflammatory cytokines, Lp-PLA2, and NIHSS had the highest predictive value for AIS adverse prognosis at the early stage. Therefore, the detection of peripheral blood inflammatory cytokines combined with serum Lp-PLA2 could increase the predictive value of NIHSS for poor early prognosis in patients with AIS.
In conclusion, we observed high expression levels of IL-6, IL-1β, TNF-α, and Lp-PLA2 and low expression of IL-10 in patients with AIS, with the alterations intensified in a dependent manner as the disease exacerbated in patients with AIS. Also, we demonstrated the high predictive value of the combined evaluation of peripheral blood inflammatory cytokines, Lp-PLA2, and NIHSS for adverse early prognosis in patients with AIS, which provided theoretical guidance for the assessment of the condition of patients with AIS and the prediction of early prognostic development. On the other hand, we also recognize that this study has its limitations. First, this study is a single-center study with limited sample selection and the possibility of potential selection bias. Second, due to the short sample collection time, we did not thoroughly assess the relationship between peripheral blood inflammatory cytokines and serum Lp-PLA2 expression in patients with AIS, which limited the investigation of long-term prognostic development. Third, we did not adequately control for potential confounding variables, such as underlying health status or lifestyle factors, which might influence inflammatory cytokine and Lp-PLA2 levels in patients with AIS and the controls. Fourth, this study only focused on the levels of these biomarkers before treatment. At last, due to the limitation of the sample collection period, we are temporarily unable to obtain enough patient samples as an external validation cohort for further research and analysis. To fill these research gaps, we will conduct further multicenter studies to expand the sample size and exclude potential selection bias for probing the impact and predictive value of these indicators for the long-term prognosis of patients with AIS. In addition, we will include more potential confounding variables for analysis, further explore changes in biomarker levels at the end of treatment and during follow-up, and conduct external cohort validation, along with analysis of the interplay between peripheral blood inflammatory cytokines and Lp-PLA2, so as to provide effective help for the exploration of the pathogenesis of AIS.
Footnotes
Data Availability Statement
The data supporting the findings of this study can be obtained from the corresponding author upon request.
Submission Declaration and Verification
The work presented in this study has not been previously published, is not being considered for publication elsewhere, has been approved for publication by all authors and the relevant authorities, and will not be published in any other form without consent.
Ethical Approval
This study adheres to the ethical criteria outlined in the World Medical Congress Declaration of Helsinki, complies with the relevant norms and regulations for clinical research, and is in accordance with the guidelines provided by the Enhancing the Quality and Transparency of Health Research network. This study was approved by the academic ethics committee of Chengdu Seventh People's Hospital [the ethical approval number (KY2024-048-01)]. All patients involved in this study were fully informed of the purpose of this study and signed informed consent before sampling.
Authors’ Contributions
All authors contributed to the study conception and design, and provided feedback on previous versions of the article. All authors reviewed and approved the final version. C.Z. and Q.H. were responsible for ensuring the integrity of the entire research, defining knowledge content, conducting literature research, experimental research, data analysis, article preparation, and editing. L.Y. and Y.L. ensured the integrity of the research and defined knowledge content, contributing to literature research, experimental research, and statistical analysis. Y.Z. and S.L. contributed to data acquisition. J.L. was involved in conceptualization, writing, review, and editing. J.J. oversaw study concepts, design, and article review.
Author Disclosure Statement
The authors have no conflicts of interest to declare.
Funding Information
This research is partially granted by Sichuan Medical Youth Innovation Research Project (No. Q23089).
