Abstract
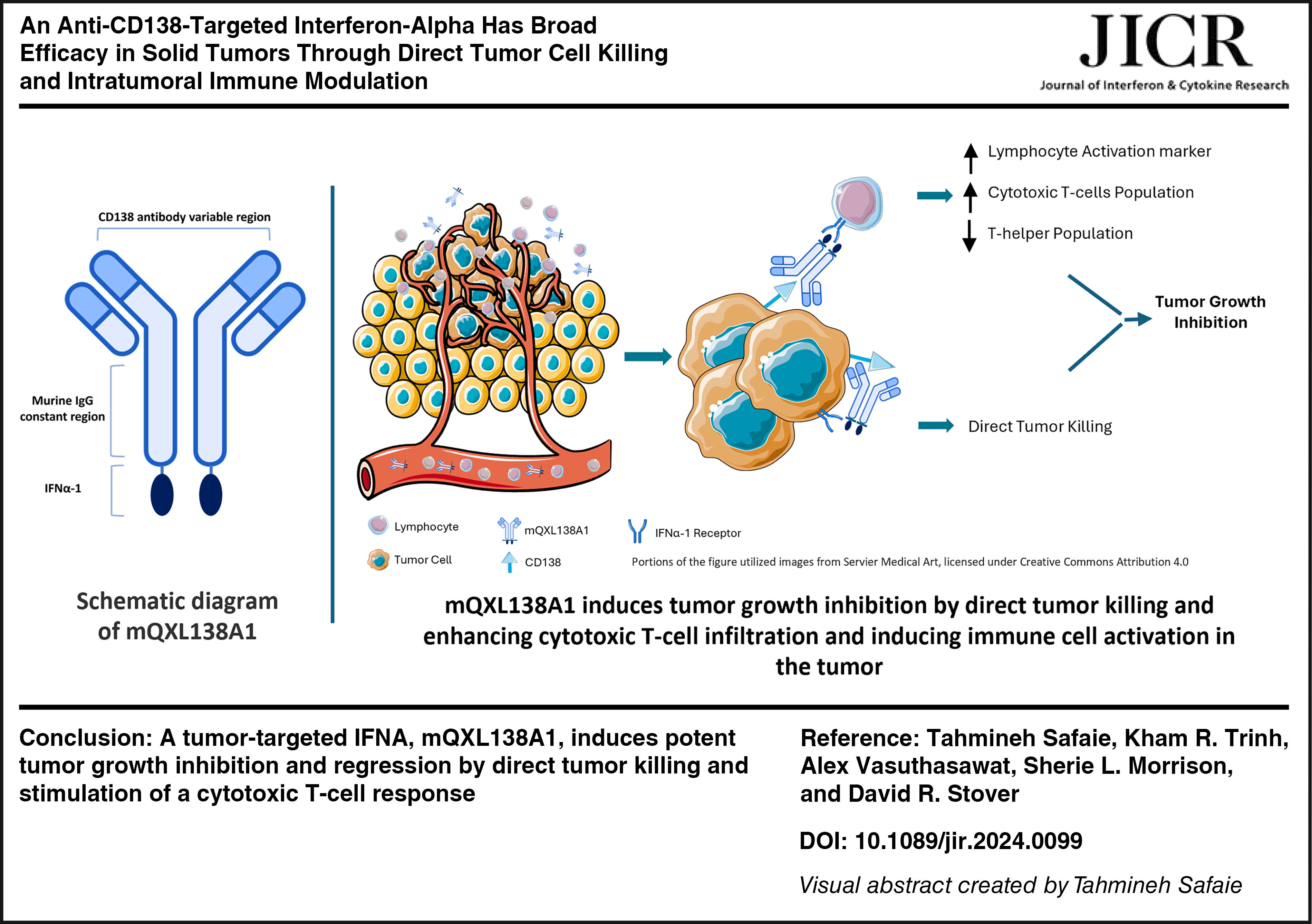
Among cytokines, interferons (IFNs) have demonstrated antitumor effects through several mechanisms, making them attractive candidates for cancer immunotherapies. However, the application of interferons in cancer treatment comes with some obstacles, such as high toxicity owing to systemic exposure and a short half-life. The mQXL138A1 fusion protein is a murine interferon alpha-1 (IFN-α-1) fused to an antimurine Syndecan-1 (CD138) antibody designed with the aim of targeted delivery of IFN-α-1 to CD138-expressing tumor cells to enhance treatment efficacy and decrease toxicity. mQXL138A1 maintained its CD138 binding properties and exhibited IFN-dependent activity in vitro. mQXL138A1 exhibited antitumor efficacy across various syngeneic mouse models and minor levels of toxicity. Tumor growth inhibition was achieved through a combination of direct tumor cell killing and a shift in the immune activation profile of the tumor microenvironment. Although this study showed that tumor-targeted IFN-α-1 had broad antitumor properties across a range of solid tumor indications, toxicities associated with systemic activity remain an issue.
Get full access to this article
View all access options for this article.
