Abstract
There is no sensitive and effective method to predict radiation-induced myocardial damage (RIMD). The aim of this study was to explore effective plasma biomarkers for early prediction of RIMD after radiotherapy (RT) in lung cancer patients and in a rat model. Biomarker levels were measured in plasma samples collected before and after thoracic RT from 17 lung cancer patients. For the animal model, a single radiation dose of 40 Gy was delivered to the cardiac apex of female Wistar rats. Control rats received sham irradiation (0 Gy). Dynamic plasma biomarker detection and histopathological analysis to confirm RIMD were performed in rats up to 6 months after RT. In lung cancer patients, the plasma caspase-3 concentration was significantly increased after thoracic RT (P = 0.0479), with increasing but nonsignificant trends observed for caspase-1, CCL2, vascular endothelial growth factor (VEGF), interleukin-1β, and IL-6 (P > 0.05). Changes in caspase-3, VEGF, and IL-6 correlated significantly with mean heart dose (P < 0.05). In the RIMD rat model, caspase-1, caspase-3, CCl-2, VEGF, CCl-5, and TGF-β1 levels were significantly elevated in the first week post-RT (P < 0.05), which was earlier than pathological changes. Myocardial tissue of the RIMD rats also showed significant macrophage infiltration at 1 month (P < 0.01) and fibrosis at 6 months postradiation (P < 0.0001). Macrophage infiltration correlated significantly with plasma caspase-3, CCL2, CCL5, VEGF, and TGF-β1 levels from 3 weeks to 2 months post-RT. Increased plasma caspase-1, caspase-3, CCl-2, and VEGF levels were detected before RIMD-related pathological changes, indicating their clinical potential as biomarkers for early prediction of RIMD.
Introduction
Cardiotoxicity is a well-recognized, serious adverse effect of thoracic radiotherapy (RT). The association of radiation exposure with long-term cardiotoxicity is well established, with decades of follow-up data demonstrating increased morbidity and mortality related to cardiac disease in long-term survivors of breast cancer, lymphoma, and lung cancer who received RT (Aleman and others 2007; Darby and others 2013; Dess and others 2017; Atkins and others 2019). Although overt cardiac disease most typically occurs 5 years or more after radiation exposure, prior studies have indicated that subclinical structural and functional abnormalities may occur early after RT (Recht, 2017; Saiki and others 2017; Clasen and others 2021). Early prediction of radiation-induced myocardial damage (RIMD) may provide critical guidance for the prevention and treatment of cardiotoxicity. However, currently, there is no effective method predicting RIMD soon after RT in the clinic.
Pathology is considered the gold standard approach for accurate evaluation of RIMD, but its feasibility and accessibility are limited. If any, pathological changes caused by RT usually occur later, possibly missing the optimal treatment opportunity (Christersdottir and others 2019). In contrast, plasma biomarkers are relatively easy to measure in patients and can be dynamically detected. Therefore, there has been a growing interest in biomarkers that can be used to diagnose or predict cardiotoxicity in the field of radiation oncology. Early detection of such biomarkers in patients, indicating increased risk of RIMD, would be useful for reducing the detrimental effects of RT-induced cardiotoxicity through aggressive and targeted therapeutic interventions.
So far, several studies have evaluated early changes in biomarker levels in patients receiving thoracic RT, particularly high-sensitivity cardiac troponin T and N-terminal pro-B-type natriuretic peptide (Gomez and others 2014; Chalubinska-Fendler and others 2019; Demissei and others 2019). However, the research to date has not yet provided conclusive evidence of biomarker relevance; hence, their potential value as RIMD markers remains uncertain.
Therefore, in the present study, we evaluated the effectiveness of several plasma biomarkers for early prediction of RIMD that have not been investigated in this context previously. Prior studies have indicated that aberrant activation of the NLR family pyrin domain-containing protein 3 (NLRP3) inflammasome is associated with the pathogenesis of various inflammatory diseases, such as diabetes, cancer, and Alzheimer's disease, and its activation leads to caspase-dependent secretion of proinflammatory cytokines such as interleukin-1β (IL-1β), IL-1α, and IL-6 (Liu and others 2018; Christersdottir and others 2019; Huang and others 2021) and the expression of chemokines (CCL2 and CCL5) and adhesion molecules (Gosling and others 1999; Moore and others 2013; Christersdottir and others 2019).
Vascular endothelial growth factor (VEGF) is known to be a principal proangiogenic factor that enhances the production of new blood vessels and plays an essential role in embryonic development, tissue remodeling, and cancer progression (Matsumoto and Ema 2004; Siveen and others 2017). Transforming growth factor-β (TGF-β) plays multiple homeostatic roles in the regulation of inflammation, oxidative stress, tissues injury, and remodeling and is widely used in the diagnosis and treatment of various diseases characterized by inflammation and fibrosis (Hanna and Frangogiannis 2019; Ong and others 2021). We postulated that these processes are integral components of the pathophysiologic changes that occur early in the pathogenesis of RIMD. Accordingly, these inflammatory factors that are reflective of a diverse array of relevant biologic processes may provide important insight into the risk for RIMD.
In this study, we chose a panel of inflammatory factors and measured their plasma concentrations before and after RT in human lung cancer patients and a rat model of RIMD, applying pathology as the gold standard for RIMD detection, to explore correlations between the dynamic changes of these inflammatory factors after RT and the risk of RIMD.
Methods
Ethical approval of the study protocol
This study was approved by the Ethics Committee of Shandong Cancer Hospital (protocol no. SDTHEC2022011007). The animal experiments and procedures were performed with the approval of the Shandong Cancer Hospital and Institute of Medicine Institutional Animal Care and Use Committee.
Patient selection and human plasma collection
Lung cancer patients treated with thoracic RT were enrolled in this prospective longitudinal cohort study from January 2021 to March 2022. All patients were treated with intensity-modulated radiation therapy. The population prescribed dose ranged from 40–70 Gy in 1.8–2.0 Gy fractions. The mean radiation dose delivered to the heart was evaluated on dose-volume histograms in the treatment-planning system. Plasma samples from both before and after RT were available for a total of 17 lung cancer patients, who were then included in the analysis of this study. Blood samples were collected before and after RT (median, 2 months) and stored at −80°C. In addition, electrocardiography (ECG) was performed for all patients at both the pre- and post-RT visits.
Rat model of RIMD
Female Wistar rats (200 ± 20 g, 8–10 weeks old) were maintained in our institution's animal facilities. Rats were subjected to image-guided RT using the Small Animal Radiation Research Platform (SARRP) and irradiated with a single dose of 40 Gy. The cardiac apex was irradiated using a 10 × 10-mm2 collimated beam operating via a CBCT image at 220 kV, 13 mA with copper filtration. An area of the heart apex measuring ∼8 mm2 was used as the target area for irradiation (Supplementary Fig. S1). Control rats aged 8–10 weeks received sham irradiation (0 Gy), and the relevant experiments were carried out 1 week after the sham irradiation.
The SARRP Control and MuriPlan software programs were used to precisely target hearts with precise irradiation doses. Rats were anesthetized by intraperitoneal injection of 1% pentobarbital sodium for the duration of each RT. Subsequently, plasma samples and heart tissues from rats were collected at various time points after RT for further analysis. Four rats were used at each time point for any given study.
Rat plasma collection
Whole blood was collected into heparinized collecting tubes containing ethylenediaminetetraacetic acid from the tail vein of each rat at various time points after RT. Plasma was obtained by centrifugation for 10 min at 2,000 rpm using a refrigerated centrifuge. Plasma samples were stored at −80°C until further analysis.
Quantification of plasma biomarker levels
The concentrations of the following potential biomarkers in human and rat plasma samples were analyzed: caspase-1, caspase-3, CCl-2, CCl-5, VEGF, TGF-β1, IL-1α, IL-1β, IL-6, and NLRP3. The levels of these cytokines were measured by enzyme-linked immunosorbent assay using a microplate reader (HBS-1096C; De Tie) according to the manufacturer's protocol. In brief, diluted samples and standards were added to the plate wells. After incubation, the plate was washed to remove nonspecific proteins. Then, antibody specific to the factor were added. After removing unattached material by washing the wells, Streptavidin-horseradish peroxidase (HRP) solution was added. After incubation, the plate was washed and the substrate solution, which reacts with HRP, was added. The intensity of the color evolved by the reaction corresponded to the factor concentration of the sample. The optical density value of samples was detected in a microplate reader at 450 nm wavelength. The levels of these cytokines were calculated according to the standard curve.
Masson's trichrome and immunohistochemical staining
Cardiac tissues of rats were fixed in 10% formalin overnight, embedded in paraffin, and later sectioned into 3–5 μm thick sections. Sections were subjected to Masson's trichrome and immunohistochemical (IHC) staining. Macrophages in cardiac tissues were stained using a rat-specific anti-CD68 antibody (1:500 dilution, ab-125212; Abcam). The tissue section was incubated with anti-CD68 antibody for 3 h at room temperature. HRP-conjugated goat anti-rat IgG (1:200 dilution, ab-97057; Abcam) was used as the secondary antibody and incubated for 30 min at room temperature. The extent of myocardial fibrosis was visualized by Masson's trichrome staining. Cardiac cross sections were stained using a Masson's Trichrome kit (Abcam; ab-150686) following the protocol included.
This staining procedure serves to stain collagen and mucus blue, while muscle and cytoplasm should stain red with nuclei staining blue-black. All slices were scanned with a digital scanner (Zeiss) for image acquisition. For each section, whole heart fibrosis was evaluated by drawing an area of interest around the stained cardiac tissue and then counting the number of CD68-positive cells per unit area in the region of interest. The positively stained fibrosis area and the number of positively stained macrophages were semiquantitatively analyzed by an experienced pathologist.
Statistical analysis
For the analysis of biomarker levels in patients' plasma, a paired t-test was used to identify differences in normally distributed data before and after RT, and Wilcoxon signed-rank test was used to analyze the differences in non-normally distributed data before and after RT. Linear regression was used to evaluate the correlations between biomarker changes pre- and post-RT and the mean heart dose in lung cancer patients. The biomarker, macrophage, and fibrosis levels in rats at different time points after RT were compared with those in the control group using one-way analysis of variance (ANOVA). The measurement data are expressed as mean ± standard deviation (SD) values. Correlations between biomarker levels and pathological findings were explored using Spearman correlation analysis. A 2-sided alpha level of 0.05 was applied to assess statistical significance. All analyses were performed using SPSS version 23.0 and GraphPad Prism 9.02.
Results
Altered biomarker levels in plasma of lung cancer patients after RT
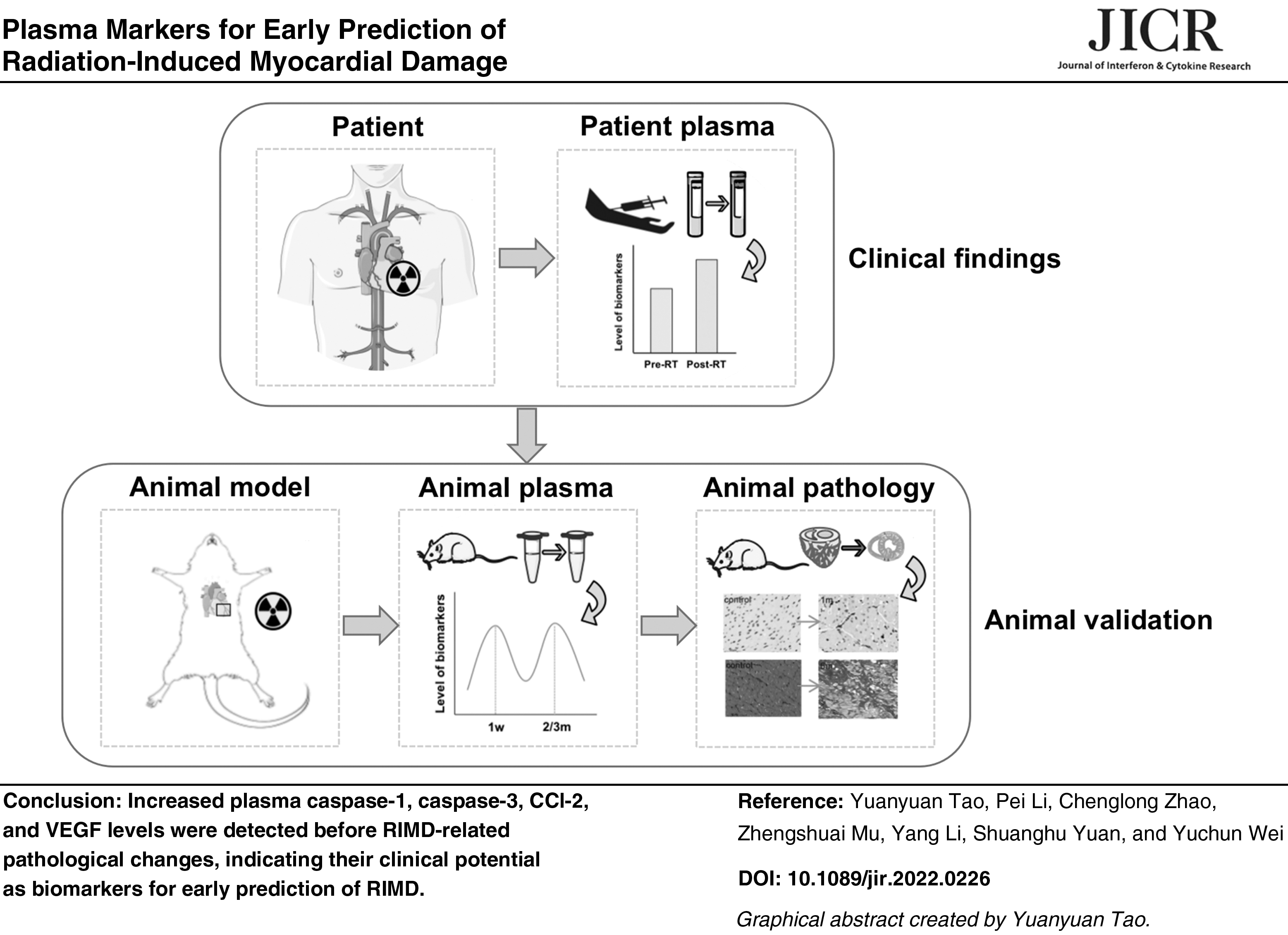
The clinical characteristics of the lung cancer patients included in this study are summarized in Table 1. Potential biomarker levels were measured in plasma samples collected from these lung cancer patients before and after RT (median 2 months). Changes in the levels of individual biomarker from pre-RT to post-RT are presented in Fig. 1. In lung cancer patients, the plasma levels of caspase-1, CCL2, VEGF, IL-1β, and IL-6 showed increasing trends after RT; however, no statistically significant differences in the levels of these biomarkers were found between the 2 time points. In contrast, a significant increase in the plasma level of caspase-3 was observed from pre-RT to post-RT (3.95 ± 2.34 versus 6.93 ± 5.43, P = 0.0479).
Changes in plasma biomarker levels from pre-RT to post-RT in lung cancer patients. Histograms depict biomarker levels at each time point; gray lines show individual changes in biomarker levels from pre-RT to post-RT (n = 17).
Patient Characteristics (n = 17)
Heart diseases were coronary artery disease and myocardial ischemia.
ECG changes were T-wave changes (2), sinus tachycardia (2), ST changes (1), prolonged PR interval (1), and poor R-wave progression (1)
ECG, electrocardiography; PR; RT, radiotherapy; ST.
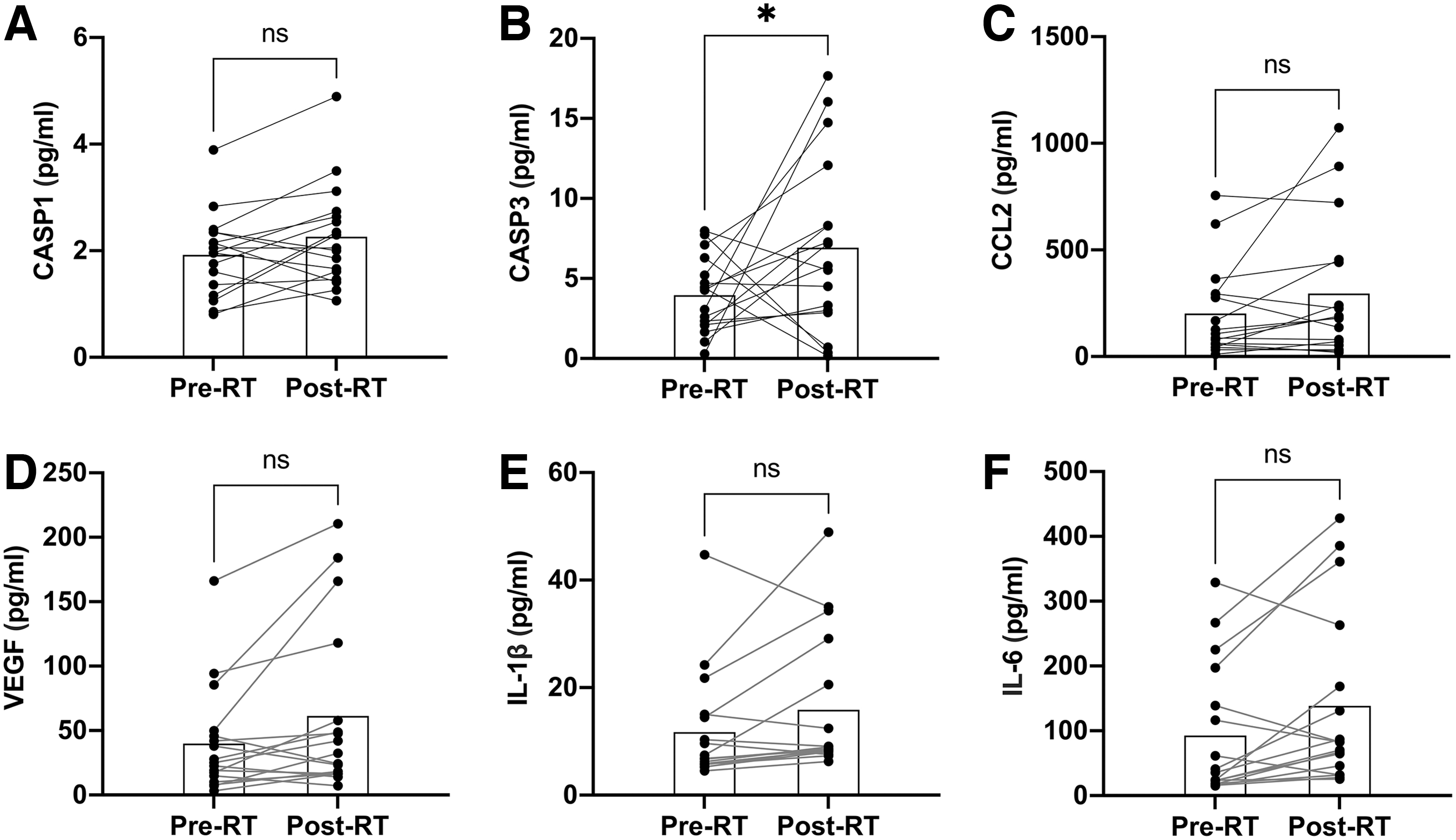
Among the 17 patients, 9 (52.9%) of the patients developed >30% elevation in plasma caspase-3. The proportions of patients who developed >30% increases in caspase-1, CCL2, VEGF, IL-1β, and IL-6 were 35.3%, 35.3%, 41.2%, 52.9%, and 64.7%, respectively. In addition, the changes in caspase-3, VEGF, and IL-6 levels were found to be significantly correlated with the mean heart dose in lung cancer patients. However, there were no significant associations between mean heart dose and levels of caspase-1, CCL2, and IL-1β (Fig. 2). ECG changes occurred in 7 patients after RT, with the 2 most common changes being T-wave changes and sinus tachycardia. As shown in Supplementary Fig. S2, the percentage of patients with elevated biomarker levels among patients with ECG changes was higher than that among patients without ECG changes.
Associations between mean heart dose and changes in plasma biomarker levels after RT in lung cancer patients.
Increased levels of biomarker in rat plasma after RT exposure
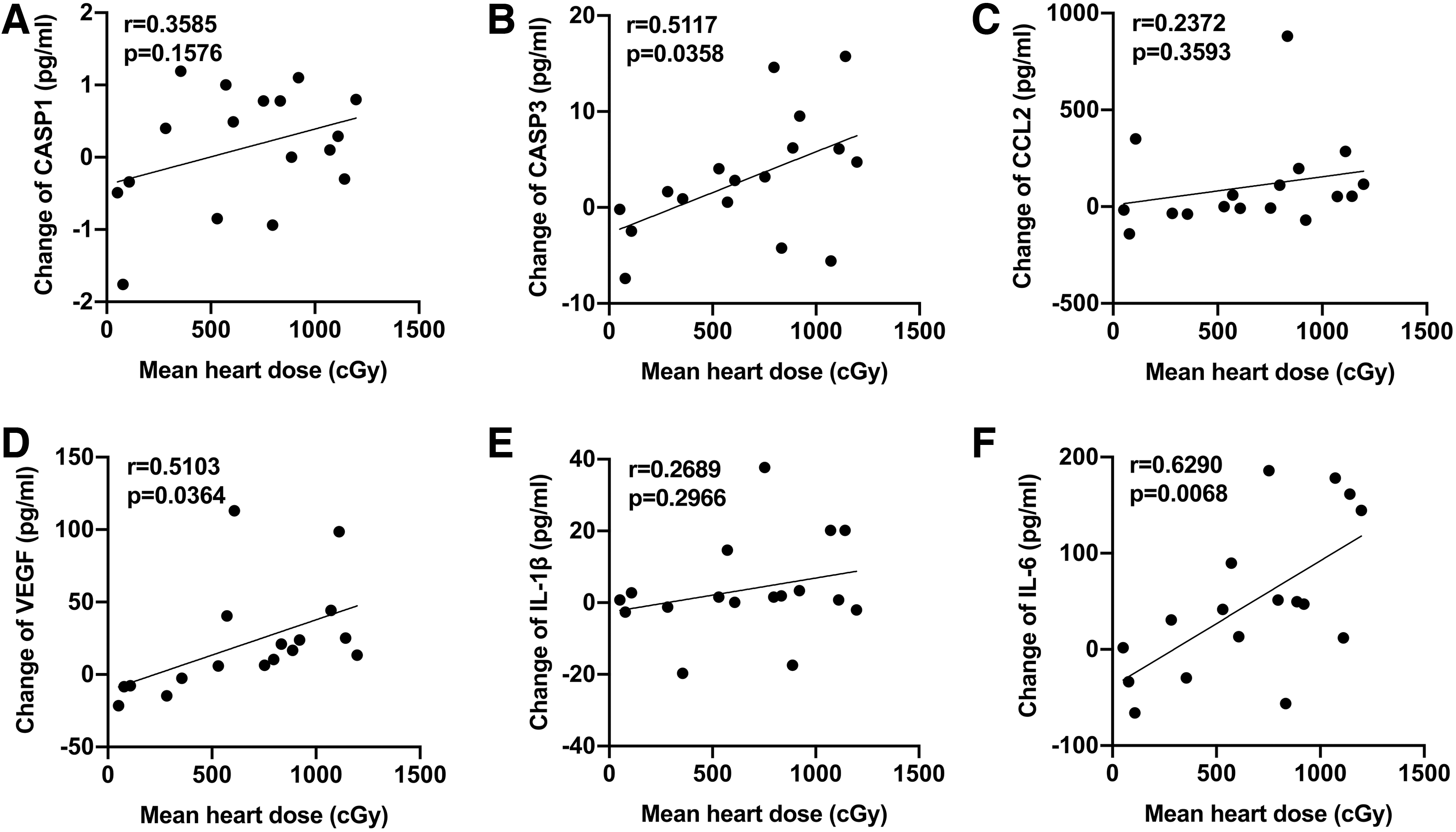
Based upon the observed trends of increasing biomarker levels in human plasma, we evaluated the levels of biomarkers in rat plasma at various time points after RT. As shown in Fig. 3, the plasma levels of caspase-1, caspase-3, CCl-2, CCl-5, VEGF, and TGF-β1 were significantly increased after RT compared with the control group (P < 0.05). Following irradiation of the heart, the plasma biomarker levels of caspase-1, caspase-3, CCl-2, CCl-5, and TGF-β1 increased to peak levels at 3 or 5 days after RT. Thereafter, the plasma levels of these biomarkers gradually returned to baseline values before reaching a second peak at the second or third month after RT. The plasma VEGF level also increased gradually after RT and peaked at the second month after RT in the rat RIMD model.
Levels of biomarkers in rat plasma at different time points after RT.
Increased macrophage infiltration in rat myocardium after RT
We examined the degree of myocardial inflammation by counting CD68-positive cells (a macrophage marker) in sections of rat irradiated myocardium after RT. As shown in Fig. 4, control rats showed a small number of CD68-positive cells, likely representing resident tissue macrophages. In comparison, the number of CD68-positive cells in the irradiated myocardium was increased significantly at 1 month after RT (P < 0.01) and reached a peak value at the sixth month (P < 0.0001), supporting a continuous inflammatory reaction in response to RT. In addition, we also evaluated macrophage infiltration in the nonirradiated myocardium after RT and found that no significant increase in macrophages within the nonirradiated area of the heart after RT compared with that in the control rats (Supplementary Fig. S3).
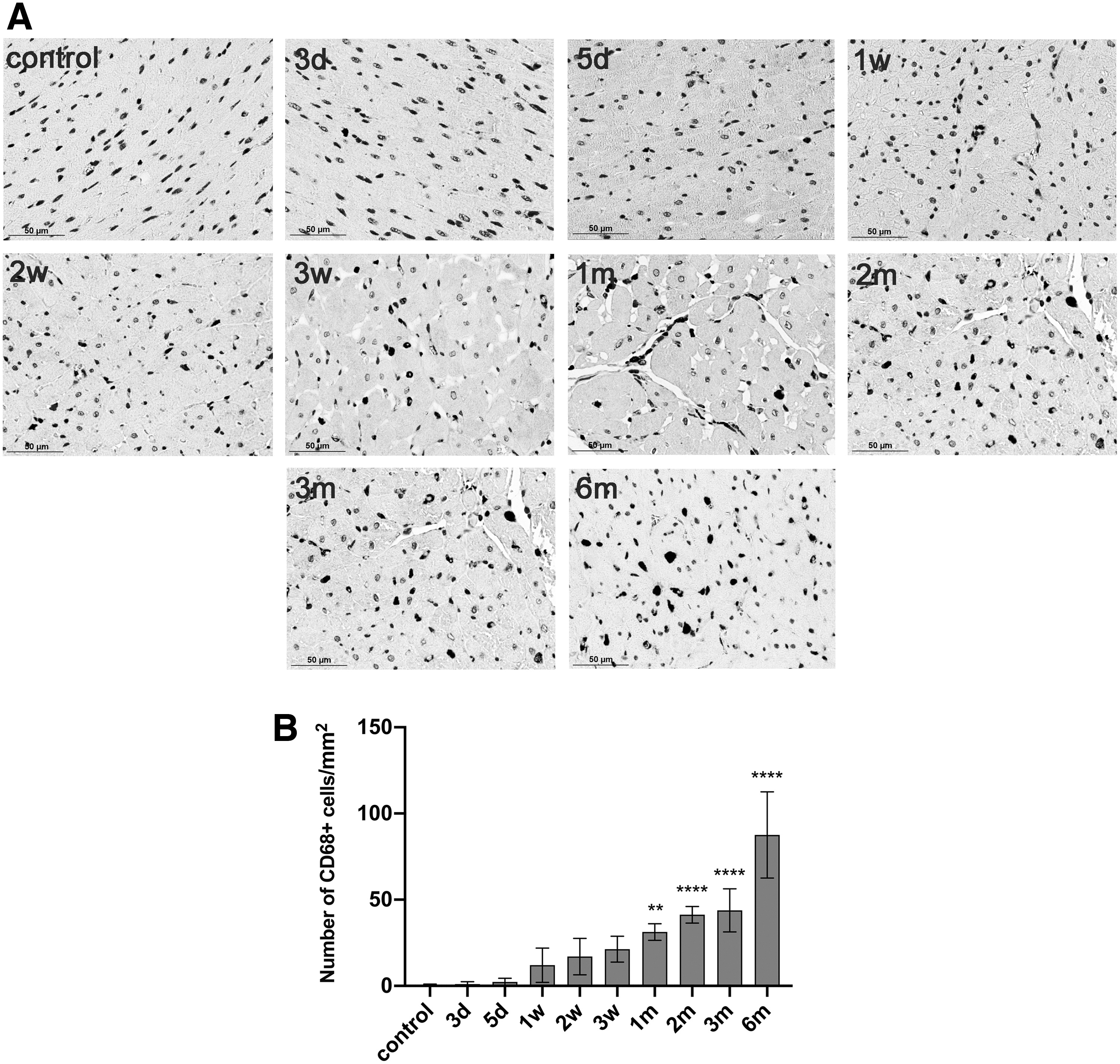
Assessment of myocardial macrophage infiltration in irradiated myocardium at different time points after RT.
Development of myocardial fibrosis after RT
RT-induced damage can develop chronically and, in most tissues, is characterized by collagen deposition and excessive formation of fibrous tissue, at which point damage is considered irreversible. We evaluated the levels of fibrosis in myocardial tissue of RIMD model rats at various time points post-RT using Masson trichrome staining. Figure 5A shows representative images of trichrome staining at different time points post-RT, and Fig. 5B shows the quantitative results for fibrosis. No fibrosis was observed in nonirradiated myocardium. Although fibrosis appeared to be increased from the second month post-RT, the fibrosis levels only reached statistical significance at the sixth month post-RT (P < 0.0001).
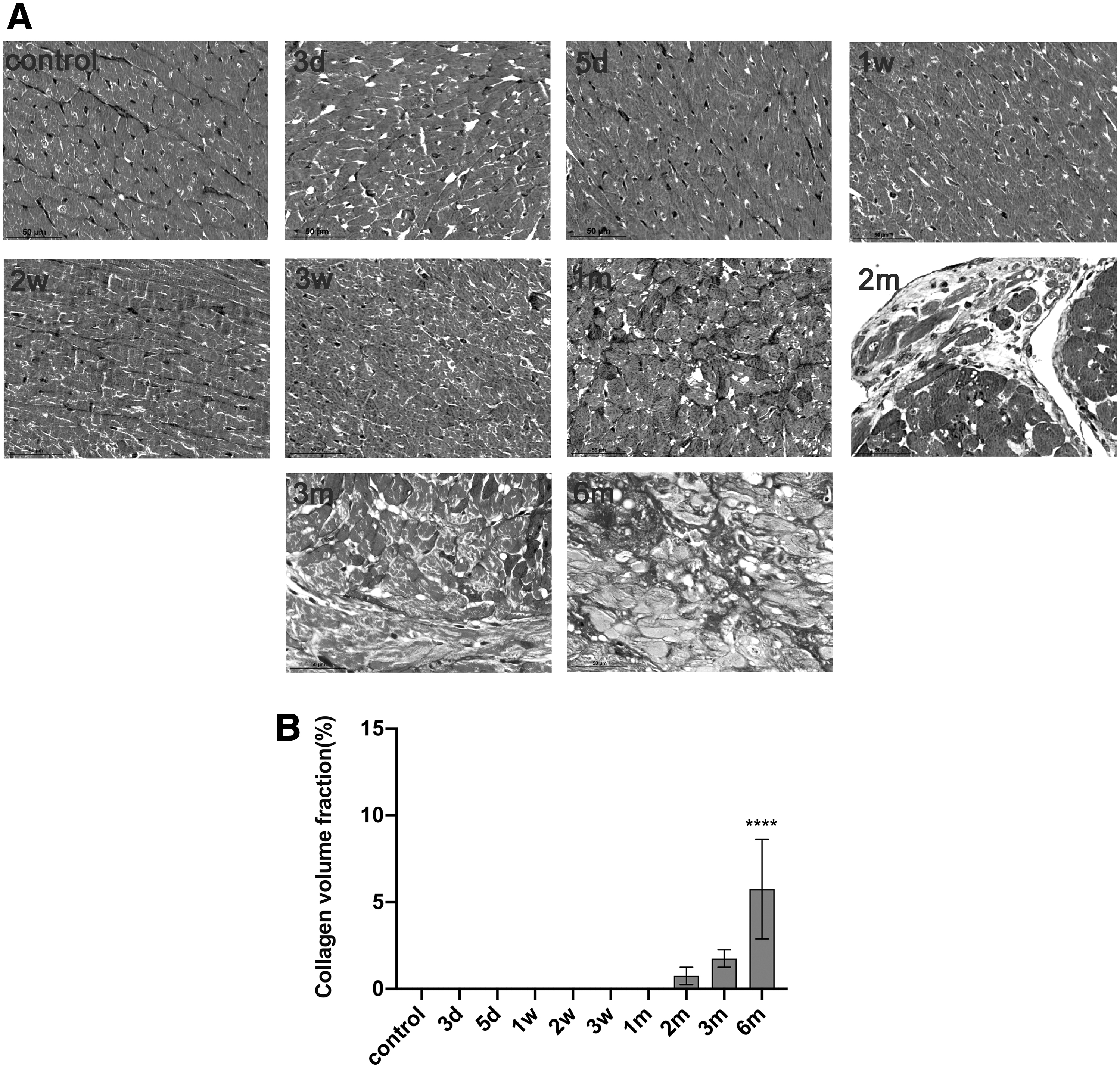
Masson trichrome staining of rat cardiac tissue at different time points after RT.
Correlations between plasma biomarker changes and RIMD-related pathological changes
Finally, we explored the correlations between changes in plasma biomarkers and pathological changes in the model of RIMD. As shown in Fig. 6, the release of CCL2, caspase-3, and TGF-β1 was significantly correlated with the infiltration of macrophages from 3 weeks to 2 months after RT. The increased plasma CCL5 level was found to be significantly correlated with the infiltration of macrophages from 3 weeks to 3 months after RT. A significant association was also found between the increase in plasma VEGF and macrophage infiltration from 1 week to 2 months after RT. Notably, the increased release of biomarkers occurred earlier than the infiltration of macrophages (1 week versus 1 month). Given the relatively late appearance of fibrosis, correlation analysis between changes in biomarker levels and fibrosis was not performed.
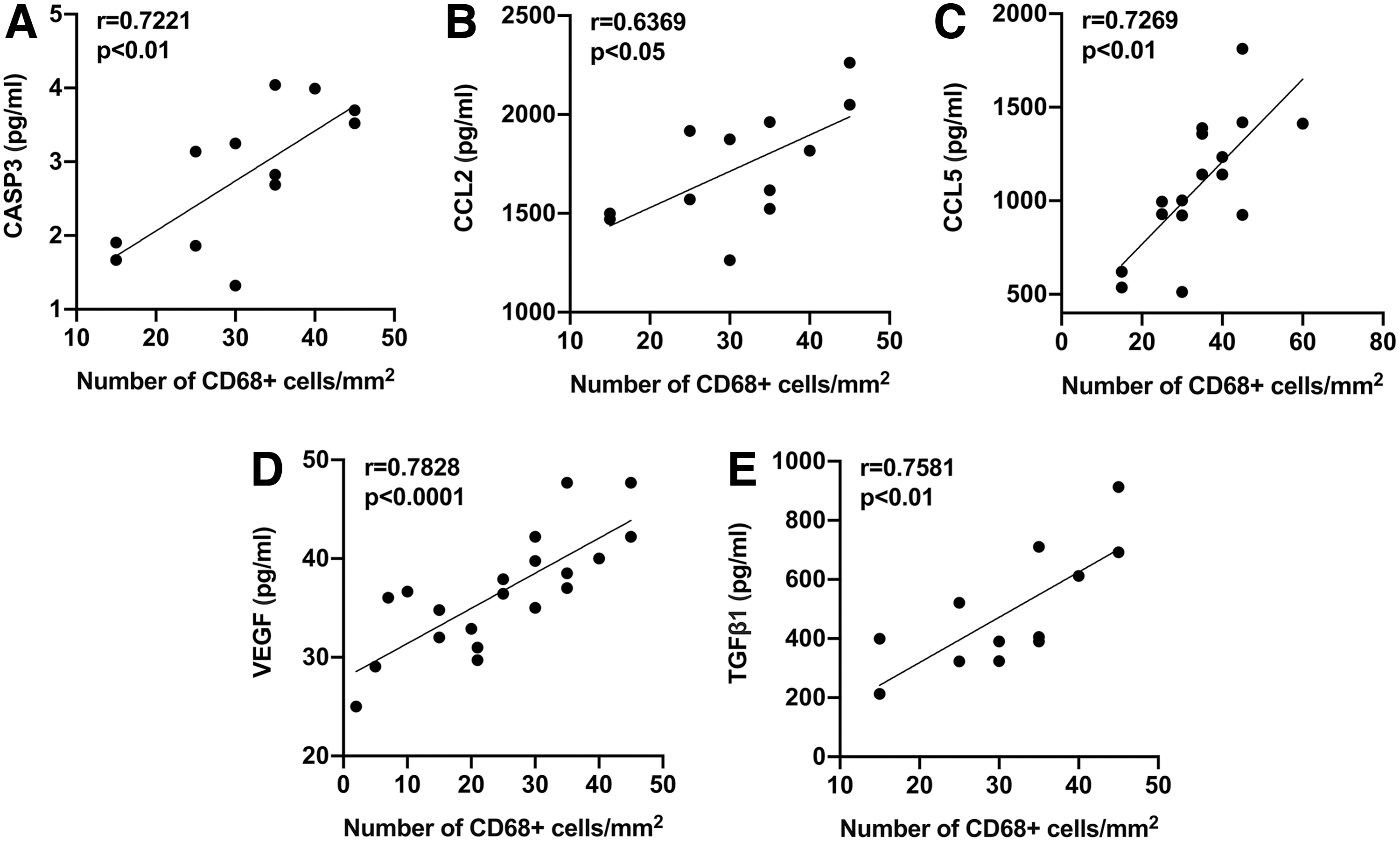
Correlations between post-RT biomarker levels and macrophage levels in myocardial tissue in the RIMD rat model.
Discussion
In this study, we evaluated the early changes in a diverse panel of plasma biomarkers after RT in lung cancer patients and a rat model. Our analyses identified sensitive plasma biomarkers that could predict RIMD in patients and rat models, including caspase-1, caspase-3, CCL2, and VEGF. IHC and Masson trichrome staining confirmed the development of myocardial damage after RT, providing pathological support for the effectiveness of the identified biomarkers for early prediction of RIMD.
Plasma biomarkers can aid in the detection of subclinical cardiotoxicity and play an important role in risk prediction for RIMD. Our study found that plasma caspase-1, caspase-3, CCL2, and VEGF levels were increased after RT in lung cancer patients and rat models, and the increases in these biomarkers were significantly correlated with the mean heart dose of radiation. Numerous studies have confirmed that the mean heart dose is an important risk factor for radiation-induced cardiotoxicity (Dess and others 2017; Atkins and others 2019; Chung and others 2021). In addition, we found that plasma biomarker levels were increased most significantly in the first week and the second/third month after RT in preclinical models, which may suggest that cardiac irradiation induces a temporary inflammatory response as well as a late inflammatory response.
Several previous studies have also explored the changes in biomarker concentrations in plasma or irradiated tissues after thoracic RT, including caspase-1, CCL2, CCL5, VEGF, placental growth factor, and others (Sievert and others 2015; Christersdottir and others 2019; Demissei and others 2019; Dreyfuss and others 2021). Most of these studies detected biomarkers at a specific time point after RT. However, biomarkers were dynamically detected in our preclinical study, and thus, our findings may better reflect the change rule for biomarkers after RT.
Early changes in plasma biomarker levels after radiation exposure can provide important mechanistic insights for advancing our understanding of the pathophysiological processes of RIMD. Prior studies indicated that the first response in the development of RIMD is activation of the coronary microvascular endothelium [to a large part related to the activation of the nuclear factor-κB (NF-κB) signaling pathway], with increased expression of chemokines and adhesion molecules, which promotes leukocyte infiltration (Halle and others 2010; Christersdottir and others 2013, 2019; Herrmann 2020). In the rat model of RIMD, the significant increase in plasma VEGF was presumably based on the stimulation of endothelial cell proliferation in response to radiation-induced damage. Elevated levels of caspase-1 and caspase-3 were observed in rat plasma, supporting increased inflammasome activity and apoptosis in acute radiation injury.
CCL2 and CCL5 are chemokines associated with inflammasome activation, and significant increases in plasma CCL2 and CCL5 levels suggest ongoing recruitment of inflammatory cells, including monocytes/macrophages (Gosling and others 1999; Moore and others 2013). The TGF-β1 level in rat plasma also increased significantly after RT, and increased TGF-β1 expression is known to induce the production of excessive amounts of collagen and to often correlate with disease severity (Herrmann 2020; Ong and others 2021). In lung cancer patients receiving thoracic RT, the caspase-3 level increased significantly after RT. Of the 9 patients with elevated caspase-3, 6 (66.7%) had pre-existing cardiac disease, including hypertension, diabetes, and heart disease. Among 8 patients without elevated caspase-3, only 4 (50.0%) had pre-existing cardiac disease. Thus, pre-existing cardiac disease may be associated with an increased likelihood of caspase-3 elevation.
In addition, increasing trends in caspase-1, CCL2, and VEGF levels in patient plasma were observed. However, the observed increases in plasma biomarkers in patients were not significant, which may indicate that the trends of decreasing mean heart dose and increased cardiac protection in the current treatment era have resulted in less cardiomyocyte damage and stress. However, it should be noted that 35%–55% of the lung cancer patients developed >30% elevation of caspase-1, caspase-3, CCL2, and VEGF levels from pre-RT to post-RT. The impact of such elevations on the long-term risk of cardiac events merits further investigation. In this study, consistency was observed between ECG changes and elevation of biomarker levels after RT. The patients with ECG changes were at greater risk for elevated biomarker levels. Our studies indicate that vascular endothelial dysfunction and inflammation may be relevant in the early pathogenesis of RIMD.
An important feature of preclinical models of RIMD is the ability to histologically evaluate myocardial damage, an analysis which can rarely be done in humans (Boerma and others 2005; Sridharan and others 2016). The significant increase in CCL2 in plasma of irradiated rats suggests ongoing recruitment of inflammatory cells including monocytes/macrophages. Consequently, we stained rat heart tissues for the macrophage marker CD68. We observed significantly increased numbers of macrophages in irradiated myocardial tissues from the first month after RT, further supporting an inflammatory reaction in this tissue after RT. No significant increase in macrophage infiltration was observed in nonirradiated myocardium after RT, indicating no increase in macrophages occurs with aging in myocardial tissue.
Furthermore, significant correlations were observed between the infiltration of macrophages and the levels of CCL2, CCL5, caspase-3, TGF-β1, and VEGF from 3 weeks to 2 months after RT. The results of our correlation analysis show the feasibility of using biomarkers to predict myocardial damage.
Increasing evidence shows that fibrosis is also an important mechanism of RIMD (Boerma and others 2008; Sharma and others 2018; Dreyfuss and others 2021). RT-related myocardial fibrosis can be initially asymptomatic, often leading to delayed diagnosis (Heidenreich and others 2003). Histologically, this fibrosis is characterized by thick collagen bands crisscrossing the heart tissue and separating and replacing cardiomyocytes (Sharma and others 2018). In our study, the most severe fibrosis occurred in the myocardial interstitium at the sixth month after RT, which is consistent with previously published data (Dreyfuss and others 2021). Pathological analysis of rat heart tissue after RT confirmed the existence of myocardial damage, and the increased plasma levels of biomarkers were detected significantly earlier than the pathological changes.
Thus, our results provide pathological support for the predictive value of plasma biomarkers for RIMD. As plasma biomarkers, caspase-1, caspase-3, CCL2, and VEGF are expected to facilitate early detection of RIMD before obvious pathological changes.
Our study has a number of limitations. First, to elicit detectable myocardial damage within a practical timeframe, we used a single dose of 40 Gy in our rat model, which is significantly greater than the routine RT dose applied clinically. Second, plasma biomarkers were not dynamically detected in lung cancer patients. Third, the plasma biomarker concentrations in patients were not stratified according to tumor location, but correlation analysis was performed to evaluate the relationship between mean heart dose and biomarker levels. Finally, given the limited number of patients included in the human plasma biomarker analysis, further validation in larger cohorts is needed to verify the relationships between these biomarker changes and the risk of heart disease in patients treated with thoracic RT.
Conclusion
The results of the present study verified the effectiveness of plasma biomarkers for early prediction of pathologically confirmed RIMD in clinical samples and rat models. Mechanistic biomarkers indicative of vascular function and inflammation, including caspase-1, caspase-3, CCL2, and VEGF, may be effective biomarkers for early prediction of RIMD. In the future, these inflammatory biomarkers also may provide valuable targets for therapeutic interventions for patients receiving thoracic RT.
Footnotes
Authors' Contributions
Y.W. and S.Y. designed the study; Y.T. analyzed data and wrote the article; C.Z. contributed to the semiquantitative analysis of pathological sections; P.L., Z.M., and Y.L. revised the article. All authors have read and approved the final version of the article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported by the Natural Science Foundation of Shandong Province (ZR2021QH008), Bethune Charitable Foundation (flzh202116), the Natural Science Foundation of China (82203218), the Natural Science Foundation of China (NSFC81872475, NSFC82073345), and the Jinan Clinical Medicine Science and Technology Innovation Plan (202019060).
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
