Abstract
There remains a dearth of data regarding the association between chronic inflammation and hypertension (HTN) in sub-Saharan Africa, a region that accounts for >70% of the global burden of HIV infection. Therefore, we assessed the levels of biomarkers among HIV+ individuals and its associations with HTN in Tanzania. A cross-sectional study was conducted at one of the largest clinics in Tanzania and data from 261 HIV+ patients were analyzed. Standardized tools were used to collect data. Blood pressure was measured using Omron® M2 blood pressure monitor. Enzyme-linked immunosorbent assay was used to test for inflammatory markers [C-reactive protein (CRP), interleukin (IL)-6, IL-18, soluble tumor necrosis factor receptor type I (sTNFRI), sTNFRII]. Bivariate and multivariable analysis was conducted to examine association between the biomarkers and HTN. We further conducted age–sex–alcohol-adjusted models to control for any confounders. The prevalence of HTN was 43% with a high prevalence reported in female (70%) participants and those older than 55 years of age (77%). Being women, older than 55 years of age, married, and being overweight was associated with HTN. The highest correlations were observed between TNR2 and CRP (ɤ = 0.13, P = 0.044), and TNR2 and IL-18 (ɤ = 0.13, P = 0.034). Participants who had elevated CRP levels were 2 times more likely to experience HTN in the age-adjusted model [odds ratio (OR) = 3.5, 95% confidence interval (CI) = 1.1–11.3], age–sex-adjusted model (OR = 3.3, 95% CI = 1.0–10.9), and the full model (OR = 2.9, 95% CI = 0.8–10.0). Our study shows that high CRP levels are significantly associated with the higher prevalence of HTN notwithstanding all other markers, which showed a positive association with HTN despite not being significant. These findings point to the importance of creating awareness, education, and screening for HTN among HIV patients in high epidemic countries. More rigorous studies are needed to know the exact pathway mechanisms of inflammation in HIV patients.
Introduction
Hypertension (HTN) is a major cause of mortality worldwide and an important risk for cardiovascular events (Zhou and others 2018). It is estimated that at least 970 million people have HTN worldwide and 9.4 million deaths occur owing to HTN complications (Cai and Harrison 2000). Reports indicate that sub-Saharan Africa (SSA) region has the highest age-adjusted prevalence of HTN in the world (WHO 2013), and an incidence of 1.5 times higher than that observed in Europe, the United States, and Australia (Benjamin and others 2017).
Although the prevalence of HTN in SSA has dramatically increased, it is important to note that HIV-infected adults on ART have a higher risk (Kavishe and others 2015) compared with the non-HIV-infected persons. The novel pathologic mechanism of HTN in HIV-infected people on ART is multiple; may be a result of microbial translocation, immune suppression and reconstitution, lipodystrophy, and chronic inflammation (Fahme and others 2018) coupled with traditional risk factors such as age, obesity among others (Guwatudde and others 2015).
Studies comparing the prevalence of HTN in HIV-infected individuals and HIV-uninfected controls show conflicting results of lower prevalence (Clark and others 2015; Benjamin and others 2016), or higher prevalence (van Zoest and others 2016) compared with controls. However, in any case, HIV-infected adults with HTN have a 10-year cardiovascular disease (CVD) risk >20% and increased risk of all-cause mortality than HIV-uninfected adults (Mutimura and others 2008; Fahme and others 2018).
HIV infection is known to cause an ongoing chronic inflammation even with effective ART. More recently, subsets of lymphocytes and their cytokines are observed to cause vascular remodeling, resulting in increased blood pressure (Baran-Gale and others 2013). Studies using animal models have demonstrated adverse events preceding the development of HTN, such as increase in systolic and arterial pressure, inhibition of endothelial nitric oxide, and increase in angiotensin II receptors to be independently influenced by inflammation (Pacurari and others 2014; Satou and others 2018). The role of inflammation in the pathogenesis of cardiovascular events is now widely accepted (Ikeoka and others 2010; Abe and others 2013; Upadhyay 2015).
The risk factors for HTN are well characterized in developed countries, whereas it not clearly understood in SSA (Kwarisiima and others 2016; van Zoest and others 2016). People of African descent are reported to have HTN-prone genes (RodrõÂguez-ArbolõÂ and others 2017), therefore identifying modifiable risk factors may reduce the epidemic rise of HTN. Several studies have reported traditional risk factors like older age, family history, male sex, sedentary lifestyle, high salt intake, and obesity to predict the occurrence of HTN (Hendriks and others 2012; Tibazarwa and Damasceno 2014; Ataklte and others 2015; Guwatudde and others 2015). However, there are limited data in SSA-relating inflammation and HTN. The details of inflammatory mechanisms and precise inflammatory mediators are yet to be fully understood (Chamarthi and others 2011).
Furthermore, classifying participants beyond traditional risk factors, based on inflammatory markers may improve diagnosis and inform prevention, treatment, and management of cardiovascular risks (Willerson and Ridker 2004; Duprez and others 2012; Golia and others 2014). As reported from studies conducted in high-income countries, there are several inflammatory markers associated with HTN. For this study we assessed C-reactive protein (CRP), interleukin (IL)-6, IL-18, soluble tumor necrosis factor I (sTNFRI) and sTNFRII owing to their ability to predict non-AIDS events (Savoia and Schiffrin 2006; Montecucco and others 2011; Tsounis and others 2014).
Methods
Study design, site, and population
A cross-sectional study was carried out from March to May 2018 at the Infectious Disease Clinic (IDC) in Dar es salaam, Tanzania. IDC operates a large care and treatment center for people living with HIV (PLHIV), which attends to ∼100 adult PLHIV per working day. The study included adult HIV participants older than 18 years of age, excluding pregnant women, patients who had been on anti-inflammatory drugs within the past 3 months, and those on antihypertensive drugs.
Sample size and sampling technique
The sample size for this study was calculated by taking most probable prevalence of HTN as 50% and permissible error as 5% with 95% confidence interval. Fixing the attrition rate as 5%, the minimum sample size was calculated as n = 403. This study dubbed HIV and Chronic Disease Study recruited 406 HIV-positive study participants on ART. Purposive sampling was used for recruitment as this method also allowed less disruption of participants as they assessed their routine HIV care.
Data collection
The WHO stepwise approach for noncommunicable diseases surveillance was used for data collection at enrolment (Riley and others 2016). Additional information on CD4 counts, as well as the types of ART and duration on ART were abstracted from the participants' health records. Demographic and behavioral characteristics such as age, sex, physical activity, alcohol use, and smoking habits were collected through face-to-face interviews by a trained nurse counselor. Anthropometric assessments such as weight, height, and waist circumference were also taken by a trained nurse.
Blood pressure measurements
Blood pressure measurements were taken using an Omron® M2 (HEM-7121-E) automatic blood pressure device when a participant was seated and relaxed. Readings were taken 2 times consistently from the left arm at 3-min interval and the average reading was used in the analysis. One was considered hypertensive if the average reading was ≥140/90 mmHg; prehypertensive if the average reading was between 121/80 and 139/89 mmHg; stage 1 HTN as 141/90 mmHg to 159/99 mmHg and stage 2 HTN as >160/100 mmHg (Casey and others 2019).
Anthropometric measurements
Participants' weight and height were measured using calibrated Seca 762 classic mechanical medical weighing scales and UNICEF standard height boards to the nearest 0.1 kg and 0.1 cm, respectively. In measuring height, participants were requested to stand with the feet (without shoes) upright, arms hanging loosely on the sides, without head gear and standing with the back against the stadiometer, feet slightly apart with the back of the heels, the middle of the shoulders, buttocks, and the back of the head touching the wall. In measuring weight, the scale was placed on a flat surface and zero checked before every measurement. Participants were weighed without shoes and wearing minimal clothing. Weight and height were used to calculate body mass index (BMI) and the following cutoffs were used based on the WHO guidelines: underweight (BMI <18 kg/m2), normal (BMI ≥18 and ≤25 kg/m2), overweight (BMI >25 and <30 kg/m2), and obese (BMI ≥30 kg/m2).
Waist and hip circumference and waist–hip ratio were used to assess the risk for CVD and were taken to the nearest 0.1 cm (World Health Organization 2008). Waist-to-height ratio (WHR) was calculated, a cutoff >50% was considered to represent an elevated WHR defined as abnormal WHR in analysis.
Blood sample collection and handling
Venous blood samples were collected into ethylenediaminetetraacetic acid tube by a trained phlebotomist and then centrifuged for 20 min at 1000g. Serum was separated immediately and stored in cryo vials at −80°C until testing. The stored blood was used for analysis of anti-inflammatory markers; IL-6, CRP, IL-18, sTNFRI, and sTNFRII using enzyme-linked immunosorbent assay (ELISA) reader Multiskan Ascent reader 354-90593.
Laboratory procedure for IL-6 using ELISA
Direct ELISA for IL-6 was performed using reagents from R&D Systems, Catalog number PD6050; we adopted their procedure as well. In brief, a standard solution was prepared and serially diluted into 5 concentrations each half of the previous starting from concentrations of 300 pg/mL. Then 100 μL of sample, standard and controls were added into a precoated 96-well plate containing IL-6 monoclonal antibody in duplicates. Human IL-6 conjugate was added followed by a substrate solution after washing. Finally, a stop solution was added changing the color of the well content into yellow from blue. Optical density was determined using a 450 nm and corrected by using a 630 wavelength.
Laboratory procedure for CRP
Direct ELISA for CRP was performed using reagents from R&D Systems, catalog number PD6050; following manufacturer's instructions. In brief, a standard solution was prepared and serially diluted into 5 concentrations each half of the previous starting from a concentration of 50 ng/mL; the samples were also diluted 100-folds (10 μL of sample +990 μL of diluents). Then 50 μL of sample, standard and controls were added into precoated 96-well plate containing CRP monoclonal antibody in duplicates. Human CRP conjugate was added followed by a substrate solution after washing. Finally, a stop solution was added changing the color of the well content into yellow from blue. Optical density was determined using a 450nm and corrected by using a 630 wavelength.
Laboratory procedure for IL-18
Direct ELISA for IL-18 was performed using reagents from Medical & Biological Laboratories (MBL), code number 7620, following manufacturer's instruction. In brief, a standard solution was prepared and serially diluted. The samples were also diluted at 1:5 (50 μL of sample +200 μL of assay diluent). Then 150 μL of sample, controls and standards were transferred to a 96-well plate, then 50 μL of sample, standard and controls were added into precoated 96-well plate containing IL-18 monoclonal antibody. Human IL-18 conjugate was added followed by a substrate solution after washing. Finally, a stop solution was added changing the color of the well content into yellow from blue. Optical density was determined using a 450nm and corrected by using a 630 wavelength.
Laboratory procedure for sTNFRI and sTNFRII
Direct ELISA for both sTNFRI and sTNFRII was performed using reagents from R&D Systems, catalog number PDRT200, following manufacturer's instruction. In brief, a standard solution was prepared and serially diluted. The samples were also diluted at 1:10 (50 μL of sample +450 μL of calibrator diluent). Then 200 μL of sample, controls and standards were transferred to a precoated 96-well plate containing IL-18 monoclonal antibody. Human IL-18 conjugate was added followed by a substrate solution after washing. Finally, a stop solution was added changing the color of the well content into yellow from blue. Optical density was determined using a 450nm and corrected by using a 630 wavelength.
A standard curve of absorbance against concentration was plotted for each biomarker; the best-fit line was determined by regression analysis. A y = mx + c equation was generated. The rest of the results were obtained by substituting y as the value of absorbance and calculating for X (concentration) for each patient result. The final concentration was obtained by multiplying with the dilution factor.
Ethical Approval
Approval for this study was obtained from the Muhimbili University Institutional Review Board 12–06/AEC/Vol.XII/86 and the University of West Florida Institutional Review Board. Before enrolment in the study, participants provided written informed consent. All ethical procedures were conducted and maintained throughout the study period.
Data analysis
Data from case report forms and ELISA reader were entered on Excel sheet and cleaned, and then data were exported to STATA 14 (StataCorp, College Station, TX) for coding and statistical analysis. Data for prehypertensive individuals were dropped and analysis was carried out on 261 participants of which 138 were normotensive and 113 were hypertensive. Descriptive statistics were used to summarize participants' characteristics. Bivariate analysis was used to look for association between the studied variables and the outcome measures. All variables with P ≤ 0.25 were subjected to a multivariable analysis with logistic regression. The magnitude of association was measured using adjusted odds ratio, and 95% confidence interval. A value of P < 0.05 was considered statistically significant.
The study further examined the use of biomarkers for predicting HTN. The biomarkers were log transformed, as they were continuous, and standardized to a mean of 0 and standard deviation (SD) of 1 to facilitate the comparison of β coefficients. Elevated biomarker level was defined empirically as ≥1 SD above the mean. The biomarkers were modeled into 3 BMI-adjusted models: the first model was age adjusted, the second was age and sex adjusted, whereas the third model was adjusted for age, sex, marital status, eating fruits, and alcohol use. Under secondary analysis the multivariable-adjusted model including multiplicative interaction terms for biomarker levels and age, sex, BMI, and waist circumference were tested for effect modification.
Results
Overall, a total of 261 participants [men = 62 (24%); women = 199 (76%)] were enrolled into the study. The baseline characteristics of the study are given in Table 1 stratified by sex. The prevalence of HTN was 113 (43%) [men = 34 (30%); women = 79 (70%)]. Majority of the hypertensive were above the age of 55 years, 87 (77%). More results are as given in Table 2.
Participant Social and Clinical Characteristics by Hypertension
The sum of the counts in strata of a given variable may be less than the total sample size because of missing responses. Unless indicated by ^, missing data for each variable is <5%.
Participant Social and Clinical Characteristics by Gender
The sum of the counts in strata of a given variable may be less than the total sample size because of missing responses. Unless indicated by ^, missing data for each variable is <5%.
ART, antiretroviral therapy; BMI, body mass index; CVD, cardiovascular disease.
Table 3 provides the univariate and multivariate logistic regression for the participant characteristics associated with HTN. The variables that were found to be associated with HTN in the univariate analysis were sex, age, marital status, BMI category, and waist circumference. Independent determinants of HTN were found to be among those older than 55 years [adjusted odds ratio (aOR) = 3.4, 95% confidence interval (CI) = 1.8–6.3], married participants in comparison with those never married (aOR = 3.8, 95% CI = 1.1–12.9) and those who were overweight (aOR = 2.5, 95% CI = 1.2–5.2). Borderline association was observed in among women being less likely to have HTN (aOR = 0.5, 95% CI = 0.2–1.0).
Adjusted and Unadjusted Odds Ratio of Hypertension Among HIV Patients in Tanzania
Bolded p values < 0.05.
CI, confidence interval; OR, odds ratio.
Age- and sex-adjusted Spearman correlation among the biomarkers are given in Table 4. The highest correlations were observed between TNR2 and CRP (ɤ = 0.13, P = 0.044), and TNR2 and IL-18 (ɤ = 0.13, P = 0.034) (Figures 1, 2). Table 5 provides the 3 multivariable model of biomarkers. Participants who had elevated CRP levels were 3 times more likely to experience HTN in model 1 (OR = 3.5, 95% CI = 1.1–11.3), model 2 (OR = 3.3, 95% CI = 1.0–10.9) but no statistical significance difference was observed in model 3 (OR = 2.9, 95% CI = 0.8–10.0).
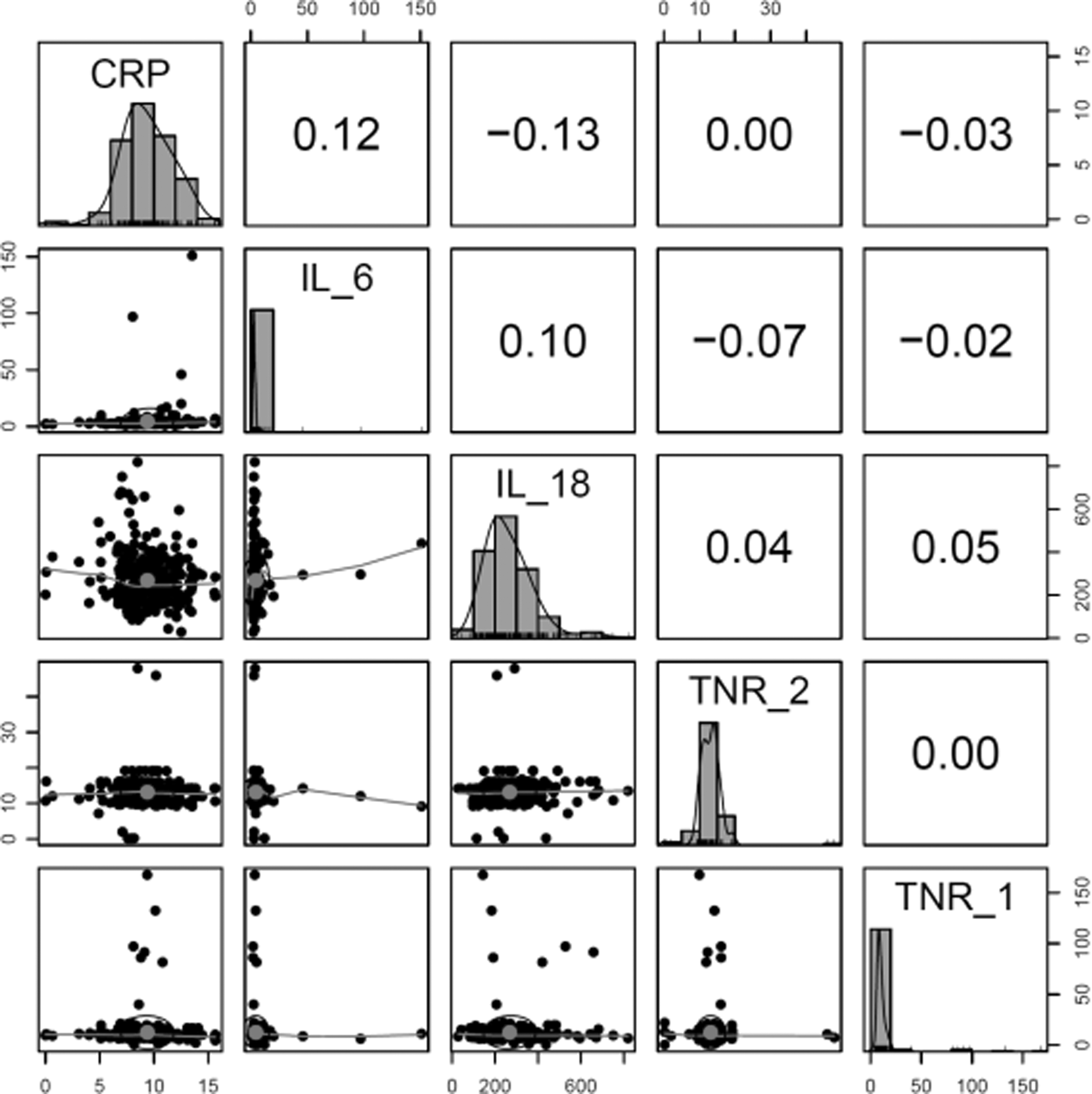
Correlation of biomarkers among HIV patients in Tanzania. CRP, C-reactive protein; IL, interleukin; TNR, tumor necrosis factor receptor.
Scatter plot of sex by biomarkers among HIV patients in Tanzania.
Age and Sex Correlation Among Biomarkers Among HIV Patients in Tanzania
CRP, C reactive protein; IL-6, interleukin 6; TNR, tumor necrosis factor receptor.
Biomarker Levels Modeling of Hypertension Among HIV Patients in Tanzania
Model 1: age adjusted.
Model 2: age–sex adjusted.
Model 3: age–sex-adjusted model controlled for marital status, eating fruits and alcohol use.
Discussion
The aim of the study was to determine the prevalence of HTN and associated biomarkers among PLHIV in Tanzania. The prevalence of HTN was 43% with a higher prevalence in female (70%) participants and those older than 55 years of age (77%). HTN remains a widespread issue of concern in SSA with studies reporting a prevalence as high as 38% (Addo and others 2007; Bromfield and Muntner 2013; Ataklte and others 2015), which is consistent with our study findings. The prevalence of HTN in this study was higher than the national steps survey conducted among 25- to 65-year-olds in Tanzania, which indicated a prevalence of 24.5% (men 24.8% and women 26.4%) (Marry Mayige 2013). Similarly, a community-mixed method study on the epidemiology of HTN in Northern Tanzania showed a prevalence of 28% with a higher prevalence of 74.4% among women (Galson and others 2017).
Although the prevalence was lower than what was reported in our study, it was consistent with the prevalence reported among women (Galson and others 2017). Another study conducted in Mafia Island Tanzania reported a prevalence of 49.5% (Muhamedhussein and others 2016). This study focused on participants 41–50 years only compared with our study that included those younger than 40 years. The variation could be because of differences in the HIV status of participants. HTN has been observed to be common in PLHIV with prevalence ranging from 8.7% to 45.9% in low- and middle-income countries (Van Zoest and others 2017). This could be the result of microbial translocation, immune suppression and reconstitution, lipodystrophy, and chronic inflammation (Fahme and others 2018) coupled with traditional risk factors such as age and obesity among others (Guwatudde and others 2015). In addition, the effect of HIV disease severity, duration of HIV, and antiretroviral (Van Zoest and others 2017) could contribute to higher prevalence.
In the context of HTN among PLHIV, the prevalence of HTN in this study is slightly different from what has been previously reported in other studies conducted among PLHIV in SSA including that from Cameroon (38%) (Dimala and others 2016) and Kenya (26%) [36]. The differences may be because of variation in population characteristics and settings. However, the findings seem to be consistent with a study carried out in South Africa that reported a prevalence of 46% (Manne-Goehler and others 2017). In contrast, a study conducted among 34,111 highly active antiretroviral therapy (HAART) naive participants in Tanzania reported a lower prevalence, 12% (Njelekela and others 2016). Similarly, Rodriguez-Aborli observed 10% of PLHIV developed HTN after 7 months of ART (Rodríguez-Arbolí and others 2016).
However, no association was observed between ART intake and HTN. The ART risk of HTN remains controversial in many studies. A large population survey reported decreased odds of HTN with ART exposure and a possible protective effect of long-term ART on cardiometabolic risk, after observing an association in mean high-density lipoprotein levels and ART duration (Kazooba and others 2017). In addition, the variations in prevalence could be because of differences in study population, design and methodology, cutoffs, and references for definition of HTN. Despite the difference, HTN remains common in PLHIV with higher prevalence in women and older populations and an important comorbidity that affects the outcomes (Van Zoest and others 2017).
The variables that were found to be associated with HTN in the univariate analysis were sex, age, marital status, BMI category, and waist circumference. Compared with men, women have a higher risk of developing HTN despite the HIV status. This was clearly observed in our study where the prevalence was 70% in women. According to American Heart Association, although HTN is not directly related to gender, health issues including pregnancy, use of contraceptives, and menopause could increase the risk of high blood pressure among women (American Heart Association 2016). The study findings are consistent with a study conducted on the burden of HTN in SSA, which indicated older age and body mass index among others as factors associated with HTN (Guwatudde and others 2015).
CRP is a marker that has been strongly linked with HTN (Hage 2014). In in vivo and ex vivo studies, CRP has been observed to induce oxidative stress and endothelial dysfunction underscoring the role of inflammation in the genesis of HTN (Jialal and others 2011). Elevated levels of CRP, IL-6, and
So both CRP and IL-6 are considered to reliably correlate with HTN; however, they are also exacerbated in the presence of obesity (Odegaard and Chawla 2013). In this study, adjustment of age–sex and alcohol attenuated the inflammation and HTN association leaving overweight as a potential driver of HTN. Obesity is a significant predictor of HTN (Muhamedhussein and others 2016) but it is also characterized with a significant inflammatory response (Lakoski and others 2011). Adipose tissue on its own is known to be immunogenic, in lean individuals, macrophage in their adipose tissue are immune modulating and release tolerogenic cytokines. However, macrophages in obese adipose tissue increase in quantity from 10% to 50% of the total cells and release inflammatory cytokines such as IL-6 and TNF (Agarwal 2014).
This is evidenced in a large cohort of treated and HIV-suppressed persons, whereby obesity was independently associated with monocyte activation markers, IL-6 and CRP (Conley and others 2015). Some studies suggest that adjustment of confounders such as obesity may be misleading, because its possible factors considered as confounders are actually in the inflammation and HTN causal pathway (Lakoski and others 2006). This is evidenced in studies where adjustment of obesity only weakens the CRP, IL-6, and HTN relationship, but it does not attenuate it.
Our findings should be interpreted in the context of the study design. They confirm an association between HTN and inflammation; however, this association is attenuated after adjustment of other risk factors, leaving obesity independently influencing HTN. The cross-sectional nature of the study limits the ability to establish causal relationships and the underlying mechanisms remain yet to be fully established. The measurement of blood pressure was conducted at a single visit that cannot show the true picture of HTN prevalence.
Conclusion
Our study shows that higher CRP is significantly associated with the higher prevalence of HTN. Those respondents being overweight and in the age group older than 55 years are more likely to develop HTN. These findings point to the importance of creating awareness through social mobilization or health education and promotion at health facility level, education and screening for HTN among HIV patients especially for older age and overweight patients in high epidemic countries and more rigorous studies are needed to know the exact pathway mechanisms of inflammation in HIV patients. Further research is needed to understand the role of inflammation in HIV, associated anti-inflammatory biomarkers, and the progression of CVD among HIV-1-infected individuals.
Footnotes
Authors' Contributions
P.M. conceived the study, interpretation of data, and drafted the article and critical review of the article. P.M. and F.H. performed the analysis. L.N. and M.M. contributed in managing the study, data collection, and critical review of the article. A.Z. and S.L. helped in preparing the first draft of the article. Y.O., P.O., S.B., E.K., A.M. and A.K. helped in critically reviewing the article and meeting journal specifications. All authors approved and read the final article. All authors participated in the critical appraisal and revision of the article.
Acknowledgments
The authors thank all clients who participated in the study. Their sincere gratitude also goes to health workers at IDC for cooperation and assistance in recruitment and specimen collection. The authors thank the AIMS institute for providing time support to the lead author to perform the study. Furthermore, the authors acknowledge Dr. Zelalem Haile and Carol Kingori from Ohio University who participated in the protocol development and the study design. The authors acknowledge Biotechne for ensuring the efficient shipment of the Biomarker kits to Muhimbili Laboratory for specimen testing. Finally, the lead author acknowledges the following fellowship programs; CODESRIA, the NIH-funded Program to Increase Diversity in Behavioral Medicine and Sleep Disorders Research (PRIDE) and HIV Intervention Science Training Program for Underrepresented New Investigators (HISTP program) for providing scholarship guidance.
Disclaimer
The views, findings, and conclusions represented in this article are those of the authors and do not necessarily represent the official views of the institutions with which they are affiliated.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
The study received partial funding from Centre for Research and Economic Opportunity. The funder had no role in the study.
