Abstract
Objective:
This pilot study evaluated the effects of Citrus depressa Hayata fruit extract (CFEx) on thigh muscle cross-sectional area (CSA) and composition in subacute stroke patients with hemiparesis who were undergoing rehabilitation.
Design and Intervention:
This double-blinded, placebo-controlled, randomized pilot trial included 40 subacute stroke patients with moderate-to-severe hemiparesis, and they were randomly assigned to receive CFEx or placebo supplements for 12 weeks. The thigh muscle CSA was measured by computed tomography as total muscle area defined by Hounsfield units (HU) values of −29 to 150 HU. The total muscle area was divided into muscle area with fat infiltration and normal muscle area to evaluate muscle composition (−29 to 29 and 30 to 150 HU, respectively).
Results:
At baseline, the total muscle area and normal muscle area in the paretic thigh were lower than those in the nonparetic thigh. The nonparetic normal muscle area was significantly higher in the CFEx group than in the placebo group at 12 weeks, whereas the total muscle area was not different.
Conclusions:
The thigh muscle CSA and composition in the paretic side have already deteriorated in patients with moderate-to-severe hemiparesis at the subacute stroke stage. CFEx supplementation during rehabilitation might improve the nonparetic thigh muscle composition in subacute stroke patients. Findings of this study are needed to be verified by a large-scale randomized trial since this study was a pilot study with a small sample size.
Trial registration: UMIN Clinical Trial Registry (UMIN ID: UMIN000012902).
Introduction
Stroke is a disease leading to long-term disability, and one third to two thirds of survivors have gait difficulties due to hemiparesis. 1 The muscle mass and strength of stroke patients with hemiparesis rapidly decrease caused by neurological deficits, local inflammation, and disuse because of bed rest and reduction of physical activities after onset of stroke. 2,3 Reduction of muscle mass is particularly observed in the paretic extremity compared with the nonparetic side. 4 Furthermore, intramuscular fat in the paretic lower extremity increases with muscle mass reduction in the subacute to chronic stroke survivors. 5,6
Intramuscular fat or muscle composition can be assessed by attenuation in the muscle area by computed tomography (CT). 7 Changes in muscle composition might be associated with declined physiological performances with aging. 8,9 The relationship between intramuscular fat and muscle strength has also been demonstrated in chronic stroke survivors. 10 Since muscle strength at the paretic side correlates with walking speed, 11 muscle composition may affect mobility function in stroke patients.
Resistance exercise is effective in improving muscle mass and composition in both chronic stroke survivors and healthy individuals. 12 Furthermore, concomitant use of dietary supplementations, which are known to inhibit muscle atrophy such as amino acids during rehabilitation, has also been shown to be useful for increasing skeletal muscle mass even in subacute stroke patients. 13 However, to the best of our knowledge, few studies have investigated the effects of dietary supplementations on muscle composition in subacute stroke patients.
Polyphenols derived from plants are known to improve muscle mass or function. 14 Citrus depressa Hayata, which is called as shiikuwasha in Japan, is a well-known traditional citrus fruit from Okinawa, Japan, and has polymethoxylated flavones, such as nobiletin (5,6,7,8,3′,4′-hexamethoxyflavone). 15 Studies have demonstrated that Citrus depressa Hayata fruit extract (CFEx) attenuates skeletal muscle atrophy induced by dexamethasone in aged rats, 16 and nobiletin has a preventive effect on fat deposition in skeletal muscle in mice. 17 These findings suggest that CFEx supplementation may improve both muscle atrophy and muscle composition.
The purpose of this pilot study was to investigate the effects of CFEx supplementation on muscle mass and composition in subacute stroke patients with rehabilitation.
Materials and Methods
Participants
Consecutive patients with either ischemic or hemorrhagic stroke who had moderate-to-severe hemiparesis and were immediately after admitted to the Tokyo Bay Rehabilitation Hospital, Chiba, Japan, were recruited in this study. In this study, we assessed the severity of lower extremity paresis by using the knee extension test score based on the Stroke Impairment Assessment Set (SIAS), which is a standardized measurement of stroke impairments according to the previous study. 18 –21 The knee extension score of the SIAS was rated from 0 to 5. Score 0 means complete paralysis, score 3 means the paretic knee could not be fully extended in the sitting position, and score 5 means no paresis. 21 We defined a knee extension test score ≤3 based on the SIAS as moderate-to-severe hemiparesis.
The inclusion criteria were as follows: aged ≥50 years with first-ever stroke, hospital admission ≤2 weeks, a knee extension test score ≤3 based on the SIAS, and requiring a standard manual wheelchair for mobility. The exclusion criteria were as follows: presence of cognitive deficits (thereby making it impossible to obtain informed consent), contraindications to exercise therapy due to complications or previous diseases, difficulty swallowing supplements, receiving parenteral or enteral nutrition, taking citrus supplementation, and allergy to the peel of citrus species.
This study was approved and monitored by the institutional review board of the Tokyo Bay Rehabilitation Hospital (No. 074) and was performed in accordance with the principles of the Declaration of Helsinki. The study protocol was retrospectively registered at the UMIN Clinical Trial Registry at January 2014. All participants provided written informed consent before participating in the study.
Intervention
The CFEx capsule contained 100 mg CFEx (Biletin; Arkray Co., Ltd., Kyoto, Japan) and excipients, including starch and crystalline cellulose. A CFEx capsule included 6.0 mg nobiletin and 2.6 mg tangeretin. CFEx is a powdered food obtained by extracting shiikuwasha fruits with hydrous ethanol, mixing with cyclodextrin for increased solubility, and drying. The safety dosage of CFEx has been demonstrated at 650 mg/day for 4 weeks in healthy subjects (unpublished data); therefore, the dose in the present study was determined to be 200 mg/day, that is, two capsules per day, considering the total amount of intake during the test period.
The identical-appearing placebo capsule contained the same amount of indigestible dextrin and cyclodextrin. CFEx and placebo capsules were supplied by Morinaga Milk Industry Co., Ltd. (Tokyo, Japan), after both capsules were prepacked in boxes and consecutively numbered according to the allocation table. Nurses administered two capsules from assigned boxes per day (at lunch and dinner) throughout the 12-week study period and recorded patient compliance. All participants received stroke rehabilitation depending on the degree of paralysis for each participant in accordance with Japanese guidelines for the management of stroke 2009 in the convalescent rehabilitation ward. 22
Study design
This 12-week, double-blinded, placebo-controlled, randomized pilot trial had a parallel design that was in conformity with CONSORT 2010 (Supplementary Table S1). Participant registration was performed by Morinaga Milk Industry as a registry center; hospital staffs and participants were blinded to the randomization. Once enrolled in the study, participants were randomly assigned to the CFEx group or the placebo group using a permuted block design with a computer random number generator. The block size was four, and stratification by sex was applied.
The random allocation was performed by a person who was independent of this study. After the research doctor had obtained the patient's consent, doctor faxed participant information, which listed name, date of birth, and sex to Morinaga Milk Industry. Morinaga Milk Industry registered participants and then gave numbers to doctors according to the allocation table. The randomization list was kept confidential at the Morinaga Milk Industry until statistical analyses. Success of blinding was not confirmed for both hospital staffs and participants after intervention.
Before the study, we performed a power analysis using G*Power version 3.0.10 (Franz Faul, Universität Zu Kiel, Kiel, Germany). The analysis of covariance was employed to calculate the sample size using an alpha of 0.05 and a power of 0.80. As this study was an exploratory study and the effect size of CFEx was unknown, we assumed an effect size of 0.4 (large effect size). 23 Twenty-six participants were required per group. Since this study was a pilot study, sample size was set 50% of the main planned trial. 24 The desired sample size was calculated as 13 for each group. Considering dropout, we decided to include 20 participants for each group in this study.
Outcome measures
The primary outcome was the cross-sectional area (CSA) of the paretic and nonparetic thigh muscles measured by CT scan. CT scanning was performed with a six-detector row scanner (Brilliance 6; Koninklijke Philips N.V., Amsterdam, the Netherlands) at 20 cm above the upper edge of the patella to quantify the skeletal muscle area of both thighs. Six slices of the thigh image were obtained by one rotation of the CT scan, and the slice interval was 1.5 mm. Obtained images were analyzed with OsiriX 8.0 (OsiriX Foundation, Geneva, Switzerland). Moreover, the total muscle area was defined as the summation of the areas where the Hounsfield units (HU) ranged from −29 to 150 HU. 25,26 As the intramuscular adipose tissue is related to low HU, we defined the muscle area with fat infiltration with −29 to 29 HU and the normal muscle area with 30 to 150 HU, as previously described. 25,26 CT scan was performed at baseline and at the end of 12-week intervention. All slices of the thigh images were analyzed. Then the average of the values of the six images was used in the statistical analysis.
Secondary outcome measures included muscle strength of the lower extremity, activities of daily living (ADL), motor function, and some biomarkers such as albumin, prealbumin, insulin-like growth factor 1 (IGF-1), and testosterone. For the muscle strength of the lower extremity, we measured isometric knee extension strength at 60° knee flexion using a handheld dynamometer (JTech PowerTrack II; JTech Medical, Salt Lake City, UT, USA). 27
We used the motor score of the Functional Independence Measure (FIM), a standard relevant scale for measuring disability, 28 for the assessment of ADL. The motor domain of the FIM contains 13 ADL-related items, and each item is scored from 1 to 7 (total score = 91). The 10-m Walk Test, Timed Up and Go Test, 30-s Chair Stand Test, and Fugl-Meyer Assessment were performed to assess motor function, as previously described. 29 –32 The biomarkers were measured by bromocresol green method for albumin, nephelometric assay for prealbumin, electrochemiluminescence immunoassay for IGF-1, and chemiluminescent immunoassay for testosterone. Each secondary outcome was measured every 4 weeks from the start of the intervention (i.e., at 0, 4, 8, and 12 weeks). Moreover, we measured triglyceride, total cholesterol, low-density lipoprotein (LDL)-cholesterol, and C-reactive protein (CRP) levels every 4 weeks as exploratory outcomes. These biomarkers were measured by using enzymatic method for triglyceride, total cholesterol, and LDL-cholesterol, and latex agglutination method for CRP.
Statistical analysis
The results are expressed as mean ± standard deviation. Statistical analysis was performed primarily to determine the efficacy of CFEx in subacute stroke patients, and data analysis was performed on an intention-to-treat basis. Demographic and clinical baseline variables and the amount of physical therapy were compared between the two randomized groups using the unpaired t test and Fisher's exact test for continuous and categorical variables, respectively. Thigh muscle areas were compared between the paretic and nonparetic sides at 0 week using the unpaired t test.
Intergroup comparison was performed at 4, 8, and 12 weeks using covariance analysis, with covariate values at 0 week and sex as the allocation factors. 33 The paired t test was employed to compare changes in outcome measures relative to the baseline value. Statistical tests of serum CRP and free testosterone levels were performed after logarithmic transformation because they were not normally distributed. Statistical analyses were performed using IBM SPSS version 22.0 for Windows (IBM Corp., Armonk, NY, USA).
Results
Recruitment began in November 2013 and continued until July 2015. A total of 40 participants were equally randomized into the CFEx and placebo groups. Two patients in the CFEx group (one patient was transferred to an acute hospital and another patient was discharged home early) and six patients in the placebo group (three patients were transferred to acute hospitals, and among them, one patient was transferred just before the first evaluation; other three patients were discharged home early) did not complete the 12-week study period. The flow of participants is shown in Figure 1.
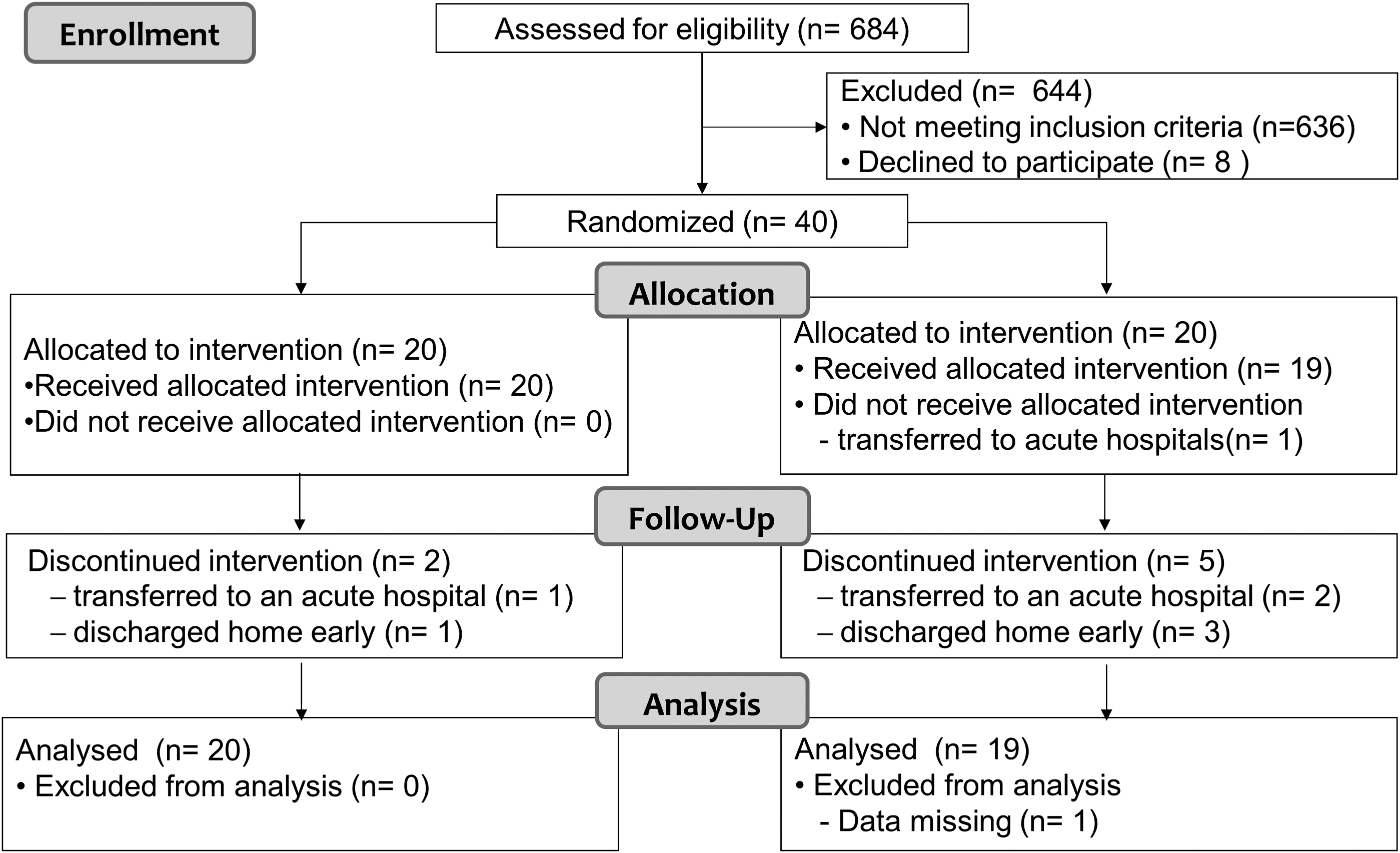
Study participation flowchart.
There were no significant differences in demographic and clinical background characteristics, including days from stroke onset and degree of paresis, which was evaluated by the Fugl-Meyer Assessment of the lower extremity (Table 1). The mean amount of physical therapy during test period was not different between the CFEx and placebo groups (78 ± 10 vs. 74 ± 10 min/day; p = 0.228). At the baseline, the total muscle area and normal muscle area in the paretic thigh were significantly lower than those in the nonparetic thigh in all participants (both p < 0.001, n = 39), but they did not differ between the two groups (p = 0.836 and p = 0.862, respectively; Fig. 2).
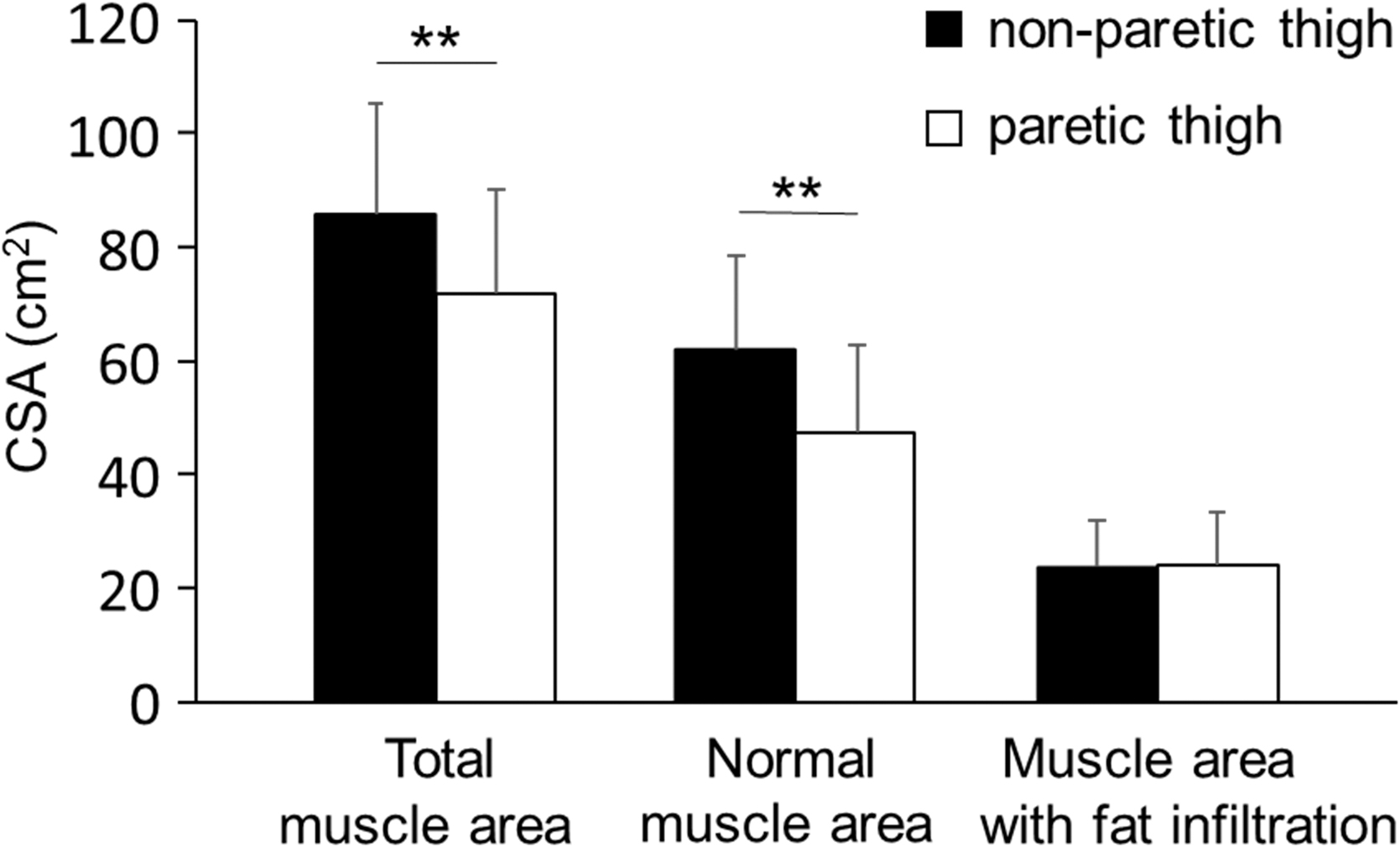
Cross-sectional muscle area of the paretic and nonparetic thighs at 0 week. The total muscle area, normal muscle area, and muscle area with fat infiltration had computed tomography values of −29 to 150, 30 to 150, and −29 to 29 HU, respectively (n = 39). White bar, paretic thigh; black bar, nonparetic thigh. Values are expressed as mean ± standard deviation. **Significantly different between thighs (p < 0.001). CSA, cross-sectional area; HU, Hounsfield units.
Demographics of Study Participants
Values are presented as mean ± standard deviation or n (%).
BMI, body mass index; CFEx, Citrus depressa Hayata fruit extract; FMA, Fugl-Meyer Assessment.
Findings of thigh muscle CSAs are shown in Table 2. No significant differences were observed in the total muscle area in the paretic and nonparetic thighs between the groups at the 12th week (p = 0.587 and p = 0.443, respectively). Similarly, the muscle area with fat infiltration did not show a statistically significant difference in both thighs between the two groups (p = 0.068 in the paretic thigh and p = 0.439 in the nonparetic thigh). In contrast, the normal muscle area in the nonparetic thigh was significantly higher in the CFEx group than in the placebo group (p = 0.045). Furthermore, the total muscle area and normal muscle area in the nonparetic thigh were significantly increased after supplementation treatment compared with the baseline in the CFEx group (both p < 0.001). However, the muscle area with fat infiltration was slightly but not significantly increased after supplementation compared with the baseline in the placebo group (p = 0.068 in the paretic thigh and p = 0.071 in the nonparetic thigh).
Changes in Cross-Sectional Muscle Area of the Thigh During 12 Weeks of Supplementation
Values are presented as mean ± standard deviation (n).
p < 0.01, significantly different between baseline (0 week) and each time point based on paired t test within the group.
p < 0.05, significantly different between the groups based on analysis of covariance.
HU, Hounsfield units.
Findings of secondary outcome measures are shown in Table 3. No intergroup differences in the secondary outcome measures during the study time were observed, but significant improvements in muscle strength, motor FIM score, and serum albumin were found within each group during the study period. Serum CRP levels were significantly lower in the CFEx group than in the placebo group at 12th week. There were no differences between the groups for triglyceride, total cholesterol, and LDL-cholesterol. No adverse events related to the intervention were observed in both the CFEx and placebo groups throughout the study.
Changes in Activities of Daily Living, Motor Function, and Serum States During 12 Weeks of Supplementation
Values are presented as mean ± standard deviation (n).
p < 0.05, ** p < 0.01, significantly different between baseline (0 week) and each time point based on paired t test within the group.
p < 0.05, ## p < 0.01 significantly different between the groups based on analysis of covariance.
CS-30, 30-s Chair Stand Test; CRP, C-reactive protein; FIM, Functional Independence Measure; IGF-1, insulin-like growth factor 1; LDL-C, low-density lipoprotein–cholesterol; T-CHO, total cholesterol; TUG, Timed Up and Go Test.
Discussion
This pilot trial suggested that rehabilitation with CFEx supplementation for 12 weeks effectively improved muscle composition in the nonparetic thigh in subacute stroke patients. Furthermore, the total muscle area in the nonparetic thigh was significantly increased after supplementation compared with the baseline in the CFEx group, but not in the placebo group. Notably, the total muscle area did not significantly differ between the groups.
It has been reported that muscle mass declines rapidly in the paretic lower extremity immediately after stroke onset. 2 Similarly, the total muscle area in the paretic thigh was already lower than that in the nonparetic thigh at 0 week in the present study. We also observed a reduction in the normal muscle area, probably because of muscle composition changes in the paretic thigh compared with the nonparetic thigh at 0 week. In the present study, the numbers of weeks from the onset of stroke to baseline in the present study were 6 to 7 weeks. A previous study observed changes in muscle composition in the paretic thigh in stroke survivors ∼3 months after onset, 5 and this present study suggests that intramuscular fat in the paretic thigh might have already altered in the subacute stage of stroke patients who had moderate-to-severe hemiparesis.
In addition, we found that the muscle area with fat infiltration in the paretic and nonparetic thighs was slightly, but not significantly, increased in the placebo group during the 12 weeks, and the total muscle area in the placebo group was not reduced. The findings indicate that rehabilitation without high-intensity resistance exercise might not have completely maintained muscle composition.
Twelve weeks of CFEx supplementation increased the normal muscle area in the nonparetic thigh. Although the cause of changes in muscle composition is poorly understood in stroke patients, exercise is one of the factors that possibly affects muscle composition. 34 Since there was no difference in the total amount of physical therapy between the groups, it has been suggested that the amount of exercise did not contribute to the difference in muscle composition in this study. Inflammation and dyslipidemia might also contribute to the change in muscle composition. The relationship of muscle composition with whole-body inflammation and lipid profile such as triglycerides, total cholesterol, and LDL-cholesterol has been shown in healthy subjects. 35,36
In addition, an increase in inflammatory marker has also been reported in the paretic lower extremity in stroke patients. 37 Nobiletin, which is included in CFEx, has been shown to improve inflammation and dyslipidemia in animal studies. 17,38 CFEx supplementation significantly reduced serum CRP levels at 12 weeks and did not reduce triglycerides, total cholesterol, and LDL-cholesterol in the present study. Improvement of muscle composition with CFEx supplementation might have been contributed by improving inflammation but not dyslipidemia.
The improvement effects of CFEx supplementation on muscle composition were different between the paretic and nonparetic sides in the present study. The reasons for this difference remain unclear because few reports are available on the effects of dietary supplementation on muscle composition in subacute stroke patients. However, one study showed that concomitant use of tea catechin, a type of polyphenol, increased muscle mass during low-intensity exercise, whereas tea catechin or exercise alone was not effective in sarcopenic older adults. 14 Muscle composition was improved by CFEx supplementation only in the nonparetic thigh in the present study. Muscle composition is known to improve by physical activity as well as muscle mass. 39 The improvement effect of CFEx on muscle composition might be affected by the difference in exercise load of rehabilitation between extremities due to paralysis.
Improvement of CFEx supplementation in muscle atrophy induced by dexamethasone in aged rats has been reported. 16 Although CFEx supplementation increased the total muscle area in the nonparetic thigh in the CFEx group, intergroup differences in the total muscle area in both thighs were not observed in the present study. CFEx supplementation might not have effectively increased muscle mass in subacute stroke patients with rehabilitation.
Although the relationships between muscle composition and muscle strength in chronic stroke survivors 10 or lower extremity performance in older adults 9 have been reported, no significant intergroup differences in muscle strength or mobility function that was evaluated by 10-m Walk Test and Timed Up and Go Test were found in the present study. Within the course of rehabilitation after stroke, patients learn compensatory strategies, 40 which involve the use of assistive devices such as orthosis for their ADL to improve their abilities. Mobility function seems to be influenced by several factors, such as endurance, balance, sensory system, and behavior learning in subacute stroke patients. Participants in the present study used different types of devices 41 and rehabilitation strategies for each patient. The change in muscle composition might not have been large enough to cause a difference in mobility function and muscle strength of participants having these differences.
Limitation
There is a limitation when interpreting the findings of this study. Findings of this study are needed to be verified by a large-scale randomized trial since this study was a pilot study with a small sample size. In addition, the long-term follow-up studies are needed to confirm safety and efficacy of CFEx on stroke patient.
Conclusions
The present study demonstrates that muscle CSA and composition in the paretic thigh might have already deteriorated in the subacute stage of stroke patients. Although the findings of this pilot study with a small sample size should be interpreted with caution, 12 weeks of CFEx supplementation could improve nonparetic muscle composition in subacute stroke patients receiving rehabilitation. Further large-scale trials should be conducted to verify these findings.
Footnotes
Authors' Contributions
Conceptualization: Y.S., H.N., H.H., and K.Ko. Data curation: K.H., Y.S., and H.H. Formal analysis: Y.O. Investigation: K.H., Y.S., and K.Ka. Supervision: K.Ko. Writing—original draft: K.H. Writing—review and editing: K.H., Y.O., and Y.S.
Ethical Approval
This study was approved and monitored by the institutional review board of the Tokyo Bay Rehabilitation Hospital and was performed in accordance with the principles of the Declaration of Helsinki.
Consent to Participate
All participants provided written informed consent before participating in the study.
Author Disclosure Statement
K.H., Y.O., K.Ka., and K.Ko. have no financial or personal relationships with Morinaga Milk Industry Co., Ltd. Y.S., H.H., and H.N. are employees of Morinaga Milk Industry Co., Ltd.
Funding Information
This study was financially supported by Morinaga Milk Industry Co., Ltd.
Supplementary Material
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
