Abstract
Increasing awareness of gonadotoxicity in cancer treatments and infertility risk is essential for counseling young cancer patients. While fertility preservation options are available in many countries, limited data on gonadotoxicity hinder recommendations, especially for soft tissue cancers. This review, part of the FertiTOX project (www.fertitox.com), organized by FertiPROTEKT (www.fertiprotekt.com), aims to address this knowledge gap to improve fertility preservation guidance.
We performed a systematic literature search on gonadotoxicity in soft tissue sarcoma (STS) cancer treatments. Only patients without metastases or recurrent disease were considered. “Suspected infertility” was defined based on low ovarian reserve parameters, low inhibin B levels, high gonadotropin concentration, gonadal dysfunction, amenorrhea, oligomenorrhea, azoospermia, or oligozoospermia due to limited infertility data. The study quality was assessed using the Newcastle–Ottawa Scale.
The search yielded 3309 abstracts, with 138 undergoing full-text analysis. Eight studies on STS were included. Suspected infertility was observed in 20 of 28 females (71.4%, range 0–100%) and 38 of 63 males (60.3%, range 34.8–100%) with STS. Six of the eight studies received high-quality scores on the NOS, while two received a fair score.
Our data suggest a high risk of infertility from chemotherapy in pre- and postpubertal STS survivors. This underscores the importance of considering fertility preservation measures when counseling these patients.
Introduction
Soft tissue sarcoma (STS) encompasses a heterogenous group of solid tumors of mesenchymal origin. 1 STS accounts for approximately 1% of all malignant tumors in adults and about 8% of malignant tumors in children. 2 The most prevalent histological subtype is rhabdomyosarcoma in children 3 and liposarcomas, leiomyosarcomas, or undifferentiated pleomorphic sarcoma in adults. 4 They are often characterized by an aggressive tumor biology. However, there has been a marked improvement in prognosis in recent years, driven by advances in cancer treatment. 4 Advances in medical science have led to improved 5-year survival rates for STS, approximately 73%. 5
Therefore, fertility preservation has become an increasing concern 6 and medical guidelines and clinical practice take fertility preservation more and more into account. However, most current clinical practice guidelines for STS, such as those from ESMO, 7 ASCO, 8 or NCCN, 1 do not address fertility preservation. The ESMO 9 and ESHRE 10 fertility preservation guidelines do not provide specific information on fertility preservation assessment or recommendations for STS, unlike the entity of osteosarcoma and Ewing sarcoma. The handbook of the network FertiPROTEKT is one of the few sources that contain specific recommendations for fertility preservation in soft tissue tumors. 11
Data on the gonadotoxicity of chemotherapies for STS are very limited. This limitation is of clinical significance as oncologists as well as reproductive physicians need to estimate the risk of infertility when they counsel patients about fertility preservation measures. Although the gonadotoxicity of specific chemotherapies can be estimated using risk calculators, which are even available online (www.oncofertilityrisk.com), the risk of infertility also needs to be provided on a disease-specific basis. The specific chemotherapy and its total dosage may not be defined at the time of fertility preservation counseling, the chemotherapy may even be adjusted during the course of therapy, and the accuracy of the prediction still needs to be validated. It is therefore of great clinical importance for reproductive physicians to be able to provide estimates of gonadotoxicity and the expected risk of infertility to be able to advise patients on fertility preservation measures.
We therefore aimed to analyze the current state of evidence regarding gonadotoxicity associated with chemotherapy treatment of STS. The review is part of the FertiTOX project 12 (www.fertitox.com), organized by FertiPROTEKT (www.fertiprotekt.com), which aims to fill the data gap on gonadotoxicity of cancer therapies to enable more accurate counseling regarding fertility preservation.13,14 Only females and males without metastases or recurrent disease were considered. Due to the frequent prevalence of STS in children and adolescents, studies with prepubertal cancer patients were also included.
Materials and Methods
Protocol registration
Our study protocol was registered at the international Prospective Register of Systematic Reviews, PROSPERO (Registry number CRD42023385402). The Preferred Reporting Items for Systematic reviews and Meta-Analysis (PRISMA) 15 were used. The FertiTOX project 12 and the associated retrospective and prospective data analyses have been approved by all Swiss ethics committees.
Sources and search strategy
A complex literature search was designed and conducted in the medical bibliographic databases MEDLINE via Embase and Cochrane Library to identify relevant documents on the topic. For the update search, the 50 most relevant references were downloaded from Google Scholar and manually compared with the previous references. In addition, forward and backward citation tracking was performed for individual studies. The original search was performed in December 2022 and the last updated on February 8, 2024.
A primary literature search strategy was developed in Embase by a medical information specialist and tested against a list of key references. Following refinement and consultation, complex search strategies were created for each information source based on database-specific controlled vocabulary (thesaurus terms/subject headings) and text words. Synonyms, acronyms, and similar terms were included in the textword search. The search was restricted to publications since 2000.
The search terms used were as follows: (1). soft tissue neoplasm (myosarcoma, leiomyosarcoma, liposarcoma, nerve sheet neoplasm, angiosarcoma); (2) cancer treatment (chemotherapy, radiotherapy); and (3) gonadotoxic effects or impact on fertility parameters. Studies involving only animals, plants, or fungi were excluded from the MEDLINE and Embase searches using a double-negative search strategy based on Ovid’s “humans-only” filters. The detailed final search strategies are presented in Supplementary Data S1. In addition to the electronic database searches, reference lists and bibliographies of relevant publications were reviewed for relevant studies.
Study selection process
The identified citations were imported into Deduklick 16 and duplicates were removed. Three investigators (MS, AR, and FP) performed the screening of titles and abstracts and tested against the inclusion and exclusion criteria (Table 1) using the Covidence software (www.covidence.org). 17 Cancer treatments were assessed on their gonadotoxicity. Gonadotoxicity with clinical relevance was defined as suspected infertility, subdefined by the criteria in Table 2.
Inclusion and Exclusion Criteria
Definition of Suspected Infertility
Data extraction
Three investigators (MS, AR, and FP) independently abstracted and reviewed the extracted data in detail. Key variables of interest were as follows: characteristics of the study populations (study design, age and number of patients at diagnosis and outcome, length of follow-up), tumor (type, localization), oncological treatment (applied regimen and dose of chemotherapy), and fertility parameters (Table 4). Discrepancies were discussed and resolved by consensus.
Quality assessment
We assessed the quality of each study using the Newcastle–Ottawa Scale. 18 The following three parameters were considered for the scoring of the individual study: subject selection (0–4 stars), comparability (0–2 stars), and study outcome (0–3 stars). The rating consisted of the following: good quality (= 3 or 4 stars in selection domain AND 1 or 2 stars in comparability domain AND 2 or 3 stars in outcome/exposure domain), fair quality (= 2 stars in selection domain AND 1 or 2 stars in comparability domain AND 2 or 3 stars in outcome/exposure domain), and poor quality (= 0 or 1 star in selection domain OR 0 stars in comparability domain OR 0 or 1 star in outcome/exposure domain).
To independently assess the risk of bias, all included studies were reviewed by MS, AR, and FP. Disagreements were resolved by consensus. Six19–24 of the eight studies were rated good, corresponding to a low risk of bias. The study of Rendtorff et al. (2012) 25 and Kenney et al. (2001) 26 got a lower rating in terms of a fair-quality score due to lacking a comparison group, no evidence about the outcome not being present at study start and limited follow-up (Table 3).
Newcastle–Ottawa Quality Assessment Form for Cohort Studies
Characteristics of the Included Studies
AMH, anti-Müllerian hormone; LH, luteinizing hormone; AFC, antral follicle count; RMS, rhabdomyosarcoma; CT, chemotherapy; RT, radiotherapy; FSH, follicle-stimulating hormone; STS, soft tissue sarcoma; GnRH, gonadotropin-releasing hormone; Y, year/s.
Results
Study characteristics
The database search yielded a total of 3309 abstracts, 138 of which were included in the full-text analysis (Fig. 1). Of these, 8 studies, each on STS, could be included in the review (Table 4, Fig. 1). The Embase database yielded the most abstracts, followed by Medline and Cochrane. The main reasons for excluding the 3164 abstracts were lack of clear reference to fertility, lack of subdivision by tumor entity, and lack of an original work. The 8 reviewed studies reported anti-Müllerian hormone (AMH), ovarian reserve parameters, gonadotropin values, and gonadal dysfunction as female fertility outcomes. Male fertility parameters included sperm quality, inhibin B levels, gonadotropins, testicular volume, and gonadal dysfunction. Four studies were conducted with males, 3 with females, and 1 with both genders. The study size in studies with females varied from 22 to 561 cancer survivors, of which 1 to 16 were STS patients. In studies with males, the number of participants ranged from 17 to 561, of which 2 to 23 were STS patients. The studies examined included a total of 28 females and 63 males with STS.
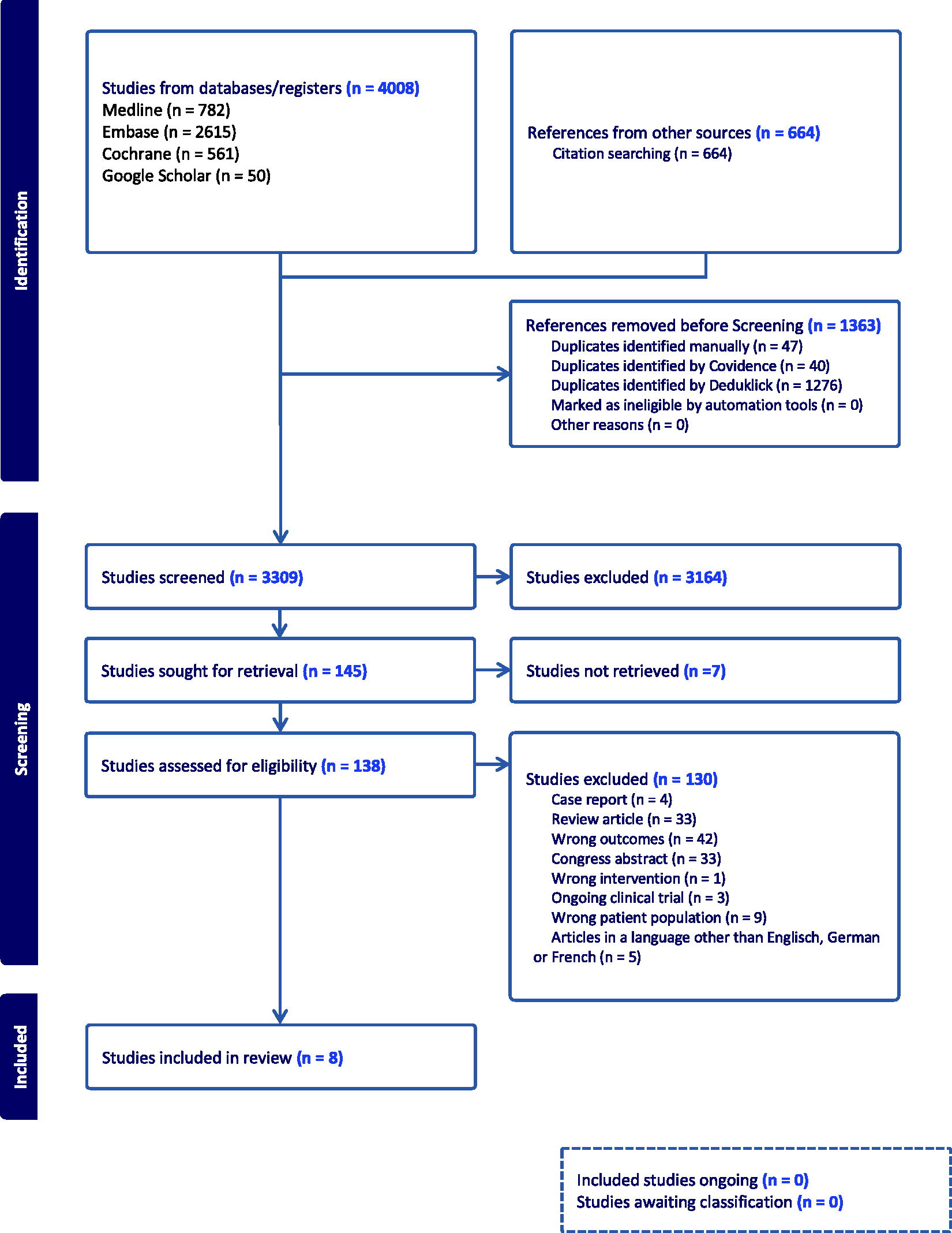
Identification of studies via databases and registers.
The age structure at diagnosis or start of treatment varied from <1 to 14 years for females and from <1 to 21 years for males. As a result, most of the participants were prepubertal, although the puberty status was not stated for each participant. Certain information, such as follow-up duration, age at diagnosis, or the treatment regimen used, applied to the entire study population in certain studies, which in some cases included heterogeneous tumor entities. Specific information on the STS patients was sometimes missing. As a result, this information may be slightly different for the subset of patients included in the analysis.
Data analysis
Suspected infertility was seen in 20 of 28 (71.4%, range 0–100%) of female (4 studies)20–23 and in 38 of 63 (60.3%, range 34.8–100%) of male (5 studies)19,21,24–26 STS patients (Table 4). Of the 8 studies, 5 studies19,20,22,23,26 contained information on the chemotherapeutic agent used, always alkylants such as cyclophosphamide or ifosfamide. One study, 24 used the alkylating agent dose score ADD, with 61.2% of cancer survivors receiving an alkylating agent and 38.8% receiving chemotherapy without alkylating agents. The remaining 2 studies21,25 did not contain specific information on the chemotherapeutic agent used and referred, in part, to common protocols, for example, to internationally recognized chemotherapy protocols. 21 The study of Rendtorff et al. 25 is the only one of the studies to use the term “suspected infertility;” however, the study was small and included only one female cancer survivor after STS. The extent of suspected infertility varied between the studies, although most of them showed that it was present. Only 1 study 20 showed no fertility impairment after chemotherapeutic treatment of soft tissue cancer. The data analysis shows a higher level of suspected infertility in the studies with females (71.4%) compared with those with males (60.3%).
Discussion
The aim of this review was to analyze the data on gonadotoxicity of chemotherapy for STS and to contribute to the improvement of fertility counseling in this context. Our data suggest that chemotherapeutic treatment of STS carries a high risk for fertility in both pre- and postpubertal females and males.
Our review has some strengths that should be emphasized. We also included prepubertal patients in our analysis. This considers the fact that STS are common in children and young adults. In addition, we focused on the effect of chemotherapy and excluded other gonadotoxic therapies such as radiotherapy or immunotherapies. By using the outcome parameter “suspected infertility,” we have considered various clinically relevant parameters and thus the heterogeneous data situation regarding fertility parameters in the existing studies in this field.
The weaknesses of our study must also be acknowledged. First, the exact pubertal status was not always known for all study participants. Second, the heterogeneity of the fertility parameters as well as the limited number of studies led us to use the broader outcome parameter “suspected infertility.” Interestingly, one of the reviewed studies used this term. 25 Balcerek et al. also used the term in their work on fertility after treatment of leukemia and solid tumors. 27 They defined the criteria for suspected infertility as AMH levels below 0.1 ng/mL in females, follicle stimulating hormone levels above 10 IU/L, and inhibin B levels below 80 pg/mL or azoospermia in males. And third, due to the frequently missing information on the exact chemotherapy regimen and dose used, we were unable to differentiate further according to the regimen used, dose, or age.
The age structure of the included studies reflects that STS occurs frequently in children and young adults. Most cancer survivors were prepubertal at the time of diagnosis or treatment. The assessment of gonadal function appears to be possible in this age group using conventional parameters such as AMH and gonadotropin levels, provided that sufficient follow-up is carried out. 28 Follow-up was adequate in most studies with males being reassessed after the onset or after puberty. In those with females, the follow-up time line was insufficient. According to our data, the prepubertal situation does not appear to provide protection against gonadotoxicity. For example, all prepubertal males in the study by Kenney et al. 26 showed a severe restriction in the semen analysis in the sense of crypto- or azoospermia. This was also the case >5 years after gonadotoxic treatment, which postulates irreversible gonadal damage. In contrast to earlier data, 29 more recent studies30–32 seem to show that prepubertal gonads are not protected but are more sensitive to gonadotoxic therapies, which supports our findings.
Assessment of treatment-related infertility in these cancers is challenging, as the gonadotoxic effects of their therapeutic interventions in STS have been less studied compared with more common cancers. The degree of gonadotoxicity depends on the specific therapeutic interventions used. 33 In early-stage disease, resection of the tumor is the treatment of choice, with or without neo- or adjuvant radiotherapy, dependent on the histological subtype and risk situation. Chemotherapy is usually applied for high-risk sarcomas, unresectable or R1-resected sarcomas, and advanced-stage disease. 7 Within the chemotherapy regimen, characteristics such as dosage and the choice of chemotherapeutic agents significantly influence the extent of gonadal damage. 33 Treatment for STS often involves polychemotherapy regimens with combinations containing alkylating agents, such as cyclophosphamide or ifosfamide, or anthracyclines, such as doxorubicin. 7 According to the ESMO guideline, 7 there is no formal evidence that alkylant-based polychemotherapy is superior to monochemotherapy with an anthracycline (doxorubicin) in terms of overall survival.
However, it may result in a better response and longer progression-free survival, which is why polychemotherapy is often used and may be a regimen of choice. For both these cytotoxic agents, a significant, dose-dependent gonadotoxicity has been demonstrated, which supports our results.34–38 The included study by Kenney et al. 26 shows that no or only reversible fertility impairment is to be expected at a cyclophosphamide dose <7.5 g/m2. In contrast, all males with doses >25 mg/2 were azoospermic. Consistent with this, gonadotropins were frequently, but not always, elevated. Interestingly, all males showed normal testosterone levels, possibly explained by a lower chemosensitivity of the Leydig cells. 30 The specific individual gonadotoxic risk is sometimes difficult to quantify. In addition to various guidelines,9,10,39 there are tools, such as the classification by Wallace et al. from 2005, 40 which help to assess the gonadotoxicity of chemotherapies and to counsel accordingly. The development of reliable calculation tools to assess the individual risk of gonadotoxicity, dependent on the therapy in question, but also taking into account patient- and disease-related factors, would be of significant benefit. This issue would require further investigation through targeted, large-scale prospective studies. One approach to reduce gonadotoxicity could be to lower the dose of chemotherapy. An ongoing study by Miyachi et al. is currently investigating the oncological safety of a dose reduction of cyclophosphamide and the effect on gonadotoxicity in rhabdomyosarcoma. 41
Apart from the extent of gonadotoxicity, the question of reproductive outcomes in STS patients arises in clinical practice of counseling in fertility preservation. These are poorly described in the literature for STS. Accordingly, none of the studies included in our review considers outcomes related to pregnancy rates, risks of oncologic therapy for pregnancy and offspring. In accordance with most studies, chemotherapy, unlike radiotherapy,42 does not appear to be associated with adverse pregnancy and offspring outcomes after cancer treatment,43,44 The work of Green et al. in the Childhood Cancer Survivor Study examined the pregnancy outcomes of females who survived childhood cancer.43 This included 480 females with STS. No increased late miscarriage or reduced live birth rate was found, regardless of whether alkylants or other chemotherapeutic agents had been used. After radiotherapy of the pelvis, however, there was an increased risk of low birth weight <2500 g. Pregnancy after chemotherapy is therefore generally considered safe in survivors of STS, provided the oncological prognosis is good.43,45
In the individual case of counseling for fertility preservation, several questions remain unanswered. The effect of age on risk of infertility could not be evaluated due to limited data. Furthermore, additional radiotherapy or immunotherapy and patient-specific factors such as the existing internal or urological comorbidities can have a negative influence. 9 Also, the most important, what is the effect of new treatment protocols currently developed?
As the drugs used in interdisciplinary therapy concepts for STS, and in particular for rhabdomyosarcomas in children, have remained practically unchanged over the last few decades, the results of our study are very up-to-date and can be used as a reference for today’s clinical routine. Nevertheless, progress in molecular genetics will enable us to investigate newer drugs with a different mechanism of action in the future and therefore intervening with gonadotoxicity and fertility. 46
However, counseling on fertility preservation measures will remain an important part of oncological treatment, which is likely to stay gonadotoxic despite advances in cancer therapy. In fact, counseling will become increasingly challenging as personalized treatments make it more difficult to estimate the individual risk of infertility. Large-scale studies such as the FertiTOX project, 12 organized by FertiPROTEKT (www.fertiprotekt.com), which collect comprehensive data, are therefore an important step toward improving counseling and protecting the fertility of cancer patients.
Conclusions
In conclusion, our review shows that data on the gonadotoxicity of chemotherapy for STS are very limited. However, there are sufficient data to estimate a high risk of chemotherapy-induced infertility in pre- and postpubertal females and males. This underlines the importance of comprehensive counseling and fertility protection for this complex tumor entity. Further research is needed to increase the quantity and quality of data and to assess the gonadotoxicity of new treatment protocols.
Footnotes
Acknowledgment
The authors would like to thank Dr. Elizabeth Kraemer for the linguistic revision.
Authors’ Contributions
M.v.W. and S.W. designed the systematic review. T.K. set up the templates for literature search. Literature was searched by A.R., F.P., and M.S. Data analysis was performed by M.S. Oncological advice was given by A.K. The article was written by M.S. All authors revised the final article.
Author Disclosure Statement
The authors have stated that there are no conflicts of interest in connection with this article.
Funding Information
The review is part of the project FertiTOX. FertiTOX reviews are funded by the Swiss cancer league (ID KLS-5650-08-2022).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
