Abstract
Purpose:
To investigate the actual status of fertility preservation techniques in oncofertility in Japan and to clarify the involvement of embryologists in this field.
Methods:
This survey was conducted online, targeting embryologists working at 622 facilities registered with the Japan Society of Obstetrics and Gynecology for assisted reproductive technology.
Results:
The response rate was 56.6%. In total, 56.8% of facilities used some form of cryopreservation as fertility preservation therapy for patients with cancer; patients’ age range was widely defined at each facility. The most common renewal frequency of cryopreserved specimens for patients with cancer was at 1-year intervals. The most common renewal methods were during patient visits to the hospital and contact by letter. Knowledge levels regarding fertility preservation therapy were not high among many embryologists, but respondents recognized the important role of embryologists in oncofertility.
Conclusions:
This study is the first to clarify the importance of embryologists in oncofertility. Many embryologists felt that their knowledge of fertility preservation was limited and considered it necessary to improve their education, including public certification. Guidelines for long-term storage systems, including methods for renewal of cryopreservation, need to be established.
Introduction
In recent years, cancer diagnostic methods and multidisciplinary treatment methods such as chemotherapy, radiotherapy, and bone marrow transplantation have advanced, and with the remarkable improvement in cancer treatment results, rates of complete remission among pediatric, adolescent, and young adult (AYA) patients with cancer have improved substantially. However, the antitumor effect of high-dose chemotherapy and radiation therapy has been reported to significantly reduce fertility.1–3 In 2004, efforts to preserve fertility to improve the quality of life among pediatric and AYA began to be discussed in Japan, and in 2012, the Japan Society for Fertility Preservation was established as the first oncofertility association in Japan. 4
In the field of oncofertility, cryopreservation technology for germ cells such as gametes (oocytes/sperm), embryos (fertilized oocytes), and ovarian tissue is essential. Similar to other health care providers, embryologists have a huge role in this field. Cryopreservation of embryos (fertilized oocytes) has a long history, and it is a desirable method for fertility preservation from the perspective of the long-term prognosis of offspring. The laboratory techniques in this field are nearly the same as those in assisted reproductive technology (ART). Furthermore, more than 130 live births after transplantation of frozen-thawed ovarian tissue have been reported worldwide. 5 In 2019, the American Society for Reproductive Medicine (ASRM) stated that “freezing of ovarian tissue has already passed the research stage,” 6 making this a method with great potential for the future. Because ovarian tissue freezing requires laparoscopic surgery for patients with an unstable general condition, and because it is often targeted toward pediatric and adolescent patients with cancer, only a few facilities in Japan can currently perform ovarian tissue cryopreservation. Few embryologists are speculated to have mastered the techniques involved in the cryopreservation of ovarian tissue. Regarding the cryopreservation of unfertilized oocytes, guidelines of the ASRM 7 and the American Society of Clinical Oncology 8 recognize unfertilized oocyte freezing as a technology that can be applied clinically. In 2014, the Japan Society of Obstetrics and Gynecology (JSOG) approved the clinical application of medically adapted oocyte freezing. Embryologists who have mastered embryo (fertilized oocyte) cryopreservation techniques can easily carry out cryopreservation of unfertilized oocytes.
Presently, no consensus exists on ART laboratory technology in oncofertility, such as when and how to freeze germ cells and tissues or how to thaw and fertilize them. In fact, fertility preservation therapy is performed in accordance with the philosophy of each ART facility, and technical disparities in fertility preservation therapy are assumed to exist among regions and facilities. To solve this problem, the present situation of ART laboratory technology related to oncofertility in Japan should be understood and these issues need to be clarified. Thus, the purpose of this study was to investigate the actual status of fertility preservation techniques in oncofertility in Japan and to clarify the role of embryologists in this field.
Materials and Methods
This survey was approved on January 7, 2021 (institutional review board’s approval no. 5093) by the Bioethics Committee of St. Marianna University School of Medicine. Survey respondents were embryologists working at 622 facilities registered with the JSOG for in vitro fertilization and embryo transfer (ART-registered facilities). The questionnaire was designed to investigate the implementation of fertility preservation therapy and the involvement of embryologists (Table 1). The questionnaire comprised 13 multiple-choice or open-ended questions. For multiple-choice questions, if the response did not match any of the provided choices for an item, the respondent was permitted to provide their own response. Of the 13 items in the questionnaire, 5 items were related to respondent information and ART facilities, and 5 items addressed information on the status of fertility preservation therapy. The other three items pertained to embryologists in oncofertility. This survey was conducted in an online format by sending a request letter to the hospital director or clinical department director of each facility and enclosing the QR code for the survey. The system design for the online survey was commissioned to an online research company (Macromill, Inc., Tokyo, Japan). The response period was from February 26 to March 24, 2021. In this survey, consent was obtained at the beginning of the questionnaire for use of the responses in the present research. Then, the questions were made available to respondents who gave their consent to participate in this study. In addition, it was possible to withdraw consent at any time even after starting the survey. Regarding privacy protection, because responses and tabulation were done in an online format, access restrictions were set and managed by assigning individual IDs and passwords.
Questionnaire on Fertility Preservation Therapy and Embryologists’ Involvement
Results
Survey response status and respondent/facility background
The responses were obtained from embryologists at 352 out of 622 ART facilities registered with the JSOG (response rate 56.6%). Consent was obtained from all respondents for research use. In total, 65.1% of respondents were women and 34.9% were men. Respondents’ ages ranged from 24 to 82 years; 6.8% were under age 30 years, 36.9% were in their 30s, 41.8% were in their 40s, 11.9% were in their 50s, and 2.6% were age 60 years or older. Embryologists had a mean ± standard deviation (SD), min–max 14.8 ± 6.8 (0–34) years of experience, with 77.0% having more than 10 years of experience. In addition, the number of embryologists at each respondent’s facility varied from 0 to 59, with a median of 3 (Supplementary Table S1). Embryologists were the target population for this survey; however, facilities that responded that they had zero embryologists did not have full-time embryologists; we assumed that medical doctors responded to this survey. We obtained survey responses from facilities in all 47 prefectures (Supplementary Fig. S1).
Status of fertility preservation therapy
In total, 200 facilities (56.8%) were implementing some form of cryopreservation as fertility-preserving therapy for patients with cancer (Fig. 1). Of these, 151 (75.5%) were certified by the JSOG as medically approved (Fig. 2A), 127 (63.5%) were registered for unfertilized oocytes, and 149 (74.5%) for embryos (fertilized oocytes). Fewer facilities (41 facilities, 20.5%) were registered for ovarian tissue (Fig. 2B). Regarding age restrictions for patients with cancer who were eligible for cryopreservation, 55.4% (93/168) of facilities had age restrictions for embryo (fertilized oocyte) cryopreservation, 62.8% (86/137) for unfertilized oocyte cryopreservation, 73.3% (33/45) for ovarian tissue cryopreservation, 12.8% (24/187) for sperm cryopreservation, and 13.0% (9/69) of facilities had age restrictions for testicular sperm cryopreservation (Fig. 3A). In those facilities with age restrictions, the median age restriction was 16.0–45.0 years for embryos (fertilized oocytes), 16.0–44.0 years for unfertilized oocytes, 1.5–40.0 years for ovarian tissue, 14.0–57.5 years for sperm, and 8.5–57.5 years for testicular sperm (Fig. 3B). The frequency of extended renewal of cryopreserved specimens for fertility preservation in patients with cancer was annual renewal at most facilities (Fig. 4A). The most common procedures for renewing the cryopreservation period in oncofertility were having the patient come to the facility (56.8%) and contacting the patient via a letter (54.9%); 28.2% of respondents said that they contacted the patient by phone, 14.7% by email, and 4.1% by managing the procedure through an app (Fig. 4B).
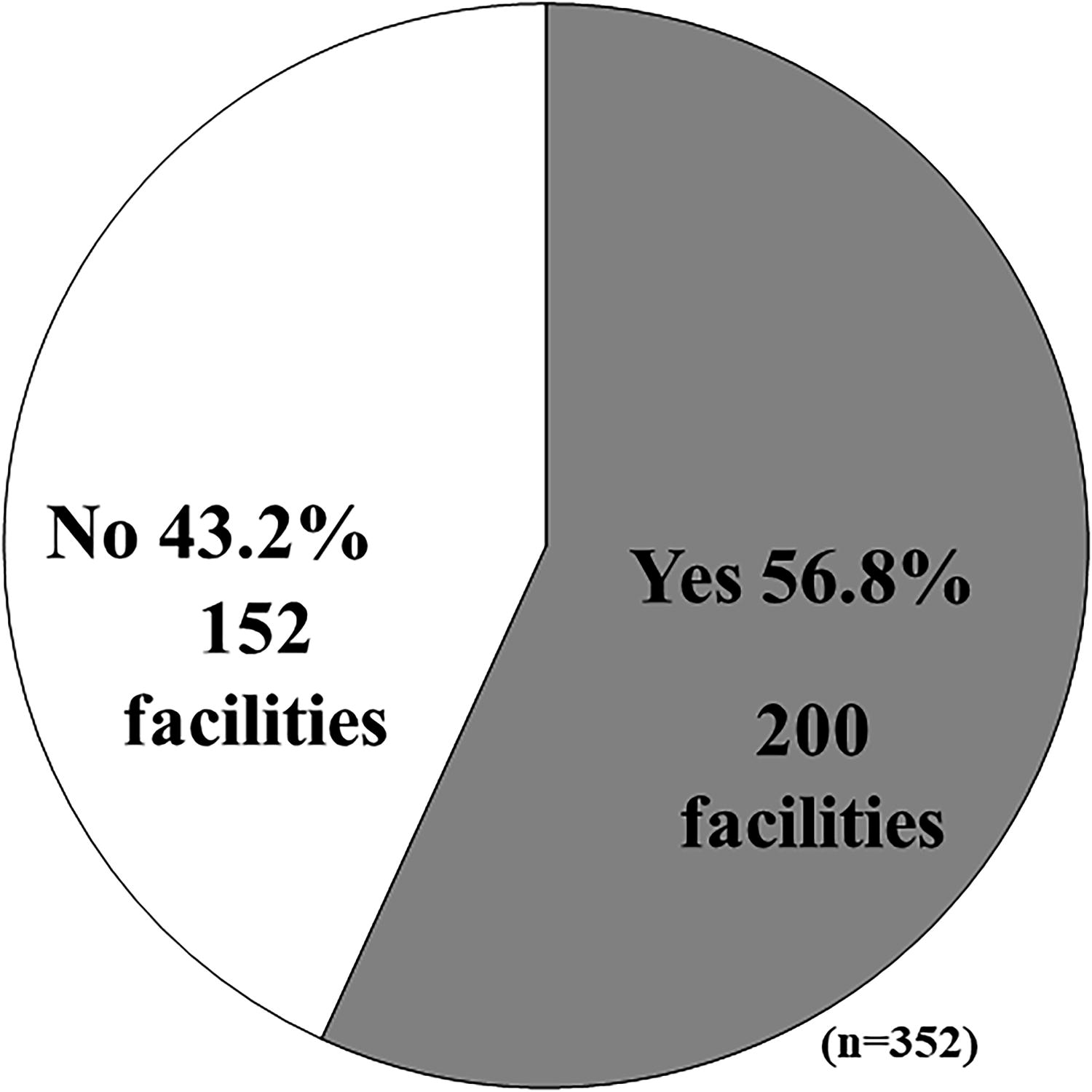
Does your institution offer fertility preservation therapy for patients with cancer using cryopreservation of germ cells and tissues?
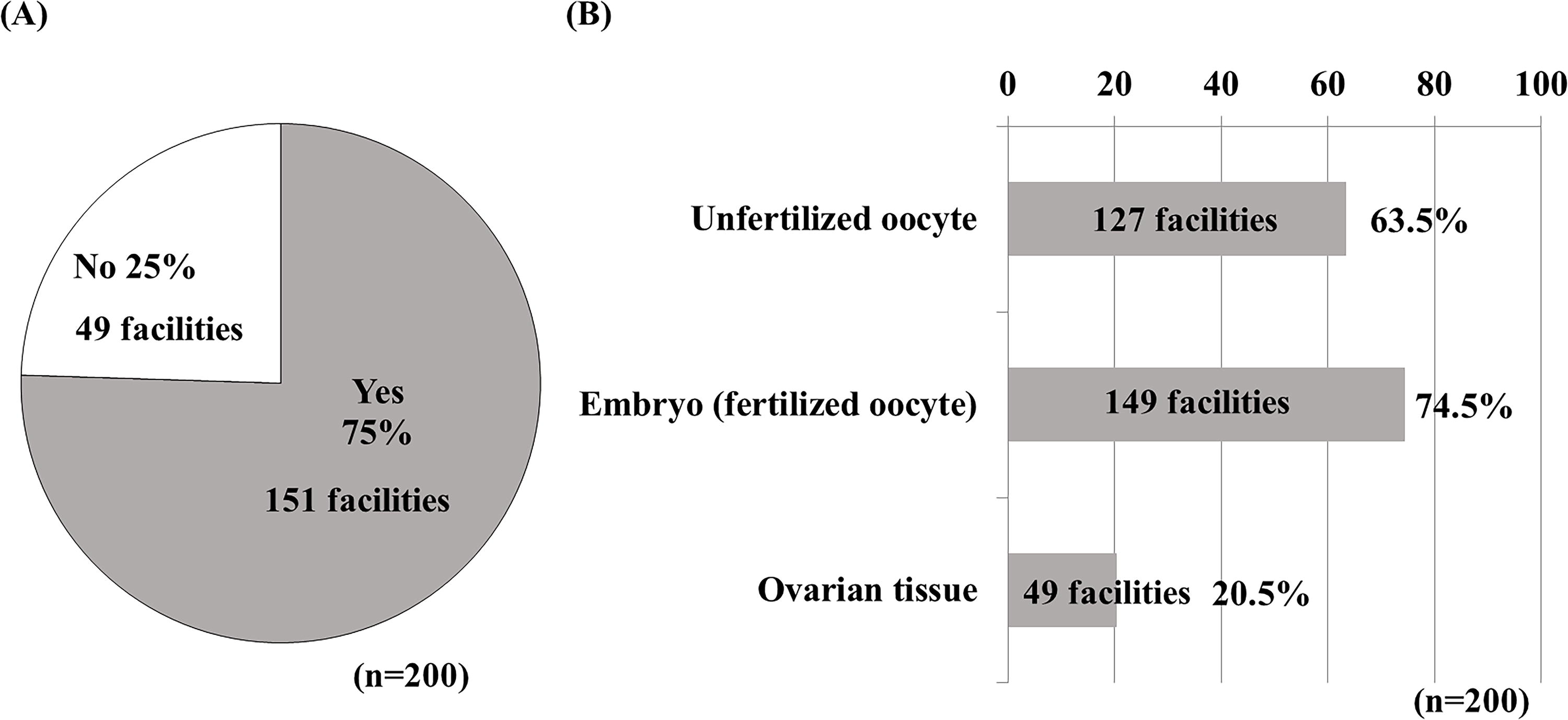
Registration status of facilities registered as medically approved by the Japanese Society of Obstetrics and Gynecology that perform cryopreservation for fertility preservation.
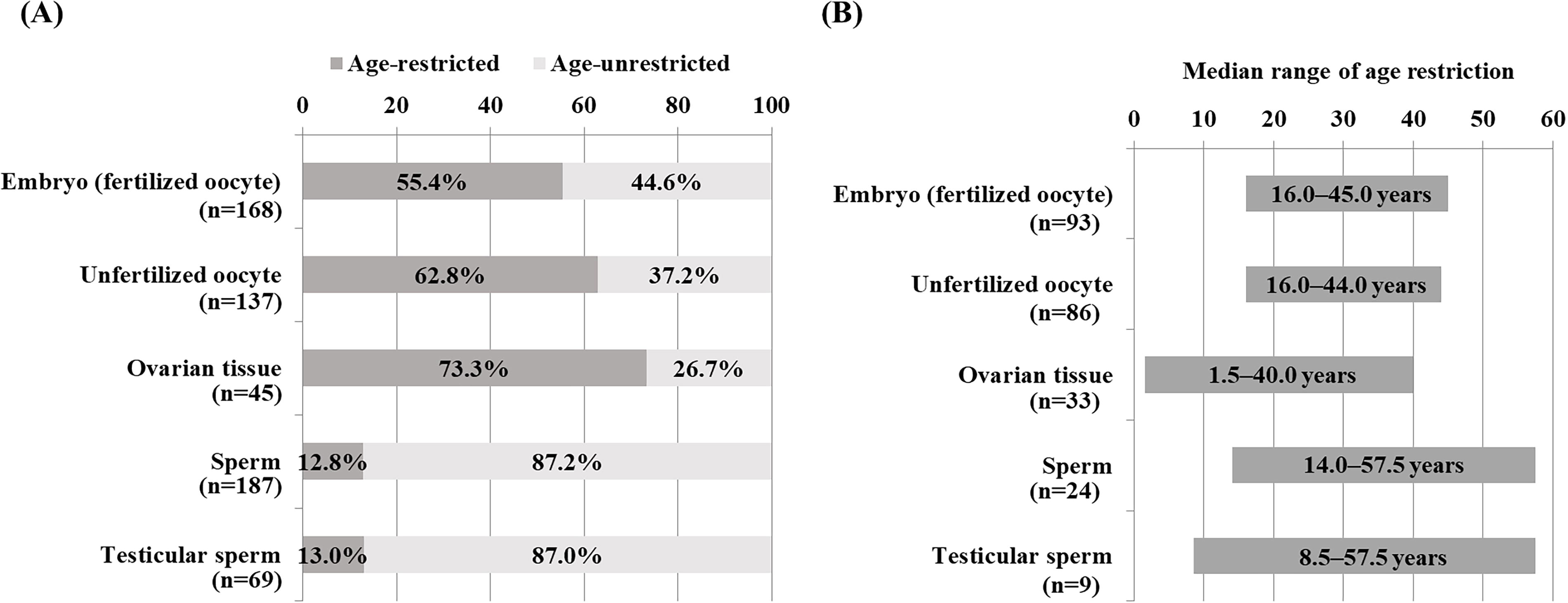
Age restrictions for patients with cancer undergoing cryopreservation as fertility preservation therapy.
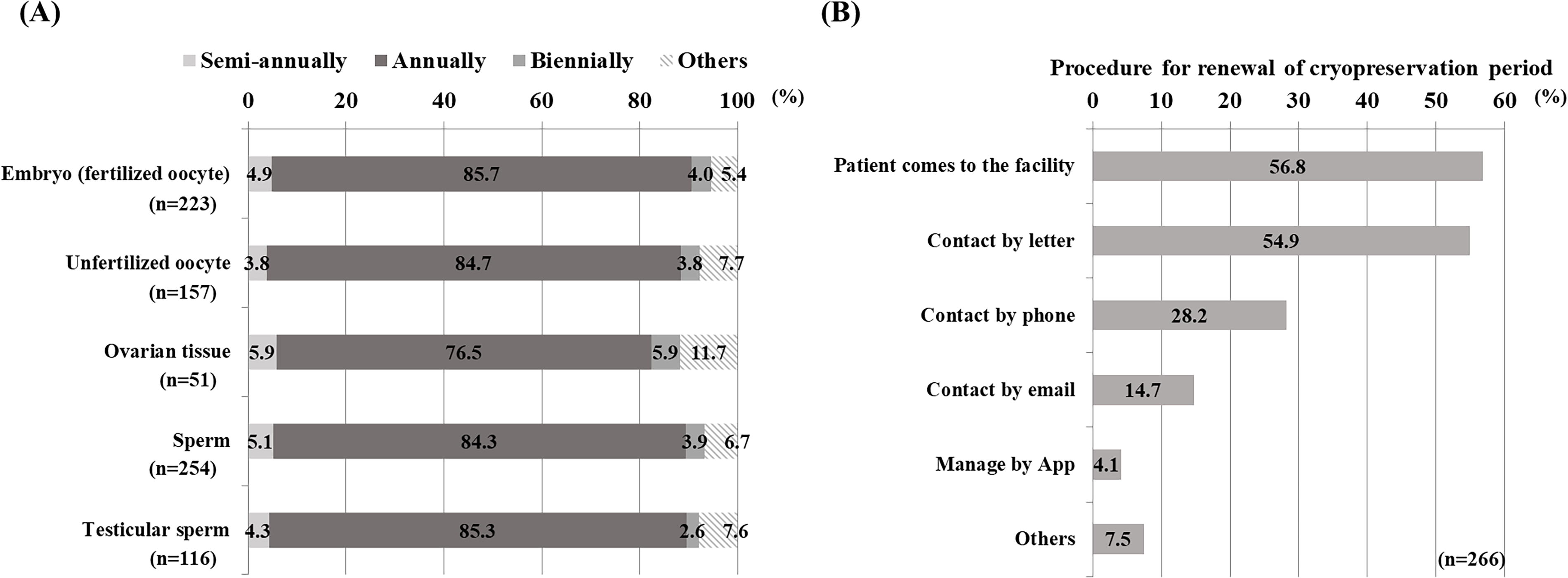
Extended renewal of cryopreserved specimens for fertility preservation in patients with cancer.
Involvement of embryologists in the practice of fertility preservation therapy
When embryologists were asked to self-assess their level of knowledge regarding fertility preservation therapy (100% was defined as needing no supplemental knowledge), 128 (36.4%) answered 50%, followed by 119 (33.8%) who answered 70% (Fig. 5). In total, 62.8% of embryologists reported a knowledge level of 50% or less.
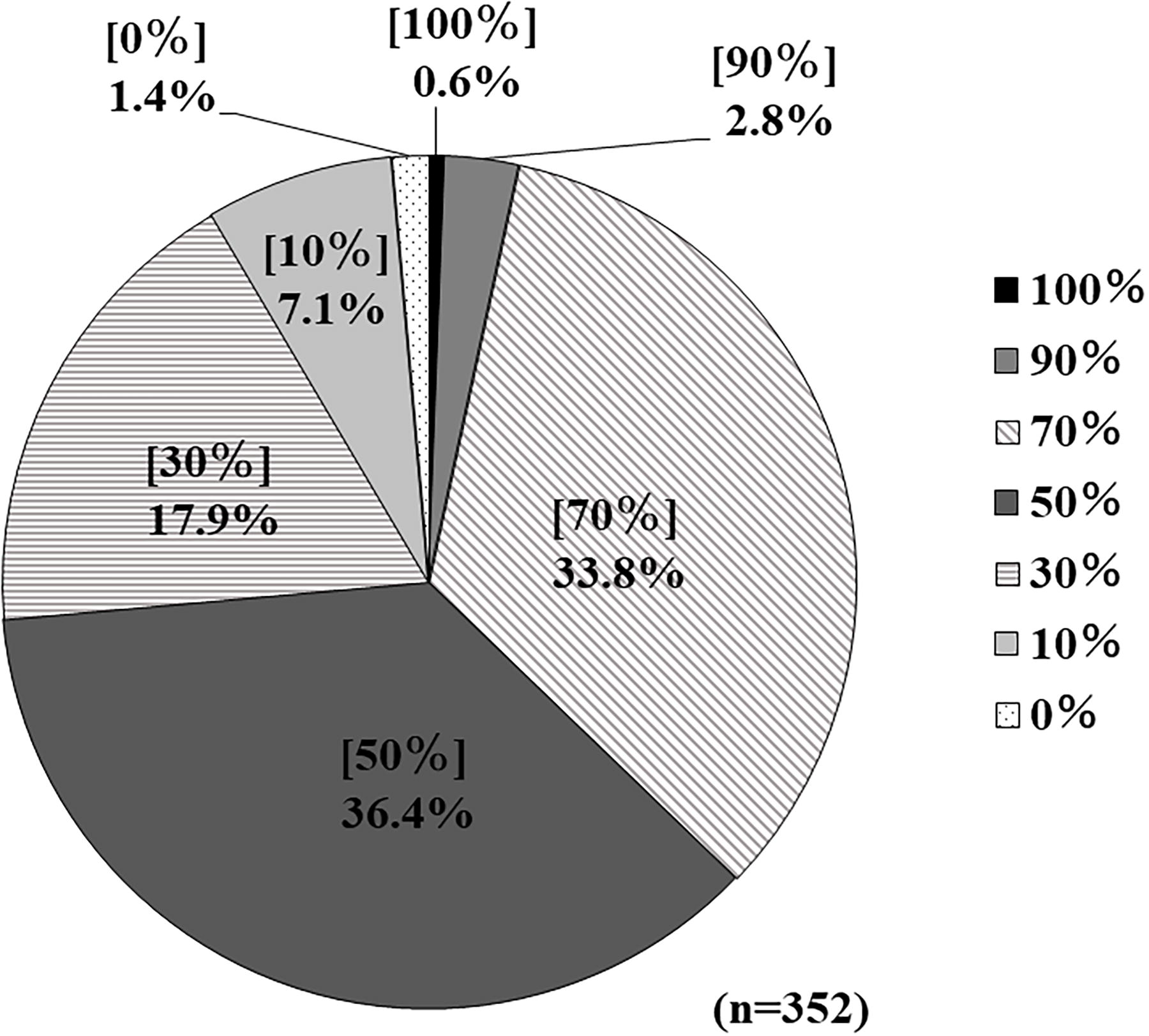
Embryologists’ level of knowledge about fertility preservation therapy. Embryologists were asked to self-rate their level of knowledge about fertility preservation therapy, with 100% indicating no supplemental knowledge was needed.
Open-ended responses were received from 41 facilities regarding difficulties in the ART laboratory when performing cryopreservation of germ cells and tissues for patients with cancer (Fig. 6). The most common response was that they could not contact the patient during the cryopreservation renewal procedure (n = 43.9%,18/41), followed by concerns about the long-term storage and management of frozen germ cells and tissues (n = 39.0%, 16/41). In addition, respondents had the following opinions: 9.8% (4/41) said they need more close contact with cancer treatment facilities; 7.3% (3/41) reported that in facilities with a small number of embryologists, it is difficult to learn special techniques for oncofertility, such as ovarian tissue cryopreservation, and to acquire embryologists with such skills; 4.9% (2/41) said they lacked the time to provide information on reproductive medicine to patients with cancer; 4.9% (2/41) said they lacked knowledge about oncofertility; and 4.9% (2/41) said there was insufficient time before oocyte retrieval and cryopreservation.
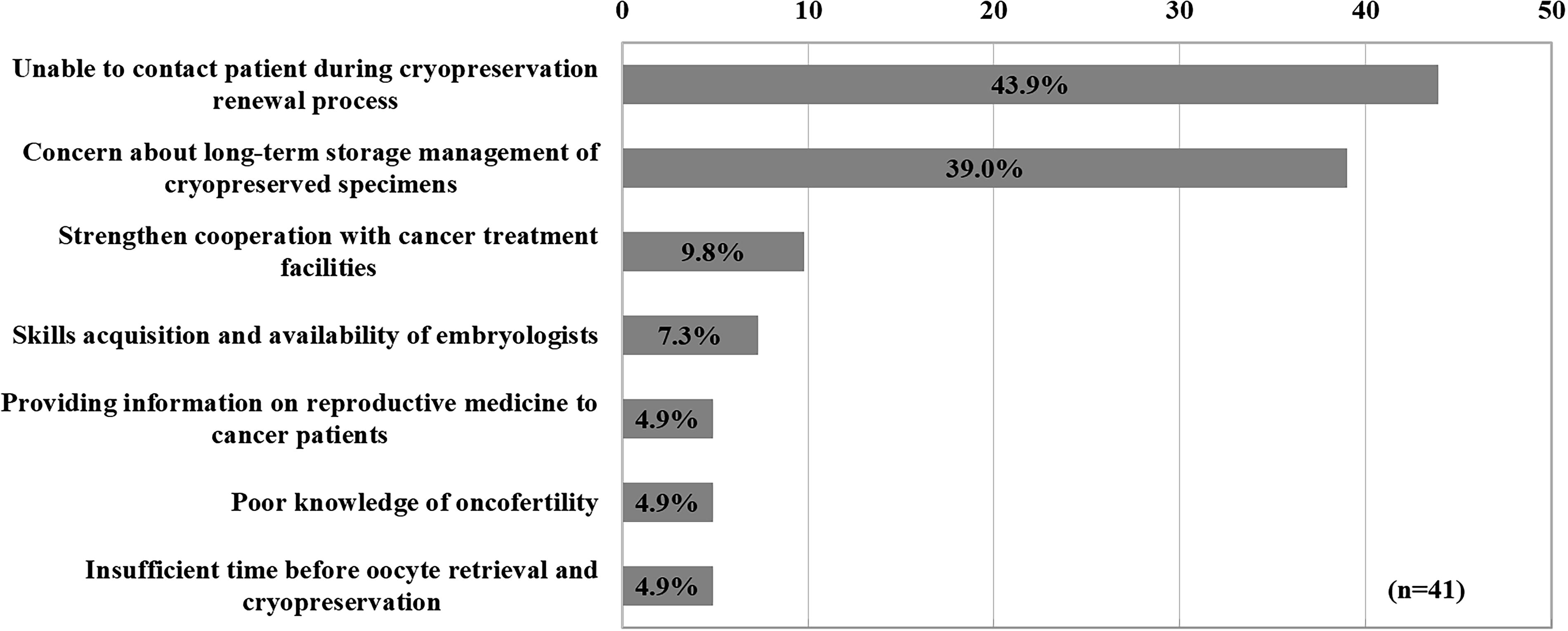
Difficulties in the laboratory when cryopreserving germ cells and tissues of patients with cancer.
When asked an open-ended question about the role of embryologists in oncofertility, many respondents indicated that embryologists have a critical role to play in oncofertility. A keyword search of the content of the free-text descriptions revealed many opinions regarding stress owing to the weight of responsibility, the importance of specialized skills and knowledge, and the need for public qualifications and a continuing education system (Fig. 7).
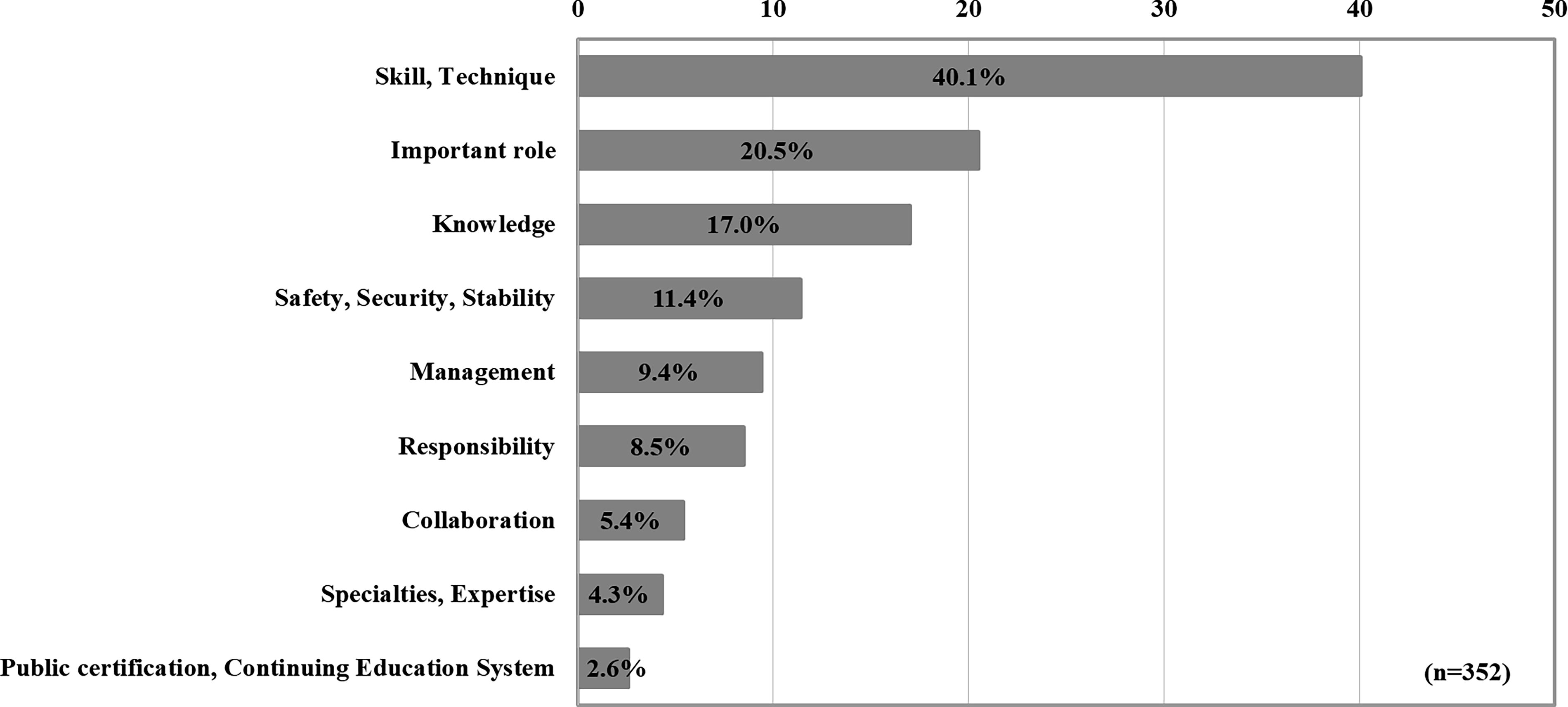
Main keywords in open-ended responses regarding the role of embryologists in oncofertility.
Discussion
Although several studies have reported on the status of fertility preservation therapy in Japan,9–13 this was the first nationwide survey of Japanese embryologists on the status of fertility preservation therapy and the involvement of embryologists in oncofertility. There is no mention of fertility preservation therapy in Japanese guidelines for reproductive medicine; consequently, there is no discussion regarding the relationship between laboratory work and oncofertility. 14 In addition, few studies have focused on fertility preservation therapy among embryologists worldwide. In fact, the European Society of Human Reproduction and Embryology and ASRM guidelines clearly state the importance of being able to provide cryopreservation techniques for fertility preservation therapy, but these guidelines do not specifically mention embryologists’ involvement in oncofertility.6,15,16 In our survey, we obtained responses from experienced embryologists from 352 facilities in all 47 prefectures of Japan, with ART facilities of varying sizes. We consider this survey to be of high quality and helpful in understanding the actual state of embryology technology in oncofertility in Japan and clarifying the role of embryologists as technicians in this field.
According to survey responses, in fertility preservation therapy for patients with cancer, 56.8% of responding facilities used some form of cryopreservation. Many facilities perform cryopreservation of oocytes, sperm, and embryos, which is also routinely performed in ART. Of these, 75.5% were certified by the JSOG as medically indicated, but we found that even facilities without such certification were performing cryopreservation for fertility preservation. In this regard, because subsidies for fertility preservation therapy among patients with cancer were expanded in Japan from April 2021, the number of facilities that are medically approved by the JSOG—a condition for receiving subsidies for egg and embryo cryopreservation—is expected to increase in the future. However, we found that fewer facilities were performing ovarian tissue cryopreservation compared with cryopreservation of oocytes, sperm, and embryos as fertility preservation therapy for patients with cancer. This means that patients are referred to higher-level medical institutions such as university hospitals when they are diagnosed with cancer, but few ART facilities in Japan collaborate in the cryopreservation of ovarian tissue. The reasons for this may include the need for specialized equipment and facilities for the collection and transplantation of ovarian tissue under laparoscopic conditions and the need for higher-level techniques for freezing and thawing. The situation in other countries is not well known; the ASRM requires that facilities be able to provide cryopreservation of embryos and oocytes as a condition for fertility preservation therapy. However, because the ASRM does not necessarily require cryopreservation of ovarian tissue, 6 we assume that not many facilities in the United States can provide ovarian tissue cryopreservation for patients with cancer, at least as in Japan.
Regarding age restrictions for eligible patients in oncofertility, many facilities had age restrictions for embryo, unfertilized oocyte, and ovarian tissue cryopreservation, but many facilities did not have age restrictions for sperm and testicular sperm cryopreservation. In facilities with age restrictions, we inferred that pediatric patients with cancer were included because the limit for testicular sperm and ovarian tissue cryopreservation was low whereas the age limit for embryo, unfertilized oocyte, and sperm cryopreservation was set mainly for AYA. In Japan, there are no national or organizational regulations regarding age restrictions in oncofertility. The present survey revealed that age restrictions are widely set for both germ cell and tissue cryopreservation at each facility. A survey of data collected from 30 European countries reported that some countries have age restrictions on the use of cryopreserved specimens: 17 countries have age restrictions for oocyte cryopreservation, 13 for embryo cryopreservation, and 7 for ovarian tissue cryopreservation. 16 European data on age restrictions and the upper age limit for oocyte cryopreservation are 42–55 years, 45–55 years for embryos, and 40–50 years for ovarian tissue; these values are higher than those in our survey in Japan. Regarding the age restriction for cryopreservation, the special adoption system may be a good reference for ethical discussions, such as consideration of the developmental environment of the future born child. Many local governments in Japan limit the age difference between parents and children to approximately 40–45 years from the perspective of parents being able to support their children physically and financially until they reach adulthood. For the same reason, further discussion on this point may be needed because few facilities in this survey set an age restriction for sperm and testicular sperm cryopreservation, and even those that did set such restrictions had a high upper age limit of 57.5 years.
The most common renewal frequency of extended cryopreservation for various germline cells and tissues in patients with cancer was set at 1-year intervals. The advantage of a longer interval is that patients can save time by not having to visit the hospital for procedures so they can concentrate on the treatment of their primary disease. However, if the interval is too long, there is less opportunity to check on the patient’s underlying disease status and their willingness to continue germline cryopreservation. Patients with a certain frequency should be contacted not only for cryopreservation renewal but also so that the attending reproductive physician can ascertain the treatment status of the primary disease; most facilities set this interval to 1 year. As for the method of renewal, 56.8% of facilities chose to have the patient visit the clinic in person. However, some facilities have patients renew without a visit, such as by letter, phone call, or email. Although not included in the survey, many facilities in routine clinical practice likely choose methods that do not require in-person visits for cryopreservation renewal of embryos (fertilized oocytes) or gametes (oocytes and sperm) for general ART patients. In fertility preservation among patients with cancer, establishing a system that allows patients to confirm their survival, treatment status of the underlying disease, and whether they wish to continue cryopreservation of their germ cells and tissues without having to visit the clinic in person would reduce the burden on patients.
Our survey revealed that many embryologists have low levels of knowledge about fertility preservation therapy. This may be owing to the fact that cryopreservation of embryos, unfertilized oocytes, sperm, and testicular sperm is commonly performed in ART, and the method is not very different for patients with cancer, as well as the fact that few facilities can perform ovarian tissue cryopreservation, a technique that is unique to oncofertility. In other words, embryologists are able to provide cryopreservation techniques in oncofertility without having knowledge of fertility preservation therapy, which may explain the lack of interest in fertility preservation therapy. Future research should investigate what kind of knowledge Japanese embryologists lack about fertility preservation therapy.
The survey revealed two main concerns among embryologists regarding oncofertility. One was that it is sometimes impossible to contact patients with cancer during the cryopreservation renewal process. The reasons for this are that, unlike patients undergoing infertility treatment, patients with cancer concentrate on the treatment of their primary disease and do not have a high level of awareness about the cryopreservation procedures that they have undergone. Moreover, embryologists do not know what level of care is needed for patients with cancer, making it difficult to contact them for renewal procedures. The second concern was about the long-term storage and management of cryopreserved specimens from patients with cancer. The reason for this is thought to be that patients with cancer require a longer cryopreservation period than infertile patients, and there are concerns about the ability of laboratories to store and manage cryopreserved specimens until the date when they can be used by patients with cancer. This includes concerns about whether staff fluctuations during that time would affect the management of cryopreserved specimens and their related information. The Japanese Society for Reproductive Medicine has established some institutional requirements for the management of cryopreserved specimens in ART in Japan, but other detailed requirements have not been established. Recently, Japanese guidelines for reproductive medicine have been published, providing broad recommendations for the safe long-term management of cryopreserved specimens by referring to the guidelines of overseas organizations. 14 Even now, however, detailed information such as the ASRM guidelines 17 is not available in Japan, and this is considered to be an issue for the future.
In this survey, many embryologists recognized that the role of embryologists in oncofertility is very important, and many commented on the importance of specialized skills and knowledge. The survey also clarified the need to establish a public qualification for embryologists and a continuing education system to ensure the quality of embryologists’ professional skills and knowledge.
Conclusions
We clarified the implementation status of embryologists’ techniques in oncofertility. Although most respondents were experienced embryologists, many had a low level of knowledge about fertility preservation therapy. To create a medical environment in which reproductive medical technology can be uniformly provided as fertility preservation therapy to patients with cancer in all 47 prefectures of Japan, education for embryologists regarding fertility preservation therapy should be improved. We also revealed difficulty among respondents in contacting patients with cancer during the renewal period of cryopreservation and concerns about the long-term storage and management of cryopreserved specimens, suggesting the need to develop guidelines and crisis management strategies to address these issues. The results of this survey suggest that embryologists play an important role in reproductive medicine in general, including oncofertility, and that an educational system should be established in consideration of public certification of embryologists.
Footnotes
Acknowledgment
The authors thank the ART-registered facilities of the Japanese Society of Obstetrics and Gynecology for their cooperation in this survey.
Authors’ Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by all authors. H.T. and O.O. drafted the article and prepared the figures and tables. N.S. oversaw the research. All authors contributed to the interpretation of the findings, editing of the article, and the approval of the final submitted version. All authors accept responsibility for the decision to submit for publication.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was funded by a Health Labour Sciences Research Grant from the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
