Abstract
Background:
N-acetylcysteine (NAC) is a classical mucolytic agent that, in addition to its mucolytic activity, also exhibits antioxidant activity. This could be beneficial in treating chronic inflammatory airway diseases, including asthma.
Background:
We evaluated the ability of NAC to modulate airway defense mechanisms, airway reactivity, inflammation, and remodeling after 10 days of administration [20 and 60 mg/(kg·d)] in an experimental guinea pig model of allergic inflammation.
Methods:
The concentrations of inflammatory cytokines (interleukins: IL-4, IL-5, IL-10, IL-12, and IL-13), granulocyte macrophage-colony stimulating factor (GM-CSF), interferon-gamma (IFN-γ), and tumor necrosis factor-alpha (TNF-α) were measured in bronchoalveolar lavage fluid using a multiplex detection method. The concentration of remodeling marker transforming growth factor beta-1 (TGF-β1) was measured in lung homogenates using enzyme-linked immunosorbent assay. In vivo, changes in specific airway resistance and number of cough efforts were determined. Tracheal smooth muscle reactivity was evaluated in vitro. Ciliary beat frequency (CBF) indicated mucociliary clearance.
Results:
A 10-day administration of NAC at a higher dosage led to a significant decrease in the regulatory cytokines IL-4, IL-5, and GM-CSF. NAC, in both dosing schedules, decreased the levels of TGF-β1. NAC at a higher dosage reduced the number of chemically induced cough reflexes and CBF. NAC did not affect airway hyperreactivity parameters.
Conclusion:
NAC is a multifactorial drug, and under our experimental conditions of allergic inflammation, it showed positive effects on the levels of regulatory cytokines and growth factors, which probably led to a reduction in the intensity of airway defense mechanisms.
Introduction
The most well-known mucolytic is N-acetylcysteine (NAC), but its mucolytic activity has not been well established to date. 1 The pharmacokinetics of NAC depend on the route of its administration. Aerosol administration of NAC dissociates the disulfide bonds of mucin and other gel components cross-linked by disulfide bonds, decreasing mucus viscosity. 2 Hyperresponsive asthmatics may develop bronchospasm as an adverse effect after aerosol administration of NAC. 3 Oral NAC has low bioavailability, 4 and is deacetylated to cysteine, whose thiol group has reducing and antioxidant properties. 5 After oral administration, NAC is not found in respiratory tract secretions. 6 Cysteine is a substrate in glutathione (GSH) biosynthesis, a crucial intra- and extracellular antioxidant. 7 NAC is attributed to antioxidant effects because it contains free thiol groups. A related NAC compound, N-isobutyrylcysteine, which has higher levels of free thiol groups compared with NAC, has shown no effect on exacerbation rates in Chronic obstructive pulmonary disease, 8 unlike NAC,9,10 which calls into question the credibility of the free thiol group hypothesis for NAC activity. Another property of NAC that could be beneficial in modulating asthma pathophysiology is its anti-inflammatory activity. The anti-inflammatory action of NAC in smokers was manifested by a reduction in lysozyme and lactoferrin concentrations, 11 and also by its ability to suppress the activity and number of neutrophils and macrophages, which were recorded in the bronchoalveolar lavage (BAL) fluid of smokers.12,13 NAC can inhibit the adhesion of bacteria to ciliated epithelial cells under in vitro conditions.14,15 Consequently, in this study, we focused on the ability of NAC to modulate airway defense mechanisms and airway reactivity in the context of changes in the levels of established inflammatory and regulatory cytokines, namely interleukins (IL-4, IL-5, IL-10, IL-12, and IL-13) granulocyte macrophage-colony stimulating factor (GM-CSF), interferon-gamma (IFN-γ), tumor necrosis factor-alpha (TNF-α), and remodeling marker transforming growth factor beta-1 (TGF-β1).
Material and Methods
Ethical approval
Experimental procedures were carried out under the Slovak Republic and European Union regulations for using laboratory animals following guidelines on animal welfare. The study was approved by the Ethics Committee of the Jessenius Faculty of Medicine in Martin, Comenius University in Bratislava (decision No. EK 19/2020).
Experimental animals
Male Dunkin-Hartley guinea pigs were purchased from an approved animal breeding facility (Velaz, Prague, Czech Republic). The weight of the animals was from 200 to 250 g. The animals underwent a quarantine period of one week. Animals were kept in an animal house under standard conditions (constant temperature 22 ± 2°C, humidity 50%–60%, 12-hour light/dark cycles, access to water and food ad libitum).
Chemicals
NAC was obtained as the commercially available product (ACC injekt; solution for injection; Sandoz Pharmaceuticals, Slovenia). Citric acid, histamine dihydrochloride, acetylcholine chloride, salmeterol xinafoate (SAL), budesonide (BUD), and ovalbumin ([OVA], chicken egg albumin grade V) were purchased from Sigma-Aldrich Chemicals (St. Louis, MO, USA). Codeine (COD) phosphate was obtained from Slovakofarma (Hlohovec, Slovakia). Aluminum hydroxide and chemicals used to prepare the Krebs-Henseleit solution (sodium chloride, potassium chloride, calcium chloride, magnesium sulfate, potassium hydrogen phosphate, sodium hydrogen carbonate, and glucose) were supplied by Centralchem (Bratislava, Slovakia).
All chemicals for nebulization (citric acid, histamine dihydrochloride, OVA, SAL, and BUD) were dissolved in 0.9% saline.
Experimental design
Animals were randomized into groups in the experiment, with each group consisting of ten animals (n = 10).
Experimental groups:
Control group (OVA–): healthy animals, administration of saline 1 mL/kg·d peroral (p.o.) for 21 days; Negative control group (OVA+): OVA-sensitized animals, no treatment; Therapeutic group NAC 20 mg: OVA-sensitized animals, administration of NAC 20 mg/kg·d p.o. for last 10 days of sensitization scheme; Therapeutic group NAC 60 mg: OVA-sensitized animals, administration of NAC 60 mg/kg·d p.o. for last 10 days of sensitization scheme.
Experimental model of allergic airway inflammation
The experimental model of allergic airway inflammation in guinea pigs was achieved by sensitizing the animals to the allergen ovalbumin for 21 days. The sensitization protocol was adapted based on the methodology outlined by Franova et al. 16 An injectable suspension of OVA (consisting of 5 mg of OVA electrostatically adsorbed on 100 mg of aluminum hydroxide in 0.5 mL of saline) was injected into an animal intraperitoneally (first and fourth day of sensitization) and subcutaneously (first and ninth day of sensitization). Inhalations of OVA were performed on the 12th (1 minute), 15th (1 minute 30 seconds), 18th (2 minutes), and 20th (3 minutes) day of sensitization.
Tracheal smooth muscle (TSM) reactivity in vitro
Experimental animals were euthanized via cervical dislocation. Tracheal samples were positioned within the chamber of a system designed for organ tissue bathing (Experimetria IS-02 Eight-channel tissue bath system, Experimetria Ltd., Budapest, Hungary). The system chambers were filled with Krebs-Henseleit’s buffer solution (in mM): NaCl 112.9, KCl 4.7, CaCl2 2.8, MgSO4 0.5, NaHCO3 24.9, glucose 11. The system was persistently supplied with a gas composition of 95% O2 and 5% CO2. It was kept at 36 ± 0.5°C and a pH of 7.5 ± 0.1. In the first 20 minutes, the tracheal strips were adjusted to a tension of 4 g. After this phase, the tension of the strips was modified and brought down to a baseline of 2 g (adaptation phase: 20–40 minutes). Each tracheal sample underwent a washing process at intervals of 10 minutes. The reactivity of the TSM was evaluated based on the alterations in the amplitude of its isometric contraction (measured in grams). This was done in response to the stepwise addition of 100 μL of the bronchoconstrictor histamine or acetylcholine in concentrations ranging from 10 to 1 mM to the tracheal samples situated in the organ baths.
Airway reactivity in vivo
The calculation of specific airway resistance (sRaw) is based on the Pennock method. 17 sRaw under in vivo conditions was assessed in double chamber plethysmograph Hugo Sachs Elektronik type 855 (Hugo Sachs Elektronik, Hugstetten, Germany). In a body plethysmograph, an animal in a conscious state was subjected to an aerosol of the bronchoconstrictor histamine (0.1 mM) for 30 seconds, which was nebulized by Pari Jet Nebuliser (Paul Ritzau, Pari-Werk-GmbH, Germany). Following each inhalation, the alterations in air pressure between the chambers of the plethysmograph were utilized to compute the mean in vivo sRaw values using the Hugo Sachs Elektronik Pulmodyn Pennock plethysmograph software (Hugo Sachs Elektronik, Hugstetten, Germany).
Sensitivity of cough reflex in vivo
For assessing the sensitivity of the cough reflex, we utilized double chamber plethysmograph Hugo Sachs Elektronik type 855 (Hugo Sachs Elektronik, Hugstetten, Germany) with attached Pari Jet Nebuliser (Paul Ritzau, Pari-Werk-GmbH, Germany). Conscious experimental animals were exposed to an aerosol of tussogenic substance (citric acid 0.3 M) and inhaled for 3 minutes. In this period, the pneumotachograph attached to the nasal chamber identified the total count of cough efforts. The total number of cough efforts was compared by observation of two trained observers.
Cytokine concentration analysis in BAL fluid
Following the cervical dislocation, a cannula was inserted to flush out the right bronchus with warm saline (37°C) at a volume of 10 mL/kg body weight (b.w.). Retrieved BAL fluid was centrifugated at 1500 g for 15 minutes at a temperature of 4°C (IEC CL31R Multispeed Centrifuge, Thermo Fisher Scientific, Waltham, MA, USA). The collected supernatant was used for qualitative and quantitative analysis of selected inflammatory and regulatory cytokines (IL-4, IL-5, IL-10, IL-12, IL-13, GM-CSF, IFN-γ, and TNF-α) by a multiplex assay technology (Bio-Plex 200 protein array system, Bio-Rad Laboratories, Hercules, CA, USA) using a commercial Bio-Plex Pro Rat Cytokine Th1/Th2 Immunoassay Panel kit (Bio-Rad, Hercules, CA, USA) following the instructions provided by the manufacturer. Output data were processed by a high-speed processor and analyzed using Bio-Plex ManagerTM 6.0 software (Bio-Rad Laboratories, Hercules, CA, USA).
TGF-β1 concentration analysis in lung tissue homogenates
Total protein concentrations for TGF-β1 in the homogenates from lung tissue were determined using the detergent compatible (DC) protein assay (Bio-Rad, Hercules, CA, USA). The absorbance of the homogenates was measured spectrophotometrically at 750 nm using a Varioskan Flash Spectral Scanning Multimode Reader (Thermo Fisher Scientific, Waltham, MA, USA).
The TGF-β1 concentration was measured in lung homogenates using an enzyme-linked immunosorbent assay kit, namely Guinea Pig Growth Factor Beta (MyBioSource, San Diego, USA).
The homogenates for TGF-β1 were prepared by weighing 100 mg from the lung tissue. The tissue was washed in ice-cold 0.01 M phosphate buffer with pH 7.0–7.2 and immediately homogenized with 1000 μL of lysing solution (Tissue Extraction Reagent I, Invitrogen, Thermo Fisher Scientific, MA, USA) with protease inhibitors (Protease Inhibitor Cocktail, Sigma-Aldrich, USA) using Laboratory Homogenizer Stuart SHM2/EURO (Bibby Scientific, UK). Lung homogenates were centrifuged in a centrifuge (7500G, 10 minutes, 4°C) and then the supernatant was collected and stored in a freezer (−80°C).
The prepared homogenates were subjected to two freeze–thaw cycles and were rapidly centrifuged at 1500 g for 15 minutes at room temperature. The created supernatants were taken up and stored in sterile tubes at −80°C.
The created absorbances were measured spectrophotometrically at 450 nm on the Varioskan Flash Spectral Scanning Multimode Reader (Thermo Fisher Scientific, Waltham, MA, USA). The final concentrations were analyzed by calibration curve with SkanIt Software for Varioskan Flash version 2.4.5 (Thermo Fisher Scientific, Waltham, MA, USA).
Ciliary beat frequency (CBF) in vitro
Right after the animal was euthanized, a midline incision was carefully executed on the upper segment of the trachea. A cytology brush (2.5 mm in diameter) was gently rotated within the trachea to collect a sample of the tracheal cilia. After collecting the sample, the brush was transferred into a basal medium (RPMI 1640 Medium, Thermo Fisher Scientific, Waltham, MA, USA). The biological material was moved from the cilia suspension onto a microscopic slide in the subsequent step. This was done in preparation for creating specimens used to study the CBF. The cilia suspension and the microscopic slides were kept at a constant temperature (37°C) using Thermo-Shaker 100C for microtubes and PCR plates (SIA Biosan, Latvia). Evaluation of specimens at a microscopic level was achieved using high-speed imaging. This involved a high-speed video camera (Basler A504kc, Basler AG, Germany) connected to an inverted phase contrast microscope (Zeiss Axio Vert. A1, Carl Zeiss AG, Göttingen, Germany) and a computer. A 40 × objective lens was used, providing a total magnification of 400 ×.
Ciliary Analysis software (LabVIEWTM) was used for high-speed video microscopy analysis. This software helped with image processing and detected the region of interest (ROIs) near the surrounding of beating cilia dependent on the variations in the intensity of gray levels. The time-based recording of the intensity variance curve for the chosen appropriate ROIs was processed by the fast fourier transformation algorithm to conduct an automated and real-time analysis of the kinetics of movement.
Statistical analysis
Statistical data were calculated, and graphs were created using the GraphPad Prism Software (version 8.1.0., San Diego, CA, USA). All obtained data were expressed as the mean ± the standard error of the mean using a one-way analysis of variance followed by Dunnett’s multiple comparison test. If the measured p-value was less than 0.05, the difference was considered statistically significant (*p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001).
Results
TSM reactivity in vitro
The effect of NAC administered for the last 10 days of the experiment at two therapeutic doses (20 and 60 mg/kg b.w, orally) on TSM reactivity was tested in vitro and compared with salmeterol. The alterations in the contraction intensity of isolated TSM samples were measured after adding 100 μL of the constrictor mediators histamine and acetylcholine in cumulative doses (10 nM-1 mM). The reduction in TSM reactivity was not statistically significant (Fig. 1).
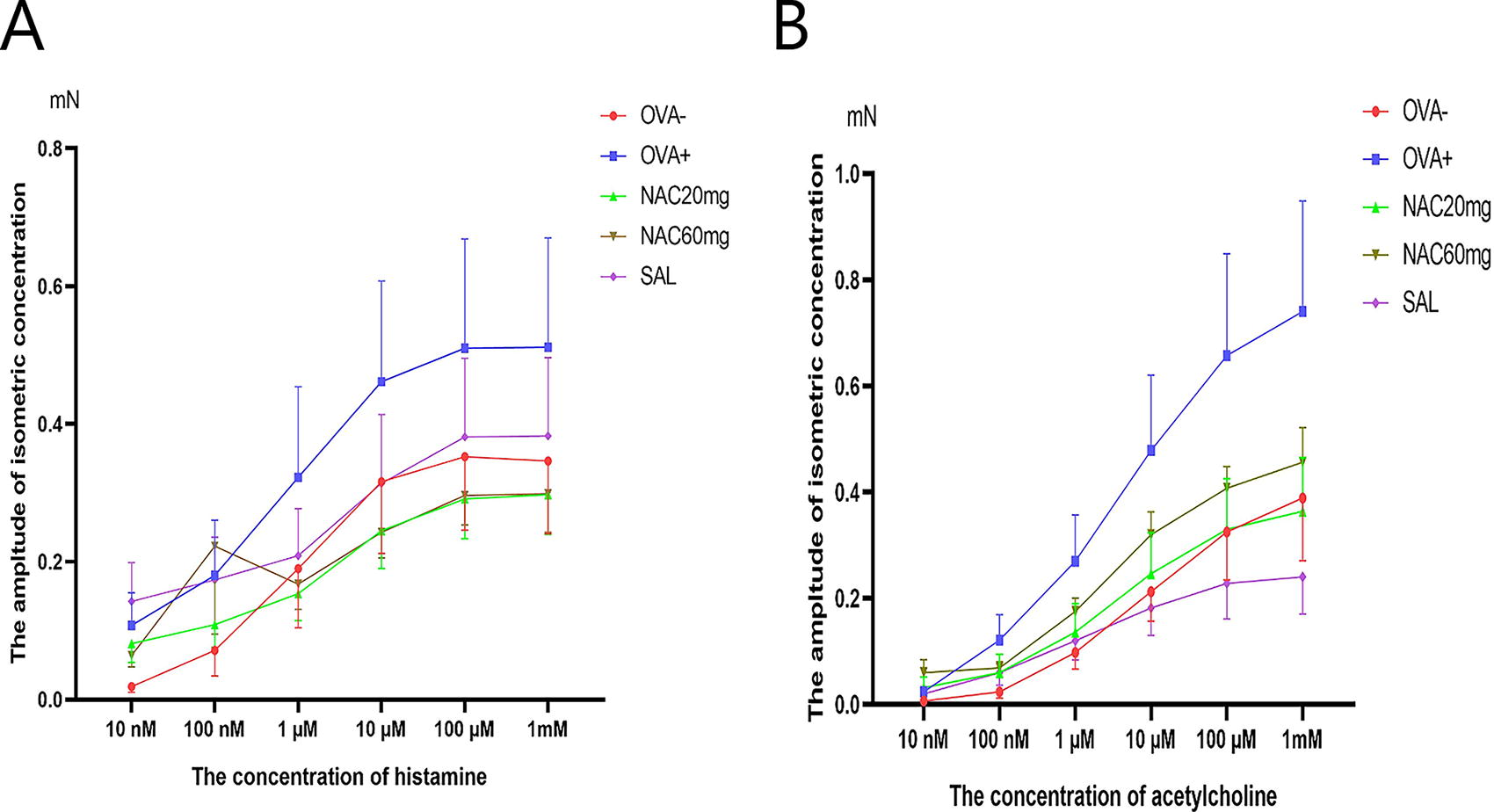
TSM reactivity after 10 days of NAC administration and a reference anti-asthmatic drug. The control group is represented by healthy animals receiving saline for 21 days (OVA-), and the negative control group is represented by animals sensitised to ovalbumin for 21 days (OVA+). OVA-sensitised animals were administered NAC in a 20 mg/kg b.w. p.o. (NAC20mg), 60 mg/kg b.w. p.o. (NAC60mg) or salmeterol (SAL) for 10 days. The changes in TSM reactivity were measured 24 h after the last OVA challenge or substance administration.
Airway reactivity in vivo
The effect of NAC on the reactivity of airway smooth muscle was evaluated by observing alterations in the sRaw parameter following the inhalation of a histamine aerosol (0.1 mM, 30 seconds) and compared with salmeterol. Twenty-one days of OVA sensitization led to significant elevation of sRaw in the negative control group (OVA+) under basal conditions (Fig. 2, OVA+ vs. OVA-, p < 0.05). Administration of NAC did not significantly affect this parameter at any tested doses.
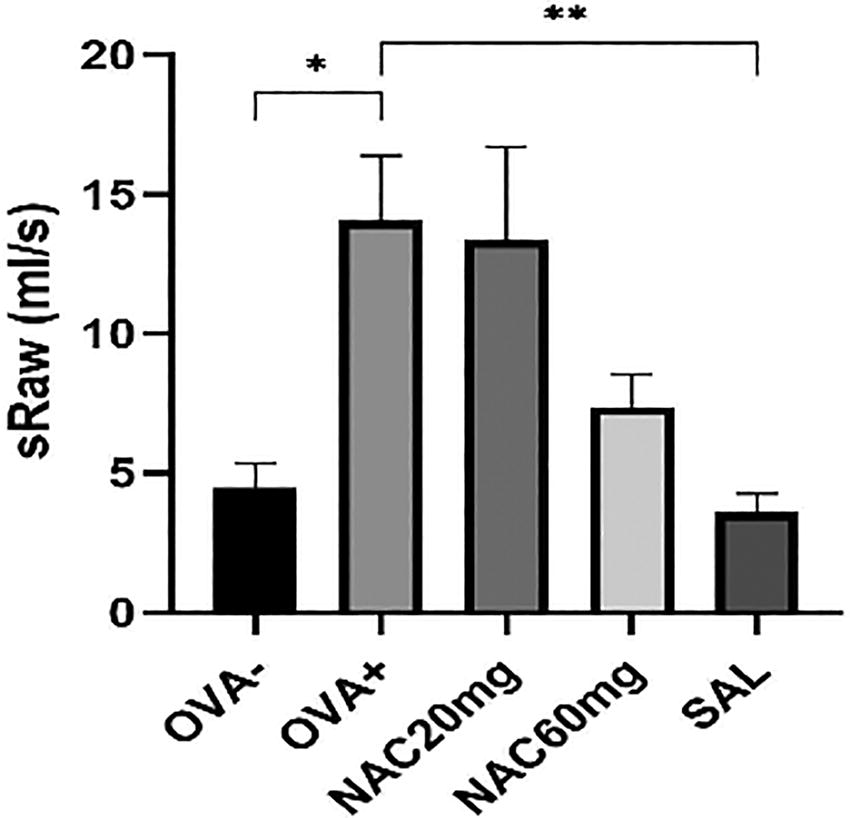
Evaluation of sRaw after 10 days of NAC administration and a reference anti-asthmatic drug. The control group is represented by healthy animals receiving saline for 21 days (OVA-), and the negative control group is represented by animals sensitised to ovalbumin for 21 days (OVA+). OVA-sensitised animals were administered NAC in a 20 mg/kg b.w. p.o. (NAC20mg), 60 mg/kg b.w. p.o. (NAC60mg) or salmeterol (SAL) for 10 days. The changes in sRaw was measured 24 hours after the last OVA challenge or substance administration. The data are expressed as the mean ± S.E.M; n = 7-10; *P < 0.05 OVA+ vs. OVA-; **P < 0.01 OVA+ vs. tested substance.
Sensitivity of cough reflex in vivo
A 30-second inhalation of citric acid (0.3 M) was used to induce a cough reflex reaction in conscious animals to determine the sensitivity of the cough reflex. The therapeutic effect of NAC was compared with codeine. Ten-day treatment with 60 mg/kg NAC (NAC 60 mg) in OVA-sensitized animals resulted in a significant reduction in cough efforts (Fig. 3, OVA+ vs. NAC 60 mg, p < 0.05) compared to OVA-sensitized animals. The antitussive effect of NAC was lower than that of the reference opioid codeine (Fig. 3, OVA+ vs. COD, p < 0.0001).
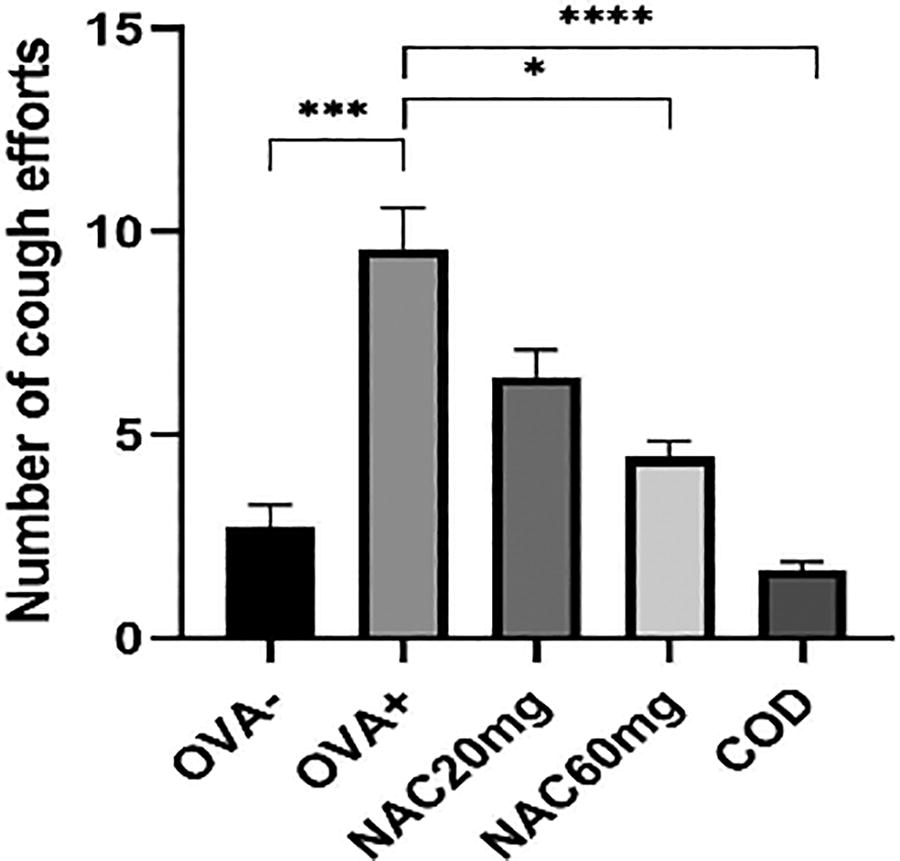
Number of cough efforts after 10 days of NAC administration and a reference antitussive drug. The control group is represented by healthy animals receiving saline for 21 days (OVA-), and the negative control group is represented by animals sensitised to ovalbumin for 21 days (OVA+). OVA-sensitised animals were administered NAC in a 20 mg/kg b.w. p.o. (NAC20mg), 60 mg/kg b.w. p.o. (NAC60mg) or codeine (COD). The number of changes in cough efforts was measured 24 hours after the last OVA challenge or substance administration. The data are expressed as the mean ± S.E.M; n = 6-10; ***P < 0.001 OVA+ vs. OVA-; **** P < 0.0001, *P < 0.05 OVA+ vs. tested substance. Number of cough efforts after 10 days of NAC administration and a reference antitussive drug. The control group is represented by healthy animals receiving saline for 21 days (OVA-), and the negative control group is represented by animals sensitised to ovalbumin for 21 days (OVA+). OVA-sensitised animals were administered NAC in a 20 mg/kg b.w. p.o. (NAC20mg), 60 mg/kg b.w. p.o. (NAC60mg) or codeine (COD). The number of changes in cough efforts was measured 24 hours after the last OVA challenge or substance administration. The data are expressed as the mean ± S.E.M; n = 6-10; ***P < 0.001 OVA+ vs. OVA-; **** P < 0.0001, *P < 0.05 OVA+ vs. tested substance.
Cytokine concentration analysis in BAL fluid
Concentrations of cytokines (IL-4, IL-5, IL-10, IL-12, IL-13, GM-CSF, IFN-γ, and TNF-α) were evaluated in samples of BAL fluid. The therapeutic effect of NAC was compared with budesonide. Ten days peroral administration of NAC in the dosage 60 mg/kg after sensitization significantly decreased the level of IL-4 (Fig. 4A, OVA+ vs. NAC 60 mg, p < 0.01), IL-5 (Fig. 4B, OVA+ vs. NAC 60 mg, p < 0.05) and GM-CSF (Fig. 4F, OVA+ vs. NAC 60 mg, p < 0.01) compared to OVA-sensitized animals.
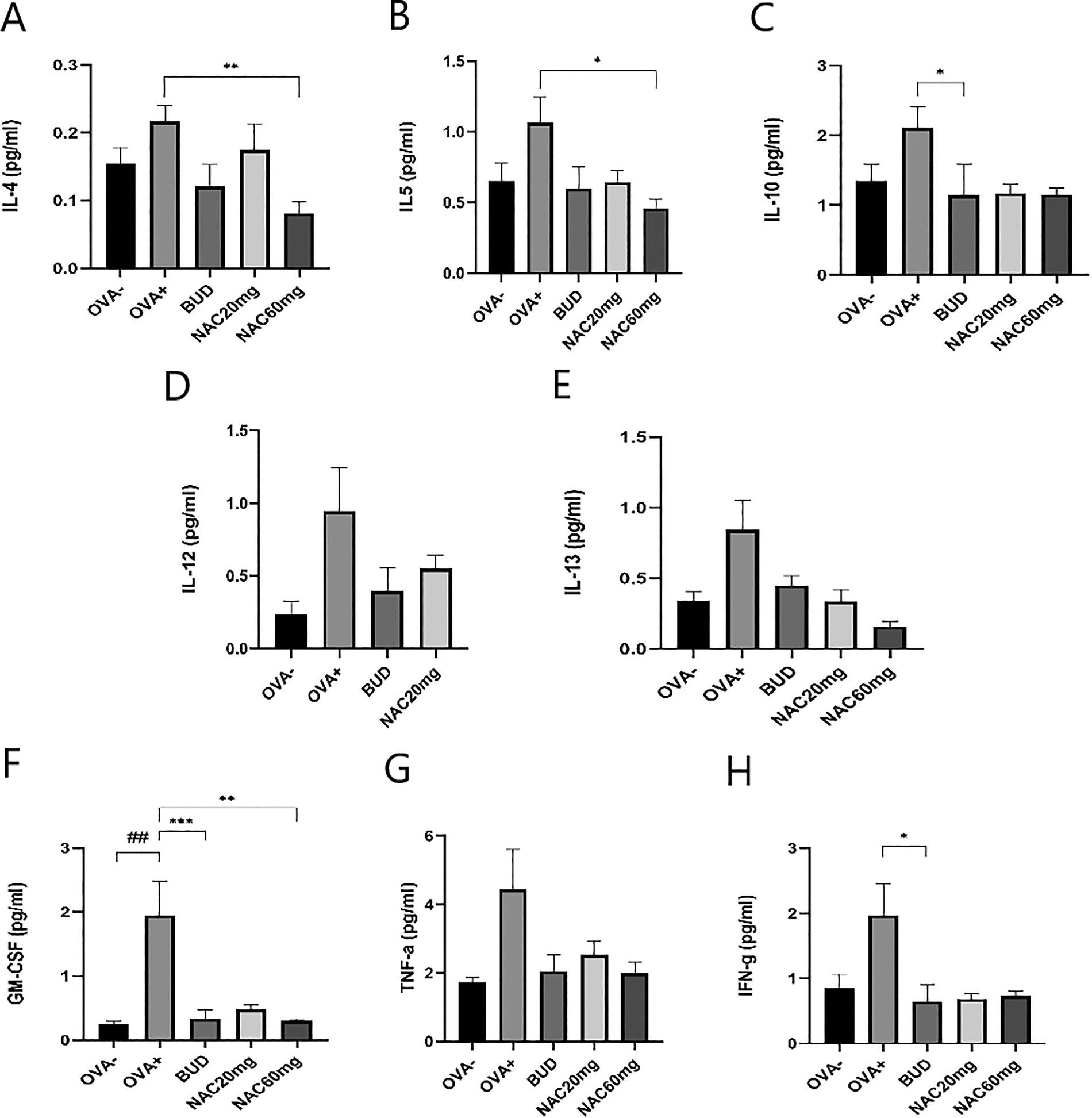
The concentration of IL-4, IL-5, IL-10, IL-12, IL-13, GM-CSF, IFN- γ and TNF-α in BAL fluid after 10 days of NAC administration and a reference anti-inflammatory drug. The control group is represented by healthy animals receiving saline for 21 days (OVA-), and the negative control group is represented by animals sensitised to ovalbumin for 21 days (OVA+). OVA-sensitised animals were administered NAC in a 20 mg/kg b.w. p.o. (NAC20mg), 60 mg/kg b.w. p.o. (NAC60mg) or budesonide (BUD) for 10 days. The data are expressed as the mean ± S.E.M; n = 8-9; ##P < 0.01 OVA+ vs. OVA-; ***P < 0.001,**P < 0.01, *P < 0.05 OVA+ vs. tested substance.
TGF-β1 concentration analysis in lung tissue homogenates
The TGF-β1 concentrations were evaluated in lung homogenate samples. The therapeutic effect of NAC was compared with budesonide. Ten days of peroral administration of NAC in both dosing schedules after sensitization significantly decreased the level of TGF-β1 (Fig. 5, OVA+ vs. NAC 20 mg, p < 0.001; OVA+ vs. NAC 60 mg, p < 0.001) compared with OVA-sensitized animals.
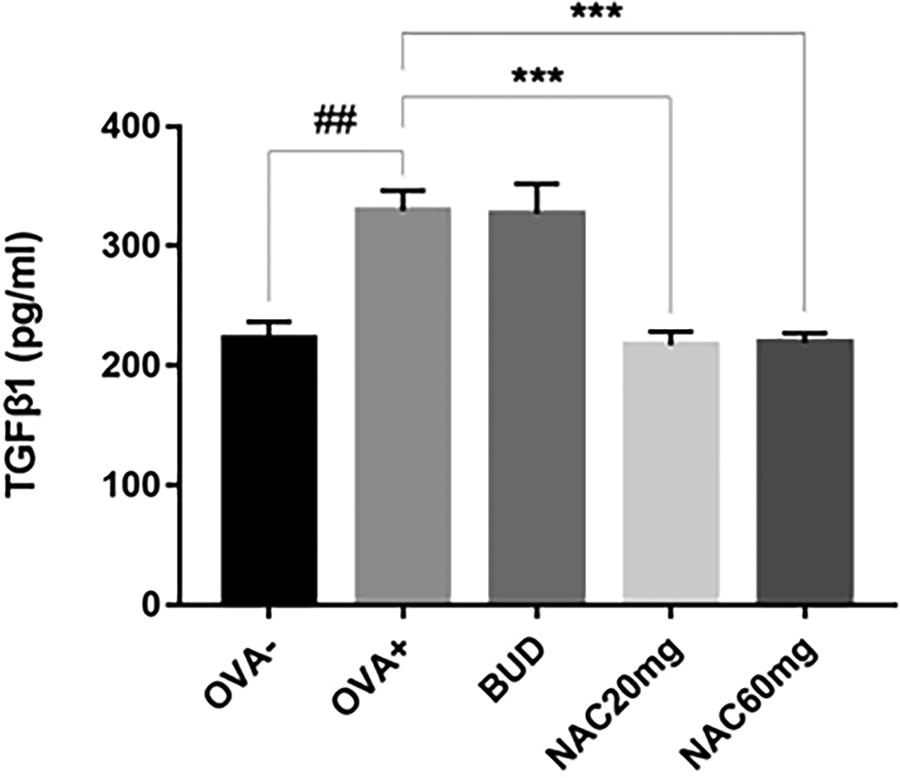
The concentration of TGF-β1 in lung homogenates after 10 days of NAC administration and a reference anti-inflammatory drug. The control group is represented by healthy animals receiving saline for 21 days (OVA-), and the negative control group is represented by animals sensitised to ovalbumin for 21 days (OVA+). OVA-sensitised animals were administered NAC in a 20 mg/kg b.w. p.o. (NAC20mg), 60 mg/kg b.w. p.o. (NAC60mg) or budesonide (BUD) for 10 days. The data are expressed as the mean ± S.E.M; n = 8-9; ##P < 0.01 OVA+ vs. OVA-; ***P < 0.001 OVA+ vs. tested substance.
CBF in vitro
The ability of effective ciliary movement was evaluated as changes in CBF. The allergen-induced suppression of epithelial cilia activity was reversed by salmeterol. Unlike the ciliostimulatory effect of salmeterol, the administration of NAC in the dosage of 60 mg/kg significantly decreased CBF compared to the OVA-sensitized group of animals (Fig. 6, OVA+ vs. NAC 60 mg, p < 0.001).
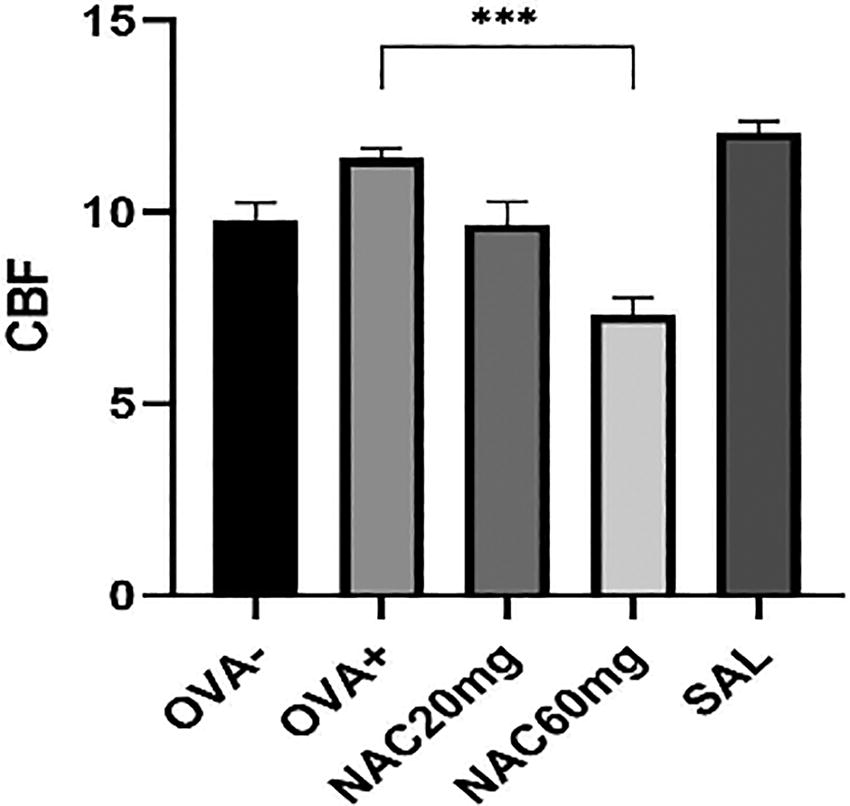
CBF after 10 days of NAC administration and a reference anti-asthmatic drug. The control group is represented by healthy animals receiving saline for 21 days (OVA-), and the negative control group is represented by animals sensitised to ovalbumin for 21 days (OVA+). OVA-sensitised animals were administered NAC in a 20 mg/kg b.w. p.o. (NAC20mg), 60 mg/kg b.w. p.o. (NAC60mg) or salmeterol (SAL) for 10 days. The changes in CBF were measured 24 hours after the last OVA challenge or substance administration. The data are expressed as the mean ± S.E.M; n = 7-10; ***P < 0.001 OVA+ vs. tested substance.
Discussion
NAC is a classical mucolytic agent that depolymerizes mucin glycoproteins while exhibiting antioxidant activity.2,18 The aim of our experimental study in a guinea pig model of allergic inflammation was to comprehensively evaluate the efficacy of 10 days of NAC administration on airway defense mechanisms, airway reactivity, the progression of allergic inflammation, and remodeling. The anti-inflammatory effect of NAC was demonstrated in an OVA-sensitized murine asthma model, where reduced levels of eosinophil peroxidase, the p65 subunit of nuclear factor kappa B (NF-κB), were measured in the lungs of NAC-treated mice, along with inhibition of goblet cell hyperplasia and pro-inflammatory cytokine production. 19 In our experimental model, administration of NAC at a higher dose significantly reduced the levels of pro-inflammatory Th2-cytokines IL-4, IL-5, and GM-CSF in BAL fluid, similar to the reference anti-inflammatory drug budesonide. GM-CSF promotes interleukin-1 beta (IL-1β) secretion by blocking antioxidant responses. 20 Allergic airway inflammation is directly related to oxidative stress. Activation of proinflammatory stimuli leads to the induction of reactive oxygen species (ROS) via activation of nicotinamide adenine dinucleotide phosphate oxidase, resulting in increased NF-κB activation. In this context, NAC is a GSH precursor, and it has been shown that elevated GSH levels repress NF-κB, a transcription factor involved in developing pulmonary inflammation. 21 ROS may result from inflammation and simultaneously initiate and augment the inflammatory response. Thus, it is possible that NAC treatment prevented oxidative stress from exacerbating airway inflammation. TGF-β1 is considered one of the major mediators involved in tissue remodeling in asthmatic lungs. 22 In our experiment, we found that NAC reduced TGF-β1 in both dosing schedules. The mentioned fact suggests that there is a rationale for its administration in allergic inflammation because, in addition to having an anti-inflammatory effect, it can probably inhibit remodeling. Coughing and mucociliary clearance are important airway defense mechanisms, and excessive coughing and mucus production are often associated with a poorer disease prognosis in asthma. 23 Lower mucociliary clearance has been demonstrated in patients with asthma compared to controls. 24 In our study, NAC treatment at the higher dose significantly reduced CBF and a number of cough efforts. The effect of NAC is multifactorial, and findings suggest that its beneficial effects on influencing the course of asthma as an inflammatory disease are not necessarily related to its demonstrated effect on changes in mucus properties. Therapeutic doses of orally administered NAC did not allow penetration of NAC into the airway lumen due to its low bioavailability. Thus, the effect of NAC was at the level of a mucoregulatory protective agent, not a mucolytic. We propose that the anti-inflammatory and potential anti-remodeling effects of NAC contribute to a reduction in both the quantity and viscosity of mucus. This alteration may lead to a slowing of CBF and a decrease in the frequency of coughs. However, it is crucial to examine the properties of sputum, such as its quantity and viscosity, in order to determine whether NAC has a beneficial impact on the progression of asthma or if its effects are primarily one-dimensional. If NAC reduces only the viscosity of sputum without affecting its amount, a decline in CBF could result in fewer coughs, while also increasing the risk of thin mucus accumulating in the peripheral regions of the lungs, potentially raising the likelihood of infections. The reduction in CBF following high oral doses of NAC demonstrated in this study is consistent with our previous study conducted under the same physiological conditions. 25 The main clinical feature of asthma is reversible airflow limitation. 26 Therefore, our study included the determination of the effect of NAC on airway hyperresponsiveness. The effect of NAC on suppressing airway hyperresponsiveness has been demonstrated in several experimental studies in rodents where it was administered orally, 27 inhaled, 28 and intraperitoneally. 29 We determined sRaw and TSM reactivity. These findings did not demonstrate statistical significance.
Conclusions
In this study, we demonstrated the anti-inflammatory and potential anti-remodeling capacity of NAC, as well as its potential to reduce airway defense mechanisms in conditions of allergic inflammation. For a comprehensive evaluation, it is crucial to examine other remodeling markers and properties of sputum.
Footnotes
Acknowledgment
The authors wish to thank Mrs. Katarína Jesenská for her technical support in this project.
Authors’ Contributions
L.S.: Conceptualization, writing—review and editing, results analysis, and formal analysis. J.M.: Methodology, investigation, formal analysis, writing—review and editing. E.G.: Investigation, methodology, statistical analysis, results analysis, writing—review and editing. M.D.: Methodology, formal analysis. M.J.: Performed experiments related to ciliary beat frequency, and analysis of results. M.Š.: Supervision, funding acquisition. S.F.: Conceptualization, supervision, writing—review and editing, project administration, funding acquisition.
Author Disclosure Statement
The authors declare no conflicts of interest in this article.
Funding Information
This work was supported by the Slovak Research and Development Agency (Grant No. APVV-19-0033 and APVV-23-0261) and the Ministry of Education, Science, Research and Sport of the Slovak Republic (Grant VEGA 1/0314/21 and VEGA 1/0042/24).
