Abstract
Background:
The RS01X is a digital dry powder inhaler (DPI) that records inhalation parameters such as technique or adherence. This offers patients and health care providers a way to have a feedback on inhalation treatments. This study used real-life data gained from 28 healthy volunteers for adherence assessment, technique evaluation, and in vitro testing.
Methods:
The study enrolled 28 healthy volunteers. Participants were shown how to use an inhaler and provided with empty capsules. The inhalers record several inhalation parameters such as peak inspiratory flow (PIF), volume, duration, and orientation. Half of the participants were selected to be in an “intervention” group with access to their inhalation data as well as feedback to improve their inhalation. The other half were a “control” group without access to their data nor any feedback. The data were then used for in vitro testing.
Results:
Overall, 28 participants were enrolled, and inhalation data were available for 13 interventions and 15 controls. Average adherence was 82.0% and 69.5% for intervention and control, respectively. The technique of inhalation was 65.58% good, 19.89% fair, and 14.53% poor for the intervention group and 36.73% good, 26.99% fair, and 36.28% poor for the control group. The variability of PIF was 9% for intervention and 30% for control. In vitro simulations showed the importance of proper angle orientation in inhalation, which was supported by in vivo data. The fine particle fraction of active pharmaceutical ingredients was similar to the inhalation profile of the intervention in comparison with a theoretical perfect inhalation.
Conclusion:
This study results showed clear improvement in inhalation technique and adherence for patients using digital DPI. In addition, in vitro testing provided concrete data illustrating the measurable advancements digitalization offers in enhancing patient adherence and inhalation technique.
Introduction
Patient errors in the utilization of inhalation devices are well known and widespread. A meta-analysis from 2017 by Chrystyn et al. showed a proportion of errors in dry powder inhaler (DPI) devices of 60.9% with about 28.4% of those being defined as critical for the patient treatment, thereby causing a health hazard. 1 The error rate has been similar and constant since the introduction of the first inhalers in the market in the 1960s. 2 Poor inhalation technique is associated with worse health condition3,4 and was estimated to have costed €105 million across three European countries (Spain, Sweden, and the United Kingdom) in 2015.
Digital DPIs represent a compelling innovation in health care, offering an effective pathway to enhance overall treatment. These devices, such as the recent RS01X from Berry Global® used in this study, allow real-time monitoring of inhalation parameters and enable health care providers to remotely monitor inhaler usage, provide tailored guidance, and enhance treatment adherence. The internal structure, air resistance, and medicinal delivery of RS01X are the same as a traditional inhaler. Although the impact of digitalization of patient technique is well studied,5,6 in many instances, these studies lack in vitro assessments to measure the enhancement in the bioavailable dose delivered to patients benefiting from digitalization, particularly when compared with traditional inhalers.
The present study examined the real-life performance of RS01X to assess patient adherence to prescribed dosing with digitalization, and controlled in vitro experiments were performed to measure inhaler performance parameters, including emitted dose and fine particle fraction, utilizing data from patients using both “traditional” and digital DPIs.
Materials and Methods
Participants
Twenty-eight participants were selected among Solvias employees. Participants with a significant knowledge of inhalation analyses techniques were excluded from the study.
The volunteers were randomly divided in two groups. The “intervention” group (n = 13) used RS01X with access to the mobile application Respiro developed by Amiko® for RS01X. The control group (n = 15) used a RS01X device also recording their inhalation profile but without access to the application and the feedback data. Both groups followed a prescription of 1 actuation per day. The participants were free to choose whether or not to also use the inhaler during the weekends in an effort to limit the invasiveness of the study. The participants used empty size three capsules of hydroxypropyl methylcellulose. After each inhalation, the volunteers were instructed to throw away the used capsules and to utilize a new one.
After selection, volunteers were given a short explanation on how to use the inhaler by a licensed pharmacist. Both groups were also provided with a diagram excerpt showing how to use a DPI inhaler. This excerpt came from a user’s notice for the Ultibro© Breezhaler©, a device with airflow resistance comparable with the RS01X. The RS01X has a resistance of 4 kPa at 100 L/min. This was done to simulate the experience of receiving real medication delivery from a pharmacy. The intervention group received an additional explanation on how to correct and improve their inhalation technique using the application. The limits of peak flow rate to define a “good,” “fair,” and “poor” inhalation in the application were established as described in Figure 1 (left) in L/min. The flows were chosen in accordance with the published literature about this subject.7–9 The time limits were selected to be considered poor with <1 second, fair between 1 and 2 seconds, and good with more than 2 seconds. As the exact time limits depend on several factors such as gender, body size, or athletic capacity, a straightforward system of time limits of 1 and 2 seconds was chosen for simplicity’s sake. The overall inhalation was only classified as good if both the length and the force were adequate (flow between 60 and 90 L/min for >2 seconds).
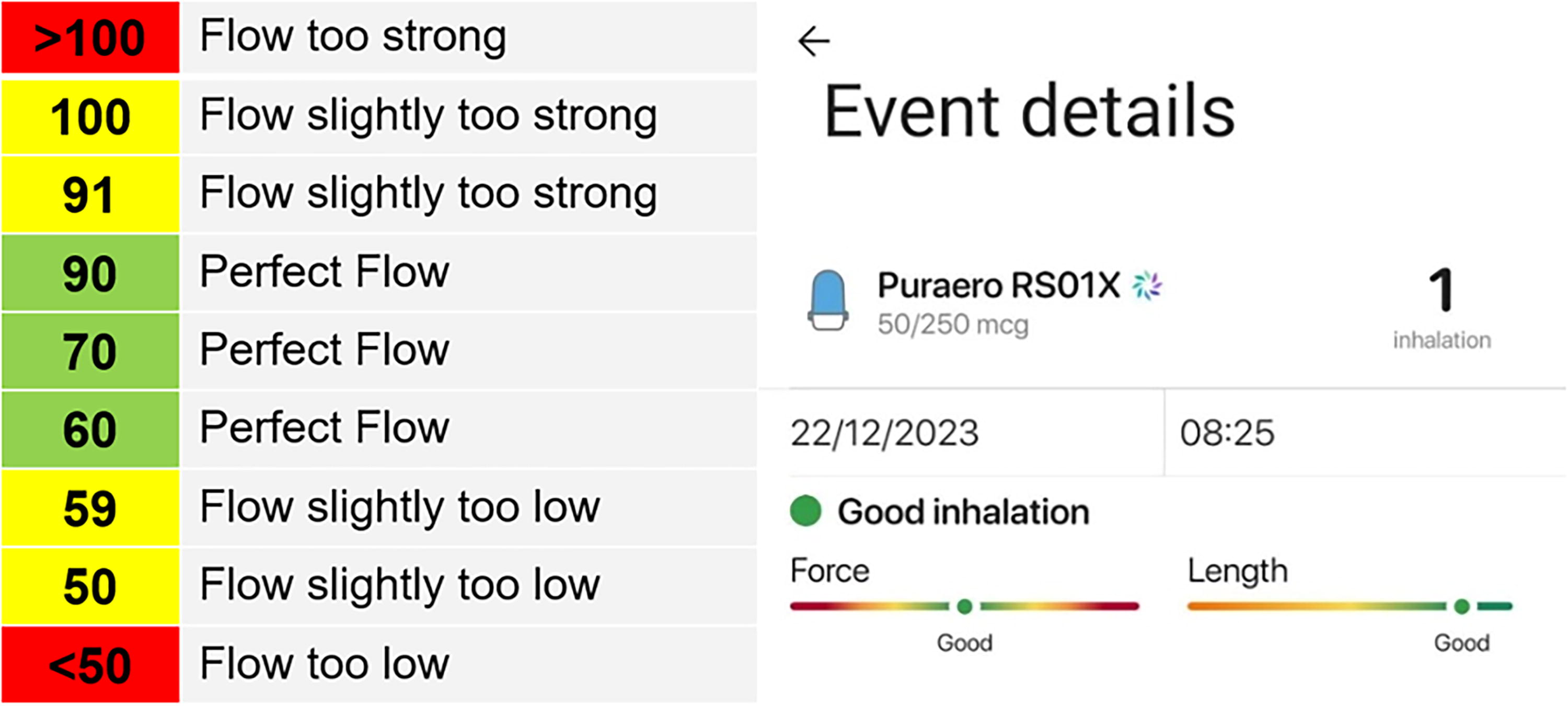
Acceptable inspiratory airflows in [L/min] (left) and view of the application showing an example of typical feedback (right).
This study involved therefore educational tests (aptitude evaluations) and behavioral observations. As the information collected was recorded in a manner that does not allow for the identification of participants, either directly or through linked identifiers, the study was determined to be exempt from IRB review under 45 CFR 46.104(d)(2).
Figure 1 (right) illustrates the feedback received by participants after each inhalation, depending on their technique.
Adherence
The adherence was calculated by excluding weekends and personal vacation, most notably the period between December 22, 2023, and January 1, 2024.
The adherence calculation involved summing up the weekly adherence and dividing by the number of scheduled days, capped at a maximum value of 1 to ensure that doses taken by users over the weekend were counted.
In vitro simulations
The in vitro simulations consisted of aerodynamic particle size distribution (APSD) measurements using a next-generation impactor (NGI) from Copley Scientific (Nottingham, UK). The delivered dose test was done by using a sintered glass funnel. The recovered dose is the amount that has left the device and capsule. The quantification was then done by reverse phase chromatography with UV detection using a Vanquish ultra-high-performance liquid chromatography from Thermo Fisher (Waltham, MA, USA).
In vitro simulations were done using three Ultibro Breezhaler’s capsules of Novartis (Basel, Switzerland). The capsules have two active pharmaceutical ingredients (API), glycopyrronium (50 µg), an anticholinergic, and indacaterol (110 µg), a beta-adrenergic agonist. This drug is used in the European Union for treating chronic obstructive pulmonary disease in adults. 10
Results
Adherence
Figure 2 shows the rolling adherence rate between the control and intervention group.
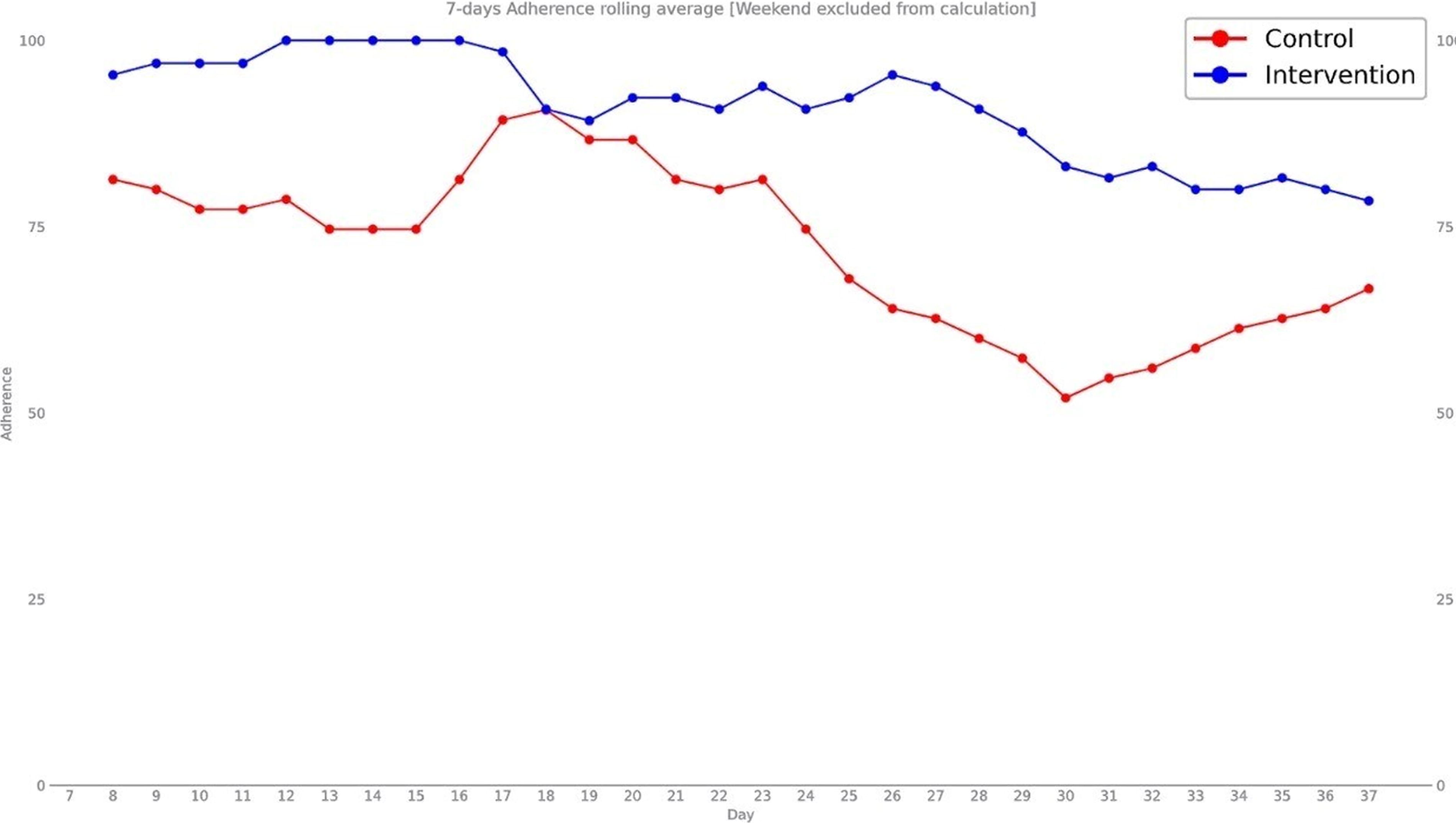
Seven days adherence rolling average starting from the first day of use of each participant until the 38th day.
Thirty-eight is the average median number of days of inhalations. At this number of inhalations, the average daily adherence for the intervention group was 91.1% and 72.4% for the control group. The total adherence rate from each participant’s first inhalation to their last was 82.0% for the intervention group and 69.5% for the control group.
Inhalation technique
Figure 3 shows the inhalation technique categorized in each of the two groups of participants. The intervention group, with access to a companion application, demonstrated a significantly higher rate of correctly executed inhalations compared with the control group. Specifically, 65.58% of actuations were classified as good, 19.89% fair, and 14.53% were deemed poor, as opposed to 36.73%, 26.99%, and 36.28%, respectively, for the control group. At the end of the study, the number of participants of the intervention group with a rate of good inhalation superior to 50% was 9 out of 13 (69%). In the control group, 5 participants out of 15 (33%) had a similar rate.
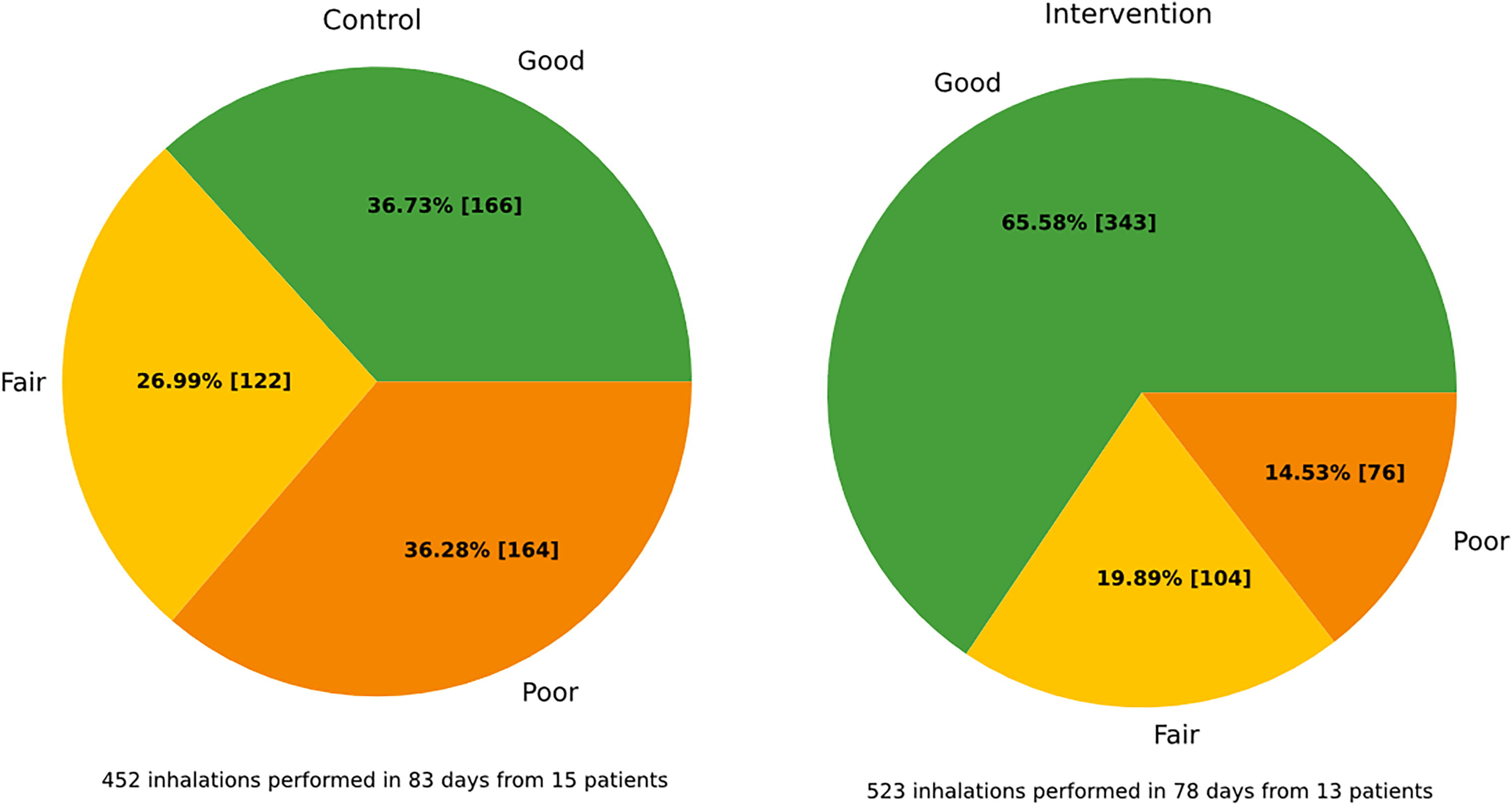
Inhaler technique comparison between control
The technique of the participants evolved over time. The first day, the rate of good inhalations was 30.77% for the intervention group and 46.67% for the control group. Starting from the fourth inhalation, the intervention group overtook the control group and had a better cumulative rate of good inhalation. After 20 inhalations, the cumulative rate of good inhalation was 57.98% and 32.65% for intervention and control, respectively.
Figure 4 shows the cumulative rate of the percentage of good inhalations over the first 20 inhalations. The trendlines have a R2 of 0.6905 for intervention and 0.9079 for control. Two participants out of 28 (7%) had <20 inhalations and were only counted up to their last inhalation. One from the intervention group (17 inhalations) and one from the control group (11 inhalations).
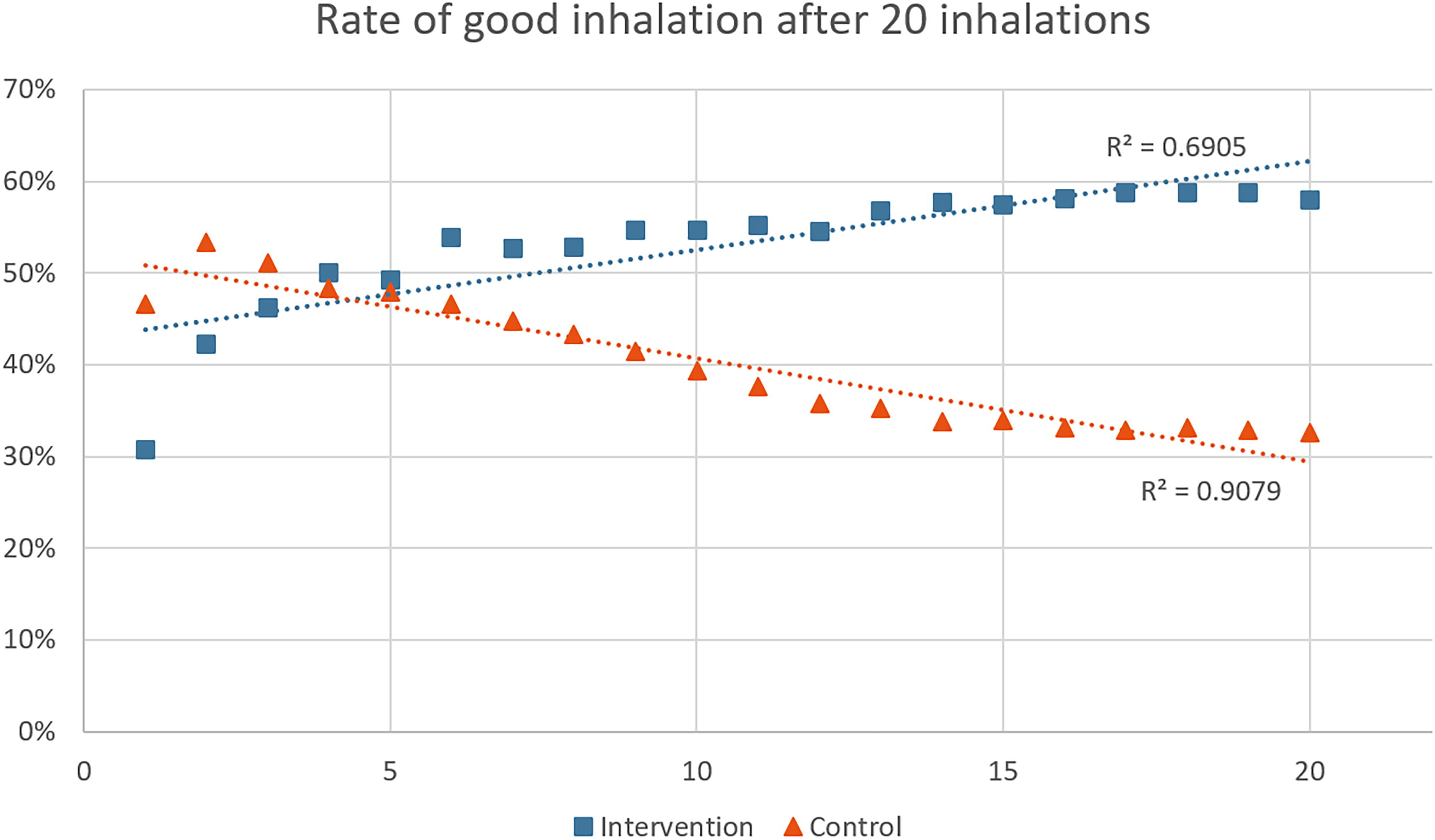
Cumulative rate of good inhalation quality after the 20 first inhalations for the two groups.
Inhalations parameters
All the inhalation parameters of every subject (975 total inhalations) were collected by the RS01X were averaged and separated by groups. The results are presented in Table 1.
Results of Averages of Intervention and Control Group
SD, standard deviation. RSD, relative standard deviation.
The average values of each participant’s peak inspiratory flow (PIF), volume inhaled, and inhalation duration were recorded by the RS01X, and the maximal and minimal values found in the study were selected as shown in Table 2.
Maximal (MAX) and Minimal (MIN) Average Values Obtained by Comparing Every Volunteer
The highest for MAX and the lowest for MIN are highlighted in bold. These values correspond to participants total averages. PIF, peak inspiratory flow.
In vitro simulations
Leveraging the user data captured by the device, the impact of poor device orientation on the delivered dose was simulated and measured in vitro. Three different real-case data were therefore selected as testing parameter, good flow with bad orientation (54.56 L/min, 2.5 seconds, and 12.98° of orientation), bad flow with bad orientation (24 L/min, 0.4 seconds and 0.67°), bad flow with good orientation (20.73 L/min, 0.55 seconds and 90.7°), and compared with a theoretical perfect flow and orientation (90 L/min, 1.3 seconds, and 90.0°). The results are shown in Figure 5. The study data showed that the average PIF between 0 and 22.5° was 62.88 L/min, 73.57 L/min between 22.6° and 45°, and 78.15 L/min between 45.1° and 90°.
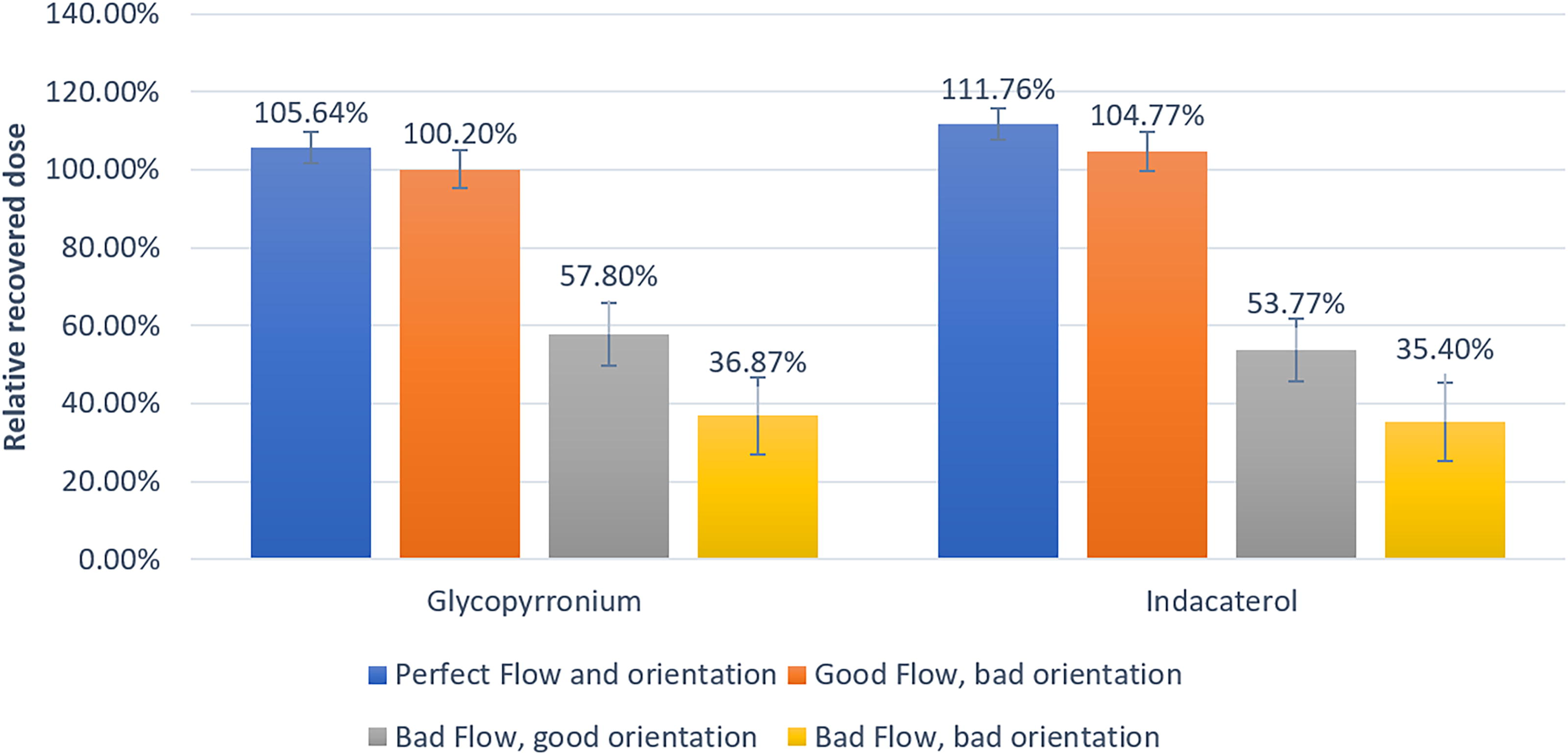
Percentage of recovered dose (n = 3 replicates) compared with the average delivery dose announced by the manufacturer for glycopyrronium (left) and indacaterol (right). The theoretical average delivered dose is 43 µg and 85 µg, respectively.
In order to evaluate the impact of a noncorrected poor inhalation technique on the performance of the device, several profiles were simulated.
A worst-case scenario (22.5 [L/min] for 0.5 seconds) was created by taking the first seven shortest inhalations with a duration of at least 0.45 seconds. The average profile of the intervention group (58.1 [L/min] for 3.2 seconds) was also simulated as well as the theoretical perfect inhalation profile (90 [L/min] during 2.7 seconds). The comparison involved assessing the emitted dose, fine particle fraction, and fine particle diameter. In these worst-case simulations, the majority of API stayed in either the device or the capsules (about 57% for each component). Figures 6 and 7 show the repartition of API in the NGI for glycopyrronium and indacaterol.
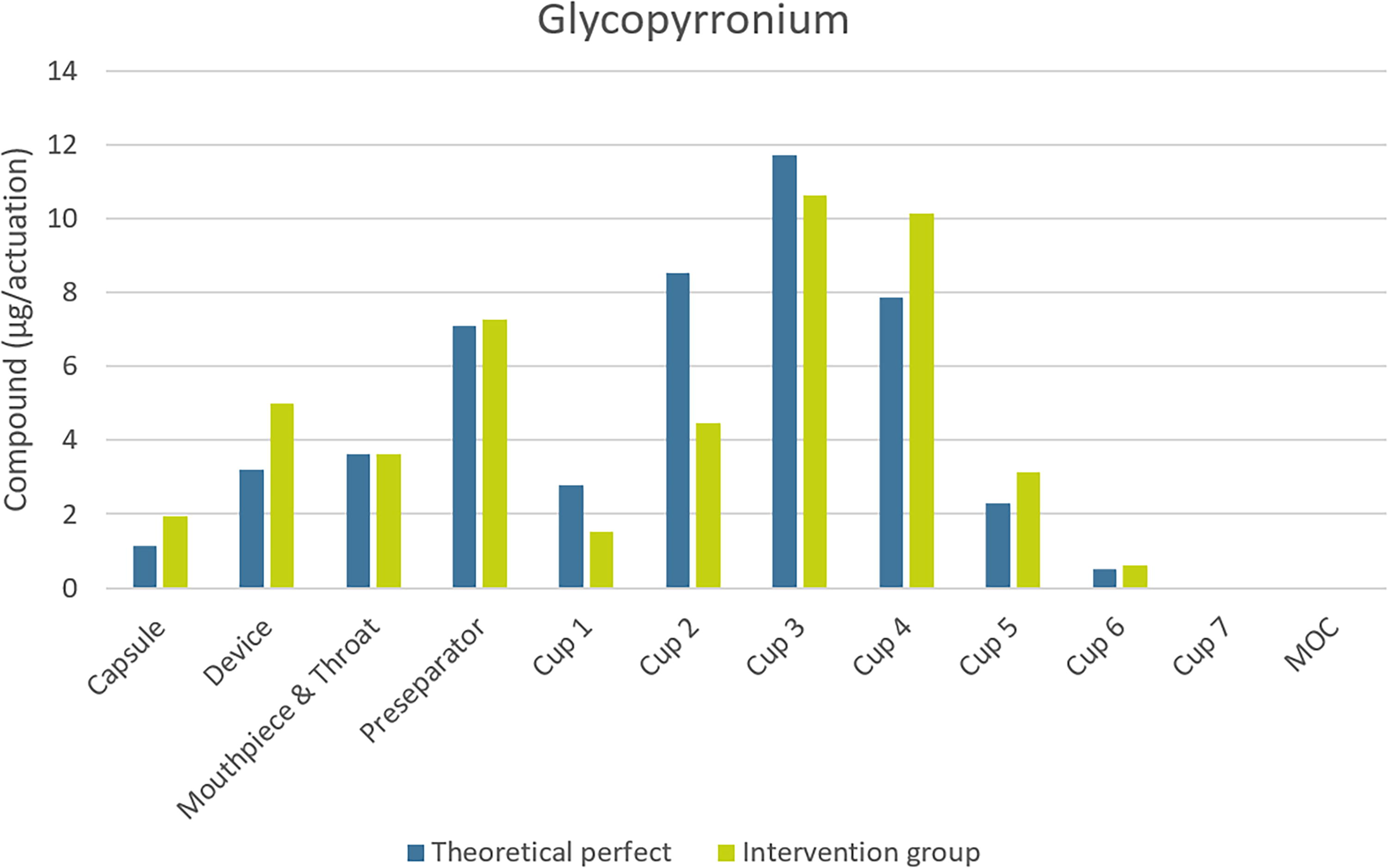
Repartition of glycopyrronium in milligrams in the different parts of the NGI with three different inhalation profile. NGI, next-generation impactor.
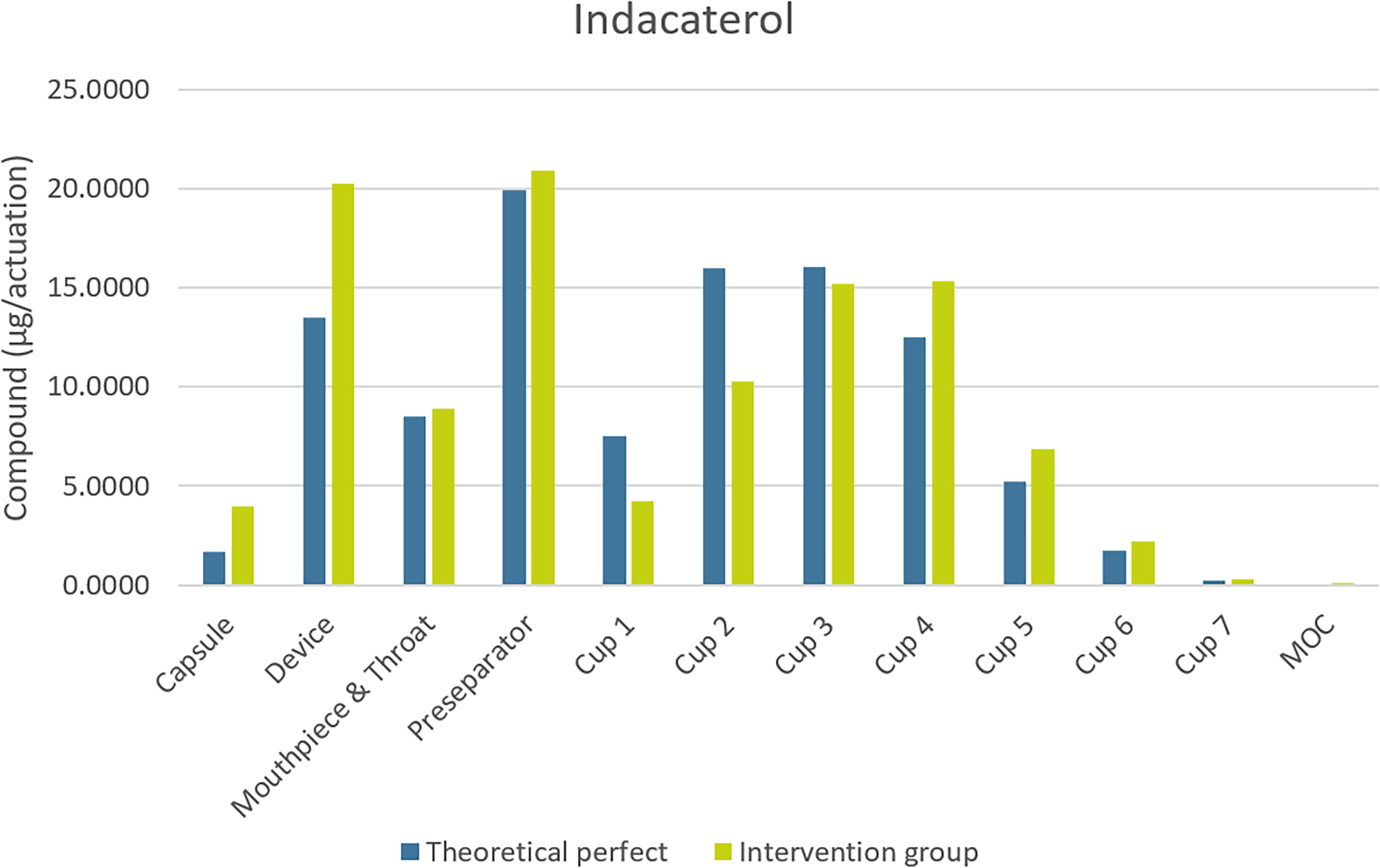
Repartition of indacaterol in milligrams in the different parts of the NGI with three different inhalation profiles.
Table 3 presents a comparative analysis of fine particle statistics between a theoretically perfect flow, the average inhalation of the intervention group, and the worst-case scenario. To illustrate the impact that the absence of feedback might induce, a comparison of two participants of the intervention and control group was done. The two participants had a similar first inhalation (48.4 [l/min], 2.42 [L], 3.59 seconds for intervention and 42 [l/min], 3.14 [L], 5.76 seconds for control) and similar physical characteristics (young women with a comparable build) were compared. After 2 months, the averages were tested again. The inhalation parameters of the control participant were 35.82 (L/min), 1.95 (L), 4.11 seconds, while the intervention participant was 70.9 (L/min), 2.32 (L), 2.48 seconds. The results are shown in Table 4.
Fine Particle Comparison Between a Theoretical Perfect Inhalation, the Intervention Group Average, and the Worst Cases Found
The “n” corresponds to the number of replicates.
Variations in Delivered Dose and Fine Particle Mass Between the Control and Intervention Cases
In parenthesis is the SD. The “n” corresponds to the number of replicates.
Discussion
Adherence
It has been established that a good adherence is associated with better health outcomes. The major 2004 study TORCH evaluated the adherence of >6000 patients for 3 years. Adherence has been significantly associated with reduced mortality and hospitalizations. 11 Patient with poorer adherence are more dyspneic and have a lower quality of life. 12 Long-term adherence is very important but typically difficult to achieve in real-life conditions.13–15
In this study, the discrepancy between the two group’s adherence rates highlights the tangible and positive influence of the digital intervention on inhalation adherence.
The main reminder was a daily notification on the participant’s phone, as well as occasional “insights” giving participants personalized inhalations advice. They have produced a largely positive effect on long-term adherence. As time passed, the control group tended to perform worse as initial curiosity and enthusiasm settled in.
It is often seen that volunteers in study trials tend to be initially more enthusiastic about their treatment than real long-time patients so the discrepancy in adherence between digital and traditional inhaler users may potentially be greater than what has been found in this study. In this study, the final difference between the control and intervention was 12.5%. This value is similar to the one found in other studies, for example, 22% in a 12-week period by Charles et al. 16
Technique
Inhaler technique is usually vastly overrated by users. A study found that ∼95% of DPI users feels that they are able to use the inhaler well with a further 90% believing that the inhaler is easy to use. 17 However, the number of user using incorrectly their inhaler is sometimes seen as >60%. 1
In this study, inhaler technique has been vastly improved by digitalization. For the majority of participants, the initial PIF was inadequate. Not only could the intervention group correct their initial inhalation strength, the feedback also prevented mistakes from taking hold over time, resulting in an unambiguous difference between the two groups. The control group did worse over the course of the study. The initial results might have been better as the initial oral explanations were still fresh but as time went on, the results became slightly worse.
Using the data collected, it can be estimated that about 65%–70% of the intervention users have a good inhalation technique while only about 35% of the control group does.
Although every participant received the same explanations and a diagram with written instructions, both too strong and too weak inhalations were identified. Inhalations with weak flow will result in low bioavailability as the API will not reach the lung’s alveoli. 18 Exceedingly strong inhalations, however, will also result in a loss of API as the drug will get stuck in the throat. 8 As a health practitioner is unable to know beforehand the inhalation strength of their patient, it is impossible to give a perfect advice on the usage of an inhaler, demonstrating the usefulness of a digital DPI.
Individual data
Individual data have shown a noteworthy variation in PIF and inspiration duration in the control group. While the average values are comparable, the dispersion is much greater in the control group, especially for PIF.
The extremes of each measure were identified in Table 2. Most extreme values were found in the control group. Meanwhile, the flow averages of the intervention group were all in between 60 and 90 (L/min), which was considered the limit for a good inhalation. Only the upper extreme for inhaled volume was in a volunteer from the intervention group but a high inspiratory volume is desirable for drug inhalation. Nevertheless, this factor is dependent on the user themselves as the inspiratory capacity depends on uncontrollable factors such as size and gender. 19
The orientation was quite similar in both groups. The application gave no feedback on the orientation, so it was not considered in the in vivo study. However, the impact of inhaler orientation was still tested in vitro.
In vitro simulations
Impact of device orientation
Although the quality of the flow is shown to be the most important parameter, a proper orientation can contribute significantly to delivering the correct medication dosage to the patient, even in the presence of a less than ideal flow. Notably, achieving an accurate flow is considerably more challenging with an improper angle. Positioning the device in a very vertical orientation led to an average PIF of 62.88 L/min, halfway to 73.57 L/min, and 78.15 in a more horizontal position.
APSD test and fine particles analysis
The size of particles is of major importance in the distribution and efficacy of inhaled drugs. It is generally admitted that for efficient lung distribution, particles size should be lower than 5 µm. For smaller particles, it is slightly less clear, but it is conventionally believed that a size smaller than 1 µm is less effective because they tend to get easily exhaled, making them unable to have a pharmaceutical impact. 20 This is the reason that APSD tests are so important. Indeed, even if a capsule is emptied during an inhalation, a bad size distribution can still lead to ineffective drug delivery.
The API repartition in the different part of the NGI was similar in the theoretical perfect inhalation and in the average of intervention. The difference in the cup repartition can be explained by the difference in the flow rate between the two simulations. Indeed, the number of fine particles was similar as shown in Table 4.
The worst cases were selected from actual inhalations performed by some of the participants. The simulation of these scenarios showed that the majority of API adhered to the preseparator section of the NGI, analogous to the bronchial tubes in vivo. The number of fine particles generated is almost negligeable, demonstrating how certain inhalations are so ineffective that, in terms of fine particle delivery, they are comparable with no inhalation at all. In contrast, the intervention group’s API, on average, exhibited a more favorable distribution, with a majority reaching the impactor stages representing the alveoli in the lungs, thereby facilitating bioavailability.
Although the overall averages of the control group were very similar to those of the intervention group (Table 1), the key point of interest lies in the variability observed within the control group. This variability includes both strong and low flow rates (Table 2), making a simple comparison of averages unrepresentative of real-life scenarios and was therefore not tested.
The results from both the theoretically perfect flow and the average of the intervention group align closely, indicating a consistently high level of inhalation proficiency within the intervention group.
Case comparison
By using two similar profiles, it is possible to illustrate the potential impact of digitalization in correcting a user manipulation. It is particularly interesting because in both cases, the capsules were emptied after manipulation, shown here by the very similar delivered dose. This means that without a digital inhaler, it would not have been possible to notice a mistake in one of the two users. However, by using the inhalation parameters recorded by the digital inhaler, it is possible to see that there is a difference in the fine particles received by each user. The control participant received 22.55% less glycopyrronium and 27.13% less indacaterol than their intervention counterpart. This specific case illustrates an example of the usefulness of digitalization.
Limitations
This study had several limitations. First, the sample of participants was small and comprised of Solvias employees who may not be representative of the typical patient population.
Furthermore, it is often seen that volunteers in study trials tend to be more enthusiastic about their treatment than real patients so the discrepancy in adherence between digital and traditional inhaler users may be greater than what has been found in this study. This was also manifested in the study by an initial enthusiasm that left adherence rate between the two groups very similar during the first days.
Finally, the capsules were empty. In real conditions, the patient has a small feedback available by checking whether the capsule has been emptied after an inhalation. However, not all patients might be doing this supplementary task. In case of children or elderly persons, they might not see very clearly if the capsule has been emptied. In the case of the Ultibro Breezhaler, for example, the capsules are colored and can be difficult to see through. Lastly, an empty capsule does not guarantee a proper inhalation as the API might be stuck in low bioavailability areas such as the throat, bronchial tubes, or even the device itself.
Conclusion
The present work has shown that participants using a digitalized DPI with a companion app significantly improved their inhalation technique and adherence compared with users using an inhaler without digital feedback.
In vitro simulations have shown the importance of device orientation with regard to the whole inhalation, and the results were supported by in vivo findings.
Furthermore, APSD simulations established the intervention group performances on par with theoretical perfect inhalation.
Likewise, the study underscores the negative impact of poor inhalation technique in the bioavailability of the API, emphasizing the critical role of patient technique and digitalization in optimizing treatment outcomes.
Footnotes
Acknowledgments
The authors thank Lonza (Basel, Switzerland) for generously supplying empty capsules for this study.
Authors’ Contributions
D.N.G.: Formal analysis, investigation, data curation, writing—original draft, and visualization. M.G.: Software, formal analysis, validation, resources, and writing—review and editing. L.P.: Software, formal analysis, and resources. C.C.: Software and resources. V.S.: Resources and writing—review and editing. M.F.: Conceptualization, validation, resources, and writing—review and editing. M.C.: Resources and writing—review and editing. Y.B.: Conceptualization, methodology, validation, data curation, writing—review and editing, supervision, and project administration.
Author Disclosure Statement
V.S., M.F., and M.C. are employees of Berry Global, the manufacturer and seller of the RS01X device. M.G., L.P., and C.C. are employed by Amiko, which markets the companion app utilized in this study. D.N.G. and Y.B. have no conflict of interest to declare.
Funding Information
RS01X devices were provided by Berry Global. Solvias employees participated as volunteers in this study.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
