Abstract
The Sugen 5416/hypoxia (Su/Hx) rat model of pulmonary arterial hypertension (PAH) demonstrates most of the distinguishing features of PAH in humans, including increased wall thickness and obstruction of the small pulmonary arteries along with plexiform lesion formation. Recently, significant advancement has been made describing the epidemiology, genomics, biochemistry, physiology, and pharmacology in Su/Hx challenge in rats. For example, there are differences in the overall reactivity to Su/Hx challenge in different rat strains and only female rats respond to estrogen treatments. These conditions are also encountered in human subjects with PAH. Also, there is a good translation in both the biochemical and metabolic pathways in the pulmonary vasculature and right heart between Su/Hx rats and humans, particularly during the transition from the adaptive to the nonadaptive phase of right heart failure. Noninvasive techniques such as echocardiography and magnetic resonance imaging have recently been used to evaluate the progression of the pulmonary vascular and cardiac hemodynamics, which are important parameters to monitor the efficacy of drug treatment over time. From a pharmacological perspective, most of the compounds approved clinically for the treatment of PAH are efficacious in Su/Hx rats. Several compounds that show efficacy in Su/Hx rats have advanced into phase II/phase III studies in humans with positive results. Results from these drug trials, if successful, will provide additional treatment options for patients with PAH and will also further validate the excellent translation that currently exists between Su/Hx rats and the human PAH condition.
Introduction
Pulmonary hypertension (PH) is currently classified into five groups (groups 1 to 5) with distinct pathophysiological characteristics, 1 but the present review focuses exclusively on pulmonary arterial hypertension (PAH), also known as group 1 PH where the arteries in the lungs become narrowed and thickened. PAH is a specific clinical group of severe and rare conditions, including different forms with similar pathological, hemodynamic, and therapeutic characteristics. 2 PAH can be considered idiopathic, heritable, associated with other diseases such as congenital heart disease, connective tissue disease, portal hypertension, or associated with exposure to certain toxins, drugs, or human immunodeficiency virus infection. 3
While a variety of rodent models of PH have been developed to replicate the important features of PAH in humans, it is generally accepted that the Sugen 5416/hypoxia (Su/Hx) challenge in rats incorporates most of the important distinguishing features of this disease.4–6 It is one of the few models that demonstrates the presence of plexiform lesions that are not reported with other commonly used PAH models in rats such as the monocrotaline challenge and chronic Hx. Furthermore, depending upon the strain of rat used for the Su/Hx challenge, it achieves the magnitude of the elevated mean pulmonary arterial blood pressure (mPAP) observed in human subjects with severe PAH.5,7,8 In mice, while Su/Hx induces many of the pathophysiological features of PAH, it fails to produce extensive angio-obliteration, neointimal lesions, and plexiform lesions in pulmonary blood vessels, and there is an overall resolution of the PAH pathology over 10 weeks.7,9
There have been several review articles describing the important features of a Su/Hx challenge model in rats,5,7 but a current review of this field is warranted as there have been significant advancements in epidemiology, hemodynamics, pathology, and pharmacology in Su/Hx rats over the past few years. In this review, we describe the current status of the pathogenesis of human PAH and its translation in Su/Hx-challenged rats, and we present the current and novel therapies for PAH that have been tested in the Su/Hx rat model that target several pathological mechanisms, with a specific focus on the ones that have been successfully advanced into clinical trials.
PAH Definition
PAH is defined as a resting mPAP above 20 mmHg, a pulmonary capillary wedge pressure (PCWP) below 15 mmHg, and a pulmonary vascular resistance (PVR) above 3 Wood units in the absence of more prevalent forms of PH such as left heart disease, chronic lung disease, and venous thromboembolism.10–12 There is significant diversity in the geographical and racial distribution of PAH with the highest percentage in older Caucasians,12–14 a higher prevalence of PAH in females,12–14 and a genetic component to the disease with ∼10% of patients having heritable PAH.12,13 Hemodynamic evaluation of PAH involves the use of echocardiography to detect whether there is an increase in right ventricular systolic pressure (RVSP), although additional parameters such as pulmonary artery diameter (PAD), pulmonary artery acceleration time (PAAT), pulmonary artery maximum velocity (Vmax), pulmonary artery velocity time integral (VTI), right ventricular anterior wall thickness (RVAWT), and tricuspid annular plane systolic excursion (TAPSE) can also be measured. 10 Right heart catheterization is used to measure mPAP, PCWP, PVR, and right heart function to validate the presence of PAH.11,15 More sophisticated assessments can also be performed such as measurement of pulmonary vascular impedance and right ventriculo-arterial coupling, and imaging of the right ventricle (RV) can be performed to help further diagnose the presence and severity of the disease.15–17
The pathology of PAH includes constriction of the pulmonary arteries due to an imbalance of constrictor and dilator mediators, hyperplasia and hypertrophy of vascular smooth muscle cells (VSMCs) in the adventitial layer of the pulmonary blood vessels, and apoptosis and proliferation of the vascular endothelium resulting in occlusion of the small pulmonary arteries with plexiform lesion formation.10,12 The presence of serine elastases and matrix metalloproteinases in the pulmonary blood vessels, along with hyperproliferation of fibroblasts and excessive collagen deposition in the blood vessel wall, results in a loss of pulmonary vascular elasticity. There is also an inflammatory component to PAH pathology with the involvement of many signaling pathways that resemble conditions observed in cancer and cellular senescence, conditions that occur in many degenerative diseases of the elderly.18,19 Collectively, these pathological changes in the pulmonary arteries contribute to increased PVR and impose strain on the right heart that if untreated will result in right heart failure (RHF).15,20
Treatments for PAH
The pathobiology of PAH is complex and involves many mechanisms such as pulmonary arterial endothelial cell (PAEC) dysfunction, pulmonary arterial smooth muscle cell (PASMC) proliferation, vascular inflammation, metabolic abnormality, excessive growth factor stimulation, ion channel defects, and germ line mutation. 21 Endothelial dysfunction is one of the hallmarks of PAH, which plays a major role in regulating the vascular tone, vascular remodeling, and inflammation through the release of vasoactive mediators. 3 Indeed, endothelial dysfunction causes an increase in vascular tone by either reducing the production of the vasodilators, prostacyclin or nitric oxide (NO), or by increasing the production of vasoconstrictor endothelin 1 (ET-1), which stimulates PASMC proliferation in pulmonary arteries and inflammation. Over the past 20 years, 13 drugs have been developed and approved for the treatment of PAH by the Food and Drug Administration (FDA) in the United States and 14 drugs have been approved worldwide, delivered via oral, inhaled, subcutaneous (SC) or intravenous (IV) routes, targeting the three main mechanistic pathways of PAH, ET, prostacyclin, and NO, to regulate the vascular tone. 22 Targeting pulmonary vascular remodeling or improving RV function is challenging, but new mechanistic pathways have emerged (Fig. 1).
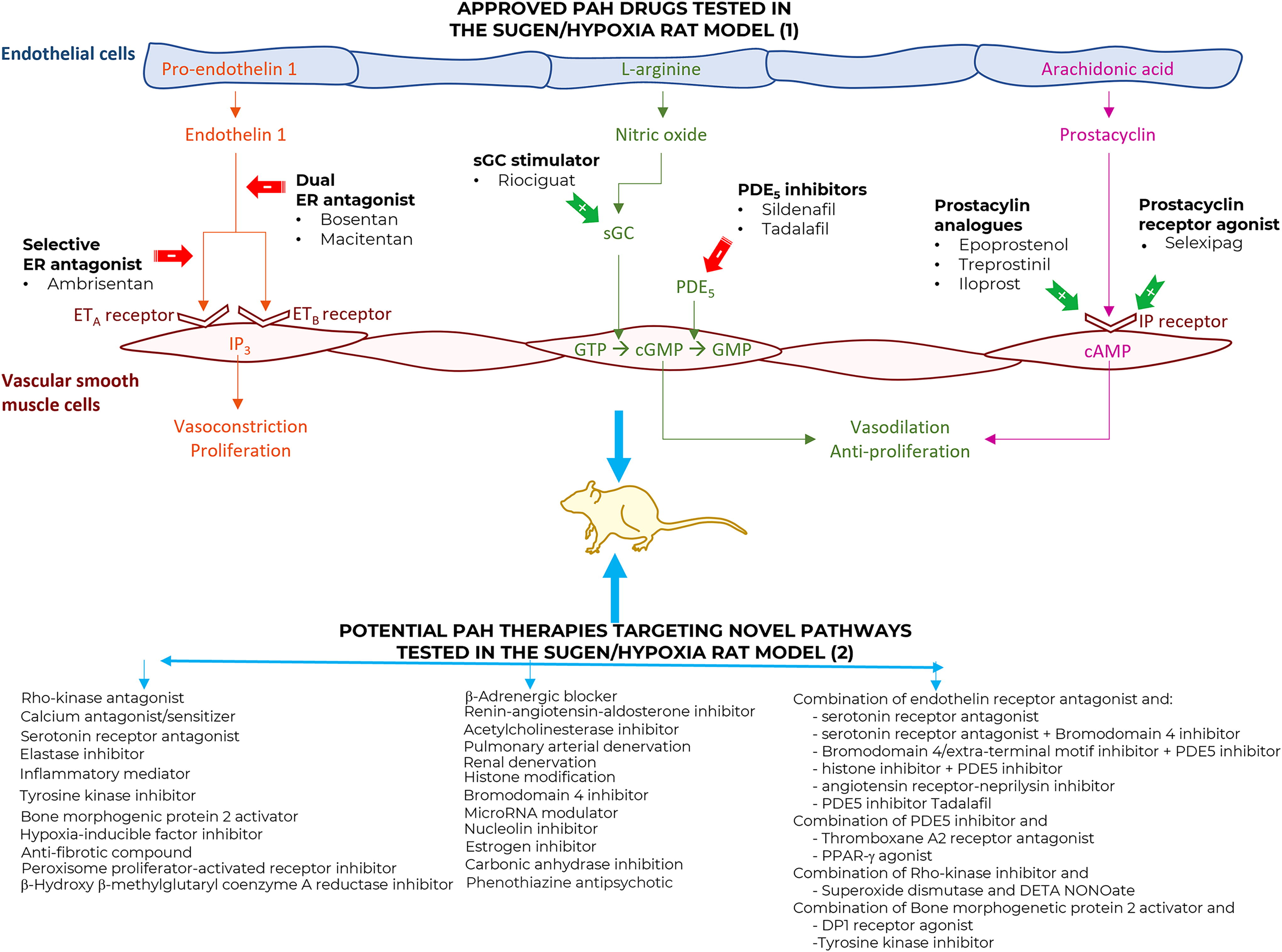
Hallmarks of pulmonary arterial hypertension (PAH) and current/emerging therapies. The initial key pathological manifestations of PAH are endothelial dysfunction and excessive proliferation of the pulmonary vascular smooth muscle and endothelial cells that lead to vasoconstriction, vascular inflammation, intimal and adventitial fibrosis, vascular remodeling with hypertrophy/hyperplasia, neointima formation, and plexiform lesions. Also, imbalances between the production of vasodilator (nitric oxide [NO] and prostacyclin) and vasoconstrictor (endothelin-1 and angiotensin) mediators contribute to the emergence of PAH. Current therapies (1) act through the three main pathways regulating the vascular tone: prostacyclin analogs (epoprostenol, treprostinil, and iloprost) or prostacyclin receptor agonist (selexipag), NO pathway (PDE5 inhibitors sildenafil and tadalafil) or guanylate cyclase stimulator (riociguat), and endothelin receptor antagonists (bosentan, ambrisentan, and macitentan). However, because of the limited effects of current therapies and with the multiple mechanisms underlying the PAH pathogenesis, new drugs targeting various pathological pathways (2) have emerged and have been tested in the Sugen/hypoxia rat model.
Current therapies
Currently, FDA-approved PAH therapies target one of the three different mechanistic pathways and are classified in five distinct classes: the ET-receptor antagonists (ERAs) bosentan, macitentan, and ambrisentan, the phosphodiesterase type 5 (PDE5) inhibitors sildenafil and tadalafil, the soluble guanylate cyclase (sGC) stimulator riociguat, the prostacyclin analogs epoprostenol, treprostinil, and iloprost, and the prostacyclin receptor (IP) agonist selexipag (Fig. 1). These therapies act predominantly on the vasoconstrictive phenotype of PAH and are associated with the regulation of the endothelial factors that affect vasoconstriction, vasodilatation, proliferation, and mitogenesis. 23 The drugs in these classes do not directly target the vascular remodeling that obstructs the pulmonary vasculature, they regulate the pulmonary vasomotor tone and indirectly inhibit cell proliferation.
Emerging therapies
Although the current approved PAH therapies that target the three principal mechanistic pathways regulating the pulmonary vascular tone (prostacyclin, ET, and NO) have led to improvements in the quality of life, exercise capacity, and life expectancy of patients with PAH, there is still no cure for this disease and no new drugs have been approved in the last few years. New approaches targeting novel molecular signaling and potential therapies are emerging. Many such therapies have been tested in the Su/Hx model (Fig. 1).
Su/Hx Rat Model
Characteristics
No single animal model can fully replicate all of the characteristics of human PAH, such as the elevation in PAP and RVSP, vascular remodeling with accumulation of PASMCs, PAECs, fibroblasts, and myofibroblasts in the pulmonary arterial wall leading to the thickening of blood vessels, loss of pulmonary vascular remodeling, and perivascular inflammation. 24 With that said, various animal models have been designed to recapitulate the pathogenesis of this human disease 25 with two distinct phases, a first phase characteristic of medial and adventitial thickening of the pulmonary artery, and a second phase defined by plexogenic arteriopathy resulting in obliteration and occlusion of the small and medium pulmonary vessels. 7 The first phase is well reproduced by the two classical PAH rat models, the Hx- and the monocrotaline-induced PH, but the second phase is absent. 7 However, the combination of vascular endothelial growth factor (VEGF) receptor 2 inhibition by Su and chronic Hx results in increased remodeling and development of plexiform lesions in the rat that simulates the second phase.26,27 The Su/Hx rat model, first described more than 20 years ago, and now with hundreds of citations, provides information about pulmonary intravascular inflammation, lung vascular immunity, and RHF. 28 Historical explanation and elucidation of the model can be found in the article of Voelkel and Bogaard. 28
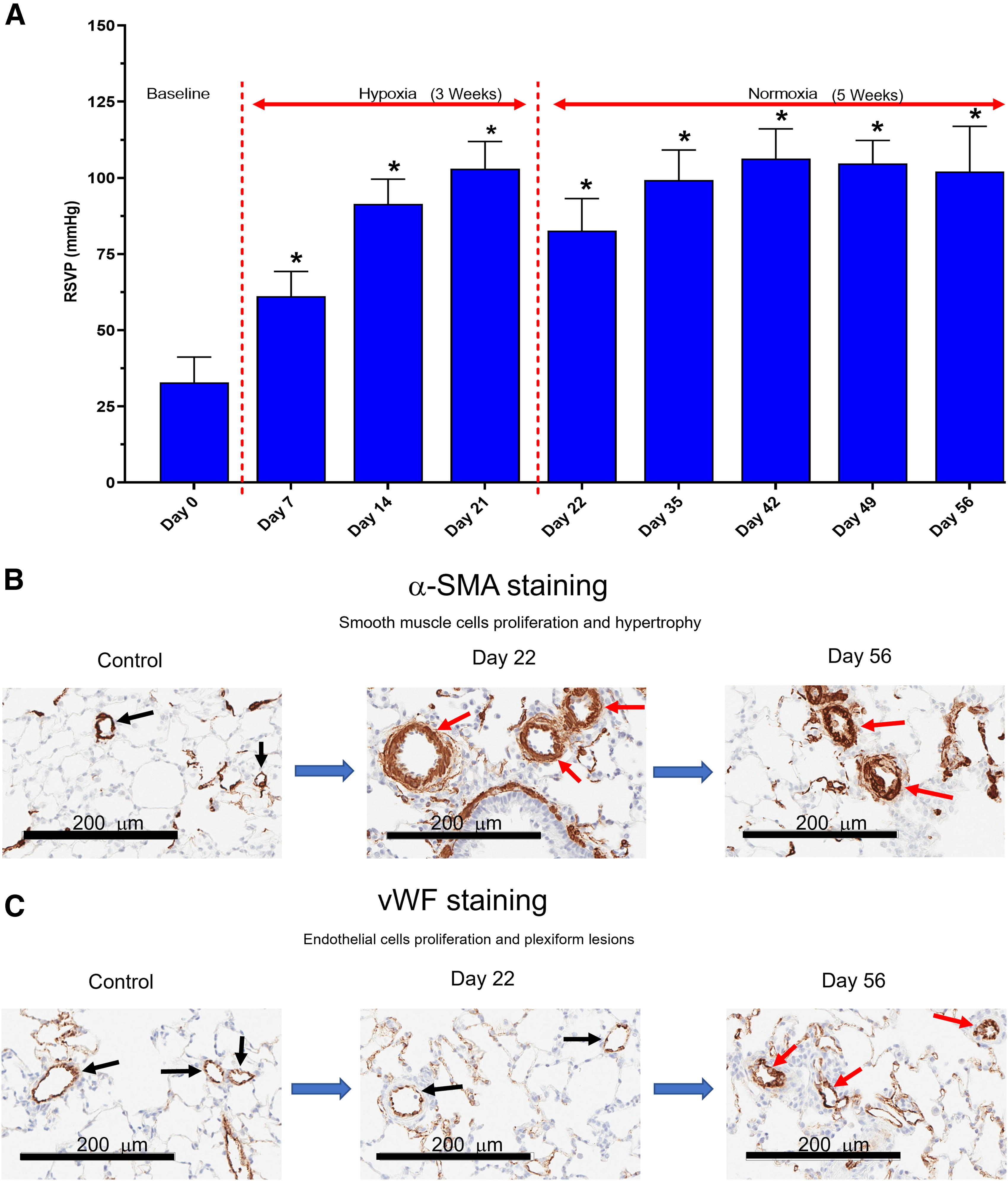
The most commonly used study design is the one-time injection of 20 mg/kg Su (SC) in 6- to 8-week-old Sprague–Dawley (S-D) rats, followed by 3 weeks Hx (10% oxygen [O2]), and subsequently a 1- to 10-week normoxia (Nx) followed by determination of pulmonary and systemic hemodynamic parameters, morphometric analyses of pulmonary vascular remodeling, such as wall thickness, muscularization, and obliteration of pulmonary arteries, and RV hypertrophy. 29 A single injection of Su and exposure to chronic Hx in rats caused pulmonary arterial medial hypertrophy, pulmonary arterial wall thickening, and sustained pulmonary vasoconstriction resulting in elevated pulmonary pressures and appearance of plexiform lesions. Also the VEGF inhibition by Su generates an initial apoptosis of PAECs followed by proliferation of PASMCs and PAECs, and apoptosis-resistant PAECs, and when combined with chronic Hx leads in the formation of plexiform lesions. 30 The double hit Su/Hx impairs pulmonary hemodynamics to a greater extent than a single hit in rats exposed to Hx or treated with Su alone.31,32 Temporal examination of pulmonary hemodynamics and histological parameters showed that RVSP progressively increased to reach a plateau between 3 and 5 weeks after Su exposure correlated with pulmonary vascular remodeling. 33 Also, the decrease in vascular density, the increase in vascular separation, and the loss of small-diameter blood vessels detected by microcomputed tomography in the Su/Hx lungs support the pruning process of small pulmonary vessels in patients with PAH. 34
Therefore, the Su/Hx rat model mimics PAH progression observed in patients with PAH allowing the evaluation of drugs on pulmonary hemodynamics, RV dysfunction, and pulmonary vascular remodeling at different relevant time points of the disease, either as a preventive or therapeutic treatment. A meta-analysis of animal PAH models from 291 publications demonstrated that the Su/Hx model provoked a more severe hypertensive response resulting in greatest RVSP and RV hypertrophy compared with chronic Hx, monocrotaline, and left pneumonectomy prior to monocrotaline. 6
Epidemiology
Rat strain
Su/Hx challenge has been performed in different rat strains, and although differences exist in the reactivity to a Su/Hx challenge, the overall response between the different strains is dependent upon a number of methodological factors such as the dose and vehicle used for the Su administration,26,35–38 the duration of Hx exposure,27,39,40 and return to Nx exposures,33,41–45 and data collected at the end of the study in the presence or the absence of high inspired O2 concentrations that are required for animal survival during the extensive surgical procedures.4,37,41,42 Also, differences in respiratory system mechanics exist among rat strains,46,47 and Fischer, Wistar-Kyoto (W-K), S-D, Lewis, and Brown Norway rats have different susceptibilities to Hx-induced PAH in terms of vasoconstriction, PVR, RV hypertrophy, and remodeling.46–52
The most comprehensive study using the Su/Hx rat model comparing the reactivity of different rat strains to the Su/Hx challenge was performed in S-D, Fischer, and Lewis rats. 8 The authors reported that a “hyperreactive” strain of S-D rats was obtained from Charles River Laboratories (Montreal, QC, Canada) that exhibited PAH pathology, mortality, and occlusive lesions in the pulmonary blood vessels with Su administration alone, whereas the other rat strains, including a subset of “normal” S-D rats obtained from Harlan Laboratories (Indianapolis, IN, USA), required a second hit with 3 weeks of Hx to initiate the PAH pathology. Unfortunately, there is no information on the genetic constitution of these “hyperreactive” rats, and we have found that not all rats purchased from this Charles River facility in Canada exhibit this “hyperreactive” profile. 41 Fischer rats have a high prevalence of mortality after Su/Hx challenge with a high mortality by 7 weeks that is preceded by right heart enlargement despite comparable hemodynamics, RV hypertrophy, and occlusive pulmonary vascular lesion severity of PH in S-D rats with an excellent survival for up to 14 weeks.8,53,54 Jiang et al. 8 concluded that “normotensive” S-D rats are suited to the study of novel therapies for PAH, while the high mortality in the Fischer rats, prone to develop RV failure in response to the Su/Hx challenge, is more relevant for the study of RV decompensation and remodeling in response to severe PAH. 54 Lewis rats unresponsive to the Su/Hx challenge are not a suitable strain for this model of PAH. 8
The Fawn-Hooded (F-H) rat, a strain with a hereditary bleeding tendency due to a genetic defect in platelet aggregation, spontaneously develops PH. 55 It is one of the few rat strains that naturally develops a PAH pathology over time, but this occurs only with advanced age of between 20 and 40 weeks, which makes it impractical to use for drug evaluation studies.4,5 An increased sensitivity to PAP, PVR, RV hypertrophy, muscularization, and wall thickness of pulmonary arteries is observed upon mild exposure to Hx in these strains when compared with S-D rats. 56
Strong differences in the severity of PAH and survival of animals can therefore be found in response to Su/Hx among strains (Table 1), and even between colonies of the same strain, suggesting a major role of the genetic background. Therefore, the selection of an appropriate strain is essential for the Su/Hx rat model of severe PAH to study the hallmarks of human PAH.
Comparison Between Rat Strain and Study Design on Mortality, Hemodynamics, and Pathology After Sugen/Hypoxia Challenge
−: None.
(+): Slight: mortality >0%–30%, mPAP 25–40 mmHg, ↑ wall thickness and muscularization of small pulmonary arteries, ↑ right heart size.
(++): Moderate: mortality >30%, mPAP 40–60 mmHg, ↑ wall thickness, muscularization and obliteration of small pulmonary arteries, ↑ right heart size.
(+++): Severe: mortality >50%, mPAP > 60 mmHg, RVSP, 80–100 mmHg, ↑ wall thickness, muscularization obliteration and significant plexiform lesions in small pulmonary arteries, ↑ right heart size.
mPAP, mean pulmonary arterial blood pressure; NR, not reported; RVSP, right ventricular systolic pressure.
Age
In humans, the incidence of PAH is higher in older subjects. 12 However, the converse is found in Su/Hx- and chronic Hx-challenged rats where the most robust response is observed in young animals and PAH pathology is diminished in older rats.59,60 As mentioned previously, the only known rat strain that naturally develops PAH pathology over time is the F-H rat but this occurs after 20 to 40 weeks of age. 4
Gender
Gender is also an important epidemiological feature of human PAH, with a higher prevalence of the disease in females60–62 but with a lower rate of survival in males, an effect likely due to a reduced RV function in male PAH subjects. 63 The presence of estrogen plays an important role in the overall PAH pathology in female human subjects and in Su/Hx-challenged rats.62,64–66 Interestingly, in male Su/Hx rats, estrogen does not appear to be important in the pulmonary pathology 66 but has been proposed to play a role in the reductions in cardiac performance and in the increase of the right heart size after Su/Hx challenge.67,68 Based upon these findings, drug targets aimed at reducing estrogen signaling may prove to be effective therapies for the treatment of PAH, as a reduction in cardiac performance, and RHF is ultimately the cause of mortality in PAH.10,12,15,17,20,63
Genetics
Heterozygous, germ line mutations in the gene encoding the type II bone morphogenetic protein receptor 2, (BMPR2) account for 70%–80% of heritable PAH cases and 10%–20% of patients with idiopathic PAH (iPAH).10–13 Functionally, loss of BMPR2 signaling promotes endothelial cell dysfunction and endothelial cell-to-mesenchymal cell transition that contributes to the proliferation of PASMCs. 69 Because of its critical role controlling the homeostasis of the pulmonary blood vessels, restoration of BMPR2 signaling has been a target for many new therapies, many of which have been evaluated in Su/Hx rats.12,13,69 Mutations have also been found in activin receptor-like kinases (ALK1 and ALK2) that utilize engolin (ENG) as a coreceptor in the BMPR2 complex.69–71 Genetic mutations have been observed in many other PAH genes such as eukaryotic translation initiation factor 2α kinase 4, aquaporin 1, and caveolin 1 (CAV1), but there is no clear evidence of disease penetrance into PAH pathology.13,72 Similar to most genetic mutations in disease, the presence of epigenetic factors will significantly affect gene expression without altering the sequence of genomic DNA. 13
Hemodynamics, echocardiography, and pathology
Hemodynamics
Virtually every study with Su/Hx challenge in rats has found an increase in mPAP or RVSP above the threshold used to define PAH in human subjects. 11 There is a wide range in values between the different Su/Hx studies that will depend upon a number of factors such as the strain of rat, the dose of Su administration and exposure to the Hx challenge, and the duration of study (Table 1). In humans, the measurement of PCWP is critical to determine if the PH falls into the group 1 category (PAH), but this is rarely done in rodent models of PAH because of the technical difficulty performing this procedure.73,74 However, as left heart hemodynamics, including left ventricular systolic pressure, are unchanged after Su/Hx challenge,31,75 the increase in mPAP and RVSP likely reflects changes in hemodynamics in the right heart.
With prolonged remodeling of the pulmonary blood vessels, their distensibility is significantly reduced, resulting in an increased right heart pressure to overcome the imposed afterload. 76 In humans, a variety of parameters can be used to measure pulmonary arterial stiffness and compliance, including noninvasive techniques such as Doppler echocardiography and magnetic resonance imaging (MRI), and are claimed to be a better predictor of clinical outcomes than the classical pulmonary vascular testing measurement.76–82 A few studies have demonstrated a reduction in pulmonary vascular compliance following Su/Hx challenge in rats,81,83 but as this parameter is not routinely measured, it remains to be demonstrated if pulmonary arterial compliance is an attractive early screening tool in Su/Hx rats to help predict the outcome of new drug evaluations in clinical subjects.
Echocardiography
Echocardiography is the gold-standard noninvasive technique to diagnose PH and is an important modality to follow the progression of the disease over time.10,84,85 Echocardiography is routinely employed to assess cardiac and pulmonary arterial hemodynamics in Su/Hx rats, including the parameters PAD, PAAT, Vmax, VTI, and heart rate (HR).85–87 From these parameters, mPAP can be derived from its inverse relationship with PAAT and cardiac output (CO), calculated as the product of stroke volume (SV) and HR.85–87 Additional measurements of RVAWT and TAPSE can also be measured by echocardiography, and they are excellent predictors of the increase in right heart size and reduction in CO, respectively.83–87 In our previous studies, the largest changes were observed in PAAT, Vmax, VTI, SV, and CO on Day 21 (immediately after the Su/Hx challenge) that decrease in magnitude over time by 5–8 weeks after challenge.41,42 Similar results have been found with the reduction in TAPSE and increase in right ventricular end diastolic diameter (RVEDD) that were largest during the Hx challenge but reverted to the baseline values by 3 weeks of Nx room air breathing. 35
Pulmonary pathology
Extensive remodeling occurs in the pulmonary blood vessels in PAH that includes an increase of wall thickness in the small to moderately sized pulmonary arteries, hypertrophy and hyperplasia of smooth muscle in the pulmonary artery vascular wall, neointimal formation with damage to the vascular endothelium and loss of CAV1 resulting in the formation of plexiform lesions, and obstruction of the small-diameter pulmonary arteries. 24 Many of these features have been found in Su/Hx rats.26,27,30,33,88–92
Pathological alterations are found in each of the three layers of the pulmonary arterial vascular wall (tunica intima, tunica media, and tunica adventitia) with alterations in the vascular endothelial cells in tunica intima thought to be the initiating event in PAH pathology.26,35,93 Following damage to the vascular endothelium and loss of many of its vital functions, including CAV1 expression, reciprocal activation of proliferative and antiapototic pathways leads to the initiation and progression of the disease. Neointimal formation is particularly prevalent in severe PAH in both Su/Hx rats and humans.24,27,35,93
In the first few weeks after Su/Hx challenge, there is an expansive proliferation of the smooth muscle in the tunica media, but this tends to decrease over time as the smooth muscle changes from a contractile to a synthetic/proliferative phenotype.4,10,26,33,45,88,94 During this transition, there is a loss of vasoconstrictor and vasodilator responsiveness in the pulmonary arteries involving the NO pathways, 31 whereas other constrictor pathways such as the Rho-kinase pathway is left intact. 92 Therefore, drug targets that are aimed at reducing smooth muscle mass in the pulmonary arteries would need to be administered early in the Su/Hx challenge to optimize their efficacy.
The tunica adventitia is the outermost layer of the vascular wall and is mainly composed of collagen and an elastic lamina. Its proliferation contributes significantly to increased wall thickness89,93 and reduced arterial compliance/increased impedance in the pulmonary arteries. 81 As previously mentioned, a reduction in compliance of the pulmonary arteries is an important predictor of clinical outcomes.76–81,95
The anatomy of plexiform lesions in Su/Hx rats closely resembles that found in human PAH, with two types of lesions identified, one present within the vascular lumen and the other present outside the vascular lumen invading gradually into the lung parenchyma. 27 The plexiform lesions formed within the vascular lumen occur predominantly at branching points in the pulmonary arteries and contains VSMCs, chromatin-rich oval core cells, matrix proteoglycans, collagen, and PAECs. 27 Small slit-like channels are also present that allow for blood flow through these structures. As previously mentioned, the Su/Hx rat model is one of the few preclinical models that demonstrates this important pathological trait found in human PAH.4,7,27,35,88
Cardiac pathology
There are significant changes in the biochemical, metabolic, and molecular signaling pathways in the right heart that occur in response to increased afterload imposed by vasoconstriction and remodeling of the pulmonary blood vessels. The RV initially adapts (adaptive phase) to the increased afterload by increasing its wall thickness and contractility, but in the vast majority of patients, these compensatory mechanisms are insufficient and RV dysfunction occurs (maladaptive phase) if left untreated, resulting in RV failure and death.10,12,15 Anatomical and biochemical changes in the adaptive phase include hyperplasia/hypertrophy of the right ventricular myocytes, an increase in blood vessel density (angiogenesis), a change from predominantly fatty acid oxidation to one of glycolysis with an associated increase in glucose oxidation, decreased mitochondrial activity, and neurohumoral activation of both the sympathetic nervous system (SNS) and the renin–angiotensin–aldosterone system (RAAS).15,20 In the transition to the maladaptive phase, there is an increase in RV muscle mass, dilation of the RV, a reduction in new blood vessel formation, ischemia, a decrease in coronary arterial blood flow, and the presence of inflammatory cells and collagen deposition in the right heart.15,20
In Su/Hx-challenged rats, most of these pathological and physiological events of both the adaptive and maladaptive phases of PAH are reproduced but are critically dependent on the severity and duration of the Su/Hx challenge and the strain of rat used for evaluation. In each version of the Su/Hx model, there is an increase in right heart mass along with an increase in PVR and pulmonary vascular remodeling. The increase in size of the RV is often measured by the increase in the Fulton index and supported by the measurement of RVAWT by echocardiography.4,41,42,86–88 There is also an increase in the myocyte cross-sectional area along with the presence of angiogenesis and new blood vessel formation in the right heart.38,96–99 Changes in the metabolic signaling pathways in the right heart have also been described in Su/Hx rats with an increase in glucose utilization that is also associated with a decrease in fatty acid oxidation,97,100–103 an increase in reactive oxygen species (ROS), 104 pulmonary fibrosis,36,41–43,53,105 and the presence of inflammatory cells.106,107 Collectively, these results demonstrate that Su/Hx rats exhibit most of the important pathological features of PAH in the heart leading up to RHF, which makes this the most appropriate model to evaluate new drug targets that are directed to this aspect of PAH pathology. 108
Overall, the key pathological manifestations (pulmonary and cardiac) of PAH in patients are mostly reproduced in the Su/Hx rat model (Fig. 2) and are summarized in Table 2.
Similarities Between Pulmonary Arterial Hypertension in Humans and the Sugen/Hypoxia Rat Model of Pulmonary Arterial Hypertension
BMPRII, bone morphogenetic protein receptor type 2; cGMP, cyclic guanosine monophosphate; MLK, mixed-lineage kinases; mPAP, mean pulmonary arterial pressure; NO, nitric oxide; PVR, pulmonary vascular resistance; Rho, Ras homologous; RhoA, Ras homolog family member A; RVSP, right ventricular systolic pressure.
Drugs Tested in the Su/Hx Rat Model
Listed below are the currently approved PAH drugs and other compounds that have been evaluated both in the Su/Hx rat model and in clinical trials with patients with PAH. In this report, the drugs have been categorized into pulmonary vasodilators, anti-inflammatory drugs, antiproliferative compounds, antifibrotic agents, and nuclear modulators based upon their primary pharmacological activities, although there is significant overlap for many of these drugs. Many other drugs have also been evaluated in different animal models of PAH and are described in recently published review articles.77,108,111
Nearly all compounds currently approved for clinical use in PAH have been shown to have beneficial effects on the multiple aspects of PAH pathology in the Su/Hx rat model (Table 3), regarded as the most translatable animal model for PAH, and other drugs both tested in this model and assessed in clinic are shown in Table 4. A total of 26 compounds are listed in Table 4, and only two compounds, selonsertib from the tyrosine kinase (TK) inhibitor class and spinolactone from the renin–angiotensin–aldosterone inhibitor class, showed positive effects in the Su/Hx rat model but had no efficacy in patients with PAH, indicating a good translatability of preclinical findings on efficacy in the Su/Hx rat model to human PAH application.
Effects of Food and Drug Administration-Approved Drugs for Pulmonary Arterial Hypertension in the Sugen/Hypoxia Rat Model
ɣ: Positive effect.
FDA, Food and Drug Administration; NT, not tested.
New Potential Drugs for the Treatment of Pulmonary Arterial Hypertension in Clinical Trials and Tested in the Sugen/Hypoxia Rat Model
ɣ: Positive effect.
: High doses only.
&: Female rat only.
#: Preventive treatment.
: Therapeutic treatment.
AE, adverse effects; BMPR2, bone morphogenetic protein receptor type 2; CO, cardiac output; Dn, denervation; HR, hear rate; Hx, hypoxia; Inhal, inhalation; IV, intravenous; mPAP, mean pulmonary arterial pressure; 6-MWD, 6-min walk distance; NE, no effect; NS, not significant; NT-pro-BNP, N-terminal-pro-brain natriuretic peptide; PASP, pulmonary arterial systolic pressure; POP, proof of principle; PVR, pulmonary vascular resistance; RV, right ventricle; Sc, subcutaneous; SV, stroke volume; TAPSE, tricuspid annular plane systolic excursion.
Pulmonary vasodilators
Pulmonary vascular tone is regulated by the balance between vasoconstrictor and vasodilatory stimuli, and a change in this balance contributes to the development of PAH in humans and in preclinical models. Increased contractility of small pulmonary arteries and arterioles in patients with PAH is a pathological feature of PAH driven by the increased secretion of vasoconstrictor substances such as thromboxane and ET-1 and/or inhibition of vasodilator mediators such as prostaglandin I2 (PGI2) and NO.201–203 Therefore, many drugs evaluated for use in PAH (and all currently approved PAH therapies) either relax PASMC or inhibit PASMC contraction. Detailed below are some of the important features of these compounds, results of evaluations in Su/Hx rats, and insights into the translation of those results to the clinic.
Prostacyclin analogs
Endothelial cells produce the endogenous prostanoid prostacyclin from arachidonic acid via the enzymes prostacyclin synthase and cyclooxygenase. Prostacyclins act as vasoactive mediators, which bind and activate the G protein-coupled receptor leading to the production of cyclic adenosine monophosphate (cAMP) and ultimately to the relaxation of the VSMCs. These G-protein coupled cell surface receptors are broadly categorized into nine different categories based upon their affinity to different prostanoid ligands. They include prostaglandin D2 receptors (DP1 and DP2), prostaglandin E2 receptors (EP1, EP2, EP3, EP4), the IP, prostaglandin F receptor (FP), and the thromboxane receptor (Tp), although further categorization of these receptors into different subtypes has been described.204–207 The endogenous PGI2, or prostacyclin, by binding to the relaxant prostanoid IP receptor induces VSMC relaxation and vasodilatation via cAMP and has antiproliferative, antiplatelet, antithrombotic, and anti-inflammatory properties. Some of the clinically approved prostacyclin analogs have selectivity for the IP receptor (epoprostenol, selexipag), whereas others (iloprost, beraprost, treprostinil) bind to several prostanoid receptors resulting in differential pharmacological activities. In addition to activation of the prostanoid receptors, iloprost and treprostinil potently bind to the peroxisome proliferator-activated receptor (PPAR) that influences much of the biology in structural, inflammatory, and immune cells that are involved with PAH pathology.208–210 In 1995, IV epoprostenol (Flolan, GlaxoSmithKline Research Triangle Park, NC, USA), a synthetic analog of PGI2, was the first approved drug by the FDA for the treatment of PAH. 211 Prostanoids with greater chemical stability, a longer half-life, and simpler modes of administration were subsequently developed.
Iloprost
Iloprost (Ventavis, Janssen Pharmaceuticals, Titusville, NJ, USA) is a synthetic prostacyclin analog that has high binding affinity to EP1, IP, and PPARs, low binding affinity to FP, EP3, and EP4, and very low binding to EP2, DP1, and Tp receptors.205,206 Although pulmonary vasodilation is obtained with inhaled iloprost in patients with PAH, it did not attenuate the increases in mPAP and right heart size, or on the number of occluded pulmonary blood vessels in Su/Hx rats. 115 This may be due to underdosing with the drug as it was administered at a dose of 0.1 µg/kg only 3 times per day for 2 weeks, and in humans it is given at 2.5 to 5 µg/kg up to 9 times daily. 115 Interestingly, EP1 receptor activation induces desensitization of the IP receptor, which may contribute to the inactivity of iloprost in Su/Hx rats on the basis of its potent activity as an EP1 receptor agonist. 206 On the contrary, inhaled iloprost inhibited fibrosis in the RV and improved some cardiac functions such as TAPSE in Su/Hx rats. 115 These results raise the intriguing possibility of a difference in the potency of iloprost by binding to cardiac and pulmonary IP and PPARs that have both been implicated in fibrosis formation in the heart.212–217
Treprostinil
Treprostinil (United Therapeutics Corporation, Research Triangle Park, NC, USA) is approved for the treatment of PAH via administration by the SC (Remodulin), IV (Remodulin), oral (Orenitram) or inhaled (Tyvaso) routes. Treprostinil activates both the receptors, prostanoid and PPAR, with a rank order of binding potency for the prostanoid receptors of EP2 > IP > DP1 > EP1 > EP3 > EP4 > FP > Tp.206,218 However, data from studies in Su/Hx rats and mice indicate that different effects are reported with treprostinil depending upon the route of administration. For example, robust inhibition of the changes in pulmonary vascular hemodynamics induced by Su/Hx is reported with IV and SC treprostinil but not when it is given four times a day (QID) by the inhaled route.42,112 Recently it was shown that treprostinil exerts cardiopulmonary effects as IV treprostinil improved RV function (increased in RV ejection fraction, stroke work, contractile index, and reduced RV systolic volume) in Su/Hx rats that may be due to a direct effect on the RV, in addition to its protective effect on the pulmonary vasculature. 113 On the contrary, inhaled treprostinil inhibited the muscularization and obliteration of small pulmonary arteries to a greater extent than IV treprostinil. 42 This may be due to the presence of locally high concentrations of treprostinil in the lung after inhalation with lower concentrations when administered by the IV or SC routes. The strain of rat used for these studies may also have been a contributing factor to these different responses to IV and SC treprostinil, as severe PAH was produced in the SC treprostinil studies, whereas mild-to-moderate PAH was observed in studies involving inhaled and IV treprostinil, which was also performed in the presence of high-inspired O2 levels on the final day of data collection.42,112
Treprostinil palmitil
Treprostinil palmitil (TP, Insmed Incorporated, Bridgewater, NJ, USA) is a long-acting prodrug of treprostinil that has been studied for administration as a nebulized lipid nanoparticle suspension (treprostinil palmitil inhalation suspension, TPIS), as a metered dose inhaler (treprostinil palmitil inhalation aerosol) and is now in clinical development as a dry powder (treprostinil palmitil inhalation powder, TPIP).41,42,218,219 Both TPIS and TPIP inhibit most of the pathophysiological features induced by Su/Hx challenge in rats, with effects generally superior to those produced by other compounds approved for the treatment of PAH such as oral sildenafil, 41 inhaled and IV treprostinil, and oral selexipag. 42 Both TPIS and TPIP maintain relatively high concentrations of TP in the lungs over 24 hours after a single dose and provide sustained pulmonary vasodilator and anti-remodeling activity that may support once a day (QD) dosing rather than QID dosing that is recommended for inhaled treprostinil.218,220 In a clinical phase I trial, TPIP was generally well tolerated and provided a sustained release of treprostinil with a pharmacokinetic (PK) profile supporting QD dosing. 221
Beraprost
Beraprost sodium (Toray Industries, Chuo City, Tokyo, Japan) is an orally administered prostacyclin analog that has been approved for the treatment of PAH in Japan and South Korea.222,223 Beraprost has a very short elimination half-life of only 35–40 minutes and has only transient clinical benefits for 6 months after QID oral administration in adults, 224 and no effect on pulmonary pressures or PVR in children. 225 It has high binding affinity to the IP receptor with lower binding affinity to the EP3 and EP4 receptors.224,226–229 Esuberaprost, is a reformulated single isomer of berapost that is the most potent IP agonist developed for therapeutic use to date. 230 Studies in Su/Hx rats demonstrate that the duration of activity of beraprost is extended by its incorporation into polyactide–glycolide polymer nanoparticles and administration by inhalation. 44 Similar findings of an extended duration of activity with beraprost were demonstrated in monocrotaline PAH in rats and chronic Hx challenge in mice when it was encapsulated in a copolymer nanoparticle. 231 These results demonstrate the benefits of reformulating active drugs in nanoparticles to extend their duration of activity.
Selexipag
Selexipag (Uptravi, Actelion Pharmaceuticals Incorporated, San Francisco, CA, USA) is an orally, nonprostanoid selective IP receptor agonist targeting the prostacyclin pathway with different PK and pharmacodynamic (PD) properties than synthetic prostacyclin (epoprostenol) and prostacyclin analogs (beraprost, iloprost, and treprostinil).10,77,232,233 The parent compound (selexipag) binds selectively to the IP receptor and its active metabolite ACT-333679 behaved as a full agonist in multiple PAH-relevant receptor cellular assays and displayed partial agonism in receptor cAMP accumulation assays resulting in limited IP receptor desensitization, β-arrestin recruitment, and IP receptor internalization. 232 Robust efficacy has been demonstrated with oral selexipag in Su/Hx rats, but the dose used in this rat study (30 mg/kg) BID is much higher than the recommended clinical dose in humans,42,116 as the IP receptor binding affinity of both selexipag and ACT-333679 is lower in rats compared with humans.206,230 Other factors such as differences in the PK profile and metabolism likely contribute to the different doses used to demonstrate efficacy in Su/Hx rats and humans.
NO–cyclic GC pathway
The endothelial dysfunction observed in PAH affects NO, the principal physiological mediator of vasodilatation, and the reduction of NO produces an increase in vascular tone. The NO-cyclic guanosine monophosphate (cGMP) pathway can be modulated by stimulators of the sGC and PDE5 inhibitors, both of which increase cGMP levels, and therefore stimulate vasodilation. Compounds in the NO-sGC-cGMP signaling pathways include the clinically approved PDE-5 inhibitors, sildenafil and tadalafil, and the sGC stimulator, riociguat. Inhaled NO also has been used to treat PAH subjects, particularly in pediatric cases but its use is limited due to cost, technical difficulties in its administration, and to the fact that not all patients respond to the therapy. 234 The NO pathway has been clearly identified in the pathology of PAH and new therapies in this drug class are in development, some of which have been evaluated in Su/Hx rats.45,77,108,111
Sildenafil
Sildenafil (Revatio, Pfizer Incorporated, New York, NY, USA) was the first oral PDE5 inhibitor approved for the treatment of PAH
235
followed by the structural distinct compound tadalafil.
236
NO is formed by the cleavage of the terminal amino group from the amino acid
An oral combination therapy of sildenafil and the thromboxane A2 receptor antagonist NTP42 in Su/Hx Wistar-Han rats resulted in improvements in all parameters assessed, including hemodynamics (PAP and RVSP), cardiac hypertrophy, fibrosis, pulmonary vascular remodeling (pulmonary vessel diameter, medial thickness, and occlusion), and pulmonary pathology and this combination acted synergistically. 242 Also, an inhaled combination of poly(lactic-co-glycolic) acid (PLGA) particles, comprising sildenafil and rosiglitazone, reduced mPAP, PVR, RV hypertrophy, collagen deposition, and muscularization of pulmonary arteries and improved cardiac function in Su/Hx S-D rats. 36 This combination therapy with PGLA particles of two distinct drugs, sildenafil and rosiglitazone, produced similar improvements in PAH pathology although administered at less frequent intervals compared with the single or dual oral combination of the two drugs. 36 Similar to drugs in the prostacyclin category, experimental studies in Su/Hx rats demonstrate that delivery via the inhaled route and incorporation in a PGLA formulation had positive effects by improving efficacy and reducing the dosing frequency compared with oral sildenafil. 123 Intratracheal (IT) administration of PLGA particles of sildenafil reduced mPAP to a greater extent than IV and IT sildenafil and for a longer time period (∼ 6 hours vs. 120 and 60 minutes, respectively). No effect on mPAP was observed with oral sildenafil. 123 Combination therapy can be a promising approach.
Tadalafil
The PDE5 inhibitor tadalafil is also given as an oral medication for the treatment of PAH under the brand name of Adcirca (United Therapeutics Incorporated, Research Triangle Park, NC, USA), which was repurposed from the initial approval in erectile dysfunction marketed as Cialis (Lilly ICOS, Indianapolis, IN, USA). In Su/Hx rats, oral tadalafil improved the pulmonary vascular hemodynamics and cardiac functions but had no effect on pulmonary vascular remodeling. 119 Although tadalafil attenuated the Su/Hx-induced remodeling and dysfunction in the RV, its effects were inferior to that produced by the ERA macitentan. 119 Importantly, tadalafil and macitentan alone or in combination attenuated the majority of the plasma metabolic changes induced by Su/Hx challenge that included effects on the NO and fatty acid pathways by reducing plasma levels of citrulline and long-chain fatty acid carnitines. 119 These results support not only the findings with sildenafil in Su/Hx rats demonstrating marked effects on right heart pathology and function with lesser effects on pulmonary vascular remodeling but also the view that combination therapies are generally superior to monotherapy for the treatment of PAH.10,119,243
Riociguat
Oral riociguat (Adempas, Bayer Healthcare Pharmaceuticals, Whippany, NJ, USA) is the first sGC simulator approved for the treatment of PAH. It directly stimulates the key enzyme of the NO signaling pathway sGC, even in the absence of NO, improving cGMP synthesis and producing pulmonary vasodilatation with antiaggregation and antiproliferation effects.3,234,244,245 Riociguat is also approved for the treatment of inoperable chronic thromboembolic pulmonary hypertension (CTEPH). 246 sGC is often dysfunctional in PAH 247 and riociguat directly activates sGC, is readily absorbed, and has oral bioavailabity of 94%. 246 In Su/Hx rats, oral riociguat improved pulmonary vascular hemodynamics, RV hypertrophy, and pulmonary vascular remodeling with effects generally superior to that produced by oral sildenafil. 45 Riociguat also inhibited the formation of occlusive vascular lesions in the pulmonary arteries under conditions of severe PAH induced by the Su/Hx challenge. 45 Robust effects were also observed on the histopathology and right heart functions in both Su/Hx-challenged rats and pulmonary arterial banding in mice,45,248 suggesting that the agents acting through the NO-sGC pathway have effects on right heart functions separate from effects on the pulmonary vasculature. In clinical trials, mixed results have been observed with oral riociguat mainly due to adverse side effects such as hypotension, especially when it has been combined with oral sildenafil.1,249,250
Inhaled NO
Inhaled NO (INOMAX, INO Therapeutics, LLC, Bedminster, NJ, USA) has been given to patients with PAH with mixed success. 251 It is often used to treat patients with primary PH and in subjects with secondary PH associated with congenital or acquired heart disease, chronic obstructive lung disease, and acute lung injury/acute respiratory distress syndrome. 251 However, long-term treatment with inhaled NO is not a viable option as potential toxicity occurs with the formation of toxic nitrogen oxides, peroxynitrite, and hydroxyl radicals. 251 In Su/Hx rats, inhaled NO demonstrates acute pulmonary vasodilation, but its effect was inferior to that observed with other pulmonary vasodilators such as the Rho kinase inhibitor fausidil or the prostacyclin analog iloprost. 92 Furthermore, inhaled NO has little effect on pulmonary vascular remodeling.92,252 On the basis of these results in Su/Hx rats, inhaled NO would not be considered a good option for long-term use in the treatment of PAH. However, combination therapy with the Rho-kinase inhibitor fausidil and diethylenetriamine NONOate (DETA NONOate), a long-acting NO donor, formulated in a peptide liposomal carrier, improved the pulmonary hemodynamics and pulmonary vascular remodeling in Su/Hx rats, suggesting that this combination therapy may have benefit for the treatment of PAH. 91
ET antagonists
ET-1, secreted by the endothelial cells, is the most abundant ET and is a potent vasoconstrictor and VSMC mitogen. It exerts its effects on two receptor subtypes, ETA and ETB receptor. 253 ET-1 expression level is upregulated in the lungs of patients with PAH, 202 which led to the evaluation of oral ERAs in PAH.
Three ERAs, bosentan, ambrisentan, and macitentan, are currently approved for use in the treatment of PAH. Each of these compounds improves hemodynamics, RV function, and exercise capacity and reduces mortality in PAH subjects.77,254–256 Pharmacologically, bosentan and macitentan are classified as dual ET receptor antagonists, which bind to the ETA and ETB receptors, while ambrisentan has greater selectivity for the ETA receptor.77,255,256 However, there is no difference in the efficacy between dual ETA/B receptor antagonists and selective ERAs in patients with PAH. 256 Hepatotoxicity, edema/fluid retention, and anemia are the major adverse effects (AEs) reported with these ERAs. 255
Bosentan
Bosentan (Tracleer, Actelion Pharmaceuticals, South San Francisco, CA, USA) was the first ERA approved for the treatment of PAH. The only study with bosentan in the Su/Hx rat model was performed in combination with the compound LCZ 696 (sacubitril/valsartan). 117 LCZ 696 is a combination of the angiotensin II receptor blocker valsartan and the neprilysin inhibitor sacubitril, and the oral combined therapy of bosentan and LCZ 696 attenuated the increases in mPAP, PVR, RV hypertrophy, pulmonary vascular remodeling (wall thickness and muscularization of pulmonary arteries), and inflammatory cell infiltration in the lungs of male Wistar Su/Hx rats. 117 Cardiac function was also improved in Su/Hx rats probably through plasma level elevation of cGMP and atrial natriuretic peptide (ANP). Moreover, the antiproliferative effect of LCZ 696 on PASMCs derived from patients with iPAH was more pronounced in the presence of bosentan. 117
Ambrisentan
In Su/Hx rats, oral ambrisentan (Letairis, Gilead Sciences Incorporated, Foster City, CA, USA) improved the pulmonary hemodynamics and reduced the occlusive vascular lesions in the small pulmonary arteries, with more pronounced effects when administered prophylactically during the Hx challenge rather than by therapeutic administration after the Su/Hx challenge. 122 These findings suggest that the early administration of ERAs may be important to optimize the efficacy of this drug class.
Macitentan
Several studies have been performed with macitentan (Opsumit, Actelion Pharmaceuticals, South San Francisco, CA, USA) in Su/Hx rats, with positive effects observed on pulmonary vascular hemodynamics, RV hypertrophy, medial wall thickness, and obliteration of pulmonary blood vessels.99,102,120,257,258 Macitentan also improved RV functions that were associated with metabolic changes in the heart of Su/Hx rats, characterized by increased glucose uptake and to a lesser extent increased fatty acid uptake. 102 There was an early improvement in PAH hemodynamics and pathology with macitentan that decreased over time, and the efficacy of macitentan relative to that of bosentan and ambrisentan might be related to the better tissue penetration and binding kinetics of this compound. 258 There was also an improvement in microvascular density in both the lungs and RV of Su/Hx rats treated with macitentan, results that support the view that ET-1 influences angiogenesis by an effect involving both ETA and ETB receptors.102,121,259 In humans, clinical trials (SERAPHIN, phase III, NCT00660179) indicate that macitentan significantly decreased morbidity and mortality with an efficiency comparable with bosentan and ambrisentan but with fewer AEs due to its unique PK profile.260–263 Recently, macitentan treatment resulted in significant improvements of RV function and structure, and cardiopulmonary hemodynamics in a clinical trial (REPAIR, phase IV, NCT02310672). 264 Overall, the effects of macitentan in Su/Hx rats show excellent translation to humans both in terms of efficacy and AEs.
Rho-kinase antagonists
The RhoA/Rho-kinase signaling pathway is believed to be a therapeutic target for PAH, and this enzyme is involved in a variety of cellular functions associated with PAH pathology, particularly the contraction of pulmonary VSMCs and vascular remodeling. 265 In addition, it is a calcium (Ca++) sensitizer that opens up ATP-dependent K+ channels in pulmonary arteries and promotes the proliferation of PASMC, which is an activity also observed with the inodilator levosimendan in the Su/Hx rat model. 266 Rho-kinases mediate the effects of many upstream mediators of PAH such as ET-1, thromboxane A2, and serotonin, endothelial NOS, and inhibition of these pathways represents a viable target for PAH.92,265,266
Fasudil
Fasudil is a calcium channel blocker (CCB) that primarily acts through the inhibition of the Rho-kinase signaling pathway leading to vasodilation through the activation of myosin phosphatase. The potent Rho kinase inhibitor fausidil (HA-1077, Woolsey Pharmaceuticals Incorporated, New York, NY, USA) has been preclinically evaluated in Su/Hx rats and has advanced into clinical trials. In Su/Hx rats, IV fasudil caused greater pulmonary vasodilatation than inhaled NO, IV iloprost, and IV bradykinin, 92 suggesting that inhibitors of the Rho-kinase pathway may have benefits over other pulmonary vasodilators in patients who respond poorly to conventional vasodilator therapy. Experiments in Su/Hx rats have been also performed following inhaled fasudil administered in peptide-modified liposomal carriers and in combination with other drugs such as superoxide dismutase and the long-acting NO donor, DETA NONOate.91,143 In both studies, the combination therapies produced a more pronounced reduction in mPAP and RV hypertrophy, and decreases in the medial arterial wall thickness, muscularization, and collagen content of the pulmonary arteries than either drug given alone. Also, the cyclic peptide augments the therapeutic effect of fasudil formulated in liposomes in a series of in vitro, ex vivo, and Su/Hx rat studies by increasing lung residence time, reducing particle clearance, and elevating the fraction retention of liposomal fasudil in the lungs. 144 Recently, fasudil dichloroacetate, an oral drug synthetized from fasudil and the metabolic modulator dichloroacetate, showed synergistic effects on pulmonary hemodynamics, pulmonary and RV remodeling, in the Su/Hx rat model. 267 All these results demonstrated that a combination therapy represents an interesting approach and yields a better protection for the treatment of PAH than when the drugs are given as monotherapy. In patients with PAH with various etiologies, IV, inhaled, or oral fasudil produced hemodynamic effects (PAP, PVR, CO).133–142
Calcium antagonist/sensitizer
PAH is characterized by vasoconstriction and hyperproliferation of PASMCs leading to pulmonary vascular wall thickening, and Ca2+ is a key intracellular signaling element for both contraction and proliferation of VSMC. 268 Drugs acting directly on Ca2+ channels such as CCBs reduced pulmonary vascular cell proliferation and vasoconstriction leading to relaxation of pulmonary arteries.
Tetramethylpyrazine
Tetramethylpyrazine (TMP), also known as ligustrazine, a compound isolated from the traditional Chinese herb ligusticum and the fermented Japanese food natto, 196 blocked the entry of extracellular calcium and inhibited the release of intracellular stored calcium in the VSMCs. 269 Oral administration of TMP in either preventive or therapeutic paradigms reversed PAH-related increases in RVSP, RV hypertrophy, and pulmonary vascular remodeling. 196 Similar preventive and therapeutic effects of TMP were observed in two other PAH animal models, the chronic Hx and monocrotaline rat models. 196 In a small clinical study, oral administration of TMP (ChiCTR-IPR-14005379) for 16 weeks in patients with CTEPH and PAH significantly improved the six-minute walk distance (6-MWD) test and PAD. 196 A larger randomized, single-blinded clinical study (120 subjects) to evaluate the efficacy and safety of TMP in the treatment of PAH has been initiated. 197
Levosimendan
A calcium sensitizer is a molecule that regulates the contractile force without inducing any changes in the calcium transient. The small calcium binding protein troponin C that activates the actin-myosin interaction is a molecular target for calcium sensitizers. 270 The troponin-targeting drug levosimendan was initially discovered and developed by Orion (Espoo, Finland), and IV levosimendan is approved in over 60 countries (not yet in the United States) for use in patients with decompensated heart failure. 270 Although principally used in the management of acute heart failure syndromes, levosimendan may play a role in PAH. Levosimendan is involved in cardiac contractility, dilatation of coronary and pulmonary circulation, and cardiac protection.38,96 In the Su/Hx rat model, oral treatment of levosimendan before (prevention) or 6 weeks after (therapeutic) Su/Hx challenge did not affect mPAP, CO, or RV hypertrophy, but occlusion lesion development, cardiomyocyte size, and gene expression of heart failure markers, ANP and brain natriuretic peptide (BNP), were significantly reduced in the preventive group. 38 Only chronic treatment attenuated pulmonary vascular remodeling and prevented the development of RV failure. In a pilot study conducted in Germany and Sweden, including a limited number of patients with PH with various etiologies (iPAH, PH due left heart disease, and chronic thromboembolic disease), acute (24 hours) and long-term (2 months) IV infusions of levosimendan lowered PAP and PVR. 198 In subsequent clinical studies, levosimendan infusion also reduced mPAP, PVR, and the biochemical marker N-terminal pro-BNP (NT-proBNP), and increased the 6-MWD of the patients with iPAH associated with RHF, 199 and IV levosimendan improved 6-MWD, NT-proBNP, echocardiographic parameters of RV function in patients with PAH of diverse causes. 200 The preclinical data associated with the beneficial effects of levosimendan in patients with PAH of various etiologies warrant larger studies. However, we must note that in two case reports, levosideman increased pulmonary pressures in two patients with iPAH and did not lead to a substantial improvement of their conditions.
Transient receptor potential channel
In pulmonary arterioles, increased cytosolic calcium induced proliferation of PAECs involved in the formation of occlusion lesions and caused contraction, proliferation, hypertrophy, and hyperplasia of PASMCs. 53 Increases in cytosolic calcium require calcium influx through transmembrane channels and the transient receptor potential channel 4 (TRPC4), an ion channel involved in the formation of a store-operated calcium entry complex, promotes contraction and proliferation of lung vascular cells. 53 After induction of PAH in TRPC4-knockout Fischer 344 rats, TRPC4 inactivation reduced mortality associated with severe PAH by reducing occlusive remodeling and the susceptibility to heart failure. Francis et al. 271 demonstrated a TRPC4-dependent increase in the permeability of isolated Fischer rat lungs after store depletion with the guaianolide thapsigargin and demonstrated that TRPC4 provides a calcium source associated with endothelial dysfunction in the pathophysiology of PAH.
Anti-inflammatory drugs
Perivascular inflammation is a prominent feature of PAH with high levels of cytokines, chemokines, and inflammatory mediators detected in patients with PAH that correlate with clinical outcomes.106,107,272–274 Furthermore, virtually all lineages of inflammatory cells are detected in the pulmonary circulation. Numerous treatments targeting the inflammatory and immune system have been explored as potential therapies for PAH, several of which have been evaluated in Su/Hx rats.106,107,111,272 Unfortunately, many of these compounds have failed in clinical trials, and it may require specific targeting of different PAH phenotypes and selective timing of administration for these compounds to show success. Listed below are the anti-inflammatory drugs evaluated in Su/Hx rats that have advanced into clinical trials.
Serotonin inhibitors
The monoamine neurotransmitter serotonin, or 5-hydroxytryptamine (5-HT), has long been associated as a major factor contributing to the pathology of PAH. In addition to its well-documented effects inducing pulmonary vasoconstriction, serotonin also induces the proliferation of PASMCs, activates pulmonary arterial fibroblasts, promotes adventitia fibrosis, induces hydrogen peroxide formation and oxidative stress, and may synergize with BMPR2.
275
This has led to the development of therapies aimed at suppressing serotonin signaling either through antagonism of the different serotonin receptor subtytpes or by interference with serotonin biosynthesis through modulation of the rate-limiting enzyme
RP5063
RP5063 (brilaroxazine, Reviva Pharmaceuticlas, Santa Clara, CA, USA) is a multimodal serotonin receptor modulator with high affinity for the 5-HT 1A/2A/2B/7 receptors and moderate binding affinity for the 5-HT transporter (SERT). Clinical phase I trials with this compound have been completed for the treatment of PAH and a phase II study has been initiated. In the Su/Hx rat model, RP5063 (10–20 mg/kg oral, BID) demonstrated nonsignificant effects against the increases induced by the Su/Hx challenge in systolic PAP, RVSP, and RV hypertrophy 37 but significantly affected wall thickness, muscularization, and obliteration of small and medium pulmonary arteries with a partial reduction of the plexiform lesion formation. In comparison, oral sildenafil had no effect on plexiform lesion formation even though effects were observed on the wall thickness and muscularization of the pulmonary arteries. 37
KAR5585
Instead of blocking selective 5-HT receptors, a new approach is to reduce systemic 5-HT tone by reducing the ligand for these multiple receptors through a partial blockade of the rate-limiting enzyme for 5-HT synthesis, tryptophan hydroxylase 1 (TPH1).189,278 KAR5585 (rodatristat ethyl, Altavant Sciences, Cary, NC, USA) is a TPH1 inhibitor blocking peripheral 5-HT production, 189 which has demonstrated safety in healthy adult subjects (NCT02746237) 279 and has advanced into a phase IIB clinical trial for patients with PAH (ELEVATE 2, NCT04712669). 278 Rodatristat is the active component of the oral prodrug rodatristat ethyl. In Su/Hx rats, therapeutic treatment with KAR5585 (100 mg/kg) significantly affected the wall thickness only, while preventive treatment had significant effects on mPAP, wall thickness, and vessel occlusion. 189 In a second study, rodatristat ethyl did not significantly decrease mPAP but reduced pulmonary vascular occlusions. 190 Interestingly, synergistic effects were reported in both studies with a therapeutic combination of KAR5585 and the ET antagonist ambrisentan resulting in greater benefits on histological (vessel wall thickness/occlusion) and hemodynamic (mPAP) parameters than individual therapies.189,190 Again, combination therapy was more effective than monotherapy in reducing both pulmonary vascular remodeling and vessel occlusion, suggesting that when TPH1 inhibitors are combined with therapies directed toward vasodilation, an additive effect can be obtained on improving the symptoms and the pathophysiology of PAH.
Elastase inhibitors
Neutrophils are the dominant cellular source of neutrophil elastase (NE), but it is also produced by macrophages and VSMCs, 280 and elastase activity is associated with pulmonary vascular pathology by degradation and remodeling of the extracellular matrix. 157 There is growing interest in defining the importance of neutrophils and NE in the pathology of PAH, especially as neutrophil extracellular traps (NETs) and extensive NETosis are found in the plexiform lesions, which suggests a functional role for NE in PAH pathology. 281 Recent data have linked pulmonary vascular remodeling to the formation of NETs that are composed of chromatin fibers coated with the granular and cytoplasmic content from neutrophils such as myeloperoxidase, NE, and α-defensins. 281 Importantly, increased NETosis has been found in patients with PAH, particularly in the occlusive plexiform lesions and intrapulmonary thrombi of their small pulmonary blood vessels. 281
Elafin
Elafin, synthetized and secreted by the tracheobronchial epithelium, Clara cells, and alveolar type II epithelial cells, is a serine protease inhibitor expressed in lungs. 282 In a Su/Hx rat model of severe PAH that demonstrates a large increase in RVSP with extensive occlusions in the distal pulmonary arteries, SC administration of elafin for 2 weeks had protective effects against the hemodynamics and histopathology that was induced by the Su/Hx challenge. 157 Elafin is a potent antimicrobial and anti-inflammatory agent, 283 and in lung organ culture, elafin is proapoptotic and decreases neointimal lesions. 284 A phase I study of elafin in healthy subjects has been conducted, but additional studies will be required to determine whether the effects of elafin are due to inhibition of NE or to an increase in BMPR2 signaling on vascular endothelial cells. Nonetheless, the promising preclinical results support the development of elafin as a treatment for PAH to reverse obliterative vascular remodeling.
Inflammatory mediators
The contribution of inflammation to PAH pathogenesis has been previously reported in various preclinical studies, including the Su/Hx rat model and in several human studies.109,285,286 Gene expression of PAH-related inflammatory molecules, such as interleukin-6 (IL-6), monocyte chemoattractant protein-1, matrix metallopeptidase 9, cathepsin-S, and RANTES, is upregulated with the progression of pulmonary vasculopathy and differentially expressed in lungs of Su/Hx rats. 109 Targeting key inflammatory mediators such as tumor necrosis factor-α (TNF-α), IL-6, and leukotriene B4 (LTB4) ameliorated the development of PAH in the Su/Hx rat model.107,192,287
IL-6 receptor antagonists
Inflammation plays a major role in PAH pathobiology, 24 and the proinflammatory cytokine IL-6 is increased in the serum and lungs of patients with iPAH.288–290 Administration of IL-6 ligand and overexpression of IL-6 in mice lead to PH by promoting the development and progression of pulmonary vascular remodeling,289,291 while IL-6-deficient mice are protected from Hx-induced lung inflammation and pulmonary vascular remodeling. 188
Circulating levels of IL-6, IL-6 receptor (IL-6R), and the soluble form of the IL-6R (sIL-6R) expression are significantly increased in PASMCs and in the lungs of Su/Hx rats and daily treatment with IL-6R or the complex IL-6R/sIL-6R for 2 or 3 weeks prevented the development of PH as shown by a reduction of mPAP, Fulton index, and pulmonary vascular remodeling (wall thickness and muscularization). 192 Collagen deposition and inflammatory cell infiltration were also significantly decreased in lungs and RV of Su/Hx rats with the IL-6R/sIL-6R antagonist, 20S,21-epoxy-resibufogenin3-formate (ERBF) treatment. 192 The humanized monoclonal antibody tocilizumab (IL-6 receptor antagonist), approved for the treatment of rheumatoid arthritis, improved PAH symptoms in a patient with mixed connective tissue disease and severe PAH. 292 This result led to a phase II TRANSFORM-UK clinical trial for evaluation of the efficacy and safety of tocilizumab in patients with PAH. 293 However, IV tocilizumab (8 mg/kg) over 6 months in patients with PAH (NCT02676947) demonstrated no change in PVR or significant effects on exploratory second endpoints, including 6-MWD and NT-proBNP, despite evidence that the drug led to expected changes in biomarkers of target engagement in plasma with increased IL-6 and decreased C-reactive protein levels. 191
Tumor necrosis factor-α
Circulatory levels of inflammatory cytokines, including the key inflammatory mediator TNF-α, are elevated in familial and patients with iPAH. 290 Transgenic mice raised in Denver (CO, USA) and overexpressing TNF-α in the lung develop spontaneous PAH characterized by increased RVSP and RV hypertrophy. 294 In rat lung of monocrotaline-induced PAH, TNF-α is elevated, 295 and the soluble TNF-α inhibitor etanercept, administered preventively or therapeutically, significantly decreased mPAP by reducing inflammatory cell infiltration. 296 Therapeutic biweekly treatment by intraperitoneal injection of 2.5 mg/kg etanercept for 3 weeks in Su/Hx rats reversed PAH by reducing RVSP, RV hypertrophy, and muscularization of small arterioles, but surprisingly, this treatment had no effect on wall thickness. 287 This reduction of PAH with etanercept was associated with restored BMPR2, phospho-Smad 1/5, and Notch 3 expression. No study is currently investigating the safety and efficacy effect of etanercept in patients with PAH despite the positive results in the PAH rat models.
Leukotriene B4
LTs are important eicosanoid products of leukocytes and mediators of inflammation. LTB4, one of the LT groups, is produced from arachidonic acid metabolism through the 5-lipoxygenase (5-LO) enzymatic pathway and is implicated in several inflammatory diseases.297,298 5-LO expression is pronounced in pulmonary macrophages and PAECs of patients with iPAH and in Su/Hx lung tissue samples.107,299 LTB4, which induces VSMC proliferation and PAEC apoptosis, is significantly elevated in the bronchoalveolar lavage fluid of animals with PAH and in blood of patients with PAH.95,97 Prevention and interventional treatments with the nonspecific 5-LO inhibitor diethylcarbamazine (DEC) administered intraperitoneally every other day for 21 days (prevention) and for 2 weeks of 10 doses in total (intervention) ameliorated the development of PH, pulmonary inflammation, angio-obliterative remodeling (reduction in wall thickness and number of obliterated pulmonary arterioles) and reduced eicosanoid metabolites (LTC4, LTD4, hydroxyeicosatetraenoic acid (15-HETE, 12-HETE, 8-HETE) and 6-keto-prostaglandin F1 alpha (6kPGF1-α)) in Su/Hx rat lungs. 107 No current human studies of DEC appear to be in progress, but a phase II LIBERTY trial (NCT02664558) in patients with severe PAH treated with ubenimex (sold in Japan as Bestatin), an inhibitor of LTA4 hydrolase that blocks downstream the production of LTB4, failed to demonstrate efficacy. 298
Antiproliferative compounds
Vascular remodeling, a hallmark pathological feature of PAH, is characterized by endothelial dysfunction, proliferation of PASMCs and/or PAECs, extracellular matrix and collagen deposition, and perivascular inflammatory infiltrates, all of them contributing to PA remodeling in PAH.10,24 The dominant effect of compounds in this class of antiproliferation is inhibition of the increase in wall thickness, proliferation of VSMCs and PAECs, and mesenchymal transition of PAECs. Some of these new targets affect cardiac tissue to inhibit cardiomyocyte disarray and right heart fibrosis formation.
TK inhibitors
Many of the TKs targeted for PAH involve the actions of cell surface receptors such as platelet-derived growth factor (PDGF), epidermal growth factor (EGF), fibroblast growth factor (FGF), VEGF, and nerve growth factor. These compounds are effective anticancer agents18,300 but are associated with AEs. 301 Some of these TK inhibitors have been evaluated in Su/Hx rats,107,148,149 and although some of these have advanced into clinical trials (Nilotinib—NCT01179737; Imatinib—NCT00477269, NCT00902174, NCT01392495; GB002—NCT04456998, NCT03926793, NCT04816604),145,148,150,302–305 others have been discontinued due to serious AEs. 306
GB002-PDGFR inhibitor
GB002 (seralutinib, Gossamer Bio Incorporated, San Diego, CA, USA) is a novel potent small-molecule kinase inhibitor targeting PDGFRα/β, colony-stimulating factor 1 receptor, and the tyrosine kinase receptor, and upregulating BMPR2 protein expression. 145 In the phase II, TORREY study (NCT04456998), administration of seralutinib (90 mg) by dry powder inhaler twice daily (BID) for 24 weeks was well tolerated, significantly reduced PVR and NT-proBNP, and significantly improved 6-MWD. 146 Inhaled seralutinib also had a significant effect on pulmonary vascular remodeling by redistributing the pulmonary arterial blood vessel volumes to smaller vessels. 147 A phase III study (NCT05934526) has been initiated based on these results. The nonselective PDGFR inhibitor PK10453 administered by inhalation was originally developed by Pulmokine (Rensselaer, NY, USA) and showed positive activity in rats challenged with monocrotaline or monocrotaline plus pneumonectomy. 307 GB002 has been evaluated in Su/Hx rats where it reduced the mPAP and RVSP and attenuated the increase in pulmonary arteriole muscularization induced by the Su/Hx challenge.148,149 Importantly, there was restoration of BMPR2 protein expression in the lungs of GB002-treated animals along with parallel decreases in plasma levels of NT-proBNP and platelet-derived growth factor (PDGF-BB), and a decrease in proinflammatory cytokines.148,149 These studies show that inhibitors of the PDGF pathway are viable targets for PAH and strategies such as delivery by inhalation may help to minimize side effects, including cardiotoxicity, that are present with drugs in this class.301,306
AGI296-PDGFR inhibitor
A novel approach for drug discovery in PAH involves the use of induced pluripotent stem cell-derived endothelial cells (iPSC-ECs) generated from patients with PAH. This approach can help to determine the molecular and genetic mechanisms of PAH, to predict the pharmacological profile of a compound in a patient, and finally to predict the response of the patient to this compound. iPSC-EC from six patients with PAH were exposed to 4500 compounds for phenotypic drug screening, 308 and in silico analyses of transcriptomic datasets revealed that the compound AG1296, an ATP-competitive selective inhibitor of PDGF receptors, was associated with an anti-PAH gene signature. AG1296 (50 mg/kg), administered SC, reduced pulmonary arterial neointimal lesions in lung organ culture and almost totally reversed pulmonary arterial occlusion in the Su/Hx rat model. 308 AG1296 also significantly decreased RVSP, and trends were reported for reduced RV hypertrophy and increased CO. 308
MAZ51-VEGF inhibitor
There are three VEGF receptors (VEGF-1, VEGF-2, and VEGF-3) that mediate the pharmacological effects of the different VEGF ligands. 309 MAZ51 (Virginia Commonwealth University, Richmond, VA, USA) is a selective inhibitor of the VEGF-3 receptor. Daily SC administration of MAZ51 at 8 mg/kg reduced the increase in RVSP, RV hypertrophy, and obliteration of pulmonary blood vessels when administered immediately after the Su/Hx challenge but worsened the severity of the PAH pathology when delivered several weeks later. 107 These results indicate a minor role for VEGF inhibition as a therapy for the treatment of PAH because of its limited efficacy and its potential to aggravate PAH pathology if administered when the disease is already established.
Imatinib
The PDGF receptor inhibitor imatinib (Novartis Pharma AG, Basel, Switzerland) is an antiproliferative agent used to treat chronic myeloid leukemia and gastrointestinal stromal tumors.310,311 IV administration of imatinib in Su/Hx rats showed some effects on RVSP at high doses (20 and 50 mg/kg) but not at a low dose of 5 mg/kg, which is equivalent to the clinical dose (400 mg/day) used for the treatment of patients with leukemia and PAH. 151 Following promising results in several case reports,312–314 clinical studies (NCT00477269, QT1571, NCT00902174) with oral imatinib in patients with PAH showed effects on hemodynamics and walk distance,150,302,303 but serious AEs and safety issues precluded the approval by FDA of imatinib in the treatment of severe PAH.150,315 Despite these concerns, a phase II clinical study (NCT 04416750) is underway to reevaluate imatinib for PAH treatment. 316 Recently, an inhaled formulation of imatinib AER-901 (Aerami Therapeutics, Durham, NC, USA), which specifically targets the lungs and may reduce the side effects of the oral formulation, has been granted orphan drug designation by the FDA. A phase I clinical trial (NCT04903730) was completed in December 2022, 150 and Aerami plans to advance AER-901 into a phase II trial.
Sorafenib
Sorafenib (Nexavar, Bayer Leverkusen, North Rhine-Westphalia, Germany) is a kinase inhibitor drug approved for the treatment of several cancers, including renal cell carcinoma and advanced primary liver cancer. 317 It is an activator of adenosine monophospate-activated protein kinase (AMPK) and it has inhibitory activity against many protein kinases, including VEGF and PDGF receptors. 317 A study in Su/Hx rats showed inhibitions of the changes in pulmonary vascular hemodynamics and the pulmonary vascular remodeling induced by the Su/Hx challenge.40,318 In a 16-week small exploratory phase IB study with 12 patients with PAH receiving continuous administration of IV prostacyclin analog with or without sildenafil (NCT00452218), oral administration of sorafenib significantly improved exercise capacity (6-MWD) but did not ameliorate CO. 152 We must note that cardiac toxicity with TK or multikinase inhibitors is a serious concern, and cardiac ischemia, left ventricular dysfunction, and hypertension have been reported. 319 However, in a more recent small clinical study of patients with severe PAH and/or RHF, 153 pulmonary hemodynamics (PAP and RVSP) were improved with sorafenib. Moreover, no cardiac toxicity was reported in the nine patients refractory to medical therapies, including epoprostenol, sildenafil, and bosentan.
GS-444217
Oxidative stress is greatly reinforced in patients with PAH with increased activity of ROS, driving pathological pulmonary vascular and right ventricular remodeling through activation of mitogen-activated protein kinases (MAPKs) such as p38 and c-Jun N-terminal kinase (JNK).155,320 Oxidative stress activates apoptosis signal-regulating kinase 1 (ASK1), a constitutively expressed MAP3K that promotes p38 and JNK activation. The ASK1 inhibitor GS-444217 (Gilead Sciences, Foster City, CA, USA), also known as MAP3K5 inhibitor, administered orally in the Su/Hx rat model, dose-dependently reduced PAP, RV hypertrophy, muscularization of the pulmonary arteries, and fibrotic gene expression in the RV. These effects were similar to the effects of the PDE5 inhibitor sildenafil, but the efficacy of ASK1 inhibition was probably due to reductions in structural remodeling of the pulmonary vasculature and not due to vasodilation. 155 In a second Su/Hx rat study, Wilson et al. 156 confirmed that GS-444217 reduced pulmonary vasculature and lung vascular remodeling of the RV. This beneficial effect was a result of a decrease in fibroblast activation, migration, proliferation, and cytokine release. However, the first-in-class ASK1 inhibitor selonsertib (GS-4997, Gilead Sciences, Foster City, CA, USA), administered once daily for 24 weeks, did not lead to a significant reduction in the primary endpoint (PVR) or secondary endpoints (6 MWD, RV function, and the cardiac biomarker NT-proBNP) in a phase II clinical trial for patients with PAH (ARROW trial NCT02234141). 154
BMPR2 activators
BMPR2 is a serine/threonine receptor kinase, highly expressed in the vascular endothelium, that forms a complex with the coreceptors ALK1, ALK2, and ENG. 321 It is activated by circulating ligands of the transforming growth factor-β (TGF-β) super family, including brain morphogenetic protein 9 (BMP 9) and BMP 10. 322 Mutations in BMPR2 signaling occurs in a high percentage (70%–80%) of familial PAH but also in 10%–20% of patients with iPAH.12,70,71,322 BMPR2 signaling controls endothelial cell dysfunction, endothelial cell-to-mesenchymal cell transition, proliferation and differentiation of VSMC, and collagen deposition in cardiomyocytes.57,70,71,323 BMPR2 signaling has been strongly linked to the pathobiology of PAH leading to the development of numerous therapeutic strategies to increase BMPR2 activity.12,70–72,322 Some of the compounds developed in this drug class have progressed into clinical trials (FK506—NCT01647945; ABI-009—NCT02587325; sotatercept—NCT03496207, NCT04896008), and compounds in development have been evaluated in Su/Hx rats.124,128,324–326 Details of the activity of some of these compounds are described below.
Tacrolimus
Tacrolimus (FK506, Stanford University, Stanford, CA, USA) stimulates BMPR2 signaling via inhibition of calcineurin and release of the BMPR2 repressor FK506-binding protein 12 (FKBP12), a repressor of BMP signaling. FK506 was superior to the calcineurin inhibitor cyclosporine in vitro assays and to the FKBP12 ligand rapamycin in potentiating BMP in PAECs. 57 Furthermore, FK506 is a potent inhibitor of the mammalian target for rapamycin complexes 1 and 2 (mTORC1, mTORC2) that are downstream signaling proteins for the phosphoinositide 3-kinase/Akt/mTOR (PI3K/AKT/mTOR) pathway, an important signaling cascade involved in cell proliferation, survival, and growth. 327 Positive results have been demonstrated in early clinical trials in PAH subjects with both FK506 (NCT01647945)124,125 and ABI-009, an mTOR inhibitor (nab-sirolimus) bound to albumin in lipid nanoparticles to promote retention in the lungs (NCT02587325). 328 Tacrolimus almost completely reversed the pathophysiological changes induced by Su/Hx challenge in rats after only 3 weeks of therapeutic oral administration (8 weeks after Su injection) when the full pathology of the disease had developed. 57 In mice subject to pulmonary arterial banding, FK506 increased RV capillarization, reduced fibrosis formation, and improved cardiac functions, suggesting that FK506 has effects on cardiac pathophysiology separate from its effects on the pulmonary vasculature. 323 There appears to be an excellent translation between these positive results with low-dose oral tacrolimus in Su/Hx rats and early results in clinical trials. Importantly, preclinical data show a positive interaction with mTORC1 signaling and activation of the DP1 prostanoid receptor, 329 which suggests that a combination therapy of tacrolimus and a prostanoid DP1 receptor agonist such as trepostinil or TP may have added benefit and enable a further reduction in the dose of tacrolimus to minimize its side effect potential.
Rapamycin
Although rapamycin (Rapamune, Wyeth Pharmaceuticals-Pfizer, New York, NY, USA) is classified as an mTOR inhibitor, it has a different mechanism of action compared with tacrolimus, as it is not a calcineurin inhibitor, but it inhibits IL-2-driven T cell proliferation and other cytokine-receptor signal transduction mechanisms via an action on mTOR. 330 Through this mechanism, rapamycin blocks the activation of T and B cells, and it directly binds to the mTOR complex to release the FKBP12 binding protein and to activate BMP signaling.330,331 IT administration of rapamycin, 3 times per week for 5 weeks following Su injection and 3 weeks of Hx, showed protective effects in Su/Hx rats with severe PAH by reducing RVSP, RV hypertrophy, and by ameliorating arterial remodeling and autophagy activation in endothelial cells. 127 The protective effects of rapamycin likely involve activation of the autophagic pathway, as there were increased levels of microtubule-associated light chain-3 protein, a key biomarker of the autophagic pathway, in both PAECs and pulmonary blood vessels. 127 Rapamycin (1 mg, BID), administered for 8 weeks to a patient with pancreatic cancer, liver metastases, and severe PAH symptoms, resulted in a substantial improvement not only in the sizes of the pancreatic mass and hepatic metastases, but also in the 6-MWD, RV function, and RV size. 126 Recently, an intraperitoneal combination therapy for 2 weeks with rapamycin (2 mg/kg) and a low dose (10 mg/kg) of the TK inhibitor imatinib resulted in greater attenuation of RVSP, Fulton index, thickening of pulmonary vascular wall, and muscularization of pulmonary vessels when compared with either drug alone in the Su/Hx rat model. 332
Sotatercept
Sotatercept (ACTRIIA-FC, Acceleron-Merck, Kenilworth, NJ, USA) is an immunoglobulin G Fc domain fusion protein that is a ligand trap for ALK A/B (ACTRIIA or ACTRIIB) and the growth and differentiation factors (GDFs) 8/11. Both ACTRIIA and ACTRIIB combine with ALK4, ALK5, or ALK7 to preferentially activate SMAD2/3 that possess pro-proliferative, promigratory, and proangiogenic effects on PAECs and PASMCs. 39 Furthermore, ALK and guanosine diphosphate ligands share receptors in common with BMP and TGFβ, and by sequestering these ligands may yield a more complete coverage to treat PAH pathology. 39 To test this hypothesis, Su/Hx-challenged rats were treated with ACTRIIA-FC in different experimental paradigms that induced moderate-to-severe PAH pathology. 39 Almost complete abrogation of the hemodynamic, pulmonary vascular remodeling and increase in right heart size was reported with ACTRIIA-FC administered intraperitoneally at doses of 1, 3, and 10 mg/kg but was less pronounced in Su/Hx rats with severe PAH pathology that involved the formation of plexiform lesions. 39 Importantly, ACTRIIA-FC was superior to oral sildenafil in Su/Hx rats with moderate and severe pathology in both studies.39,130 Sotatercept advanced into clinical studies for efficacy and safety (phase II) where a reduction in PVR was observed in patients receiving therapy for PAH with World Health Organization Functional Class (WHO FC II/III) in the PULSAR study 128 (NCT03496207, and where an increase in peak O2 uptake (VO2 max), and a decrease in PAP was reported in patients with WHO FC III in the SPECTRA study (NCT03738150). 129 Sotatercept was further investigated in an extensive phase III clinical program that comprised four studies to establish efficacy and safety across a broad spectrum of patients with PAH (Clinical trials),131,132 including patients with WHO FC II/III in the STELLAR study (NCT04576988), patients with WHO FC II/III in the HYPERION study (NCT04811092), patients with WHO FC III/IV at high-risk mortality in the ZENITH study (NCT04896008), and for a long-term safety/tolerability in the SOTERIA study (NCT04796337). Positive results from the phase III STELLAR trial showing improvements in 6-MWD, PVR, and NT-proNTB level 326 led to the recent approval by the FDA of sotatercept under the brand name of Winrevair. It should be noted that another selective TGF-β ligand trap, the selective immunoglobulin-Fc fusion protein of TGFβ, also improved PH and vascular remodeling in preclinical PH models (Su/Hx and monocrotaline). 333
Hypoxia-inducible factor inhibitors
Hypoxia-inducible factor (HIF) is a transcription factor that drives the biological responses to oxygen deprivation but may also play a role to optimize oxygen delivery to normoxic tissue for maintenance of the alveolar architecture.334,335 HIF consists as a heterodimeric complex, composed of the oxygen-sensitive α subunit (HIF-1α, HIF-2α, and HIF-3α) and an oxygen-insensitive β subunit (HIF-β). HIF has been implicated as an important regulator of PAH pathology with relatively high levels of HIF-1α found in PAECs, PASMCs, and RV. 334 Levels of both HIF-1α and HIF-2α are elevated in patients with PAH 336 and in rodent models of PAH, including Su/Hx rats,43,337 where pharmacological studies with HIF inhibitors show a profound effect on most of the pathobiology of the disease. 334
Celastramycin
Following the screening of 5562 compounds for inhibition of PASMC proliferation, celastramycin (Tohoku University, Sendai, Miyagi, Japan), an HIF-1α inhibitor, was identified as the most potent compound and was evaluated in 3 rodent models of PAH, including Su/Hx rats. 338 Intraperitoneal administration of celastramycin reduced the increased mPAP, RVSP, PVR, and right heart size, improved pulmonary vascular remodeling (pulmonary vascular wall thickness) and cardiac functions (TAPSE and CO), and enhanced walking distance in Su/Hx rats. Mechanistic studies also showed a decrease in the protein levels of HIF-1α in PAECs by celastramycin, providing a strong link of efficacy with HIF inhibition. 338
HIF-2α translational inhibitor (compound 76)
The predominant role of HIF-2α versus HIF-1α activation in the pathogenesis of PAH was previously demonstrated 43 and compound 76 is a selective translational inhibitor of HIF2-α, acting at the level of the RNA. 339 Intraperitoneal administration of compound 76 attenuated the increase in RVSP, improved pulmonary artery diastolic function, and markedly reduced the increases in wall thickness and percentage of occlusive pulmonary blood vessels in Su/Hx rats. 43 Compound 76 also showed beneficial effects on cardiac pathology with a reduction in right heart size measured by the Fulton index and RVAWT, and a decrease in collagen deposition in the RV. These results were also demonstrated in a severe PAH mouse model (Elgin1Tie2Cre mice) 340 and in the monocrotaline-exposed rat model of PAH. 43
PT2567
Peloton Therapeutics (UT Southwestern Medical Center, Dallas, TX, USA), acquired by Merck (Rahway, NJ, USA), developed PT2977, a novel HIF2α inhibitor. Prevention and intervention strategies with PT2977 in the Su/Hx rat model reversed pulmonary vascular hemodynamics (RVSP, PVR, Fulton index) and pulmonary vascular remodeling (smooth muscle hypertrophy, vessel muscularization and small arterial occlusion, perivascular/vascular inflammation, and vascular fibrosis), and decreased circulating proinflammatory cytokines (TNF-α) and the expression of PAH-associated genes in lung and cardiac tissues. 341 The PDE5 inhibitor sildenafil demonstrated only a small beneficial effect in both prevention and intervention studies. 341
Taken together, these results with compound 76, celastramycin, and PT2977 illustrate that inhibition of both HIF1-α and HIF2-α provides protective effects against PAH pathology in Su/Hx rats. Combination studies with HIF1-α and HIF2-α inhibitors have not been performed to demonstrate if synergy exists for improvement in this PAH pathology.
PPAR inhibitors
PPARs are a family of three nuclear receptors (PPARα, PPARβ/δ, and PPARγ) that affects many biological processes, including lipid and energy metabolism, epidermal wound repair, inflammation, and atherosclerotic plaque formation. 208 PPARγ has been implicated as a therapeutic target in PH, 210 although both the PPARβ/δ and PPARγ subtypes may also have a role on PAH pathology via interactions with the prostacyclin analog.208,342,343 Two PPARγ agonists, rosiglitazone (Avandia, GlaxoSmithKline, London, UK) and pioglitazone (Actos, Takeda Pharmaceutical, Osaka, Japan), are currently approved for the treatment of type 2 diabetes by reducing insulin resistance and improving glycemic control, but fluid accumulation and cardiac effects limit the prolonged use of these drugs.344,345
Pioglitazone
In a Su/Hx rat study, oral pioglitazone fully reversed the changes in pulmonary vascular hemodynamics, right heart size, and echocardiography parameters TAPSE and PAAT after Su/Hx challenge, with significant effects also on muscularization, obliteration, and plexiform lesion formation in the pulmonary arteries. 97 Improvements after pioglitazone treatment were also noted in the RV with a decrease in collagen content, and mitochondrial disarray, and a reversal of intramyocellular lipid accumulation. Importantly, pioglitazone produced these effects through microRNA (miRNA)/messenger RNA (mRNA) networks (pre-mIR-197 and premIR-146b) present in tissues from human end-stage PAH. 97 An early clinical trial to evaluate the incidence of insulin resistance in patients with PAH with pioglitazone was terminated due to the difficulty in finding eligible subjects (NCT00825266). It will be critical to compare these impressive results in Su/Hx rats with its efficacy in clinical trials given the fact that the clinical dose will be limited because of the significant toxicity associated with this drug class. 344
Rosiglitazone
Rosiglitazone (Avandia, GlaxoSmithKline Pharmaceuticals, Brentford, UK) is also an oral antidiabetic drug in the thiazolidinedione class. Due to the increased risk of heart problems, the drug is no longer available in Europe and is rarely used in the United States. However, delivery of this PPARγ agonist directly to the lung has been performed in the Su/Hx rat model. IT administration of PLGA-based particles of rosiglitazone (0.3 mg/kg) produced pulmonary vasodilatation over an extended period of time (6 hours) in the Su/Hx rats, while the same IT dose of free rosiglitazone reduced mPAP by ∼30% within 15 minutes. Oral rosiglitazone had no vasodilatory effect. These results demonstrate the superiority of lung delivery over oral administration for the treatment of PAH and poor absorption from the gastrointestinal tract for the oral drug. 243 This prolonged reduction of mPAP by the rosiglitazone particles is due to the localized availability and slow release of rosiglitazone in the pulmonary vasculature. Also, a combination therapy comprising PLGA particles of two distinct drugs, the PDE5 inhibitor sildenafil and the PPARγ agonist rosiglitazone, at much lower doses than that of the human doses, caused a pronounced reduction in mPAP, improved cardiac function by increasing CO and lowering total PVR, decreased right heart remodeling, and disease progression by reducing collagen deposition and muscularization of the pulmonary arteries in the Su/Hx rat model. 36 Inhaled combination therapy could be a viable alternative to currently available vasodilator-based combination therapy for PAH. 36
β-hydroxy β-methylglutaryl coenzyme A reductase inhibitors (statins)
Statins, β-hydroxy β-methylglutaryl coenzyme A reductase inhibitors, are clinically approved compounds used to lower cholesterol, but based upon their antiproliferative, antithrombotic, anti-inflammatory, and antioxidant effects, statins were thought to also offer a potential novel approach to the treatment of PAH.158,346 However, while statin treatment appears to be safe, it may have no effect on PAH. 347 Simvastatin showed early promise in a PAH clinical trial (NCT00180713) reducing RV mass measured by cardiac MRI and NT-proBNP, but the slight improvement in 6-MWD did not achieve statistical significance and the beneficial effects of simvastatin were not sustained over 12 months. 158 Likewise, in a separate clinical trial with atorvastatin (NCT00615823), the primary endpoint of a change from baseline in the 6-MWD was not met. 348 On the basis of these results, interest in the development of statins for the treatment of PAH has dwindled. However, statins (simvastatin) have been evaluated in Su/Hx rats.
Simvastatin
Treatment of Su/Hx rats with oral simvastatin markedly reduced the increase in mPAP and RV hypertrophy that was associated with caspase-3 activation and microvascular endothelial cell apoptosis. 346 There was also CAV1, CAV2, and phospho-CAV restoration in the pulmonary blood vessels and a reduction in Rho kinase activity. These positive results in the Su/Hx rat model were not confirmed in clinical trials when simvastatin was given for 6 months or over a 12-month period,158,159 suggesting that tolerance to repeat dosing with simvastatin occurs in PAH subjects.
Antifibrotic compounds
Both nintedanib (OFEV, Boehringer Ingelheim Pharmaceuticals, Ridgefield, CT, USA) and pirfenidone (Esbriet, Genentech-Hoffman La Roche, South San Francisco, CA, USA), two antifibrotic agents approved for the treatment of idiopathic pulmonary fibrosis (IPF), have been tested in Su/Hx rats.160,162,349,350
Conflicting results were observed with the multiple TK inhibitor nintedanib, which targets PDGF, VEGF, FGF receptors, and the Src family of protein TKs (nonreceptor TKs). Nintedanib did not improve pulmonary hemodynamics or right heart function (no effects on RVSP, PVR, CO, RV hypertrophy, muscularization, and occlusion of pulmonary arterial vessels) in Su/Hx W-K rats. 160 Nintedanib did not affect pulmonary vascular remodeling but improved RV adaptation with amelioration in cardiac contractility, decrease in RV dilation, and reduction in the RV hypertrophy and collagen content in Su/Hx rats, 349 while nintedanib improved hemodynamics and reduced vascular remodeling involving neointimal lesions and medial wall thickening in the study of Tsutsumi. 350 In patients, nintedanib at 100 mg BID in two patients with PAH and at 150 mg in two other patients with PAH decreased the cardiac index (CI), accompanied by an increase in PVR, 6–9 months after starting treatment. 160 Nintedanib failed to improve pulmonary hemodynamics and right heart function in the Su/Hx rat model and in four patients with severe PAH and caused deterioration of these variables in the clinical setting, indicating that nintedanib may have deleterious effects in PAH. 160
Treatment with pirfenidone had beneficial effects on pulmonary vascular remodeling with reductions in PASMC proliferation (in Su/Hx rats and in tissues from patients iPAH) and in collagen deposition in the RV of Su/Hx rats but had no effects on mPAP and the Fulton index. 162
These results demonstrated contrasting effects of nintedanib and pirfenidone in Su/Hx rats, and these compounds have not been advanced as monotherapy into clinical trials in PAH subjects. Only one clinical study (phase IIB trial; NCT02951429) investigated the use of pirfenidone in combination with sildenafil in patients with advanced IPF and risk of PH, but this combination did not provide any benefit versus pirfenidone alone after up to 52 weeks of treatment. 161
Neurohormonal modulators
With elevated PAP and PVR, PAH is also characterized by progressive pulmonary vascular remodeling and increased RV afterload, which ultimately leads to RHF and death. 351 Indeed the high demands on the RV cause changes in RV contractility, RV hypertrophy, fibrosis, ending up in RV failure and death, 352 and RV dysfunction is the strongest predictor of mortality in PAH. 353 Approved PAH medications are predominately pulmonary vasodilators that modulate the ET, NO, and prostacyclin pathways as discussed above, and although these therapies primarily address the vasoconstrictive phenotype of PAH, therapies targeting RV failure are not widespread. Cardiovascular autonomic dysfunction characterized by increased SNS activity and parasympathetic nervous system (PNS) withdrawal is associated with disease progression and poor prognosis in PAH. 354 Neurohormonal regulation plays a major role in pulmonary vascular tone and RV function, 110 and increased neurohumoral activation during PAH supports the use of β-blockers, aldosterone antagonists, or acetylcholine esterase inhibitors. Clinical trials on neuronal modulation in PAH are listed in the review from Vaillancourt et al. 355
β-adrenergic blocker
In patients with PAH, RV dysfunction is associated with reduced PNS activity, while SNS activation is markedly augmented and associated with increased mortality.354,356 RHF is the leading cause of death in PAH, suggesting that the dysfunctional RV represents a target for β-blockers to maintain RV function and could be useful in patients with PAH and RV dysfunction. 357
Carvedilol
Carvedilol is a nonselective α1-/β1-/β2-adrenoceptor blocker marketed as Dilatrend by F. Hoffmann-La Roche Ltd. (Basel, Switzerland) and as Coreg by GlaxoSmithKline (Brentford, UK) for the treatment of hypertension, angina pectoris, postmyocardial infarction, and chronic heart failure. Carvedilol administered orally and daily for 4 weeks in the Su/Hx rat model had no effect on pulmonary hemodynamics (RVSP), RV hypertrophy, RV remodeling (RV wall thickness and RV cross-sectional area), and RV function, 167 confirming the absence of effect on RVSP and pulmonary vascular remodeling reported in a previous study. 168 However, while carvedilol improved the RV function in the study of Boogard et al., 168 no RV beneficial effect was reported in Zelt’s study although the dose (15 mg/kg/day) and duration of treatment (4 weeks) were similar in both studies. In patients, treatment with carvedilol in a small pilot study (NCT00964678) significantly improved the RV ejection fraction and SV without changes in exercise capacity (6-MWD) or in the marker BNP for ventricular failure, 6 months after starting treatment. 163 In a double-blind, randomized, controlled trial (PAHTCH, NCT01586156), carvedilol at the same dose and duration of the pilot study increased RV function but did not improve CO or exercise capacity. 164 Also, no statistical differences in survival or time to clinical worsening were observed in a large PAH cohort with the use of β-blockers, including carvedilol.165,166 Overall, there is no clear evidence for using β-blockers to target the SNS in patients with PAH confirming the absence of positive findings in preclinical animal studies using the Su/Hx rat model.
Renin–angiotensin–aldosterone inhibitors
The primary function of the RAAS is to maintain blood pressure, but the RAAS also plays a major role in the regulation of inflammation, proliferation, and fibrosis in pulmonary diseases, and this system is activated in patients with PAH. 358 Renin, a protease protein and enzyme secreted by the kidneys, circulates in the bloodstream and cleaves the hormone angiotensinogen synthesized and secreted mainly by the liver into the peptide angiotensin I. 359 Circulating angiotensin I is then hydrolyzed to angiotensin II by the angiotensin-converting enzyme (ACE), which is located primarily in the pulmonary and renal endothelium. Angiotensin II activates the angiotensin type 1 (AT1) receptor to initiate a vasoconstrictor response and to stimulate aldosterone synthesis by the adrenal glands. 359
Aldosterone
The steroid hormone aldosterone that binds to mineralocorticoid receptors is present in the heart and pulmonary vasculature and exerts multiple physiological effects such as cardiac fibrosis and activation of the SNS. 360 In the Su/Hx rat model, treatments with the selective steroidal mineralocorticoid receptor antagonist eplerenone (second generation) initiated simultaneously with Su injection, or with the nonselective steroidal mineralocorticoid receptor antagonist spironolactone (first generation) delivered 14 days after Su injection, reduced pulmonary pressures, collagen deposition, and pulmonary vascular remodeling. 170 However, when administered after the onset of RV dysfunction (after 3 weeks of Hx and 2 weeks of Nx), neither of these mineralocorticoid receptor antagonists significantly affected PAP or vessel remodeling in Su/Hx rats. 172 A first-in-class nonsteroidal third generation of mineralocorticoid receptor antagonist finerenone, with higher selectivity and stronger affinity for the mineralocorticoid receptor, applied orally after Su injection decreased PAP and the Fulton index, improved CO, and attenuated pulmonary vascular remodeling (wall thickness and muscularization of pulmonary arteries) in Su/Hx rats. 361 The effect of mineralocorticoid receptor antagonism was evaluated in several clinical trials (NCT01468571; NCT01712620), 171 and a crossover study (NCT01468571) in patients with PAH showed that spirolactone was safe and well tolerated but did not change collagen metabolite levels or the 6-MWD. 169 A phase IV study, in patients with RHF secondary to PAH (NCT03344159, STAR-HF), may determine if oral treatment with spironolactone causes a reduction in RV wall stress or change in cardiac sympathetic activity.
LCZ 696
A novel approach based on a combination of angiotensin receptor blocker and neprilysin inhibitor was developed to target natriuretic peptide (NP) enhancement and RAAS blockade. This optimized heart failure treatment by acting on both pathways (NPs and RAAS). The first-in-class angiotensin receptor and neprilysin inhibitor, LCZ696, is composed of two molecular moieties of the angiotensin receptor blocker valsartan (RAAS blockade) and of the neprilysin inhibitor prodrug sacubitril (inhibition of NP degradation), attenuating the damaging effects of RAAS, and augmenting the positive effects of the NPs that lead to vasodilation.362,363 Oral LCZ696 treatment QD for 5 weeks in the Su/Hx Wistar rats reduced the increases induced by Su/Hx challenge of RVSP, RV hypertrophy, RV dilatation, and wall thickness of the small pulmonary arteries. 175 Also, oral gavage with LCZ696 for 6 weeks significantly decreased RVSP, RV hypertrophy and fibrosis, and vascular remodeling (wall thickness only), and significantly increased NPs in serum and lung tissues of Su/Hx S-D rats, 176 but valsartan administered orally did not affect the pulmonary pressures, RV hypertrophy, or vascular remodeling, 176 confirming the absence of effects on mPAP and RV hypertrophy with the ACE inhibitor lisinopril and the AT2 receptor blockade ibesartan in Su/Hx rats. 346 In humans, LCZ 696 improved the pulmonary hemodynamic profile (mPAP and PVR) in two PH patients due to left heart disease and in five PH patients with advanced heart failure.173,174 These preclinical and clinical results support the evaluation of this novel dual therapy in human PAH.
Autonomic system
Vagal nerve stimulation (VNS) is another approach to alter the parasympathetic activity. Activating the parasympathetic nerve by VNS had beneficial effects in animal models for cardiovascular diseases, 364 and in the Su/Hx model using Fischer rats, a strain prone to RHF, VNS decreased PAP, attenuated pulmonary vascular remodeling, preserved RV function, improved survival, and reduced cytokines and pathological markers of inflammation. 364 However, although clinical trials have shown the safety of VNS, the efficacy of VNS in patients with heart failure remains controversial due to the high precision required for this procedure to avoid the potential for unwanted cardiac and noncardiac effects of VNS.365,366
However, a new approach, the focused ultrasound stimulation of the spleen (sFUS), for activating the neuroimmune pathway has emerged. 367 sFUS is a noninvasive method using ultrasound that was first applied to the spleen to modulate the cholinergic anti-inflammatory pathway. 367 In the Su/Hx rat model, sFUS treatment reduced RVSP, wall small pulmonary arteriole thickness, RV fibrosis, and hypertrophy, improved RV function, stopped lung inflammatory cell infiltration, lowered BNP serum levels, and downregulated several inflammatory genes and pathways involved in human PAH. 368 These effects persist for weeks after sFUS treatment discontinuation.
Pulmonary arterial denervation
Increases in PAP and PVR induced by balloon distension of the pulmonary artery in dogs or by IV infusion of the thromboxane A2 in pigs were reduced by surgical or percutaneous pulmonary artery denervation (PADN).177,369,370 PADN led to improvements in pulmonary hemodynamics (PAP and PVR), exercise capacity (6-MWD), and cardiac function in patients with PAH (chiCTR-ONC-12002085, NCT02220335).178,179 In the Su/Hx-induced PAH model, complete denervation of the pulmonary artery by the transthoracic pulmonary artery denervation (TPADN) procedure attenuated PAH progression. 180 PAP, RVSP, RV hypertrophy, medial wall thickness, pulmonary artery muscularization, and RV fibrosis were significantly reduced by TPADN. 180
Renal denervation
Another invasive strategy acting on the autonomic nervous system is the catheter-based renal denervation to disrupt the renal sympathetic nerves. Renal denervation improved pulmonary hemodynamics, RV, and pulmonary vascular remodeling in the monocrotaline dog and in rat models.371,372 In the Su/Hx rats, renal denervation reduced PVR, RV, and pulmonary vascular remodeling, RV hypertrophy, and fibrosis, but did not significantly affect RVSP, CO, RV size, or systolic function. 373 No clinical trials have been performed to treat PAH via this strategy, but renal denervation lowered blood pressure in hypertensive patients resistant to treatment in one trial, 374 while in another trial (SYMPLICITY HTN-3), no efficacy was reported in patients with uncontrolled hypertension when compared with antihypertensive therapy alone. 375
Epigenetics and modulators of DNA
Epigenetic is defined as a heritable change to the chromatin in the genome resulting in a change of gene expression without affecting the DNA sequence. 376 There are three major layers of epigenetic modifications: DNA methylation, histone modifications, and noncoding RNAs that regulate the expression of genes at the transcriptional and posttranscriptional level.
Histone modification
The principal components of chromatin, the histones, are highly evolutionarily conserved proteins. Histone posttranslational modifications such as acetylation and methylation can activate or repress gene expression. 377 Histone acetylation is mediated by the histone acetyltransferases but is also regulated by histone deacetylases (HDACs) that remove acetyl groups from histone proteins and represses DNA transcription. 377 A total of 18 human HDAC enzymes have been identified and grouped into four classes based on functional criteria. 378
In the Su/Hx model, the class I and II HDAC inhibitor trichostatin A had no effect on RVSP, Fulton index, and pulmonary vascular remodeling, and did not improve cardiac function. 379 HDAC inhibition by trichostatin A or valproic acid was harmful to the heart in the pulmonary artery banding rat model, a model that only induces RV hypertrophy. 380 However, quisinostat (JNJ-26481585), a second-generation HDAC inhibitor, highly potent against class I and II HDACs, decreased proliferation and inflammatory cytokine expression in PAECs, reduced RVSP and PVR, reversed vascular remodeling, and improved RV function in the Su/Hx rat model.381,382
The transcriptional activity of the transcription factor, myocyte enhancer factor 2 (MEF2), is decreased in PAECs of patients with PAH, and this reduction was associated with increased nuclear accumulation of two class IIa HDACs: HDAC4 and HDAC5. 383 In Su/Hx rats, increasing MEF2 activity with a selective class IIa HDAC inhibitor, MC1568, significantly reduced RVSP, Fulton index, muscularization, and obliteration of pulmonary arterioles without worsening RV remodeling and fibrosis, 383 as previously reported with nonselective HDAC inhibitors. 380
More recently, tubastatin A (Tub A), a class IIb HDAC6 inhibitor significantly improved mPAP, RVSP, PVR, and vascular remodeling (wall thickness) and induced apoptosis of PASMCs in Su/Hx rats. 384 These therapeutic effects of Tub A were similar to the effects of Tub A in combination with the ET receptor antagonist macitentan and the PDE5 inhibitor tadalafil. 384 The difference in terms of the localization between the classical HDAC inhibitors such as trichostatin A and valproic acid (localized in the nucleus) and the HDAC6 inhibitor Tub A (located within the cytoplasm) may explain the contrast between the absence and the presence of effect on pulmonary hemodynamics and vascular remodeling in the Su/Hx rat model. 384 HDAC6 promotes cardiac fibrosis and is upregulated in the lung and RV of patients with PAH and Su/Hx rats, and therefore, an HDAC6 inhibitor may represent a promising strategy in the treatment of PAH.
Bromodomain-containing protein inhibition
The epigenetic driver for PAH, bromodomain-containing protein 4 (BRD4), a member of the bromodomain and extraterminal motif (BET) family, is involved in multiple diseases, including cancer and cardiovascular diseases. 385 In addition to the vasoconstriction and obliterative vascular remodeling, PAH is also associated with inflammation, which is toxic for the cells and promotes DNA damage. Using lung tissues, distal pulmonary arteries, and PASMCs, Meloche et al. 386 demonstrated that PAH is associated with sustained DNA damage leading to activation of poly(ADP-ribose) polymerase-1 (PARP-1), which promotes cell survival and proliferation by triggering a microRNA-204 (miR-204)-dependent activation of the transcription factors, nuclear factor of activated T cells 2, and HIF-1α. PAH is regulated by miR-204, an RNA gene affiliated with the miRNA class.
PAH, linked to an increase in oxidative stress and inflammation, may result in DNA damage that is increased in human PAH lungs, remodeled arteries, PASMCs, and PAECs. 387 The anticancer drug veliparib (ABT-888, AbbVie, Chicago, IL, USA), a potent PARP-1 inhibitor, reduced mPAP, RV hypertrophy, and pulmonary arterial wall thickness when administered orally in the Su/Hx rat model. 386 In vitro results implicate PARP-1 in many PAH features, including impaired mRNA regulation, cell proliferation, and resistance to apoptosis. 386 These in vitro and in vivo findings suggest that PARP-1 inhibitors could be a new therapeutic target for human PAH.
In Su/Hx rats, an increase in miR-204 expression by nebulization of miRNA-204 led to improved mPAP and RVSP, and treatment by nebulization with either a molecular (siBRD4) or a pharmacological (JQ1) BRD4 inhibitor reversed the increases in hemodynamics (mPAP, RVSP, PVR), RV hypertrophy, and pulmonary arterial wall thickness associated with reduced VSMC apoptosis resistance and proliferation. 385 A clinically available BET inhibitor and BRD4 antagonist, apabetalone (RVX208, Resverlogix Corp., Calgary, Canada), administered orally at a clinically relevant dose alone or in combination with the PDE5 inhibitor tadalafil, and the dual ET receptor antagonist macitentan, improved pulmonary hemodynamics (mPAP, PVR) and reduced vascular occlusion in the Su/Hx rat model. 94 In a pilot study of a phase II clinical trial (APPROACH-p trial, NCT03655704), apabetalone was safe and effective in PAH. 181 Changes in PVR, CO, SV, with minimal decrease in mPAP and no changes in the 6-MWD and NT-proBNP levels, were reported in patients with PAH treated with apabetalone for 16 weeks. 181 However, a phase II trial, APPROACH-2 (NCT04915300), is ongoing to test the safety and efficacy of apabetalone in the PAH population.
miRNA inhibition
miRNAs have roles in various diseases and both long noncoding (lncRNAs) and small noncoding (miRNAs) RNAs that regulate gene expression have been implicated in pulmonary vascular pathophysiology.388–391 Dysregulation of miRNAs is linked to the hyperproliferation and apoptosis resistance of PAECs, PSMCs, and pulmonary arterial adventitial fibroblasts. 388 This has resulted in the development of inhibitors of both lncRNA and miRNA, and several miRNAs have been evaluated in Su/Hx rats showing robust effects on PAH pathology.392–394 There has been a significant disparity in preclinical models of PAH for the lung and plasma miRNA expression for 4 potential miRNA candidates (miR-29, miR124, miR-140, miR-204), with the Su/Hx rat demonstrating the best correlation to human PAH. 395 Ongoing clinical trials will determine which miRNAs increase in PAH (miR-126, miR-133—NCT03905083, multiple miRs—NCT04489251, multiple miRs—NCT04610788) and will assess the viability of targeting this approach for treating PAH.
miRNA-145, miRNA-124, and miRNA-96
IV administration of an inhibitor for miRNA-145, delivered in a liposomal formulation to Su/Hx rats, attenuated the increases in RVSP and right heart size, and decreased the wall thickness of pulmonary arteries with the greatest effects on the larger sized (100–200 μm) vessels. 392 There was also a reduction in the number of occluded pulmonary arteries and in plexiform lesion formation, and improvements were also observed in cardiac function. 392 Because of the accumulation of anti-miR-145 in the lungs, its low toxicity, and the improvement of hemodynamics and pathology in Su/Hx rats, this formulation appears to be attractive for development to treat PAH as a therapy by exploiting RNA interference.
Downregulation of miRNA-124 in pulmonary vascular and circulating progenitor endothelial cells from patients with heritable PAH and iPAH results in a switch from oxidative phosphorylation to aerobic glycolysis (Warburg effect). This feature is also found in Su/Hx-challenged rats that was characterized by endothelial hyperproliferation and reduced BMPR2 expression supporting the relevance of this mechanism to human PAH pathology. 393 These strategies, aimed to increase miRNA production, may have beneficial effects for the treatment of PAH.
MiRNA-96 is the only known miRNA that targets the serotonin 5-HT1B receptor, 394 and its effects were evaluated on the pathophysiology of PAH in Su/Hx rats. When miRNA-96 was administered IT every week for 3 weeks at a dose of 50 µg/rat, 394 the increases in RVSP, right heart size, and remodeling and occlusion of the pulmonary arteries induced by the Su/Hx challenge were significantly attenuated. There was also an increase in BMPR2 signaling in lung tissue of miRNA-96-treated rats suggesting that restoration of BMPR2 may have contributed to the beneficial effects of the drug. When studied 3 weeks after the last dose, miRNA-96 had no effects on the increases in RVSP and right heart size, or occlusion of the pulmonary arteries, but had modest effects on the pulmonary vascular remodeling. 394 These results suggest that miRNA-96 would likely have to be administered once a week to maintain activity but underline the importance that inhaled delivery is a viable method for miRNA administration that overcomes the inconvenience of IV infusion.
Nucleolin inhibition
Midkine, a 13-kDa heparin binding growth factor, regulates cell proliferation and migration, and serum midkine levels are increased in patients with PAH. 396 Nucleolin-specific small interfering RNA (siRNA) suppressed midkine-induced EGF receptor activation in PASMCs and midkine-induced migration and proliferation of PASMCs under Hx were abolished by the nucleolin inhibitor AS1411. 396 AS1411 is a nucleolin aptamer with clinical efficacy and low toxicity in several human clinical anticancer trials. 397 In the Su/Hx model, rats receiving AS1411 (10 mg/kg) showed significant reductions in RVSP, RV hypertrophy, and pulmonary vascular occlusion. 396 Therefore, midkine may play an important role in the pathogenesis of PAH, and the midkine-nucleolin-EGF receptor axis may represent a novel therapeutic target for PAH.
Estrogen inhibition
Estrogens induce cellular changes through several different mechanisms that may involve binding to the estrogen receptor (ER) located in the nucleus (genomic) or in the plasma membrane (nongenomic). 398 The classical genomic component occurs over hours, whereas the nongenomic actions occur within second to minutes. 398 In preclinical models of PAH, both exogenously administered and endogenous estrogen is involved in the pathology of PAH,67,399–401 resulting in the development of antagonists and synthesis inhibitors of estrogen. Several of them have been evaluated in Su/Hx rats and have advanced into clinical trials (metformin—NCT01352026).
Apelin
Although PAH is more common in women as mentioned previously, female patients with PAH survive longer than male patients due to the superiority of the RV function in females.63,402,403 RV function is critical to PAH survival and estrogen levels are correlated with RV function.404,405 Similar to PAH in human, sex differences exist in the Su/Hx rat model, with RV function significantly superior in females.67,406 Su/Hx female rats exhibited higher SV and CI, a better RV compliance, and a reduced increase in PVR than Su/Hx males.67,406 Ovariectomy aggravated the effects induced by the Su/Hx challenge, while repletion of the female sex hormone 17β-estradiol (E2) after ovariectomy attenuated them. In addition, repletion was associated with increased expression of the procontractile, anti-inflammatory, antiapoptotic, and prosurvival peptide apelin.67,406 E2 exerts its protective effects on RV function and remodeling via the ER-α, and recently, a cardioprotective E2/ER-α/BMPR2/apelin axis in the RV has been identified. 183 E2, via ER-α, activates BMPR2 signaling to upregulate apelin, the potent effector of cardiac contractility.183,407,408 Apelin expression is decreased in patients with PAH with RV failure and in RV homogenates from Su/Hx, monocrotaline, and artery banding models, suggesting a protecting role of apelin in RV failure development. 183 Recently, it was demonstrated that an apelin analog (0.5 µM/kg) administrated by IV (BID) in Su/Hx rats improved cardiac function, ameliorated RV hemodynamic pressures, reduced cardiac fibrosis and hypertrophy, and pulmonary vascular lesions. 184
Previously, clinical studies have been conducted in patients with heart failure demonstrating that IV infusions of apelin caused coronary vasodilatation, PVR reduction, increases in CO, and myocardial contractility (NCT00901719, NCT00901888, NCT01049646, NCT01179061).409,410 More recently, short-term IV apelin infusion with increasing doses reduced PVR and increased CO and SV in patients with PAH (NCT01457170) but did not affect mPAP and HR. 182 Interestingly, the effects of apelin treatment were enhanced in a subgroup of patients on PDE5 inhibitor therapy, suggesting that a combination of these drugs may be a new potential therapeutic strategy for PAH. 182
Metformin
Metformin acts through a variety of different mechanisms to improve blood glucose levels such as stimulation of AMPK and inhibition of the enzyme aromatase synthesizing estrogens through aromatization of androgens. Experiments in Su/Hx rats demonstrated that daily oral administration of metformin (100 mg/kg) inhibited the increase in RVSP and right heart size, reversed the pulmonary vascular remodeling, and prevented the formation of occluded pulmonary vascular lesions in female Su/Hx-challenged rats. 185 In a follow-up study to this, 64 the changes in pulmonary hemodynamics, right heart size, and pulmonary vascular remodeling induced by Su/Hx challenge were inhibited by treatment with an aryl hydrocarbon receptor antagonist, CH223191. In contrast, no significant difference in RVSP, RV hypertrophy, and pulmonary vascular remodeling was observed with a higher dose of metformin (300 mg/kg) in male S-D rats. 411 Sex differences have been previously described in the response to metformin.412,413 These results indicate an important function for endogenous estrogen in the pathology of PAH in female Su/Hx rats and demonstrates good translation to the role of endogenous estrogen in humans. Metformin is approved for the treatment of type 2 diabetes mellitus and was advanced into a phase II clinical trial for PAH (NCT01352026). However, while this trial was withdrawn due to the difficulty in recruiting eligible patients, a second 8-week trial in adult patients with iPAH or heritable PAH without diabetes mellitus showed that metformin therapy was safe and well tolerated, improved RV function, and reduced RV triglyceride content. 186
Anastrozole
In female Su/Hx rats, administration of the aromatase inhibitor anastrozole (Arimidex, AstraZeneca, Cambridge, UK) reduced the increases in RVSP, PVR, and right heart size; remodeled and obliterated pulmonary blood vessels; but had no effect on these histopathological changes in male rats. 66 The effects of anastrozole strongly correlated with a decrease in the levels of circulating estrogen and an increase in BMPR2 gene expression in the lungs of female rats, with no effects observed in male rats. These results further support the important role for endogenous estrogen in PAH and suggest that aromatase inhibitors may have a therapeutic potential for the treatment of this disease, particularly in female subjects. In a “proof-of-principle” study in male and female subjects with PAH, anastrazole administered for 3 months reduced estrogen levels, improved the 6-MWD, and was safe and well tolerated but had no effect on TAPSE (assessment of RV systolic function) or NT-proBNP levels (NCT01545336). 187 A longer and larger phase II clinical trial up to 12 months (NCT03229499) with this compound was completed, but to date, no final data were issued. 414
Drug repositioning/repurposing
Because of the high costs and slow pace of new drug discovery and development, repurposing of existing drugs to treat rare diseases such as PAH is becoming an attractive strategy. Indeed, there is a lower risk in terms of development costs due to their known safety and PK profiles and consequently, shorter development timelines. 22 If the dose and delivery route are unchanged, phase I clinical trials may not be needed and these drugs can then be rapidly evaluated in phase II clinical trials. 22 Several drugs have already been repositioned or repurposed for the treatment of PAH such as the prostaglandin epoprostenol, CCBs, and the PDE5 inhibitors sildenafil and tadalafil. 22 In the elegant review by Grinnan et al., 22 new drug candidates being repurposed and repositioned for clinical use in PAH are listed and some of them have already been discussed in this review.
Carbonic anhydrase inhibition
Carbonic anhydrase is an enzyme that catalyzes the hydration of carbon dioxide to bicarbonate and protons. Carbonic anhydrase inhibitors cause metabolic acidosis, and acidosis induced by ammonium chloride (NH4Cl) prevented Hx- and monocrotaline-induced PH and reduced RVSP, Fulton’s index, and pulmonary arteriolar remodeling in S-D rats.194,415
Acetazolamide
The carbonic anhydrase inhibitor acetazolamide (ACTZ; Diamox, Alembic Pharmaceuticals, Vadodara, Gujarat, India) has been used since the 1950s in multiple clinical conditions, including glaucoma, idiopathic intracranial hypertension, congestive heart failure, altitude sickness, and epilepsy. 194 ACTZ and NH4Cl induced metabolic acidosis in Su/Hx rats and early and late treatments with ACTZ and NH4Cl decreased mPAP, RVSP, and RV hypertrophy, while late treatment with ACTZ also reversed vascular remodeling (wall thickness). 195 Additionally, ACTZ and NH4Cl treatments reduced the increased lung and plasma mRNA levels of proinflammatory cytokines induced by the Su/Hx challenge. 195 More recently, with a design of the study slightly different, the beneficial effects of ACTZ on pulmonary hemodynamics and remodeling were confirmed in Su/Hx rats. 194 Acute hemodynamic effects of ACTZ were tested in patients with PAH undergoing right heart catheterization in a clinical trial in Europe (AcuteAZA, NCT02755259). 193 As of today, no results have been reported.
Phenothiazine antipsychotic
Thioridazine
Thioridazine is a first-generation antipsychotic drug belonging to the class of drugs called phenothiazine previously used in the treatment of schizophrenia and psychosis. The expression of the translationally controlled tumor protein (TCTP), a potent antiapoptotic and growth promoting factor, responsible for malignant transformation in many cancers, is increased in endothelial cells derived from patients with hereditary PAH with BMPR2 mutations. Continuous delivery via SC osmotic minipumps of the TCTP antagonist thioridazine slowed down the progression of PAH by reducing RVSP, RV hypertrophy, and the formation of vascular lesions in Su/Hx rats. 416
Trifluoperazine
The psychiatric medication trifluoperazine (TFP), also from the phenothiazine antipsychotic class, is used to treat certain mental disorders (schizophrenia or related psychoses). 417 TFP inhibits the AKT serine/threonine kinase/forkhead box protein O3 axis and interferes with DNA repair mechanisms and autophagy. 418 TFP has been shown to inhibit cell proliferation and to promote cell death in several types of cancer cell lines and animal models, and because proliferation of PASMCs is one of the causes of vessel thickening in PAH, it was hypothesized that TFF could also prevent PASMC proliferation in PAH. 418 TFP showed strong antisurvival and antiproliferative effects on PASMCs isolated from small lung arteries of patients with PAH, and TFP lowered mPAP, RVSP, and PVR; improved cardiac function; and reduced wall thickness of pulmonary arteries in the Su/Hx rat model. 418
Acetylcholinesterase inhibition
The acetylcholinesterase inhibitor pyridostigmine (PYR) is an FDA-approved drug used to treat myasthenia gravis. 419 This immunomodulator, by stimulating the PNS, improved survival, RV function, and reduced PVR, pulmonary vascular remodeling including RV stiffness, RV hypertrophy and dilatation, RV fibrosis, and RV inflammation in the Su/Hx rat model. 354 No safety and efficacy clinical investigations of PYR have been initiated.
Summary
In this review, we have discussed the preclinical studies performed with compounds from different classes in the Su/Hx rat model. These studies also provide the rationale for emerging new drugs that could be used in PAH and for drugs already in phase I–III clinical trials. New potential drugs for the treatment of PAH in clinical trials and tested in the Su/Hx rat model are listed in Table 4. Across all diseases, <10% of drugs that enter clinical trials succeed leading to regulatory approval. Therefore, it is necessary and essential to identify a relevant preclinical animal model for PAH. The Su/Hx rat model of PAH exhibits most of the important pathology that is found in human subjects, the most important of which is the presence of obstruction and plexiform lesions in the small pulmonary arteries that cannot be found in other standard PAH animal models such as the Hx and monocrotaline models.4–8,27 The overall pathology demonstrated in Su/Hx rats results in an increase in PVR leading to adaptations in right heart pathology, morphology, and physiology to deal with this increase in afterload.4,5,26,35 As advances have progressed in the assessment of pulmonary and cardiac hemodynamics in humans, the analysis of these physiological functions in Su/Hx rats has also improved to the extent that this rodent model can now be used to better characterize these physiological changes and to define the mechanisms involved. Over the last few years, a considerable number of compounds have been evaluated in Su/Hx rats, including those already approved for clinical use and new compounds that show promise for the future treatment of this debilitating disease. Listed below is a current update on some of the main features that have been identified in Su/Hx rats with an emphasis on their translation into PAH in human subjects.
There is a range in the overall pathology and pathophysiology over time in Su/Hx rats with an increase in wall thickness and muscularization and constriction of the small pulmonary arteries within a few weeks after Su/Hx challenge and, in models of severe disease, obstruction and plexiform lesion in the small pulmonary arteries from about 8 weeks onward. Therefore, drug targets focused on the increase in smooth muscle mass and pulmonary vasoconstriction should focus on the early phase of the disease in Su/Hx rats. Likewise, targets focused on the obstruction and plexiform lesion formation should use later time points for these evaluations. The initial assessment of right heart function in human PAH is performed with cardiac echocardiography, from which important parameters such as TAPSE, RVAWT, PAD, PAAT, Vmax, VTI, SV, and CO can be measured. With the advent of new technology, cardiac echocardiography can now be performed in Su/Hx rats, which has shown concordant changes in the hemodynamic parameters compared with human subjects with PAH. In humans, the measurement of new cardiovascular hemodynamics such as pulmonary arterial compliance may be better predictors of clinical outcomes than the classical measurement of PAP. This parameter has been successfully measured in Su/Hx rats, which opens the door for the development of novel therapies aimed at improving pulmonary arterial compliance. Current trends in drug discovery in Su/Hx rats indicate that delivery of drugs via inhalation and administered in novel liposomal or nanoparticle formulations are often a better approach to the development of both novel and established drug entities. The rationale for inhalation administration is based upon reduction of systemic AEs, delivery of the drug directly to the target organ (thereby optimizing the beneficial effects of the drug in resident, inflammatory, and immune cells in the lungs), and prolongation of the presence of the drug within the target organ. In human PAH, combination therapies appear to provide a superior effect to long-term survivability compared with monotherapy and they may have the best potential. Similar findings are demonstrated in Su/Hx rats, with many of the compounds approved for clinical use and with drug combinations using new drug entities. It will be important to find out if this translates into improved clinical outcomes in human subjects with these new combination therapies.
Future Research
Although the Su/Hx rat has many important features for the translation of basic research into human PAH pathology and pathophysiology and plays a crucial role in drug development in PAH, there are knowledge gaps with this animal model that need to be addressed with future research. For example, there is evidence that a strain of S-D rat demonstrates hyperreactivity to Su administration, but the identity of this particular strain has not been defined genetically and the biochemical pathways producing this hyperresponsiveness have not been elucidated. Without this information, it is impossible to select the use of this hyperreactive strain for drug evaluation. There is a mismatch between Su/Hx rats and humans with regard to age and the development of PAH, with the pathology and pathophysiology more evident on older subjects in humans compared with Su/Hx rats that develop more severe pathology in younger animals. On the contrary, F-H rats naturally develop PAH with advancing age. It would appear that detailed studies are required on the genetics, biochemistry, and physiology between F-H rats and other rat strains used for Su/Hx studies. From this basic research, novel targets may be identified against the specific biochemical or genetic factors involved in PAH. Similar data are also needed between the different rat strains that demonstrate hyper- versus hyporesponsiveness to a Su/Hx challenge. From a pharmacological viewpoint, a full profile of the PK, distribution, and metabolism of the drug in the strain of rat used in the Su/Hx studies would be useful to more accurately translate data in Su/Hx rats to humans. Additionally, in vitro studies of the binding potency or activity of the drug to rat enzymes or receptors in this strain of rat would help facilitate this analysis.
Conclusions
The rat model of Su/Hx has many distinguishing features that support its use for basic research in PAH. Although this animal model is not perfectly translatable to human PAH disease, it demonstrates most of the pathology and pathophysiology observed in human subjects. As the understanding of PAH has advanced in these areas, the Su/Hx rat model demonstrates translatability of the important features in humans. New drug targets continue to be advanced into clinical evaluation following preclinical studies that include the use of Su/Hx rats, and it is hoped that some of these compounds will demonstrate benefit for patients with PAH in clinical trials.
Footnotes
Acknowledgment
This work was supported by Insmed Incorporated (Bridgewater, NJ, USA).
Ethical Approval
This review did not involve human participants or animals. This is a review.
Authors’ Contributions
The authors meet the criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE) for this review. All authors (M.R.C., T.L.N., A.S., D.C., W.R.P., and R.W.C.) were involved in drafting, writing, and critically revising the content of review. All authors approved the final version for publication. Insmed Incorporated was given the opportunity to review the article for medical and scientific accuracy, as well as for intellectual property considerations.
Author Disclosure Statement
All authors are employees of the study sponsor, Insmed Incorporated. No author has an actual or perceived conflict of interest with the contents of this review. R.W.C. died during the preparation of this review.
Funding Information
Funding support for this review was provided by
