Abstract
Introduction:
During mechanical ventilation (MV), inspired gases require heat and humidification. However, such conditions may be associated with reduced aerosol delivery efficiency. The practice of turning off heated humidification before nebulization and the impact of nebulization on humidity in a dry ventilator circuit remain topics of debate. This study aimed to assess the effect of turning off heated humidification on inhaled dose and humidity with nebulizer use during adult MV.
Methods:
A bronchodilator (albuterol) and two antibiotics (Colistimethate sodium and Amikacin sulfate) were nebulized with a vibrating mesh nebulizer placed at the humidifier inlet and in the inspiratory limb at the Y-piece. Additionally, albuterol was nebulized using a jet nebulizer in both placements. Aerosol particle size distribution was determined through a cascade impactor. Absolute humidity (AH) and temperature of inspired gases were determined with anemometer/hygrometers before, during, and after nebulization, before, during, and up to 60 minutes after interrupting active humidification. Aerosol collected on a filter distal to the endotracheal tube and on impactor stages were eluted and assayed by spectrophotometry.
Results:
The inhaled dose was greater when both nebulizers were placed at the humidifier inlet than the inspiratory limb at the Y-piece. Irrespective of the nebulizer types and placements, the inhaled dose either decreased or showed no significant change after the humidifier was turned off. The aerosol particle size ranged from 1.1 to 2.7 μm. With interruption of active humidification, humidity of inspired gas quickly dropped below recommended levels, and nebulization in dry ventilator circuit produced an AH between 10 and 20 mgH2O/L, lower than the recommended minimum of 30 mgH2O/L.
Conclusion:
Interrupting active humidification during MV before nebulization did not improve aerosol delivery efficiency for bronchodilator or antibiotics, but did reduce humidity below recommended levels.
Introduction
Humidification of inspired gases is essential for patients receiving mechanical ventilation (MV). However, it can lead to an increase in aerosol particle size during nebulization with MV, thereby reducing the efficiency of aerosol delivery. Previous studies have reported greater inhaled dose of medical aerosols when administered with dry inspiratory gas compared with heated humidified gas during MV when using jet nebulizer (JN), vibrating mesh nebulizer (VMN), or pressurized metered dose inhaler (pMDI).1–10
An expert panel was commissioned to identify best practices for aerosol delivery practices for critically ill adults. 11 During the process of reaching a consensus on the best practices for aerosol administration to critically ill adults, 11 several panelists, representing a minority position, advocated turning off heated humidification before aerosol administration to increase the inhaled aerosol dose in mechanically ventilated adult, particularly for inhaled antibiotics. 12 A few panelists further proposed that aerosol generated from a nebulizer in a ventilator circuit without heated humidity would provide sufficient humidity to compensate for the absence or interruption of heated humidification. As there was a lack of published data specific to these questions, a subset of panelists agreed to perform in vitro studies at their respective institutions to determine: (1) whether interrupting heated humidity during adult MV increases aerosol deposition administered from jet and mesh nebulizers; and (2) the absolute humidity (AH) and temperature generated when nebulizing aerosol during MV when the humidifier is off and interrupted.
The available studies mostly conducted in vitro have demonstrated increases in aerosol particle size distribution which is associated with reduced aerosol delivery with heated humidification.9,10 However, most did not investigate the transition from dry to heated humidification and vice versa. Specifically, evaluations of the effect of interrupting transiently heated humidification, a potential means to reduce humidity of inspired gas lacks extensive evaluation. On the other hand, randomized controlled trials comparing patients receiving aerosols with or without heated humidification reported similar outcomes in terms of urinary salbutamol concentrations, duration of MV, and intensive care unit length of stay. 13 An in vitro study by Lin et al. investigated aerosol delivery through pMDI and spacer during MV and found no improvement when the heated humidifier was turned off, compared with aerosol delivery during heated humidification. 14 However, due to perceived differences in technology, it remained uncertain whether aerosol delivery by JN or VMN, unlike pMDI, would improve when the heated humidifier was turned off.
Moreover, limited studies assessing the effects of interrupting humidification on aerosol delivery efficiency, neither reported the temperature and humidity of the delivered gases, nor the magnitude of decrease after interrupting heated humidification, making it difficult to draw definitive conclusions regarding the benefits and risks of this practice.15,16 The impact of nebulization on AH and the temperature of inspired gas during MV has not been evaluated extensively using modern ventilators, circuits, and nebulizer.
Our objective was to investigate the effect of interrupting the heated humidifier on aerosol delivery efficiency and to quantify the impact of nebulization on the temperature and AH of the ventilator circuit. We hypothesized that interrupting heated humidity before aerosol administration during MV would not substantially increase the inhaled dose, and that nebulization alone would not provide adequate levels of heat and humidity to meet the recommended minimum AH of 30 mgH2O/L.
Materials and Methods
In vitro model
Three laboratories (Taoyuan, Chicago, and Tours) independently conducted experiments to compare the efficiency of aerosol delivery with and without heated humidification using models of adult MV (Fig. 1). The model utilized in the experiments consisted of an adult critical care ventilator equipped with a heated passover humidifier and a dual-limb heated wire circuit (Fisher & Paykel in Taoyuan and Chicago, and Hamilton Medical AG in Tours), which was connected to an endotracheal tube (ETT). Ventilator settings and medication brands varied among the three laboratories based on the clinical practices in the different institutions. Two laboratories (Taoyuan and Chicago) utilized albuterol, while the laboratory in Tours used Colistimethate sodium and Amikacin sulfate. A summary of test parameters and medications investigated by each laboratory is provided in Table 1.
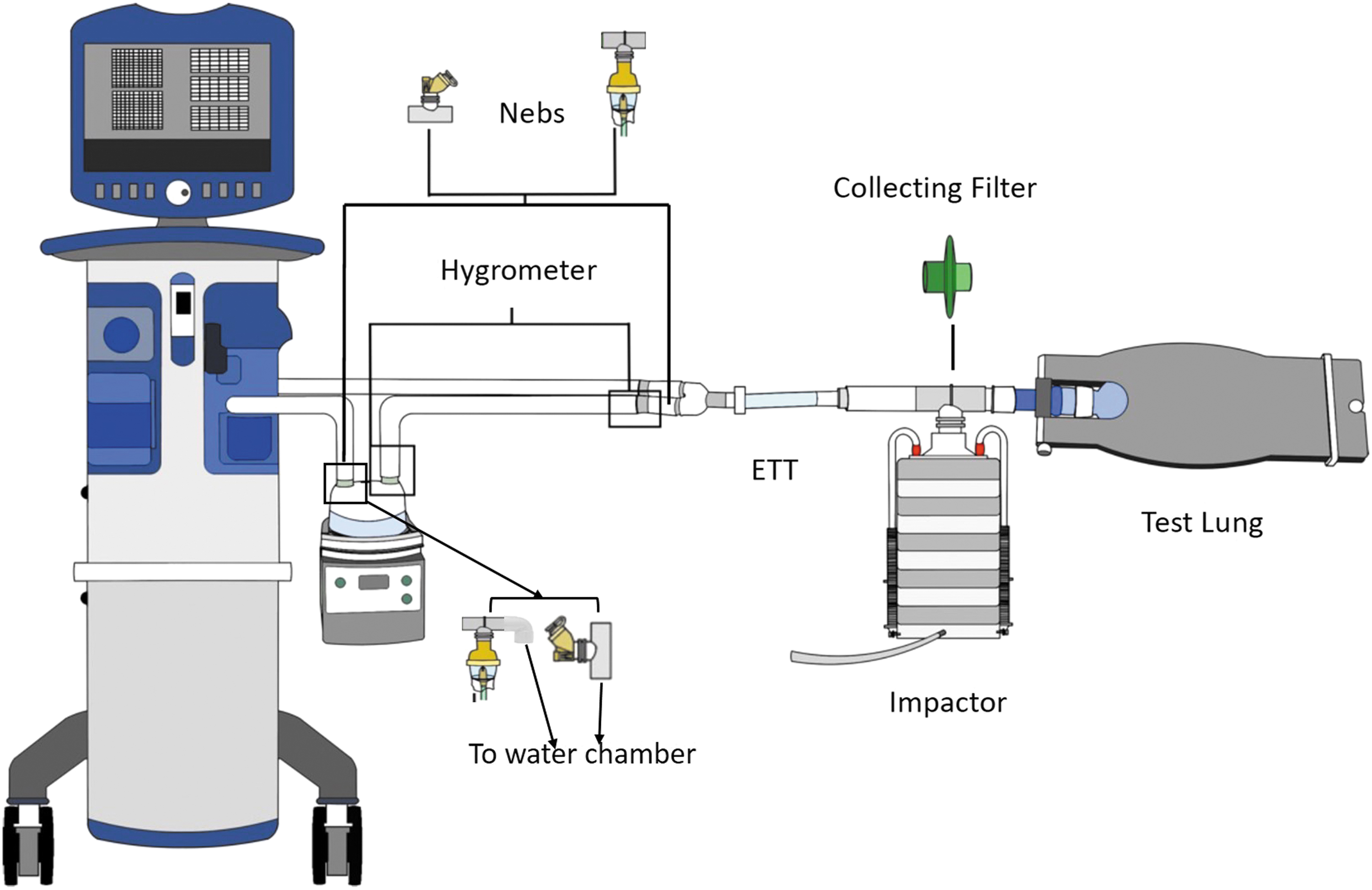
The experimental setup used in the study. An adult ventilator was attached to a heated passover humidifier and a dual-limb heated wire circuit, which were connected to an ETT. A collecting filter or an impactor was positioned between the ETT and a passive test lung. A VMN and/or a JN, was placed at the inlet of the humidifier or between the inspiratory limb and Y-piece. Hygrometer sensors were placed at the outlet of humidifier and/or proximal to the ETT. ETT, endotracheal tube; JN, jet nebulizer; Nebs, nebulizers.
Experiment Settings in the Three Labs
VC, volume controlled; PRVC, pressure-regulated volume controlled; VMN, vibrating mesh nebulizer; JN, jet nebulizer; AH, absolute humidity; D, decelerating; S, square; APSD, aerosol particle size distribution; VT, tidal volume (mL); RR, respiratory rate (breath/min); Ti, inspiratory time (s); PEEP, positive end-expiratory pressure (cmH2O); ETT, endotracheal tube; Cst, compliance (mL/cmH2O); Rrs, resistance (cmH2O/L/s).
A VMN (Aerogen Solo™; Aerogen Ltd., Galway, Ireland) was used by all laboratories, while JN (Hsiner Medical, Inc., Taichung, Taiwan), powered by compressed gas at 50 psi and operated at 8 L/min was used in Taoyuan. Nebulizers were positioned at the inlet of the humidifier and between the inspiratory limb and the Y-piece. Nebulized albuterol was administered under the following conditions: (1) in a dry circuit (dry), with no water in the chamber and the humidifier turned off; (2) at 1, 2, and 3 hours after the humidifier was turned on, with the temperature set at 37°C at the airway, (3) immediately after the humidifier was turned off (immediately off), and (4) at 30 (30 minutes off) and 60 minutes (1 hour off) after the heated humidifier was turned off. In Tours, Amikacin and Colistimethate sodium were delivered through a VMN placed at the Hamilton-H900 humidifier (Hamilton Medical AG) under the following conditions: (1) nonhumidified circuit; (2) humidifier turned on with set temperature stable (humidifier on); and (3) immediately after humidifier turned off (immediately off).
Measurements
Drug delivery
To quantify delivered drug dose, a collecting filter was placed between the ETT and the passive test lung (Fig. 1). After each nebulization, the filter was dissembled from the circuit and eluted, the drug assay was measured by a UV/VIS spectrophotometer (detailed measurements and settings in each laboratory is available in the Supplementary Appendix S1).
Aerosol characteristics
To quantify particle size distributions, a cascade impactor (ACI; Thermo Fisher Scientific, Inc., Waltham, MA used in Taoyuan and IMPAQ GS-1E, California measurements, Inc., Sierra Madre used in Tours) was placed between the ETT and the test lung, with designated operation flow rates of 28.3 and 1 L/min, respectively. Cascade impactor measurements were performed separately from filter delivery experiments using identical quantification methods.
Temperature and humidity
Two laboratories (Taoyuan and Tours) measured changes in AH (mgH2O/L) and temperature of inspired gas at the proximal position to the ETT. Hygrometer sensors (a customized hygrometer made by VADI Medical Technology Co. Taoyuan, Taiwan was used in Taoyuan and a Pt100 capacitive sensor hygrometer [KTH 350–I, Kimo®, Montpon, France] was used in Tours) were placed proximal to the ETT.
Each experiment was repeated three times (Tours laboratory) or five times (Taoyuan and Chicago) laboratories.
Statistical analysis
Statistical analysis was performed separately by each of the 3 laboratories. Results are reported as mean ± standard deviation. The inhaled dose of albuterol was calculated as a percentage of the captured dose in the collection filter to the nominal dose. Repeated measures analysis of variance and post hoc Tukey's test were used to compare the inhaled dose and aerosol particle size distribution across different conditions (dry circuit, humidifier on, and humidifier turned off), while the inhaled dose between two nebulizer positions were compared by Mann–Whitney test. A p-value lower than 0.05 was considered as significant.
Results
Inhaled dose of different medications
Albuterol
Nebulized albuterol under different circuit conditions were tested in two laboratories (Taoyuan and Chicago). Regardless of the ventilator settings, the results of inhaled dose under different conditions from both laboratories (Fig. 2) are similar. When the VMN was placed between the inspiratory limb and Y-piece and the JN was positioned at the inlet of the humidifier, the inhaled dose was greater with dry circuits compared with heated humidified circuits. However, when the VMN was placed at the inlet of the humidifier, the inhaled dose was lower with a dry circuit and the humidifier off compared with a heated humidified circuit at 1 and 2 hours (all p < 0.05). In contrast, the inhaled dose with the JN placed between the inspiratory limb and Y-piece did not show significant changes under these conditions. Regardless of nebulizer types, placing nebulizers at the inlet of the humidifier resulted in a greater inhaled dose compared with placing them between the inspiratory limb and Y-piece (p < 0.001).
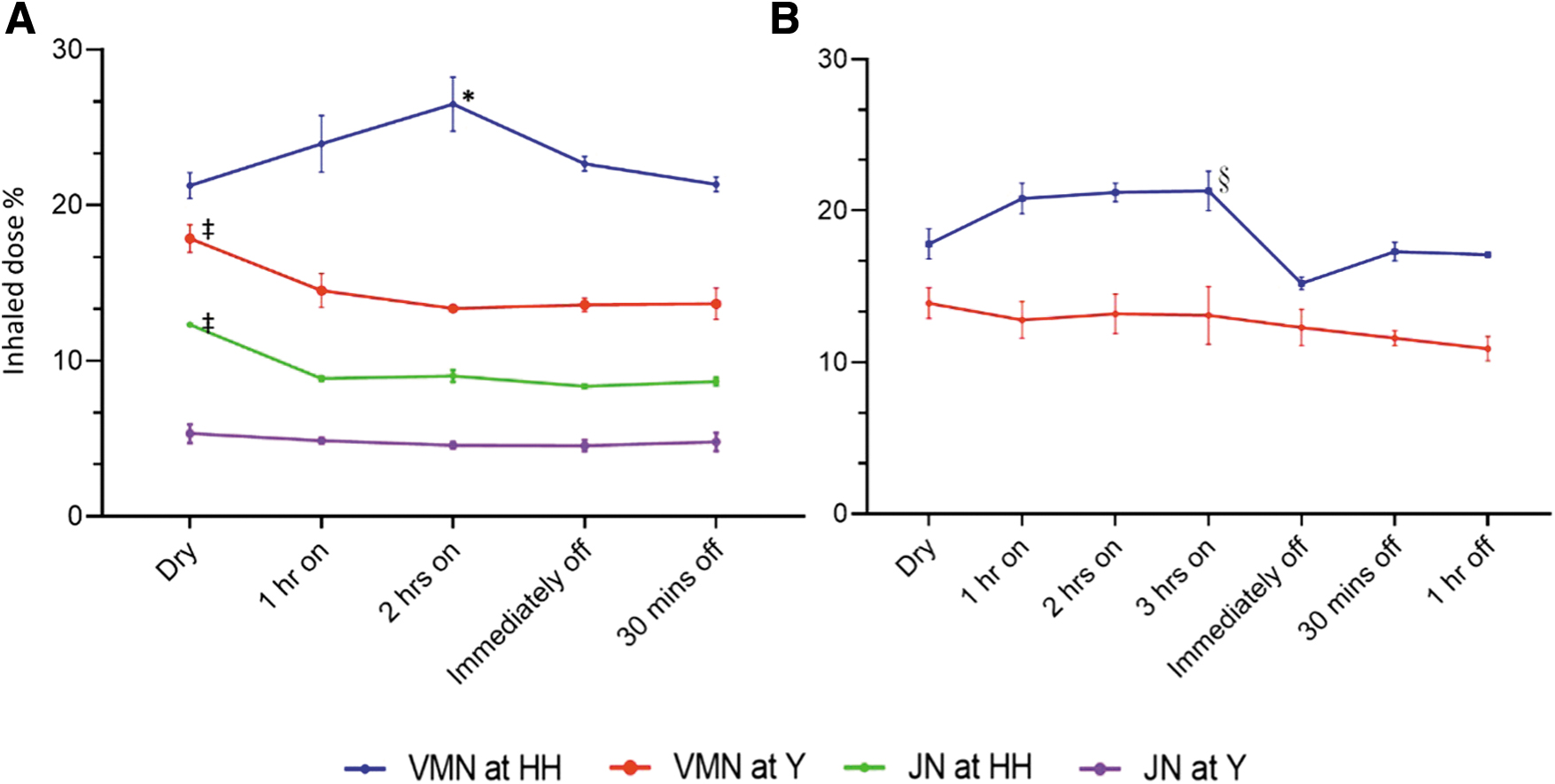
The inhaled dose (mean ± SD) of albuterol collected distal to the ETT.
Antibiotics
Regardless of the minute ventilation settings, the inhaled dose of antibiotics was higher in the dry circuit with humidifier off (Fig. 3), in comparison with both the humidified circuit and during the 30-minute automatic humidification interruption. However, there were no significant differences in the inhaled dose between the humidified circuit and the 30-minute interval with humidification turned off.
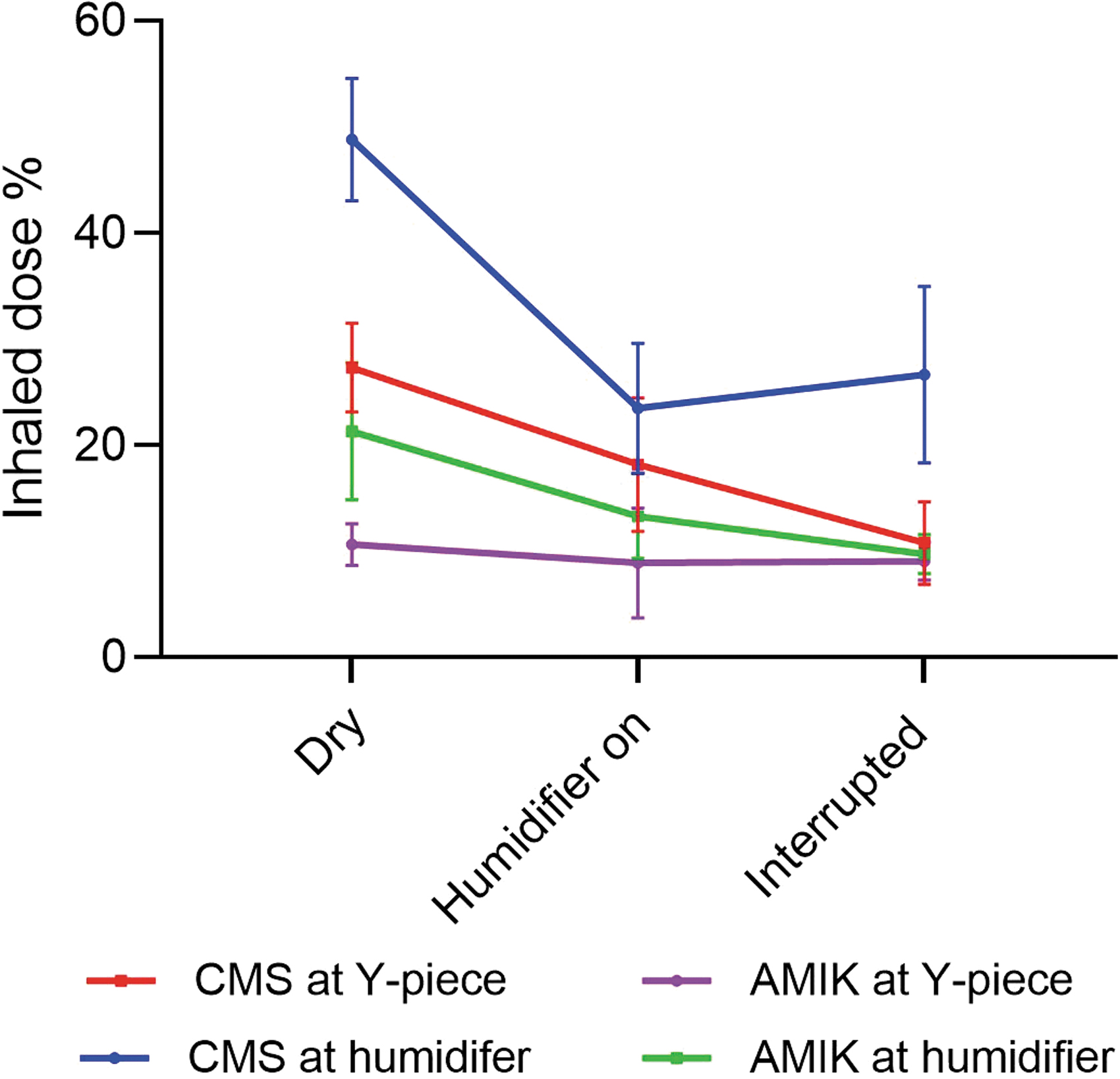
The inhaled dose (mean ± SD) of CMS and AMIK delivered distal to the ETT. VMN was placed at the inlet of humidifier and between the inspiratory limb and Y-piece with dry circuit, humidifier on, and humidifier interrupted for 30 minutes (Tours laboratory). AMIK, Amikacin; CMS, Colistimethate sodium.
Aerosol particle size
The median mass aerodynamic diameter (MMAD) of albuterol measured at the ETT outlet in Taoyuan laboratory was 1.10 ± 0.25 μm in the dry circuit, listed in Table 2. After 1 and 2 hours of heated humidification, the MMAD increased to 2.30 ± 0.28 μm. However, after 1 hour of humidification interruption, the MMAD returned to 1.17 ± 0.17 μm. In contrast, the MMAD of Colistimethate measured at the ETT outlet in Tours laboratory was 1.7 ± 0.4 μm in the dry circuit and increased to 2.1 ± 0.1 μm in the heated humidified circuit. There was no significant change in MMAD observed 30 minutes after interrupting humidification, which remained at 2.1 ± 0.2 μm. Table 2 listed the comparisons of particle size distributions on MMAD, geometric standard deviation, and fine particle fractions.
Particle Size Distributions Among the Conditions
FPF, fine particle fraction; GSD, geometric standard deviation; MMAD, median mass aerodynamic diameter.
Humidity
AH with humidifier interruption (Tours laboratory)
When the humidifier temperature was set at 37°C and 41°C, the AH decreased to below 30 mgH2O/L within 5–10 minutes after turning off the humidifier, regardless of the minute ventilation settings (Fig. 4).
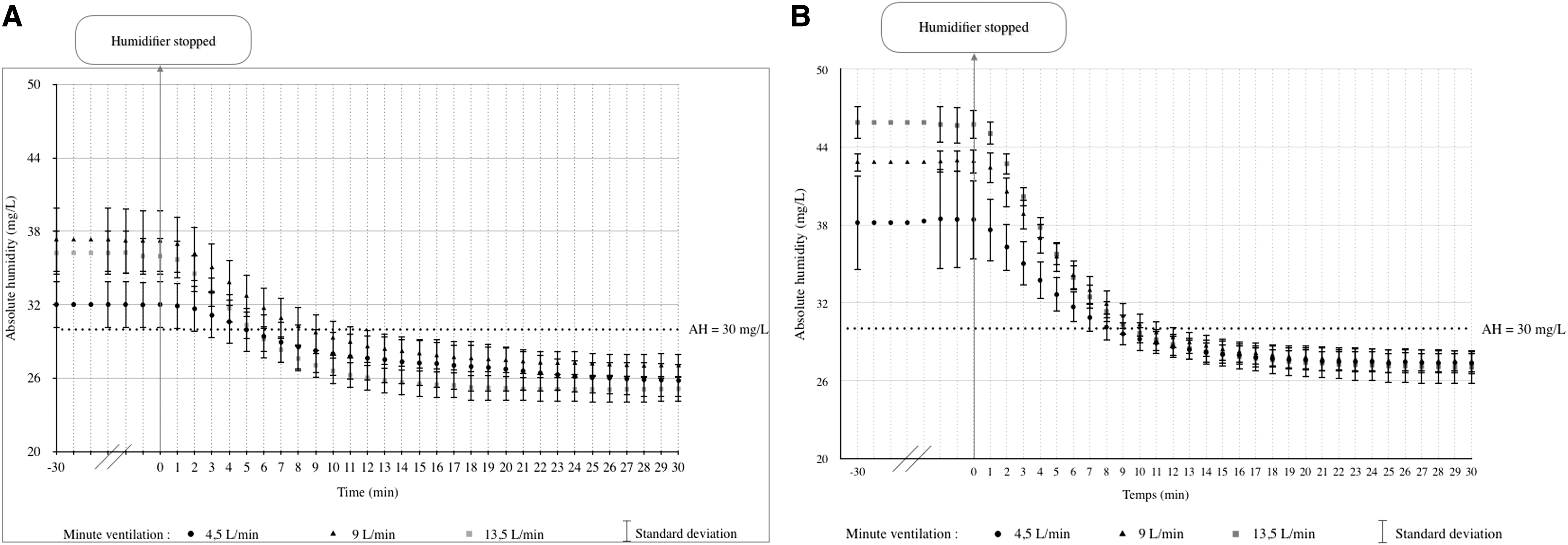
AH expressed at mgH2O/L (mean ± SD) measured distal to the circuit Y-piece. Humidifier temperature was set at 37°C
AH with nebulization (Taoyuan laboratory)
The changes of temperature and AH with the use of VMNs and JNs in dry and heated humidified circuits at various time points are illustrated in Figures 5 and 6. In the dry circuits, nebulization through JN caused a decrease in gas temperature by 10°C (Fig. 5C, D), while AH remained ∼10 mgH2O/L (Fig. 6C, D). Conversely, the gas temperature remained unaffected by nebulization with VMN placed between the inspiratory limb and Y-piece (Fig. 5B), while temperature slightly decreased with VMN placed at the inlet of humidifier (Fig. 5A). In both placements, AH increased to ∼20 mgH2O/L, which was maintained during nebulization periods (Fig. 6A, B). Furthermore, when the humidifier was turned off and nebulization started, regardless of the nebulizer types and placements, both gas temperature and AH decreased. In particular, AH dropped to around 20 mgH2O/L by the end of nebulization (Fig. 6).
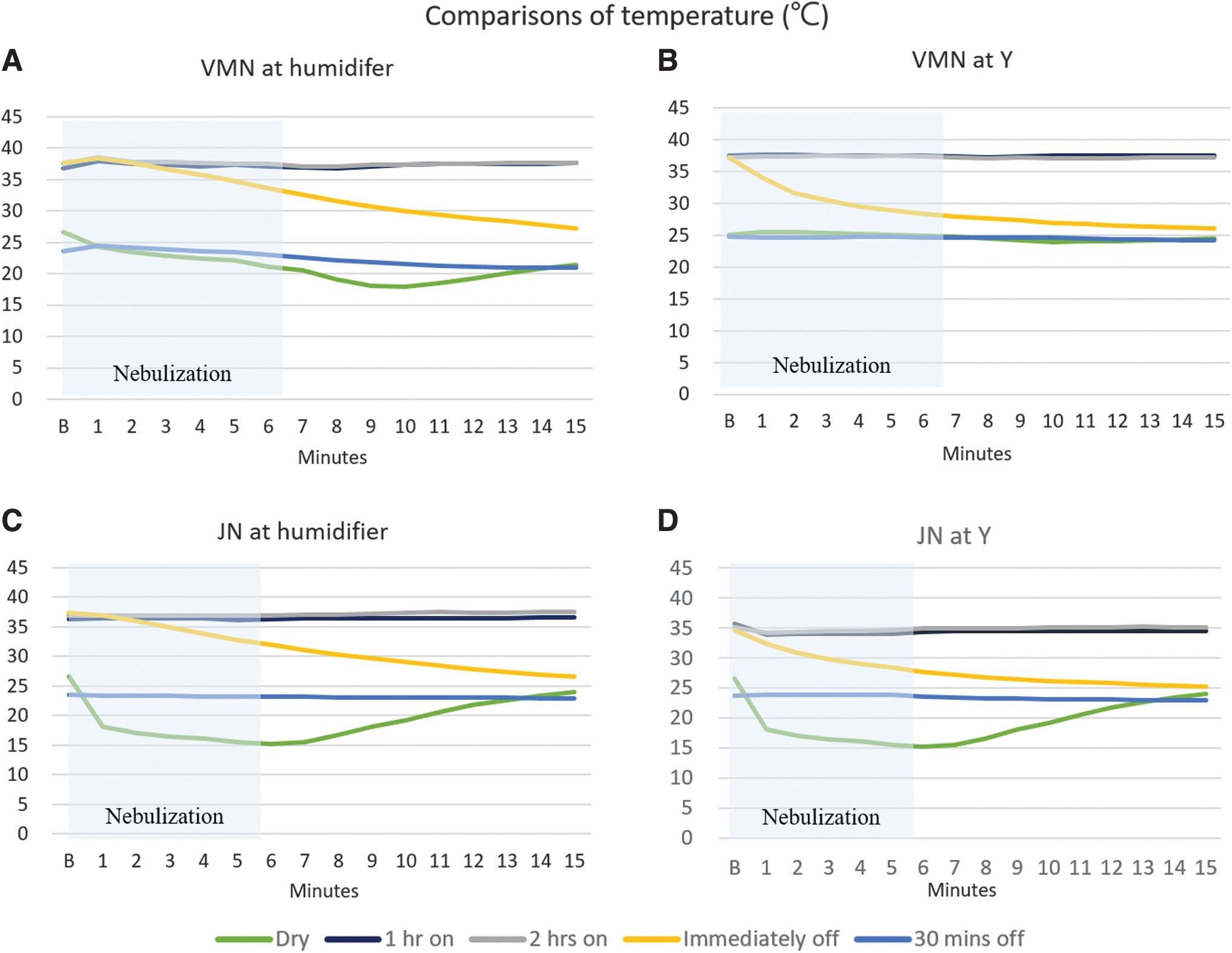
The changes of temperature measured distal to the Y-piece at various time points with nebulizers positioned at different locations. Nebulizers were placed at different locations (Taoyuan laboratory):
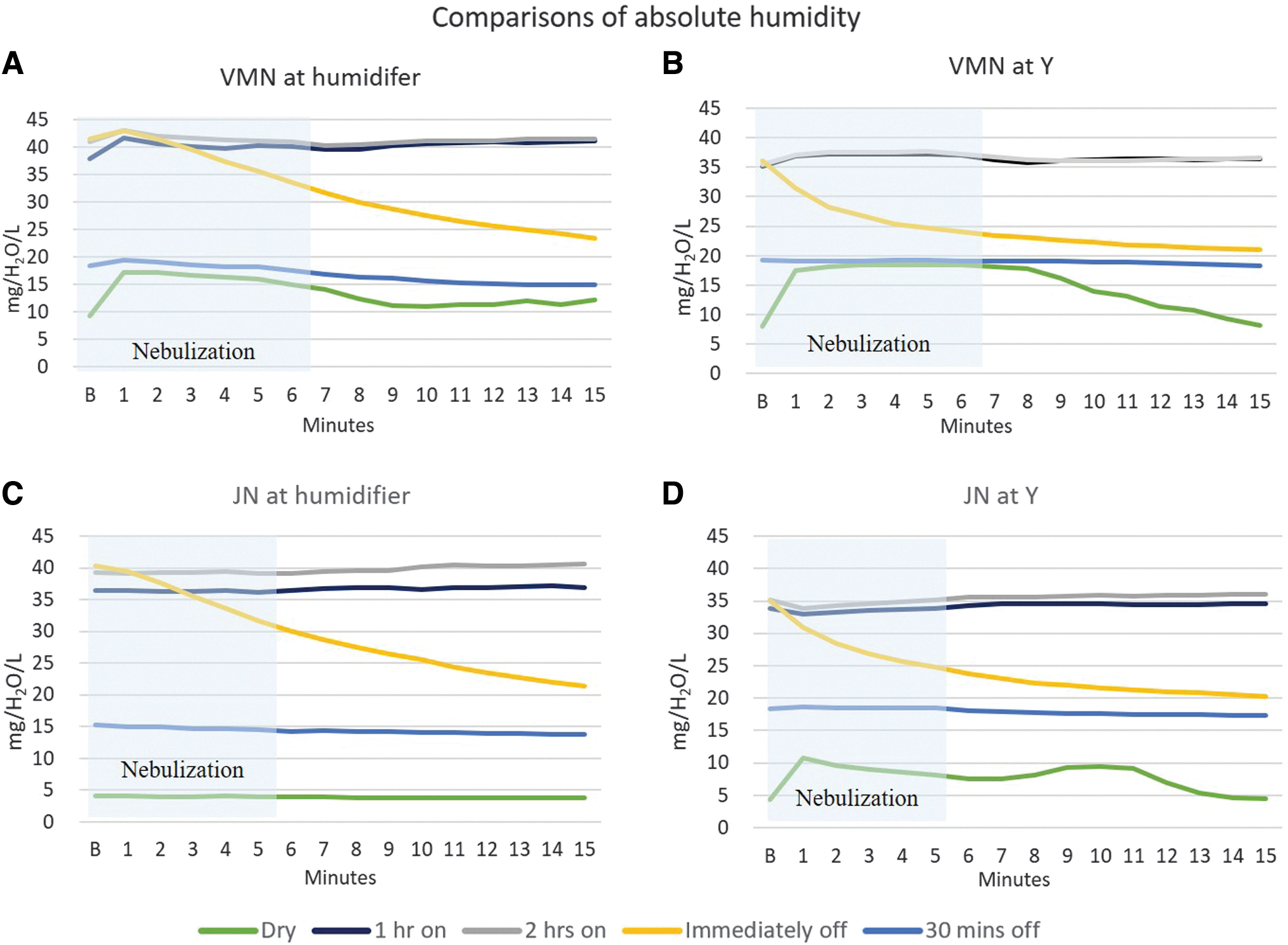
The changes of AH measured distal to the Y-piece at various time points with nebulizers positioned at different locations. Nebulizers were placed at different locations (Taoyuan laboratory):
Discussion
This study represents extensive in vitro explorations conducted by three collaborating laboratories to investigate the impact of interrupting heated humidification on nebulizer aerosol administration on drug delivery efficiency during MV. The results from all three sites, employing complementary methods, consistently demonstrated that interrupting heated humidification for 30–60 minutes did not increase aerosol drug delivery efficiency under the tested conditions. While nebulization performance was initially higher under some conditions with a new dry circuit and the heater off and without water in the humidifier, this was not consistently observed across all three laboratories. As hypothesized, there was no significant difference in nebulization delivery efficiency between aerosol administered with heated humidification and after the humidifier was turned off for 30 minutes, regardless of the types of medication tested. In addition, this study is the first to quantify changes in AH and temperature during nebulization using JN and VMN in both unheated dry and heated humidified ventilator circuits.
The findings revealed that the AH levels of 10–18 mgH2O/L produced by nebulization in a dry circuit were below the recommended minimum levels of 30 mgH2O/L to avoid airway damage during MV.
Effect of heated humidification on nebulizer efficiency
All laboratories consistently reported that the inhaled dose was greater when VMN was placed at the inlet of humidifier compared with between the inspiratory limb and Y-piece. In line with numerous previous studies using various aerosol devices (pMDI, JN, ultrasound nebulizer, or VMN),1–10,13 our findings also showed a higher inhaled dose with dry circuits compared with heated humidified circuits when aerosol generators were placed between the inspiratory limb and Y-piece. However, when VMN was placed at the inlet of humidifier, we did not find any significant differences in the inhaled dose between dry circuits and heated humidified circuits. This finding may appear contradictory to the findings by Ari et al., who reported nearly three times higher inhaled dose with dry circuits compared with heated humidified circuits when VMN was placed at inlet of the humidifier. 1 It is important to note that their study did not use bias flow. Interestingly, in another study conducted by the same authors using the same experiment setup with bias flows ranging from 2 to 5 L/min, 18 the mean inhaled doses (21.4%–23.8%) with heated humidified circuits were comparable to the inhaled dose with dry circuits (24.2% ± 1.2%) reported in the previous study. 1 In our study, all three laboratories utilized bias flow (2–4 L/min), thus our findings agree with Ari et al.'s findings.1,18
Miller et al. described the in vitro and in vivo effects of jet nebulization of antibiotics during MV, both with humidifier on, and immediately after turning it off and bypassing it for dosing. 8 In contrast to our findings, they reported consistently higher deposition when bypassing the humidifier, which was associated with even greater proportional increases in in vivo measures based on sputum samples. Factors such as use of a passover humidifier set to 35°C without heated wires and prolonged nebulization for up to 60 minutes may contribute to increased deposition after interruption of humidity. Considering advancements in ventilators, humidifiers, and nebulizers, a thorough investigation of the humidifier bypass strategy may be warranted to better understand its impact on inhaled medication delivery.
Furthermore, in vitro models simulating active exhaled humidity have reported smaller differences in inhaled dose between dry and humidified conditions. 19 This effect is even more pronounced in vivo, where patients add heat and humidity to the inspired gas. This probably explains that studies examining drug levels in urine and comparing dry and heated humidification conditions during aerosol administration have shown no significant difference in drug levels.13,14
Effect of turning off heated humidification on nebulizer efficiency, aerosol particle size, and absolute humidity
Our findings, consistent across all three laboratories, indicate that interrupting or stopping heated humidification before nebulization did not result in a higher inhaled dose compared with dry circuits or during active heated humidification. These findings align with the study conducted by Lin et al., 15 who observed similar patterns with administration of pMDI and chamber positioned proximal to the Y-piece of a ventilator circuit. Additionally, we observed substantial condensate formation in the circuit, adapter, and model lung during the 30 minutes after turning off the humidifier, which may explain the lack of increase in the inhaled dose. These consistent findings support our primary hypothesis that emitted aerosols, regardless of the aerosol devices (pMDI, JN or VMN) used or the medications (bronchodilator or antibiotic) being nebulized, are similarly affected by changes of heated humidification.
Furthermore, the MMAD obtained in our study is comparable to those reported by Miller et al. using a JN of 1.5 μm in dry circuits and 2.3 μm in humidified circuits. 8 Similarly, Liu and colleagues reported a range of 2.03–2.26 μm with Colistimethate from VMN in heated humidified circuits. 19 Despite variations in particle size distribution among different humidification conditions, we found that the MMAD was consistently below 2.7 μm, which is within the range for effective pulmonary and alveolar deposition. Interrupting humidification before nebulization resulted in an immediate drop in AH, falling below 30 mgH2O/L within 5 to 10 minutes. However, delivering aerosols after the interruption, the process of AH falling was expedited to 3 to 5 minutes. Setting the humidifier chamber to a higher temperature of 41°C did not slow down the process.
Under normal circumstances, the upper airway adds heat and humidity to inhaled gas, providing sufficient water content in the lower airway to maintain homeostasis in the lung (44 mgH2O/L at 37°C). However, compressed medical gases used during MV contain little to no water vapor and are relatively cold. Current guidelines from American Association for Respiratory Care and International organization for standardization recommend a minimum AH of 30 mgH2O/L delivered through an artificial airway during MV to prevent airway injury, regardless of humidification modalities (active or passive humidification).20,21 Based on these comprehensive findings that indicate no additional benefits with nebulization efficiency but reductions in AH, we strongly recommend keeping the heated humidifier at all times to maintain the recommended AH 30 mgH2O/L by the guidelines. Interrupting heated humidification is not conducive to improving aerosol delivery and may pose potential risks for patients, even for inhaled antibiotics delivery.
Effect of nebulization on AH and temperature in dry circuits
In dry ventilator circuits, nebulization affected both AH and temperature. When JN was positioned between the inspiratory limb and Y-piece, the temperature dropped from 25°C to 15°C, with AH peaking at 10 mgH2O/L. The drop in temperature with JN is attributed to adiabatic expansion of compressed gas. Interestingly, when VMN was placed at inlet of the humidifier in a dry circuit, the temperature also dropped to 15°C during nebulization, resulting in AH of 10 mgH2O/L. This cooling effect observed with VMN has not been previously described and may be due to evaporative cooling of the aerosol. The measured AH of 10 mgH2O/L during nebulization in dry circuits represents only a small fraction of the emitted aerosol mass, as not all droplets evaporate and the remaining particles do not contribute to the water vapor content of the inhaled gas. Nonetheless, it is important to note that inhaled gas temperatures as low as 15°C may potentially trigger bronchospasm and exacerbate airway response, which might persist beyond the period of low inhaled temperature. When the humidifier was turned off, AH dropped to near 20 mgH2O/L, which is below the recommended level of 30 mgH2O/L. The combination of low inhaled temperature and AH of nebulization in a dry ventilator circuit warrants further investigation.
Limitations
There are several limitations in this study. First, the Tours laboratory only measured humidity without nebulization to avoid potential interferences of effects with the introduction of aerosol. Second, we observed higher nebulization efficiency with Colistimethate compared with Amikacin, it should be interpreted with caution as this study was not specifically designed to compare the two drugs, although the UV spectrophotometer measurements showed good linearity for each drug. Third, our experiments did not simulate exhaled humidity, which may lead to an overestimation of the inhaled dose with dry circuit in the clinical setting compared with the bench measurements. 22 Fourth, several nebulizers were used according to the experimental requirements, specific interdevice and batch variability was outside the scope of the study. Fifth, variations in experimental settings exist among the three laboratories due to differences in clinical practice. Despite these discrepancies, the consistent outcomes underscore the robustness and validity of our findings. Lastly, we found no benefit to turning off heated humidification for administration of medical aerosols through nebulizer.
While the risk associated with short-term periods of exposure to inhaled gas with lower AH and temperature than recommended by international organizations have not been well quantified or reported (over the past four decades), such determinations are beyond the scope of this investigation, and is an area that may merit further investigation. However, subjecting patients to any additional risk in the absence of any demonstrable benefit should be avoided.
Conclusion
Interrupting heated humidification before aerosol administration by JN or VMN did not enhance nebulization efficiency with bronchodilator or antibiotics during MV. However, this interruption resulted in a reduction in AH in the inspired gas below recommended levels. Nebulization in dry circuits failed to provide sufficient humidity but led to a decrease in gas temperature. When VMN was placed at the inlet of humidifier, dry circuits did not improve aerosol deposition compared with heated humidified circuits. Based on these findings, it is not recommended to interrupt heated humidification or use dry circuits solely for increasing aerosol delivery efficiency during MV.
Footnotes
Acknowledgments
The authors extend their heartfelt gratitude to the esteemed panelists involved in the consensus of ‘Aerosol Therapy in Adult Critically Ill Patients' for their invaluable encouragement and support in undertaking this study.
Authors' Contributions
H.L., J.L., J.B.F., and S.E. conceived this project. S.J., C.S., P.K., and J.F.L. conducted the study. S.J., J.L., and H.L. performed the data analysis. S.J., J.L., H.L., and J.B.F. drafted the article. S.E. supervised the project and provided critical edits on the article. S.J., H.L., and J.L. equally contributed to the overall project. All authors reviewed and approved the article.
Author Disclosure Statement
J.L. discloses research funding from Fisher and Paykel Healthcare Ltd., Aerogen Ltd., and Rice Foundation, and speaker fees from AARC, Aerogen Ltd., Heyer Ltd., Vincent Ltd., and Fisher & Paykel Healthcare Ltd. H.L. discloses research funding from Chang Fung Memorial Foundation. S.E. discloses consultancies from Aerogen Ltd., research support, speaker fees, and travel support from Aerogen Ltd, and Fisher & Paykel Healthcare, research support from Open AI and Hamilton Medical AG. J.B.F. is Chief Science Officer for Aerogen Pharma Corp. Others do not have any conflicts of interest to disclose.
Funding Information
The experiments performed in Tours were in part funded by Hamilton Medical AG, the company was not involved in data interpretation, article preparation, and the decisions to submit; other laboratories had no funding for the experiments presented in this article.
Reviewed by:
Gerald Smaldone
Ariel Berlinski
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
