Abstract
Background:
Chronic obstructive pulmonary disease (COPD) is a preventable, progressive disease and the third leading cause of death worldwide. The epidemiological data of COPD from Gulf countries are very limited, as it remains underdiagnosed and underestimated. Risk factors for COPD include tobacco cigarette smoking, water pipe smoking (Shisha), exposure to air pollutants, occupational dusts, fumes, and chemicals. Inadequate treatment of COPD leads to worsening of disease. The 2024 GOLD guidelines recommend use of inhaled bronchodilators, corticosteroids, and adjunct therapies for treatment and management of COPD patients based on an individual assessment of the severity of symptoms and risk of exacerbations. This article reviews COPD pharmacotherapy in the Gulf countries and explores the role of nebulization in the management of COPD in this region.
Methods:
To review the COPD pharmacotherapy in the Gulf Countries, literature search was conducted using PubMed, Medline, Cochrane Systematic Reviews, and Google Scholar databases (before December 2022), using search terms such as COPD, nebulization, inhalers/inhalation, aerosols, and Gulf countries. Relevant articles from the reference list of identified studies were reviewed. Consensus statements, expert opinion, and other published review articles were included.
Results:
In the Gulf countries, pressurized metered-dose inhalers (pMDIs), dry powder inhalers (DPIs), soft mist inhalers, and nebulizers are used for drug delivery to COPD patients. pMDIs and DPIs are most prone to errors in technique and other common device handling errors. Nebulization is another mode of inhalation drug delivery, which is beneficial in certain patient populations such as the elderly and patients with cognitive impairment, motor or neuromuscular disorders, and other comorbidities.
Conclusion:
There is no major difference between Gulf countries and rest of the world in the approach to management of COPD. Nebulizers should be considered for patients who have difficulties in accessing or using MDIs and DPIs, irrespective of geographical location.
Introduction
Chronic obstructive pulmonary disease (COPD) is a progressive and potentially debilitating disease that is prevalent globally. It is a preventable and multifactorial disease that can be treated. 1 Globally, it is considered to be the third leading cause of death. 2 A meta-analysis of the global prevalence of COPD showed that spirometry-confirmed COPD prevalence, was 12.2% (10.9%–13.4%), and pooled prevalence was 15.7% (13.8%–18.6%) and 9.9% (8.7%–11.1%) in men and women, respectively. 3 As of 2019, ∼380 to 455 million people were diagnosed with COPD4,5 and accounted for 3.23 million deaths worldwide. 2 According to WHO, low- and middle-income countries account for 90% of COPD deaths in people 70 years of age and below. 2
This review will focus primarily on the epidemiology and management of COPD in the Gulf countries and the local challenges encountered with it. It also focuses on the role of nebulization in the management of COPD in these countries, as an option to overcome some of the local challenges of pharmacological management of COPD. For this purpose, a literature search was conducted using PubMed, Medline, Cochrane Systematic Reviews, and Google Scholar databases (before December 2022). Consensus statements, expert opinion, and other published review articles were included.
COPD Epidemiology in the Gulf Countries
In the Middle East and North Africa (MENA) region or the Gulf countries, the data on COPD epidemiology, including the age or sex-standardized prevalence, incidence, morbidity, and mortality rates are very limited, suggesting that COPD remains underdiagnosed and underestimated. 5 The estimated prevalence of COPD in the MENA countries has increased from 2.56% in 1990 to 3.13% in 2017. 6
In the Kingdom of Saudi Arabia (KSA), between 1990 and 2019, the age-standardized prevalence rate of COPD increased by 49% in the population, the prevalence increasing by 37.4% and 33.8% in men and women, respectively. Age-standardized incidence of new COPD cases increased by 43.4%. COPD mortality increased by 47.9%, which accounted for 1.65% of the total all-cause deaths in KSA. This constitutes 57% of the deaths due to respiratory diseases, including asthma and interstitial lung disease. 5 A recent study reported COPD prevalence in the United Arab Emirates (UAE) to be between 3.7% and 5.3%, whereas prevalence of asthma was 12.3% to 13.0%. In the Middle East, the prevalence of adjusted asthma was 4.4% to 7.6%. 7
The landmark BREATHE study, conducted to estimate the prevalence of COPD symptoms in the general adult population in 10 countries in the MENA (Algeria, Egypt, Jordan, Lebanon, Morocco, KSA, Syria, Tunisia, Turkey, and UAE) region and in Pakistan,8,9 reported that the adjusted prevalence of COPD was 3.6% (95% CI: 3.5–3.7 [range: 1.9% in UAE to 6.1% in Syria]) and was more frequent in men compared with women (5.2% vs. 1.8%, p < 0.0001). COPD prevalence was more in men than in women (5.2% vs. 1.8%; p < 0.0001), and in men it varied from 2.6% in UAE to 9.7% in Syria. Among women, COPD prevalence was higher in Lebanon (4.7%).8,9 Cigarette smokers (p < 0.001) and waterpipe users (p < 0.026) had a significantly higher frequency of respiratory symptoms. The BREATHE study showed that COPD prevalence in the MENA region was lower than that reported in the Western countries. This difference was attributed to under-reporting and nonsmoking-related risk factors. 9
Overall, the proportion of persons with a current or past history of smoking of tobacco (cigarettes, waterpipes, or both) was 31.2% [95% CI: 30.9–31.6] in the MENA region. Tobacco smoking was significantly higher (p < 0.001) in men (48.0%) compared with women (13.8%), and no differences were observed between the age groups. Rate of smoking ranged from 15.3% in Morocco to 53.9% in Lebanon. Cigarette smoking rates were 28.8% [28.4–29.2] and waterpipe usage was 3.5% [3.4–3.6]. Waterpipe usage was more frequent in KSA (8.5% of participants). Cigarette smoking was higher, and the mean number of pack years smoked was 18.5 ± 20.5 (mean ± SD) for women and 29.1 ± 26.2 for men. Smoking, the major risk factor for COPD, is a major health concern in the MENA region. 10 This study reported that usage of available therapies was exceptionally low, implying that COPD was underdiagnosed and inadequately evaluated and treated in this region.11,12
COPD Risk Factors in the Gulf Countries
COPD presents with continuous and progressive respiratory symptoms and clinical manifestations that include breathing difficulty due to airflow limitation; cough and/or phlegm; airways occluded by mucus, inflammation, and swelling of airway lining; and lung destruction. 2 Risk factors include tobacco smoking (cigarette, water pipe smoking, or both), indoor and outdoor air pollutants, biomass fuel, occupational dusts, fumes, and chemicals. Some of these risk factors are specific to Gulf countries.2,13 Smoking is one of the leading causes of COPD and in the Gulf countries it is part of the culture, particularly the waterpipe smoking (Shisha). The BREATHE study revealed that the overall prevalence of smoking was 31.2% and COPD was 3.6%, ranging from 1.9% in UAE to 6.1% in Syria.7,14 In KSA, about 4.2% of adults 40 years of age and above live with COPD. 15 Other studies in KSA have reported the prevalence to be between 2.4% and 14.2%. Prevalence of COPD in Lebanon was 5.3% in the BREATHE study 14 and 9.7% in the Waked et al. study. 16
COPD Management in the Gulf Countries
In Gulf countries, management of COPD is complicated by the fact that COPD is underdiagnosed, inadequately evaluated, and undertreated. The use of inhaled long-acting bronchodilators, corticosteroids, and oxygen therapy for COPD is exceptionally low. However, COPD symptoms are one of the key reasons for significant health care consumption, which includes more physician visits and hospitalization. 12
In the BREATHE study, 50.5% of subjects, defined as having COPD by GOLD criteria, were on medication for their respiratory symptoms. Among these, only half of the population was using either bronchodilators or inhaled corticosteroids [ICSs] or both as per the GOLD guidelines. Among the COPD patients, the use of both long-acting bronchodilators and ICS, either alone or in combination, was seen in less than 5% of subjects. 12
For COPD, no proven cure is available, and management involves slowing disease progression, treating symptoms, and preventing exacerbations. Many national and international guidelines, including the GOLD and Saudi Thoracic Society (STS) guidelines for COPD, recommend using inhaled therapy for drug administration,17–19 since it provides the advantages of quicker onset of action, reduced dose, and less systemic side effects. 20
The 2024 GOLD report provides guidance for the treatment and management of COPD patients, including the use of inhaled bronchodilators, corticosteroids, and adjunct therapies, thought to potentially have some benefit in COPD.1,2,6,19 It recommends spirometry for making an accurate measurement of not fully reversible airway obstruction and a confident diagnosis of COPD in patients with chronic respiratory symptoms (dyspnea, cough, expectoration, and/or exacerbations). 19 As per this report, guidance for pharmacological therapy is based on assessment of symptoms and risk of exacerbations (Fig. 1). 19
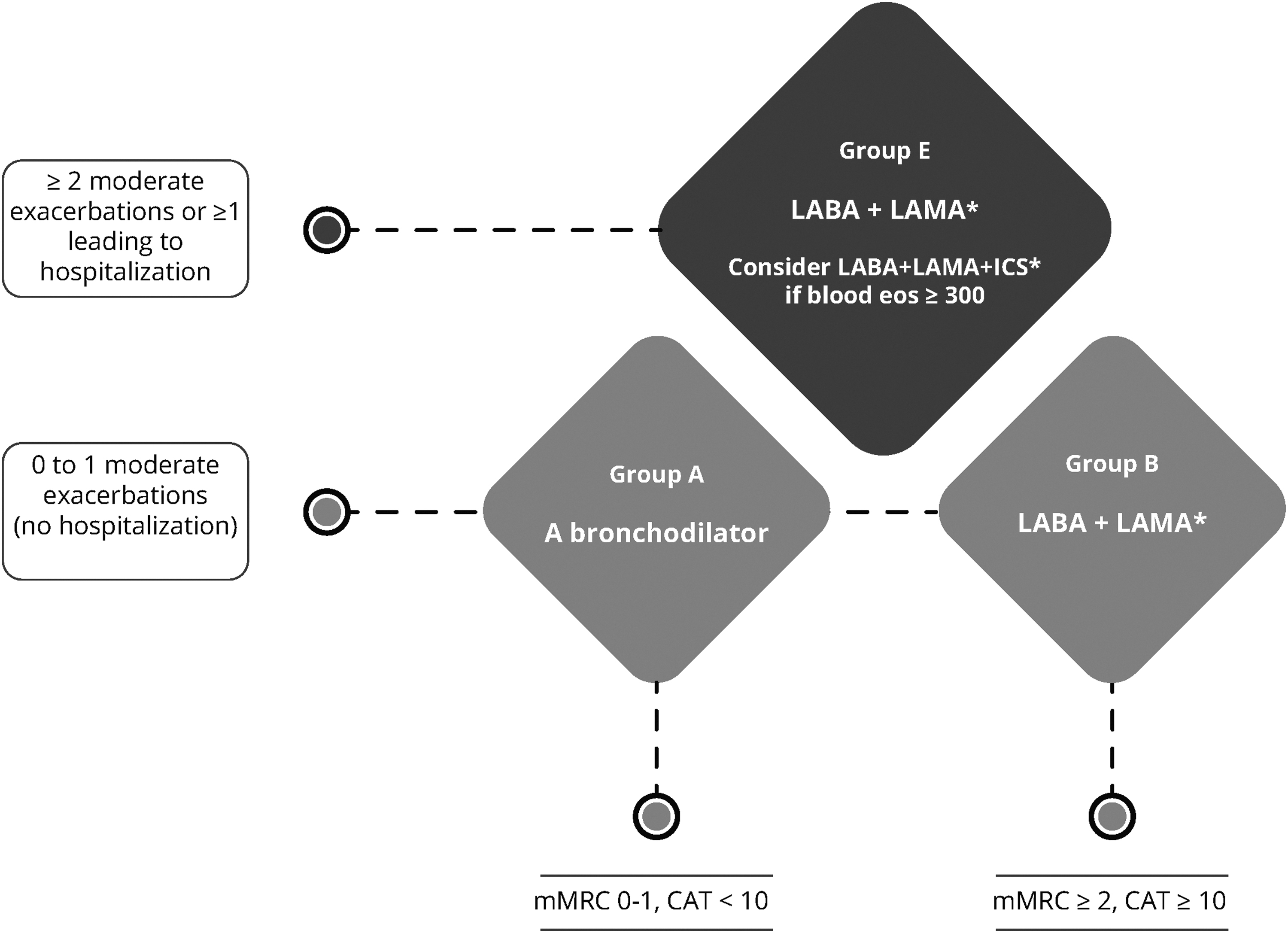
Pharmacological management of COPD as per the GOLD guidelines based on symptoms and risk of exacerbations. Initial Pharmacological Treatment: *single inhaler therapy may be more convenient and effective than multiple inhalers. Reproduced and modified from GOLD Guidelines Committees. 19 CAT, COPD assessment test; COPD, chronic obstructive pulmonary disease; eos, blood eosinophil count in cells per microliter; ICS, inhaled corticosteroids; LABA, long-acting beta2 agonist; LAMA, long-acting muscarinic antagonist; mMRC, modified Medical Research Council dyspnea questionnaire.
Similarly, the STS guidelines for COPD from KSA, which is based on the GOLD guidelines, recommend pharmacological therapy for COPD mostly based on symptom control of symptoms and acute exacerbation prevention. COPD patients are categorized into three clinical classes (Fig. 2) 21 :
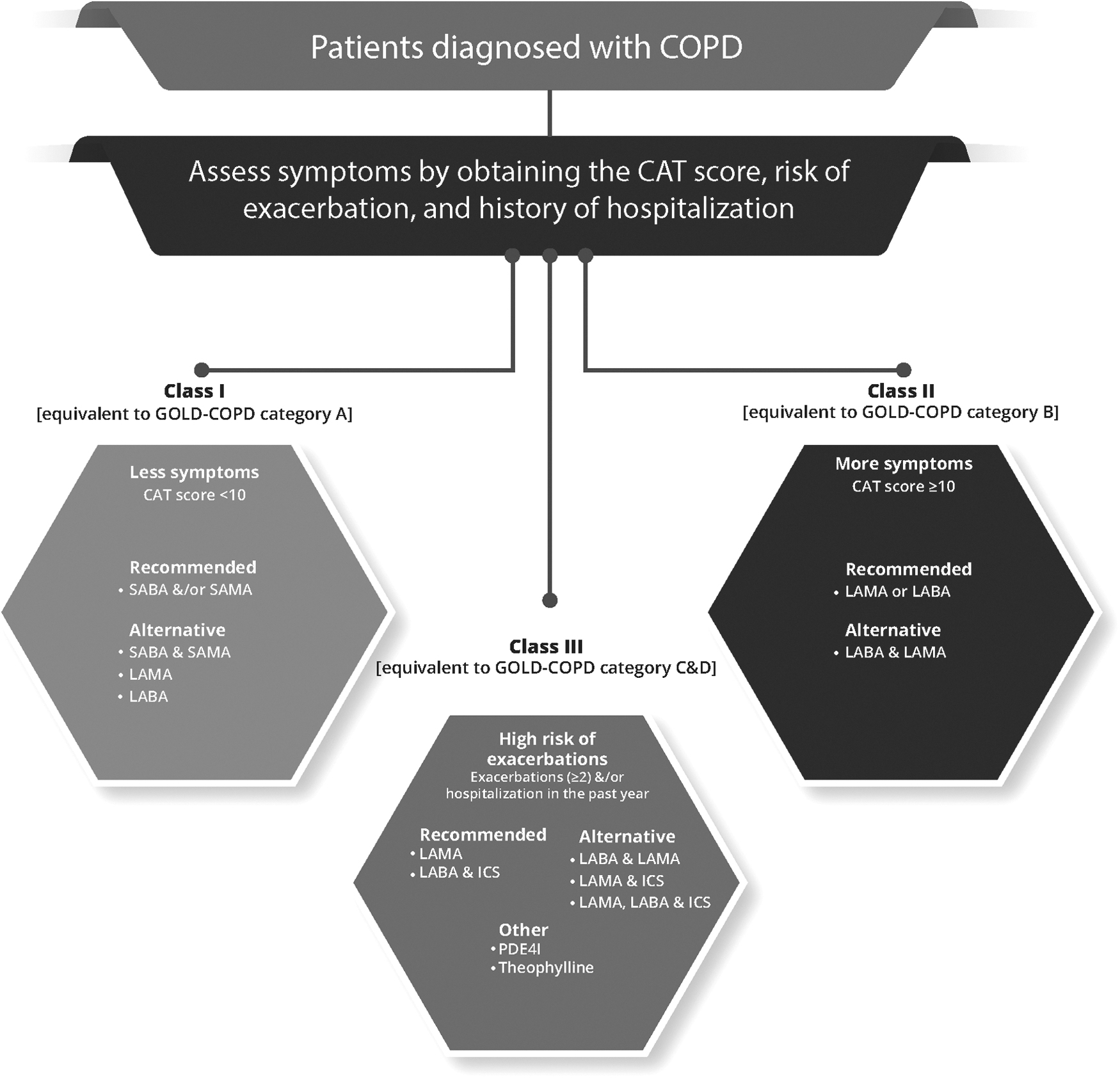
Management of COPD as per the STS guidelines. Information from Khan et al. 21 CAT, COPD assessment test; COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroids; LABA, long-acting beta2 agonist; LAMA, long-acting muscarinic antagonist; PDE4I, phosphodiesterase-4 inhibitor; SABA, short-acting beta2 agonist; SAMA, short-acting muscarinic antagonist; STS, Saudi Thoracic Society.
Class I: Patients having less symptoms (CAT <10) and 0–1 exacerbation in the previous year with no hospitalization. This is equivalent to GOLD group A.
Class II: Patients with more symptoms (CAT ≥10) and 0–1 exacerbation in the previous year with no hospitalization. This is equivalent to GOLD group B.
Class III: With increased exacerbation risk, manifested by ≥2 exacerbations in the previous year and/or hospitalized exacerbation. This is equivalent to GOLD group E as per 2023 GOLD guidelines (previously it was GOLD-COPD category C and D).
Pharmacological Therapies in the Gulf Region
Pharmaceutical agents help in reducing COPD symptoms, decrease exacerbations, enhance exercise tolerance, and improve health status. Selection of a pharmacological therapy depends on its clinical response, side effects, availability, and cost. 19 Other approaches include smoking cessation, use of vaccines, and oxygen therapy.
Bronchodilators
Bronchodilators are mostly prescribed and used in COPD due to their effects on symptom reduction, improved expiratory flow, and exercise performance. They improve the FEV1 and other spirometry parameters. 19 Commonly used bronchodilators include the short-acting beta2 agonists (SABAs), long-acting beta2 agonists (LABAs), long-acting muscarinic antagonists (LAMA), and oral methylxanthines (e.g., aminophylline, theophylline), although the latter are now used infrequently. Bronchodilators alone and in various combinations with another bronchodilator and/or an ICS that are available in one or more countries in the Gulf region are shown in Tables 1–3.
Pharmacological Agents Available for Treating Chronic Obstructive Pulmonary Disease in United Arab Emirates and the Kingdom of Saudi Arabia
DPI, dry powder inhaler; KSA, Kingdom of Saudi Arabia; LABA, long-acting beta2 agonist; LAMA, long-acting muscarinic antagonist; SABA, short-acting beta2 agonist; SAMA, short-acting muscarinic antagonist; UAE, United Arab Emirates.
Combination Therapies for Chronic Obstructive Pulmonary Disease Available in the Kingdom of Saudi Arabia
Available as SMI.
ICS, inhaled corticosteroids; LABA, long-acting beta2 agonist; LAMA, long-acting muscarinic antagonist; SABA, short-acting β2 agonist; SAMA, short-acting muscarinic antagonist; SMI, soft mist inhaler.
Hand-Held Inhalers for Chronic Obstructive Pulmonary Disease Available in the Gulf Countries
Short-acting inhaled bronchodilators
SABAs as bronchodilator medications, delivered either by a hand-held inhaler or by nebulization, are recommended in the management of COPD to relieve acute bronchospasm. Studies have reported that during a COPD exacerbation (ECOPD), SABAs and short-acting muscarinic antagonists (SAMAs) can generate 15% to 30% improvement in FEV1 and FVC over a period of 60 to 120 minutes, and both agents generate similar bronchodilation.22–24 The choice of the inhaler device is individualized to patient's needs, preferences, and demonstrated ability or lack of ability to use hand-held devices appropriately.
SABAs are effective for 4 to 6 hours and both regular and as-needed SABAs improve FEV1 and symptoms of bronchospasm. 19 Also, the onset of bronchodilation is faster with a SABA compared with a SAMA, a difference that may be relevant while making the decision as to which should be used as rescue therapy for worsening symptoms or an acute exacerbation.
LABAs and muscarinic bronchodilators (LAMAs)
LABAs and LAMAs are recommended in patients with severe COPD for long-term treatment.24,25 In patients with stable COPD, LABAs and LAMAs are indicated in patients with symptomatic COPD (Medical Research Council ≥2 and CAT ≥10) to reduce dyspnea and improve lung function and health-related quality of life (HRQoL). They are also indicated to help reduce ECOPD.26–28 Among LABAs, while formoterol and salmeterol are administered as twice-daily dose, indacaterol and olodaterol are ultra-long-acting beta-agonists administered once daily. All these medications reduce symptoms and improve FEV1, lung function, health status, and quality of life (Figs. 1 and 2).19,21 The STS guidelines recommend the use of ipratropium bromide as a rescue medicine for acute bronchospasm (Figs. 1 and 2).19,21
LAMAs are known to improve COPD symptoms and health status.29,30 They have shown greater effectiveness than a LABA in reducing exacerbations, including those leading to hospitalization31,32 and to improve exercise tolerance combined with pulmonary rehabilitation.30,33,34
Of the various LAMAs, tiotropium, umeclidinium, and revefenacin are administered once daily, while aclidinium is given twice daily. Glycopyrronium is approved in some countries for once-daily dosing, and in a few others, it is approved for twice-daily dosing. LAMAs improve symptoms, health status and effectiveness of pulmonary rehabilitation, and reduce exacerbations and related hospitalizations. Clinical trials have shown a greater effect on exacerbation rates for a LAMA (tiotropium) versus a LABA treatment (salmeterol). 31
Anti-inflammatory agents
Inhaled corticosteroids
ICs are recommended for consideration with a long-acting bronchodilator (LABD) for patients with GOLD Grade E with an eosinophil count ≥300 cells/μL. 19 In the available guidelines, the use of a short course of systemic corticosteroids is recommended for treating exacerbations of COPD (ECOPD).19,30,35 Using corticosteroids in addition to bronchodilators can significantly decrease treatment failures and prolonged hospital stays, in addition to its effect on improving COPD symptoms and lung function. 19 IC monotherapy is not recommended for the treatment of COPD. However, when given in combination with a LABA, the efficacy of ICs is enhanced in COPD. 19 Table 1 provides the list of pharmacological agents available for treating COPD in UAE and KSA.
Combination therapies
Combination therapy using different bronchodilators may enhance bronchodilation and reduce the risk of side-effects compared with increasing the dose of respective monotherapies. 19 Combination therapies (SABA/SAMA, LABA/LAMA, or LABA/ICS) are more effective in reducing symptoms and exacerbations, in improving FEV1, lung function, and health status, and show no greater side effects compared with respective monotherapies (SABA, SAMA, LABA, or LAMA). 19 Table 2 provides combination therapies available in KSA. Inhaled therapies with triple drug combination (e.g., Trelegy Ellipta, Breztri, Aerosphere) are more effective in improving lung function and reduce exacerbations compared with respective double-drug combinations.36,37 In the Gulf countries, some of the hand-held inhalers available include single-drug bronchodilators, such as tiotropium bromide (Spiriva Respimat), and combinations, such as umeclidinium bromide/vilanterol (Anoro Ellipta), tiotropium bromide+olodaterol (Spiolto Respimat), and others (Tables 1 and 3).
Phosphodiesterase-4 inhibitors and macrolide antibiotics
Roflumilast does not have a direct bronchodilator effect, but when used in combination with systemic corticosteroids, it decreases moderate-to-severe exacerbations in patients with severe- to very-severe COPD and chronic bronchitis. This effect is more pronounced in patients with a previous history of hospitalization due to an acute exacerbation. 19 Macrolides, such as azithromycin, are beneficial in COPD patients with smoking habits and severe exacerbations. 19
Nonpharmacological Approaches
Management of COPD patients using nonpharmacological approaches includes smoking cessation, vaccination, long-term supplemental oxygen therapy, and pulmonary rehabilitation, and should be considered in addition to pharmacological therapies.19,38,39
Smoking cessation is the only intervention that significantly slows the accelerated decline in lung function that usually characterizes COPD in patients who continue to smoke, in addition to reducing the disease progression and mortality. The combination of patient counseling (including simple advice to quit smoking), education and pharmacotherapy (nicotine replacement therapy, bupropion sustained release, nortriptyline and varenicline alone or in combination) has been shown to be effective in promoting smoking cessation and sustained abstinence in smokers with COPD.19,38–40 Studies have shown that quitting smoking at early stages of COPD helps to reverse loss of lung function. 38
In UAE, although a smoking cessation program is recommended as an important nonpharmacological treatment approach, the personal experience of one of the authors indicates that there is limited success with this approach because of an inadequate number of such programs, lack of skilled health care professionals with expertise in psychotherapy and cognitive behavior therapy, and a limited pool of well-trained consultants to operate these centers.
In COPD patients, reduced exercise capacity can lead to hospitalization, poor HRQoL, and risk of mortality. Pulmonary rehabilitation should be considered in addition to pharmacological treatment, since it can reduce symptoms, hospitalization, mortality and improve exercise capacity, and overall health status. Physical exercise, awareness, training, and education about disease management may improve quality of life and reduce mortality.19,38,39 In the Gulf countries, one of the key challenges with rehabilitation programs for patients with COPD is that physiotherapists are less familiar with the disease, in the opinion of one of the authors.
Viral infections such as influenza and pneumococcal infections can result in exacerbations and hospitalization in COPD patients. Influenza and pneumococcal vaccination are recommended for these patients, as it can reduce the occurrence of lower respiratory tract infections, exacerbations, hospitalization, and risk of death.19,38,39 In the Gulf countries, there are no standardized vaccination programs and no long-term tracking and monitoring of COPD patients regarding their vaccination status.
Supplemental oxygen (O2) therapy is another crucial part of treatment in advanced COPD. It can be used as a long-term O2 therapy (≥15 hours a day), during exercise, during sleep, or at high altitude. The clinical benefits include prolongation of survival in those with severe resting hypoxemia, increase in exercise capacity, and a reduction in symptoms, particularly breathlessness.19,41
In UAE, according to the experience of one of the authors, COPD patients have a poor understanding and awareness about oxygen therapy. Many COPD patients who are prescribed supplemental oxygen therapy refuse to use supplemental oxygen while performing outdoor physical activities and prefer to use oxygen therapy at night or while they are at home. Consequently, COPD patients prescribed oxygen therapy tend to stay at home and minimize their physical activity.
Devices for Aerosol Delivery of Bronchodilators and Inhaled Steroids
Inhalation therapy, using various inhalation devices, or aerosol-based drug delivery systems are the preferred modes and cornerstone of drug delivery in many of the respiratory disorders. Many studies have discussed the role of inhaled drug delivery in COPD patients.42–46 Various medications, such as short- and long-acting beta2-adrenergic and antimuscarinic bronchodilators, corticosteroids, and combination products, are all available as aerosolized therapies for COPD.22,47 While selection of the appropriate medication is important, choice of the right inhalation device for the right patient is also essential and critical for successful treatment of COPD. The following inhalation devices are most commonly used for drug delivery in COPD patients—pressurized metered-dose inhalers (pMDIs), dry powder inhalers (DPIs), soft mist inhalers (SMIs), and nebulizers. In Saudi Arabia, inhalers are available in the form of pMDIs (evohalers), breath-actuated devices (Easyhaler) and DPIs (turbuhaler, discus, aerohalers, and handihalers).20,21 The several types of inhalation devices, each having its own distinct characteristics and inhalation technique, can cause confusion for both patients and clinicians.22,48,49
Certain COPD patients may find pMDIs, DPIs, and SMIs to be easy to use. Others may face challenges while using these hand-held devices; such patients include those with neuromuscular disabilities, cognitive or ventilatory impairments, and those in need of caregiver support,42,50,51 particularly elderly patients.52,53 The use of DPIs is specifically challenging for those with poor peak inspiratory flow rate (PIFR). Inhaler technique needs to be assessed regularly. 19
Recent innovations have led to the development of new drugs and newer technologies and devices for drug delivery in COPD patients. For example, glycopyrrolate and revefenacin, both LAMAs, have been approved for use in a nebulizer for long-term treatment of COPD. 19 Budesonide, an ICS, is available in the UAE for use as a nebulizer. All these epitomize that nebulization may be a suitable treatment option for delivery of maintenance medication for COPD in addition to hand-held devices.42–46
Pressurized metered-dose inhalers
The pMDIs are one of the most common and widely used hand-held inhalation devices, which contain propellants that aerosolize the drug and deliver it through a pressurized dispenser. 20 They can be used with or without a spacer. The pMDIs containing hydrofluoroalkane propellants release the drug as a fine mist at a reduced flow velocity ensuring better aerosol delivery than older chlorofluorocarbon pMDIs.20,22,54–56 This reduced velocity is of benefit in those patients who have difficulty with the coordination while inhaling the medication. 52 The use of a spacer allows for creation of smaller particle aerosols that can flow at reduced velocity resulting in better lung deposition of the drug and response to treatment.20,22,54–56
Dry powder inhalers
DPIs are passive inhalers that are dependent on the inspiratory airflow generated by the patient for delivery of the aerosolized drug. 57 As a breath-actuated device, a DPI eliminates the key problem associated the pMDIs, which is the coordination required between device actuation and inhalation. However, for DPIs, a higher inspiratory flow rate is required for dispersing and dispensing the drug easily compared with pMDIs, for which the propellant provides the force for aerosol delivery.58–60 The drug delivery flow rate from pMDIs is low (∼30 L/min) compared with DPIs, which is higher (>60 L/min), and the rate varies based on the inhaler device.61,62
In patients with severe COPD, using DPIs that offer increased airflow resistance may be difficult because they may fail to achieve the required inspiratory flow rate sufficient enough to create an effective aerosol.20,22,61,62 In some patients, higher error rates are seen due to incorrect device handling and inappropriate preparation of the dose and inhaler. Another challenge with DPIs is that patients may exhale into the inhaler before inhalation, and this can increase humidity in the device. This may affect the device, drug product, and dosing efficiency over a period of time.20,22,56
The DPI systems are of four types based on the dose delivered: single-unit dose inhaler; disposable inhaler (with a premetered amount of a single dose); multiunit devices (which deliver individual doses from premetered replaceable blisters, disks, dimples, or tubes); and multiple-dose reservoir inhalers. 63
Soft mist inhalers
SMIs are nonpropellant-based inhalation devices, which aerosolize the drug particles that are delivered as a fine mist. Like pMDIs, they are portable, useful in patients with lower inspiratory flow rates, offers better lung deposition of the drug and ensures accurate and reproducible dosing. However, they require coordination between device actuation and inhalation, coordination while assembling the cartridge, and involve multiple steps of device preparation, which makes it difficult and cumbersome to patients.64–66 Currently, Respimat® is the sole commercially available SMI.65,66 In UAE, SMIs (Respimat) are available as Spiriva Respimat (tiotropium) and Spiolto Respimat (tiotropium plus olodaterol). However, SMIs are not available for use in other Gulf countries.
Nebulizers
Nebulizers are drug delivery devices that support inhaled therapy, with the liquid medicament delivered as a fine aerosol. They offer various advantages compared with DPIs and pMDIs. 67 Patients can inhale the medication more comfortably since the aerosol in a nebulizer is produced consistently and requires only tidal-volume breathing. Nebulizers make it possible to modify the dose, are effective with tidal breathing, require minimal patient coordination and minimal inspiratory efforts, and therefore are beneficial in patients with poor coordination or inadequate PIFR, and can be used with supplemental oxygen.19,67,68
Some of the common complaints about nebulizers are related to the deficiencies of the device itself. Hand-held nebulizers have advantages over traditional nebulizers that require a stationary power source 69 as they are small in size and portable. Although traditional nebulizers such as jet nebulizers are easy to use and less expensive than newer high-efficiency nebulizers, such as ultrasound and vibrating mesh nebulizers, they are bulky and need a compressor, pressurized gas or electricity as an energy source, treatment and drug delivery is prolonged as more time is required for preparation and set up of the device (8–10 minutes may be required for dose delivery), and additional equipment is needed for cleaning.22,68 Other problems with nebulizers include possible interaction between device and drug formulation, since surface tension and viscosity of the formulation can impact the aerosol formation. Table 4 summarizes the properties of different types of nebulizers.
The Properties of Different Categories of Nebulizers
Some of the nebulizers that are currently available in the Gulf countries include: Jet nebulizers (e.g., Omron CompAir, Beurer IH21), ultrasonic nebulizers (e.g., Philips InnoSpire Essence, Omron Ultra AIR Pro), and vibrating mesh nebulizers (e.g., Philips InnoSpire Go, Beurer IH55) (Fig. 3).
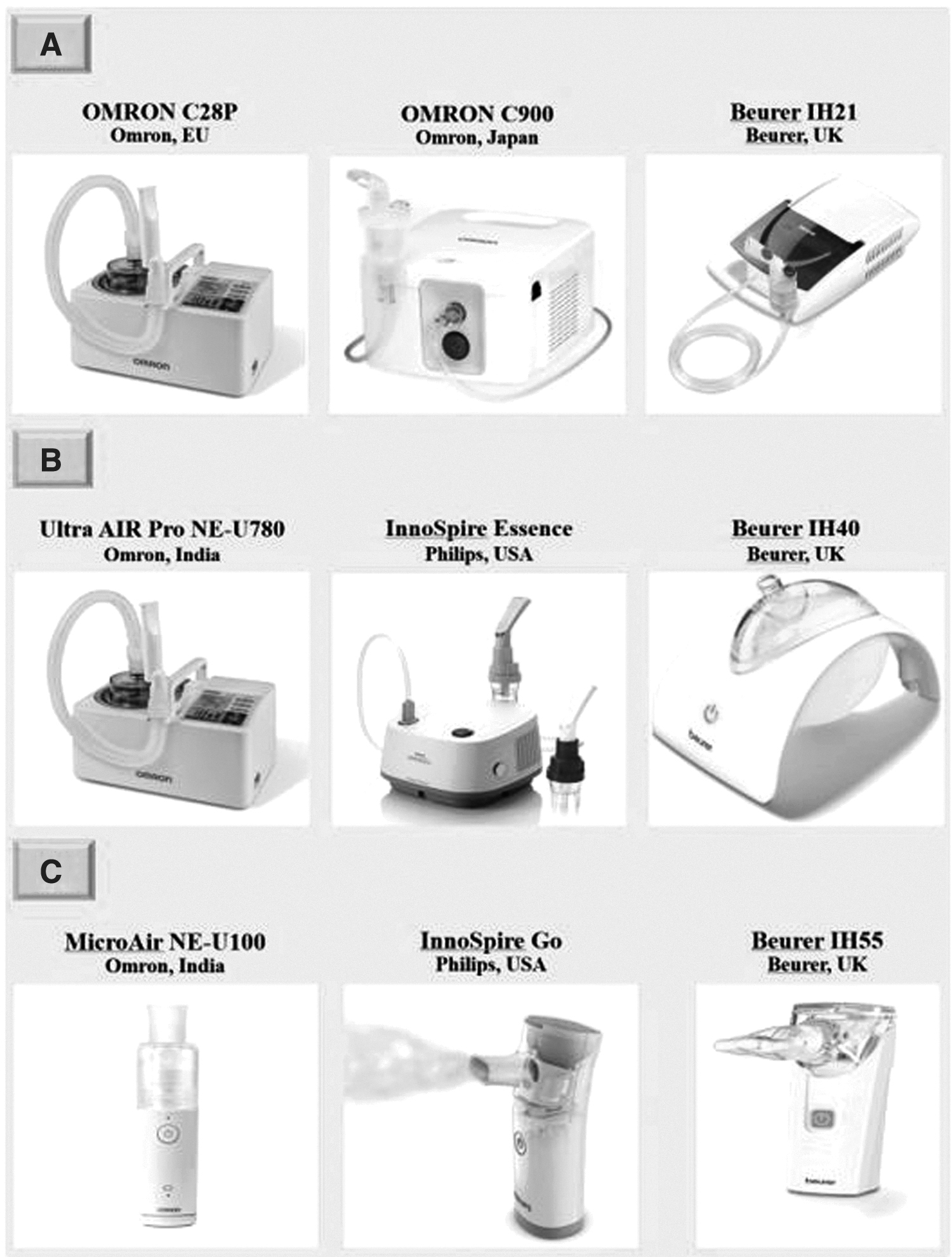
Jet, ultrasonic, and vibrating mesh nebulizers available in the Gulf Countries.
Nebulizers as Maintenance Therapy in COPD
Maintenance therapy with a nebulizer is advised in COPD patients with the following conditions
22
:
Those with cognitive function constraints (Alzheimer's, other dementia, etc.), motor complications (Parkinson Disease, dyskinesias, etc.), neuromuscular disease (severe pain, muscle weakness, etc.), arthritis, stroke, and altered consciousness that prevents normal use of hand-held inhalers. Those facing challenges with pMDI or DPI use in an ideal manner even after getting suitable training and instructions, having difficulty coordinating their breath while using a pMDI, or fail to generate sufficient inspiratory flow (PIFR) for better aerosol delivery. Patients with insufficient symptom control––those using hand-held inhalers regularly, those using hand-held inhalers irregularly or noncompliant, and those who favor using nebulizers.55,70–73 Elderly patients, people with severe disease, or with physical disability, or cognitive impairments may fail to use hand-held inhalers effectively; and in such patients, nebulizers may be of benefit.
Despite instructions, patients who fail to coordinate their inhalation with actuation of a pMDI or create insufficient PIFR while using a DPI should be advised to use nebulizers. 50 In patients who are prescribed inhaled medication, it is incumbent on prescribing physicians to assess their patients' ability to use a particular type of delivery device when determining which medication to be prescribed. It is also important to give clear instructions regarding proper inhaler technique to both patients and caregivers and to assess their use of the device to confirm correct use. 50
Pitfalls in the Diagnosis and Management of COPD in the Gulf Countries
Our review found that there is underdiagnosis and underreporting of COPD in Gulf countries. This may lead to inadequate initiation of treatment for the patients suffering with debilitating disease. There is a need to increase awareness about COPD in the region and to provide the latest tools and treatments to physicians to help their COPD patients.
In the Gulf countries, under-prescribing by physicians and barriers to medication access are two major reasons for inadequate use of certain specific treatments. Creating a larger awareness about the importance of GOLD guidelines and treatment recommendations, especially among community medicine practitioners, or awareness about local guidelines may help in overcoming this situation.
In the Gulf region, there are many shortcomings in the nonpharmacologic management of COPD, which include underdiagnosis, failure to initiate appropriate therapy at an appropriate time by general practitioners or internal medicine doctors; unavailability of spirometry for confirmation of diagnosis; patient noncompliance; different attitudes about disease and its management among patients from low socioeconomic class and sociodemographic backgrounds; absence of well-structured pulmonary rehabilitation programs; lack of dedicated/trained staff for delivering standard rehabilitation programs; and unaffordability of noninvasive ventilation, oxygen machines, and medications. 11
Patients should be advised about the benefits of smoking cessation and provide them the counseling and other therapeutic aids that may help them to quit smoking.
Summary of the Role of Nebulizers in COPD Management
Many studies and literature reviews on the role of inhaled pharmacotherapy in the treatment of COPD have highlighted the importance of nebulizers and have indicated that future long-term treatment of COPD patients will need continuous innovation and development of novel nebulizer devices and drugs for clinical situations where the current hand-held inhalers may be insufficient. 43 While selecting or prescribing an inhaled therapy, practitioners should consider factors such as available drug formulations, different drug combinations and devices, patients' pulmonary function capacity, their skills, and preferences. Available data suggest that when patients are able to use their prescribed hand-held inhaler devices appropriately, treatment delivered by these devices provide similar clinical benefits in the long term irrespective of the device prescribed. On the other hand, when hand-held inhalers are incorrectly used and patients are unable to learn to use them correctly, nebulized medications may play an important role. Health care providers and patients together can enhance the benefits of these inhaled therapies for COPD. 43
Conclusion
There is no major difference between the Gulf Countries and the rest of the world in the approach to management of COPD. Nebulizers should be considered for patients who have difficulties in accessing or using MDIs and DPIs, irrespective of geographical location.
Footnotes
Acknowledgment
The authors thank Shantha Kumar V, Ph.D, from Viatris for medical writing assistance and editorial support.
Authors' Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Author Disclosure Statement
None of the authors has any relevant disclosures related to this review. DPT has consulted with and received grants from Mylan, Sunovion, Boehringer-Ingelheim, Theravance, AstraZeneca, and Novartis. IZB has provided consultation to Astra Zeneca, Boehringer Ingelheim, CSL Behring, GE Healthcare, GlaxoSmithKline, Grifols, Mylan, Theravance Biopharma, and Verona Pharma, and obtained grants from AMGEN, GE Healthcare, Mylan, and Theravance Biopharma. JG and NB have no relevant conflicts of interest. AK and US are employees of Viatris Inc.
Funding Information
No funding was received for this article.
Reviewed by:
James Fink
Adam Wanner
