Abstract
Abstract
Background:
To evaluate the in vitro dose delivery characteristics of approved asthma and chronic obstructive pulmonary disease (COPD) therapies delivered via the ELLIPTA® dry powder inhaler across inhalation endpoints representative of the target patient population, using the Electronic Lung (eLung™) to replicate inhaler-specific patient inhalation profiles that were previously recorded in vivo.
Methods:
Selected profiles, representative of the range of inhalation endpoints achieved by patients with all severities of asthma and COPD, were replicated using the eLung breathing simulator in conjunction with an oropharyngeal cast. A Next Generation Impactor was coupled to the eLung to determine the aerodynamic particle size distribution of the ex-throat dose (ETD) of asthma and COPD therapies delivered via the ELLIPTA inhaler. Delivered dose (DD), ETD, and fine particle dose (FPD; defined as a mass of active substance less than 5 μm) were determined for fluticasone furoate (FF)/vilanterol (VI) 100/25 μg and 200/25 μg (asthma and COPD), umeclidinium (UMEC)/VI 62.5/25 μg (COPD only), FF 100 μg and 200μg monotherapy (asthma only), and UMEC 62.5 μg monotherapy (COPD only).
Results:
Inhalation profiles replicated by eLung covered a wide range of peak inspiratory flow rates (41.6–136.9 L/min), pressure drops (1.2–13.8 kPa), and inhaled volumes through the inhaler (0.7–4.2L). DD was consistent across the range of patient representative inhalation parameters for all components (FF, VI, and UMEC) of each therapy assessed; although ETD and FPD were also generally consistent, some small variation was observed. Dose delivery was consistent for each of the components, whether delivered as mono- or combination therapy.
Conclusions:
The in vitro performance of the ELLIPTA inhaler has been demonstrated for the delivery of FF/VI, UMEC/VI, FF monotherapy, and UMEC monotherapy. Across a range of inspiratory profiles, DD was consistent, while ETD and FPD showed little flow dependency.
Introduction
D
To use any DPI efficiently, patients must be able to apply sufficient inspiratory effort to emit the dose from the inhaler, and to disperse the powder formulation to create a sufficient mass of particles within the respirable size range (fine particle dose [FPD]; typically considered as mass of particles <5 μm aerodynamic diameter).(2) Some asthma and COPD patients may not be able to achieve the required inspiratory flow rate to use some DPIs efficiently,(3–5) particularly those with severely impaired lung function, for example, in very-severe COPD.(6,7)
It is therefore important to assess the inhaler-specific inhalation characteristics of the target population to confirm whether patients can achieve the minimum inspiratory effort required to use the inhaler efficiently and consistently. Furthermore, consistent performance across the range of inhalation parameters of the target population ensures that these variations do not influence or control the dose delivery and the resultant respirable mass of particles, as it has been suggested that there are correlations between FPD determined in vitro, lung deposition, and clinical response.(8)
The Electronic Lung (eLung™: a trademark of the GSK group of companies) is a breathing simulator designed for the characterization of inhalation devices,(9,10) and is capable of replicating patient inhalation profiles recorded in vivo using an inhalation profile recorder. It can be used in combination with an oropharyngeal cast, and coupled with a Cascade Impactor to allow dose delivery characteristics (including aerodynamic particle size distribution) to be assessed in vitro, using more patient-relevant conditions than standard impactor testing.
To determine whether asthma and COPD patients are capable of achieving the required inspiratory flow to use the ELLIPTA inhaler, the maximal effort inhaler-specific inhalation profiles for the target population have been characterized, and these are described separately (Prime et al., J Aerosol Med Pulm Drug Deliv, in press). In this second publication, the objective was to evaluate the in vitro dose delivery characteristics of approved therapies delivered via the ELLIPTA inhaler, using the eLung to replicate patient-specific inhalation profiles representative of patients with asthma or COPD, categorized into subgroups of varying disease severity.
Materials and Methods
Maximal effort inhaler-specific inhalation profiles were recorded and characterized from patients with mild to severe asthma or mild to very-severe COPD in two studies (Study 1: GSK study RES113817, clinicaltrials.gov NCT01345266; Study 2: RES117178, NCT02076269). Patient demographics and the results of these evaluations are described separately (Prime et al., submitted in parallel). Generally, the patients with asthma tended to be younger than those with COPD (median age range of 40.9–46.2 vs. 61.6–64.4, respectively), while those with COPD were more likely to be male than female, but no significant differences in terms of age or BMI were seen within each of the asthma or COPD groups across the disease severities.
The aerodynamic particle size distribution of the fraction of the dose passing beyond the throat (ex-throat dose [ETD]) was assessed for each of the following approved therapies, where the stated dose is equal to the nominal blister content: FF/VI 100/25 μg (label claim delivered dose: 92/22 μg), FF/VI 200/25 μg (label claim delivered dose: 184/22 μg), 11 UMEC/VI 62.5/25 μg (label claim delivered dose: 55/22 μg), 12 FF 100 μg monotherapy (label claim delivered dose: 90 μg), FF 200 μg monotherapy (label claim delivered dose: 182 μg), 13 or UMEC 62.5 μg monotherapy (label claim delivered dose: 55 μg). 14 FF/VI and UMEC/VI were delivered using the two-strip configuration of the ELLIPTA inhaler, whereas FF monotherapy and UMEC monotherapy were delivered using the single-strip configuration.
For each therapy, five inhalation profiles were sourced for the relevant device configuration and disease state(s) by sorting the appropriate full dataset of profiles by peak inspiratory flow rate (PIFR), then selecting individual profiles to represent the absolute minimum and maximum PIFRs, plus three intermediate values at the median and interquartile points. These inhalation profiles were replicated using the eLung.
The eLung was used in combination with a specific throat cast based on a previous imaging study (PP2), 15 which was considered more representative of human anatomy than the standard metal throat that would typically be used during quality control testing of inhaled products. While the general population will vary in oropharyngeal anatomical structure, the throat cast selected was deemed suitable for use in these experiments as the average cross-sectional area closely matched the median observed in imaging studies (RES113817, RES117178; GSK data on file) of healthy individuals, as well as asthma and COPD patients.
The inlet was modified with a rubber mouthpiece adaptor to enable coupling of the ELLIPTA inhaler to the cast. Prior to use, the internal surface of the anatomical cast was coated with an ethanolic Brij solution in glycerol. This was done by placing a volume of coating solution within the throat, sealing the inlet and exit, inverting in all orientations to ensure absolute coverage, and then allowing draining of any surplus solution from both inlet and exit ends prior to use. A Next Generation Impactor (NGI) was coupled to the eLung, to enable the aerodynamic particle size distribution of the ETD to be characterized. NGI cups were coated with a polyethylene glycol solution to prevent particle bounce.
In the companion report to this one (Grant et al, J Aerosol Med Pulm Drug Deliv, in press), a constant flow rate of 30, 60, or 90 L/min (4 L inhaled volume) was used in a standard impactor set-up. In this study, it was desirable to replicate complete patient inhalation profiles and to use the NGI just to characterize the resultant ETD. In order to maintain NGI stage effective cut-off diameters, the impactor was operated at a constant flow rate of 60 L/min; using this set-up, the eLung equipment permits separation of the flow conditions at which dose emission and dose characterization take place.
Dose emission takes place under variable flow conditions, representative of the patient inhaling through the inhaler, whilst constant flow is maintained through the impactor, which is a requirement of impactors to maintain effective cut-off diameters of the impactor stages. A flow rate of 60 L/min through the impactor was selected to be consistent with the GSK registered flow rate used during standard aerodynamic particle size distribution testing.
Each dose was actuated from the ELLIPTA inhaler as the eLung breathing simulator replicated the selected inhalation profile. Multiple doses (six for FF monotherapy and FF/VI; five for UMEC monotherapy and UMEC/VI) were aerosolized into the eLung set-up during one experiment replicate. To assess the total delivered dose (DD), ETD, and FPD, individual parts of the eLung/impactor apparatus were rinsed with a suitable solvent to recover the deposited mass. The solutions were assessed using high performance liquid chromatography (HPLC), performed on Agilent 1100 Series equipment. This process was repeated in triplicate for each product and inhalation profile combination. FPD was interpolated from NGI stage deposition data using a validated template.
Results
There was wide variation between the inhalation characteristics in the selected inhalation profiles: the nominal PIFRs across both the single-strip and two-strip configurations of the ELLIPTA inhaler ranged from 41.6 (very-severe COPD) to 136.9 L/min (moderate asthma); peak pressure drops ranged from 1.2 (very-severe COPD) to 13.8 kPa (moderate asthma); and nominal inhaled volume ranged from 0.7 L (very-severe COPD) to 4.2 L (mild COPD). Overall, inhalation characteristics for the profiles that were recorded from patients within the same disease severity subgroups were similar between the single-strip and two-strip configurations where these comparisons could be made (Fig. 1; Table 1). Although the data sets for each product and profile combination were relatively small (n = 3), low variability was observed.
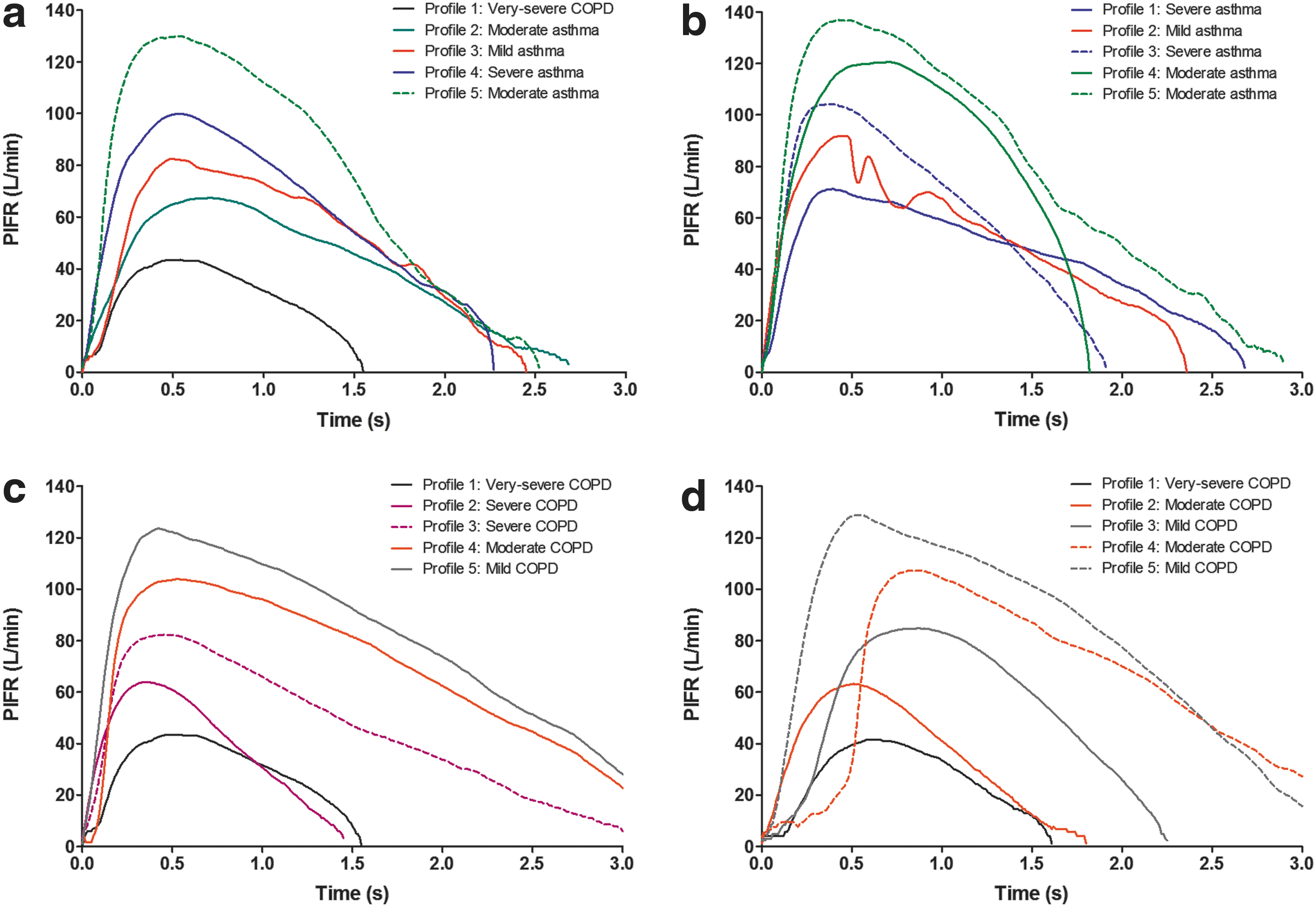
Flow rate versus time for each patient representative maximal effort inhalation profile replicated for the assessments of
COPD, chronic obstructive pulmonary disease; FF, fluticasone furoate; PIFR, peak inspiratory flow rate; UMEC, umeclidinium; VI, vilanterol.
DD, ETD, and FPD
Mean DD, ETD, and FPD (% nominal blister content) for each component of the therapies tested are presented in Tables 2, 3, and 4, respectively. There was little evidence of trends in DD, ETD, or FPD for each component across the inhalation profiles tested, with DD close to that of the stated label claims (Fig. 2). For the FF and UMEC components, DD, ETD, and FPD were consistent regardless of whether the component was delivered as part of a combination therapy or as a monotherapy. Similarly, the DD, ETD, and FPD of VI appeared to be unaffected regardless of whether it was delivered as a component of FF/VI or UMEC/VI. The DD (as % nominal blister content) of FF and VI tended to be slightly higher than those of UMEC, but this did not translate to differences in FPD; the FPDs for UMEC and VI were similar, while the FPD of FF was slightly lower than for either UMEC or VI.
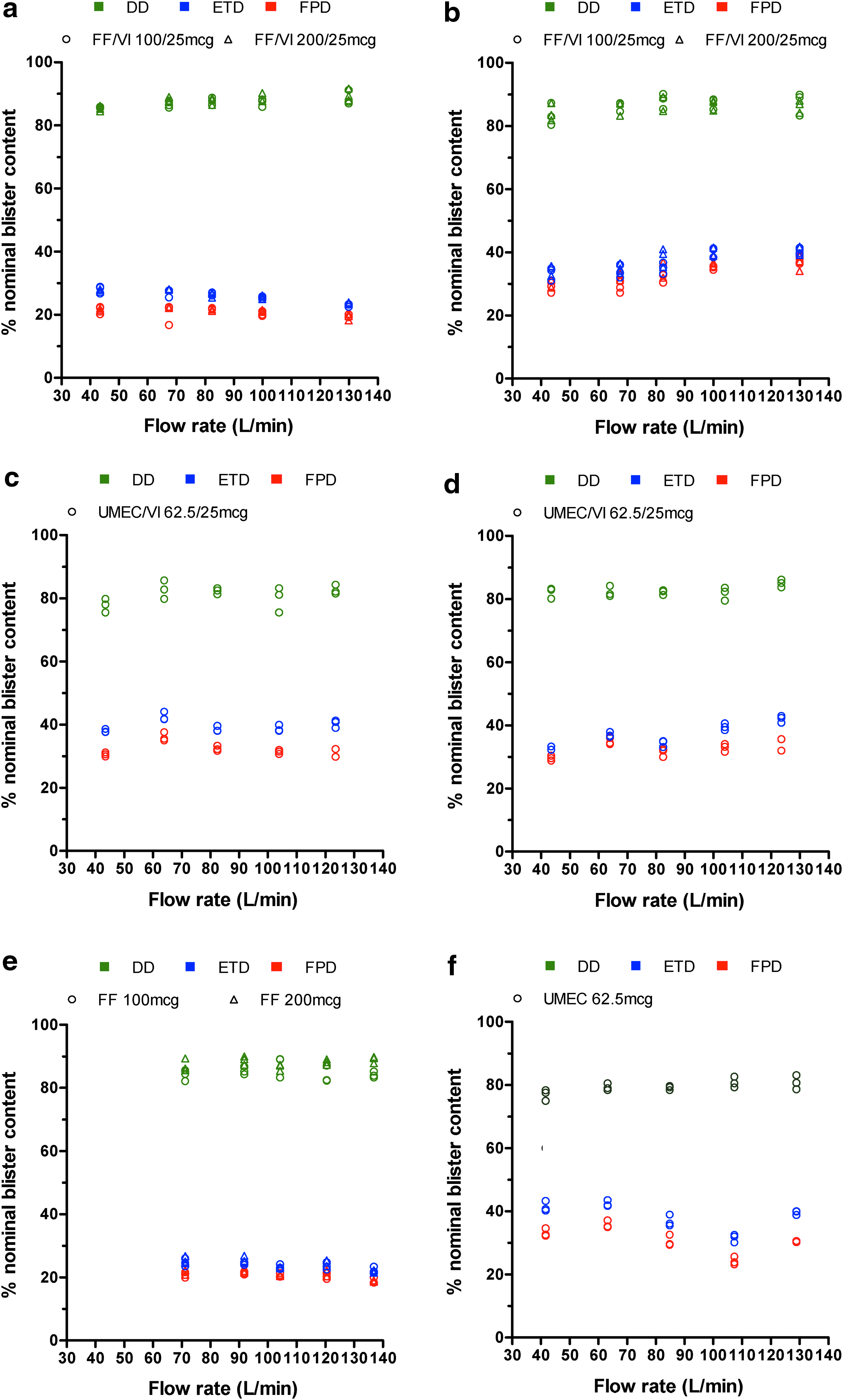
DD, ETD, and FPD (% nominal blister content) versus PIFR (L/min) for
All data are mean (range) unless otherwise stated.
COPD, chronic obstructive pulmonary disease; DD, delivered dose; FF, fluticasone furoate; PIFR, peak inspiratory flow rate; UMEC, umeclidinium; VI, vilanterol.
All data are mean (range) unless otherwise stated.
COPD, chronic obstructive pulmonary disease; ETD, ex-throat dose; FF, fluticasone furoate; PIFR, peak inspiratory flow rate; UMEC, umeclidinium; VI, vilanterol.
All data are mean (range) unless otherwise stated.
COPD, chronic obstructive pulmonary disease; FF, fluticasone furoate; FPD, fine particle dose; PIFR, peak inspiratory flow rate; UMEC, umeclidinium; VI, vilanterol.
Discussion
In this article, we have described how the eLung apparatus and an anatomically representative throat cast were used to assess the dose delivery characteristics of FF/VI, UMEC/VI, FF, and UMEC delivered via the ELLIPTA inhaler. This method enabled these assessments to be performed in a more realistic manner than standard in vitro methodologies allow, and over a greater range of inhalation endpoints, by replicating maximal effort inhalation profiles collected from the target patient population.
The set-up also allowed for exploration of a wider range of PIFRs than could be achieved with standard NGI testing, as well as achieving variable flow through the device. These in vitro assessments suggest that patients are capable of achieving the inspiratory effort required for consistent performance of the ELLIPTA inhaler when used for the delivery of the asthma and/or COPD therapies FF/VI, UMEC/VI, FF monotherapy, and UMEC monotherapy, at currently approved doses.
The inhalation profiles replicated during the eLung assessments were chosen to represent patients for whom that therapy is indicated. The replicated profiles were selected based on PIFR to represent the absolute range of previously observed inhalation parameters in asthma and COPD of all severities. From the previous assessment of inhalation profiles (Prime et al., submitted in parallel), it was established that very-severe COPD was associated with the largest negative impact on PIFR and other inhalation endpoints, and therefore it is these patients who are most likely not to be able to achieve the required inspiratory effort for efficient and consistent dosing when using a DPI. Therefore, an inhalation profile representative of a patient with very-severe COPD was used in the assessments of all COPD relevant therapies (FF/VI, UMEC/VI, and UMEC monotherapy).
For FF/VI, the other four profiles used were derived from patients with asthma, although there is overlap between the inhalation profiles and the disease severity subgroups represented (e.g., a ‘moderate asthma’ profile may also be representative of the inhalation endpoints that could be achieved by a patient with mild COPD). Despite the large differences in PIFR and other inhalation parameters for very-severe COPD patients compared with asthma patients, the DD, ETD, and FPD were similar for each of the profiles.
For UMEC/VI and UMEC monotherapy, which are only indicated in COPD, the inhalation profiles were representative of patients with COPD of all severities (including very-severe) so it was not possible to make the same comparison. Despite the trend for PIFR (and other parameters) to significantly decrease with increasing disease severity, no trends indicating that DD, ETD, and FPD decreased with increasing severity were identified for any of the therapies studied. It has been shown that the flow acceleration rate may have a significant effect on the performance of some DPIs, independently of the PIFR achieved.(16) The flow acceleration rate varied significantly between the profiles used in this study, however, in general no effect on DD, and little effect on ETD or FPD was observed.
For the individual FF and UMEC components of each of the therapies tested, the DD, ETD, and FPD were consistent whether they were delivered alone or simultaneously with another compound as part of a combination therapy. Although there was some slight variation between FF, UMEC, and VI for the DD, ETD, and FPD, these differences were not considered to be clinically important; this finding is supported by observations from Phase III clinical trials in which clinical efficacy was demonstrated, even in very-severe COPD patients, for FF/VI,(17,18) and UMEC/VI and UMEC monotherapy(19–22) delivered via the ELLIPTA inhaler. Efficacy of FF monotherapy has also been demonstrated, but only in asthma patients as this treatment is not indicated in COPD.(23–25)
Although the data set was not large enough to perform formal statistical analysis, these findings provide a useful indication that the ELLIPTA inhaler performs consistently across a wide range of patient relevant inhalation profiles and should therefore be suitable for the majority of patients. This is supported by the fact that there was no tail off in performance for any of the profiles replicated, including that of the very-severe COPD patient group with the absolute minimum observed PIFR. It is further supported by previous in vitro dose delivery assessments, which showed that the delivered dose from the ELLIPTA inhaler for all products assessed was close to the stated label claim at flow rates of 30, 60, and 90 L/min under standard impactor conditions (Grant et al., J Aerosol Med Pulm Drug Deliv, in press).
The results of in vitro dose delivery characteristics of the ELLIPTA inhaler described herein were generated through replication of inhalation profiles previously recorded in a study using a maximal effort instruction for comparison with inhaler-independent lung function assessments (Prime et al., submitted in parallel). It is recognized that the ‘long, steady, deep’ inhalation instruction is described in the patient information leaflet. Despite this difference, our assessments included a wide range of inhalation profile parameters and therefore the results should still be relevant to a wide range of adult patients with mild to severe asthma and mild to very-severe COPD.
The selection of inhalation profiles based on PIFR is relevant because of the variety of profiles covered, and the fact that these profiles, which were obtained in vivo, will also cover a range of other inhalation parameters that are affected by disease severity (e.g., inhaled volume and acceleration rate). It should also be noted that this study did not replicate any profiles previously recorded from patients experiencing exacerbations or in remission from an exacerbation. Future studies may focus on representing patients with inhalation parameters that may differ from those already investigated, and may include pediatric subjects for whom these results are not applicable.
Finally, it should also be noted that the effectiveness of any inhaled therapy is not just dependent on the dose delivery characteristics of the inhaler, but also on patient inhaler preference, ease of use, and inhaler handling, which have been reported elsewhere for the ELLIPTA inhaler.(26–28) As noted previously, this study only performed three replicates for each of the inhalation profiles. This number could perhaps be increased in any future experiments to ensure a wider range of data. In addition, as this study included data for only one example of throat geometry, a wider study exploring the impact of variability in this area may be worthy of future investigation, particularly with regard to the potential effect on throat size of imaging obtained in a supine versus upright position.
Conclusions
The in vitro dosing performance of the ELLIPTA DPI has been established through replication of a range of patients' inhaler-specific maximal effort inhalation profiles, which was achieved using the eLung breathing simulator, in conjunction with an anatomically representative oropharyngeal cast of average size. Using a range of inhalation parameters representative of patients with all severities of asthma or COPD (PIFR: 41.6–136.9 L/min; inhaled volume: 0.7–4.2 L), dose delivery of all therapies was consistent and reproducible with little flow dependency.
Footnotes
Acknowledgments
These studies were funded by GSK. Inhalation profiles were derived using data from studies RES113817 (![]() NCT01345266) and RES117178 (NCT02076269).
NCT01345266) and RES117178 (NCT02076269).
All listed meet the criteria for authorship set forth by the International Committee for Medical Journal Editors. MH, DP, RL, and BG were involved in the conception and design of the study; MH, CP, and SC were involved in the acquisition of data; and MH, DP, RL, CP, SC, and BG were involved in data analysis and interpretation.
The authors acknowledge the late Dr. Patricia KP Burnell, and Emma Lindo, Richard Atkinson, Maribel Bogalo-Huescar, members of the RES113817 and RES117178 clinical study teams and members of Inhaled Delivery Science (GSK, Ware, UK) for their contributions towards this work. Editorial support for development of early drafts of this manuscript, in the form of development of the manuscript first draft, editorial suggestions to draft versions of this article, assembling tables and figures, collating author comments, and referencing was provided by Diana Jones of Cambrian Clinical Associates Ltd. and was funded by GSK.
Editorial support in the form of development of final drafts of this manuscript in consultation with the authors, editorial suggestions to draft versions of this article, assembling tables and figures, collating author comments, copyediting, fact checking, referencing, and graphic services was provided by Jacqueline Phillipson, PhD, and Laura Maguire, MChem at Gardiner-Caldwell Communications (Macclesfield, UK) and was funded by GSK.
Author Disclosure Statement
All authors are employed by and hold stock in the study sponsor, GSK.
