Abstract
Abstract
Background:
Research on the use of a pressurized metered dose inhaler (pMDI) with spacer (pMDI/spacer) in children has indicated oral inhalation via the spacer mouthpiece is more efficient than the combination of oral and nasal inhalation that occurs when a pMDI/spacer is used with a facemask. Changes in pMDI formulations and developments in spacer and facemask designs have highlighted the need for new comparative studies of spacer use, particularly focusing on the age at which children can be taught to transition from use of a pMDI/spacer with facemask to use of the spacer mouthpiece.
Methods:
Twelve children aged 3–5 years (7 males) with stable asthma were recruited. Of these, 10 children (6 males) completed both arms of the study. A transmission scan of each compliant subject was taken using a 37 MBq 99mTc flood source. Actuations (2–3) of a 99mTc-radiolabeled albuterol pMDI were administered through an antistatic spacer (OptiChamber Diamond) via either a facemask (medium LiteTouch facemask), or the spacer mouthpiece. The subject's inhalation pattern was simultaneously recorded using a pMDI Datalogger, and narrative data relating to tolerance and compliance were documented. Anterior and posterior planar scintigraphic scans were taken immediately after aerosol administration.
Results:
Mean (SD) lung deposition (% total dose) was 18.1 (9.1)% with the facemask and 22.5 (7.9)% with the spacer mouthpiece (p>0.05). Peripheral lung deposition (expressed as peripheral:central (P:C) ratio) was higher in 7 out of 10 children with the facemask compared with the spacer mouthpiece: 1.3 (0.26) vs. 1.2 (0.35); (p=0.11). Head and neck deposition was higher with use of the facemask compared with the spacer mouthpiece: 19.7 (10.6)% vs. 10.8 (5.3)% (p=0.011).
Conclusions:
Lung deposition achieved using the spacer with facemask was higher than previously reported, with a difference of only 4.4% of total dose measured compared to the deposition with mouthpiece. This may be due to a combination of factors including pMDI formulation, and use of an antistatic spacer with a flexible, well-fitting facemask.
Introduction
A
In infants and children up to 4–5 years of age who cannot use a mouthpiece, a facemask is the preferred interface between the spacer and the face of the child.(7,8) Facemasks attached to spacers have made it possible to deliver aerosols even to young infants.(9) The design of the facemask is critical, because leaks between the facemask and the face of the child can greatly diminish the amount of aerosol inhaled.(10,11) Results from in vivo and in vitro studies indicate that the facemask can be the most important component of an aerosol delivery system.(12–15) The precise age at which children can reliably and consistently be trained to use a pMDI/spacer with a mouthpiece has not been established.(16–18)
Thus, although a pMDI/spacer with a mouthpiece is generally thought to result in higher lung deposition when delivering aerosols to infants and young children, guidelines indicate that the pMDI/spacer should be used with a facemask.(7,8) There is limited clinical information on the impact of aerosol delivery through a pMDI/spacer facemask in comparison with a mouthpiece. Roller et al. investigated lung deposition following use of a pMDI spacer with mouthpiece using 99mTc-radiolabeled HFA beclomethasone dipropionate (QVAR) in children aged 5–17 years.(19)
In vitro tests indicate that the development of antistatic spacers and contemporary soft well-sealing facemasks has improved aerosol delivery,(20) in comparison with older spacers and facemasks.(21) As scintigraphic imaging has been previously used to assess lung deposition of inhaled bronchodilators and corticosteroids in children,(19,22) it is of interest to use this technique to evaluate contemporary spacer–facemask combinations in terms of lung deposition.
Our primary objective was to compare drug delivery when inhaled from a pMDI/spacer with either a facemask or mouthpiece, by measuring the lung and oropharyngeal deposition of 99mTc-radiolabeled albuterol in children diagnosed with asthma aged 3–5 years. Data relating to the breathing pattern and parameters of breathing associated with use of the 2 inhalation methods were also recorded using an electronic monitor: the pMDI Datalogger (Fig. 1).
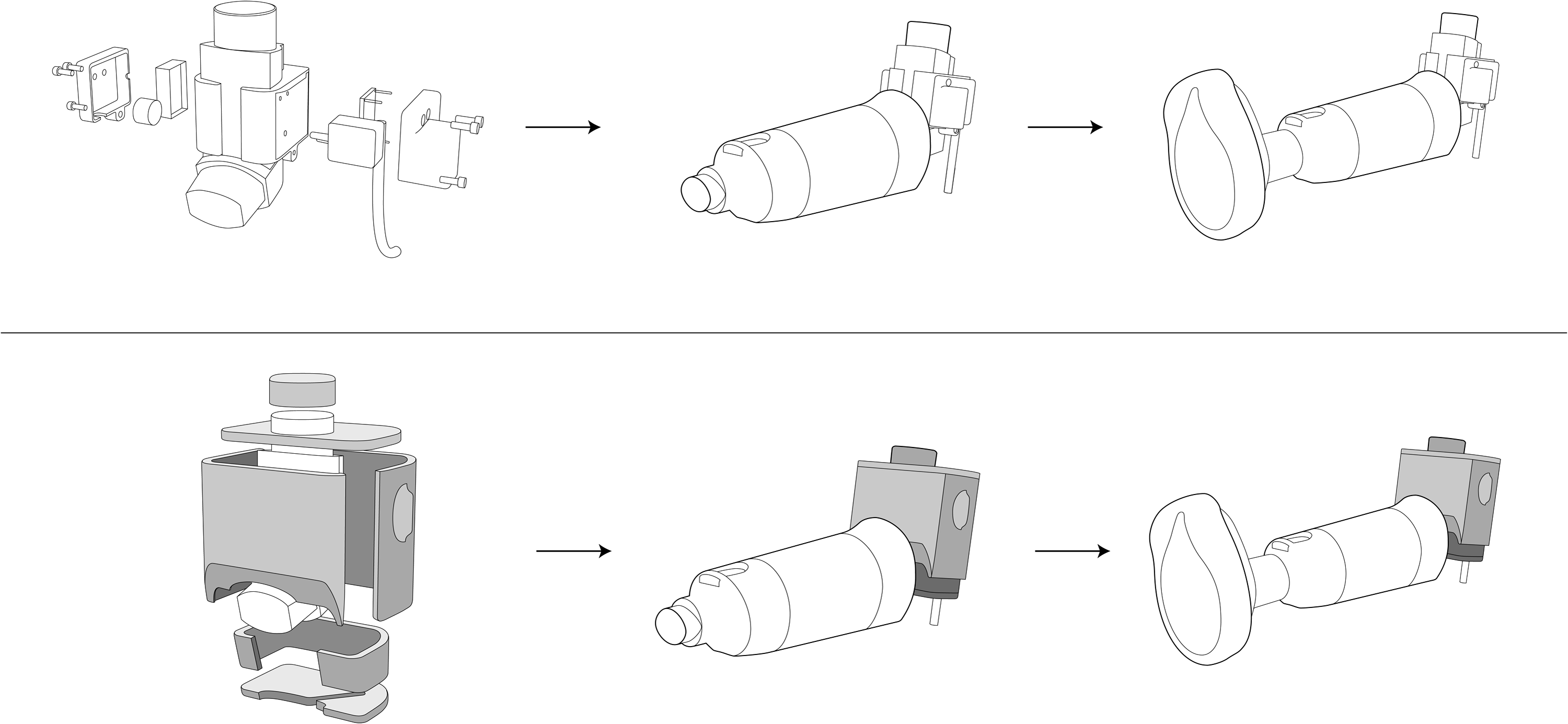
pMDI Datalogger with pMDI, Diamond spacer, and medium LiteTouch facemask. Lower portion of the figure shows setup with a lead shield (shaded areas) for gamma scintigraphy tests.
Materials and Methods
Study design
The study was performed at Princess Margaret Hospital in Perth, Australia, and designed as a randomized, open label, cross-over study during which 99mTc-radiolabeled albuterol was administered to children (3–5 years) with mild, stable asthma, using an albuterol-HFA pMDI (ProAir 90 μg HFA; Teva Respiratory LLC, Horsham, PA, USA) and a spacer (OptiChamber Diamond (Diamond); Respironics Respiratory Drug Delivery (UK) Ltd, a business of Philips Electronics UK Limited, Chichester, West Sussex, UK). The spacer was used with both the standard mouthpiece and a facemask (medium LiteTouch facemask; Respironics Respiratory Drug Delivery (UK) Ltd). The order in which the spacer interfaces were used was based on a randomized balanced block design.
The study was conducted in accordance with the principles of the Declaration of Helsinki and in accordance with Good Clinical Practice. The study protocol was approved by the human research ethics committee at Princess Margaret Hospital. Informed consent was obtained from the parents of the children participating in the study.
The subjects underwent a pre-study evaluation including a clinical examination, and were trained in the optimal use of the study device. All subjects completed a screening visit and two study visits.
During each study visit, every subject participated in the following:
• Training in the use of the study device (pMDI spacer with facemask and/or with pMDI spacer without facemask if necessary); • An anterior transmission scan; • An inhalation of study drug with either mouthpiece or facemask attached to the spacer; • A simultaneous anterior/posterior gamma camera scan.
The subjects were randomly allocated to the use of either a facemask or a mouthpiece on the first deposition visit. Study subjects were asked to return within 3–7 days for a second deposition visit when they performed the second arm of the study.
The Diamond spacer was small and transparent, and could be used with either a mouthpiece or a facemask.(23) The medium LiteTouch facemask, intended for use with the Diamond spacer, included a soft, contoured silicone edge and a rigid, transparent shell.
During the screening visit and prior to inhalation of radiolabeled drug on the study visits, a placebo pMDI and spacer were used to practice the inhalation technique, and to ensure that the subjects were accustomed to the equipment. Subjects were asked to inhale the drug in tidal breaths through the spacer using both the mouthpiece and the facemask. Aerosol was administered via the pMDI/spacer by the study investigators or other medical personnel. The pMDI was actuated at the beginning of an inhalation.
HFA-albuterol radiolabeling and validation
Radiolabeling of HFA-albuterol was performed using a method previously described by Leach.(24) Sodium pertechnetate (Na99mTcO4) was extracted into chloroform as tetraphenylarsonium pertechnetate (AsPh4TcO4), followed by evaporation of the chloroform under a gentle stream of nitrogen. A commercial canister of albuterol-HFA from the same batch as those used in the study was weighed and cooled in liquid nitrogen for 60 sec. The top was cut off and the contents were rapidly poured into the pre-chilled 99mTc lined canister. The canister was crimped with a new valve and then reweighed to determine the propellant loss. At all stages of the process, potential contamination was monitored with a scintillation meter (Type 540, Mini Instruments Ltd, Burnham-on-Crouch, UK).
Prior to commencing the in vivo studies, a pre-study validation of the radiolabeling method was performed by comparing the ratio of both the emitted drug and radiolabel in the fine particle fraction from the radiolabeled pMDI canister with the mean of the drug in the fine particle fraction from 10 commercial (nonradiolabeled) pMDI canisters. The radiolabeling method was considered acceptable once a set of 10 sequential radiolabeling runs was performed, with the ratios mentioned previously falling within the range 0.8–1.2.(25)
During the in vivo phase of the study, for quality control, a comparison was made between the 99mTc distribution in the freshly radiolabeled pMDI, and both the particle size distribution of albuterol obtained from a nonradiolabeled (commercial) pMDI and the activity distribution in all the previously radiolabeled pMDIs. This was performed on each study day and was intended to ensure that the radiolabeling procedure was successful, and that the radiolabeled pMDI canister was usable. The activity per actuation was measured, and if the calculated activity per actuation was sufficient to ensure that the correct dose for the subject could be attained in 1–3 actuations, the distribution of radioactivity in the aerosol was then determined by cascade impaction, as outlined below.
Measuring the particle size distribution before and after radiolabeling was undertaken to confirm that the contents of the commercial canister were not changed by the radiolabeling procedure. Measurement of the distribution of radioactivity in the radiolabeled aerosol ensured that the patterns of lung deposition seen using gamma scintigraphy would reflect the patterns of drug deposition in the lung. Particle sizing was conducted using an 8-stage nonviable Anderson Cascade Impactor (Copley Scientific, Nottingham, UK). The pMDI with radiolabeled albuterol was initially discharged twice into a glass fiber filter, and the activity per actuation was determined by measuring the activity of the filter in a radioisotope calibrator (CRC-SRB, Capintec, Ramsey, NJ) after these actuations. The pMDI was primed and then inserted into the mouthpiece adaptor of the cascade impactor. Ten doses of HFA-albuterol (90 μg/actuation) were drawn into the cascade impactor at a continuous flow of 28.3 L/min, in order to optimize analytical sensitivity and to minimize bounce effects.(26) The pMDI was shaken for 5 sec between each actuation. The activity of the entire canister was measured before and after the ten actuations to enable further assessment of the activity per actuation.
Particles were either deposited on the standard USP “throat,” the jet stage, or one of the eight impaction plates and absolute filter, depending on aerodynamic size. Particles were recovered by washing each stage with 20 mL methanol into a 25 mL volumetric flask containing 2.5 mL of 0.1 M sodium hydroxide (NaOH), and making up to volume with methanol. This process was conducted behind a lead glass screen. The activity of each flask was measured in the radioisotope calibrator. The percentage of the total activity detected on each component was calculated. The 25-mL volumetric flasks were then placed in a lead box and the activity was allowed to decay to undetectable levels before the drug particle size distribution was determined spectrophotometrically. The absorbance of albuterol was measured in each wash by UV spectrophotometry at λ=246 nm (Shimadzu UV 1601, Kyoto, Japan). A standard curve for albuterol was determined by measuring the absorbance of solutions containing 5, 10, 20, 30, and 40 μg/mL albuterol in methanol/10% 0.1 M NaOH. This was linear (r2=0.99) for concentrations up to 40 μg/mL. The mass median aerodynamic diameter (MMAD) of albuterol from each canister used for inhalation studies (n=20) was calculated from these results using the method described in ISO standard 27427.(27) The percentage of drug and activity recovered was divided into 4 fractions: (1) actuator, (2) USP throat, (3) sum of impactor jet and plate 0 to plate 2, and (4) sum of impactor plates 3 to 7 and the absolute filter (Fig. 2); fraction (4) was defined as the fine particle fraction (FPF).
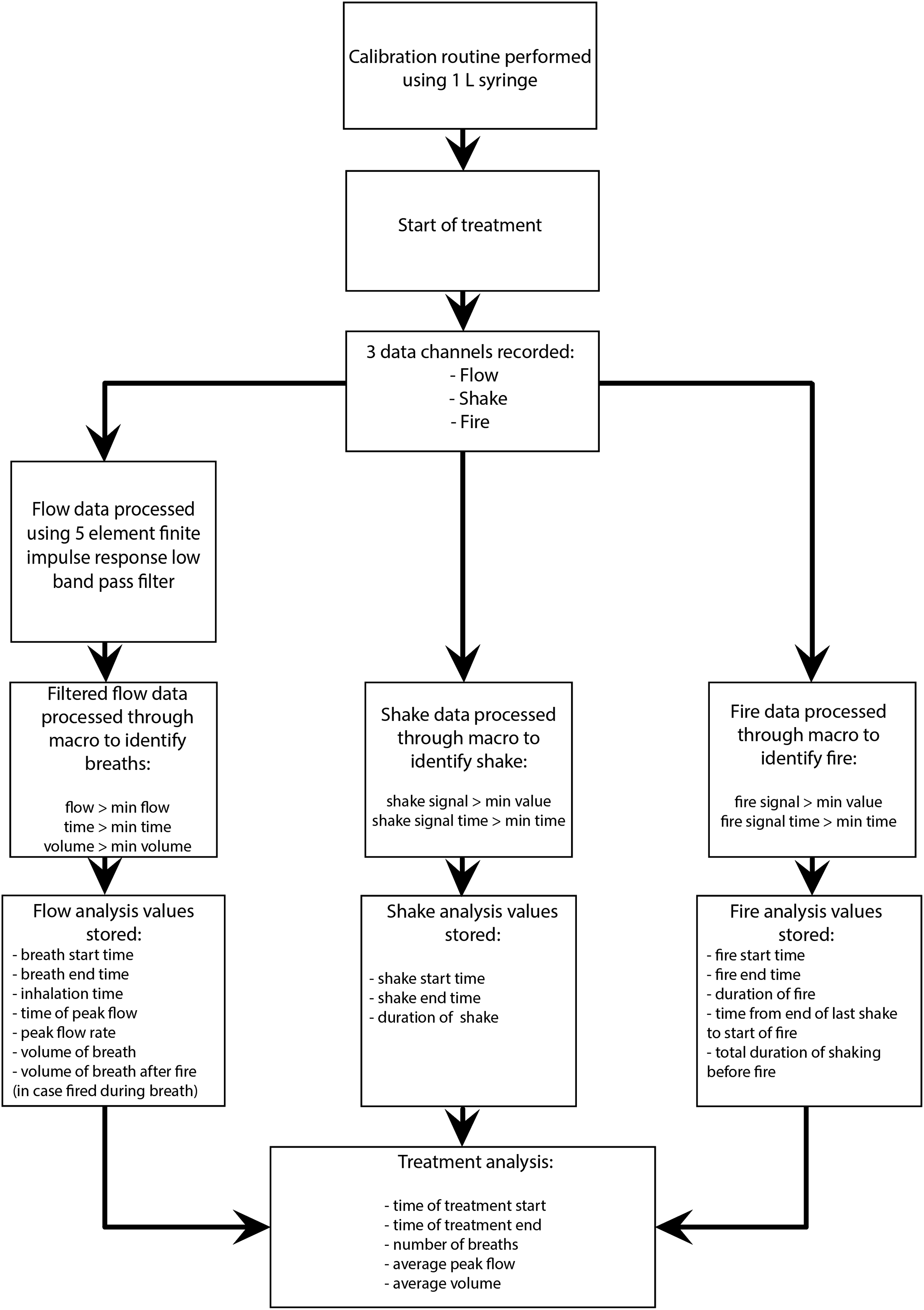
Collection and processing of pMDI Datalogger data.
Scintigraphy
A double-headed gamma camera (Siemens ECam, Siemens, Perth, Australia) was used for scintigraphic imaging. Each subject had an initial 2 minute anterior transmission scan in the supine position, using a uniform flood source containing 37 MBq technetium (99mTc). Attenuation factors were derived for each subject to compensate for absorption by body tissues, as described by Macey and Marshall.(28) After the transmission scan, each subject inhaled two or three doses of 99mTc-radiolabeled albuterol (180–270 μg albuterol) so that the dose of radiation administered was within the range of 2–4 MBq. Immediately after inhaling the 99mTc-radiolabeled albuterol, simultaneous anterior and posterior planar scintigraphic images (120 sec acquisition time) of the chest and abdomen, together with lateral images of the head and neck (the child's head was turned to one side), were obtained using the double-headed gamma camera.
Areas of interest were defined on each of the images, and separate count rates were determined for these areas, namely right and left lungs, stomach, esophagus, and oropharynx/mouth/nose/face. Each count rate was corrected for background counts and tissue attenuation, and the geometric means of the corresponding anterior and posterior count rates were then calculated. Gamma camera scans of the pMDI/spacer and filters were also obtained, and the count rates were corrected for background counts and decay time.
The dose deposited in the lungs was expressed as a percentage of the total dose ex-valve; this was either adherent to the actuator, spacer, or facemask, deposited in the body, or exhaled and captured on a filter in a funnel held close to the exhalation valve of the spacer, with air being drawn through it by means of a vacuum pump. The pulmonary regional distribution was determined by calculating the ratio of peripherally and centrally deposited activity. The central region was defined as half the width of the lung and the central one-third of the height, and the remaining region was described as the peripheral region.(29)
The maximum level of radiation associated with the scintigraphic procedure (2–4 MBq in children <6 years) was approved by the Radiation Safety Officer at Royal Perth Hospital, and equated to the additional radiation exposure from a 12 hour plane flight, or <3 weeks natural background radiation.
pMDI Datalogger
The pMDI Datalogger was a recording instrument attached to a modified plastic pMDI actuator boot with a custom designed lead shield to enable parameters of pMDI/spacer use to be monitored (Fig. 1). The Datalogger comprised:
• An ultrasonic detector, used to monitor activation of the pMDI; • An accelerometer, used to monitor shaking of the pMDI; • A flow sensor, used to monitor inhalation through the pMDI.
The sensors were wired to an electronics enclosure where the signals were converted for communication to a PC via a USB port.
The pMDI Datalogger housed the three sensors on the outside of the plastic pMDI actuator boot and allowed the operator to use the pMDI in a normal manner with minimal interference from the instrumentation. Data was sampled using a Measurement Computing data acquisition software package (Measurement Computing Corporation, Norton, MA); measurements from each of the sensors were taken 100 times a second to give a detailed recording of the breathing pattern. During use, the device remained connected to a PC via a USB cable, through which the unit was powered.
Breathing patterns and subject narratives
The pMDI Datalogger provided a detailed data output, which included information on the performance and timing of device shaking and actuation, peak inspiratory flow rate (L/min), inspiratory volume per breath, mean inspiratory volume across breaths per treatment, and number of breaths taken per treatment.
Raw data recorded by the pMDI Datalogger was placed into a Microsoft Excel worksheet and processed using a macro to remove noise from the signal, as outlined in Figure 2. This filtered data was analyzed with a focus on the three aspects of device use (shake, flow, and fire) and key breathing pattern parameters.
The analyzed information, presented in a graphical form (Fig. 3), is intended to aid data interpretation and may be complemented by descriptive data relating to the subject's cooperation during the administration of the aerosol. For the purpose of this study, key breathing pattern parameters of interest included inhalation time, inhalation duration:exhalation duration, (I:E) ratio, volume of inhalation (L), and peak inhalation flow (L/min).
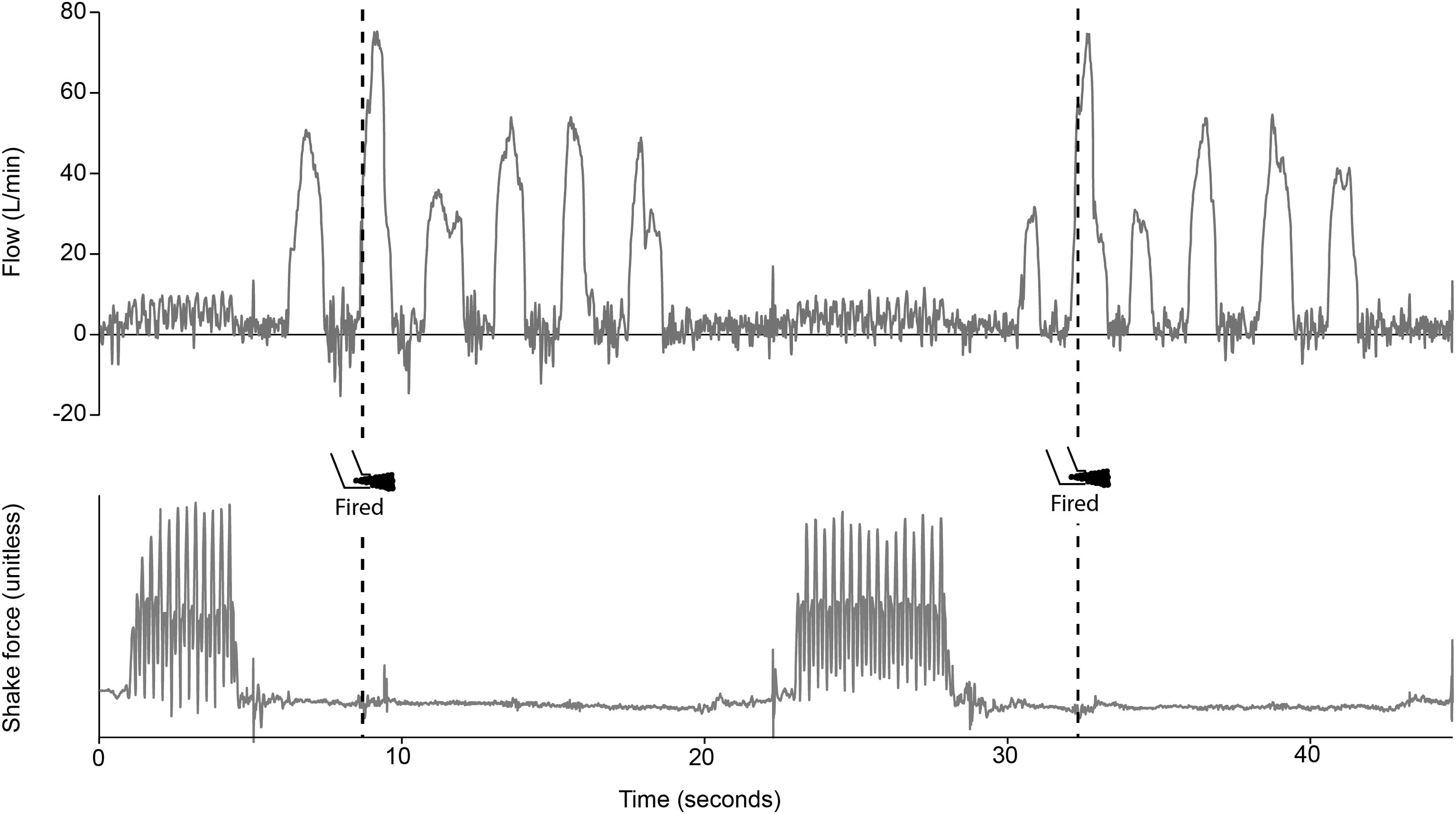
pMDI Datalogger output from macro: example analysis. Well-tolerated treatment via pMDI/spacer with facemask (Subject 1). Note the thorough shake (bottom analysis) and short delay between the end of shaking and beginning of inhalation (top analysis), followed by the well timed actuation (firing) of the pMDI at the beginning of the second breath. The recording shows the procedure for both actuations required to give the correct dose of 99mTc-radiolabeled albuterol to the subject.
Descriptive narratives were considered to be an important addition to the data provided by the pMDI Datalogger, given that a subject's level of cooperation has previously been shown to be a principal determinant of in vivo drug delivery.(30) During recording visits, qualitative data in the form of descriptive subject narratives were collected by the study investigators. This involved a subjective account of the subject's tolerance to/cooperation with treatment administration, and observed compliance with correct technique. Any departures from protocol were also noted.
The data presented here also allow comparison of key breathing pattern parameters and corresponding drug delivery, achieved with the use of a pMDI/spacer with facemask, compared with that achieved when the same subjects inhaled via the spacer mouthpiece.
Sample size and statistical analysis
The sample size was calculated based on the assumption of α=0.05, β=0.1, and δ=10%.
Estimates of variability were obtained from a study conducted by Wildhaber et al.(22)
• Sample size calculation based on lung deposition using a facemask in children <4 years (16.4±5.5%) (n=8). Minimum sample size=7 subjects.
• Sample size calculation based on lung deposition using a mouthpiece in children aged 4–7 years (28.2±6.7%) (n=5). Minimum sample size=10 subjects.
Based on these calculations, 12 subjects were recruited to participate in the present study. Efforts were made to enroll an equal number of subjects per year of age.
The percent ex-valve dose deposited in the lung; head and neck region (comprising deposition in the oropharynx/mouth/nose/face); extrapulmonary region (comprising deposition in the stomach, esophagus, and oropharynx/mouth/nose/face); and deposited on the actuator, spacer, mouthpiece, facemask, and exhalation filter was assessed. The ratio of central to peripheral lung counts determined immediately post-dosing was also assessed.
Comparison of data between mouthpiece or facemask interfaces was achieved using paired 2-tailed t-tests to look for differences in the means of the results for each parameter measured with each interface.
Results
Study subjects
Twelve children were assessed for eligibility, and randomized to use of the Diamond spacer with mouthpiece, or Diamond spacer with medium LiteTouch facemask for the initial recording visit. No subjects were excluded or lost to follow-up. Six of the recruited 12 subjects were randomized to undertake the initial inhalation recording via the pMDI/Diamond spacer with facemask, and 6 subjects first undertook the inhalation recording using the pMDI/Diamond spacer mouthpiece.
Full data sets (Diamond spacer mouthpiece and medium LiteTouch facemask) comparing subject deposition patterns were available for 10 subjects, (6 male, mean age at visit 1=4.77 years). One subject (female, age 3.42 years) failed to complete both arms of the trial and her deposition data is not included in the analysis. One child (male, age 4.5 years) refused to participate after the initial screening visit and induction onto the trial.
HFA-albuterol radiolabeling and validation
Validation that the radiolabel distribution matched that of the drug was calculated in two ways. At the time this study was conducted, the validation guidelines of Snell and Ganderton(25) were in use; the radiolabeling method was validated using these guidelines, and in vivo studies commenced on that basis. These guidelines state that the ratio of the FPF of the radiolabel to that of the commercial drug should be within 0.8 to 1.2, and Table 1 demonstrates that this was the case. However, more recent guidelines for validation of radiolabeling methods were subsequently published,(31) and the labeling method was retrospectively tested against these new parameters. The ratio of the FPF of the radiolabel to that of the commercial drug was within 0.85–1.18, which meets the first part of the requirements; however, there was some activity not associated with drug on stages 6 and 7, and this may have been as a result of the formulation of the HFA propellant in these pMDIs. In addition, the ratio of the radiolabeled albuterol in the FPF to that of albuterol in the FPF of the commercial product was within 0.85–1.18. The mass transfer of the contents of the commercial canister to the new freshly crimped radiolabeled canister was over 90% in all cases, and we specified >90% mass transfer of contents as acceptable.
The percentage of drug and activity contained in the actuator, USP throat, large particle fraction, and fine particle fraction are shown in Figure 4.

Radiolabel validation. Activity in labeled drug (□), radiolabeled drug ( ), and unlabeled drug (■).
), and unlabeled drug (■).
The fine particle fraction of both albuterol and radiolabeled albuterol was calculated as the percentage of the ex-valve dose in particles <4.7 μm in diameter (sum of Anderson stages from plate 3 to filter), and the MMADs were calculated as 2.56 μm and 2.77 μm, respectively.
Subject narratives
Complete subject narrative information was available for all of the 12 subjects (Table 2). One subject (subject 11) was too intolerant of the procedure to allow the full imaging protocol to be completed. One subject (subject 5) refused to participate at all. Narrative information provided an additional reference which complemented the breathing pattern data collected via the pMDI Datalogger and the scintigraphic images collected using the gamma camera.
Overall, the subjective narrative information suggested that there was little difference in subject tolerance and compliance when using the two interfaces (Table 2). Most of the subjects demonstrated good tolerance with both interfaces, but good compliance with correct technique was less evident with the facemask, although most subjects still managed ‘good’ or ‘satisfactory’ compliance with this interface. Compliance was assessed by the operator administering the aerosol, in terms of depth of breathing, movement of the child, number of breaths taken after each actuation, and mouth sealing around the mouthpiece.
Assessment of scintigraphic dose deposition
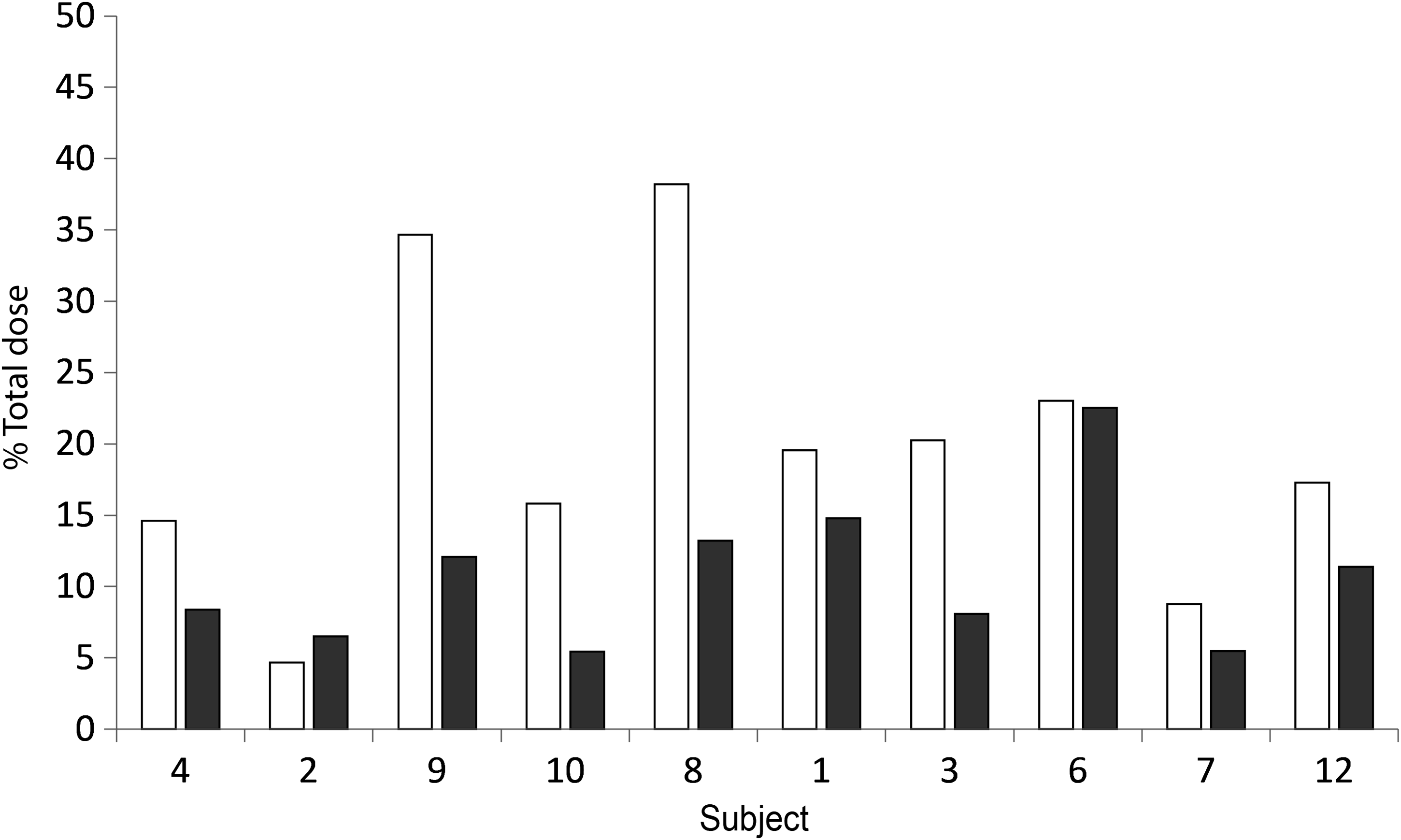
Radiolabeled albuterol deposition in the lung as a percent of total dose measured for each interface and each subject is shown in Figure 5. Mean (SD) lung deposition (% total dose) was 18.1 (9.1)% with the facemask and 22.5 (7.9)% with the spacer mouthpiece (p>0.05). Although the lung deposition for the majority of subjects was greater with use of the mouthpiece compared with the facemask, and the overall mean lung deposition for mouthpiece use was greater (Fig. 6), the difference was not statistically significant (Table 3). Visual inspection of Figure 5 indicated no age-related trends in lung deposition for the two interfaces. In contrast, the by-subject head and neck deposition was higher for the majority of the subjects with use of the facemask (Fig. 7), and the mean difference was statistically significant in favor of lower deposition in the head and neck region with use of the mouthpiece (facemask; 19.7 (10.6)% vs. mouthpiece; 10.8 (5.3)%, p=0.011). (Table 3).
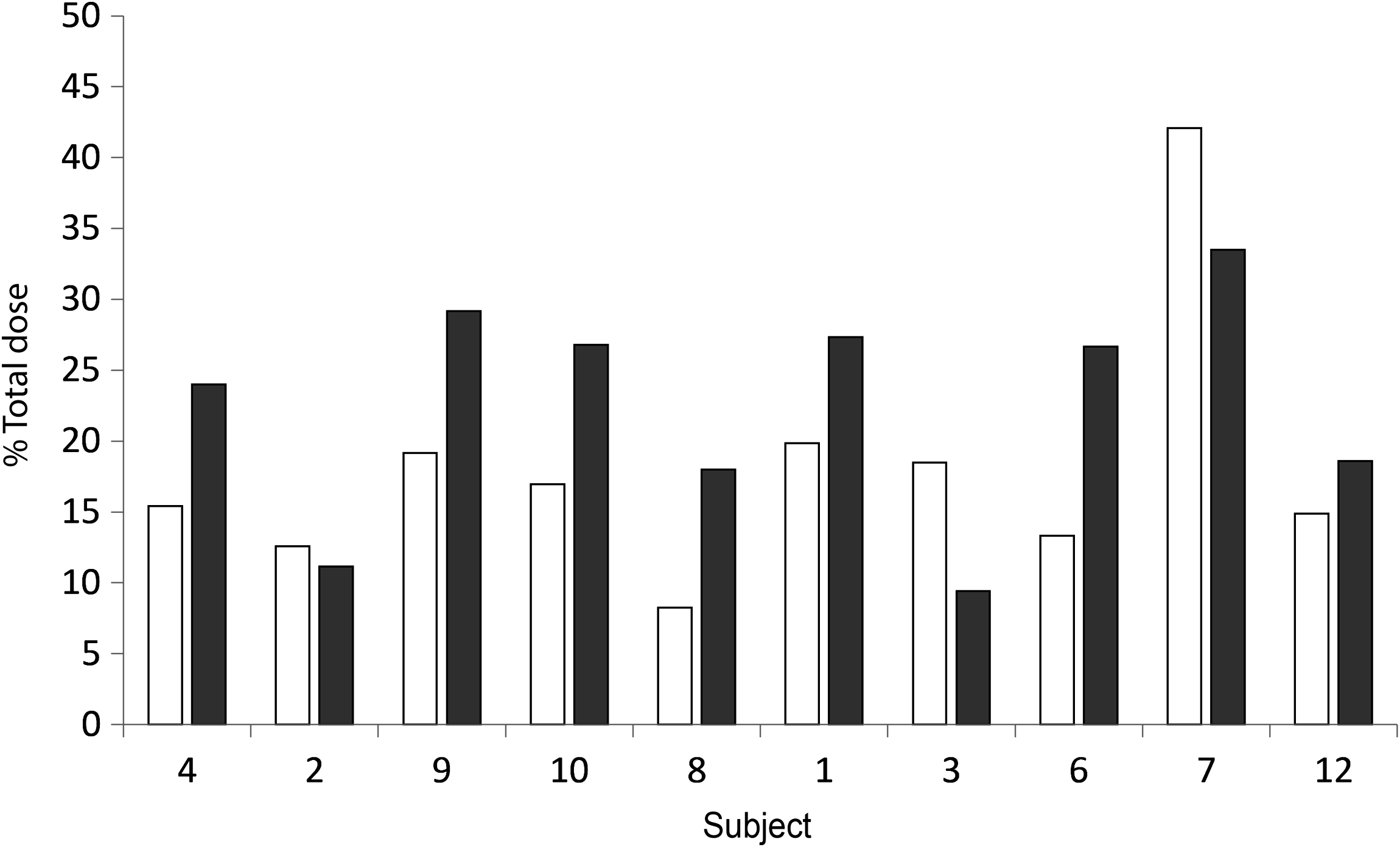
Percent of total dose deposition; lung dose. Facemask (□) and mouthpiece (■). Sorted by age; youngest first.
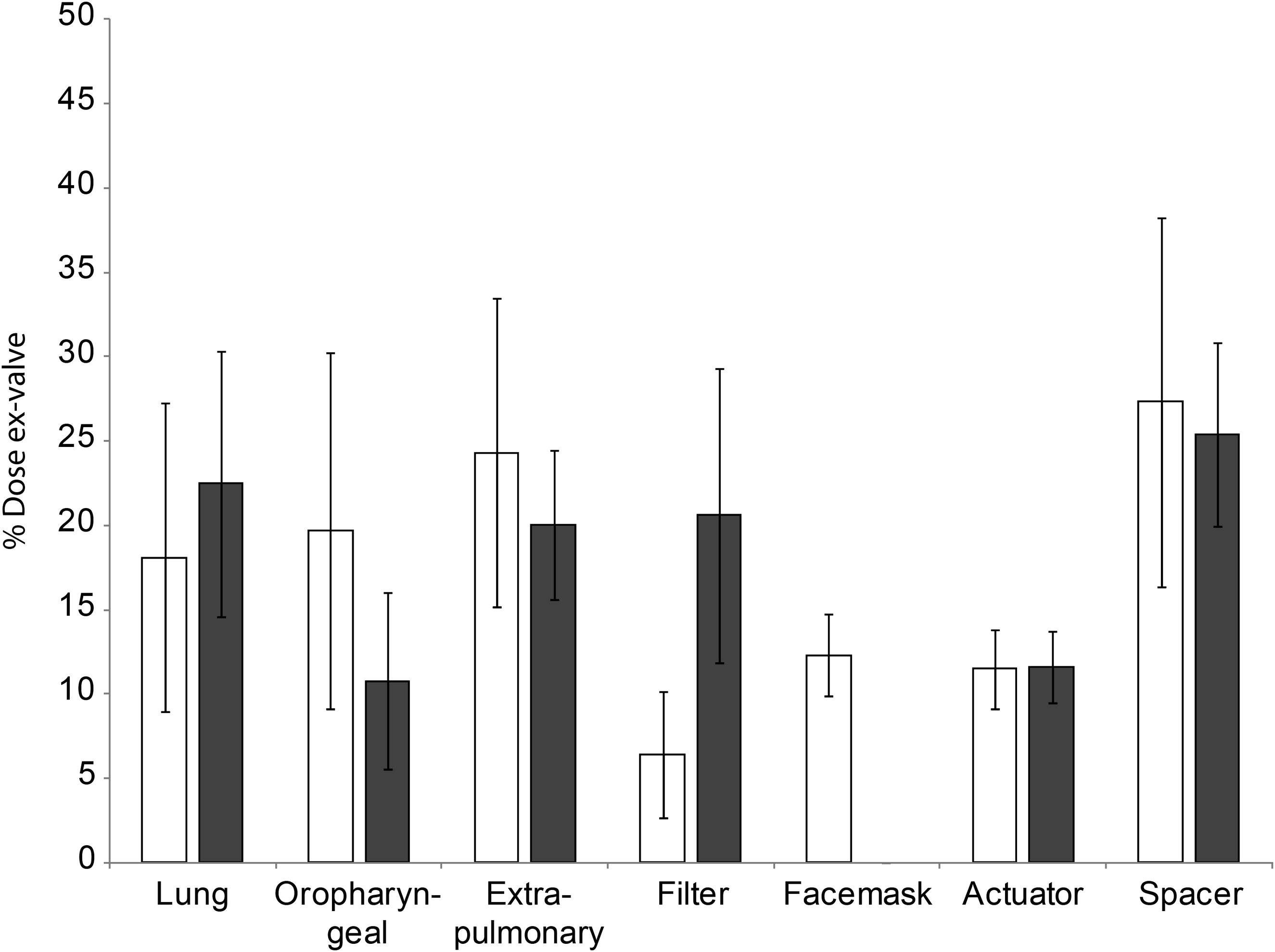
Percent of total dose deposition; mean dose deposited throughout system. Facemask (□) and mouthpiece (■). Error bars represent standard deviation.
Percent of total dose deposition; head and neck dose. Facemask (□) and mouthpiece (■). Sorted by age; youngest first.
Figures in
The dose of drug deposited in the extra-pulmonary regions has the potential to cause drug-dependent side-effects. For this reason, the extra-pulmonary deposition was also compared for the two interfaces. By-subject extra-pulmonary dose deposition is shown in Figure 8, and indicates no clear association between spacer interface and extra-pulmonary dose, though there was a trend toward greater deposition through use of the facemask. Comparison of mean extra-pulmonary dose deposition (Fig. 6) also showed a greater extra-pulmonary dose with the facemask compared with the mouthpiece, but once again this difference was not statistically significant.
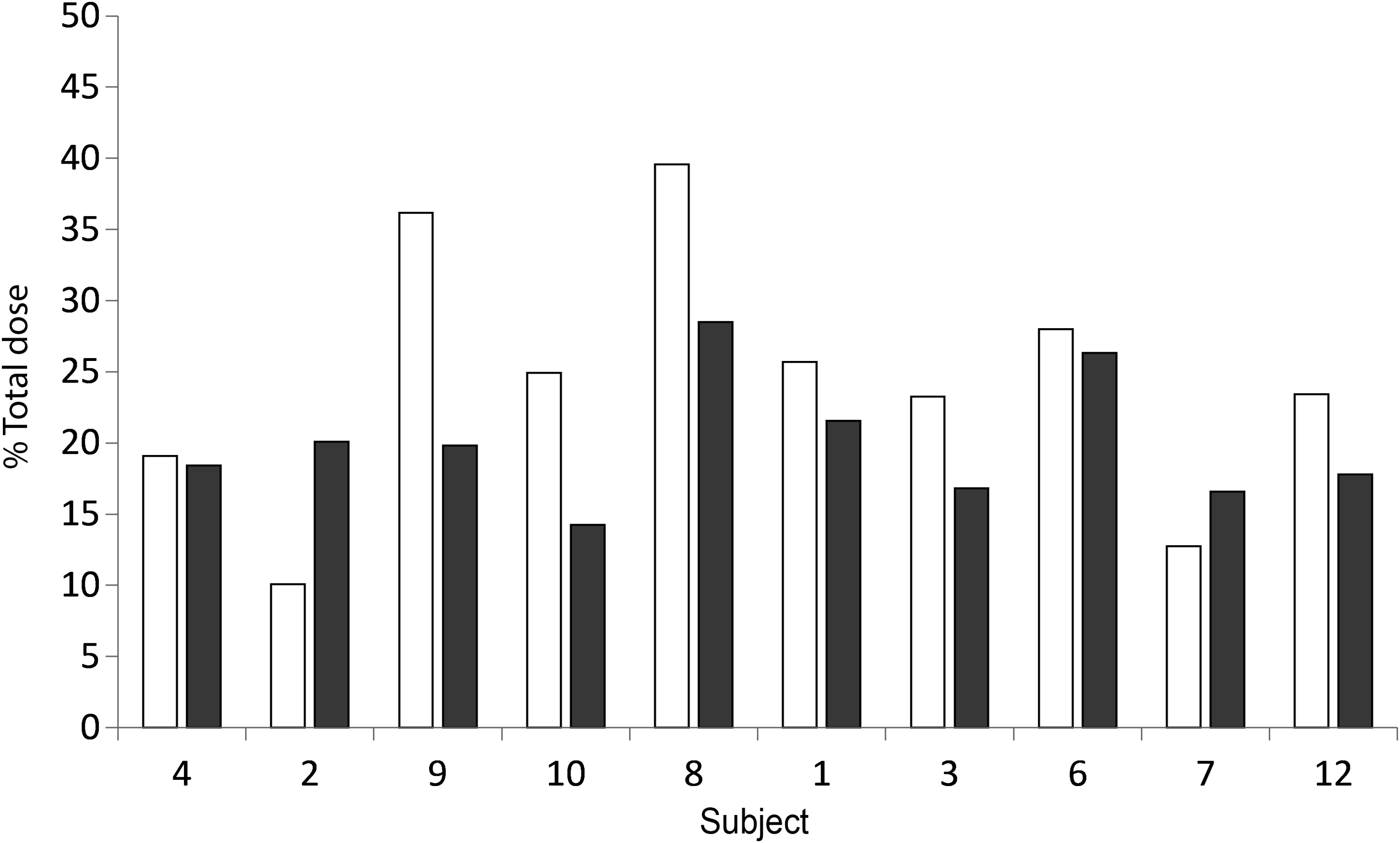
Percent of total dose deposition; extra-pulmonary dose. Facemask (□) and mouthpiece (■). Sorted by age; youngest first.
There was a statistically significant difference in the dose detected on the exhalation filter (p=0.003); the dose deposited for the facemask interface was most likely influenced by the significant deposition of dose on the inside surface of the facemask (Fig. 6) and face. The actuator and spacer dose did not differ between the two methods of aerosol delivery, and the mean peripheral to central (P:C) lung deposition ratios were not significantly different (facemask; 1.3 (0.26) vs. mouthpiece; 1.2 (0.35); p=0.11). Individual subject P:C ratios are given in Figure 9.
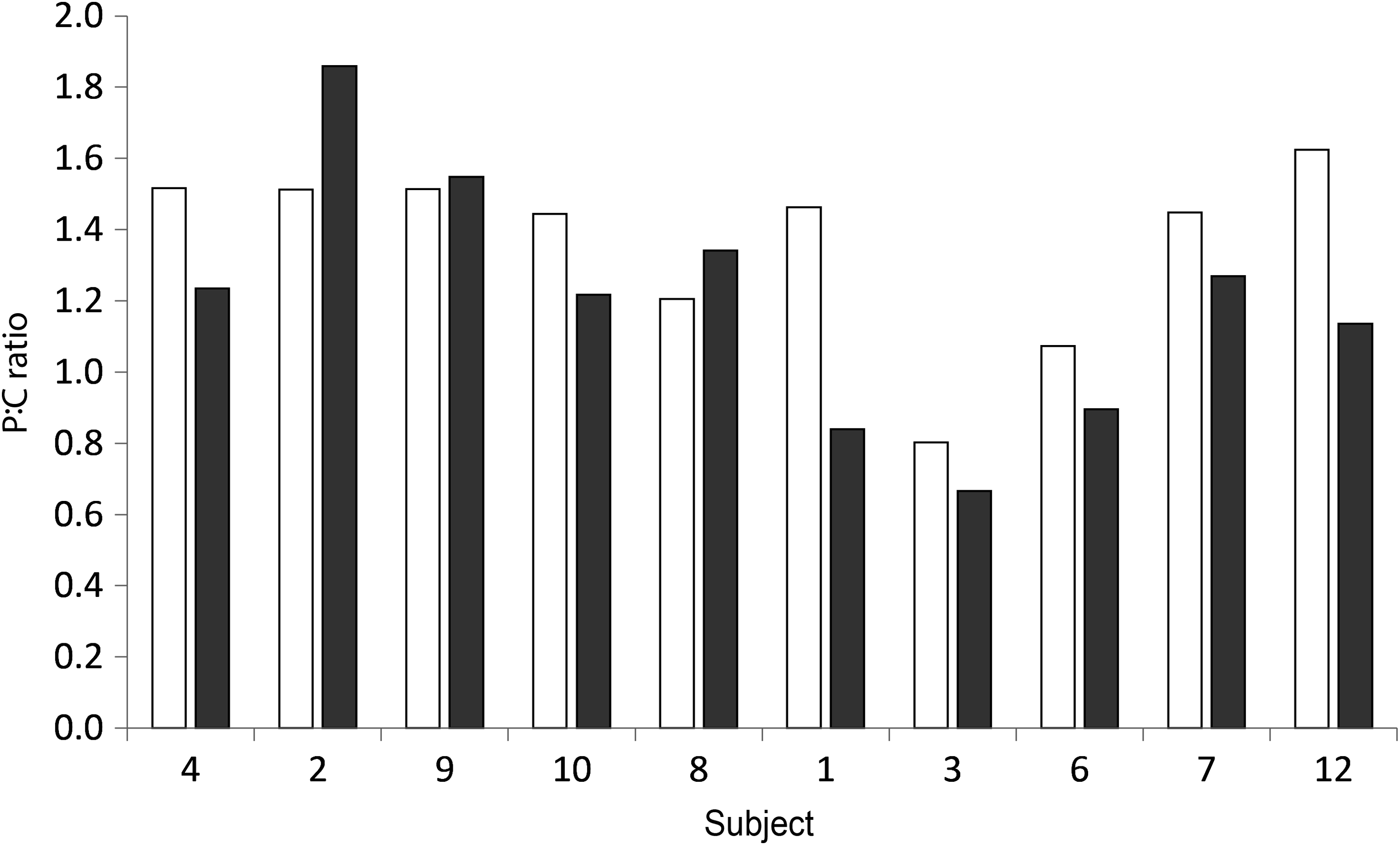
P:C ratio. Facemask (□) and mouthpiece (■). Sorted by age; youngest first.
Example scintigraphic traces are shown in Figure 10. Gamma camera images following an inhalation by a subject with poor tolerance and poor compliance are shown in Figure 10A. Figure 10B shows gamma camera images following compliant inhalation by a subject with good tolerance to the procedure. Note the higher ratios of head and neck:lung and extra-pulmonary:lung deposition associated with the noncompliant inhalation via the facemask (Fig. 10A (i)) compared with the compliant inhalation via the facemask (Fig. 10B (i)). Similarly, the ratio of extra-pulmonary:lung deposition with a noncompliant inhalation via the mouthpiece (Fig. 10A (ii)) was also higher than that observed for the compliant inhalation via the mouthpiece (Fig. 10B (ii)). Similar findings are also demonstrated in Figures 5, 7–9, in which subject 2 (the noncompliant patient in Fig. 10A) received a lower lung dose and a higher P:C ratio than subject 7 (the compliant patient in Fig. 10B), both with the facemask and the mouthpiece. Comparison of the scintigraphs shown in Figure 10 with the associated narratives in Table 2 demonstrate the value of the narrative information.
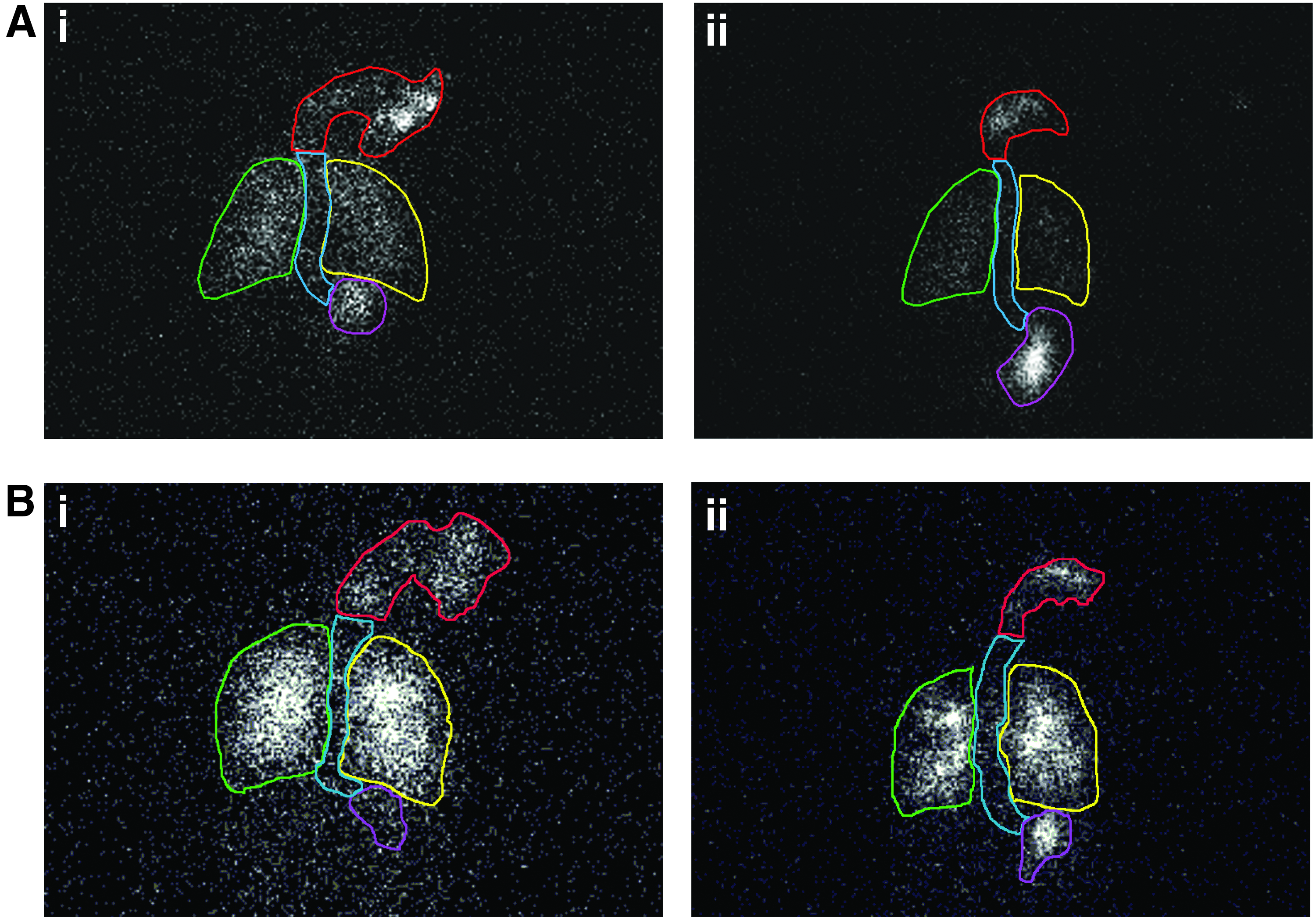
Example scintigraphs; (i) Facemask, (ii) Mouthpiece.
Breathing pattern parameters summary data
Key breathing pattern parameters recorded from subjects using the pMDI/spacer with a facemask and pMDI/spacer mouthpiece were recorded with the exception of; one subject with either interface (Subject 5), subjects 8 and 10 when using the facemask interface, and subject 11 when using the mouthpiece interface. A mean of the remaining subjects' recorded parameters was calculated (Fig. 11 and Table 3). The results indicated that the parameters measured were substantially similar for the study population, regardless of whether a mouthpiece or facemask was used to inhale the aerosol. However, examination of the individual pMDI Datalogger analyses (an example of which is given in Fig. 3) showed obvious differences between subjects. For example, the analyses for subject 2 (Fig. 10A is the corresponding scintigraph) supported the observations noted in the subject narratives, namely that breathing was erratic or absent after pMDI shake and actuation, with breath volumes of 60 to 360 mL. In contrast, the analyses for subject 7 (Fig. 10B is the corresponding scintigraph) showed regular breathing, which was well timed following pMDI shake and actuation, with breath volumes of 340 to 840 mL.
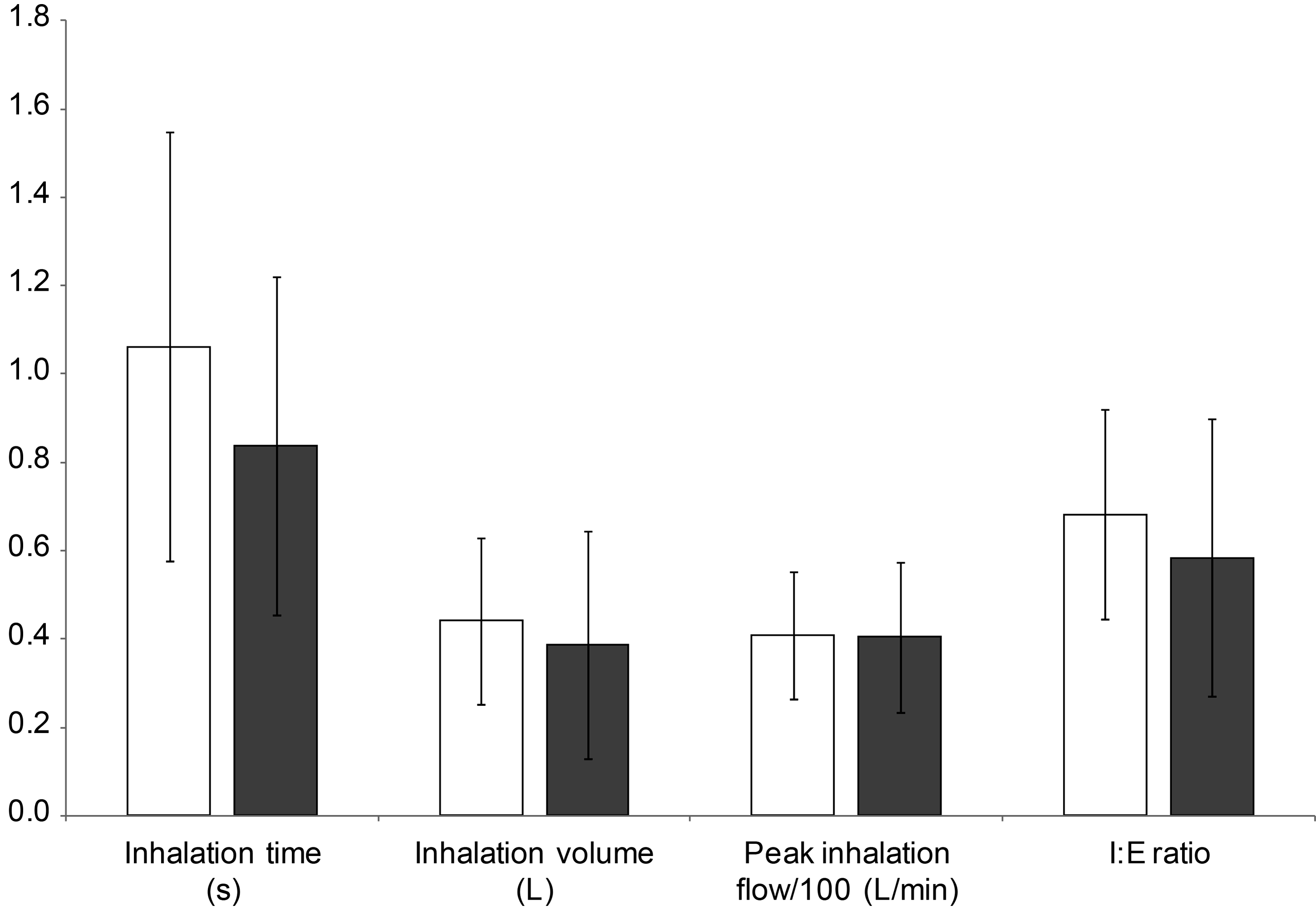
Mean values for key breathing parameters for subjects using pMDI/spacer with facemask (□) and pMDI/spacer with mouthpiece (■). Error bars represent standard deviation.
Comparison of scintigraphic dose deposition and narratives
A by-subject visual comparison of lung deposition and narrative data showed that overall, the narratives provided a very rough guide to the lung deposition results. The narrative data included the two subjects without scintigraphic lung deposition data. Poor tolerance and compliance were recorded in the narrative data for the subject with the lowest combined facemask and mouthpiece deposition (subject 2). The results for the subject with the highest combined facemask and mouthpiece deposition corresponded with a narrative that reflected a child that was very tolerant/compliant during the administration.
Discussion
There was no significant difference in the lung deposition of radiolabeled aerosol between use of the different interfaces, which may be surprising considering the tendency of older facemask designs to result in a reduction in delivered dose due to leakage.(14,15) The similarity in mean lung dose, and lack of age related trend in by-subject lung dose with either interface suggests that leakage during use was not a significant issue in this study. The facemask was held gently but firmly to the child's face through the actuation of the pMDI and subsequent tidal breathing to clear the spacer, to minimize the chances of leakage as the fit was being continuously monitored. There was a significant difference in head and neck deposition with a lower deposition through use of the mouthpiece. This is not surprising, as the head and neck deposition included deposition on the face and in the nasal cavities due to nose breathing by some patients when using the facemask. Example differences in facial and nasal depositions have been shown in Figure 10. Avoidance of filtration of aerosol within the nasal cavities is one of the main advantages for use of the mouthpiece interface over the facemask interface. The significant differences seen in the exhalation filter and facemask/mouthpiece deposition were also not surprising considering the difference in design and operation of the two interfaces.
Validation of the radiolabeled aerosol showed that the aerosol characteristics of the HFA-albuterol pMDI used were not affected by the radiolabeling process, providing justification that the results found could be assumed to be valid for daily-life use of a pMDI spacer with facemask or mouthpiece interface in a pediatric population. It could be argued that the lead shield used to minimize exposure of the subjects and study personnel to radiation could have made the pMDI spacer assembly more awkward to use, and thus could have affected the technique of administration by impairing natural shaking and firing of the pMDI. However, as the effect was equal for both interface configurations tested, it should not have interfered with the comparison of facemask and mouthpiece interfaces.
This study provided the first paired comparison of lung and head and neck deposition of radiolabeled aerosol when either a facemask or mouthpiece interface was used to inhale aerosol from a pMDI with spacer by young children. There is limited clinical information on the impact of aerosol delivery through a pMDI/spacer facemask in comparison with a mouthpiece. In a scintigraphic study of lung deposition of beclomethasone in children aged 5–17 years, the mean lung deposition in the youngest age group (5–7 years) following tidal breathing was 35.4%, whereas the head and neck and gastrointestinal deposition was 24.0%.(19) These results are not far from the present results, considering the difference in age range and aerosol droplet sizes (HFA albuterol 2.77 μm and HFA beclomethasone dipropionate 1.16 μm).(19) Tal et al. measured lung deposition following use of a spacer with facemask using 99mTc-radiolabeled CFC albuterol in children aged 3 months to 5 years.(32) The mean lung deposition was 1.97%, with no data on lung deposition achieved using a spacer mouthpiece in the same age group. More recently, Zar et al. showed that lung deposition with a spacer facemask was comparable to that achieved with a mouthpiece by using 99mTc-DTPA radiolabeled aerosol.(33) However, the results are difficult to generalize, because the aerosol was delivered from a jet nebulizer rather than a pMDI, resulting in different aerosol delivery characteristics compared with a pMDI.
The data recorded by the Datalogger used in the study provided an additional frame of reference against which the results of the radiolabeled aerosol deposition could be compared. The mean results shown in Figure 11 were substantially similar for the two interfaces, which may reflect the similar lung deposition seen with the radiolabeled aerosol. However, the differences between individual Datalogger recordings and their comparison with the individual depositions of radiolabeled albuterol for subjects 2 and 7 indicate that this type of data may be of use when performing aerosol deposition studies, as it could help explain the wide variation in results for individual subjects. The detailed nature of the breathing pattern recordings demonstrated in Figure 3 means that the parameters shown in Figure 11 represent only a small portion of the breathing pattern characteristics that could be derived from the recordings. The investigation of potential correlations between breathing pattern characteristics and radiolabeled aerosol deposition results for individual patients would require more space than could be accommodated in the present article, but will be the subject of further research.
Conclusions
Lung deposition was higher than anticipated for children in the age group of 3–5 years, and interestingly, deposition with the facemask appeared to be comparable to that using the mouthpiece. This was possibly due to the fact that some of the younger children in particular were not used to the mouthpiece (although training was provided during screening), and were more comfortable using the facemask. The significantly reduced head and neck deposition with the mouthpiece is of note, however, and potential contributing factors were increased nasal deposition due to nose breathing by some patients, and facial deposition of drug, both of which occurred with the facemask, but not the mouthpiece. This extra-pulmonary deposition would not be of major concern with normal use of albuterol, but may be a consideration if corticosteroids are being administered.
Our conclusions, based on this limited data, are to allow younger patients to use a facemask while they are unfamiliar or uncomfortable with a mouthpiece, to ensure therapeutic efficacy. However, attempts should be made to familiarize and train them in the use of a mouthpiece whenever possible, because consistent inhalation technique using the mouthpiece will continue to ensure therapeutic efficacy while reducing extra-pulmonary exposure of drug.
Footnotes
Acknowledgments
We acknowledge Humanfactors Design Works, New York, USA (Steve Getz and Donald van Royen) for assistance in developing the Datalogger; the technologists of the Nuclear Medicine Department at Princess Margaret Hospital, Perth, Western Australia, for running the gamma camera and assistance in data acquisition (Karen Hindley and Jill Summers); and PS5 Consultants Ltd, Portsmouth, UK, for editorial assistance (Rachael Giles and Sophia Kuperman).
Author Disclosure Statement
William Ditcham, Jasminka Murdzoska, Christina Roller, and Sunalene Devadason are employees of the University of Western Australia, School of Paediatrics and Child Health, Perth, W.A., Australia. Guicheng Zhang is an employee of Curtin University, School of Public Health, Perth, W.A., Australia. Dirk von Hollen is an employee of Respironics, Inc., a Philips Healthcare company, Murrysville, PA, USA; and Kurt Nikander is an employee of InDevCo AB, Nyköping, Sweden, but was an employee of Respironics, Inc. during the study. The study was partially funded by Respironics, Inc. and by a grant from the Princess Margaret Hospital for Children Foundation.
