Abstract
Abstract
Background:
Pressurized metered dose inhalers (pMDIs), combined with a valved holding chamber (VHC) and facemask, are often used for young asthmatic children. When using a VHC with facemask, a tight seal between the facemask and the patient's face is crucial for efficient pulmonary aerosol delivery. Realistic parameters for in vitro bench testing are not well known. A custom instrumented OptiChamber Diamond VHC, known as the Facemask Datalogger, was developed to measure the real-time application of force and the air flow through the VHC and facemask.
Methods:
Thirty asthmatic children aged 1–4 years from the Kinderhaven outpatient clinic, who were prescribed a pMDI/VHC with facemask for regular use, were included in the study. Using the Facemask Datalogger, the parent applied the facemask to the face of the child during normal tidal breathing, and force and flow were recorded. This was repeated three times. A video of the procedure was made and scored on cooperation (scale of 1–5).
Results:
Mean application force was 4 N (±1.5) equal to 411 g (±156) when expressed as a weight equivalent; intrasubject variability in application force was 23% (±23); intersubject variability in application force was 39%; time needed to empty the VHC was 4.5 sec (±2.9); breaths needed to empty the VHC were 2.9 (±1.1); and mean cooperation score was 4.3 (±0.8). Age was correlated with time (r=−0.49; p=0.006) and breaths needed to empty the VHC (r=−0.75; p<0.001), and observer cooperation score (r=0.65; p<0.001).
Conclusions:
The Facemask Datalogger is useful for measuring application force and air flow in vivo. Mean application force was lower than assumed in other studies. Older children emptied the VHC faster, with fewer breaths and better cooperation. The data from this study can be used in the future development and testing of new facemasks and VHCs.
Introduction
A
When using a VHC with facemask, a tight seal between the facemask and the patient's face has been shown to be crucial to efficient pulmonary aerosol delivery.(4) Young children often dislike the use of a facemask, which can cause them to struggle during treatments. This can make it difficult for the caregiver to maintain a tight seal between the facemask and the face of the child; a poor facemask-to-face seal is associated with a substantial decrease in the lung deposition of aerosol.(4–7) The greater the force required, the less comfortable it will be for the child and the more he or she is likely to struggle, to the detriment of the inhaled dose. Achieving a good facemask-to-face seal is therefore critical, and depends in part on the design of the facemask.(8) Although a number of VHC-facemask combinations are commercially available, current research has focused on the development of new facemasks, intended to improve inhalation therapy in young children. In vitro testing is an important part of the facemask development process, prior to in vivo application. In vitro tests allow for a wide range of isolated parameters to be independently investigated in a systematic way, and are particularly useful when assessing aspects of VHC-facemask system design, such as dead space, and factors influencing the facemask–patient interface, such as age-dependent facial geometry, seal efficiency, application force, and application angle.(9) In vitro testing has some weaknesses, particularly relating to the dependence on assumptions regarding the in vivo situation, when modeling parameters in vitro. It may be unknown, for example, how great an application force is required to form a tight seal with any given facemask. In addition, sinusoidal breathing patterns are typically used to simulate breathing during in vitro testing.(2) In the development of facemasks, good equipment and data derived from in vivo studies are required for the accurate simulation of the in vivo situation.(10) Such tools should improve the efficiency of laboratory-based facemask development.
A custom-built Force/Flow Facemask Datalogger, which incorporates a modified antistatic VHC and a detachable facemask, has been developed for use in vivo. The Force/Flow Facemask Datalogger can measure and record several in vivo parameters of subjects' breathing patterns, which can be translated to, and used in, in vitro studies. These parameters include the application force (measured as weight) applied through the facemask with VHC, the flow rate and volume of air inhaled through the VHC, and the time needed to empty the VHC. Previous studies conducted with different electronic datalogger devices have demonstrated the feasibility of this method. (11,12)
Our first aim was to assess the usefulness of the Force/Flow Facemask Datalogger for collecting in vivo breathing pattern and application force data from young children. The primary endpoint of interest was the force with which the caregiver applied the VHC-facemask system to the child's face (application force). A secondary aim was to use the data recorded by the VHC-facemask Datalogger to evaluate the breathing patterns of young children inhaling via a VHC-facemask system.
Materials and Methods
Study design
Thirty children with doctor-diagnosed asthma, aged 1–4 years, who were prescribed a pMDI/VHC with facemask for regular use, were enrolled in an observational pilot study from the Kinderhaven outpatient asthma clinic, Rotterdam, the Netherlands. Children with facial malformations or acute asthma symptoms at the time of the study were excluded. The study consisted of a single visit to the clinic from each patient, which was organized to coincide with a regular checkup appointment. Patients who did not have a regular checkup scheduled were visited at home. The study was approved by the local ethical committee, and written informed consent was provided by the parents prior to the study procedure.
Force/Flow Facemask Datalogger
The Force/Flow Facemask Datalogger is constructed from a modified OptiChamber Diamond VHC [Respironics Respiratory Drug Delivery (UK) Ltd., Chichester, UK] fitted with the LiteTouch facemask [medium; meant for children aged 1–4 years; Respironics Respiratory Drug Delivery (UK) Ltd.] (Fig. 1). The Force/Flow Facemask Datalogger can be customized for use in other VHC-facemask combinations.
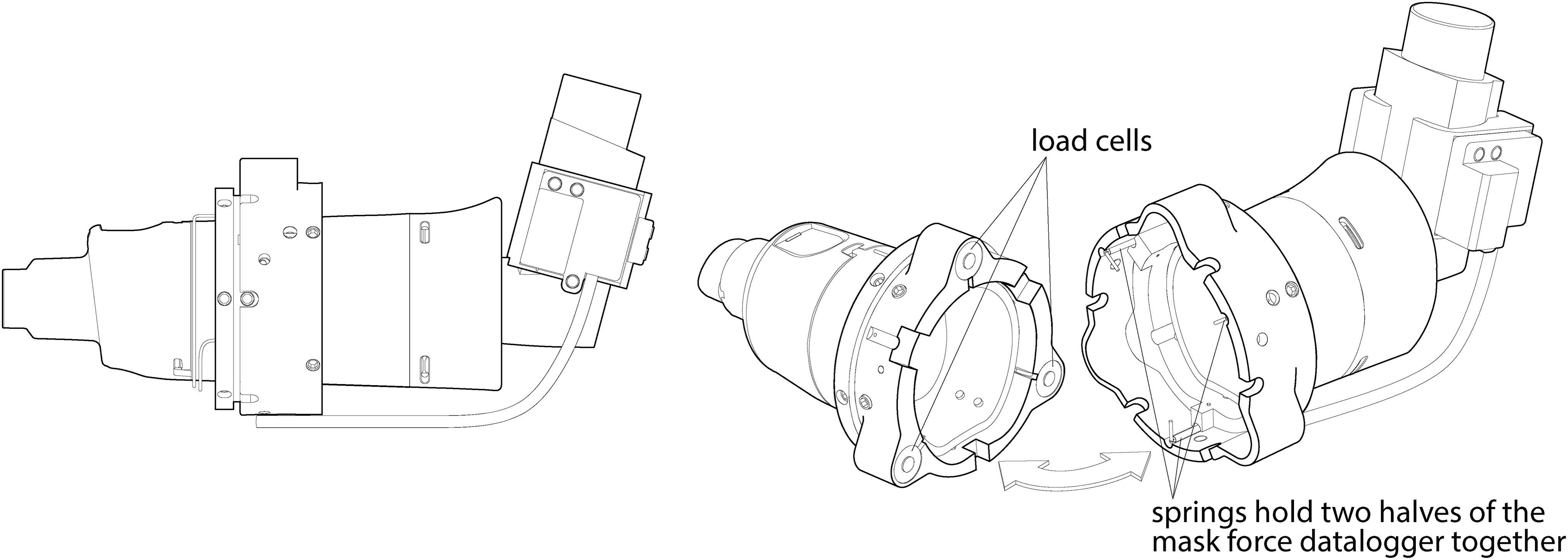
The Force/Flow Facemask Datalogger.
The Force/Flow Facemask Datalogger instrumentation comprised a set of “flow” instruments to record information on flow through the VHC, and a set of “force” instruments to record information on facemask-to-face application force. The flow instrumentation was attached to a custom inhaler actuator boot, and recorded the flow through the actuator. The force instrumentation was permanently attached to the two halves of the OptiChamber Diamond VHC, and recorded the amount of force transmitted from the distal end of the VHC (pMDI actuator end) to the proximal end of the VHC (facemask end), via three load cells placed around the circumference of the VHC. The force/flow instrumentation was connected to a PC via wires, which communicated the signals detected (Fig. 1). Force information recorded comprised a continuous average of the signals from the three load cells, indicating the force with which the facemask was applied to the face over the course of the treatment. Flow information recorded comprised a continuous signal of the air flow passing through the actuator over the course of inhalation.
Once the measurements were recorded, they were placed into a Microsoft Excel worksheet and processed using a macro to remove noise from the signals (Fig. 2). The filtered data were analyzed to determine the mean, minimum, and maximum force for each treatment (taken from the start of treatment up to the point at which 320 mL had been inhaled), and the time and number of breaths required to empty the VHC.
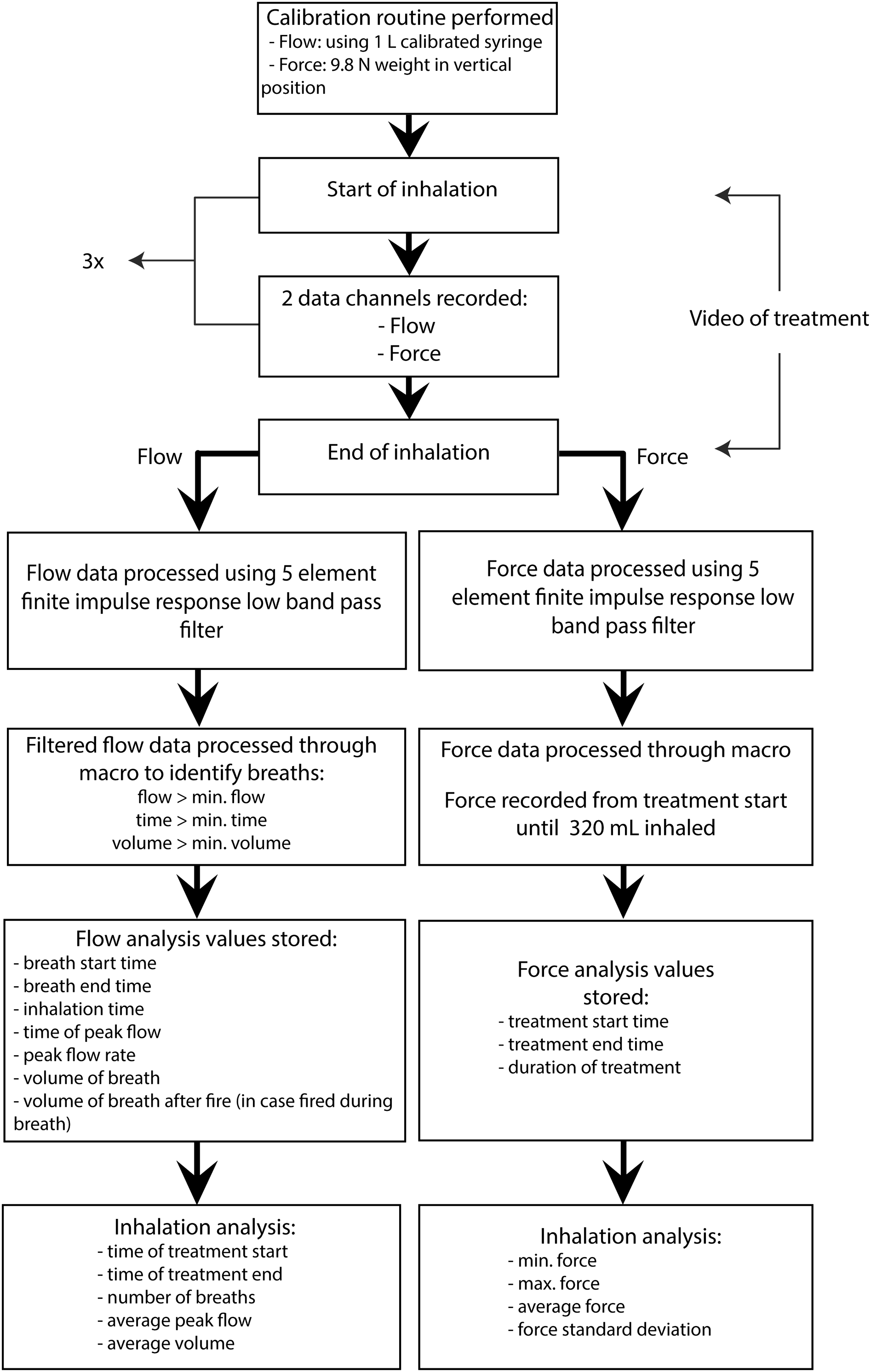
A macro in Microsoft Excel was used to filter the noise from the force and flow measurements.
Study procedure
Prior to each patient session, the Force/Flow Facemask Datalogger instrumentation was verified via a calibration routine (Fig. 2). The Force/Flow Facemask Datalogger was designed such that the entire facemask and one-way valve system could be removed and replaced for each child, to minimize the risk of cross-contamination.
The child sat on the lap of the parent throughout the study procedure. The parent applied the facemask to the face of the child and was asked to simulate a normal inhalation procedure. All parents and children were familiar with the inhalation procedure when using a VHC with facemask, and had been well instructed before as part of routine care. No extra instructions regarding the facemask seal were given at the time of the study, to reflect a real-life situation as closely as possible. Each measurement was repeated three times with each child (Fig. 3), and the entire procedure was videoed for later scoring on cooperation. A measurement was considered complete if at least twice the volume of the VHC-facemask system (2×160 mL) was replaced; this volume was considered sufficient to withdraw all aerosol from a VHC; the volume was determined by filling the VHC, including the facemask, with water, and measuring this amount of water. (11,13,14)
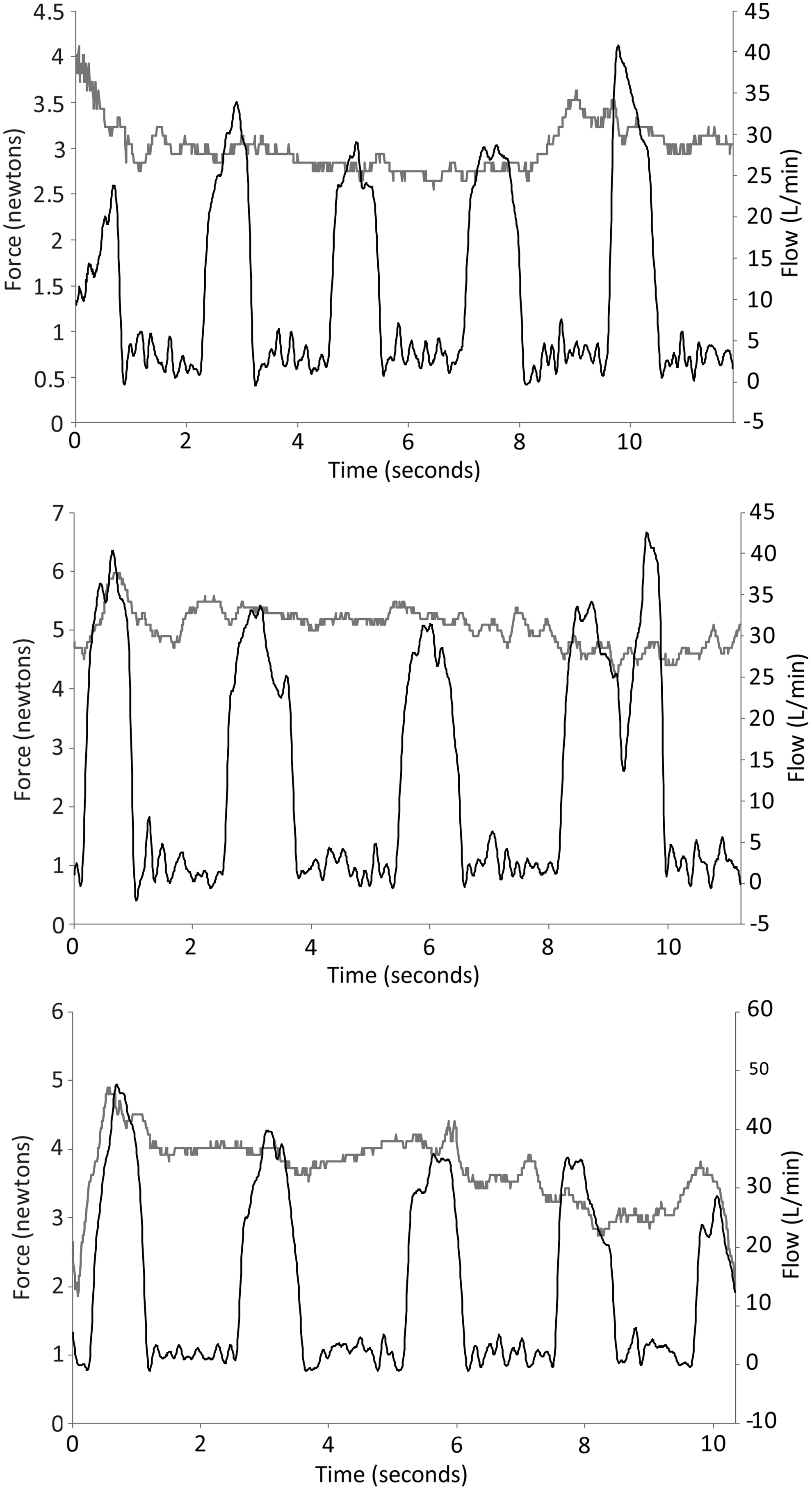
Three examples of treatments recorded by the Force/Flow Datalogger. All three examples are from consecutive treatments with the same patient and show the repeatability of the measurements with this particular patient.
Observations and scoring of acceptability
Acceptability of the Force/Flow Facemask Datalogger was scored on an evaluation form using a visual analogue scale (VAS) design with a range of 1 to 5, where 1 indicated a low/poor result and 5 indicated high/good result (see Fig. 4). Three skilled independent observers scored cooperation for each measurement from the recorded videos. Observers were asked to “Place a mark on the lines below to indicate how good the cooperation is in each measurement,” as shown in Figure 4. The observers were either physicians or nurse practitioners, who were specialists in the treatment of children with asthma.
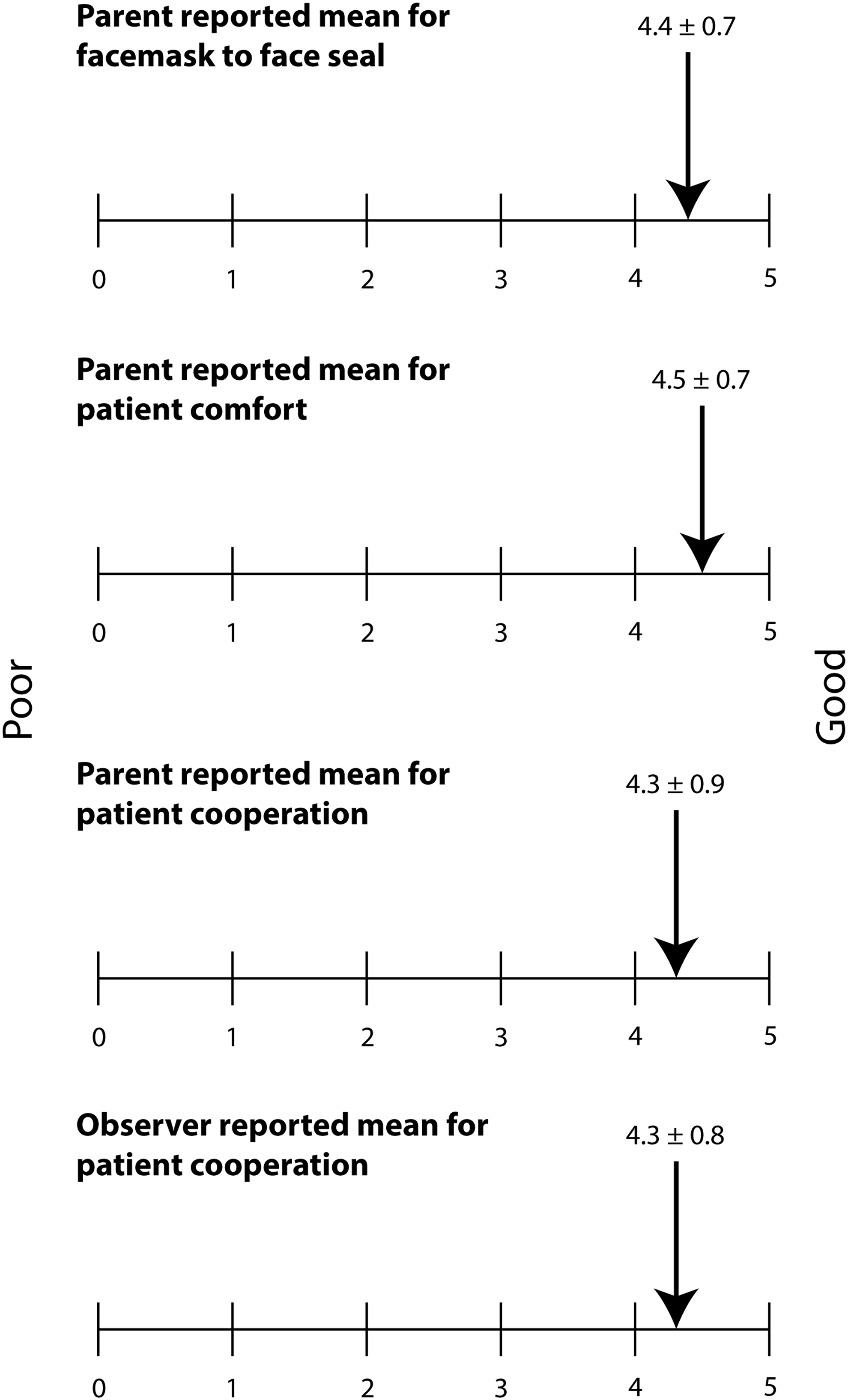
Mean VAS scores from parents and observers. Cooperation was scored from 0 to 5: 0=no measurement possible; 1=continuously crying, screaming, or struggling against the procedure; 2=crying, screaming, or struggling, but not continuously; 3=not very good cooperation, but no crying or screaming; 4=good cooperation, but clearly not comfortable; 5=good cooperation and quiet breathing during the whole measurement.
Statistical analysis
The primary endpoint was mean application force, which was expressed in newtons and force equivalent weight. Secondary endpoints were: the volume of air inhaled through the spacer, the time needed to replace this volume, the degree of cooperation scored by parents and observers, and breathing patterns/volumes. For all variables, mean values (±SD) were calculated for each patient and for the group overall. Inter- and intrasubject variability in application force was expressed as coefficient of variation (%). Correlations were calculated with a Spearman correlation test. p values of <0.05 were considered significant. An interclass correlation coefficient between the scores of parents and the mean scores of observers was calculated, where a coefficient of more than 0.8 was considered a good agreement between scores.
Results
Thirty children with asthma [21 boys; mean age 3.2 years (1.0); mean weight 15.1 kg (3.3); mean height 96.3 cm (9.3)] completed the study procedure. Mean values for data collected via the Force/Flow Facemask Datalogger are shown in Table 1. Data from 90 complete study procedures were collected. Breaths to empty the VHC could not be calculated for two measurements due to technical problems with the Force/Flow Facemask Datalogger in one instance, and crying of the child in another.
Mean tidal volume through the VHC was 200 mL, and was higher in older patients (r=0.69; p<0.001). The intrasubject coefficient of variation for tidal volume was 22.6%.
Overall, the Force/Flow Facemask Datalogger was well accepted by the children and their parents (Fig. 4). Most children cooperated well during the study procedure, as scored by parents and observers; only two children cried. There was close agreement between the mean cooperation scores recorded by parents and observers (interclass correlation coefficient: 0.903). Due to technical problems, one study procedure measurement was not videoed, and therefore not scored by observers.
Age was negatively correlated with the child's time to empty the VHC (r=−0.49; p=0.006), and breaths to empty the VHC (r=−0.75; p<0.001); the younger the child, the longer it took to empty the VHC, with more breaths (Fig. 5). The child's age was also correlated with the observer-assigned cooperation score (r=0.65; p<0.001). The cooperation score had no significant correlation with time taken to empty the VHC, breaths to empty the VHC, or applied force. There was no significant correlation between age and force applied by the parent (r=−0.28; p=0.132).
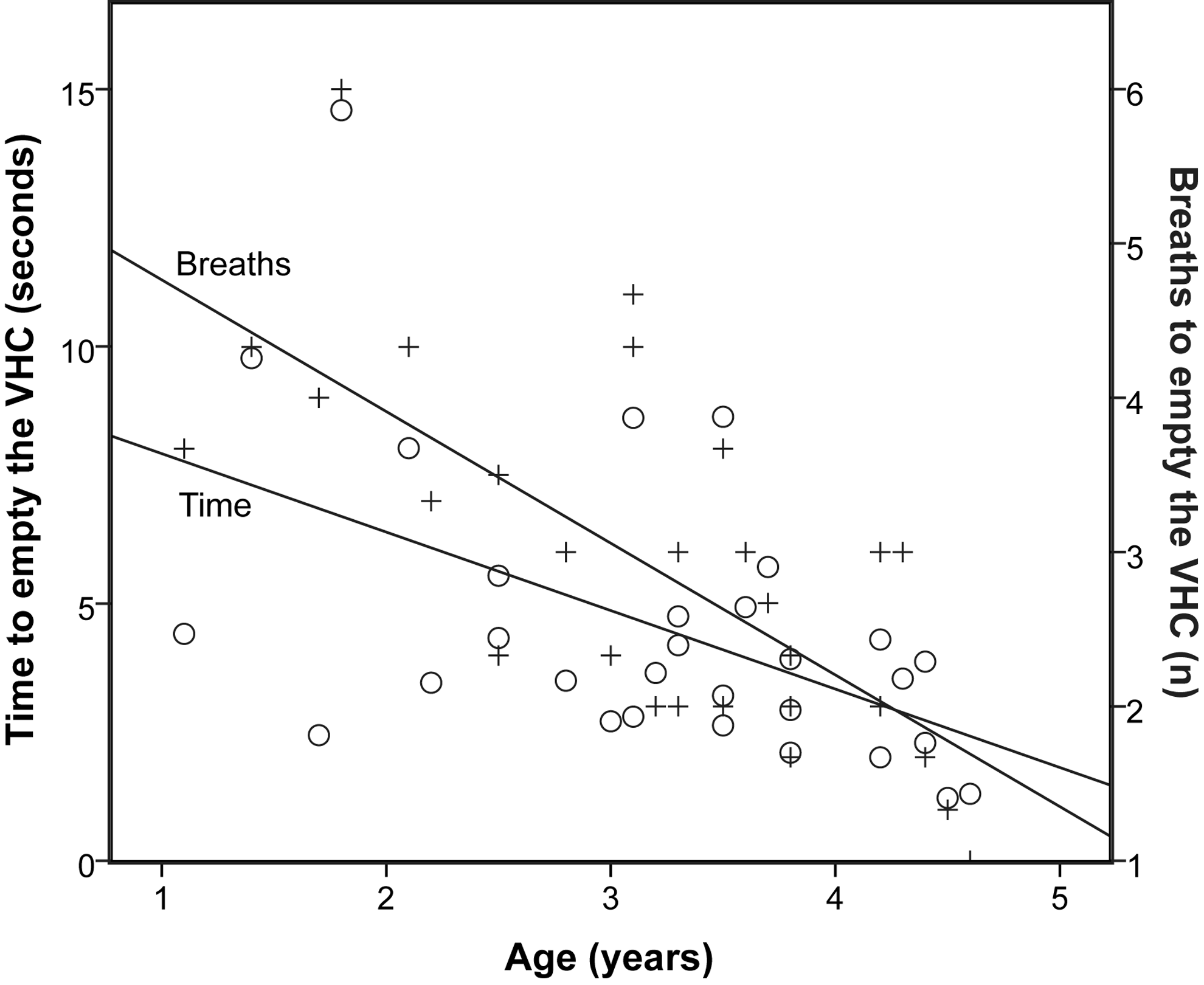
Correlation between age and time to empty the VHC (○), and breaths to empty the VHC (+).
Figure 6 shows an example measurement recorded from a cooperative child. With each breath, an approximately equivalent amount of air was moved, indicating a good facemask-to-face seal. Figure 7 shows an example measurement from a child with poor cooperation, who struggled and cried during the procedure. This child moved his head away from the facemask, reflected in the drop in force on the facemask (t=2 sec). When the facemask was replaced on the face (t=7 sec), the crying was reflected by a continuous rise in the volume of air moved. Figure 8 shows a measurement from a child with a poor facemask-to-face seal. Under these conditions, the child's mean time to empty the VHC was around 25% longer than the overall group mean time to empty the VHC.
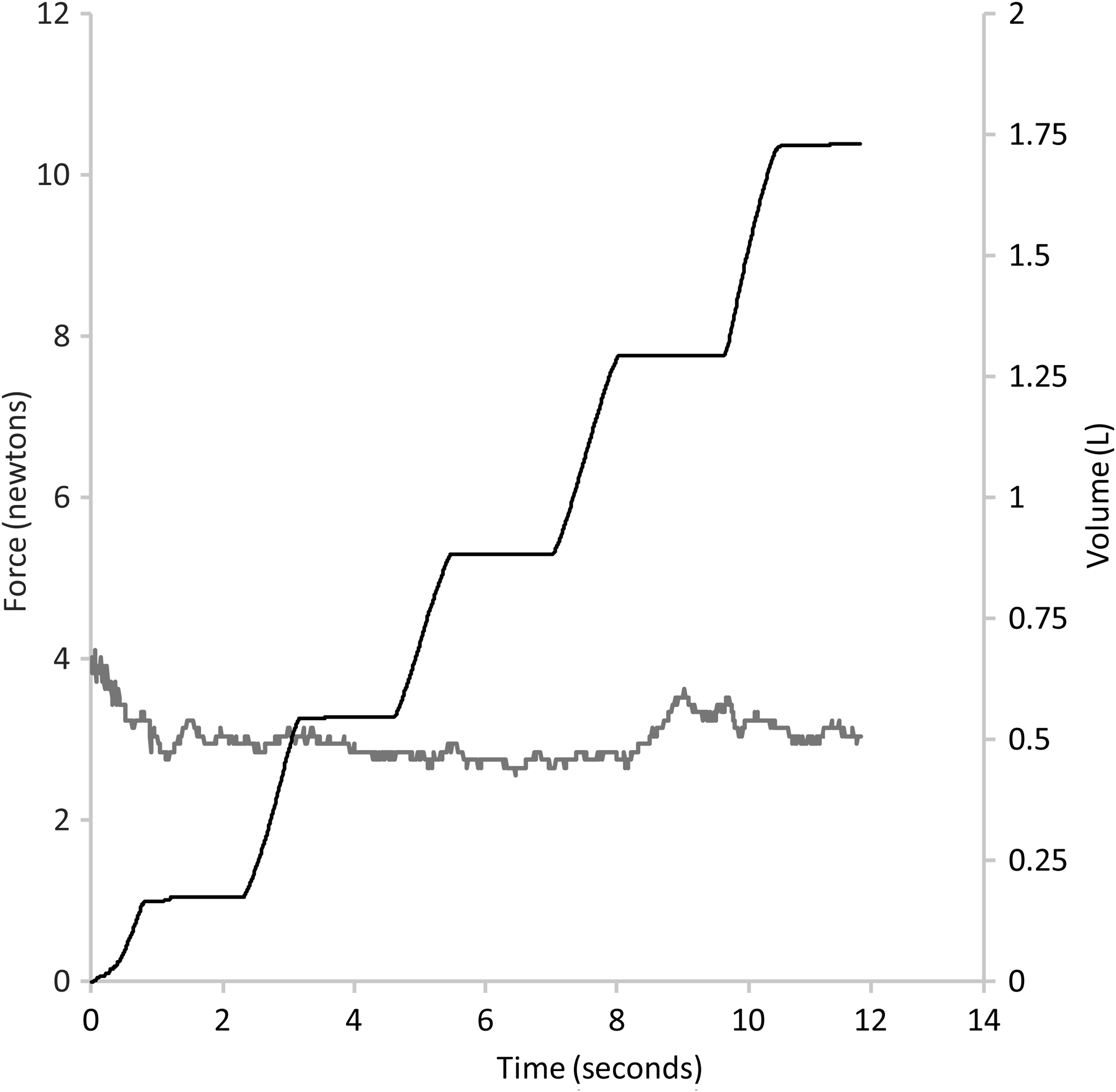
Force/flow data recorded during a study procedure with a cooperative child.
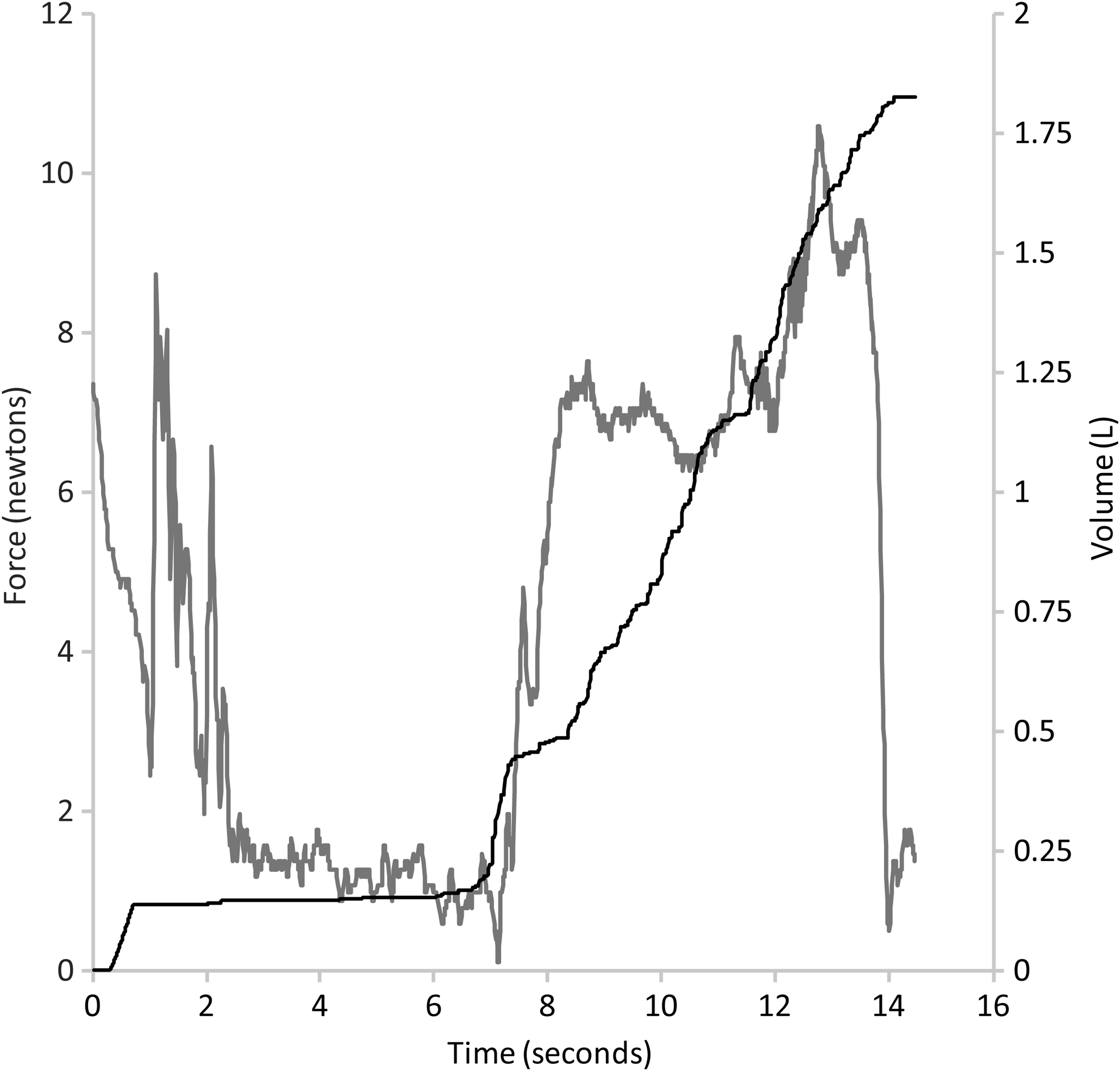
Force/flow data recorded during a study procedure with an uncooperative child, crying and struggling.
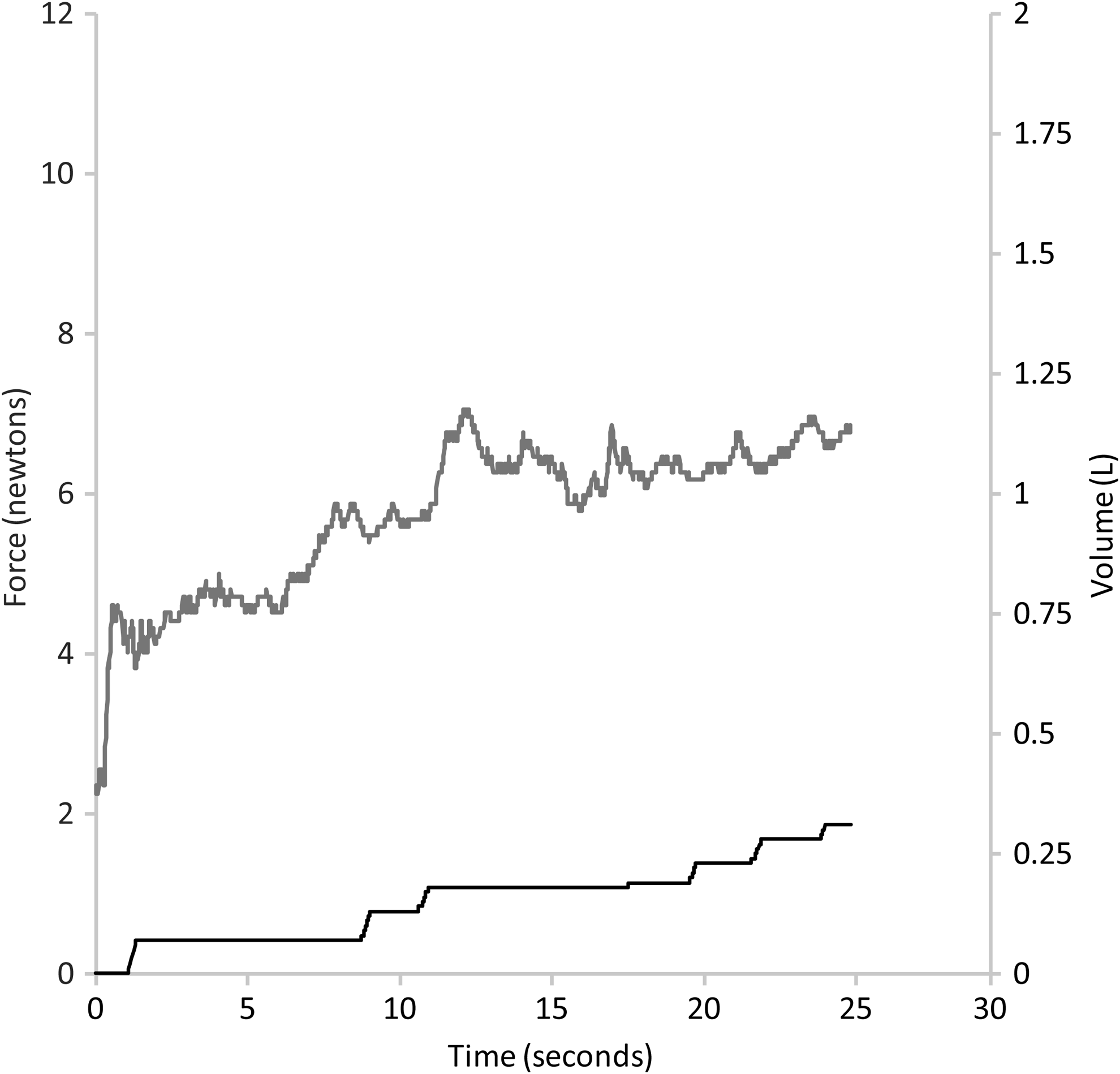
Force/flow data recorded during a study procedure with a poor facemask-to-face seal.
Discussion
The present article reports results of the first study using the Force/Flow Facemask Datalogger in vivo, and demonstrates that the Datalogger is a useful tool for the collection of in vivo data from young children during the use of a VHC-facemask system.
This is the first study to investigate the application forces associated with the use of a VHC-facemask system in vivo. The mean application force observed in this study was considerably lower than that used in former in vitro studies.(9,15) Shah et al. used weight equivalent forces varying between 680 g (6.66 N) and 3.4 kg (33.3 N) during in vitro tests into the effect of applied force on the dead space of various facemasks; these forces were chosen based on experts' opinions.(9) The forces measured in our study are realistic forces, derived in a real-life in vivo situation. This might explain the differences compared with other studies. Another explanation for the differences might be the different designs of the facemasks. The LiteTouch facemask has a soft plastic cushion that surrounds the rim. This might make it easier to achieve a good facemask seal, at low application forces, compared with hard facemasks with a flat rim. A soft cushion also causes less discomfort for the child. Differences in applied forces should be further studied by comparing different facemasks.
There was a relatively high intersubject variability in applied force. This might be explained by differences in the facial anatomy of individual children, or by differences in cooperation between children. Within subjects, the force applied by the parent was fairly consistent, as indicated by a low intrasubject variability.
In the older child, in addition to better cooperation, the time taken to replace twice the volume of the VHC (time to empty the VHC) was shorter, with fewer and larger breaths. The negative correlation between age and time to empty the VHC, and age and breaths to empty the VHC, can be explained by the tidal volumes of older children; older children with a larger tidal volume required fewer breaths to replace 320 mL.
Because the air flow sensors of the Force/Flow Facemask Datalogger were attached at the distal end of the VHC, the breathing patterns recorded consisted only of the air that was inhaled by the child through the VHC. Any air inhaled through leaks around the seal was not recorded. Therefore, the data reflect, as closely as possible, the true amount of air inhaled during normal pediatric inhalations via VHC-facemask systems. Our tidal volume results differ from those found in a study using an in vitro model of infant breathing.(2) In the present study, the observed number of breaths required to empty the VHC result was slightly higher than that reported by Schultz et al.(14) This may be explained by the different definitions of “empty” in the two studies [2×the volume of VHC and facemask (our study) versus the point at which there was no significant difference in drug delivery (Schultz et al.(14))], the age range of the subjects, the use of a facemask in our study rather than a mouthpiece, and the use of different VHCs, and hence different volumes.(14) The results for time to empty the VHC indicated that breathing through the VHC for 10 sec is sufficient to empty twice the volume of the VHC in the age group studied. This is useful information to consider when instructing parents in the use of OptiChamber Diamond VHCs.
In contrast to our expectations, children's cooperation scores were not significantly correlated with any other outcome. We assumed that good cooperation would result in a lower force, but we did not find such correlation. This might be caused by the fact that most children cooperated very well. There were only two children who cried during the study procedure.
A limitation of the study was that it was not possible to objectively measure facemask seal leakage in vivo, because the Force/Flow Facemask Datalogger only recorded the volume inhaled through the VHC. Objective measurement of facemask seal leakage would require a sealed box around the patient, or use of a radiolabeled aerosol.(7,15) Despite the relative safety of radiolabeled aerosols, their use raises ethical issues, particularly in young children.(16) The video recordings and air flow and volume measurements of the current study are considered a satisfactory means by which to identify probable facemask leakage. As shown in Figure 7, facemask seal leakage is reflected by the smaller amounts of air moved with each breath, and the irregular recording of air movement. This highlights the importance of a good facemask-to-face seal; without a good seal, it takes much longer to empty the VHC, which reflects impaired drug delivery, and might also impact negatively on compliance.
Applied force results are unlikely to be directly translatable to clinical practice. Results of the present study indicated no correlation between applied force and time to empty the VHC, suggesting that the force with which a parent presses the facemask to the face of the child does not influence the time required for inhalation of the actuated aerosol. However, it is uncertain whether this lack of association is related to the design of the LiteTouch facemask and low application force required. Further research is required, preferably comparing different facemask designs, in order to determine the relationship of applied force to dead space, seal efficiency, and aerosol delivery. As this study was conducted with one type of facemask, the applied force results cannot be directly translated to delivery via other facemasks. Results of the present study may be an underestimation of the forces required with other facemasks, because of the soft cushioning surrounding the LiteTouch facemask seal rim. However, these results give a new insight into the range of forces applied by parents, which turned out lower than previously assumed.
Few studies comparing different facemasks have previously been conducted. As new facemasks have been and continue to be developed, a comparative study between these facemasks would be of value. Facemask design and acceptability to the child are very important aspects contributing to the effective treatment of asthma. As there are presently minimal data regarding the force applied through VHC-facemask systems in vivo, the results of this study should be a useful addition to future facemask development. The new Force/Flow Facemask Datalogger may be an effective tool for use in further facemask research, because it can be used to compare different facemasks, both in vivo and in vitro.
Footnotes
Acknowledgments
The authors acknowledge Magda Spierings, Jantine Hiddinga, and Aukje Bos for their technical assistance. We also acknowledge Humanfactors Design Works, New York, NY (Steve Getz and Donald Van Royen) for assistance in developing the Datalogger, and PS5 Consultants Ltd., Portsmouth, UK, for editorial assistance (Nick Smith, Rachael Giles and Sophia Kuperman).
Author Disclosure Statement
Kirby Tong Minh and Annemieke J. von Königslöw are medical students of the Erasmus University, and Hettie M. Janssens is an employee of Kinderhaven/Havenziekenhuis and the Erasmus Medical Center/Sophia Children's Hospital, Department of Pediatric Respiratory Medicine and Allergology, Rotterdam, The Netherlands. Dirk von Hollen is an employee of Respironics, Inc., a Philips Healthcare company, and Kurt Nikander was an employee of Philips Respironics at the time of the study. This study was funded by Respironics, Inc.
