Abstract
Abstract
Background:
Inhaler mishandling is a common issue among patients suffering from asthma and is associated with poor clinical outcomes and greater consumption of health-care resources. Ease of use can improve inhaler technique and, possibly, patients' preference for their inhaler device, which in turn may lead to better adherence to therapy.
Methods:
This study investigated usability characteristics of NEXThaler® versus two other dry powder inhalers (DPIs; Diskus® and Turbuhaler®). Sixty-six adult patients with asthma (mean age 42.9±17.7 years) and with no previous experience of using a DPI were included in a randomized crossover comparison of the three devices. The main measures of usability were the number of steps failed for each device and the number of people who were able to use the device successfully (effectiveness), the time it took patients to set up the device and the time to read the instructions for use (IFU; efficiency), and patient preferences (satisfaction). Inhaler technique was evaluated after the IFU leaflet was read.
Results:
NEXThaler was found to be superior to the other two DPIs in terms of the number of device use failures (p<0.001), time to set up (p<0.001), and time to read IFU (p<0.001). Additionally, the proportion of participants who completed a successful inhalation without any errors at all was significantly higher for NEXThaler than for Diskus and Turbuhaler (p<0.001). Patients rated NEXThaler as the easiest to use and most preferred inhaler to own (p<0.001).
Conclusions:
NEXThaler displayed better usability compared with Diskus and Turbuhaler. The improved usability and higher satisfaction with the device may contribute to increased patient adherence to asthma treatment.
Introduction
D
NEXThaler® (Chiesi Farmaceutici S.p.A.) is a DPI with an innovative full dose feedback system incorporating a novel breath-actuated mechanism (BAM): a click is heard and a dose counter decreases by one count, only after effective full dose delivery.(18,19) NEXThaler® provides a high lung deposition throughout the entire bronchial tree (56% of the emitted dose in patients with asthma),(19,20) and the BAM is activated at a consistent inhalation flow that is independent of disease severity and the level of asthma control, i.e., 35.0 and 35.4 L/min in patients with controlled and partially controlled/uncontrolled asthma, respectively.(21,22) At the time of writing this article (October 2013), NEXThaler is marketed in Germany, Spain, and The Netherlands, and has been approved in the following countries: Austria, Belgium, Bulgaria, Greece, France, Hungary, Italy, Luxembourg, Poland, Slovenia, and Slovakia.
The current study was conducted to compare usability on first and second use of NEXThaler versus DPIs delivering inhaled corticosteroid/long-acting β2-agonist fixed combinations currently on the market, Diskus® (GlaxoSmithKline) and Turbuhaler® (AstraZeneca), and to explore patient preferences and perception of device characteristics. The study assessed all three devices by examining three key components of usability: efficiency, effectiveness, and satisfaction (as detailed in ISO 62366:2008: Medical devices—Application of usability engineering to medical devices).
Patients and Methods
Patients
Adult patients of 18 years and older were eligible to enter the study if they had a physician diagnosis of asthma and were regularly using a pressurized metered dose inhaler (pMDI) for at least 3 months prior to enrollment. As asthma is common in people of all ages, patients were selected in order to have a similar number of subjects in different age groups. All patients had to be naive to DPIs. Patients were excluded from the study if they reported an asthma attack that required either a visit to the general practitioner or a hospital admission within the previous 3 months. Patients gave their written informed consent before any assessment was performed.
Outcome measures of usability
The outcome measures for effectiveness of device handling were the number of steps failed and the number of people who were able to use the device successfully without making any critical errors. The outcome measures for efficiency were the time required to set up the device (from the point the participant picked up the device to commencement of inhalation) and the time to read the instructions for use (IFU). Satisfaction was measured with a preference questionnaire, which included questions relating to easiest device to use and preference to own.
A number of preliminary usability activities were conducted by an independent organization (Lloyd's Register Consulting, London, UK) to define steps for all three devices and to develop a comprehensive set of potential credible use errors (i.e., opportunities for step failures), which were included in a master errors log to be used for measurement of effectiveness. A critical error was defined as any error that prevented a dose being delivered. The preliminary activities consisted of heuristic evaluations, systematic human error reduction and prediction approach (SHERPA), and a literature review exploring known issues and errors with DPIs. The heuristic evaluations were conducted by an independent organization (Lloyd's Register Consulting) to assess the devices against a set of well recognized heuristics as defined by Nielsen,(23) and capturing usability issues and potential errors. The SHERPA was used to build on errors identified through the literature review and heuristic evaluations, and was completed by a team of usability experts using the SHERPA error taxonomy and consequence classifications as defined by Embrey.(24)
Trial procedure
Recruited patients were included in a randomized crossover comparison of NEXThaler, Diskus, and Turbuhaler. The trials were conducted by an independent organization (Lloyd's Register Consulting) in professional viewing facilities at three sites in the United Kingdom with a two-way mirror to allow observation of the trials without disturbing participants. All the trials were video- and audio-recorded.
The name of each inhaler was anonymized throughout the trial to reduce any potential negative or positive bias toward specific brands. All medications were removed from the inhalers, and patients were informed that they were testing empty devices. During the trial, the original manufacturer's IFU was used for each device. All branding and logos were removed from the IFUs, as well as any information deemed irrelevant for the purpose of the trial (e.g., details of medication, storage).
The study was counterbalanced using a Latin square to set the presentation order used by each participant. An equal number of participants were given NEXThaler to use first, second, and third. The same approach was adopted with Diskus and Turbuhaler. This approach was used to ensure that the study was counterbalanced in terms of the order of exposure to each inhaler and to mitigate the potential of learning effects. Each time patients were presented with an inhaler, they were instructed to familiarize themselves with the device only by reading the IFU without any verbal explanation. During this time, the experimenter left the room but measured the time the patients took to familiarize themselves with the IFU (maximum allowed time=5 min). When the experimenter returned to the test room, patients were asked to use the device by carrying out steps as detailed in the IFU and thinking aloud to explain what they were doing, any aspects of the device they were checking, as well as any problems they were having. During this process, the experimenter identified errors using the master errors log developed in the preliminary work as reference, but did not provide any feedback to patients. Patients were asked to reuse the inhaler device for a second time. The number of total and critical steps failed during each operation was used as a measure of effectiveness. Patients used the device for the third time without verbalizing what they were doing. Timings for device setup during the third inhalation were used as a key measure of efficiency.
After the second use of each inhaler, users were asked to rate how confident they were they had received a correct dose, and whether they could correctly read the dose counter. Confidence was rated on a 5-point Likert scale from 1 (strongly disagree) to 5 (strongly agree).
These steps were repeated for each inhaler and, once all the inhaler devices had been used, a preference questionnaire was administered. The questionnaire consisted of nine questions based on the preference questionnaire detailed in Schulte et al.,(25) and participants were told that they could freely look at or use the inhalers while answering the questions. The last two questions of the questionnaire were used as key measures of satisfaction, as these two questions were directly related to ease of use and patients' overall preference with the devices.
Statistical analyses
Results were analyzed by using SPSS 20.0 software. Data were reported as means±standard deviation (SD). Confidence scores (i.e., confidence in dose administration) were analyzed using a paired samples t test, and the correct identification of remaining doses data were analyzed using a chi-square test.
Effectiveness data were analyzed with a repeated-measures analysis of variance (ANOVA) using inhaler type (NEXThaler, Diskus, Turbuhaler), error type (critical versus noncritical), and usage (first use, second use) as within-subject factors and using the number of steps failed for each device as the dependent variable. Chi-squares were used to analyze the number of people with successful usage for each device. Efficiency data (i.e., time to set up the device) were analyzed with a repeated measures ANOVA using inhaler as the main within-subjects factor with three levels (NEXThaler, Diskus, Turbuhaler) and time as the dependent variable. Satisfaction was investigated using the preference questionnaire, and the data (i.e., the number of “yes” responses) were analyzed using a chi-square test.
A formal sample-size calculation was not performed, because the study was exploratory in nature. The total number of 60–70 patients was chosen according to previous similar studies.(25,26)
Results
Patients
The study included a total of 66 subjects (32 men, 34 women) with a mean age of 42.9±17.7 years. With regards to the distribution of patients by age, the study included: 16 (24%) patients aged 18–24 years; 18 (27%) patients aged 25–44 years; 18 (27%) patients aged 45–64 years; and 14 (21%) patients aged ≥65 years. Patients had an average history of asthma of 17.0±13.6 years.
Effectiveness of device handling
Mean number of steps failed for each device and for each error type at first and second use are illustrated in Figure 1. The overall number of errors decreased from first to second use with each inhaler (p<0.001). Bonferroni comparisons showed that the number of step failures for NEXThaler was less than for Diskus (p<0.001) and Turbuhaler (p<0.001) for critical and any errors made in both first and second use. It was also revealed that patients made fewer step failures for Diskus than for Turbuhaler (p<0.001) in both first and second use for both critical and any errors. Error frequency of each step required for a complete inhalation maneuver for each device is detailed in Table 1. The proportion of participants who completed a successful inhalation without any errors at all was significantly higher for NEXThaler than for Diskus and Turbuhaler on both first and second use (p<0.001) (Fig. 2). Participants completed more successful inhalations without any critical errors for NEXThaler and Diskus than for Turbuhaler in the first use (p<0.05). On the second use, the percentage of patients completing an inhalation without critical errors was 71% for NEXThaler, 59% for Diskus, and 47% for Turbuhaler. Mean confidence scores in correct dose delivery were 8.78±1.53 for NEXThaler, 7.57±1.90 for Diskus, and 5.86±2.00 for Turbuhaler. Paired-samples t tests showed that patients were more confident that they had received the correct dose with NEXThaler than with Diskus and Turbuhaler (p<0.001). Patients also showed more confidence in Diskus than in Turbuhaler (p<0.001). Analysis on patients' ability to accurately read the dose counter showed that patients had more difficulty with Turbuhaler than with the other two devices (p<0.01).
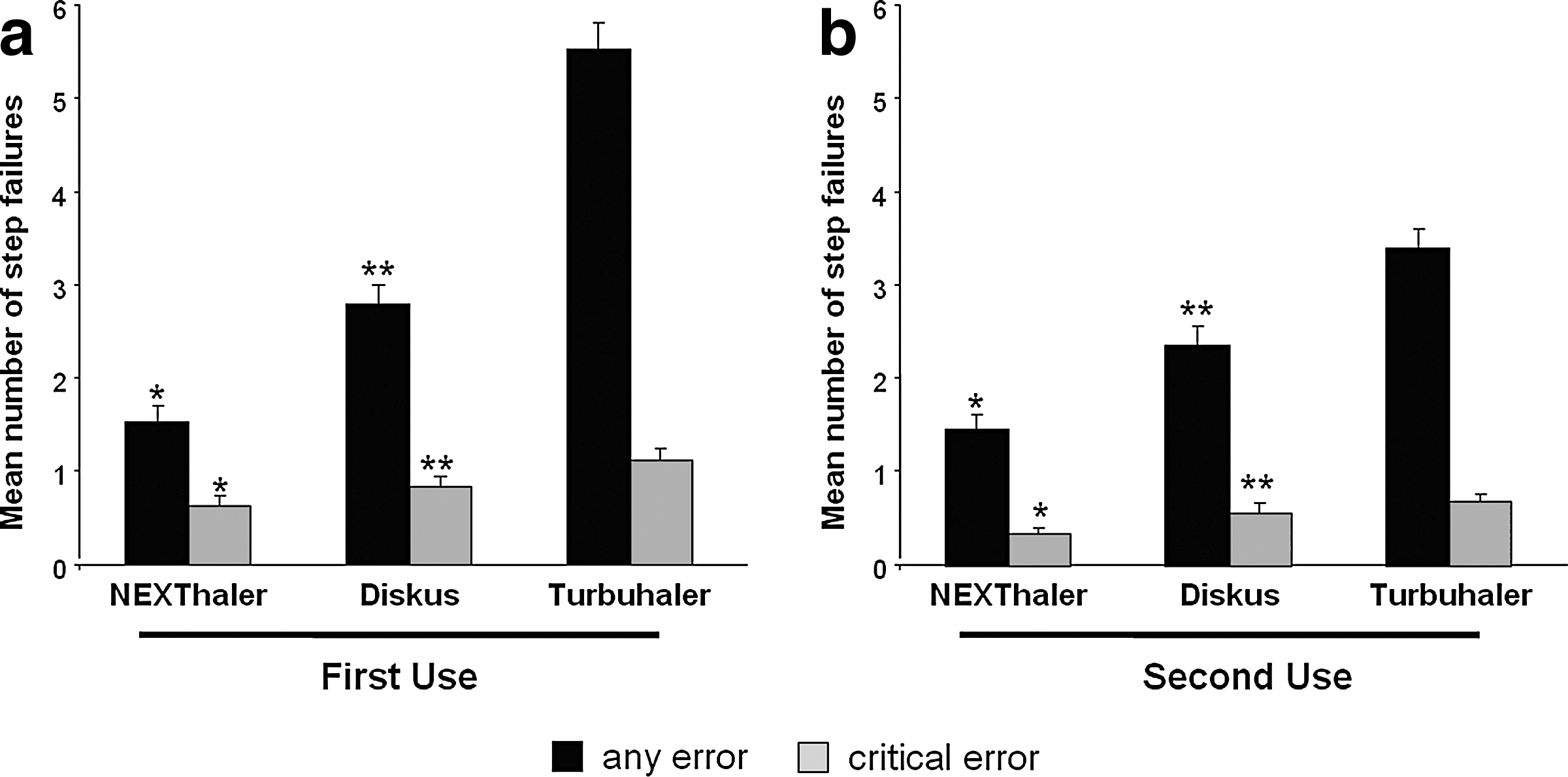
Mean number of step failures per inhaler on
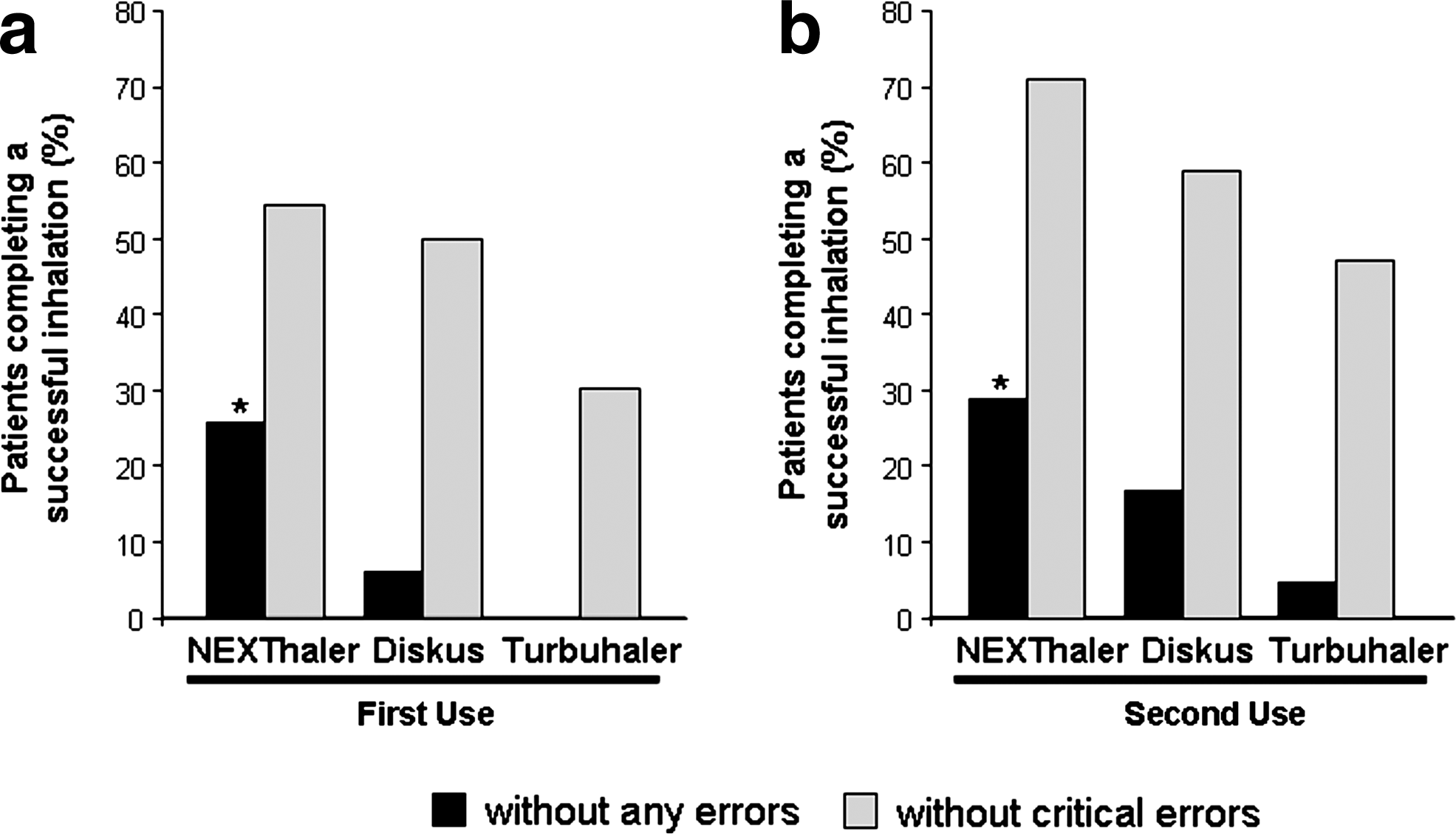
Percentage of patients who completed a successful inhalation on
A comprehensive set of potential critical and noncritical errors was defined for each inhalation step. The table lists the proportion of patients making at least one error (both critical and noncritical). Values in brackets indicate the proportion of patients making critical errors.
Efficiency of device handling
Patients took significantly more time to set up Turbuhaler (8.69 sec) and Diskus (7.85 sec) than NEXThaler (6.02 sec) (p<0.001, Bonferroni adjustments). The difference in time to set up Turbuhaler and Diskus was not significant (p=0.188). Average IFU reading durations per syllable for each of the IFUs were significantly lower for NEXThaler (0.15 sec) than for Diskus (0.32 sec) and Turbuhaler (0.2 sec) (p<0.001).
Satisfaction of device handling
Patients' preferences with respect to different device features are presented in Table 2. NEXThaler was rated as easiest to open, prepare, and set a dose (p<0.001 vs. Diskus and Turbuhaler), easiest to tell how many doses are left (p<0.001 vs. Turbuhaler), and easiest IFU to understand (p<0.001 vs. Turbuhaler). Responses to overall preference questions were used as key measures of satisfaction. Patients rated NEXThaler as the easiest to use (p<0.001 vs. Diskus and Turbuhaler) and most preferred inhaler to own (p<0.001 vs. Diskus and Turbuhaler). Results on overall ease of use and overall preference to own were correlated (47 out of 49 patients who rated NEXThaler as the easiest to use rated NEXThaler as the most preferred inhaler to own).
p<0.001, NEXThaler vs. Turbuhaler.
p<0.001, Diskus vs. Turbuhaler.
p<0.001, NEXThaler vs. Diskus.
Discussion
The aim of the current study was to investigate usability characteristics (effectiveness, efficiency, and satisfaction) of NEXThaler versus Diskus and Turbuhaler. This study was performed in DPI-naive asthmatic patients who familiarized themselves with the devices only by reading the IFUs without any verbal explanation. It is known and reported in the literature that IFU reading is the only way of instruction for a significant number of experienced inhaler users in real life.(10,27) Training limited to reading of the IFU only is not recommended, and the ideal scenario is for patients to be trained on how to use the device by health-care givers. However, literature data report that up to 25% of patients have never received inhaler technique instructions by health-care professionals.(17) Other authors have already evaluated device handling after reading of written instructions(28) but, to our knowledge, this is the first study investigating also IFU readability. Recent research has shown that the most important features for inhaler selection are ease of use, preference, and consistent dose delivery.(5) Also, ideally inhalers need to provide clear feedback to the patient that the dose was released and that inhalation has occurred correctly.(8) NEXThaler is a DPI that has been designed to overcome some of the limitations of the currently marketed devices. The full dose feedback system provides accurate dose metering and consistent full dose release independently of the inspiratory flow.(18,19) NEXThaler has a unique “open-inhale-close” operating sequence that is at least one inhalation step less than that of other existing DPIs and easy for patients to use. Complex operating systems requiring several handling steps can increase the chance of making an error and lead to unintentional nonadherence of patients using their medication.(29) Our findings showed that more patients carried out steps leading to a successful inhalation without making any errors at all for NEXThaler. The correct inhalation steps for each device were identified during preliminary usability activities and were comparable to those reported in similar articles assessing DPIs such as Diskus and Turbuhaler.(10,30,31) Overall, similar steps were failed for NEXThaler and Diskus, specifically those involved with checking the dose counter, inhalation, and exhalation away from the mouthpiece. Similar step failures were observed also for Turbuhaler, but with a high proportion of participants failing to complete the priming step successfully. Categorization of patients by age indicated that the frequency of errors was similar in all age groups. As for checking the dose counter before use, error frequency in this step tended to increase from first to second use with both NEXThaler and Turbuhaler. The main reason given by participants for failing to check the dose counter on the second use was that they had just checked it after finishing the first inhalation. This could be interpreted as an artifact of the study design, because the two inhalations were conducted sequentially one after the other.
It can be difficult to compare studies on error rates due to the lack of a gold standard for the evaluation of proper inhalation technique, different definitions of errors, and variations in the characteristics of selected patients. Alternative study designs have been used in previous usability studies, such as assessing error frequency with the inhaler that patients were regularly using for treatment or with an inhaler that they had never used before. In our study, we enrolled patients who were pMDI users only and had no previous experience of using a DPI. Consistently with other publications,(10,30–32) this study showed that there were fewer errors (both any errors and critical errors) when using Diskus compared with Turbuhaler. Although we found that error frequency during the loading step of Diskus and Turbuhaler was similar at first use, data recorded at second use indicated a lower loading error frequency with Diskus than Turbuhaler, which is comparable to results reported in similar studies involving experienced inhaler users.(10) This is the first study reporting failure rates for loading NEXThaler that ranged from 1.5% to 4.6%. Dose loading is an important step of the inhalation technique that can affect effective use of DPIs, and a difficult loading process can explain the increased level of errors observed with the different inhalers tested. Another important step for proper use of a DPI is to inhale forcefully from the start. Melani et al. reported that 22–28% of patients made a slow and not forceful inhalation when using their regular DPI.(10) In our population of DPI-naive patients, error frequency for this step ranged from 27.3–34.9% at first use to 16.7–33.3% at second use. Although our study was based on a subjective assessment of the inhalation technique, the findings are in line with those of observational studies that used objective measurements of the inspiratory flow or technique.(33,34)
The specific characteristics of the device can potentially explain the significant reduction in inhaler mishandling observed with NEXThaler. For example, although all three devices have a feedback system that indicates to the user how many doses are left, only the BAM of NEXThaler would change the dose counter only after a successful inhalation has occurred. Diskus, in contrast, would change the dose counter if the steps leading to inhalation have occurred even without inhalation, whereas the Turbuhaler counter marks out the remaining doses only in tens. Indeed, participants in this study had more difficulty in accurately determining the remaining number of doses on Turbuhaler. Many participants also heard the click associated with the activation of the NEXThaler BAM and indicated that they were reassured by this audible feedback that the dose had been released. The improved feedback systems may account also for the higher confidence in dose release shown by participants toward NEXThaler. Feedback signals are important to reassure patients that they are taking their medication correctly, and inclusion of feedback mechanisms signaling that the dose has been emitted and inhaled correctly are warranted to improve inhaler characteristics.(8)
In terms of efficiency of device handling, our findings show that NEXThaler was the quickest to be set up and its IFU the quickest to be read. As previously noted, the time to set up a device was one of the key factors patients valued.(35) While patients in the real world would receive training by a medical professional, the IFU leaflets acted in place of the training in our study. Previous studies have shown that patients tend to make frequent errors when using the device for the first time with only the help of the leaflet.(25) Our results indicate that a clear and easy-to-read IFU can help patients develop and maintain a proper inhalation technique, already from the first time they are using an inhaler device. In general, participants were able to quickly grasp the information, and the improved performance observed from the first to the second use reflected the effect of familiarization with the devices.
Responses to the overall preference questions indicate that NEXThaler was preferred more than Diskus and Turbuhaler. Similar to the study by Schlaeppi et al.,(36) we found that Diskus was preferred over Turbuhaler. Our findings that Turbuhaler scored better than Diskus in factors related to its size and that Diskus was favored for its counting mechanism are consistent with those reported by van der Palen et al.(30) Patient preference for a particular inhaler has been described as a valid patient-reported outcome to be assessed and included in scientific studies, as increased preference could potentially lead to improved adherence and clinical outcomes.(7) Nonadherence to treatment is frequent among asthma patients and is associated with many adverse consequences. It has been reported that low levels of adherence are associated with more frequent exacerbations and hospital admissions.(37,38) Additionally, patients with high adherence to medical therapy experience better asthma control and improved quality of life.(39) In our study, 96% of patients who found NEXThaler as the easiest to use also preferred to own it as well, lending support to the claim that ease of use and patient preference may be linked. When the inhaler device is easier to use and provides accurate feedback, or when a dose is successfully delivered, patients are more likely to use it correctly in accordance with their treatment program.(6)
Usability and preference studies present some limitations. In our study, the inhalation technique was assessed subjectively by observers. For example, the decision that an inhalation was not as forceful as required was not confirmed by objective measurements of the breathing patterns. This limitation was possibly reduced by using a standardized checklist clearly defining the correct inhalation steps and errors for each device and by giving the same repeated instructions to the experimenters. Additionally, as the study had to be performed unblended, all brands and logos were removed from the devices and DPI-naive patients were selected in order to avoid any preference bias. Another potential limitation could be represented by having conducted the study in one country only, and this could possibly lead to country limitations in the applicability of the current findings.
For a device to be successfully used, it must be effective, efficient, and enhance user experience (satisfaction).(7) Previous research has shown that ease of use and patient preference are two important considerations for device selection.(40,41) This study has shown that although all the three devices tested will enable patients to use the device successfully, usability measures demonstrate that NEXThaler is rated as better than Diskus and Turbuhaler in terms of patient satisfaction (preference), effectiveness, and efficiency of use. Rigorous scientific studies are required to evaluate the link between patient preference and device characteristics with treatment adherence.
Footnotes
Author Disclosure Statement
T. Voshaar has been reimbursed for attending conferences and/or giving talks by Boehringer Ingelheim, Pfizer, Chiesi, UCB, GlaxoSmithKline, Novartis, Mundipharma, and TEVA. He has acted as a consultant for Boehringer Ingelheim and Pfizer. A.S. Melani has received fees as a consultant and advisory board member from Chiesi Farmaceutici S.p.A., Artsana, Novartis, and Sanofi-Aventis. M. Spinola, P. Linnane, A. Campanini, M. Scuri, and B. Ronca are full-time employees of Chiesi Farmaceutici S.p.A.
