Abstract
Abstract
Spacers and valved holding chambers (VHCs) are pressurized metered dose inhaler (pMDI) accessory devices, designed to overcome problems that patients commonly experience when administering aerosol via a pMDI. Spacers were developed in direct response to patient-related issues with pMDI technique, particularly, poor coordination between actuation and inhalation, and local side-effects arising from oropharyngeal deposition. Current clinical guidelines indicate the need for widespread prescription and use of spacers, but, despite their apparent ubiquity, the devices themselves are, unfortunately, all too commonly "disused" by patients. An understanding of the background from which spacers developed, and the key factors influencing the optimization of the spacer and the later VHC, is crucial to developing an appreciation of the potential of these devices, both contemporary and future, for improving the delivery of pressurized aerosols to patients. This review, informed by a full patent search and an extensive scientific literature review, takes into account the clinical and laboratory evidence, commercial developments, and the sometimes serendipitous details of scientific anecdotes to form a comprehensive perspective on the evolution of spacers, from their origins, in the early days of the pMDI, up to the present day.
Introduction
I
Here we review the scientific history of spacer devices, informed by a full patent search, an extensive review of scientific literature, and first-hand experience of this evolving field. This focus is distinct from an approach based on market share or product availability, and follows a chronological structure; the timeline presented in Figure 2 provides a basis for this chronological approach.
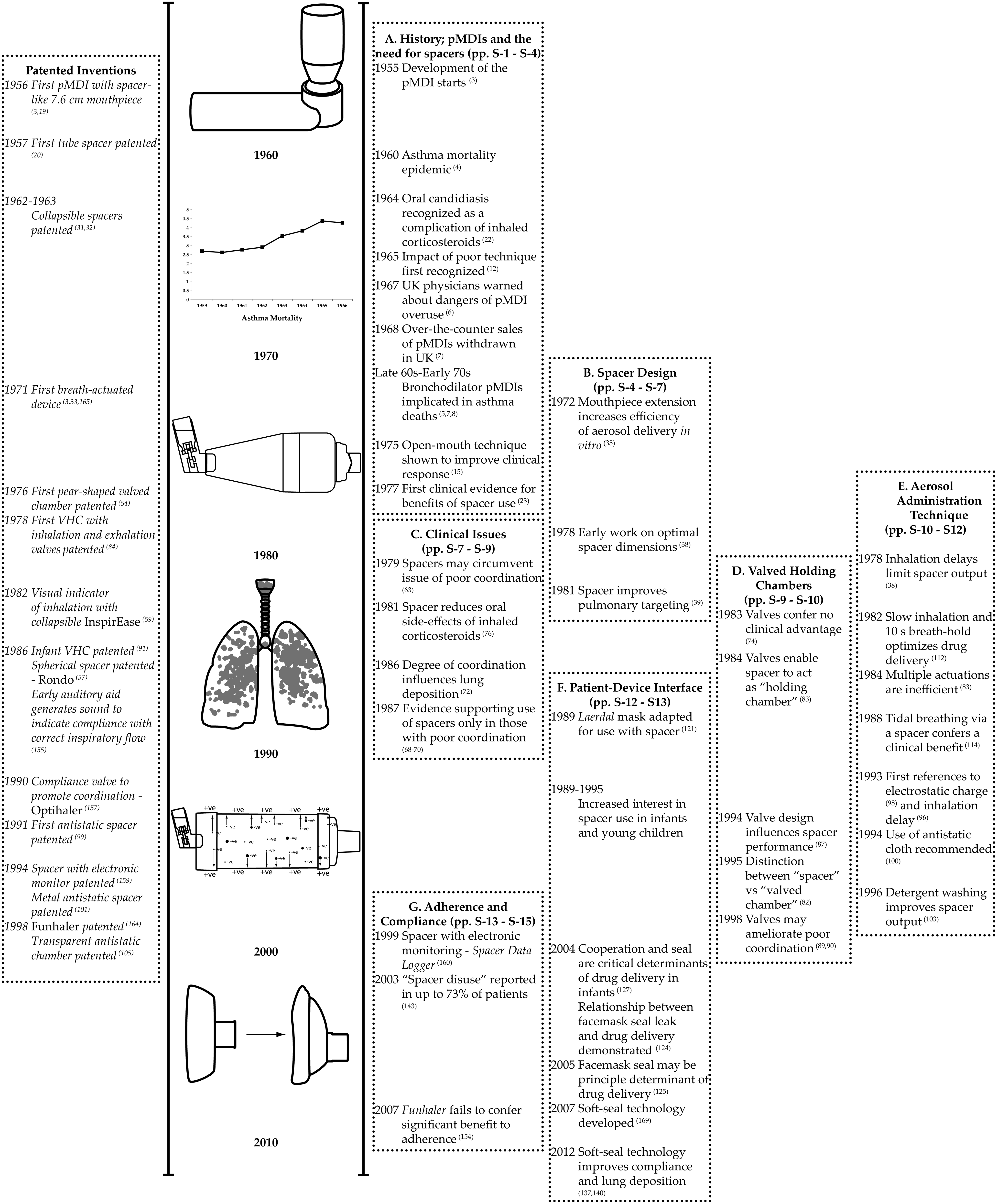
Timeline of key events in the evolution of spacers. Patented inventions (
The literature covers a period of over 60 years; thus, there is an inherent use of terms no longer considered accurate. For the purpose of this article, the term “spacer” refers to all accessory devices, including pMDI mouthpiece extensions and VHCs. Terminology regarding drugs has also developed; to avoid confusion, drugs are referred to by their contemporary class, or generic name, rather than the historical class that was used in the original source.
A. History; pMDIs and the Need for Spacers
Origins of the pMDI
The pMDI originated when, in the spring of 1955, 13-year-old Susie, the daughter of the then managing director of Riker Laboratories, Inc., asked her father, “Daddy, why can't they put my asthma medicine in a spray can like they do hair spray?”(3) At that time, patients administered their bronchodilators via small, hand-held jet nebulizers, which were equipped with a rubber bulb. These devices had no means by which to meter a reproducible dose.
By the summer of the same year, clinical trials with the prototype pMDI were in progress, and, having demonstrated their effectiveness, the first pMDIs, isoproterenol sulfate and epinephrine bitartrate (Medihaler-Iso and Medihaler-Epi, Riker Laboratories, Inc., Los Angeles, CA), were launched in March 1956(3) (Fig. 3). The pMDI remains unrivaled as a convenient, reliable, and relatively inexpensive (per dose) means by which to deliver inhaled medications. However, pMDIs are not without their own issues, which include patients' difficulties with their correct, coordinated use; many patients, including the very young, confused, intellectually impaired, or those experiencing acute severe breathlessness, will be unable to inhale in anything other than tidal breaths. It is some of these issues that spacers are designed to overcome.
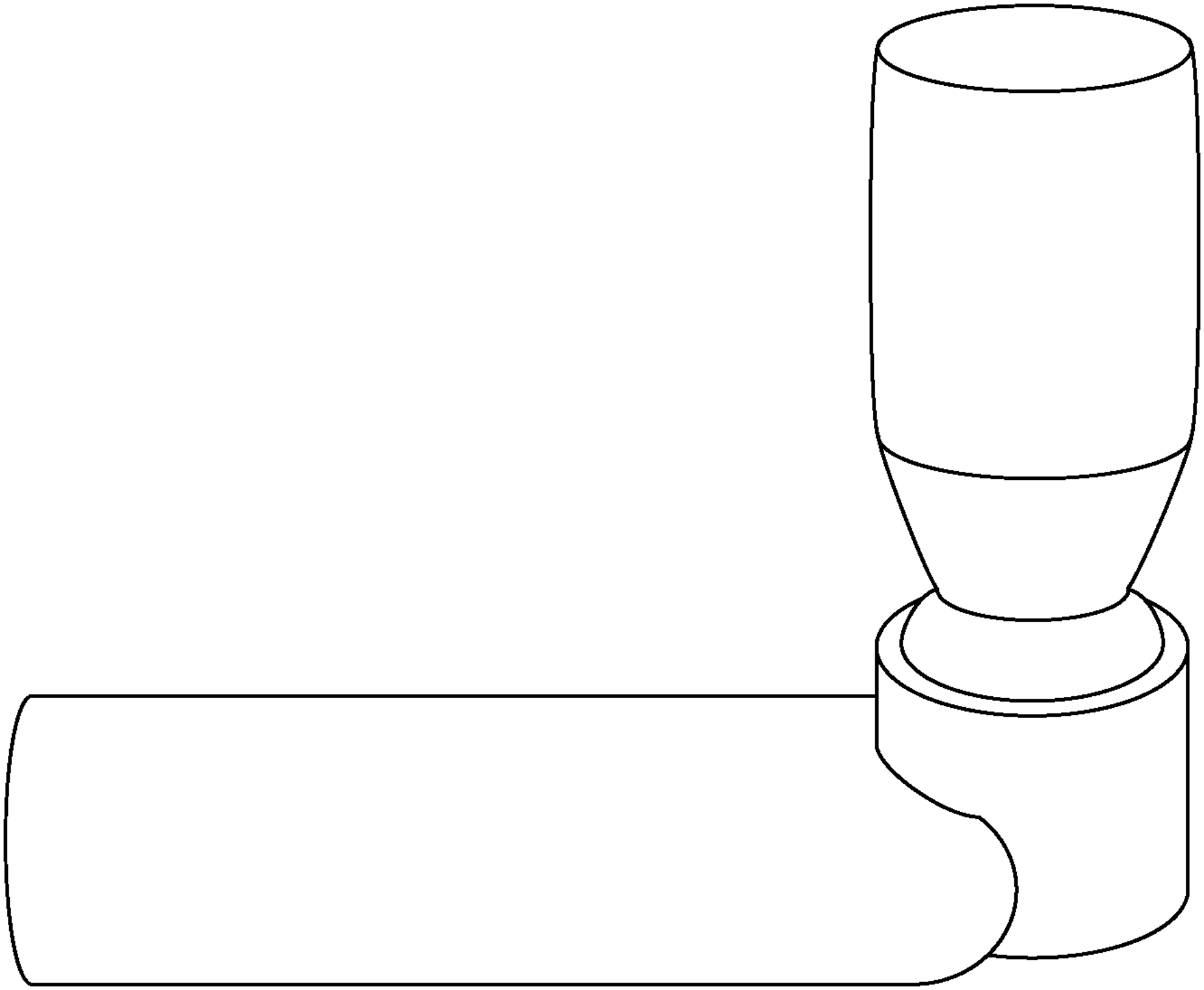
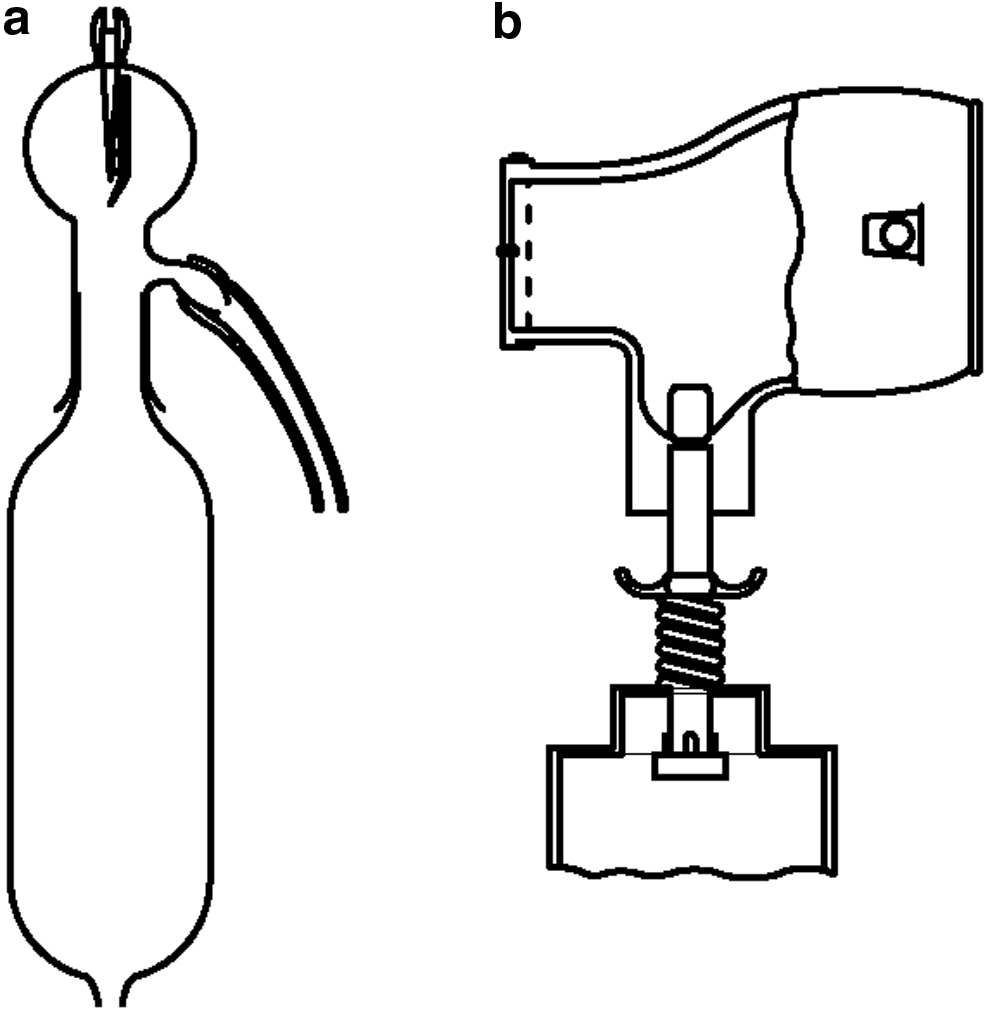
Medihaler pMDI. The original pMDI was equipped with an elongated mouthpiece, arguably making it the first tube spacer. Maison, 1956.(19)
Early problems and challenges with pMDI use
Shortly after the launch of the pMDI, a striking epidemic of asthma-related deaths hit the UK, Norway, Australia, and New Zealand (Fig. 2). In the UK, asthma mortality in those aged 5–64 years almost doubled. Most disturbingly, mortality among younger people showed the steepest increase; the asthma death rate in patients aged 5–35 years more than trebled, while a dramatic eightfold increase was observed in those aged 10–14 years.(4) Although initial suspicion focused on suggestions of an etiological role for corticosteroids,(5) several detailed analyses of mortality data shifted concern to the overuse of bronchodilator pMDIs. In a review of asthma mortality trends, it was noted that the UK epidemic coincided with a fourfold increase in pMDI sales, between 1961 and 1966.(4) By 1969, the epidemic was abating; this corresponded with a greater awareness of the dangers associated with excessive use of pMDI bronchodilators. In the UK, for example, an official warning regarding safe use of pMDIs, issued to all UK physicians in June 1967,(6) was followed by the nationwide withdrawal of over-the-counter sales of bronchodilator pMDIs in December 1968.(7)
Retrospective data implicated overuse of aerosolized bronchodilators, primarily the β-agonists isoprenaline and orciprenaline.(8) It has been widely suggested that patients and physicians developed an overreliance on bronchodilators, such that symptom severity was disregarded, and patients did not seek appropriate care. Lack of appreciation of the role of aerosolized β-agonists in asthma management, alongside a poor understanding of correct technique, may also have contributed to the overuse of (perhaps incorrectly administered) pMDIs. Systemic absorption of large doses of nonselective β-agonists might also have played a part in patients' eventual cardiorespiratory collapse. Since this initial episode, other countries have witnessed similar epidemics, although the role of β-agonists in these epidemics remains controversial.(9–11) Regardless of the explanation, it is clear that the “aerosol hypothesis”(10) was a crucial stimulant to increasing the education of physicians, the recognition of the potential dangers of β-agonists, and an appreciation of the need to understand the safe and effective use of pMDIs.
By the mid-1960s, the complexities of pMDI technique had also gained some recognition. A need for synchronized actuation and inspiration, later termed “hand–lung coordination,” was noted as early as 1965, when it was reported that, despite “adequate instruction,” patients frequently used aerosols incorrectly.(12) Over the subsequent decade, the impact of poor technique on therapeutic effect became clear; in one study, 75% of patients failed to gain maximal benefit from self-administered bronchodilators,(13) whereas another study found that a significant minority were “unable” to use their pMDI, due to inefficient technique, “in spite of careful tuition.”(14) An early attempt to address patients' difficulties with pMDI technique was the use of the “open-mouth” method, whereby aerosol was fired into an open mouth from a distance of several centimeters. Intended to facilitate aerosol entrainment and reduce oral impaction, this method improved patients' bronchodilator response(15) and doubled the delivery of radiolabeled aerosol to the lungs, even in those with good coordination.(16) For this reason, the open-mouth technique was recommended prior to the widespread availability of spacers,(17) and later as an alternative to spacers in patients with good coordination.(18) Some authors found that the degree of improvement in lung deposition observed with use of the open-mouth technique was comparable to the degree of improvement associated, in other studies, with use of a tube or pear-shaped spacer.(16) However, although this technique was also anecdotally reported to reduce the development and persistence of oral candidiasis,(16) a local side-effect caused by oral impaction of inhaled corticosteroids, the method would not resolve either the issue of poor coordination or patients inhaling in tidal breaths. Thus, although by the mid-1970s problems relating to poor hand–lung coordination and the oral impaction of aerosol were well-recognized, they were far from resolved.
Early spacer patents and development
Despite the fact that it took 2 decades, from the introduction of the first pMDIs, before there was widespread appreciation of their clinical challenges for the patient, the development of spacer devices began far earlier. The original pMDI was equipped with an elongated 7.6-cm mouthpiece, arguably making it the original tube spacer (Figs. 2 and 3).(19) Just a year later, the first accessory device was designed specifically as a simple tube spacer.(20)
A pMDI emits large, high-velocity droplets, which are prone to impaction in the upper airways. This issue was recognized as early as 1958, when Franklin
In 1977, Freigang, a Canadian pediatrician, published a seminal case series that described “beclomethasone aerosol by reservoir bottle” or “BARB.”(23) Developed to improve the delivery of aerosolized corticosteroids in children, BARB provided the first evidence of the clinical efficacy of spacers. A 1.2-L vinegar bottle served as a reservoir, while a facemask with a one-way valve was used as an interface (Fig. 4). Remarkable clinical benefits were reported, including, at the time of publication, no further hospital admissions in any of the 14 cases. Initially a “homemade” device, Dr. Freigang explained, “I soon tired of gluing my fingers together—and the following year a plastics company manufactured the device.”(24) Freigang's work may have started a long tradition of homemade spacers, driven initially by demands for a convenient supply of devices and more recently by the requirement for cost-effective spacers for use in developing countries.(25–28)
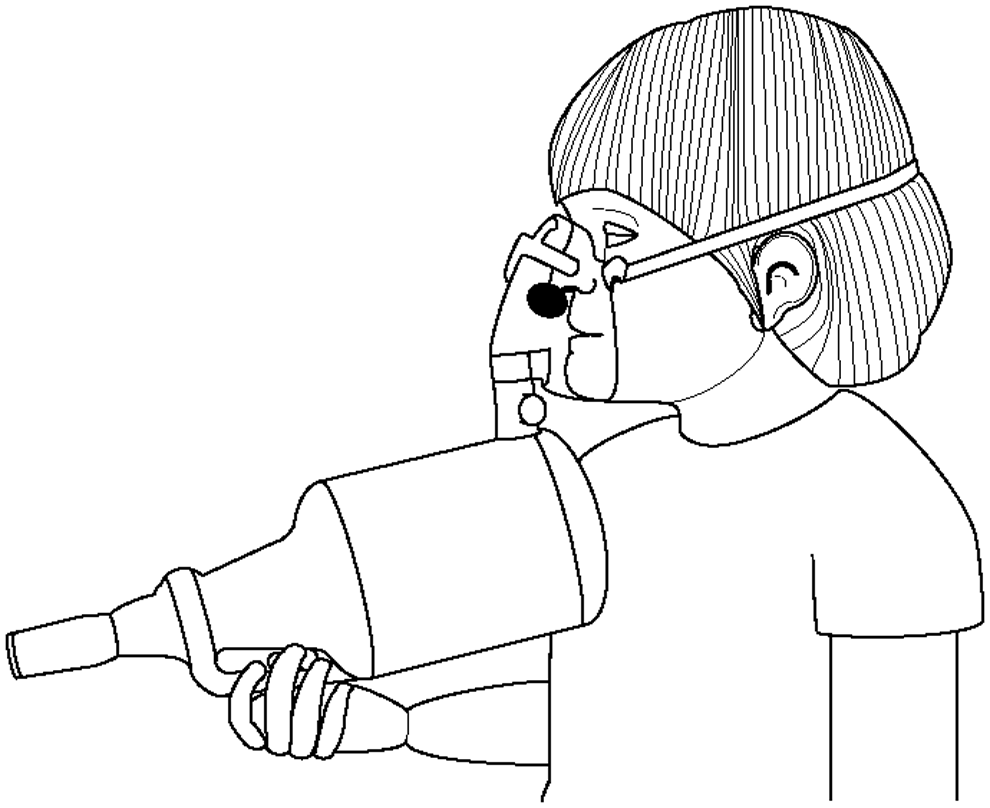
Beclomethasone aerosol by reservoir bottle (BARB). Spacer system comprised a 1.2-L vinegar bottle and facemask with one-way valve. Freigang, 1977.(23)
Between 1957 and the development of the first commercial valved spacer in 1976, an abundance of spacer patents were filed (Figs. 2 and 5). These novel designs, which focused on improved convenience and portability, included spacers with foldout mouthpieces(29,30) and spacers that were completely collapsible.(31,32) The delay between the patenting of early spacers and their commercial development reflects the lag time between the introduction of pMDIs and widespread recognition of their associated challenges, together with a lack of experimental support for spacers. One of the earliest commercially available devices, based on Hart's 1963 patent(32) and manufactured between 1967 and 1971, was a smoking cessation aid that was designed to deliver aerosolized lobeline hydrochloride (Antisol Anti-Smoking Aerosol, Aerosmoke Ltd., Newbury, Berkshire, UK). The device incorporated an extended, spacer-like mouthpiece, which folded conveniently over the pMDI.
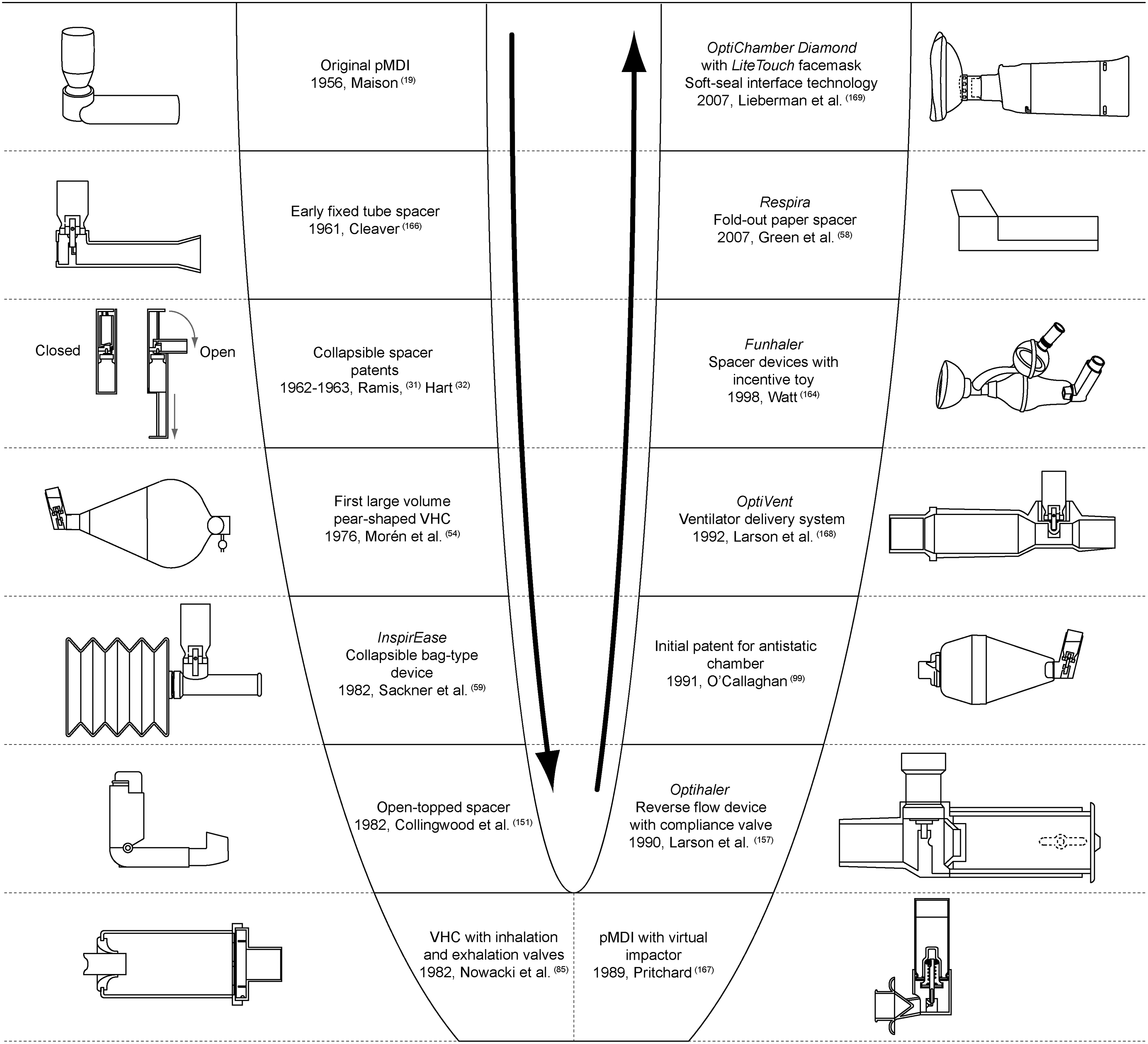
Historical development of spacer design.
Perhaps surprisingly, the commercial success of spacers was preceded by the development of the first “breath-actuated” device (Duohaler, Riker Laboratories, Inc.), which was launched in 1970 and, like the spacer, was intended to overcome the issue of poor coordination.(3,33) However, the device was poorly received, in part due to an objectionably loud clicking sound made on actuation,(3) and clinical studies of the time failed to demonstrate any significant benefits over a conventional pMDI.(34) Even though they were designed to overcome some of the same pMDI-related issues as spacers, breath-actuated pMDIs would fail to gain popularity for a further 2 decades.
B. Spacer Design
Early formal spacer experiments began in the mid to late 1970s, following, and probably influenced by, the burgeoning use of inhaled corticosteroids, growing recognition of patients' difficulties with pMDI use, and the reported effectiveness of the open-mouth technique. Attention initially focused on the establishment of optimal design parameters, and later turned to the contribution of valves, the influence of procedural issues, such as inhalation delay, and the effect of static charge. Studies determining
Early investigation of spacer dimensions
Optimal spacer dimensions were an early concern for investigators, and yet areas of controversy still remain. Initial investigations into the influence of mouthpiece length on the efficiency of “pulmonary” delivery used an
In the late 1970s, Morén investigated the influence of spacer dimensions on the
Spacer volume; practicality and clinical considerations
Even at the earliest stages of spacer evolution, there was recognition of the cumbersome and impractical nature of large-volume spacers, and the need to balance optimal dimensions for aerosol delivery, with clinical convenience. For example, in the design of an early commercial spacer, Corr
Deposition studies were not entirely supportive of the shift toward favoring small-volume VHCs; a review of radiolabeling studies indicated that larger-volume spacers (volume greater than or equal to 750 mL) were associated with greater whole-lung deposition, compared with a pMDI alone, and also tended to improve pulmonary targeting, compared with smaller devices.(42) As predicted by Morén,(38) the large volume reduced loss to the internal walls, and the pear shape allowed deceleration and evaporation. However, it should be noted that the authors of this review highlighted that, whereas “engineers are likely to opt for the most efficient (device)” in terms of aerosol output, “clinicians have to take other factors into account to obtain optimum patient compliance.” Factors such as ease of use, pocketability, and, perhaps most importantly, clinical response must clearly trump any small improvement in
A decade later, the respirable dose delivered via cylindrical spacers of different lengths (5–50 cm) and diameters (3–10 cm) was investigated by Barry and O'Callaghan.(43) Whereas spacer diameter correlated positively with respirable fraction, the effect of length plateaued at 20 cm, equivalent to approximately 1-L volume, beyond which extension conferred little benefit. Holding space for deceleration was considered the principal means by which spacers increased the fraction of respirable particles. This study highlighted that output was likely to be determined by the pMDI–spacer combination, rather than a universally optimum spacer.(43)
Work by Bisgaard supported this “plateau” effect of spacer length; the stable respirable fraction of approximately 30%, which was observed with tubes longer than 13 cm, fell when the length of the spacer was reduced.(44) This phenomenon is likely related to plume velocity, which is expected to have slowed to the equivalent of inspiratory flow within a 10-cm distance from the actuator (45) (Fig. 6); for infant inhalation flows, a spacer with a length of 13–20 cm would therefore be expected to provide a sufficient distance over which the plume could decelerate. Taken together, these results indicate that both diameter and length influence
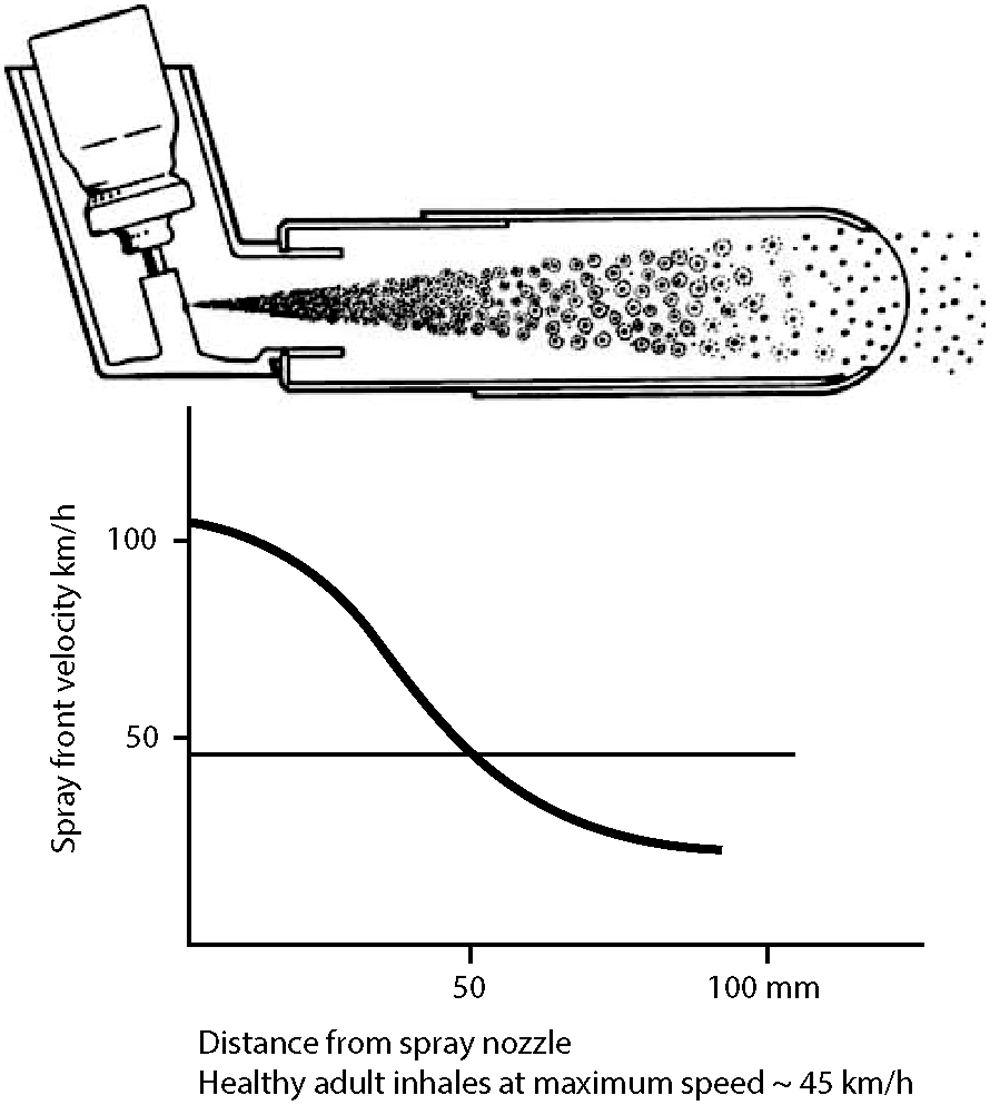
The late 1970s tube spacer (Inhalet, Astra) was designed to extend the distance from the mouthpiece to the patient's mouth such that aerosol reaching the patient's mouth would be equivalent to the speed of tidal inhalation. From Nikander.(45)
Despite the conclusion, by some, that small-volume devices (150–200 mL) are comparable to large spacers,(46) respirable dose has been shown to correlate with volume up to approximately 1 L, after which the relationship plateaus.(43) This is consistent with recent reviews, which suggest that larger chambers confer advantages in terms of aerosol quality.(47,48) Such advantages must be balanced with the impracticality of larger devices; several studies investigating adherence to a prescribed regimen of spacer use have indicated that convenience, or rather the perceived inconvenience associated with use of an accessory device, may be important factors contributing to “spacer disuse.”(49,50) If the cumbersome nature of large-volume spacers results in high levels of disuse, any improvement in aerosol quality, relative to a small-volume device, is likely to become irrelevant.
Conflicting results and study design
Consideration of study design may resolve some contradictory results; studies supporting the equivalence of large- and small-volume spacers tend, in general, to be
The combinations of pMDI and spacer used in different studies may also be a source of inconsistency in the results; whereas some spacers, particularly contemporary devices, may be used with a range of pMDIs, others are manufactured for use with specific pMDI drug formulations. Such manufacturer-recommended spacer–formulation combinations are frequently disregarded in the experimental literature, which might account for some of the observed discrepancies in results. Results of an investigation into the relative influence of parameters of breathing (tidal volume, respiratory rate) and pMDI/spacer combination on aerosol deposition
Size, shape, and clinical evidence
It should be noted that the evidence that indicates an effect of spacer size on clinical response is sparse, and improvements in particle size or lung deposition may not equate to clinically significant differences.
After the patenting of the first nontubular spacer, a large-volume, cone-shaped device, in 1976 (Fig. 5),(54) the relationship between spacer shape and drug delivery attracted increasing interest. Lindgren
By the mid-1960s, the development of high-speed photography facilitated the qualitative assessment of pMDI plumes.(3) This revealed that different pMDIs emit plumes with distinct morphology; given that consideration of the theoretical shape of a pMDI spray had influenced the pear-shaped spacer design, this realization had obvious implications for the generalizability of conclusions concerning spacer shape, as Barry and O'Callaghan noted in 1995.(43) More recent commercially developed patents have included a spherical device (Rondo, Leiras Takeda Pharmaceuticals, Turku, Finland),(57) a fold-down paper device (Respira, Respira Design, www.respiradesign.org),(58) and inflatable bags with no fixed shape (InspirEase, Key Pharmaceuticals, Inc., Division of Schering-Plough, Kenilworth, NJ).(59) Whether one particular design confers any real clinical advantage remains unclear.
C. Clinical Issues
Despite experimental support for the value of spacers, the need to demonstrate the clinical relevance of any observed improvement in drug delivery was paramount. When considering studies that have attempted to relate use of a spacer, compared with a pMDI alone, to improvements in clinical outcome, it is important to appreciate some of the challenges in experimental design. The first concerns the assessment of coordination, because the association between assessed technique and therapeutic response remains relatively weak. Between-study comparisons are confounded by differences in the definition of correct technique, and studies evaluating the relationship between pMDI competency and bronchodilator response in adults have yielded inconsistent results,(60,61) despite there being an apparently positive relationship in children.(62)
Studies seeking to demonstrate the effectiveness of using a pMDI with spacer, compared with a pMDI alone, have generally used β-agonist pMDIs, rather than, for example, inhaled corticosteroids. It is plausible that this use of β-agonists may limit the interpretation of such studies, given that it is expected that patients inhaling high doses of bronchodilator drugs will be close to the plateau of the dose–response curve. However, regardless of this concern, several studies have demonstrated improvements in clinical measures when a pMDI with spacer is used, compared with a pMDI alone.(54,55)
Spacers and hand–lung coordination
By the late 1970s, it was recognized that use of a spacer might mitigate imperfect hand–lung coordination. Bloomfield
Clinical improvements; conflicting evidence and current guidelines
Similar results have been found in a number of other clinical studies. In children with asthma, terbutaline delivered via a tube spacer, compared with a pMDI alone, elicited better responses in a range of clinical parameters, when measured at 20 min and 60 min post inhalation.(64) Immediate postinhalation measures did not differ between the methods. Hidinger and Kjellman demonstrated that children with asthma achieved a significant improvement in PEFR and a higher mean maximum PEFR, after terbutaline was administered via a spacer, compared with the response to terbutaline from the pMDI alone.(65) Terbutaline was also shown to result in a greater bronchodilator response when administered via a pear-shaped spacer (volume, 750 mL), compared with either a nebulizer or a pMDI alone.(66) Whether the observed improvement—in the region of a 12–17 L/min increase in PEFR at each time point—would confer substantial clinical benefit is uncertain.(65)
Other studies assessing the effectiveness of various pMDI accessory devices have failed to replicate such positive results.(67–70) Gomm
The association between coordination and lung deposition, which has been used as a proxy for the relationship between coordination and clinical response, was comprehensively addressed by Newman
Following the recognition of such problems with experimental design, in 1987 a spate of studies indicated that clinical benefit might be restricted to “poor coordinators.” In one study, improvements in FEV1 were observed only in patients with “incorrect technique,”(69) whereas a different study reported that lack of improvement in PEFR was attributed to “good” technique,(68) although, in the latter study, coordination was not formally assessed. Another study concluded that, when compared with the open-mouth method, a spacer conferred only a “marginally clinically significant” benefit.(70) This lack of clinical improvement was consistent with the recommendation that spacers were “not necessary” in most cases, though “should be of use for less coordinated patients,” in addition to those taking high doses of β-agonists via a nebulizer.(66) These conclusions also reflected those of a 1984 review of double-blind crossover studies investigating the clinical efficacy of spacers,(74) which indicated that large spacers, but not small spacers, consistently improved FEV1, regardless of the presence of a valve. These reviewers suggested that the benefit of large cone spacers may be from alterations in regional lung deposition; this explains an observation that is otherwise surprising, given that total dose was approaching the dose–response plateau.
Given that clinical research has generally focused on the benefits of spacers in the context of delivering β-agonists, much of the literature may be less relevant to other inhaled drugs, such as corticosteroids or anticholinergics. The oral impaction of inhaled corticosteroids may be complicated by oropharyngeal candidiasis and dysphonia. Mouth rinsing and counseling regarding voice strain were early strategies intended to minimize the risk of such local complications.(75) Subsequently, the same group published evidence that use of an early small-volume spacer reduced these side-effects, consistent with reductions in oral deposition(76); worryingly, asthma control “deteriorated slightly,” suggesting that use of the device was also associated with a reduced lung dose. A later, more comprehensive randomized trial, which also used a slightly larger-volume spacer, indicated that spacer use improved both oral and respiratory measures, and facilitated weaning from oral corticosteroids.(77) This is consistent with contemporary guidelines, which recommend that a spacer is prescribed for all patients using high doses of inhaled corticosteroids, regardless of technique.(78) Although the mechanisms underlying the observed increased risk of dental caries in asthma patients remains a subject of debate,(79) it is plausible that reducing the oral impaction of β-agonists with a spacer might represent a similar preventative measure.
Few studies have compared spacers with conventional pMDIs in settings that relate closely to real life. Pedersen assessed PEFR response to terbutaline, inhaled via pMDI alone or with a spacer, in children with exercise-induced asthma.(62) The author's intention was to mimic a day-to-day situation, by recruiting participants from a physical activity program for children with exercise-induced asthma, and administering the bronchodilator if, and when, an attack occurred.(62) Although both treatments resulted in a significantly greater improvement in FEV1, when compared with placebo, spacer use was associated with a significantly greater response in PEFR than the pMDI alone. Pedersen suggested this may have been secondary to fewer procedural errors when a spacer was used, compared with the pMDI alone, an advantage that would be highly relevant in an acute asthma attack.
It has been proposed that spacers afford a “margin of error”(72) in terms of the impact of technique on drug delivery; this may be deemed unnecessary in coordinated patients, particularly if they are administering bronchodilators and are already at the plateau of the dose–response curve. Conversely, international guidelines highlight the clinical value of accessory devices, recommending their use when administering higher doses of inhaled corticosteroids,(78) and more generally in children,(80) particularly those under 5 years of age.(81) In addition to the more commonly prescribed pMDI products, the use of pMDIs to deliver novel drugs remains an area of research. A spacer might be indicated when administering formulations with a high cost, a low therapeutic index, or unwanted local side-effects; there have been previously marketed pMDIs, such as triamcinolone acetonide and flunisolide, that had flavors so unpleasant they were not licensed for sale without a spacer.
Spacers have some clear clinical advantages. Their use reduces the oropharyngeal deposition and local side-effects of inhaled corticosteroids. Spacers have been associated with improved clinical response, although this may be dependent on patients' preexisting level of coordination. While spacers are likely to be of value in patients with dubious or inconsistent inhalation technique, they are considered essential in infants and very young children, who inhale aerosol in tidal breaths.(80,81)
D. Valved Holding Chambers
Despite established advantages, spacers require some degree of hand–lung coordination, and their value in patients breathing tidally is limited. These issues may be overcome with the use of a VHC, a spacer with a low-resistance one-way valve. With inhalation, the valve opens, facilitating coordination and entrainment of the aerosol cloud; a VHC can be said to “capture” the aerosol, and has thus been described as partially “breath-actuated.”(41) Some VHCs also incorporate an exhalation valve, allowing comfortable exhalation and preventing rebreathing. The distinction between a “spacer” and “valved chamber” is ascribed to Dolovich in 1995,(82) although the capacity of a valve to transform a spacer into a “holding chamber to contain the aerosol before inhalation” had been noted over a decade earlier.(83)
The first valved spacer, patented in 1976 (Nebuhaler, Astra Pharmaceuticals), was a large-volume, pear-shaped device, equipped with both an inhalation and exhalation valve.(54) Two years later, Newhouse filed a patent for the first small-volume VHC.(84) The inhalation valve of this device was designed such that inspiratory flow caused the rim of a circular, flexible rubber flap to deflect, allowing air to pass. In later patents, this was replaced with an elastomeric valve, containing a single slit,(85) which can be seen as a precursor to contemporary crosscut valves.
Evidence supporting the value of VHCs
Radiolabeling studies reported that a VHC might improve drug delivery, in comparison with a pMDI alone, or a spacer without a valve.(82) However, the comparison of a small-volume tube spacer, which did not incorporate a valve, with a large-volume, pear-shaped VHC made it impossible to interpret which design feature conferred this benefit.
Several early references actually proposed that turbulence at valves may be detrimental to drug delivery, and suggested that an “adequate” spacer, without a valve, allowed for greater pulmonary deposition.(86) An
VHCs in the pediatric population
In the early 1990s, aerosol delivery to infants and young children received increasing attention. The first infant VHC, a modified standard VHC with attached facemask, was patented in 1986 by Trudell (Aerochamber with mask, Trudell Medical, London, Ontario, Canada).(91) The VHC was equipped with a modified pediatric tracheostomy facemask; the most important feature of the adaptation was considered to be “the presence of a seal attached to the mask to assure a tight fit between the mask and the infant's face.”(92) The device also had a lower dead space, and very low resistance valve, compared with the other available adult VHCs.(91,92) A double-blind clinical trial in young patients (<3 years) with asthma indicated significant improvements in symptoms when the modified VHC was used to deliver 100 μg of albuterol, compared with a placebo.(92) Later reports indicated that other infant-specific spacers were effective and well-tolerated,(93,94) although few studies validated their use against other methods. In 1992, simulated infant/child breathing patterns were used to model aerosol delivery in the pediatric population.(25) Whereas previous studies had suggested that standard valves opened and closed with low tidal volumes, this work helped establish the importance of light valves in the context of the very low tidal volumes observed in infants. A limitation of some early pediatric VHCs (for example, the Babyhaler, Glaxo Wellcome, Middlesex, UK) was its large dead space,(43) given the tidal volumes of the target population and the inverse relationship between dead space and aerosol delivery per breath.
Current understanding of VHCs vs. spacers
The presence of valves
E. Aerosol Administration Technique
Hand–lung coordination and inhalation delay
The effect of poor coordination, despite the use of a spacer, was first indicated in 1978, when a 5-sec delay between actuation and inhalation was shown to increase within-device deposition.(38) Although there was early evidence that spacers could not fully compensate for poor technique, the belief that accessory devices enabled “delivery of medication, whether or not the discharge of aerosol is synchronized with inhalation” persisted for over a decade.(40)
It is likely that the results of some early work perpetuated this fallacy, with one study suggesting that responses to terbutaline were unaffected by delays of 5 sec.(95) In the same study, a delay of 30 sec was associated with a reduced bronchodilator response, but a delay of this duration was considered to be irrelevant to clinical practice. In contrast, the availability of
The impact of delay on respirable dose raises two issues regarding inhalation delays in “real life.” Firstly, although a 30-sec, or even a 10-sec, delay may sound unnecessary, if a caregiver actuates the pMDI before encouraging a patient to inhale, such delays soon become realistic. Secondly, for patients inhaling in multiple tidal breaths, an inherent delay is introduced.
Barry and O'Callaghan also demonstrated that other forces, in addition to gravitational sedimentation, were responsible for the effect of delay on spacer output; in a 1993 study by O'Callaghan
Taken together, the literature suggests that clinically relevant delays result in a reduction in spacer output of a magnitude likely to significantly impact on the proportion of the nominal respirable dose available to the patient. Work on inhalation delay has played a historical role in highlighting the fact that a spacer cannot entirely mitigate poor coordination, and raises awareness of the impact of static charge on drug delivery.
Electrostatic charge
The issue of static charge first entered the history of spacers serendipitously. O'Callaghan explained (written communication, October 2012) that while he was conducting laboratory-based research at the Royal Children's Hospital, Melbourne, Australia in the late 1980s, he noted that spacer output appeared to be influenced by which of the laboratory's research assistants prepared the device. The disparity persisted, despite repeating the experiments and carefully assessing technique, measurement, and drug assay. Further investigation revealed that whereas one assistant used the new spacer straight out of its container, the other, whose experiments yielded dramatically increased drug output, washed the spacer prior to the experiment. Suspecting that this affected the static charge on the inside of the spacer, further experiments were conducted, after the devices were pretreated with an antistatic fabric spray. Using the spray was similarly associated with substantial increases in spacer output, leading the group to conclude that electrostatic interactions between the spacer and charged aerosol particles reduced the availability of aerosol. In 1991, O'Callaghan patented the first antistatic spacer, which, in its preferred embodiment, involved applying an internal coat of antistatic spray.(99) Alternative, more inventive, proposals included coating the inner walls with “a sticky substance, such as honey or a greasy compound, such as petroleum jelly.”(99) The same group went on to demonstrate that treatment with antistatic spray increased the respirable dose of sodium cromoglycate by 244%, compared with that available from an untreated spacer.(98)
Static charge, quantified by direct measurement of field strength, was shown to correlate inversely with aerosol delivery; new spacers, compared with used spacers, had a higher charge and a lower respirable output. The authors suggested wiping spacers with an antistatic cloth prior to use, the earliest practical recommendation for minimizing static.(100)
The first spacer made from a conducting material, stainless steel, which abrogated the need for antistatic treatment, was patented in 1994 (Nebuchamber, Astra Pharmaceuticals).(101) Compared with conventional plastic “static” spacers, a higher filter dose was delivered via the steel antistatic device; the steel antistatic device also performed favorably in terms of dose consistency and respirable fraction.(102) The half-life for passive fallout was up to twice as long as that observed with conventional static spacers, supporting the earlier concept that, in the absence of static charge, inhalation delays should be less problematic.(98)
Wildhaber
Although the steel Nebuchamber eliminated static charge, it was opaque, precluding visual feedback from actuation and inhalation. Transparent antistatic spacers manufactured from charge-dissipative polymers were later designed to facilitate such feedback; the first was patented in 1998(105) and later marketed commercially (Zerostat, Cipla Ltd., Mumbai, India). Several charge-dissipative spacers are now commercially available, reflecting the advantages of a durable, transparent, antistatic spacer.
Static charge has been associated with increased dose variability,(106) although this has not been universally replicated.(107) Dompeling
In summary, static charge on plastic spacers is known to reduce drug delivery
Multiple actuations
The time-consuming nature of inhaled therapies can impact negatively on patients' adherence. To reduce treatment times, patients may actuate several puffs of aerosol into the spacer before inhaling. The first study investigating “multiple actuations” indicated this practice to be inefficient.(83) Four consecutive actuations into the spacer, followed by an immediate inhalation, produced a lung deposition profile more favorable than that achieved with a pMDI alone. However, the practice was associated with higher within-spacer retention and a small, but significant, reduction in whole-lung deposition, compared with a single actuation via the spacer. Based on this work, it was recommended that patients administer single, rather than multiple, actuations when using a spacer.(83)
O'Callaghan
Optimal inhalation technique
In contrast to pMDI technique, relatively little effort has been directed toward the optimization of inhalation via a pMDI with spacer. A 1982 study showed that a slow inhalation (30 L/min), followed by a 10-sec breath-hold, achieved significantly greater lung deposition than either a short breath-hold after slow inhalation or a 10-sec breath-hold after rapid inhalation.(112) Similarly, an
F. Patient–Device Interface
Facemasks were initially used as an interface between spacers and young children in the 1970s,(23) and are now established as necessary components of spacer systems in those patients who are unable to perform the required breathing maneuvers. However, the degree to which a facemask complicates spacer-mediated aerosol delivery was recognized only recently, and remains an area of ongoing research.
Several age-specific factors require special consideration in the very young. Infants are preferential nose-breathers, which can reduce lung delivery by approximately 50%.(116) Young children are unable to understand or maintain a “breath-hold,” although it appears that tidal breathing may result in equivalent clinical responses.(117) Children also tend to hyperventilate into a facemask,(26,118) complicating the interpretation of
Early patient–device interfaces
Early approaches to aerosol delivery in infants included the untimed firing of aerosol onto the buccal mucosa(119) and the use of a polystyrene coffee cup as an extension device.(120) Both methods require actuation directly into the child's face, which is poorly tolerated and associated with poor compliance.(121) With the commercial introduction of VHCs with facemasks in the mid-1980s,(91) the clinical literature reflected an increasing concern for the development of effective and acceptable interfaces. O'Callaghan
Facemask seal and patient tolerance
The need for a good facemask-to-face seal, although not directly investigated, was raised again in 1992(25); however, it was not until 2001 that the first systematic assessment of the relationship between seal and drug delivery was conducted.(122) Delivery via pediatric VHC facemasks and an anesthetic facemask was compared. Larger leaks reduced subsequent
Young patients' intolerance to therapy and resultant distress has been posited as the most common cause of a poor seal in real-life settings,(126) supported by the finding that the degree of cooperation is a primary determinant of drug delivery
Novel methods and devices
Several novel aerosol delivery devices have been developed with the intention of improving the acceptability of aerosol therapy to the very young. Such devices include a facemask with incorporated pacifier (SootherMask, InspirX, Inc., Somerset, NJ), designed to promote calm acceptance of aerosol administration.(134,135) The facemask covered only the nose, so that aerosol would be inhaled nasally, while the infant suckled on the pacifier. Although it has been suggested that the pacifier facemask is compatible with the delivery of aerosol via a VHC,(135) so far published clinical studies have investigated use of the facemask only with a nebulizer.(136)
Alternative methods of improving aerosol delivery to young children have focused on improving the design and seal of conventional facemasks. Hydrogel, applied to the facemask rim, has been used in adult patients receiving continuous positive-pressure ventilation, and may have potential for use with spacer facemasks.(27) However, although the gel forms an effective seal, this method would complicate therapy, which might have a negative influence on adherence and compliance. Facemasks with air-cushioned rims also produce a good seal, and are used in anesthetics. Similar designs have been suggested for use with a spacer, although the thin plastics are fragile and not considered practical for repeated use.(27) A promising recent development is soft-seal technology, used in the seal of a contemporary VHC facemask [LiteTouch facemask, Respironics Respiratory Drug Delivery (UK) Ltd., a business of Philips Electronics UK Limited, Chichester, West Sussex, UK]. Intended for use with existing VHCs, the soft-seal technology facemask has achieved favorable results, in terms of a low-pressure seal and leak resistance.(137)
Current understanding of appropriate patient interfaces
Few studies have investigated the optimal age at which to transfer patients from a facemask to the spacer mouthpiece. Given the difficulties with facemask seal and tolerance, the prevailing advice has been to transfer to a mouthpiece “as soon as possible”—in practice, between 3 and 8 years.(138) However, some evidence indicates that the optimization of facemask design might remove this pressure, and potentially improve pulmonary targeting. Early results suggested that, in children aged 3 years or older, lung deposition achieved with a spacer and facemask was comparable to that achieved with the spacer mouthpiece,(139) but the generalizability of these results is limited by the use of a nebulizer, rather than a pMDI.
In summary, a good seal, which is easier to achieve with a calm child, is vital for effective drug delivery via a facemask. Coaching parents about the importance of a seal is likely to be of value, while facemasks that have flexible rim geometry and require minimal pressure should minimize distress and leakage.
G. Adherence and Compliance
Adherence, compliance, and contrivance
Guidelines recommend the use of spacers in order to ameliorate poor coordination, deliver aerosols to pediatric populations, and administer inhaled corticosteroids.(81,141) However, it is well-established that patients struggle to comply with the correct use of inhalation devices, arguably related to the complex and involved nature of administering aerosols, as opposed, for example, to swallowing a pill.(142) Consequently, “spacer disuse” is a common occurrence in clinical practice.(143) Since the early 1980s, a range of compliance aids have been patented; in a manner characteristic of the general history of spacers, commercial development of these compliance aids has lagged significantly, while experimental support for the efficacy of many such designs is limited.
Patient-related issues resulting in a suboptimal therapeutic response can be described in terms of adherence and compliance. In this context, adherence refers to a patient's tendency to follow a regimen, or use of a device, as prescribed. Compliance, sometimes referred to as competence, concerns the correct use of a treatment or device. It is established that patients who have received instruction in the correct use of inhalation devices, either in person or via video recordings, can demonstrate better compliance than those who received written instruction only.(144) In the general clinical setting, patients are dependent on health-care professionals for such tuition. Since the early 1980s, it has been recognized that health-care professionals themselves are frequently unable to demonstrate correct technique, with either conventional or contemporary respiratory devices,(145,146) a finding that has obvious implications for the quality of tuition available to many patients. Although there are multiple steps in the administration of aerosol via inhalation devices, one of the “most frequent,” and it has been suggested, “single most important” has long been recognized as the ability to coordinate pMDI actuation with inhalation.(147)
“True adherence” [(% adherence to prescribed regimen×% compliance with correct device use)/100] is a measure of a patient's overall device use. Measuring true adherence enables poor adherence to be distinguished from poor compliance,(148,149) introducing an opportunity for targeted interventions. A further issue is contrivance, which describes the purposeful or knowing misuse or disuse of a device or treatment, and complicates both adherence and compliance. Contrivance is recognized as a particular problem in the context of spacers.(143) A full review of the numerous interventions that have been evaluated in the pursuit of improved adherence and compliance with respiratory devices is beyond the scope of the present article.
Compliance aids
Given the difficulties with the regular and correct use of accessory devices, there have been a variety of attempts to incorporate spacer-related compliance aids into the design and manufacture of spacers. Attention initially focused on ensuring adequate inhalation, with the development of collapsible bag spacers that gave a visual indication that the volume of the spacer had been inhaled. An inflatable reservoir bag device, patented in 1982, was the first of its kind.(59) In terms of clinical response, collapsible spacers have been reported as equivalent(70) or even superior to fixed spacers.(150) It has previously been suggested that the correct use of a collapsible spacer may require less skill than a fixed spacer(69); alternatively, positive results may have reflected the provision of immediate feedback, or the opportunity for “rebreathing” if initial inhalation was inadequate.(69) Spacers with an open upper surface were also patented in 1982,(151) and later developed commercially (Synchroner, Fisons, Loughborough, UK/Aerotube, Sanofi, Paris, France). These devices collapsed for ease of storage, and, given that aerosol became visible to the patient if an uncoordinated inhalation was performed, also provided feedback.(152) More recently, a spacer with a visual incentive device, designed to improve both adherence and compliance, was developed (Funhaler, Infamed, Perth, Australia).(153) Although a pilot study demonstrated significant improvements in adherence, the improvement was not sustained in a subsequent 3-month trial,(153,154) which suggested that the “novelty factor” was accountable for initial results.
Auditory aids signaling various features of correct inhalation have been patented since the mid-1980s. These included a spacer with an indicator, which generated sound throughout inhalation,(91) and a device with a whistle triggered by excessive inhalation flow, designed to encourage a slow inhalation.(155)
Over the last 2 decades, valves have been recruited to the cause of compliance. Early VHCs had solid valves, the deflection of which helped caregivers to count the number of breaths taken by children inhaling tidally from the VHC(114); when soft valves were introduced, observation of valve movement was impaired. In 1988, Trudell patented an inhalation device with facemask that was intended to facilitate assessment of the patient's breathing by observation of a convex “bubble” of plastic; the bubble, which maintained its shape at rest and during exhalation, deflected inwardly when the patient inhaled.(156) A modification of this device, which comprises a silicone flap that deflects upon inhalation, was later developed commercially (Flow-Vu, Trudell Medical International). A 1990 VHC (Optihaler, Healthscan Products Inc., Cedar Grove, NJ) also incorporated a compliance valve, which was intended to limit inspiration until the pMDI had been actuated, encouraging coordination.(157)
Contemporary devices generally use multiple compliance/adherence aids, which can include a design of a convenient size, an adaptor suitable for a range of pMDIs, and a storage function, allowing the VHC to contain the aerosol canister. Toy-like VHCs, in addition to their intentionally child-friendly design, have been developed to incorporate compliance-related features such as a collapsible balloon as a visual indicator of inhalation.(158)
Feedback and adherence
Feedback, particularly that provided by information gathered through electronic monitoring, may represent a means by which to increase spacer adherence. To date, no commercially available spacer incorporates electronic monitoring and feedback. A spacer with an electronic monitor, designed to improve compliance and dose consistency, was patented in 1994, but not developed.(159) Compliance with the key features of treatment could be monitored with the Spacer Data Logger (Medic-Aid Ltd., Bognor Regis, UK), which was designed for use in a research environment.(160) Despite also remaining commercially undeveloped, the Spacer Data Logger, which also provided feedback to patients, was trialed in the domiciliary setting, where it was associated with improved adherence and compliance, and a reduced total treatment time.(161) Similar devices can be incorporated into pMDIs; for example, the MDILog (Medtrac Technologies, Lakewood, CO), an electronic monitor that was evaluated with contemporary VHCs.(162)
The potential value of electronic monitors, and the feedback they provide, is exemplified by a recent systematic review of interventions designed to promote adherence in respiratory patients.(163) The four studies that demonstrated a statistically significant improvement in adherence combined electronic monitoring with the provision of adherence-related feedback.(163) With further technological refinement, a combination of electronic monitoring and adherence-related feedback might offer a means by which to improve spacer use. Such devices might be relatively expensive, but this might be offset by reduced health-care utilization due to exacerbations. Ideally these kind of devices would not only remind and advise the patient or the parents, but also deliver compliance- and adherence-related information to physicians and pharmacists.
The future of compliance; patient-centered interventions
Understanding and addressing patients' and caregivers' issues with therapy is arguably the most effective means by which to improve true adherence. Contrivance, or “spacer disuse,” has been reported as a problem in up to three-quarters of patients, including over 90% of parents of preschool asthma patients, with time constraints and inconvenience being frequently cited reasons.(50) Children's intolerance of, and distress related to, facemask application pressures may be an important barrier to compliance and adherence,(132) especially when administering treatment to a distressed child, which will prolong therapy and might minimize efficacy. Measures specifically addressing the problem of delivering aerosol via a facemask and spacer are likely to be important aids to true adherence in this group; contemporary facemask designs that seek to address both intolerance to facemask application and the generalized inconvenience of spacers may prove to be important adherence aids. Novel devices such as the Funhaler, incorporating incentive toys, proved promising in pilot studies, but, as mentioned above, did not improve long-term adherence.(153,154) In the future, it is likely that efforts to improve true adherence will have to address not only laboratory measures of drug delivery, but patient-identified problems with compliance and adherence.
Conclusion
The evolution of spacers has been a complex process, influenced by scientific, commercial, and clinical insights. Today, guidelines recommend that a spacer or VHC should be prescribed to all children using a pMDI, as well as any patient administering inhaled corticosteroids, and those whose ability to coordinate or perform a breathing maneuver is dubious. Current devices can minimize the effect of issues such as inhalation delay, static charge, and dependence on tidal breathing. However, despite almost half a century of development, the optimization of spacer design is far from complete; between 67% and 81% of patients reportedly contrive to knowingly disuse their spacer,(49,50) a clear indication that accessory devices remain unpopular with, and insufficiently acceptable to patients. Ideally, the spacer should be smaller, readily pocketable, and less obtrusive when being used in order to make spacers more patient-friendly. Future developments must focus particularly on the development of more effective and acceptable interfaces, with reference to breathing technique and true adherence. Electronic monitoring and feedback might aid the latter, although patient-identified issues should provide the principal influence for future development. This is appropriate, given that, from their origins in Susie's question, pMDI accessory devices have evolved to meet the needs of respiratory patients.
Footnotes
Acknowledgments
The authors acknowledge Elna Berg (Emmace Consulting AB, Södra Sandby, Sweden), Sunalene Devadason (School of Paediatric and Child Health, University of Western Australia, Perth, Australia), Myrna Dolovich (McMaster University, Hamilton, Ontario, Canada), Chris O'Callaghan (Institute of Child Health, University College London, London, and Great Ormond Street Hospital, London, UK), Steve Newman (Scientific Consultant, Hunstanton, Norfolk, UK), and Olof Selroos (Semeco AB, Ängelholm, Sweden) for their guidance and technical assistance. The authors also acknowledge Stephen Keen (PS5 Consultants Ltd., Portsmouth, UK) for his editorial assistance. This review was sponsored by Respironics Respiratory Drug Delivery (UK) Ltd.
Author Disclosure Statement
Kurt Nikander was an employee of Philips Respironics, Respiratory Drug Delivery, Parsippany, NJ, USA during drafting and is now an employee of InDevCo AB, Nyköping, Sweden. John Pritchard is an employee of Respironics Respiratory Drug Delivery (UK) Ltd. John Denyer works with PS5 Consultants Ltd., and Clare Nicholls is an employee of PS5 Consultants Ltd., Portsmouth, UK, both of whom were engaged by Philips on this project.
