Abstract
Abstract
Background:
In vitro performance studies of valved holding chamber (VHC)-facemask systems are a cost-effective means of circumventing potentially confounding clinical variables. This article reports results of an in vitro investigation into VHC-facemask performance, using three age-specific soft anatomical model (SAM) faces, under clinically relevant conditions.
Methods:
A potentially standardized method was developed to assess VHC-facemask seal leakage, and evaluate the in vitro delivery efficiency of conventional and antistatic VHC-facemask systems. A custom-built test rig and VHC cradles were used to position the VHC-facemask systems against the SAM faces, with a constant, reproducible force. A standardized simulated pediatric breathing pattern (tidal volume = 155 mL; inhalation:exhalation ratio = 40:60; 25 breaths/min) was utilized. Percent facemask seal leakage, percent delivered dose, and the effect of different numbers of simulated breaths (2 to 8) were investigated.
Results:
Of the VHC-facemask systems tested, the OptiChamber Diamond VHC with LiteTouch facemask (Diamond) system had the lowest percent seal leakage with each SAM face. Percent seal leakage from the other VHC-facemask systems was similar with SAM0 and SAM2 faces; the AeroChamber Plus Z-Stat VHC with ComfortSeal facemask (AC Z-Stat) system had a substantially greater percent seal leakage with the SAM1 face. Regardless of the number of simulated breaths, the Diamond system delivered the greatest mean percent delivered dose, with the lowest coefficient of variation, with each SAM face. Percent delivered dose did not correlate well with seal leakage, particularly for VHC-facemask systems with high seal leakage. The electrostatic properties of the VHCs appeared to influence drug delivery.
Conclusions:
This study describes a potentially standardized method for the evaluation of VHC-facemask systems. Use of this method enabled a comprehensive investigation into the influence of clinically relevant variables, including age-specific facial anatomy, number of simulated breaths, and seal leakage, on the delivery efficiency of several commercially available VHC-facemask systems.
Introduction
V
National guidelines indicate that children under 5 years of age, who are unable to breathe reproducibly through a VHC mouthpiece, require a facemask.(2,3) The facemask serves as an interface between the VHC and the child. Unfortunately, although it is necessary to improve the inhalation of aerosol by young children, use of a facemask introduces additional complexities to the design and assessment of VHCs. In addition to VHC-related factors, such as volume, valve structure, and the material from which the VHC is manufactured (conventional or antistatic), consideration must also be given to aspects of the combined VHC-facemask system, particularly relating to facemask-to-face seal, facemask material, and the associated application force required to achieve a good seal.
A good facemask-to-face seal is a crucial element of VHC-facemask system performance, although it can be difficult to achieve. In the presence of a good seal, aerosol is drawn through the VHC system toward the patient, within the confines of the facemask. In this case, the pressure differential created by the child's inspiration is maximized, resulting in optimal opening of the inhalation valve within the VHC and improving the efficiency of drug delivery.(4)
A poor facemask-to-face seal has been shown to result in lower and more variable delivered dose,(5) and may negate the beneficial effects of reduced static charge on a VHC.(6) The effect of a facemask-to-face seal, or a lack thereof, on drug delivery has been verified in vivo, where facemask leakage was shown to result in a dramatic reduction in the lung deposition of radiolabeled salbutamol.(7) Understanding the consequences of a poor facemask seal on drug delivery has stimulated increasing interest in the design of facemasks with more efficient seals.(8)
Achievement of a good facemask seal with minimal dead volume depends, in part, on application force.(9,10) However, application force is poorly tolerated by young patients, and may contribute to distress during treatments. Distress can result in a crying breathing pattern, in which inhalations become irregular and convulsive, with a high flow rate and low volume. This is associated with poor drug delivery to the lungs and, potentially, a reduction in the motivation of caregivers to administer treatments. Optimization of facemask design must therefore consider the minimum application force with which a good seal can be achieved. The facemask sealing rim, particularly the geometry, the flexibility of its material, and the resultant facemask-to-face “fit,” can influence the achievement of a good seal(8) and the required application pressure. Flexible facemasks with large, inwardly-curled, sealing rim geometries are intended to facilitate seal formation, with a small functional dead volume, and minimum applied force.(8)
Interest in the complexity of VHC-facemask use is a relatively recent phenomenon. This is reflected not only in the relative lack of research in this area, but also in the methodology used for in vitro testing of facemasks. Issues with the latter, particularly in terms of the clinical relevance of existing experimental methodology, place considerable limits on the confidence with which in vitro results can be extrapolated to the clinical setting.
The drug output of VHCs can be sensitive to even subtle changes in breathing pattern. Therefore, it is preferable to utilize a tidal breathing simulator for in vitro tests.(11–13)
Many face models that have been tested with VHC-facemask systems have overlooked the complexities of human facial anatomy, including the nasal bridge, and force-responsive three-dimensional (3D) contours.(14,15) An example of this is seen in an in vitro evaluation of VHC-facemask systems that used a flat metal plate, to which the facemask was sealed.(6) Early face models were intended to be more representative of facial structure, but were manufactured from rigid materials nonresponsive to applied force.(6,16–20) Contemporary face models are manufactured from materials that mimic both the structure and soft tissues of the face.(4,21–23) Such face models should facilitate more clinically relevant in vitro testing of VHC-facemask systems.
Soft anatomical model [SAM; Respironics Respiratory Drug Delivery (UK) Ltd., a business of Philips Electronics UK Limited, Chichester, West Sussex, UK] faces have been designed as age-specific models for in vitro testing of VHC-facemask systems. The SAM faces, fixed on a horizontal test rig, facilitate the simulation of different VHC-facemask heights (vertical positioning on face model) and levels of applied force. The SAM faces have been used to assess the delivery efficiency of different VHC-facemask systems with a range of application forces and seal conditions.(4,23–25)
Interfacial “fit” between the VHC-facemask and the face model is a crucial determinant of the seal achieved. SAM0 is based on an age-specific face, which may not be generalizable to other relevant facial anatomies. Preliminary work using different SAM faces indicated that VHC-facemask delivery efficiency was influenced by a variety of factors, such as application force(4,23) and application angle.(26,27)
We report results of an in vitro investigation into factors influencing VHC-facemask performance, using three SAM faces. Particular consideration was given to: (1) use of appropriate face models; (2) use of simulated tidal breathing that was representative of breathing performance by children of the age that the SAM faces were based on; and (3) standardization of in vitro performance testing.
Materials and Methods
Overall study design and test configuration
Each VHC-facemask system comprised a VHC and a facemask. The facemask (brand and size) used with each VHC was selected based on manufacturers' recommendations, as stated in the product labeling information. The VHC-facemask systems investigated in this study are shown in Figure 1. Because commercially available VHC-facemask systems differ in terms of volume, electrostatic nature, valve design, and facemask flexibility, a representative range of systems was selected. The internal volume of the six selected VHCs varies from the small P Chamber (110 mL) to the large Volumatic (750 mL). The selected VHC-facemask systems included both conventional VHCs, which had no antistatic coating, and newer antistatic VHCs. The latter have an antistatic polymer additive, intended to reduce aerosol deposition on the walls of the VHC and to increase the amount of aerosol available to inhalation.
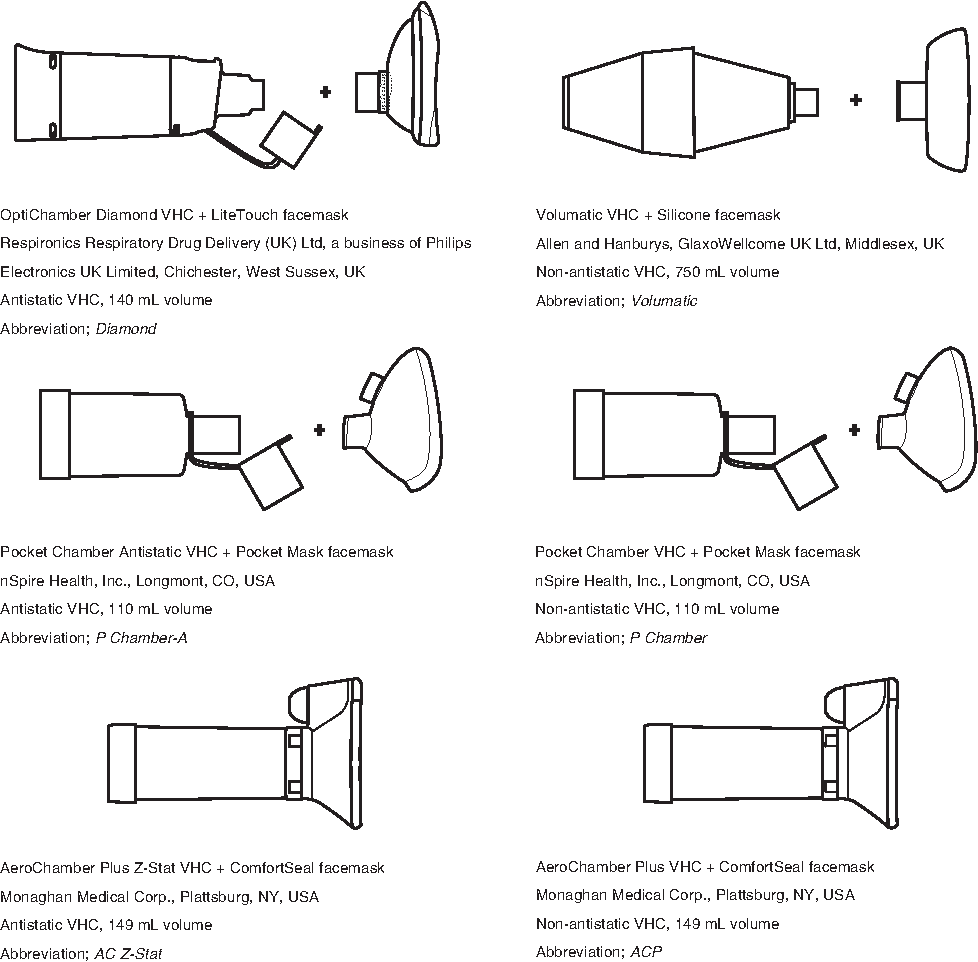
VHC-facemask systems tested.
The study comprised two parts: an assessment of seal leakage, and an evaluation of in vitro drug delivery efficiency using a validated simulated pediatric breathing pattern. The drug delivery efficiency of each VHC-facemask system was quantified after 2, 4, and 8 tidal breaths (volume, 155 mL). Three SAM faces (SAM0, SAM1, and SAM2), which represented the features of 4- and 5-year-old children, were used (Fig. 2). A custom-built horizontal test rig and customized VHC cradles were used throughout the studies (Fig. 3). This setup enabled precise reproducible positioning of the VHC-facemask systems against the SAM faces, and the substitution of VHC-facemask systems. A face model mount enabled adjustment of the relative vertical position and tilting angles between SAM faces and VHC-facemasks. A pulley system was used to deliver a constant force, produced by a 1.9-kg weight, which was applied to the VHC-facemask system, against the SAM face.
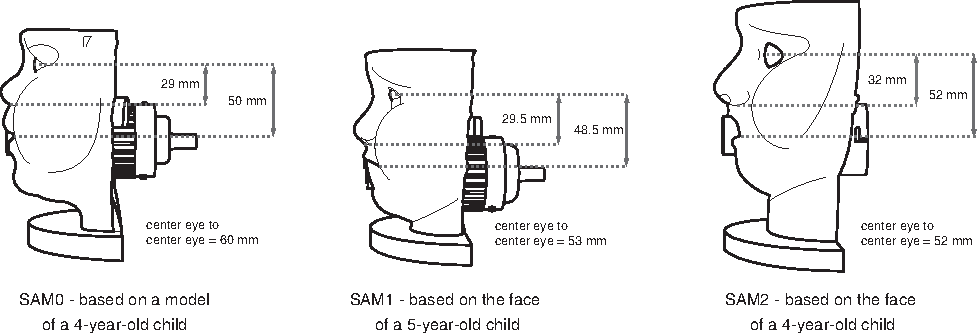
SAM0, SAM1, and SAM2 faces. Key facial dimensions are shown.
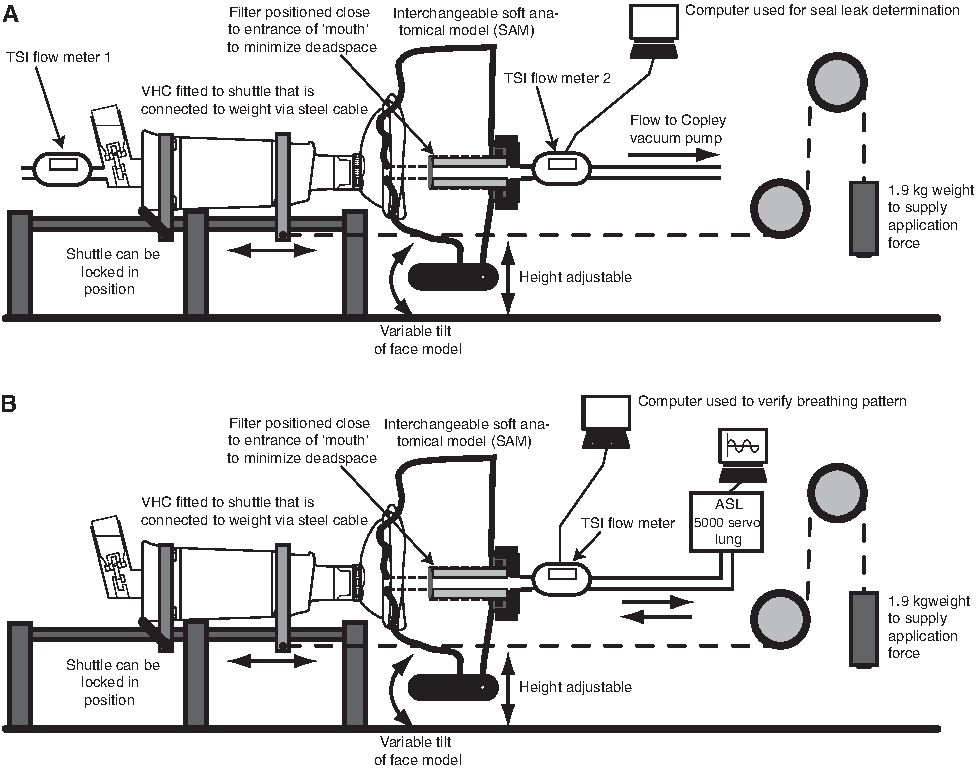
Custom test rig for optimal facemask position (seal leakage;
Development of the SAM faces
The SAM faces were developed with the intention of increasing the clinical relevance of in vitro facemask testing. A custom-made model of a 4-year-old child's face (PA Consulting Group, Melbourn, UK), which had previously been used during in vitro facemask testing,(6,20) was selected for the development of the SAM0 face. The rigid face model was digitized using a high-accuracy portable scanner, and the digitized information was converted into a neutral file format, compatible with the computer-aided design environment. The output from the neutral file was enclosed in a mold base and its volume inverted; splitting the mold base revealed a core side and a cavity side of the face. The cavity side, with anatomical detail, was left untouched; the core side was substituted with a support structure, designed to simulate facial bone structure. Particular attention was paid to the portion of the face below the forehead. The cavity side of the mold and bone structure was “printed” in a 3D prototyping machine (Dimension BST 768, STRATASYS, Eden Prairie, MN) using an acrylonitrile butadiene styrene material. The surface texture of the printed facial cavity was refined by sanding.
SkinRite (EnvironMolds, Summit, NJ), a two-part 10 durometer silicone material, was used to mold the face. SkinRite is used as a casting material for prosthetic skins, and provides excellent elongation properties, mimicking the elasticity of skin.
An airtight metal plunger system, within the support structure, was used to position a precut, 38-mm diameter, modified inhaled mass filter inside the “mouth” of the SAM face. The location of the inhaled mass filter was chosen based on existing work, which indicates that, during in vitro assessment of the inhaled mass of an aerosol delivery system with a facemask, the inhaled mass filter should be placed inside the airway.(20)
For the development of additional representative age-specific SAM faces, a portable floor-mounted high-accuracy 3D camera was used to digitize the faces of children of different ages and ethnicities. Written consent was obtained from the parents of the children scanned. A scan of a 5-year-old Caucasian boy and a scan of a 4-year-old Caucasian boy were selected for the development of SAM1 and SAM2 faces, respectively. Movement of the children during the scan resulted in noise, which was digitally removed before the molds were reverse-engineered. The airtight plunger system was incorporated in a method similar to that utilized in the construction of the SAM0 face.
Validation of simulated pediatric breathing
A flow meter (TSI, Inc., Shoreview, MN) was calibrated using a USB data acquisition board (Measurement Computing, USB-1208 LS, Norton, MA). The flow meter was connected upstream of the SAM face, which was in turn connected to a breathing simulator (ASL 5000 Servo Lung, IngMar Medical, Pittsburgh, PA).
The breathing simulator was used to simulate a pediatric tidal breathing pattern [tidal (inspiration) volume, 155 mL; inhalation:exhalation ratio, 40:60; flow rate, 25 breaths/min]. Parameters, including trachea resistance, breathing rate, and muscle pressure, were adjusted and verified based on the output curve obtained from the TSI flow meter. Flow rate data were transferred to a LabView program (National Instruments, Austin, TX), which calculated inhalation volume using the area under the curve of the inspiration peak.
SAM face leakage
To test leakage from the SAM face alone (from the “mouth” entry to the back end of the filter, without a facemask fitted), each SAM face was first installed individually onto the horizontal test rig. A precut filter (3M Company, St. Paul, MN) was placed in the “mouth” of the SAM face. Leakage was determined using two calibrated flow meters (TSI Inc.), positioned upstream and downstream of the SAM face. A constant airflow (15 L/min and 30 L/min) was applied to the flow meter downstream of the SAM face using a vacuum pump (Copley Scientific, Shoreview, MN). The difference (ΔFlow, L/min) in airflow rate was recorded, and the percent leakage (Equations 1a,b) was used to determine the leakage across the SAM face:
VHC-facemask system seal leakage
Seal leakage tests were performed to determine the position of lowest seal leakage (“optimal” position) for each combination of VHC-facemask system and SAM face.(4) Each combination of VHC-facemask system and SAM face was installed individually onto the horizontal test rig, such that the facemask visually fitted the SAM face (across the bridge of the nose). Optimal relative height for VHC-facemask against the SAM face was determined by altering the vertical position of the SAM face by 1-mm increments. A force, delivered via a 1.9-kg weight, was applied horizontally, and provided application pressure to hold the VHC-facemask system against the SAM face.(4) The ΔFlow upstream of the pMDI inlet and downstream of the SAM face was recorded to determine the VHC-facemask leakage. The leakage test was repeated at the optimal height, by tilting the SAM face in the vertical plane (−10°, −5°, +5°, +10°, where “−” signifies head down and “+” signifies head up). This range of facemask tilt has been used in a previous study, with a variety of facemask designs, and was shown to alter facemask seal leakage by an average of 30.23%.(26) The conventional ACP and P Chamber were not tested, given that these VHC-facemask systems have identical geometry to the antistatic AC Z-Stat and P Chamber-A, respectively.
Drug delivery efficiency evaluation
The optimal position, with the lowest seal leakage, for each of the four VHC-facemask systems was used for the subsequent drug delivery efficiency evaluation.
The drug delivery efficiency evaluation was designed to minimize variability, facilitating comparison between the six different VHC-facemask systems and the three SAM faces. The simulated pediatric tidal breathing pattern was controlled and monitored in real time, using a computer (HP Min311 Netbook) with wired router (CISCO Linksys Wireless-N Router, Irvine, CA). An albuterol sulfate suspension pMDI (90 μg ProAir HFA, lot no. PAA69B; Teva Respiratory LLC, Horsham, PA) was used in the tests.
Prior to use, all VHC-facemask systems were immersed in warm soapy water, rinsed with tap water and deionized water, and air-dried. Each pMDI actuator was washed, rinsed in tap water, and rinsed in deionized water, both before first use and after each study. After drying with compressed air, the actuator was replaced, and the pMDI was shaken and primed by actuating once, in accordance with the manufacturer's instructions.
Each SAM face, with a filter positioned inside the “mouth,” was mounted on the horizontal test rig, and the height adjusted to the previously determined “optimal” position. VHC-facemask systems were assembled and placed individually on the cradle, and the breathing simulator was connected to the back of the SAM face. Once the VHC-facemask system was applied to the SAM face with the 1.9-kg weight, the cradle was locked to prevent facemask seal distortion during pMDI actuation. The pMDI canister was removed from the actuator, shaken, reinserted, and actuated. Actuation was timed to coincide with the onset of “inspiration,” after at least 5 breaths had occurred. The breathing simulator cycled for 2 continuous breaths. The cradle lock was then released, and the cradle was pulled away from the SAM face and left for 20 sec.
This procedure was repeated (n=5) to ensure collection of a quantifiable amount of albuterol sulfate. After all test runs, components (pMDI actuator, VHC, facemask, and filter) were collected into individual Ziploc bags for drug deposition analysis.
The test protocol was repeated using cycles of 4 and 8 breaths. All the above procedures were repeated in triplicate (n=3).
Mass balance was checked for each test to ensure it fell within an 80% to 120% range. Delivered dose uniformity of each pMDI was monitored using dose unit sampling apparatus (DUSA; Copley Scientific) at a constant flow rate (28.3 L/min). Emitted dose was defined as the dose of albuterol sulfate deposited in the actuator, filter, and DUSA. To minimize any systematic error caused by dose variation through the life of the pMDI, separate pMDIs were used, such that each test condition was actuated from the same dose number. Albuterol sulfate was rinsed from components using deionized water, and quantified using high-performance liquid chromatography (Hitachi L-7000 series; Tokyo, Japan) with a Spherex 5 μm C18 250×4.6 mm reverse-phase column (Phenomenex, Torrance, CA).
The percent delivered dose was calculated using Equation 2:
Statistical analysis
The percent seal leakage and percent delivered dose were calculated prior to pooling results. Reporting the percent delivered dose, rather than mass deposition, is assumed to normalize variability attributed to the through-life dose output of the pMDI, and highlight variability associated with the different SAM faces and VHC-facemask systems.(28) Values are reported as means±standard deviation (n=3). The percent delivered dose was compared with the number of breaths and seal leakage data (SigmaPlot for Windows, Version 9.01; Systat Software, Inc., San Jose, CA). A three-way analysis of variance (ANOVA; SigmaStat for Windows, Version 3.5; Systat Software, Inc.) was used to examine the main effects and interactions of VHC-facemask system, SAM face, and number of breaths, as they relate to percent delivered dose. All pairwise multiple comparisons were carried out using the Holm-Sidak method; the overall significance level was 0.05. Descriptive statistics were also collected.
Results
SAM face leakage
The mean percent leakage from within the three SAM faces was 0.64% with the 15 L/min flow rate, and 1.56% with the 30 L/min flow rate.
VHC-facemask system seal leakage
In general, optimal positioning with regard to seal leakage was achieved with the SAM face in a vertical position (0° tilt). Exceptions were seen with the Volumatic VHC-facemask system with the SAM1 face, and the AC Z-Stat and Volumatic VHC-facemask systems with the SAM0 face; a −10° tilt achieved optimal positioning when these combinations of VHC-facemask systems and SAM faces were tested. When SAM faces were positioned with a “+” (face up) tilt, leakage increased. For consistency, the optimal positions with 0° face tilting, and the corresponding percent seal leakages, were chosen for the subsequent delivery performance evaluation.
The percent seal leakage results for each VHC-facemask system, at 15 L/min and 30 L/min, with each facemask placed in optimal position on the SAM0, SAM1, and SAM2 faces, are summarized in Figure 4. At both flow rates, and with all SAM faces, the Diamond VHC-facemask system had the lowest percent seal leakage. Figure 4 highlights that, in general, the AC Z-Stat VHC-facemask system was associated with the highest percent facemask seal leakage.
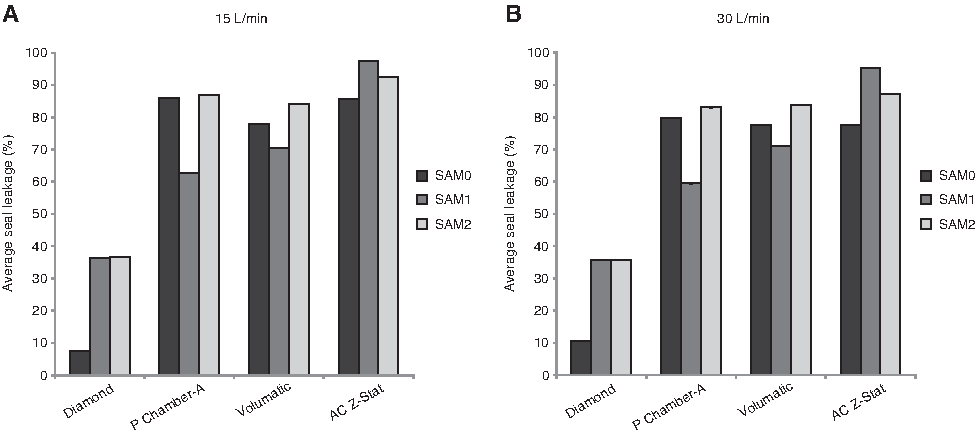
Percent facemask seal leakage for four VHC-facemask systems with SAM0, SAM1, and SAM2 faces:
In vitro evaluation of drug delivery efficiency: standardization and verification
Verification of the simulated pediatric tidal breathing pattern was achieved using a calibrated TSI flow meter. Monitoring the inhalation output from the computer showed that it took approximately 5 breaths after initialization to reach a steady-state breathing pattern. Monitoring indicated that the adjusted parameters were held constant throughout the performance evaluation. Inspiratory volume was observed to drift slightly (±3 mL) when the different VHC-facemask systems were applied. This drift was not considered to have been significant, and likely occurred due to differences in the specific resistance of different VHC-facemask systems, which may modify the feedback loop of the breathing simulator system. DUSA runs (n=51) indicated that emitted dose of albuterol sulfate was in good agreement with the label claim [emitted dose, 537.5±78.9 μg per 5 actuations; expected dose, 540 μg (5×108-μg actuations)].
Drug delivery efficiency: percent delivered dose
For each VHC-facemask system tested, the percent delivered dose with each SAM face, following 2, 4, or 8 simulated pediatric breaths, is shown in Figure 5. In most of the combinations of VHC-facemask systems and SAM faces tested, the percent delivered dose was largely independent of the number of simulated breaths. Results from the P Chamber VHC-facemask system with SAM0 and SAM1 faces were notable exceptions; the percent delivered dose showed a positive association with the number of breaths. Regardless of the number of breaths, the Diamond VHC-facemask system was associated with the greatest mean percent delivered dose, with each SAM face. Drug delivery via the Diamond VHC-facemask system also showed the lowest coefficient of variation, indicating dose consistency that was independent of the number of breaths. Following the Diamond VHC-facemask system, the AC Z-Stat VHC-facemask system, used with SAM1 or SAM2 faces, resulted in a higher mean percent delivered dose, compared with the other VHC-facemask systems tested. The ACP, P Chamber-A, and P Chamber VHC-facemask systems were associated with a low percent delivered dose and a high coefficient of variation. The percent delivered dose from the Volumatic VHC-facemask system was also low, and consistent across the number of breaths extracted.
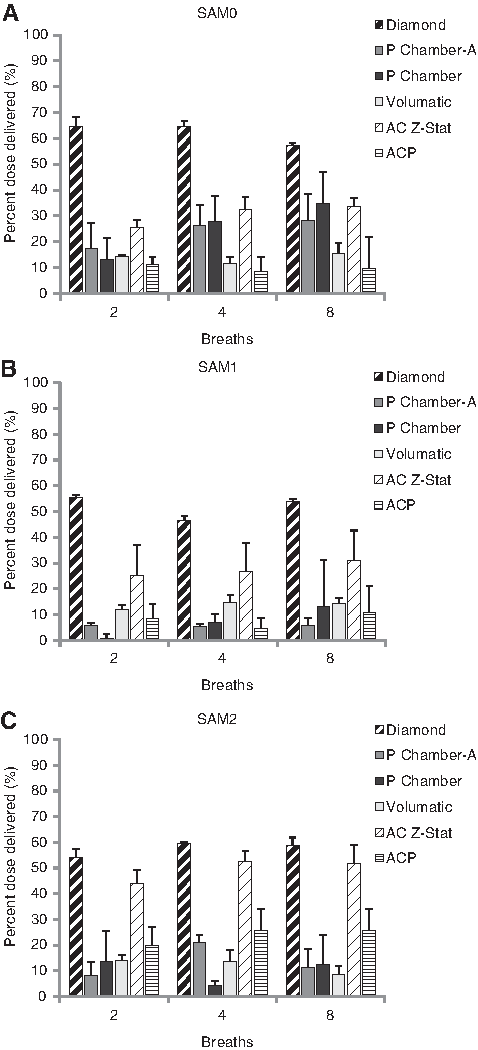
The relationship between delivery efficiency and the number of breaths for the VHC-facemask systems tested with:
Results of the three-way ANOVA indicated that mean percent delivered dose was significantly different between AC Z-Stat and ACP (p<0.001) but not between P Chamber-A and P Chamber (p=0.929) VHC-facemask systems. There was a significant difference in mean delivered dose from the three antistatic VHC-facemask systems (p<0.001). In contrast, mean percent delivered dose from the three nonantistatic VHC-facemask systems did not differ significantly (p>0.05). Comparison of the results from different SAM faces indicated that the pattern of delivery to the SAM1 face differed significantly from that to SAM0 and SAM2 faces (p<0.05); delivery to the SAM0 face did not differ significantly from that to the SAM2 face (p>0.05). No significant differences were observed between the patterns of drug delivery to 2, 4, and 8 breaths (p>0.05).
The relationship between VHC-facemask system seal leakage and delivery efficiency is shown in Figure 6. The Diamond VHC-facemask system had the lowest mean percent seal leakage and the highest percent delivered dose. Notably, in those VHC-facemask systems with high levels of seal leakage, seal leakage did not correlate well with delivery efficiency. With the SAM1 face, for example, percent seal leakage from the P Chamber-A VHC-facemask system was lower than that observed with the AC Z-Stat VHC-facemask system, despite the fact that the P Chamber-A VHC-facemask system had a significantly lower percent delivered dose.
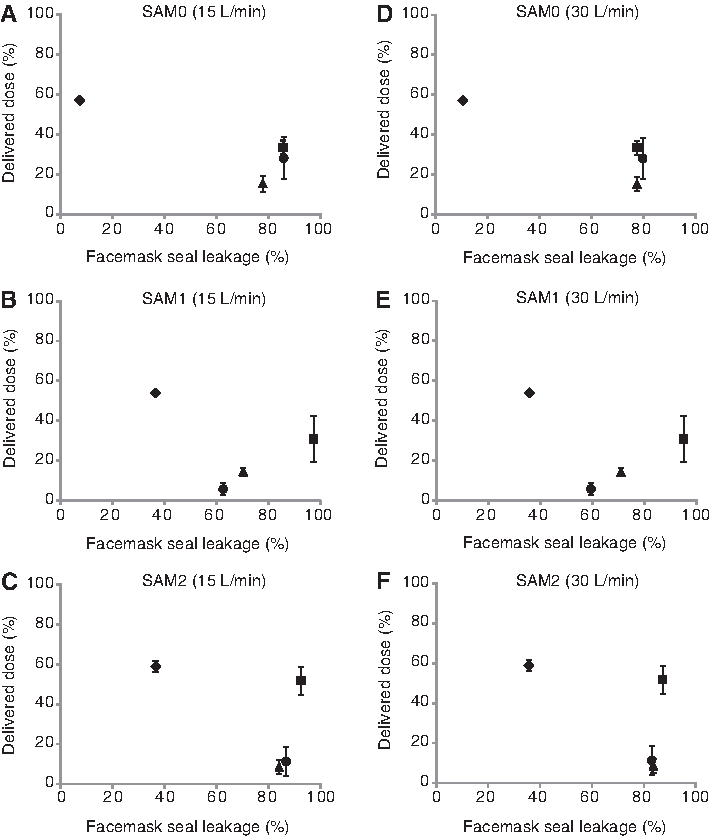
Relationship between facemask seal and drug delivery efficiency for eight tidal breaths with 15 L/min flow (
Discussion
It is well established that seal leakage is a critical determinant of VHC-facemask delivery efficiency.(6,19,29) The present study explored the impact of seal leakage on delivered dose, using six commercially available VHC-facemask systems, three different face models, and a pediatric breathing pattern with 2, 4, and 8 breaths. The standardization of VHC-facemask positioning and application pressure minimized variability from these sources.
The methodology described in the present article was developed alongside the SAM0 face, and has previously been reported in in vitro studies evaluating VHC-facemask system performance.(4,23–25) These earlier in vitro studies were limited by the use of a single face model, whereas the present study utilized two additional SAM faces. Use of different face models should increase confidence in the generalizability of the results, and confirm whether previous positive results obtained using the Diamond VHC-facemask system, which was designed using the SAM0 face, can be extended to different age-specific facial anatomies.
The results showed that VHC-facemask seal depended, to some extent, on the SAM face used; the SAM1 face differed significantly, in terms of percent delivered dose, from SAM0 and SAM2 faces. The percent dose delivered to the SAM0 face did not differ significantly from the SAM2 face. The facial size and contours of pediatric patients vary in an age-dependent manner, such that standardization of age-specific face models may be valuable to the future development and evaluation of VHC-facemask systems.
Minimal VHC-facemask system seal leakage occurred when vertical head tilt was absent (0° tilt), in the majority of cases; the 0° tilt position was therefore used for testing performance delivery. It is plausible that reduced tilt minimizes distortion of the flexible facemask sealing rim, reducing leakage around the nose. This is supported by Esposito-Festen et al., who used a round resuscitation facemask in an obligate nasal breathing infant model, and reported that leakage around the nose influenced lung dose to a greater degree than leakage around the chin.(19) Overall, of the six different VHC-facemask systems tested, the Diamond VHC-facemask system was associated with the lowest percent seal leakage. This difference was particularly apparent when used with the SAM faces modeled on 4-year-old children, compared with the SAM face modeled on the face of a 5-year-old. The lower percent seal leakage associated with use of the Diamond VHC-facemask system may reflect the soft-seal technology of the LiteTouch facemask, which is designed to achieve a good seal with minimal application pressure. Seal leakage tests were not conducted on two of the conventional VHC-facemask systems included in the present study (P Chamber and ACP), as these VHC-facemask systems have identical geometry to their antistatic counterparts (P Chamber-A and AC Z-Stat, respectively), meaning that seal leakage results would be expected to be similar.
Evaluation of drug delivery efficiency indicated that there was a surprising range in the delivered dose, particularly given the careful standardization of the test setup. In general, poor delivery efficiency, such as that observed with the P Chamber-A and P Chamber VHC-facemask systems, corresponded with a high coefficient of variation. Consistent with the results of the seal leakage tests, the Diamond VHC-facemask system had a superior drug delivery performance, compared with the other VHC-facemask systems tested.
Perhaps surprisingly, our results indicated that seal leakage does not correlate well with delivery efficiency when seal leakage is high, implying that, under conditions of poor seal, factors other than seal leakage may have a dominant influence on drug delivery.
Previous studies have documented the influence that breathing pattern, including volume and breathing rate, may have on VHC-facemask system performance.(30) To this end, the present study utilized a carefully validated pediatric tidal breathing pattern, and compared performance efficiency using 2, 4, and 8 tidal breaths. Given that increased tidal breathing might appear necessary to empty a larger-volume VHC, it might be expected that the number of tidal breaths would influence results of drug delivery efficiency, particularly with large-volume VHCs, such as the Volumatic VHC. However, our results revealed that drug delivery efficiency was largely independent of the total number of breaths, at least in the 2 to 8 breaths range, regardless of the degree of seal leakage. This is consistent with a preliminary breath-by-breath study, also from our laboratory, in which percent delivered dose following 1 breath from the Diamond VHC-facemask system was significantly higher than the delivered dose following 8 breaths via the AC Z-Stat and Vortex VHC-facemask systems.(24) This is despite the fact that the chambers of these three VHC-facemask systems are of similar volume. Further increases in number of breaths did not significantly increase the percent delivered dose.
Results also suggest that the electrostatic nature of the VHC within a VHC-facemask system may influence the drug delivery. The AC Z-Stat VHC-facemask system, for example, resulted in a significantly higher percent delivered dose than the ACP VHC-facemask system; these two systems are identical, with the exception of the antistatic nature of the AC Z-Stat VHC-facemask system. This effect was observed despite washing all of the VHC-facemask systems in soapy water, an established method for facilitating charge dissipation. It is possible that static charge may accumulate after pretreatment, perhaps influenced by the removal of detergent by rinsing.(31) Notably, the P Chamber-A and P Chamber VHC-facemask systems are also identical, except that the former is antistatic; there was no significant difference in the percent delivered dose from these systems. Direct measurement of surface charge would be required in order to further investigate the role of electrostatic charge in this phenomenon, and it is uncertain whether the differences observed in vitro would be of clinical significance.
Finally, although the use of SAM faces is intended to improve the clinical relevance of VHC-facemask studies, the existing SAM faces have their limitations. Perhaps the most important limitation concerns the positioning of the inhaled mass filter, which at present collects the total oropharyngeal dose delivered. If data are to be reliably extrapolated to clinical settings, a better understanding of drug delivery to the lungs will be necessary. The current SAM faces are of a limited age range, precluding generalization of results to other patient populations. Also, the children who were used for the modeling of the SAM faces were all Caucasian; if the use of SAM faces were to be extended to the validation and comparison of new VHC-facemask systems, it would be necessary to develop SAM faces across a broader range of ages and ethnic backgrounds.
A remaining challenge for in vitro evaluation is validation of the model against the clinical situation, so that in vitro data provide a good prediction of the results expected in vivo. Despite the advantages of the current methodology, particularly in comparison with existing methods by which VHC-facemask systems have been evaluated, the positioning of the inhaled mass filter is likely to represent a considerable limitation. If an accurate estimation of lung dose is required, evaluation that takes place distal to the oropharyngeal region is necessary. Evaluations that include accurate anatomical modeling of the upper respiratory tract, in addition to the facial profiles, while also taking into account further considerations, such as particle size distribution and drug deposition, are likely to represent an important but challenging next step.
Conclusions
In the present study, three age-specific anatomically accurate SAM faces were used to evaluate factors influencing the delivery efficiency of six commercially available VHC-facemask systems. The study utilized a potentially standardized methodology, which allowed the focus to be placed on the influence of age-specific facial anatomy, the number of simulated tidal breaths, and seal leakage, on delivered dose.
Prior to the performance evaluation, optimal positioning, at which seal leakage was minimized, was assessed using a constant flow rate. Under optimal conditions, seal leakage was lowest from the Diamond VHC-facemask system, and greatest from the Volumatic VHC-facemask system. In general, leakage was minimized when the SAM faces were in a vertical position (0° tilt). The performance efficiency of the Diamond VHC-facemask system was the highest of all the VHC-facemask systems tested.
For those systems with high seal leakage, seal leakage results did not correlate well with drug delivery efficiency. This indicates that factors other than seal leakage alone may be important influences on performance efficiency. Our results suggest that such factors might include the susceptibility of the VHC to the accumulation of static charge (dependent on whether the VHC was made from antistatic or conventional materials), and the influence of the VHC inhalation valve. The latter appears to be more important in VHC-facemask systems that have high seal leakage. VHC volume did not appear to be an important factor, and within the range of breath cycles tested, the total number of breaths did not have a significant impact on delivered dose.
This study showed that the performance of VHC-facemask systems is affected by many interrelated factors, making it difficult to isolate a single test variable in an in vitro environment. Consequently, in vitro testing of VHC-facemask systems must utilize the most realistic methods possible, such as anatomical faces and breathing simulators; otherwise critical parameters, such as seal leakage and its effect on overall performance, may not be identified. This study clearly shows how influential seal leakage can be on VHC-facemask system performance, and the critical importance of effective facemask design. In future, this method of testing should be a normal part of the assessment of new VHC-facemask systems.
Footnotes
Acknowledgments
We acknowledge PS5 Consultants Ltd., Portsmouth, UK, for drafting (Clare Nicholls) and editorial assistance (Stephen Keen). The study was sponsored by Philips Respironics, Respiratory Drug Delivery (Parsippany, NJ).
Author Disclosure Statement
Zhen Xu and Wenchi Hsu were postgraduate students at the School of Pharmacy, University of Maryland, (Baltimore, MD). Richard Dalby was the supervisor for Xu and Hsu at the School of Pharmacy, University of Maryland. Dirk von Hollen, Ashwin Viswanath, and Kurt Nikander are employees of Philips Respironics.
