Abstract
Abstract
Background:
Use of a valved holding chamber (VHC) in conjunction with a pressurized metered dose inhaler (pMDI) can reduce issues relating to poor actuation–inhalation coordination and potentially improve the lung deposition of aerosol, compared with use of a pMDI alone. However, the performance of a VHC is influenced by different device-related factors, including the size and shape of the VHC and the material it is manufactured from (conventional versus antistatic). This study aimed to provide an in vitro characterization of an antistatic VHC, the OptiChamber Diamond VHC, comparing the aerodynamic particle size distribution of aerosol delivered via this VHC with results from a second antistatic VHC and a conventional VHC.
Methods:
The pMDI drug formulations (albuterol, suspension; beclomethasone dipropionate, solution) were connected to a Next Generation Impactor, either directly (pMDI alone tests) or via a VHC (VHC tests). The pMDIs were actuated (×10 per product pair) and tested at extraction flow rates of 15 L/min and 30 L/min, without any time delay between actuation and inhalation. Dose delivery using the two pMDI drug formulations was compared, and is presented with reference to key aerodynamic particle size parameters.
Results:
Compared with tests on pMDIs alone, use of a VHC increased the dose of aerosol within the respirable range, particularly at a 15 L/min flow rate. Between-VHC comparisons indicated that the two antistatic VHCs were equivalent. Delivery of albuterol appeared to be influenced by the VHC used, but beclomethasone dipropionate seemed unaffected.
Conclusions:
The two antistatic VHCs were equivalent for both pMDI brands. Aerosol delivered from the antistatic VHCs at 15 L/min had a higher proportion of fine particles compared with the conventional VHC.
Introduction
V
The principle function of a VHC is to provide space for the development of the aerosol plume, deceleration of droplets, evaporation of propellant, and gravimetric deposition of larger, nonrespirable particles. Together, these factors may increase the efficiency of aerosol delivery and reduce the likelihood of local side effects. It has long been recognized that valves confer to a VHC the ability to “hold” aerosol, and thus reduce the need for perfect actuation–inhalation coordination.(6) Previous work using gamma scintigraphy indicated that use of a VHC, as an accessory to a contemporary pMDI, can result in favorable lung deposition, regardless of whether the patient performs a breathing maneuver (slow inhalation followed by breath hold) or inhales in tidal breaths.(3)
Despite the apparently simple holding function, numerous VHC specifications have been shown to influence delivered dose and fine particle characteristics, and must be considered in the design and manufacturing process. Factors known to influence the output from a VHC include valve design,(7,8) the presence of electrostatic charge on the surface of the VHC,(9) and, although it remains controversial, the size and shape of the VHC.(1)
The OptiChamber Diamond VHC [Diamond; Respironics Respiratory Drug Delivery (UK) Ltd., a business of Philips Electronics UK Limited, Chichester, West Sussex, UK) is an antistatic VHC made from acrylonitrile butadiene styrene, with an antistatic polymer additive that facilitates the dissipation of localized electrostatic charge over its surface. VHCs manufactured from such antistatic materials are less prone to the accumulation of static charge and may provide a more consistent dose of drug.(9) The Diamond VHC has a volume of 140 mL and a length of 14.2 cm, and can be disassembled for hand washing. The Diamond VHC has both an inspiratory valve and an expiratory valve. The inspiratory valve is a low-resistance, low dead-space, duck-billed valve, designed to minimize any impediment to air flow, which might otherwise lead to high levels of local aerosol deposition. The exhalation valve is clearly visible, which allows caregivers to view the deflection of the valve; this provides feedback on the patient's breath count and breath holding. Compliance-related feedback is provided via a warning whistle, which signals an excessively high inhalation flow rate. In addition to a stepped mouthpiece, designed to facilitate the transfer of pediatric patients to use of the mouthpiece, the Diamond VHC can be fitted with the LiteTouch facemask [Respironics Respiratory Drug Delivery (UK) Ltd.], suitable for young patients who inhale tidally from the VHC. The LiteTouch facemask incorporates soft-seal technology, which is designed to provide a high-quality seal, at minimal application pressure, with low leakage.
The Diamond VHC is marketed under 510(k) clearance in the United States, having been determined to be “substantially equivalent” to the AeroChamber Plus Z-Stat VHC (AC Z-Stat; Monaghan Medical Corp., Plattsburgh, NY) by the US Food and Drug Association. The Diamond VHC is also CE marked for use in Europe.
In this article, in vitro aerosol characteristics of the antistatic Diamond VHC, a second antistatic VHC, and an older, conventional VHC are compared. The experiments were carried out using two different drug formulations commonly prescribed to respiratory patients, and assessed key delivery parameters that are typically used to define the characteristics of aerosols and aerosol delivery devices. Results are presented with reference to these key parameters. The delivery of a suspension of albuterol and a solution of beclomethasone dipropionate was investigated, in order to ensure that the evaluation compared both solution and suspension drug formulations that are commonly used in the treatment of asthma. In addition, the antistatic performance of the Diamond VHC was confirmed by characterizing the fine particle distribution of aerosol delivered via both an unwashed and a washed Diamond VHC. To ensure the applicability of results to patients who inhale from a VHC in tidal breaths, investigations were carried out at 15 L/min and 30 L/min extraction flow rates. No delay between actuation and the start of inhalation flow was used in the present study.
Materials and Methods
Test products
Two HFA pMDI drug formulations, a suspension of albuterol (albuterol; ProAir 90 μg HFA; Teva Respiratory LLC, Horsham, PA) and a solution of beclomethasone dipropionate (beclomethasone; QVAR 100 μg HFA; Teva Specialty Pharmaceuticals), were evaluated using a Next Generation Impactor (NGI; MSP Corporation, St. Paul, MN).
ProAir is supplied as a pressurized canister, with a red plastic actuator and white dust cap. Each canister weighs 8.5 g and provides 200 actuations (NDC 59310-579-20). On actuation, 120 μg of albuterol is delivered from the canister valve, and 108 μg is delivered from the actuator mouthpiece; this is equivalent to 90 μg of albuterol base.
QVAR is supplied in a pressurized canister, with a dark mauve plastic actuator and a gray dust cap. Each canister weighs 7.3 g and contains 100 actuations (NDC 59310-177-80). On actuation, 100 μg of beclomethasone dipropionate is delivered from the canister valve, and 80 μg is delivered from the actuator mouthpiece. Neither of the pMDIs tested contained a dose counter.
Drug delivery characteristics of albuterol and beclomethasone delivered via the pMDI alone, the Diamond VHC, the AeroChamber Plus VHC (AC Plus; Monaghan Medical Corp.), and the AC Z-Stat VHC were compared.
Pretest conditioning
All pMDIs, VHCs, test equipment, and fluids were stabilized to ambient conditions for 2 hr prior to use. The pMDI actuator was cleaned before use and before each test run during the study (washed in warm soapy water, rinsed with deionized water, and air-dried). The pMDI was primed by firing a number of shots to waste, as stipulated in the instructions for use, supplied with each pMDI brand.
Prior to use and during the study, the US Pharmacopeia (USP) induction port was washed in warm soapy water, rinsed with deionized water, rinsed with methanol, and left to air-dry. All the VHCs were washed, rinsed, and dried before each test, with the exception of the unwashed Diamond VHCs, which were kept in their original packaging until just before testing.
A mixed anionic/nonionic detergent (15–30% anionic surfactants, 5–15% nonionic surfactants) was used to create the warm soapy water in which all above components were washed (Fairy Liquid, Proctor & Gamble UK, Weybridge, Surrey, UK).
NGI preparation
The NGI was operated as per British Standard EN 13544-1:2007(10) with either a 15 L/min or a 30 L/min air flow through the system. These flow rates were chosen to reflect the range of typical tidal inhalation flow rates observed in patients.(10)
The effect of NGI plate coating on particle bounce was considered; however, given that this analysis concerned differences in VHC performance, any effect of particle bounce was not thought to be critical to the primary objectives of the study. Air flow was generated by a Copley HCP5 vacuum pump (Copley Scientific Ltd., Nottingham, UK). The vacuum pump was connected to a backup filter placed on the exhaust port of the NGI. A TSI mass flowmeter (TSI Inc., Shoreview, MN) was sealed in line between the pMDI actuator and the USP induction port, with the arrow on the bottom of the meter pointing in the direction of the extraction flow. Once the correct extraction flow had been set, the mouthpiece of the pMDI actuator was connected to the USP induction port and sealed with Parafilm M (Bemis Company, Inc., Oshkosh, WI).
Test plan
The two pMDI drug formulations were assessed at two different flow rates, giving a total of four different pMDI–flow rate combinations. The tests were conducted according to a set schedule, designed to minimize any potential systematic error caused by dose trend throughout the life of the pMDI (Table 1). Each of the four tests used six pMDI canisters, with the exception of albuterol at 15 L/min (three canisters). For each test, six runs were performed per pMDI, with 10 actuations per run. Given that this study was designed to investigate between-VHC variability, 10 actuations per run were chosen, in order to minimize variability conferred by the pMDI. This number of actuations also ensured that the dose collected in each NGI stage reached the lower limit of quantification.
The test schedule for albuterol at 30 L/min involved six runs, to allow for the use of both a washed and an unwashed Diamond VHC. This constrained the test schedule design; the washed Diamond VHC tests were conducted with the same devices as the unwashed, which necessitated that washed Diamond VHC tests followed tests using unwashed Diamond VHCs. All references to the “Diamond VHC” refer to the device in the washed state, unless otherwise specified.
Tests on pMDI alone
Before use, each pMDI was primed according to the instructions given in the relevant prescribing information (ProAir, 3×actuations, shaking well before each actuation; QVAR, 2×actuations). The pMDI mouthpiece was connected to the USP induction port, as described above (NGI preparation); the pMDI canister was removed from the actuator, shaken as required, returned to the actuator, and immediately actuated (held down for 1–2 sec). This was repeated a further nine times with a 20-sec delay between repeats. The extraction flow through the NGI was run for a further 10 sec after the final actuation/20-sec delay period. The USP induction port, NGI stages, and backup filter were eluted with a suitable diluent (albuterol, 10% acetonitrile solution; beclomethasone, 60% acetonitrile solution). A predetermined quantity of diluent was used for each component (USP induction port, 10 mL; backup filter, 25 mL; NGI, 10 mL per stage). The amount of drug in each eluate was quantified using high-performance liquid chromatography (HPLC; see below, HPLC analysis ). The USP induction port and pMDI actuator were cleaned in preparation for the subsequent test.
Tests on VHCs
For the VHC tests, the NGI was assembled with a clean set of stages, and a leak test was performed. The USP induction port was fitted to the NGI inlet, and a backup filter was connected to the exhaust port. The NGI was set up as previously detailed (NGI preparation). The mouthpiece of the VHC was connected to the USP induction port using an elastomeric-lipped ISO connector (Intersurgical Ltd., Wokingham, UK). This connection was sealed with Parafilm M, so as to prevent leakage. The pMDI actuator was fitted to the VHC; the pMDI canister was then removed from the plastic actuator, prepared according to the instructions given in the relevant prescribing information, returned to the actuator, and immediately actuated (held down for 1–2 sec). This was repeated a further nine times with a 20-sec delay between repeats. The extraction flow through the NGI was run for a further 10 sec after the last actuation/20-sec delay period.
The USP induction port, NGI stages, and backup filter were eluted with a suitable diluent (albuterol, 10% acetonitrile solution; beclomethasone, 60% acetonitrile solution). Given that this study focused on ex-actuator dose, drug deposited on the pMDI actuator was not eluted. A predetermined quantity of diluent was used for each component (USP induction port, 10 mL; backup filter, 25 mL; NGI, 10 mL per stage; VHC, 30 mL). The amount of drug in each eluate was quantified using HPLC (see below, HPLC analysis ). The mouthpiece of the VHC was sealed with Parafilm M, and a quantity of diluent was dispensed into the VHC. The back end cap of the VHC was then sealed with Parafilm M and the VHC was shaken, so that all internal surfaces were fully washed. The back end cap was unsealed, and the diluent was transferred to a collection vessel. Eluate obtained was quantified using HPLC. The USP induction port and pMDI actuator were cleaned in preparation for the subsequent test.
HPLC analysis
Following each NGI run, all test components were covered and kept dry, until processed. Individual components were processed separately: each component was washed with diluent, and a sample was placed in the HPLC vial, prior to washing the next component.
The HPLC equipment (Agilent 1100 series; Agilent Technologies, Waldbronn, Germany) was fitted with the appropriate column [albuterol, Genesis Phenyl, 4 μm, 150×4.5 mm, 22°C, 1 mL/min; beclomethasone, Phenomenex Luna C18(2), 3 mm, 150×4.5 mm, 25°C, 1 mL/min]. Bracketing standards were made up and run, and the accuracy of the standards was checked. System suitability was then confirmed using five replicate injections of the bracketing standard to be used throughout the run. In order for the results of the run to be valid, the relative standard deviation of the bracketing standards had to be below 2% throughout the run, and the tailing factor (USP method) had to be no greater than predefined values (albuterol, 2.0; beclomethasone, 1.5).
Statistical analysis
The primary objectives of the statistical analysis, for both pMDI drug formulations, were to compare:
• Results for drug delivery via the VHCs to drug delivery via the pMDIs alone • Aerodynamic particle size distribution of aerosol delivered via the different VHC brands • The effect of the two different flow rates • Any differences between drug delivery via a washed Diamond VHC and an unwashed Diamond VHC • The delivery of albuterol and beclomethasone
Comparisons were made for the following endpoints:
• Delivered dose (DD)=(Throat+Stage 1+Stage 2+…+Stage 7+micro-orifice collector+backup filter) • Fine particle dose ≤5 μm (FPD) • Fine particle dose ≤1 μm (FPD1) • Mass median aerodynamic diameter (MMAD) • Dose recovered from VHC • Fine particle fraction ≤5 μm (FPF)=100×FPD/(DD+dose recovered from VHC)
Note that delivered dose, FPD, and FPD1 were normalized to the label claim for delivered dose (ProAir albuterol, 90 μg; QVAR beclomethasone, 80 μg) in order to achieve a common scale and allow comparison between pMDI drug formulations. FPD and FPD1 were calculated from stage data, using linear interpolation on the original scale between the relevant adjacent stages. MMAD was determined using Copley Inhaler Testing Data Analysis Software (CITDAS V3.10, Copley Scientific Ltd., Nottingham, UK).
The delivery method (pMDI alone, AC Z-Stat VHC, AC Plus VHC, Diamond VHC) was treated as a fixed factor, with a random intercept and a random slope included in the model to adjust for possible within-canister trends. A separate model was used for each combination of key variable and pMDI product. The mean values (least square-means) were estimated, together with the relevant differences. A log transformation of the key variables was used to calculate the relative differences and confidence intervals. In vitro equivalence between “Test” and “Reference” devices can be claimed when the 90% confidence interval for the mean ratio of the endpoint under study is within the acceptance interval of 85–118% (i.e., a maximal difference of 15%).(11)
Results
Delivered dose
The delivered dose for the pMDI alone and each VHC, at 15 L/min and 30 L/min flow rate, is shown in Figure 1. The mean delivered dose of albuterol achieved using the pMDI alone was significantly greater than that achieved with the use of any of the three VHCs (Table 2). This pattern was observed at both 15 L/min and 30 L/min flow rates. The delivered dose of beclomethasone followed a similar pattern with a flow rate of 30 L/min; a significantly lower delivered dose was achieved in the presence of a VHC compared with the pMDI alone. This is a predictable effect of using a pMDI with a VHC.
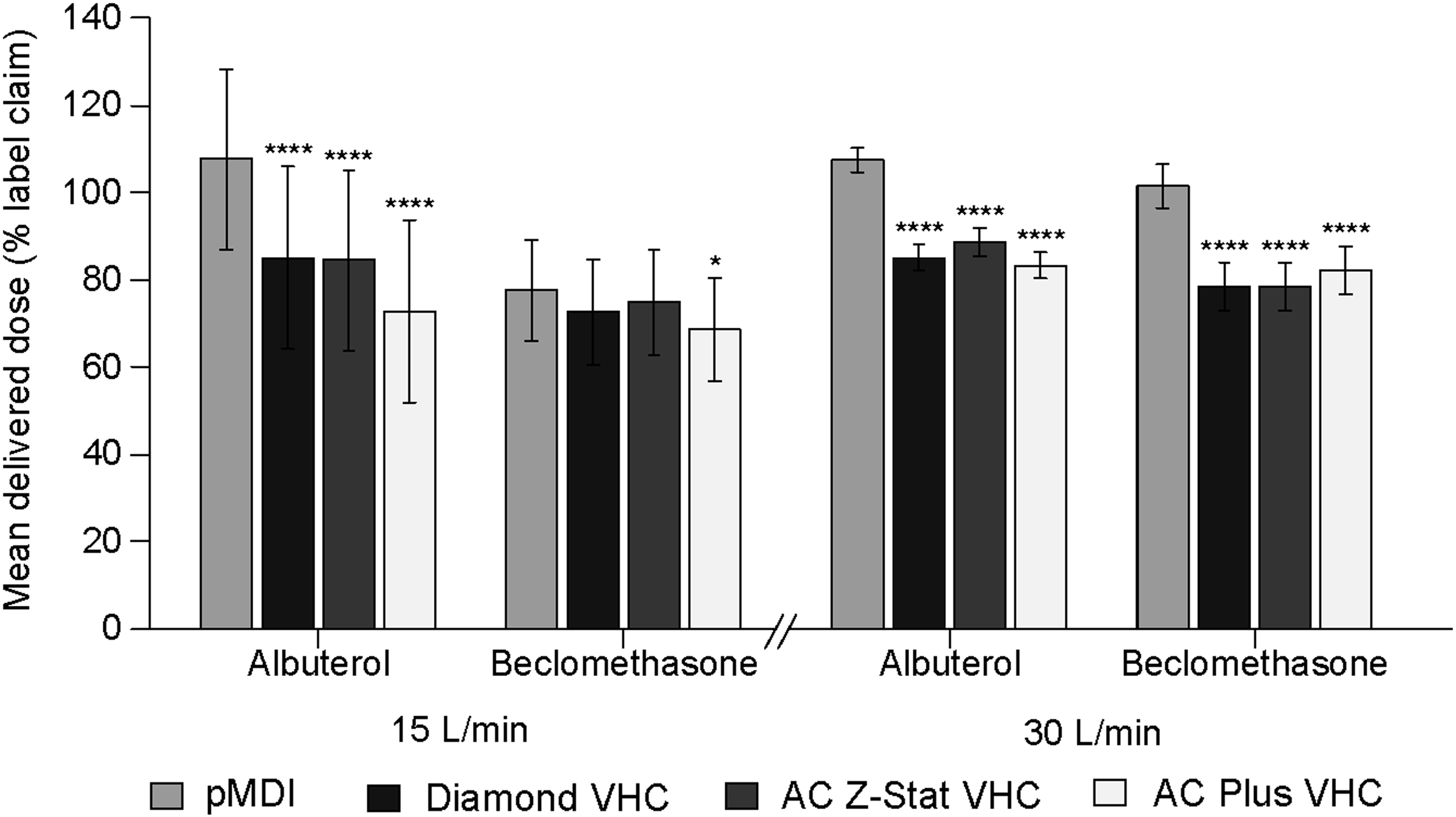
Estimated mean delivered dose as percentage of label claim. Error bars represent 90% confidence intervals about the mean. All comparisons are with pMDI alone; *p<0.05, ****p<0.0001.
% LC, % label claim.
The delivery of beclomethasone with a flow rate of 15 L/min was the exception to this pattern; the delivered dose of beclomethasone achieved with either of the antistatic VHCs did not differ significantly from that achieved with the pMDI alone, whereas use of the AC Plus VHC was associated with a significantly lower delivered dose. Overall, there was a greater variability in delivered dose with a flow rate of 15 L/min compared with 30 L/min.
Fine particle dose ≤5 μm
Drug recovered in particles with an aerodynamic diameter ≤5 μm (FPD), at 15 L/min and 30 L/min, is shown in Figure 2A. With an extraction flow rate of 15 L/min, use of each of the VHCs resulted in a significantly greater FPD of albuterol compared with the pMDI alone (Table 3). A significant increase in the FPD of albuterol was also observed at 30 L/min, although this was restricted to the antistatic VHCs. The FPD of beclomethasone delivered via each brand of VHC at 15 L/min was significantly greater than that achieved via the pMDI alone. In contrast to albuterol, at a 30 L/min flow rate, there was no difference in the FPD of beclomethasone achieved using a VHC compared with that achieved with the pMDI alone. Notably, between-VHC differences for both albuterol and beclomethasone were more pronounced at the lower flow rate.
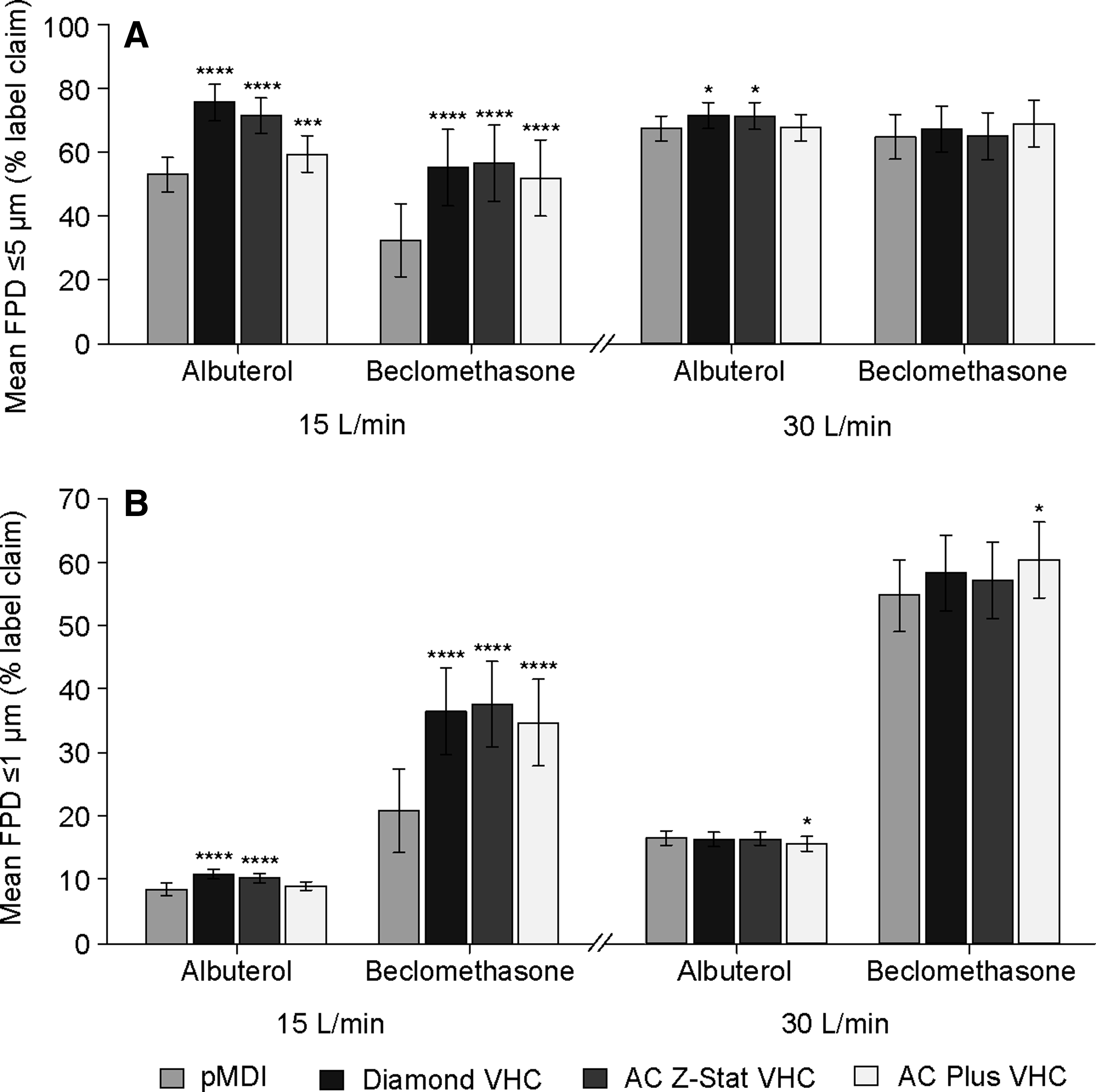
FPD
% LC, % label claim.
Fine particle dose ≤1 μm
The mean FPD1 of albuterol and beclomethasone delivered at 15 L/min and 30 L/min flow rates, relative to the label claim, are shown in Fig. 2B. Mean FPD1 of albuterol delivered at 15 L/min via the Diamond VHC and the AC Z-Stat VHC was significantly greater than that delivered via the pMDI alone (Table 4). There was no significant difference between the FPD1 of albuterol delivered via the AC Plus VHC at 15 L/min and that delivered with the pMDI alone. There was no significant difference in the FPD1 of albuterol delivered via the Diamond VHC and AC Z-Stat VHC at 30 L/min compared with pMDl alone. When the AC Plus VHC was used to deliver albuterol at 30 L/min, the FPD1 was marginally lower; this difference was statistically significant.
% LC, % label claim.
At a flow rate of 15 L/min, a substantially greater FPD1 of beclomethasone was delivered compared with the observed FPD1 for albuterol. The FPD1 of beclomethasone was significantly greater when delivered via any of the VHCs tested compared with that delivered via the pMDI alone. In contrast, at 30 L/min, a statistically significant increase in FPD1 was observed only with the AC Plus VHC compared with the pMDI alone. In general, the FPD1 of both albuterol and beclomethasone increased approximately twofold with a 30 L/min flow compared with the 15 L/min flow, regardless of the VHC used.
Mass median aerodynamic diameter
The MMAD for albuterol and beclomethasone, delivered at both 15 L/min and 30 L/min, is shown in Figure 3. Overall variation in the MMAD was low, regardless of the presence of a VHC or the type of VHC used. MMAD of albuterol delivered via the VHCs at a 15 L/min flow rate did not differ from that delivered via the pMDI alone (Table 5). At 30 L/min, there was a significant increase in MMAD when albuterol was delivered via the Diamond VHC and the AC Z-Stat VHC compared with the pMDI alone. A similar pattern was observed for beclomethasone; with a 15 L/min flow rate, use of a VHC was not associated with a change in MMAD compared with the pMDI alone. When flow rate increased to 30 L/min, the MMAD of beclomethasone delivered via each of the VHCs was fractionally lower than that observed with the pMDI alone; these differences were statistically significant.
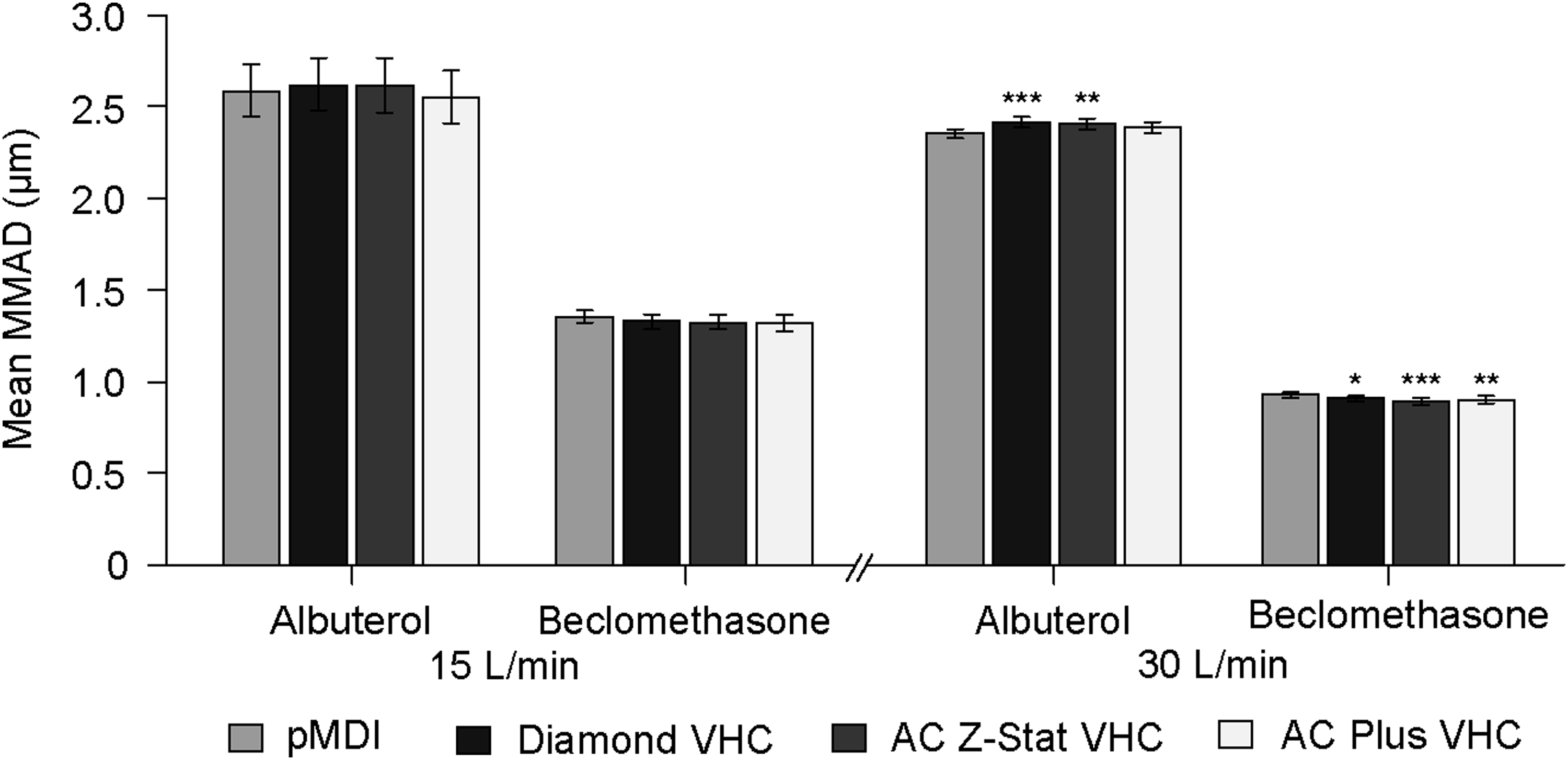
MMAD by pMDI drug formulation, VHC, and flow rate. Error bars represent 90% confidence intervals about the mean. All comparisons are with pMDI alone; *p<0.05, **p<0.01, ***p<0.001.
% LC, % label claim.
Fine particle fraction ≤5 μm
The fine particle fraction ≤5 μm (FPF) for albuterol and beclomethasone, delivered at 15 L/min and 30 L/min flow rate, via each of the VHCs, is shown in Figure 4. At the lower flow rate, use of any of the three VHCs was associated with a significantly greater FPF of albuterol (Table 6). This difference was greater with the antistatic VHCs. At 30 L/min flow rate, only the antistatic Diamond VHC and AC Z-Stat VHC were associated with an increased FPF. For beclomethasone, the increase in FPF at 15 L/min flow rate showed a similar pattern to that observed with albuterol. In contrast to albuterol, at 30 L/min, the AC Plus VHC was associated with a slight but significant increase in FPF relative to the pMDI alone.
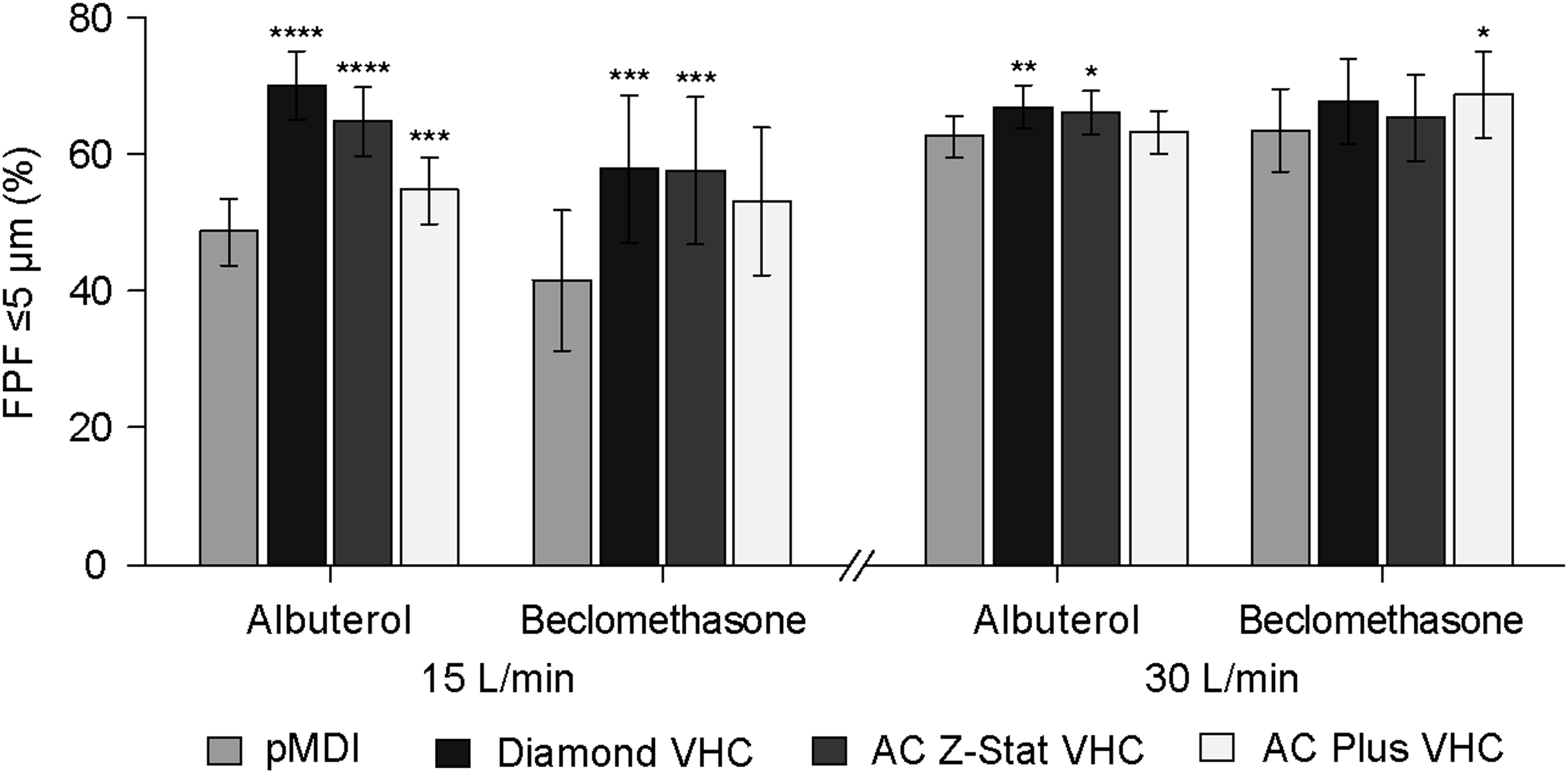
FPF by pMDI drug formulation, VHC, and flow rate. Error bars represent 90% confidence intervals about the mean. All comparisons are with pMDI alone; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
% LC, % label claim.
Mean dose recovered from VHC
The dose of albuterol and beclomethasone recovered from the VHCs following delivery at 15 L/min and 30 L/min is shown in Figure 5. At 15 L/min, a significantly greater dose of albuterol was recovered from the AC Plus VHC compared with either the Diamond VHC or the AC Z-Stat VHC (Table 7). At 30 L/min, a significantly greater dose was recovered from the Diamond VHC compared with the AC Z-Stat VHC. Compared with the AC Plus VHC, a significantly lower dose was recovered from the AC Z-Stat VHC.
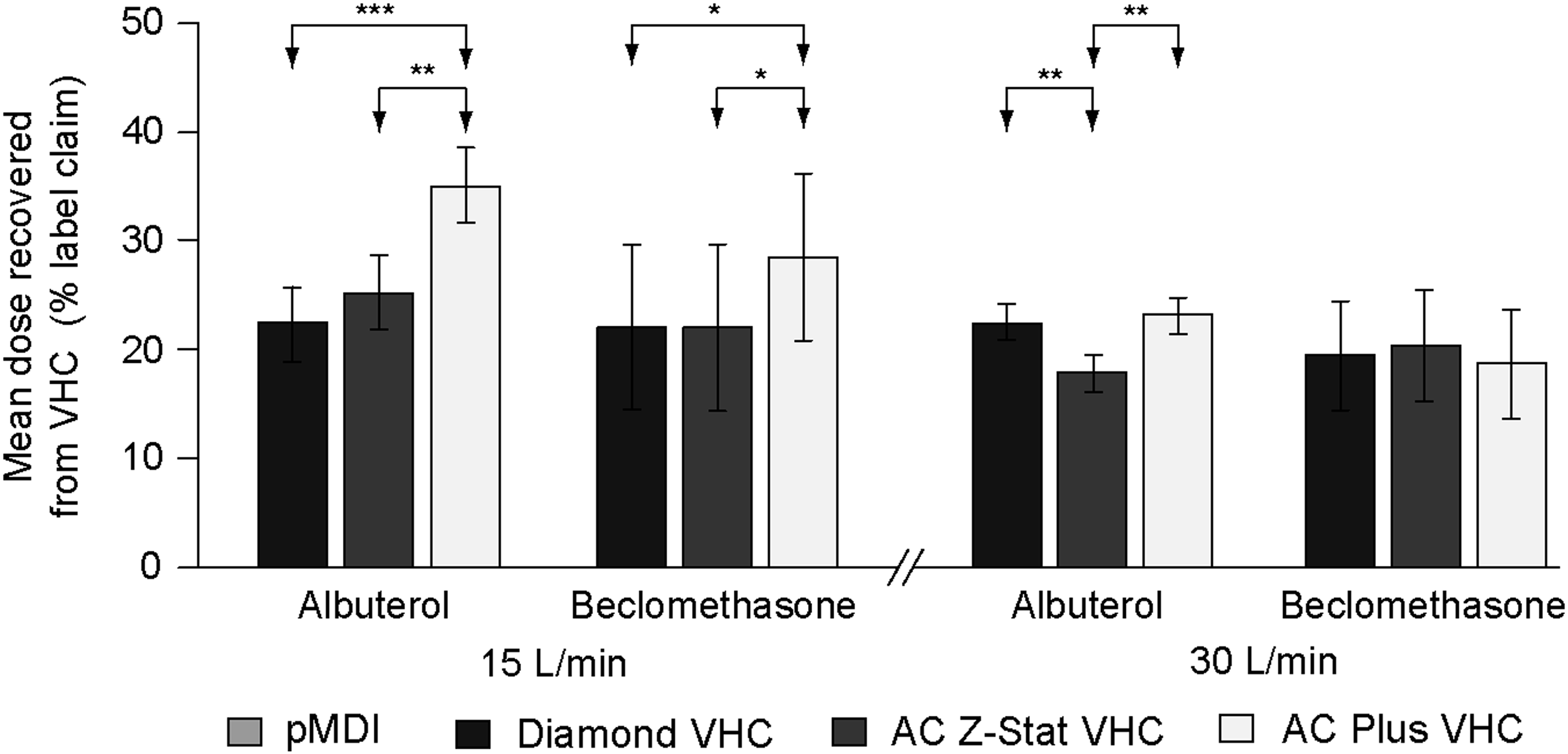
Mean dose recovered from VHC (as percentage of label claim) by pMDI product, VHC, and flow rate. Error bars represent 90% confidence intervals about the mean. Arrows indicate between-VHC comparisons; *p<0.05, **p<0.01, ***p<0.001.
% LC, % label claim.
For beclomethasone, there was no significant difference between within-chamber loss of drug with the AC Z-Stat VHC compared with the Diamond VHC. A significantly greater quantity of drug was recovered from the AC Plus VHC compared with either of the antistatic VHCs. At 30 L/min, the percentage of the label claim of beclomethasone recovered from the VHCs did not differ significantly between VHC brands.
VHC comparisons
Between-VHC comparisons for the key parameters of albuterol and beclomethasone delivery, at 15 L/min and 30 L/min, are summarized in forest plots. The 90% confidence intervals for the ratio between Diamond VHC versus AC Z-Stat VHC (Fig. 6), Diamond VHC versus AC Plus VHC (Fig. 7), and AC Plus VHC versus AC Z-Stat VHC (Fig. 8) are shown; 90% confidence intervals contained within the 85% and 118% bracket indicate in vitro equivalence.(11) At 15 L/min, between-VHC comparisons indicate that a significantly greater delivered dose was achieved with the Diamond VHC compared with the AC Plus VHC (p=0.0025). Use of the AC Plus VHC was also associated with a lower delivered dose compared with the AC Z-Stat VHC (p=0.0036). There was no significant difference in the delivered dose achieved with the Diamond VHC compared with the AC Z-Stat VHC. There were no statistically significant between-VHC differences in the delivered dose of beclomethasone. Only the AC Plus VHC at 30 L/min was associated with a slight, but statistically significant, reduction in the delivered dose of albuterol when compared with the AC Z-Stat VHC (p=0.0057). No other pairwise VHC comparisons differed significantly for the delivery of either albuterol or beclomethasone.
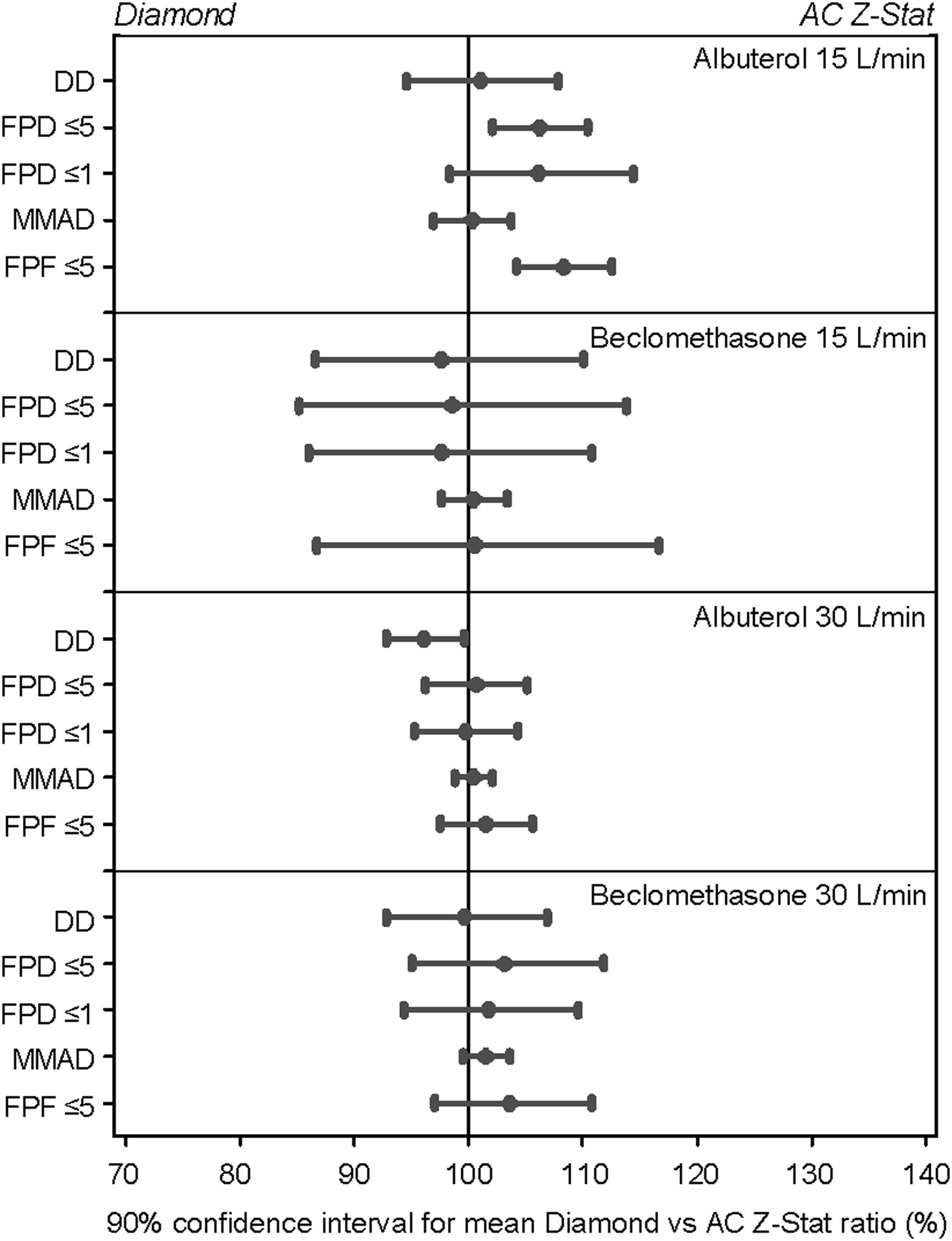
Forest plots of 90% confidence intervals for the relative difference by pMDI drug formulation and key aerosol parameters; Diamond VHC versus AC Z-Stat VHC. DD, delivered dose.
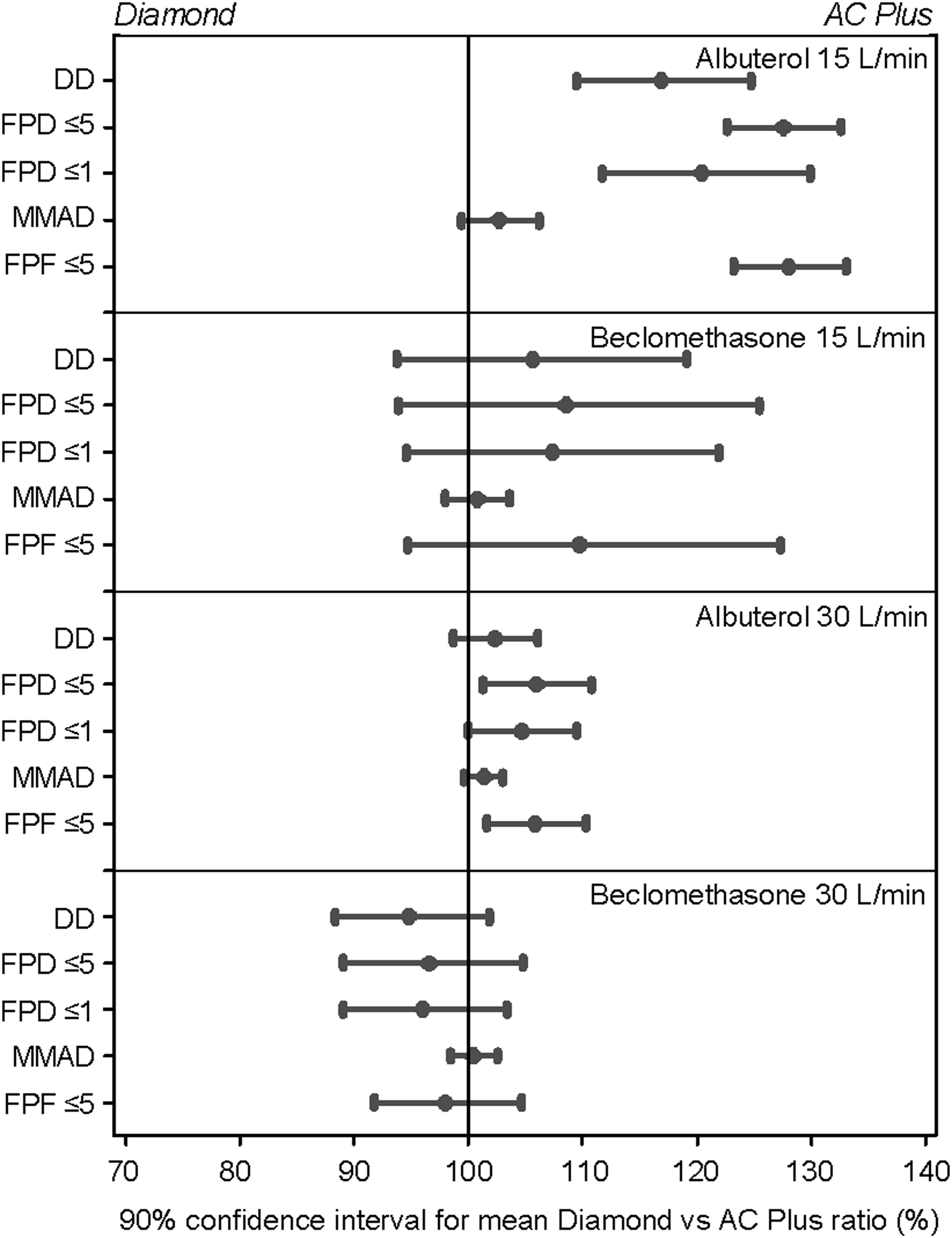
Forest plots of 90% confidence intervals for the relative difference by pMDI drug formulation and key aerosol parameters; Diamond VHC versus AC Plus VHC. DD, delivered dose.
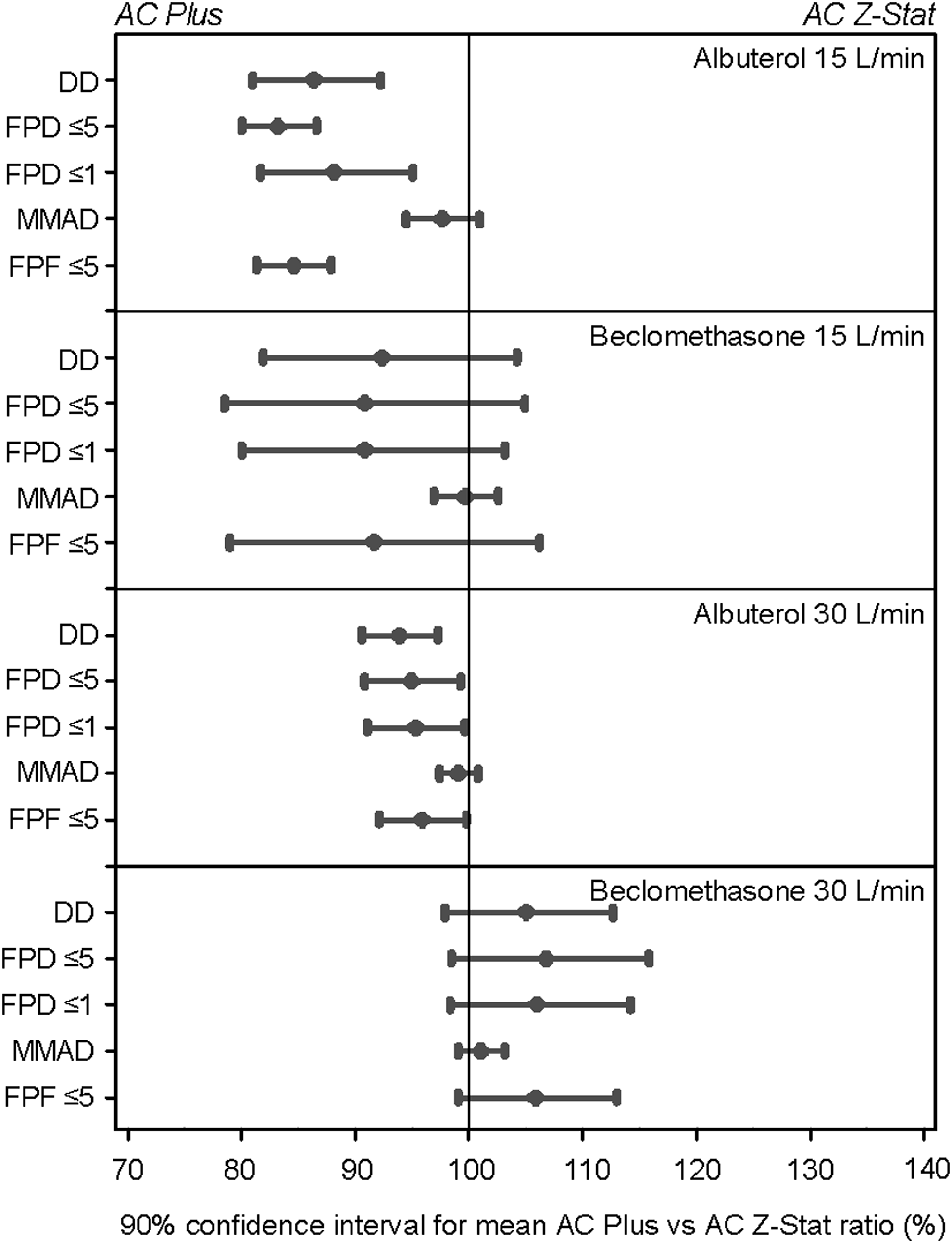
Forest plots of 90% confidence intervals for the relative difference by pMDI drug formulation and key aerosol parameters; AC Plus VHC versus AC Z-Stat VHC. DD, delivered dose.
At 15 L/min, albuterol delivered via the Diamond VHC (p<0.0001) and AC Z-Stat VHC (p<0.0001) had a greater FPD compared with that delivered via the AC Plus VHC. There was a slight, but significant, increase in the FPD of albuterol delivered via the Diamond VHC compared with the AC Z-Stat VHC (p=0.0223). At 30 L/min, there was a trend toward a lower FPD of albuterol when delivered via the AC Plus VHC compared with the AC Z-Stat VHC (p=0.0578). For beclomethasone, no between-VHC differences in FPD were observed at either flow rate.
At 15 L/min, the FPD1 of albuterol delivered via the Diamond VHC did not differ from that delivered via the AC Z-Stat VHC. The FPD1 of drug delivered via the AC Plus VHC was significantly lower than that achieved with either the AC Z-Stat VHC (p=0.0158) or the Diamond VHC (p=0.0024). At 30 L/min, there were no significant between-VHC differences in albuterol FPD1. The FPD1 of beclomethasone did not differ depending on the VHC used, regardless of flow rate. There were no significant between-VHC differences in the MMAD of either albuterol or beclomethasone, at either flow rate.
A greater FPF was achieved when albuterol was delivered via the Diamond VHC and the AC Z-Stat VHC compared with the AC Plus VHC (Diamond VHC, p<0.0001; AC Z-Stat VHC, p<0.0001). There was a slight, but significant, increase in the FPF delivered via the Diamond VHC compared with the AC Z-Stat VHC (p=0.0049). At 30 L/min, there were no significant between-VHC differences in the FPF of albuterol. No between-VHC differences in FPF were observed in the delivery of beclomethasone at 15 L/min or 30 L/min.
Diamond VHC; washed versus unwashed comparisons
All comparisons of the Diamond VHC and the unwashed Diamond VHC were carried out using albuterol, at 30 L/min flow rate. Comparison of aerosol parameters from the Diamond VHC and the unwashed Diamond VHC demonstrated no indication of differences in any of the key parameters of drug delivery (Fig. 9); for all endpoints, the 90% confidence interval values fell well within the 85% to 118% acceptance interval for in vitro equivalence.(11)
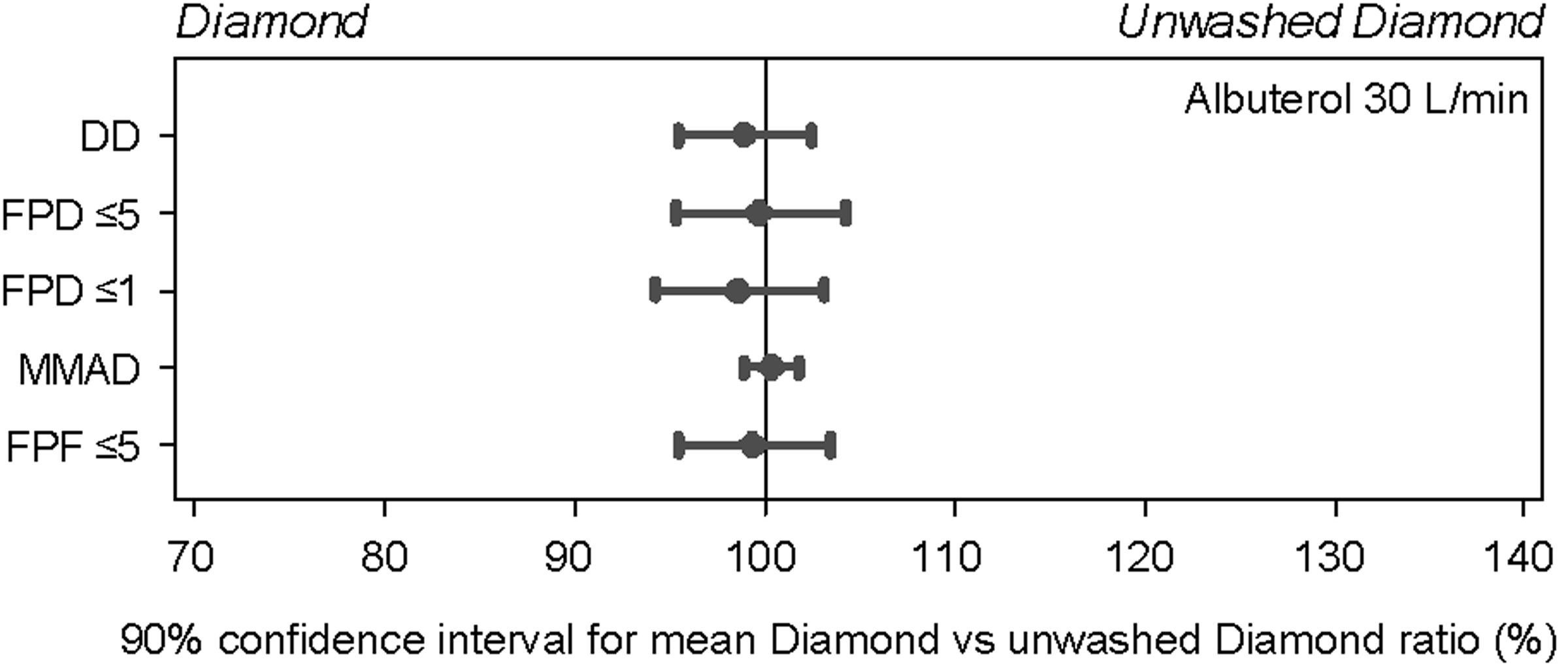
Forest plots of 90% confidence intervals for relative differences between the Diamond VHC versus unwashed Diamond VHC for key aerosol parameters. DD, delivered dose.
Discussion
The aim of this study was to conduct a characterization of aerosol delivery performance from the antistatic Diamond VHC and make comparisons with a second antistatic VHC (AC Z-Stat VHC) and a conventional VHC (AC Plus VHC). Using in vitro test methods, key delivery parameters that are typically used to define the characteristics of aerosols and aerosol delivery devices were assessed. The experiments were carried out at different flow rates using two different pMDI drug formulations that are commonly prescribed to respiratory patients.
pMDI alone versus VHC comparison
The data presented here reflect the expected pattern of a greater delivered dose when the pMDIs were used alone, and a lower delivered dose when the pMDIs were used with a VHC. Of the pMDI–VHC pairs tested here, an equivalent delivered dose of QVAR was achieved via the two antistatic VHCs, compared with the pMDI alone, when the lower flow rate of 15 L/min was used. In contrast, the conventional AC Plus VHC delivered a significantly lower dose (electrostatic effects may have contributed to this difference). This indicates that exceptions to the expected pattern of drug delivery may occur for certain pMDI formulations, under certain flow rates.
The presence of a VHC, compared with the pMDI alone, made little difference to the MMAD. Given the significant differences observed for FPD, FPD1, and FPF, which might be expected to be associated with changes in MMAD, this is perhaps surprising. The similar MMAD, regardless of differences in the FPD and FPF, reflects the reduction in the number of large particles entering the impactor associated with use of a VHC, and the greater throat deposition associated with use of the pMDI alone. The result is that, despite higher deposition in the presence of a VHC, the relative distribution pattern is very similar, and subsequently the MMAD is comparable.
The difference observed was in terms of the quality of aerosol; when a VHC was used, there was a tendency toward the delivery of a greater FPD, and greater FPF, particularly at the 15 L/min flow rate, compared with the pMDIs alone (Figs. 2 and 4). As the use of a VHC was associated with either a reduction in delivered dose or no change in delivered dose, it can be assumed that the amount of drug contained in coarse particles was reduced. Such an effect on aerosol quality is a well-recognized consequence, and one of the main advantages, of using a VHC; the evaporation of propellant from large, nonrespirable aerosol particles, during transport through the VHC, produces smaller particles with a more favorable respirable profile. In addition to potentially improving lung dose, use of a VHC has previously been shown to reduce oropharyngeal impaction of aerosol in an in vitro model, compared with a pMDI alone.(5) This effect was most pronounced at lower (<60 L/min) flow rates, similar to those achieved by patients inhaling tidally from a VHC.
In the present study, each pMDI showed a distinct pattern of deposition, which was maintained across the different VHCs tested. This suggests that, in addition to the delivery method, drug formulation is an important influence on aerosol delivery.
Comparison of VHC brands
In terms of the key aerosol characteristics, performance of the three VHC brands was broadly comparable for the two flow rates and two different pMDI drug formulations studied. Delivery via the two antistatic VHCs (Diamond VHC and AC Z-Stat VHC) was equivalent, with the Diamond VHC delivering a slightly greater FPD and FPF of albuterol at the lower flow rate.
The characteristics of aerosol delivered via the AC Plus VHC differed from those of the aerosol delivered via the antistatic VHCs, with the exception of MMAD, which showed no significant differences between VHC brands. Similar to the effect described for the comparison of pMDI alone with VHC, this is likely to have been influenced by deposition within the NGI throat.
Flow rates
Across the two flow rates, the difference in delivered dose for each pMDI–VHC combination was generally not substantial. At a lower flow rate, the FPD, FPD1, and FPF appeared to be affected to a greater degree compared with the other parameters assessed. This was particularly the case when the drug was delivered via the pMDI alone, or using the conventional VHC, the AC Plus VHC. This is consistent with recent work from our group, which suggested that, when a conventional “static” VHC is used, drug delivery may be more vulnerable to the effect of inhalation delays, particularly at lower flow rates.(12) Notably, previous work comparing the lung deposition achieved by inhaling via a pMDI with VHC using a breathing maneuver and tidal breathing found that some benefit was conferred by undertaking the breathing maneuver.(3) However, the previous study used the conventional AC Plus VHC. It is plausible that the lower flow rates likely to be associated with tidal breathing, compared with a maximal inhalation maneuver, may have less impact on lung dose if an antistatic VHC were used.
The lower flow rate had a comparable influence on output from the two antistatic VHCs. Differences in parameters were less pronounced with the antistatic VHCs than those observed with the pMDI alone or the AC Plus VHC, with the FPD1 being the most affected. Low flow rate generally appeared to influence the delivery of beclomethasone to a greater degree than the delivery of albuterol. This suggests that the solution formulation, which had a much lower MMAD than the suspension formulation, may be particularly sensitive to low inhalation flow. QVAR, the solution of beclomethasone used in the present study, is a unique formulation, with a low MMAD. Previous literature has indicated that, unlike a larger particle chlorofluorocarbon (CFC) beclomethasone formulation, the delivery of QVAR may not be affected by the use of a VHC, with or without an inhalation delay.(13) It is not known whether the use of a steroid formulated as a suspension, with a corresponding larger MMAD, would influence the results of the present study.
Washed versus unwashed Diamond VHC
The results of this study indicated that the performance of the Diamond VHC, when taken out of its original packaging and used for the first time, was not influenced by whether it was in a washed or unwashed state. A newly opened Diamond VHC can therefore be used immediately, without any washing required before initial use.
Limitations of the study
The study test plan was not fully randomized due to a lack of Diamond VHC prototypes, meaning that the tests on the washed Diamond VHC had to be conducted after the unwashed tests. However, as unwashed tests were not conducted on the other VHC brands, this prevented the use of a balanced test matrix.
The interpretation of the two inhalation flow rates deserves further consideration; the “high” flow rate of 30 L/min, which achieved a more positive respirable profile than the lower flow rate, has, in a previous lung deposition study, been classed as a “low” inhalation flow.(14) This would suggest that, in order to adequately approximate likely drug delivery to patients, in whom excessively rapid inhalation flow rates are well-documented, it would be necessary to use a wider range of higher flow rates. It should be noted, however, that the previous study was conducted using old CFC-containing pMDIs; it is likely that contemporary HFA formulations exhibit different properties. Indeed, recent in vitro work suggests that the benefit of VHC use in reducing oropharyngeal deposition becomes less pronounced at higher flow rates, and is canceled out completely once a flow rate of 60 L/min is reached.(5)
Given that patients are known to struggle to comply with correct pMDI and VHC technique in real life, the generalizability of the current results is likely to be limited. It is well recognized, for example, that patients using a pMDI with a VHC will often unwittingly introduce an inhalation delay between actuation of the pMDI and inhalation from the VHC mouthpiece. Such inhalation delays can reduce the respirable drug available for inhalation. Although not explored in the current article, we have recently undertaken an extensive in vitro analysis of the effects of inhalation delays.(12) This analysis, which used a test setup similar to that of the present article, indicated that emitted dose increased with flow rate and decreased with lengthier inhalation delays. The antistatic VHCs tested were shown to have in vitro equivalence, whereas the conventional VHC had a more pronounced flow rate dependence.(12)
Separate from any effect of poor compliance, environmental factors, including extremes of temperature and humidity, are known to affect the performance of solution and suspension HFA pMDIs.(15) The current study was undertaken in ambient laboratory conditions, and therefore results may not be generalizable to other environments.
Finally, it is widely recommended that patients, particularly the very young and those administering high doses of inhaled corticosteroids, use a VHC with facemask. Results of the present study cannot be extrapolated to tests in which a VHC is used with a facemask, rather than the VHC with mouthpiece.
Conclusions
Overall, compared with the pMDI alone, the use of a pMDI with VHC influenced the dose of aerosol within the respirable range, particularly at low flow rates. Between-VHC comparisons showed that the two antistatic VHCs, the Diamond VHC and the AC Z-Stat VHC, achieved in vitro equivalence. Between the VHCs tested, the FPD, FPD1, and FPF were greater for albuterol when the drug was delivered via the antistatic Diamond and AC Z-Stat VHCs compared with the conventional AC Plus VHC. These results indicate that use of an antistatic VHC facilitates the output of aerosol with a better aerodynamic particle size distribution compared with that available from a conventional VHC. This effect is more apparent at lower flow rates and appears to be dependent on the pMDI drug or formulation. Similarly, when the within-VHC effect of different flow rates was compared for albuterol and beclomethasone, the effect appeared to be dependent on both the VHC and the pMDI drug or formulation.
Results also indicated distinct patterns of drug delivery for the two pMDIs tested. Although there were no between-VHC differences in the delivery of beclomethasone, in any of the key aerosol parameters and at either of the two flow rates, the properties of albuterol aerosol were influenced by the material from which the VHC was made.
Footnotes
Acknowledgments
The authors acknowledge Adam Metcalf of Respironics Respiratory Drug Delivery (UK) Ltd., a business of Philips Electronics UK Limited, Chichester, West Sussex, UK, for his analytical assistance, and Kurt Nikander for his technical assistance and guidance. We also acknowledge PS5 Consultants Ltd., Portsmouth, UK, for drafting (Clare Nicholls) and editorial assistance (Stephen Keen). The study was sponsored by Respironics Respiratory Drug Delivery (UK) Ltd.
Author Disclosure Statement
Ross Hatley, Dirk von Hollen, and Lois Slator are employees of Respironics Respiratory Drug Delivery (UK) Ltd. Dennis Sandell is a self-employed statistician and the owner of S5 Consulting. Dennis is an independent consultant who was engaged by Philips on this project.
