Abstract
Abstract
Background:
Aerosolized medications that have been used in infants receiving ventilatory support have not been shown to be effective clinically among the smallest patients. The aim of this study was to characterize the delivery of aerosolized albuterol sulfate in vitro under simulated neonatal ventilatory conditions using a novel ventilator circuit/patient interface connector.
Methods:
A Babylog® ventilator (VN500®; Draeger), a novel ventilator circuit/patient interface (VC) connector (Afectair®; Discovery Laboratories, Inc.), a TwinStar® HME (Draeger) low-volume filter, and either a test lung (Draeger) or lung simulator ASL 5000® (IngmarMed) were used. Intermittent mandatory ventilation conditions were set to replicate the most typical ventilation conditions for premature infants. Continuous positive airway pressure was also used to measure aerosol delivery with active respiratory drive from the patient. Albuterol sulfate (0.5 mg/mL) was loaded into the drug reservoir of a Misty Finity® nebulizer (Airlife®; Cardinal Health) and connected to the ventilator circuit either via a “T” connector as described by the manufacturer [standard of care (SoC)] or via the VC connector. Albuterol extracted from the filters was analyzed using qualified high-performance liquid chromatography. In addition, a laser diffraction spectrometry (Spraytec®; Malvern) and white-light spectrometry (Welas model 2100; Palas GmbH) were used to determine particle size distribution (PSD).
Results:
Compared with SoC, the amount of albuterol delivered using the VC connector was significantly greater (p<0.001) under simulated neonatal ventilatory conditions. Additionally, the PSD profile of albuterol sulfate delivered using the VC connector was more representative of the PSD profile directly from the nebulizer.
Conclusions:
The use of the VC connector increased the delivery of albuterol sulfate and resulted in a PSD profile at the patient interface that is more consistent with the PSD profile of the selected nebulizer when compared with SoC. This VC connector may be a useful, new approach for the delivery of aerosolized medications to neonates requiring positive pressure ventilatory support.
Introduction
A
Materials and Methods
In vitro ventilation system and ventilatory parameters
A ventilator circuit and humidifier were set up according to the manufacturers' instructions. A Babylog® VN500® (Draeger, Luebeck, Germany) neonatal ventilator was used for all scenarios in this study. The ventilator circuit, an RT 225 infant respiratory system (Fisher & Pykel Healthcare, Berkshire, UK), was used with a heater and MR 290 humidifier (Fisher & Pykel Healthcare). Peak inflation pressure (PIP), positive end expiratory pressure (PEEP), inflation time (IT), ventilator rate (VR), tidal volume (Vt), and CPAP were set to replicate typical ventilation conditions for premature infants (Table 1). A Twin Star® HME low-volume filter (Draeger) was placed at the patient interface connection of the connector between the ventilator circuit and either an expandable test lung (Draeger) for simulated ventilation or lung simulator ASL 5000® (IngmarMed, Pittsburgh, PA) for simulated infant on CPAP to measure delivery of aerosolized drug. The flow characteristic of the filter assured low resistance to accommodate the relatively low inspiratory flows generated by premature infants.(11) After the ventilator was turned on, the breathing circuit was tested and ET tube leak was calculated. The ventilator was set on controlled mechanical ventilation (CMV) or CPAP mode. For each test, the humidifier was switched on to mimic clinical conditions.
The active lung simulator is capable of generating simulated spontaneous breathing. It was set at an RR of 40 breaths/min, an inspiratory Vt of 6.0 mL, a muscle pressure of 3.75 cm H2O, a rise time of 17%, a release time of 23%, and an inspiratory hold of 0. These settings have been shown to generate inspiratory flows of approximately 1.5–2 L/min and are representative of the spontaneous breathing of a premature infant.(12)
VC connector
The VC connector (Afectair®; Discovery Laboratories, Inc., Warrington, PA) is a ventilator circuit/patient interface connector that simplifies the delivery of aerosolized medications into ventilator circuits and is positioned at the junction between the inspiratory and expiratory limbs of the ventilator circuit and the patient interface (in place of a wye connector). The VC connector has an internal channel designed to separate the fluidic paths of the therapeutic aerosol and ventilator bias flows (Fig. 1). This design separates aerosol flow from the ventilator bias flow and, therefore, the aerosol is not exposed to the relatively high flow of gas that would decrease the concentration of the inhaled aerosol (Fig. 2). Additionally, the VC connector provides aerosol delivery to the patient under lower and more laminar flow conditions. This should decrease aerosol losses in the artificial airways related to impaction. The internal geometry of the VC connector and the common chamber surrounding the internal channel provides maintenance of PEEP during exhalation.
Cross-sections of the VC connector.
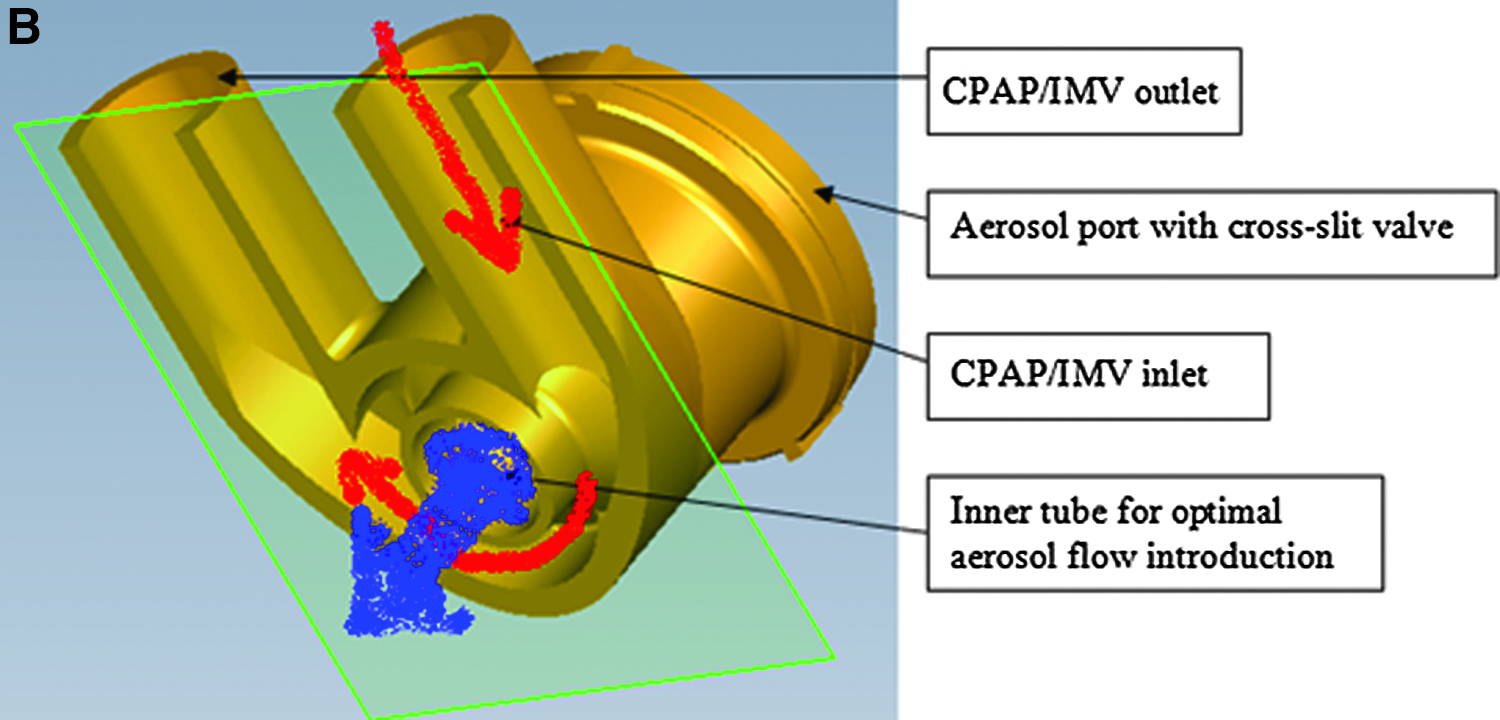
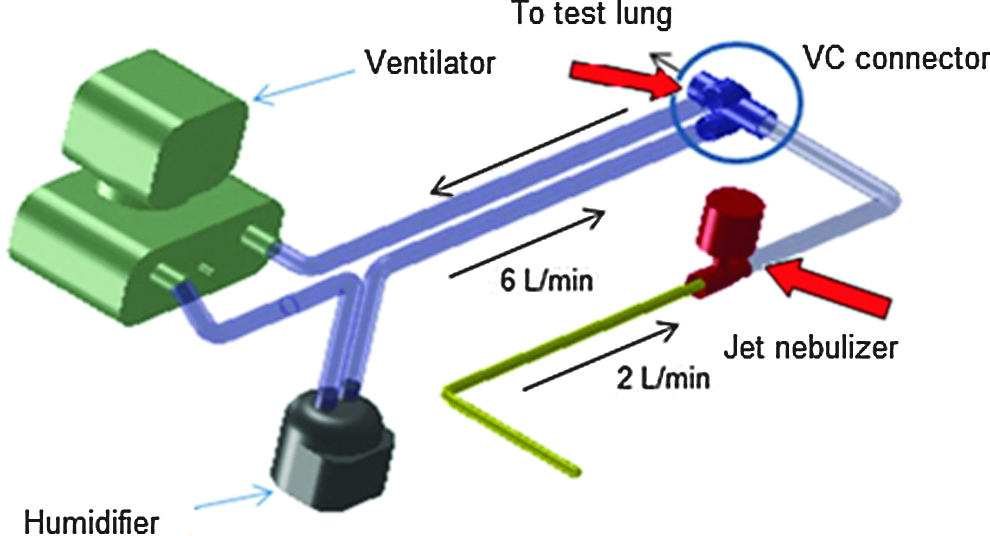
Diagram of the novel aerosol delivery system with VC connector. Arrows indicate measurements of emitted dose (Ed).
Aerosol generation and aerosol delivery
A Misty Finity® jet nebulizer (Airlife®; Cardinal Health, Dublin, OH) was used to aerosolize albuterol sulfate (Ventolin® 1 mg/mL; GlaxoSmithKline, Brentford, UK). To achieve optimal nominal volume and avoid bias caused by residual volume, albuterol was diluted to 0.5 mg/mL in water for injection, and the reservoir chamber was refilled after each run up to 10 mL to avoid any deviation in the output rate of the device. Because the aim of this study was to test this same type of nebulizer with two different delivery systems, we have established these operational criteria and defined nominal dose as the amount of the drug dispensed from the nebulizer during a 5-min sampling time to eliminate any bias derived from the level of drug in the reservoir chamber. For each of the ventilator settings, five nebulizers were tested, and after each scenario and delivery method a fresh reservoir fill of drug was used with each new device. Nebulization was achieved using 2 L/min air flow (as recommended by the manufacturer) with a duration of 5 min for each of the tested conditions. The nominal dose dispensed during 5-min aerosolization was established based on the mass-balance measurement of the nebulizer weight before and after aerosolization. The emitted dose from the nebulizer was measured by placing an HME filter directly adjacent to the output from the nebulizer for each of three runs.
For the SoC tests, the nebulizer was connected to the inspiratory arm of the ventilator circuit as recommended by the manufacturer (Cardinal Health), 20 cm from the wye connector, through the “T” connector and corrugated tube supplied by the manufacturer. For the VC connector tests, the nebulizer was connected to the VC connector through this same corrugated tube as for SoC tests. The VC connector replaced the standard wye connector. The diagram of the novel aerosol delivery system is presented in Figure 2. The aerosol delivered dose under IMV or CPAP conditions was based on the mean value out of five repetitive runs of the same ventilator conditions.
Analytical methods
Analysis of the filters was based on a high-performance liquid chromatography (HPLC) technique. The HPLC method was calibrated with a series of standard solutions of albuterol. The calibration curve was linear with a correlation coefficient of 0.9998 over the range of tested concentrations (0, 6.25, 12.5, 25, 50, 75, 100, 150, and 200 μg/mL). After the completion of each run, the HME filter was placed immediately in a sealed test tube. There were five runs for each tested ventilation setting. Filters were eluted with 5 mL of 0.1 mol/L HCl. Identification of albuterol was based on the retention time and UV spectra. Filter extraction recovery was 92% based on the three samples of 125 μg of albuterol aerosolized directly onto the HME filter. Limit of detection (LOD) was established at 0.28 μg/mL and limit of quantification at 0.84 μg/mL. To verify that 100% efficiency of the aerosol was captured on the filter, two sets of in-line filters were used for the emitted dose study. Over three runs, no drug was collected on the second filter. The data were recorded, and results were presented as mean and standard deviation (SD).
Aerosol characteristics
PSD of the albuterol aerosol for the VC connector tests was measured with a Spraytec® spectrometer (Malvern Instruments, Malvern, UK). The operating principle of the Spraytec is based on the laser light diffraction analysis, and this method has been proposed as a fast and reliable substitution of pharmacopeial impactor techniques that are also used for nebulizers.(13) Laser light diffraction analysis allows for a rapid sampling and in-line analysis of aerosols at high concentrations and determines particles in the very wide size range of 100 nm to 900 μm. The Spraytec spectrometer was operated in the open-bench configuration with the sampling rate of 1 kHz and sampling period of 400 msec. Experimental data were averaged over the sampling period and delivered as the percentiles of the cumulative volume (mass) distribution function: D10, D50, and D90. However, the method fails if aerosol concentration is too low. Hence, the low density of aerosol produced under low ventilator conditions forced the use of another measuring technique in SoC tests.
Spectrometry based on white-light scattering (Welas model 2100; Palas GmbH, Karlsruhe, Germany) was used for in-line analysis of the PSD of the albuterol aerosol for the SoC tests. This spectrometer has been successfully used in other studies related to pharmaceutical aerosols.(14,15) The method is the most suitable for aerosols of low concentrations, and it measures particles in the size range of 300 nm to 18 μm, which is sufficient for determination of medical graded aerosols. The Welas spectrometer was operated at standard conditions (sampling flow up to 5 L/min) with a sampling period of 90 sec. The data were obtained as the number distributions, which were recalculated to cumulative mass distributions. The percentiles D10, D50, and D90 were established by interpolation.
The PSD measurements of the albuterol aerosol were made at the nebulizer, at the wye for SoC tests, and at the VC connector for the VC connector tests, and at the tip of various ET tube sizes under heated and humidified conditions. The humidifier chamber was filled with water, and the heater was switched on at least 10 min before initiation of the experiment. To achieve flow conditions similar to those used when ventilating newborn infants, the expiratory outlet of the wye connector was equipped with a low-resistance flow regulator allowing for direction of 2 L/min flow through the wye and VC connector ET tube port, mimicking the inspiratory flows of a premature infant. All measurements of PSD in each test group were done in triplicates.
The equivalence of the two PSD methods was demonstrated by a comparison bridging study conducted with test silica spheres (Monospher 1000®; Merck, Darmstadt, Germany). The aerosol was produced by mechanical dispersion of the powder into the air by a 2-mL syringe. The aerosol cloud was sampled by the Welas spectrometer for 30 sec to count more than 3,000 particles passing through the optical chamber. The same aerosol cloud was analyzed by the Spraytec spectrometer operated at 1 kHz sampling rate with 500-msec sampling window. The differences in two tested PSD methods in the determination of D10, D50, and D90 values were below 0.15 μm.
Statistical analysis
For comparison of mean emitted doses as well as mean values of particle sizes, a test for homogeneity of variance (Leven test) and T test with Welch correction were used. In the analysis of particle size, the mean values measured at the tip of ET tubes were compared between different delivery systems, as well as with the mean values measured just at the output from the nebulizer. Statistical calculations were done with Statistica software v.10 (Statsoft, Inc., Tulsa, OK). Statistical analysis was performed only if at least four out of five samples exceeded LOD. A post hoc multiple comparison analysis was done for the high ventilator setting in the SoC group. As the drug was detected only on two filters, an additional five filters were analyzed, and all seven samples were included in the statistical analysis.
Results
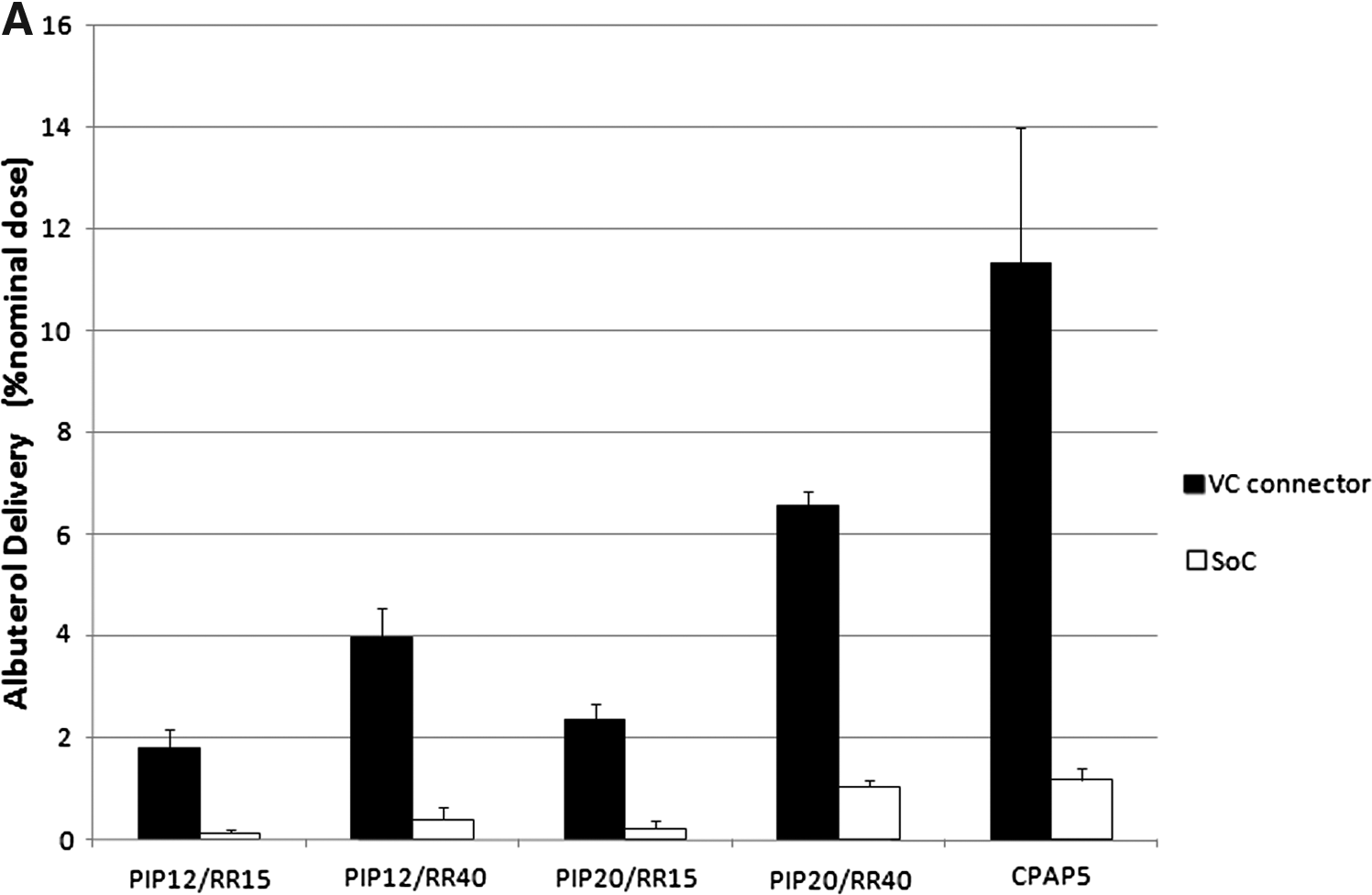
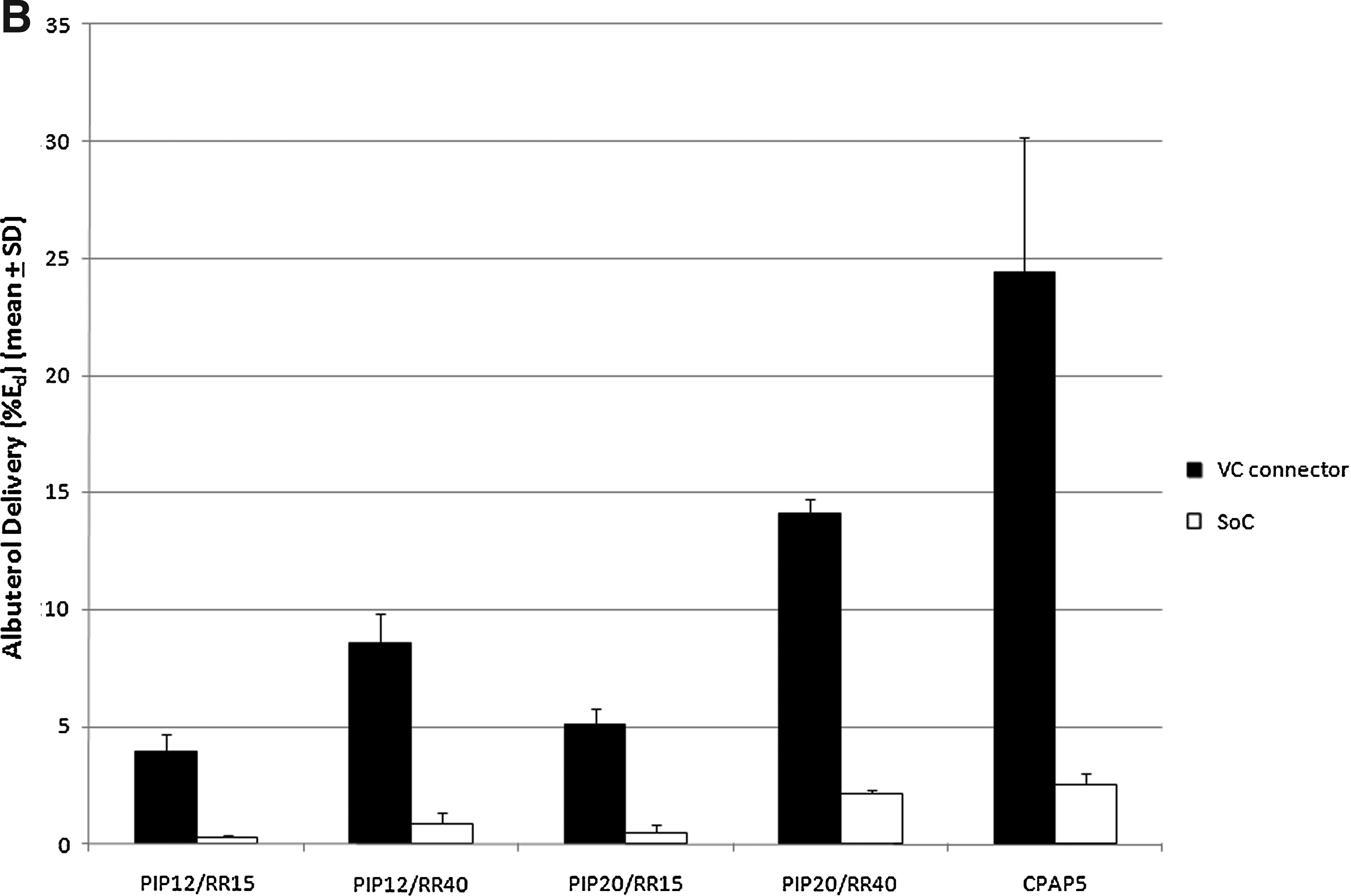
The nominal dose of drug dispensed during a 5-min sampling period was 220±0.01 μg (mean±SD). The emitted dose from the nebulizer during this time was 102.08±6.62 μg (mean±SD). Albuterol delivery under neonatal ventilation is presented in Table 2. For all ventilator settings, albuterol delivery was markedly higher when using the VC connector under all ventilator conditions tested, and these differences were statistically significant in all cases where sample size allowed for valid statistical testing (p<0.001). Highest delivered albuterol dose under neonatal IMV was achieved for PIP=20 cm H2O and VR=40 inflations/min with Vt=9.8 mL, which reached 14.44±0.61 μg (mean±SD); this corresponds to 6.5% of the nominal dose and 2.3±0.29 μg (mean±SD) with corresponding 1.05% of the nominal dose for the VC connector and SoC, respectively. For spontaneous breaths supported with CPAP, 11.3% and 1.2% of the nominal dose was delivered for the VC connector and SoC, respectively (Fig. 3A). The results are presented as a percentage of the emitted dose in Figure 3B. The amount of the drug delivered at the ET tube port entrance for the lowest ventilation parameters for SoC was below the LOD, and statistical analysis was not performed for this setting. Initially, albuterol was detected on only two out of five filters using higher ventilation settings for the SoC tests. An additional five samples for this condition were tested, and a total of seven samples were included in the final analysis.
Efficiency of the aerosol delivery.
Only values at or above limit of detection (LOD) were included in sample pool.
<0.28 μg/mL=below LOD.
Comparison via T test with Welch correction when cell sizes≥4.
Ratios of mean values.
IMV, intermittent mandatory ventilation; CPAP, continuous positive airway pressure; PIP, peak inflation pressure.
The particle size of aerosolized albuterol is presented in Table 3. The PSD of albuterol delivered using the VC connector was more similar to the PSD of the nebulizer when compared with albuterol delivered using the SoC. The D50 and D90 measured at the tip of the ET tubes differ statistically between test groups and showed larger particles for the VC connector group. There was no statistical difference between particles emitted from the nebulizer and from different sizes of ET tubes for SoC and VC connector group in D10 (Table 3).
T test with Welch correction: Control vs. SoC D50 and D90, p<0.05; Control vs. VC connector D50 and D90, p=NS; VC connector vs. SoC D50 and D90, p<0.05.
Discussion
The purpose of this study was to evaluate the performance of a novel aerosol delivery system using an in vitro model of neonatal ventilation. A neonatal ventilator that has never been tested for aerosol delivery was chosen and operated under typical neonatal conditions, including humidified and heated gases as used in real-life settings, and demonstrated that new VC connector delivers more aerosolized albuterol under neonatal IMV compared with the SoC. In addition, the novel VC connector preserved the particle size to a greater degree compared with SoC.
A previously conducted in vitro study using simulated ventilation of a pediatric model with a Vt of 100 mL showed that the aerosol-delivered dose from the jet nebulizer ranged from 3.8% to 5.2% of the nominal dose, depending on nebulizer placement and ventilator bias flow.(3) Fok et al. showed that only 0.04% of the nominal dose from the jet nebulizer was deposited in the lung of a 1-kg newborn.(16) Grigg et al. have shown lung deposition of 1.3% of the nominal dose from the ultrasonic nebulizer in larger patients with Vt of 15 mL.(17) Everard et al. tried to address the issue of neonatal small tidal volumes (11–22 mL) and short ITs among ventilated infants by testing the holding chamber with smaller volume and using longer ITs. However, this attempt was focused only to improve delivery of aerosols from meter dose inhalers (MDIs).(18) In our study, with a Vt ranging from 4.5 to 9.75 mL, delivery efficiency of SoC was 0.1% to 1.2% of the nominal dose, whereas delivery efficiency of the VC connector was 1.8% to 11.3% (Fig. 3A). When the data were expressed as percentage of the emitted dose from the Misty Finity nebulizer, the efficiency for SoC ranged from 0.5% to 2.5% and for VC connector from 3.9% to 24.4% (Fig. 3B). The difference between efficiency expressed as nominal versus emitted dose is caused by the characteristics of the small-volume jet nebulizers, which have been shown to emit only half of the drug placed in the reservoir.(3,19) In addition, previous reports that focused on testing the effect of different nebulizer placement, different types of nebulizers, different rates of bias flow, humidification, or longer ITs reported no more than a 50% improvement in emitted dose with different conditions,(3,18) whereas the VC connector had a significantly higher impact on aerosol delivery.
Our study, similar to others, showed the highest aerosol delivery under CPAP conditions with spontaneous breaths. Fink et al. studied the effect of different modes of ventilation on aerosol delivery from an MDI using an in vitro adult model and demonstrated significantly higher aerosol deposition within the lower respiratory tract with spontaneous breaths under CPAP.(20) The experiments in our study were done with heated and humidified air to closely mimic the clinical conditions for treating the premature infant. Air flow conditions and tidal volumes were driven by the ventilator based on the specified pressures. There are only two options for aerosol entrainment within the ventilator circuit: (1) placing the nebulizer within the inspiratory arm of the circuit or (2) introducing the aerosol between the wye connector and patient interface. Connecting the nebulizer to the inspiratory arm via a T-shape connector is recommended for vibrating mesh nebulizers and jet nebulizers.(21–23) It has previously been shown that aerosol delivery during mechanical ventilation depends on ventilator bias flow, where the higher the bias flow, the lower the aerosol delivery.(3) The VC connector allows for decreased exposure of the aerosol to high ventilator bias flows, and thus makes aerosol delivery less dependent on the characteristics of the ventilator used. New-generation ventilators, such as the VN500, automatically adjust bias flows, which depend on pressures and IT settings, as well as patient's lung compliance.
PSD measured at the tip of the ET tube was dependent on the size of the ET tube in both test groups, which has been shown previously.(24) Additionally, Everard et al. showed a drop in drug delivery when using a smaller ET tube (2.5 versus 3.0 mm) during in vitro testing using a Draeger Babylog neonatal ventilator circuit.(18) For both test groups in our study, aerosol particle size was gradually reduced after passing through the corrugated tube (before entering the wye and VC connector), and then it was further reduced during flow through the ET tube. This observation correlates with findings from previously published computational fluid dynamic analysis.(25)
Nevertheless, when both delivery methods were compared, it was apparent that aerosol particle size was significantly reduced in the SoC tests versus the VC connector tests. This effect is most likely caused by the higher bias flow rate in the inspiratory arm and wye connector and the dilution of aerosol emitted from the nebulizer (2 L/min) and entrained to the high (approximately 6 L/min) ventilator bias flow. The high flow rate of the aerosol in the SoC tests most likely induces impaction and settling of the droplets in the corrugated tube (in fact, the collected liquid was visible in the tubing). This leads to the reduction in PSD D50 from 5.46 μm at the nebulizer exit to 1.58 μm at the wye connector and from 5.46 to 4.57 μm for SoC and VC connector tests, respectively. Such a small particle size, in combination with short ITs and relatively low inspiratory flows, increases the risk for exhalation drug losses. Inferior lung deposition occurs in infants with bronchopulmonary dysplasia treated with smaller aerosol particles [mass median aerodynamic diameter (MMAD) of 0.83±0.01 μm] versus larger aerosol particles (MMAD of 1.88±0.01 μm).(16) It has been shown that small particles (<1 μm) are less dependent on gravitation and can be exhaled without deposition in the lungs.(26) But it is important to note that we do not know the optimal aerosol particle size to be used in mechanically ventilated newborns,(27) and future animal-based as well as clinical studies will have to answer this question.
The Spraytec spectrometer method used in this study for PSD analysis provided similar particle size results compared with previously published cascade impactor results. Fok et al. showed that jet nebulization of albuterol under neonatal ventilation conditions created an aerosol with MMAD measured at the tip of the ET tube of 0.83 μm,(16) whereas in our study, the D50 for SoC reached values of at least 0.92 μm.
There are typical study limitations linked to an in vitro approach. The emitted dose results cannot be directly extrapolated to in vivo conditions, because aerosol-delivered dose under mechanical ventilation is not equal to inhaled dose, although the ventilation parameters set up on the lung simulator were closely mimicking those of premature infants. We did not measure the emitted dose at the tip of the ET tube, as we did not want to influence the assessment with another variable. It has been shown that smaller ET tube sizes cause greater impaction of larger particles, decreasing aerosol penetration and thus potentially influencing aerosol-delivered dose, and thus inhaled dose.(25) Nevertheless, we believe that this relationship should be studied separately. Finally, we were forced to use two different methods for PSD measurements, although we have shown in the comparison bridging study that the measurement techniques are comparable in this study.
In summary, compared with SoC, utilization of the novel VC connector provides greatly increased in vitro delivery of albuterol under neonatal ventilation conditions and allows for a PSD profile that is more consistent with the PSD profile of the aerosol generator. This novel VC connector may be a useful new tool for the delivery of aerosolized medications to patients requiring positive pressure ventilatory support and may improve dosing of inhaled medications for this underserved patient population.
Footnotes
Acknowledgments
This study was supported by a local grant from PUMS, and analysis performed at WUT was supported by governmental funds for science 2010–2013 (project no. NN209 023 339). The device was provided by Discovery Laboratories, Inc.
Author Disclosure Statement
Jan Mazela is serving as consultant to Discovery Laboratories, Inc. Chris Henderson and Tim Gregory are employees of Discovery Laboratories, Inc. Both Jan Mazela and Chris Henderson are inventors of the AFECTAIR. They all own company stock and stock options. Dr. Martin Keszler serves as a consultant to Draeger and is a member of the scientific advisory board to Discovery Laboratories, Inc. There is no conflict of interest for the rest of the coauthors.
