Abstract
Abstract
In selected patients, noninvasive positive pressure ventilation (NIPPV) with a facemask is now commonly employed as the first choice for providing mechanical ventilation in the intensive care unit (ICU). Aerosol therapy for treatment of acute or acute-on-chronic respiratory failure in this setting may be delivered by pressurized metered-dose inhaler (pMDI) with a chamber spacer and facemask or nebulizer and facemask. This article reviews the host of factors influencing aerosol delivery with these devices during NIPPV. These factors include (1) the type of ventilator, (2) mode of ventilation, (3) circuit conditions, (4) type of interface, (5) type of aerosol generator, (6) drug-related factors, (7) breathing parameters, and (8) patient-related factors. Despite the impediments to efficient aerosol delivery because of continuous gas flow, high inspiratory flow rates, air leaks, circuit humidity, and patient-ventilator asynchrony, significant therapeutic effects are achieved after inhaled bronchodilator administration to patients with asthma and chronic obstructive pulmonary disease. Similarly to invasive mechanical ventilation, careful attention to the technique of drug administration is required to optimize therapeutic effects of inhaled therapies during NIPPV. Assessment of the patient's ability to tolerate a facemask, the level of respiratory distress, hemodynamic status, and synchronization of aerosol generation with inspiratory airflow are important factors contributing to the success of aerosol delivery during NIPPV. Further research into novel delivery methods, such as the use of NIPPV with nasal cannulae, could enhance the efficiency, ease of use, and reproducibility of inhalation therapy during noninvasive ventilation.
Introduction
Successful application of NIPPV in patients with acute exacerbations of chronic obstructive pulmonary disease(COPD) can often obviate the need for endotracheal intubation and improve mortality.(5–8) Despite its known benefits, the application and usage of NIPPV across various centers in the United States varies significantly depending on the availability of appropriate equipment and expertise in using this modality, whereas in Europe NIPPV is more widely used. For example, NIPPV was the initial method of mechanical ventilation in 16% of patients in 42 ICUs in Europe and Tunisia, it was the initial choice in 50% of patients with hypercapnic respiratory failure, and endotracheal intubation could be avoided in the majority (60%) of patients receiving NIPPV.(9)
Patients with acute or acute-on-chronic respiratory failure who receive NIPPV often require inhaled bronchodilators for relief of airway obstruction. One option is to remove the patient from NIPPV and administer bronchodilators by pressurized metered-dose inhaler (pMDI) and holding chamber or nebulizer as patients can tolerate brief periods of discontinuation that are needed for providing such treatments.(10) The preferable option, however, is to continue NIPPV without interruption, especially in hypoxemic or acutely dyspneic patients. In addition to bronchodilators, other inhaled drugs that could be employed in patients receiving NIPPV include antibiotics, prostacyclins, surfactant, and mucoactive agents, among others. The use of these drugs in patients receiving mechanical ventilation has been previously reviewed.(11)
Aerosol delivery to patients receiving NIPPV in the ICU is a complex task! Aerosol losses in the circuit, interfaces, and upper airways need to be minimized in order to optimize lung deposition. Delivery devices and techniques that can overcome the effects of air leaks, high inspiratory flows, and patient ventilator asynchrony are needed.(12) High aerosol losses during NIPPV result in aerosol delivery of <1–10% in adults and children in vitro,(13,14) and as low as 1 to 6% in vivo.(15) This review examines inhaled therapy to adult patients receiving NIPPV, and elucidates the effects of a complex array of variables influencing optimal drug delivery (Fig. 1). Because data on aerosol therapy in patients receiving NIPPV is not as robust as that in invasive mechanical ventilation,(12) another purpose of this article is to highlight areas for further research.
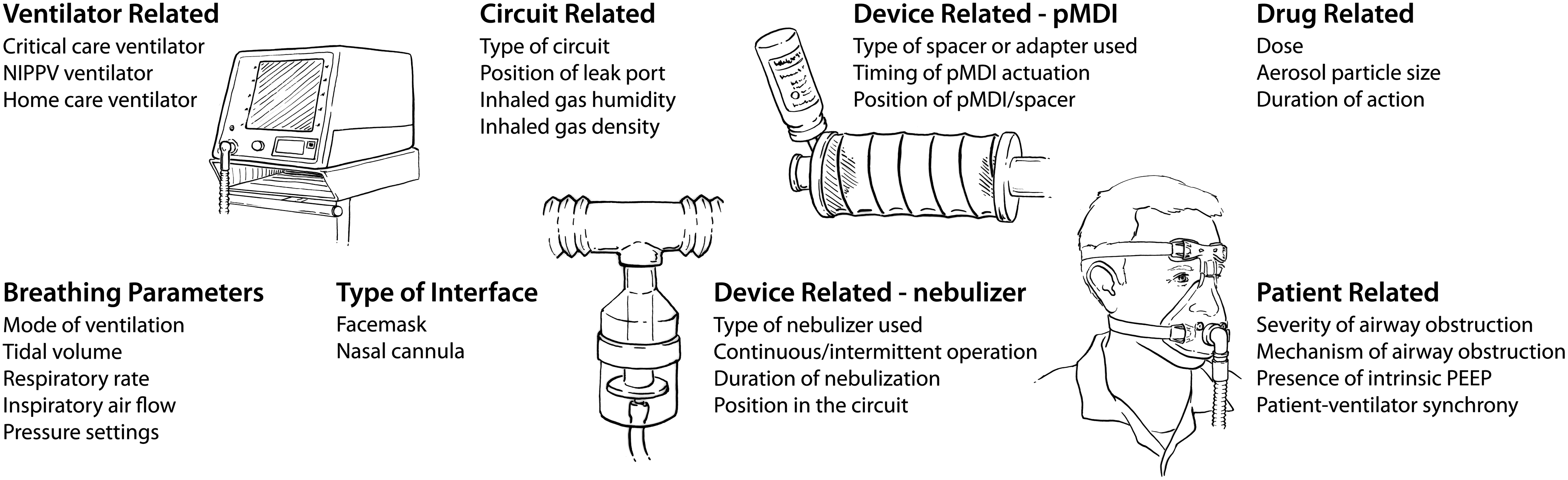
Schematic showing the various factors influencing aerosol delivery during noninvasive positive pressure ventilation (NIPPV). PEEP denotes positive end-expiratory pressure, pMDI denotes pressurized metered-dose inhaler.
Methods
A broad-based literature search was conducted in August 2011 for English language articles in Ovid MEDLINE (including in-process and nonindexed records), the PubMed publisher only records not included on Ovid, the Cochrane Central Register of Controlled Trials, and Summon published since 2005. Synonyms for noninvasive ventilation techniques and equipment were extracted from some key reviews and were deployed in combination with controlled vocabulary (where available) in each database. Also included were articles from the authors' personal reference library. Citations from published articles were also searched for relevant articles.
Factors Influencing Aerosol Delivery During NIPPV In Vitro
A multitude of factors influencing the efficiency of aerosol delivery during NIPPV are shown in Figure 1. These factors include: (1) the type of ventilator, (2) mode of ventilation, (3) circuit conditions, (4) type of interface, (5) type of aerosol generator and its configuration, (6) drug related factors, including aerosol particle size, (7) breathing parameters, and (8) patient-related factors, especially the ability to tolerate a facemask, the level of respiratory distress, hemodynamic status, the type and severity of lung disease, and synchronization of aerosol generation with inspiratory airflow.
Type of ventilator
Several varieties of ventilators that are employed for NIPPV include the following.
Portable ventilators specifically for NIPPV
These machines are typically flow-generating units that have a single tube connecting the ventilator to the facemask. An air leak is incorporated in the circuit or the mask to allow exhaled gases to escape. The inability of previous models to provide a precise fraction of inspired oxygen(16) has been rectified by provision of an oxygen blender in newer designs of such ventilators.
Critical care ventilators
Ventilators that are commonly employed in ICUs for invasive mechanical ventilation have separate arms for inspiratory and expiratory gases to minimize rebreathing of exhaled gas and they are more expensive than portable ventilators for NIPPV. With these ventilators, occurrence of air leaks during NIPPV leads to frequent and bothersome alarms.
Portable home care ventilators
are often employed for patients with chronic respiratory failure due to neuromuscular disease, but they do not tolerate large air leaks well. Recent improvements in home care ventilators, such as higher flow capability, more sensitive trigger mechanisms, and better alarm systems, have made them more suitable for application of NIPPV.(17)
Mode of ventilation
Several modes of ventilation that are employed for NIPPV could influence aerosol delivery because of differences in pressure settings and airflow rates (see details in a later section). Some ventilator modes employed for NIPPV are briefly described.
Intermittent positive pressure breathing (IPPB)
A pneumatically driven device provides a pressure-triggered and pressure-cycled inspiratory cycle to enhance the patient's tidal volume. This mode was widely employed for lung expansion therapy and aerosol delivery in the 1960s. Unlike pressure support ventilation (see below), IPPB does not sustain a constant pressure during inspiration and its beneficial effects are short lived. In normal, nonintubated subjects, the work of breathing was lower during pressure support than during IPPB.(18) Thus, IPPB is much less effective at unloading the patient's respiratory muscles compared to pressure support.
Bilevel ventilation
Bilevel ventilation employs two independent and adjustable levels of positive pressure and is commonly employed for NIPPV (Fig. 2). The difference between the set inspiratory positive airway pressure and the set expiratory positive airway pressure is the level of inspiratory pressure support (or pressure control). The inspiratory and expiratory pressures could also be applied independently. Continuous positive airway pressure (CPAP) maintains a constant positive pressure in the airways, raises the functional residual capacity, prevents airway collapse, reduces left ventricular transmural pressure and left ventricular afterload, and increases cardiac output.(19,20) Application of CPAP recruits alveoli and maintains their patency throughout the respiratory cycle, thereby reducing ventilation–perfusion mismatch and shunt.(21) During pressure support, patient-triggered and pressure targeted support is added to boost the patient's spontaneous inspiratory effort until the inspiratory flow falls below a specified value. Application of pressure support reduces inspiratory muscle effort and respiratory rate, increases tidal volume, and improves arterial blood gas values.(22,23) By reversing small airway closure and microatelectasis, pressure support could also promote more uniform pulmonary aerosol deposition.(24) In patients with COPD, application of bilevel ventilation (CPAP and pressure support) achieves a greater reduction in the work of breathing than either mode alone.(25)
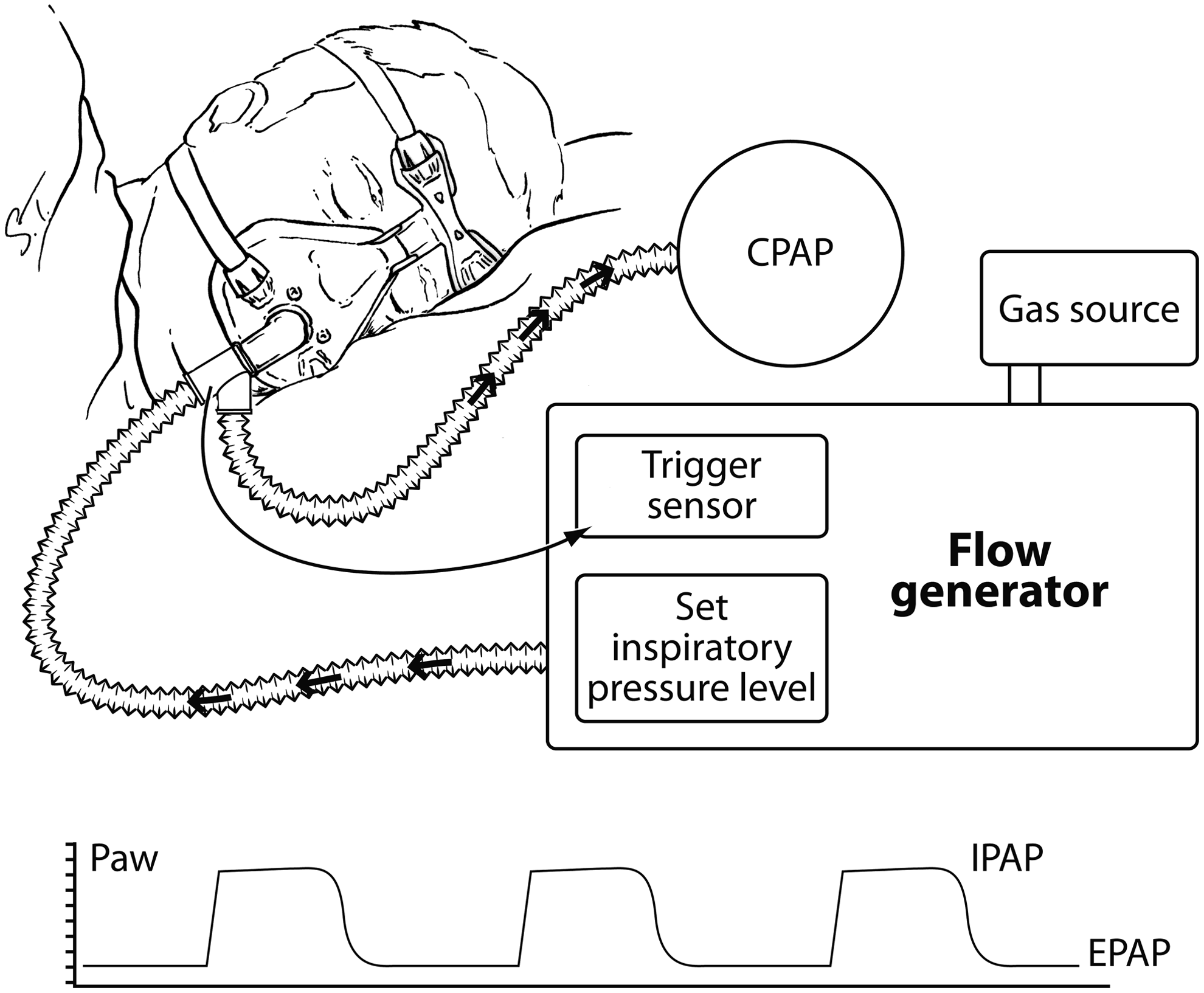
Schematic showing the principle of bilevel ventilation. A resistor maintains a continuous positive airway pressure (CPAP) level. This pressure is denoted by the expiratory positive airway pressure (EPAP) on the airway pressure (Paw) tracing. The patients breathing efforts trigger the ventilator to provide an inspiratory boost until the set pressure is achieved, and this pressure is maintained until the ventilator cycles into exhalation. The set inspiratory pressure is represented by the inspiratory positive airway pressure (IPAP) on the Paw tracing. The difference between IPAP and EPAP is the level of inspiratory pressure support.
Other modes
Various other modes, including pressure control ventilation, proportional assist ventilation,(26) intermittent percussive ventilation,(27) and high-frequency oscillation,(28) are infrequently employed for NIPPV.
Circuit-related factors
Type of circuit
Most ventilators designed for NIPPV use a single limb circuit without a true exhalation valve. A fixed leak in the circuit or mask allows passage of exhaled gases, but the potential for rebreathing carbon dioxide remains a concern. Carbon dioxide rebreathing could be diminished by increasing the leak flow, setting a higher CPAP level, by employing a nonrebreathing exhalation valve,(29) or by incorporating the leak into the facemask instead of a leak in the circuit.(30)
Humidity
During invasive mechanical ventilation, drug delivery to the lower respiratory tract from both pMDIs and nebulizers is reduced by 40% or more in a humidified compared to a dry circuit (Fig. 3).(12,31–33) Circuit humidity probably increases impaction losses by increasing the size of drug particles.(34,35) Although adding a heat and moisture exchanger between the nebulizer and the CPAP device did not influence aerosol particle size,(36) the overall effects of humidity on aerosol delivery during NIPPV are probably similar to those observed during invasive mechanical ventilation.
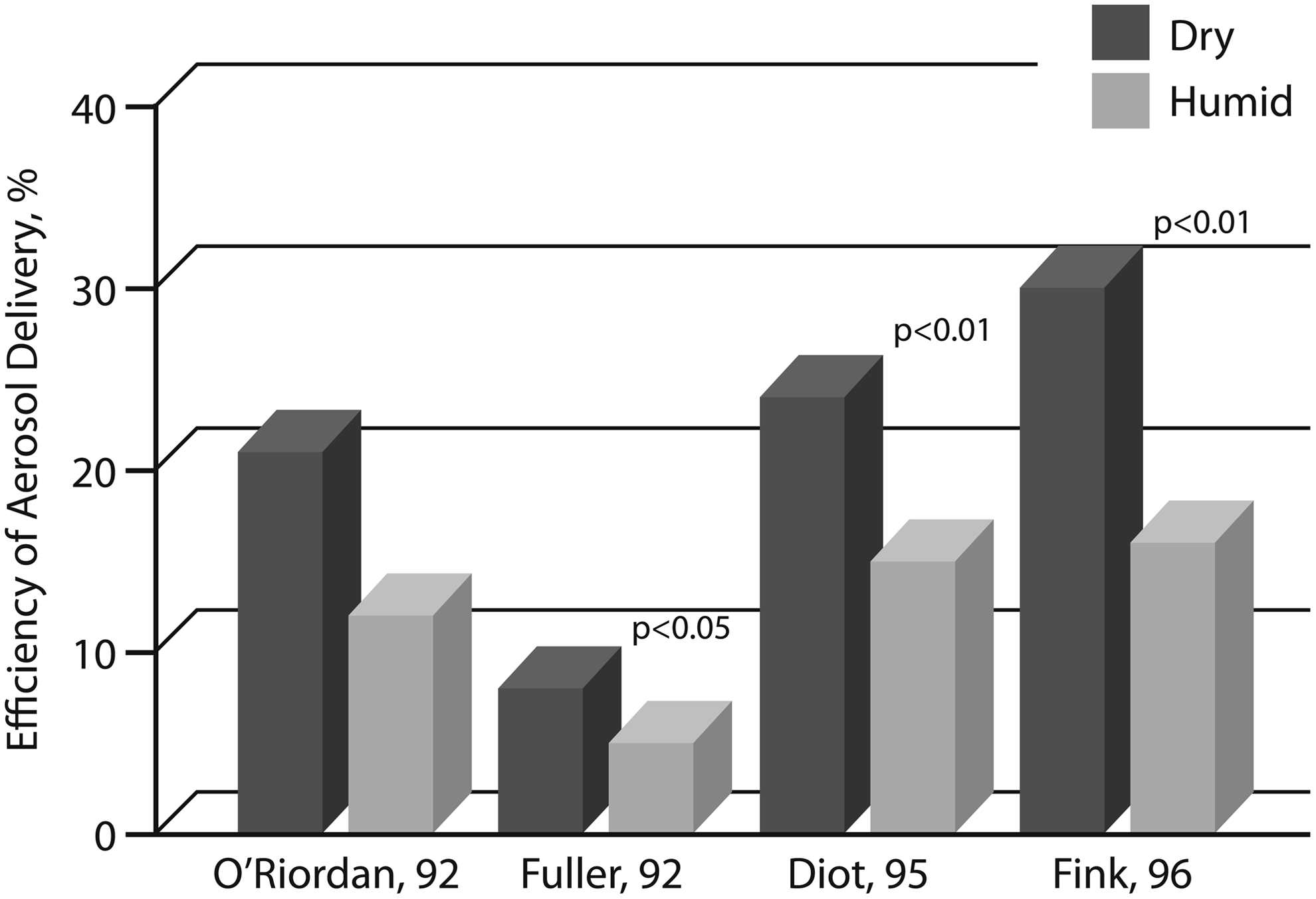
Effect of humidity on aerosol delivery in patients receiving invasive mechanical ventilation. The mean efficiency of aerosol delivery to the lower respiratory tract is shown for bench models of invasive mechanical ventilation with dry (black bars) and humidified (gray bars) circuits. Numbers after the authors' names indicate the year in which the studies were published.
During NIPPV, unlike invasive mechanical ventilation, air is humidified during its passage through the nose. The humidification capacity of the nose may be overwhelmed by the sustained high airflow rates employed during NIPPV, and several factors influence the humidity level in the inspired gas. ICU ventilators and high flow oxygen devices provide dry “wall” gases,(37,38) whereas flow generators designed for NIPPV deliver ambient air whose humidity depends on ambient temperature and humidity and the use of air conditioning. With the use of an external humidifier, the gas flow rate,(37) presence of leaks,(39) whether the patient breathes through the nose or mouth, and the type of humidifier employed(37) all impact humidity of the inhaled gas. In some patients receiving NIPPV, a cool, ambient temperature, passover humidifier may add sufficient moisture, but for most patients a heated humidifier or heat and moisture exchanger is more effective in preventing drying of the nasal and upper airway mucosa.(36,37)
High air flow rates employed during NIPPV not only rapidly cause mouth and nose dryness and throat irritation,(38) they also produce increases in nasal resistance,(39) and have the potential to induce bronchial hyperresponsiveness in the majority of patients who are receiving NIPPV because of airway inflammation. The undesirable effect of inhaled dry gas to produce an increase in airway resistance negates the potential benefits of aerosol bronchodilator therapy. Adverse effects caused by dry gases could be mitigated by appropriately humidifying the inhaled gas.(40) Thus, optimal humidification of the inhaled gas could improve patient comfort, facilitate use of the mask, and improve bronchodilator effects in those requiring NIPPV. The dose of inhaled drugs may have to be adjusted to account for the effects of humidity on aerosol delivery.(12,31–33) However, the extent of dose modification with various levels of humidification of the inhaled gas during NIPPV merits further inquiry.
Gas density
Inhalation of a less dense gas, such as a heliox, an 80/20 mixture of air and oxygen, makes airflow less turbulent and more laminar. During NIPPV in patients with acute exacerbations of COPD, heliox reduces patient's work of breathing and improves arterial blood gas levels more than NIPPV with air.(41,42) Enhanced unloading of the respiratory muscles by heliox could improve tolerance to NIPPV and provide additional benefits in patients with acute exacerbations of COPD. During mechanical ventilation, the ventilator employed must be compatible with the use of heliox.(43)
In normal, healthy subjects, heliox reduces upper airway drug deposition and increases drug deposition in the peripheral lung.(44,45) Likewise, in a bench model of adult mechanical ventilation heliox improved drug delivery from a pMDI, principally due to a reduction in aerosol deposition in the ventilator circuit and endotracheal tube.(46) In contrast, nebulizer operation with heliox reduced drug output and respirable mass.(46,47) To optimize pulmonary deposition of aerosol from a nebulizer the recommended technique is to operate the jet nebulizer with air or oxygen at a flow rate of 6 to 8 L/min and entrain the aerosol generated into a ventilator circuit containing heliox.(46) Currently, there is a dearth of information on how breathing heliox affects aerosol deposition and response to inhaled therapies during NIPPV.
Type of interface
Facemasks
Several different type of interfaces, including nasal mask, full-face or oronasal mask, total face mask, nasal pillows or plugs, mouthpieces, and a helmet are employed to apply NIPPV (Table 1), but most clinicians prefer full-face masks or oronasal masks.(1,48) Although nasal and oronasal masks improve arterial blood gases equally,(49) oronasal masks are considered as the first choice in patients with acute respiratory failure.(50) Nasal masks may be employed for patients who do not tolerate oronasal masks because of claustrophobia or a frequent productive cough.(51) Selection between various oronasal masks may not be critical because their internal volume did not influence patient's breathing frequency, ventilatory pattern, or gas exchange in the short term.(52) Aerosol deposition in nasal passages significantly reduces drug delivery to the lung,(53–55) and could reduce bronchodilator efficacy compared to inhalation with a mouthpiece.(56) Based on predicted nasal deposition of aerosol droplets 2 to 7 μm in size, 40 to 99% of the aerosol inhaled during NIPPV is likely to deposit in the nose.(57,58) For optimal aerosol delivery, the facemask should produce a tight seal(59–61) to avoid aerosol leakage and to reduce aerosol deposition around the eyes.(62)
Humidified high-flow nasal cannula
These devices are commonly employed in the ICU to enhance gas exchange and avoid mechanical ventilation.(63,64) With this device, air or blended oxygen is preconditioned with heat and humidity to allow continuous delivery through a nasal cannula with flow rates as high as 40 L/min. Humidified high-flow nasal cannula use provides a low level of CPAP depending on the air leak, and the relationship between the size of the nasal prongs and nares.(64) In a bench study, the inhaled mass of aerosol and aerosol particle size with a vibrating aperture plate nebulizer, an adult-sized nasal cannula, and heated and humidified gas flow (3 L/min) were comparable to those obtained with mouthpiece inhalation from a continuously operating jet nebulizer.(65)
Enhanced condensational growth
Longest and colleagues(66,67) applied this novel concept of aerosol delivery to a nose, mouth, and throat model during NIPPV. A submicrometer aerosol was provided to one nostril at slightly subsaturated conditions, whereas a humidified airstream saturated with water vapor was delivered to the other nostril with a temperature a few degrees above in vivo wall conditions. The nasal septum physically separated the two streams allowing the submicrometer aerosol to retain its small size and minimize nasal deposition. The two airstreams combined in the nasopharynx with condensational growth of the aerosol particles as the air stream moved downward into the lungs. With this approach, mean drug deposition in the nose, mouth, and throat model was reduced from 72.6% to 14.8%, whereas aerosol size increased from an initial mass median aerodynamic diameter of 900 nm to approximately 2 μm at the exit of the model.(67) The growth in aerosol size is predicted to enhance lung deposition and limit aerosol losses during exhalation.(67) This technique could overcome the barrier imposed to aerosol particles by the nasal passages, and lung deposition could be enhanced to ∼90% of the inhaled dose.(67) Further studies in patients are needed to corroborate these interesting in vitro findings.
Type of aerosol generator
pMDI and spacer
For a pMDI to be employed during NIPPV, the canister supplied by the manufacturer must be removed from the actuator and connected to the mask/circuit with a different adapter (Fig. 4). Several varieties of chamber or reservoir spacers are commercially available for use in mechanically ventilated patients.(12,68–71) During invasive mechanical ventilation, a chamber spacer with a pMDI was four- to sixfold more efficient for aerosol drug delivery compared with either an elbow adapter or a unidirectional inline spacer.(12,68–71) A pMDI and chamber spacer placed at a distance of approximately 15 cm from the endotracheal tube provides efficient aerosol delivery and elicits a significant bronchodilator response.(12,31,72,73) Although comparable studies have not been performed during NIPPV, it is logical to connect a pMDI and chamber spacer to the facemask.
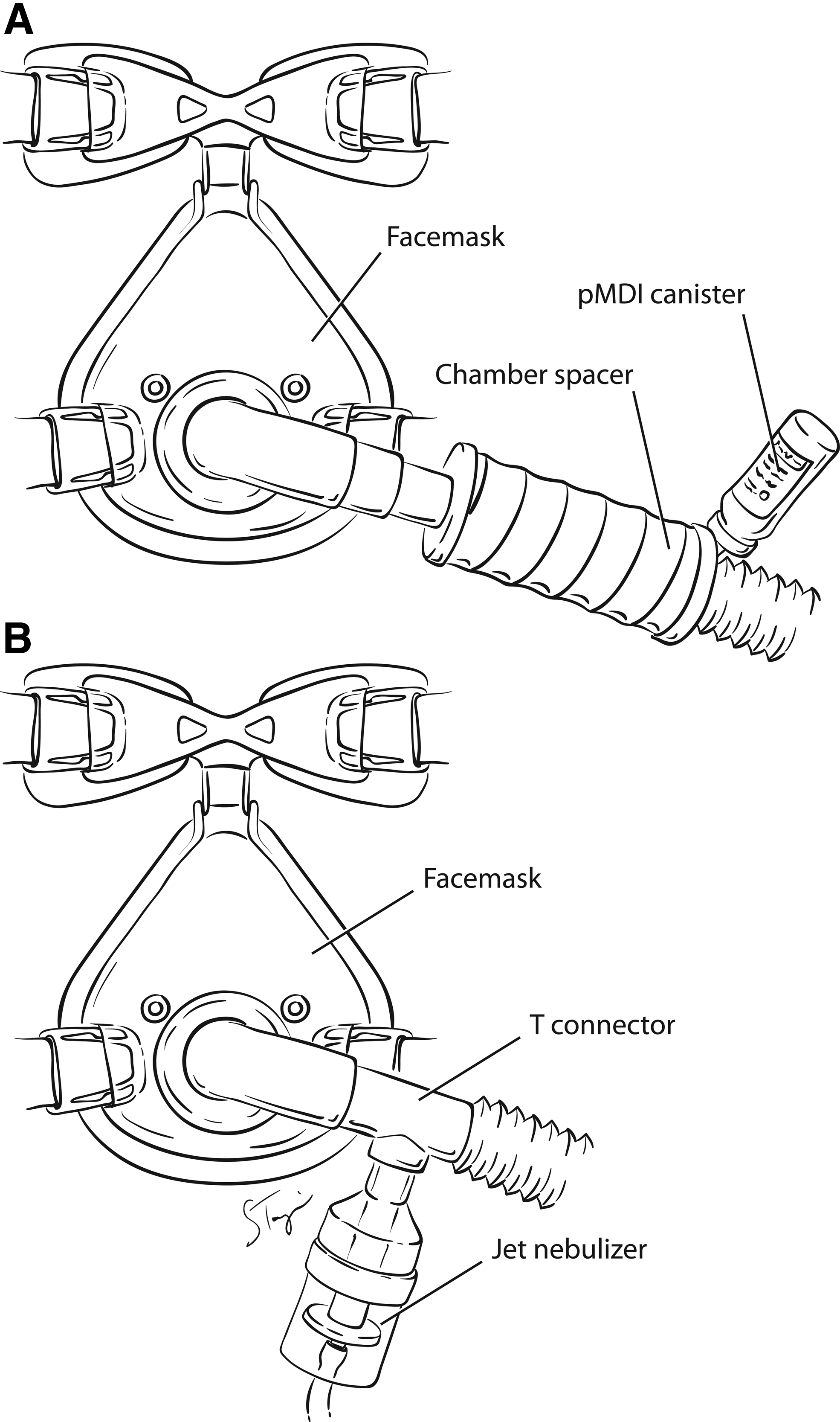
Method of connecting pMDI and chamber spacer or jet nebulizer to a facemask during NIPPV. The pMDI canister requires a third-party actuator to generate an aerosol in a ventilator circuit. A jet nebulizer can be connected to the ventilator circuit with a T-piece.
The pMDIs have now been reformulated with hydrofluoroalkane propellants, which have a similar or superior lung deposition profile to previously employed chlorofluorocarbon-pMDIs.(74–76) Newer generation pMDIs not only differ in the type of propellant, they also have different formulations, metering valve, and actuator designs. During invasive mechanical ventilation, hydrofluoroalkane-pMDIs achieve drug delivery similar to chlorofluorocarbon-pMDIs in pediatric and neonatal models of mechanical ventilation,(77,78) but in an adult model a hydrofluoroalkane–beclomethasone pMDI that produces an ultrafine aerosol had a higher efficiency of drug delivery to the lung compared to the previous chlorofluorocarbon–beclomethasone pMDI that produces a coarser aerosol.(79) During noninvasive bilevel ventilation, the use of a chlorofluorocarbon-pMDI connected to a facemask with a cylindrical chamber spacer (Aerovent, Monaghan Medical, Plattsburgh, NY) provided delivery of ∼9% of the nominal dose of albuterol.(80) Because the stems of various pMDIs differ in size, it is important to verify the quality of the aerosol produced when the canister is employed with a third-party actuator.(32) Thus, attention to in vitro characterization of aerosols produced by pMDIs is required to reduce the variability in drug delivery during NIPPV.
Nebulizer
Both jet and ultrasonic nebulizers are connected in the inspiratory limb of the ventilator circuit or directly connected to the mask (Fig. 4). During invasive ventilation, placement of a jet nebulizer at a distance from the endotracheal tube improves its efficiency compared with placing it between the patient Y and endotracheal tube;(81–84) however, similar studies have not been performed during NIPPV with a critical care ventilator. With conventional jet nebulizers, the nebulizer brand,(81,85) diluent volume, operating pressures and flows, and duration of treatment,(85,86) are well known to influence the efficiency of aerosol generation.
With ventilators designed for NIPPV, the position of the leak port in the single-arm circuit used for bilevel positive pressure ventilation influences nebulizer efficiency (Fig. 5), but it does not influence pMDI efficiency of drug delivery.(80) Nebulizer efficiency is higher with the leak port in the circuit compared to a leak port in the facemask.(80) Calvert and coworkers(87) reported that a nebulizer placed between the leak port and ventilator performs with a higher efficiency than placement of the nebulizer between the leak port and face mask. In contrast, Abdelrahim and colleagues(88) found a higher drug delivery with the nebulizer placed between the leak port and face mask than placement between the ventilator and leak port (Table 2). The difference in the results of the two studies mentioned above(87,88) may be explained by the fact that the former investigators placed the nebulizer at a farther distance from the leak port than the latter, and this maneuver could have reduced aerosol losses during exhalation (Fig. 5). Abdelrahim and colleagues(88) also found that a newer generation vibrating aperture plate nebulizer delivered two- to fourfold higher drug dose than a jet nebulizer.
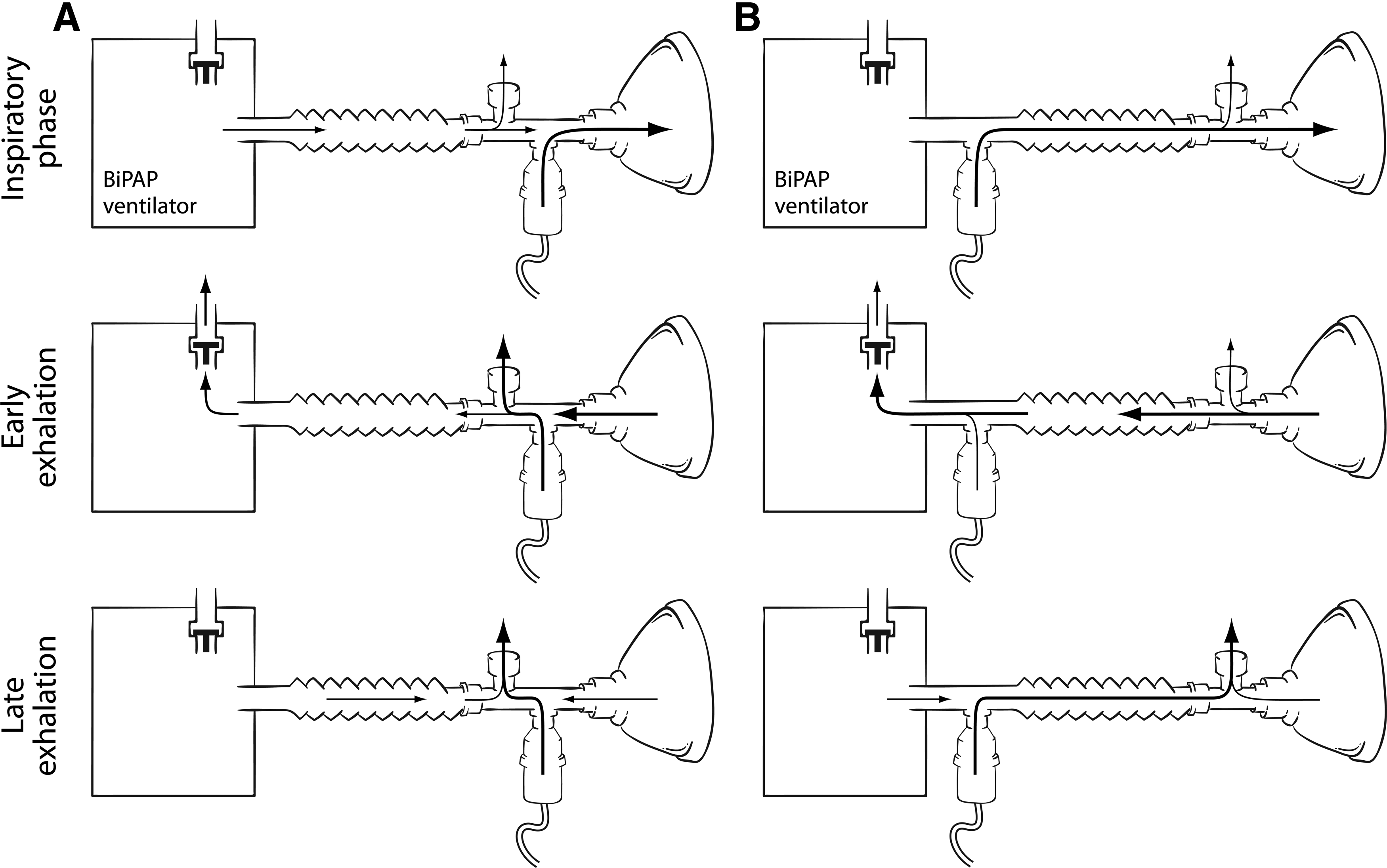
Schematic showing the effect of leak port on aerosol delivery from a nebulizer. In
NR, not reported.
p<0.05 compared to filter deposition when nebulizer was placed between leak port and filter.
Filter deposition with Aeroneb placed between the leak port and face mask was higher than filter deposition achieved with the Aeroneb placed between leak port and ventilator (farther from filter), and for both positions with the Sidestream nebuizer. Nominal dose used was 5 mg of albuterol [87,94] or 5 mg of terbutaline [88].
Dry powder inhaler
Dry powder inhalers have not been adapted for aerosol delivery during NIPPV. The efficiency of aerosol delivery with dry powder devices in this setting is likely to be low, because of the known effect of humidity in reducing aerosol delivery from such devices.(89)
Drug-related
Drug effects depend on the type of drug, its duration of action, and the size of drug particles in the aerosol. For optimal pulmonary deposition, aerosol particles should be small enough to penetrate through the upper airway, yet large enough to avoid being carried back out into the atmosphere with the exhaled breath. Devices that produce aerosols with mass median aerodynamic diameter <2 μm are more efficient for pulmonary deposition during invasive mechanical ventilation than devices that produce aerosols with larger particles.(33,90) However, some nebulizers that produce aerosols with such a small particle size may require a considerably greater time to deliver a standard therapeutic dose.(33,68) Two groups of investigators determined aerosol particle size during NIPPV with nebulizers (Table 3).(82,83) One group reported that a jet nebulizer produced a finer aerosol during NIPPV compared to nebulization with the ventilator turned off, and the size of the aerosol particles had a high likelihood of deposition in the lung.(87) The other group of investigators found that although a jet nebulizer produced a finer aerosol than a vibrating aperture plate nebulizer, but the fine particle dose with the latter was two to four times higher than that with the former device.(88)
Top: Data from Abdelrahim et al. J Pharmac Pharmacol. 2010; 62;966-972.(88)
Bottom: data from Calvert et al. J Pharmac Pharmacol. 2006; 58;1553-1557.(87)
MMAD, mass median aerodynamic diameter; GSD, geometric standard deviation; FPD, fine particle dose.
Smaller particle size with Sidestream compared to Aeroneb.
Lower fine particle dose with Sidestream compared to Aeroneb. Nominal dose used was 5 mg of terbutaline(88) or 5 mg of albuterol.(87)
Lower with NIPPV and nebulizer compared to nebulizer alone.
Breathing parameters
Application of CPAP in bench models reduces drug delivery from a jet nebulizer.(15,91) Moreover, evaporation of solvent due to the high rates of gas flow used during CPAP cools the aerosol, reduces droplet size, and increases tonicity,(92) with the potential to induce paradoxical bronchoconstricion. High flow rates during CPAP dilute the aerosol and reduce aerosol delivery due to high impaction losses.(15) In patients with cystic fibrosis, nebulization with a positive expiratory pressure device that maintained a pressure between 10 and 20 cm H2O reduced the lung deposition fraction from 10.76±4.52% without positive expiratory pressure to 6.10±3.05% with positive expiratory pressure.(93) However, positive expiratory pressure significantly reduced the particle size of the aerosol.(93) After administration of amikacin via a jet nebulizer, significantly more drug was recovered in the urine over 24 h (4.9% of the initial dose) compared to administration with the same jet nebulizer and CPAP of 6 cm H2O (1.97%).(91) These investigators reported that application of CPAP lowered respiratory rate and I:E time, thereby reducing the total time for which the continuously generated aerosol was actually inhaled.
Chatmongkolchart and coinvestigators found a fivefold variation in the amount of albuterol delivered by a jet nebulizer in vitro (between 5 and 25% of the nominal dose); delivery was highest (25%) when the nebulizer was close to the patient (between the leak port and patient connection), and inspiratory pressure was set higher (Fig. 6). At a breathing rate of 20 breaths/min, the set expiratory pressure level (5 or 10 cmH2O) did not significantly influence aerosol delivery (Fig. 6).(94)
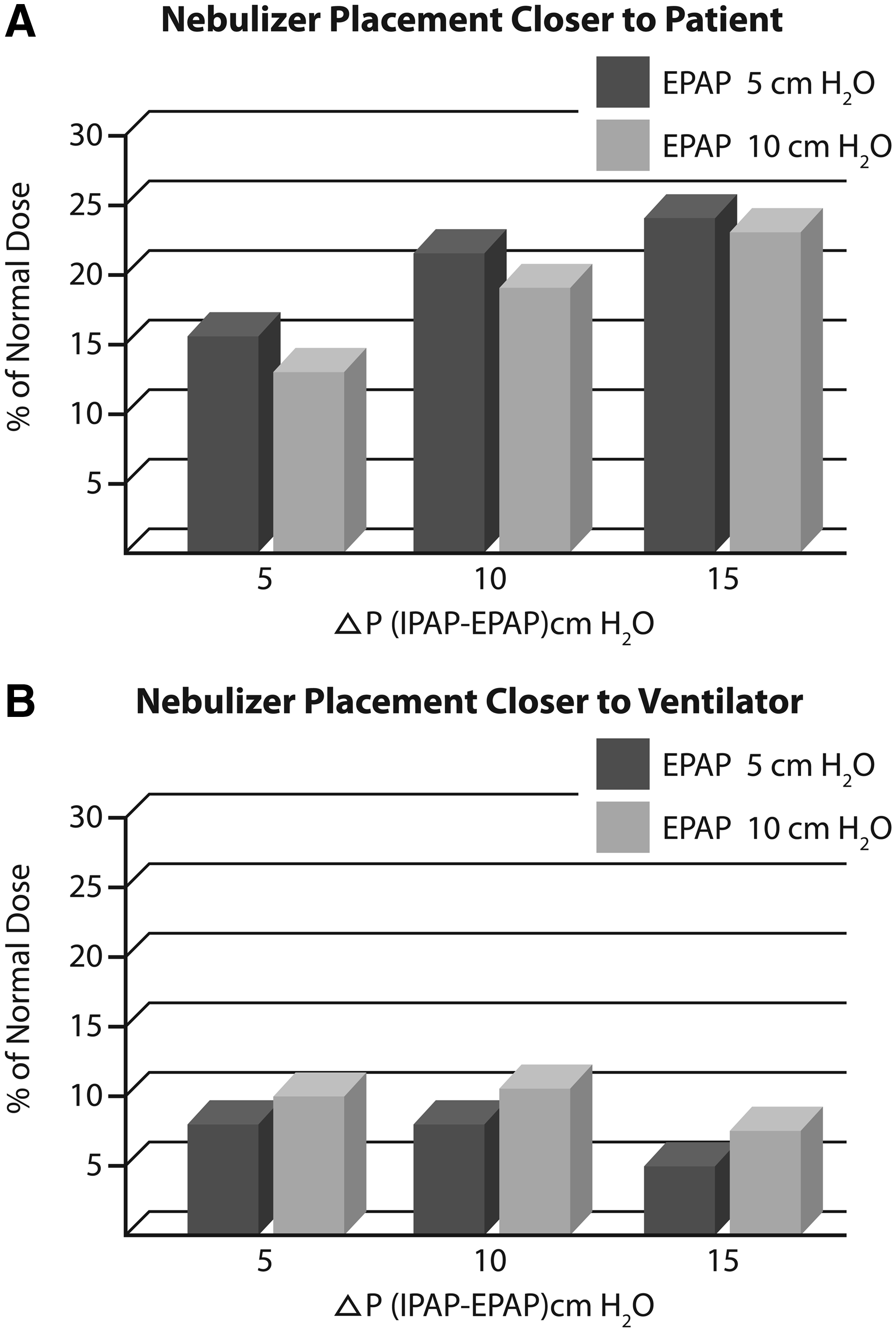
Bar diagram showing the effect of ventilator settings on the efficiency of aerosol delivery during bilevel positive airway pressure ventilation. Only data from experiments conducted at a breathing rate of 20/min are depicted as these are more relevant to patients receiving NIPPV in the ICU. The mean efficiency of aerosol delivery (as a % of the nominal dose) is shown at Expiratory positive airway pressure (EPAP) of 5 or 10 cm H2O and difference between Inspiratory positive airway pressure (IPAP) and EPAP of 5, 10, or 15 cm H2O. Inspiratory Pressure Support (ΔIPAP-EPAP) of 10 to 15 cm H2O had the highest efficiency of drug delivery when the nebulizer was placed closer to the patient, that is, between the leak port and face mask. When the nebulizer was placed farther away from the patient, that is, between the ventilator and leak port, the efficiency of aerosol delivery declined compared to placement closer to the patient, and the pressure settings had much less influence on aerosol delivery. (Data from Reference #94.)
Patient related
The type and severity of lung disease and the patient's ability to tolerate a facemask or other suitable interface are important considerations for NIPPV to be successful. In addition, synchronization of the ventilator airflow and patient's breathing is essential for optimal aerosol delivery. pMDI actuation must be synchronized with the precise onset of inspiratory airflow from the ventilator.(95,96) As short as 1- to 1.5-second delay between pMDI actuation and inhalation can profoundly reduce the efficiency of drug delivery.(68) Similar to invasive mechanical ventilation, optimum aerosol delivery from a pMDI during NIPPV requires synchronization of pMDI actuation with inspiratory airflow. When the pMDI was actuated during exhalation, aerosol delivery was <50% compared to that achieved by pMDI actuation at the onset of inspiration.(13,80)
In a ventilator circuit, nebulizers can be operated continuously or intermittently by airflow from the ventilator. Continuous aerosol generation requires a pressurized source of gas (from a wall outlet, pressurized tank, or an air compressor), whereas intermittent operation requires synchronization with inspiratory airflow. During invasive mechanical ventilation, intermittent operation of the nebulizer minimizes aerosol wastage during exhalation and is more efficient for aerosol delivery compared with continuous aerosol generation.(33,83) Similar considerations should apply during NIPPV.
Clinical Use of Inhaled Drugs in Patients Receiving NIPPV
High inspiratory flows employed during NIPPV increase turbulence and the associated high inertial forces cause greater particle impaction in central airways.(97,98) On the other hand, application of positive pressure reduces aerosol particle size,(87) increases tidal volume, and reduces respiratory rate, all of which tend to enhance aerosol delivery.(99) Moreover, increase in expiratory time due to a slower respiratory rate could enhance particle sedimentation and alter the pattern of drug deposition during exhalation.(99) Pressure support or CPAP are more helpful than IPPB in reducing work of breathing in patients with acute bronchoconstriction,(18,100,101) and this may influence the response to inhaled drugs.
Shenfield and colleagues(102) found higher plasma levels of albuterol were achieved with pMDI and IPPB compared to administration by pMDI alone in patients recovering from an acute exacerbation of asthma. However, a later study by Dolovich and coworkers(103) in patients with severe COPD and relatively fixed airway obstruction reported ∼30% reduction in delivery of 99mTechnetium-labeled pertechnate from an ultrasonic nebulizer employed with IPPB.
In healthy volunteers, NIPPV reduced radioaerosol deposition compared to spontaneous breathing.(104) Likewise, pulmonary deposition of radiolabel was not enhanced by unconventional modes of NIPPV.(27,28)
In six healthy volunteers who received pressure support ventilation with a mouthpiece, administration of amikacin by Aernoeb Pro (Aerogen, Galway, Ireland) and vertically oriented Idehaler spacer (La Diffusion Technique Française, St-Etienne, France) achieved systemic absorption of ∼10–13% of the nominal dose.(105) Similar efficiency of aerosol deposition with pMDIs and nebulizers is observed in patients receiving invasive mechanical ventilation.(32,106) In contrast, intermittent percussive ventilation reduced pulmonary deposition of amikacin compared to jet nebulization alone.(91,107)
In patients with acute asthma, CPAP alone reduced respiratory rate and dyspnea scores without significant changes in forced expiratory volume in 1 sec or arterial blood gas values.(108) The effects of bronchodilator therapy with NIPPV in patients with asthma are summarized in Table 4.
CPAP, continuous positive airway pressure; EPAP, expiratory positive airway pressure; FEF25%–75%, forced expiratory flow between 25% and 75% of the FVC; FEV1, forced expiratory volume in 1 sec;, FVC, forced vital capacity; IPAP, inspiratory positive airway pressure; IPPB, intermittent positive pressure breathing; PAP, positive airway pressure; PEFR, peak expiratory flow rate; PEP, positive expiratory pressure.
In five asthmatics who were recovering from an acute exacerbation, Shenfield and colleagues(102) administered albuterol by IPPB and reported significant improvements in forced expiratory volume in 1 sec from baseline (mean 37%, range=23 to 67%) in all patients.
A randomized crossover study that compared three times daily treatment over 2 weeks with terbutaline in eight asthmatics reported modest increases in peak expiratory flow rate with all treatments, but the highest values were achieved with the combination of terbutaline given by pMDI and conespacer connected to a facemask that provided positive expiratory pressure of 10 to 15 cm H2O compared to terbutaline alone without positive expiratory pressure, or placebo pMDI with similar levels of positive expiratory pressure.(109) In nine stable patients with asthma, application of CPAP (10 cm H2O) did not affect the shape of the dose–response curve or the magnitude of the bronchodilator response to nebulized albuterol.(15) Another group of investigators reported similar results, although they found that oxygenation improved to a greater extent when nebulized bronchodilators were administered with CPAP compared to standard nebulizer therapy.(36)
In patients with acute asthma exacerbations in an Emergency Department, patients receiving nebulized albuterol with bilevel positive airway pressure had more rapid and greater improvement in peak expiratory flow rate than patients receiving a similar dose by nebulizer alone.(110) However, it is unclear if this improvement was due to relief provided by inspiratory pressure support, enhanced drug delivery or drug effect due to NIPPV, or a combination of the two.
A prospective investigation in patients receiving standard bronchodilator and intravenous corticosteroid therapy for asthma, randomized patients to bilevel positive airway pressure (IPAP max 14 cm H2O; EPAP 3 to 5 cm H2O) applied for 3 h versus sham bilevel positive airway pressure.(111) Among patients receiving NIPPV, 80% achieved a ≥50% increase in forced expiratory volume in 1 sec compared to baseline versus 20% in those receiving sham treatment (p<0.004), and they also achieved a higher mean increase in forced expiratory volume in 1 sec (p=0.006).(111) Notably, patients were taken off NIPPV during nebulizer treatments.(111)
A prospective randomized controlled trial in 36 patients with severe asthma presenting to an Emergency Department compared three groups of patients: one group (control) received nebulization with unpressured mask, a second group received nebulization with bilevel positive airway pressure 15/5 cm H2O, and a third group received nebulization with pressures of 15/10 cm H2O.(112) After 30 min of treatment (2.5 mg fenoterol and 0.25 mg ipratropium by nebulizer), the group receiving pressures of 15/10 cm H2O had greater improvement in forced vital capacity, forced expiatory volume in 1 sec, and peak expiratory flow rate, whereas the group receiving pressures of 15/5 cm H2O showed improvement only in peak expiratory flow rate compared to those receiving nebulized drugs without positive airway pressure.(112)
Another randomized comparison of standard medical therapy versus bilevel NIPPV (median inspiratory pressure of 12 cm H2O and median expiratory pressure of 5 cm H2O) reported more rapid resolution of acute severe asthma with a reduction in bronchodilator requirements in those receiving NIPPV.(113)
IPPB enjoyed great popularity for administration of aerosols to patients with COPD; however, it fell out of routine use when it was shown to not improve response to nebulized bronchodilators in a randomized, multicenter study that followed patients over an average of 33 months.(114) In patients with chronic airflow obstruction, high-frequency oscillation reduced pulmonary deposition of radioaerosol, but a larger proportion of total aerosol deposition occurred in the lung periphery.(28)
Despite the much more frequent use of NIPPV in patients with acute respiratory failure due to acute exacerbations of COPD than in patients with acute severe asthma, there are only sporadic reports of aerosol delivery in patients with COPD receiving NIPPV. Significant bronchodilator responses occur after albuterol administration with a pMDI in stable patients with COPD receiving NIPPV with mask.(110,115) Nava and coworkers(115) compared placebo via pMDI and chamber spacer versus 400 μg albuterol with MDI and spacer versus 400 μg of albuterol by pMDI and spacer during NIPPV and 5 mg of albuterol in 5 mL saline given by IPPB in a randomized manner. With the pMDI and spacer, they found a higher change in forced expiratory volume in 1 sec after albuterol administration during NIPPV compared to placebo but the effect was comparable to that obtained with a pMDI and spacer during spontaneous breathing.(115) In another group of eight patients with COPD, the response to 400 μg of albuterol by pMDI and spacer during NIPPV was compared with the effect of placebo at identical settings (to determine the effect of NIPPV alone). Application of NIPPV increased forced vital capacity both after placebo or albuterol administration but forced expiratory volume in 1 sec increased only after albuterol.(115)
In bench models, drug delivery was increased during simulated spontaneous breaths compared to controlled mechanical breaths.(116) However, in ventilated patients with acute exacerbations of COPD, the bronchodilator response did not differ between controlled mechanical ventilation and pressure support ventilation.(117) In a similar population, the administration of nebulized albuterol when extrinsic positive end-expiratory pressure (PEEP) was employed to balance intrinsic PEEP had additive bronchodilator effects compared to zero PEEP, albuterol alone, or extrinsic PEEP alone.(118)
In summary, several investigations suggest that NIPPV and bronchodilator therapy provide added benefits in patients with acute severe asthma or acute exacerbations of COPD. However, definitive studies in larger groups of patients are needed to corroborate these preliminary findings.
During bilevel ventilation in children with cystic fibrosis, pulmonary deposition of radiolabeled aerosol was increased by the application of 10 cm H2O inspiratory pressure support.(119) In stable patients with cystic fibrosis, another group of investigators reported more peripheral deposition of aerosol when a jet nebulizer was employed with positive expiratory pressure.(93) Only a few investigators have studied the role of NIPPV in acutely ill patients with cystic fibrosis,(120,121) but the response to inhaled bronchodilators was not examined in these studies.
Drug toxicity
Most investigators have reported no serious adverse effects following administration of albuterol with a pMDI,(12,31) but a dose-dependent increase in heart rate occurs with higher doses.(73) In ambulatory patients with acute asthma, up to 16 puffs each of albuterol or fenoterol administered with a pMDI and holding chamber connected to a facemask did not produce significant arrhythmias or other serious cardiovascular side effects,(122) but higher doses of beta-agonists delivered by a pMDI can cause atrial and ventricular arrhythmias.(123–125) Continuous nebulization of beta-agonists is effective and safe in nonintubated children(126) and adults(127) with acute severe asthma; but the efficacy and safety of continuous nebulization has not been established in acutely ill patients who are receiving NIPPV. Manthous and coworkers observed sinus tachycardia or supraventricular ectopy after a cumulative dose of 7.5 mg of albuterol administered by a nebulizer in 4 of 10 intubated and mechanically ventilated patients, and most of the remaining patients developed premature atrial and ventricular contractions with a cumulative albuterol dose of 15 mg.(128) Small reductions in arterial oxygen tension may occur transiently after beta-agonist inhalation, probably secondary to pulmonary vasodilation mediated by beta-2 receptors.(129) In contrast, reduction in arterial oxygen tension was either not seen after inhalation of antimuscarinic agents(130) or the fall in arterial oxygen tension PaO2 was smaller than that produced by albuterol and salmeterol.(131)
In patients with severe airway obstruction who are hypoxemic a fall in serum potassium after beta-agonist inhalation(132) has the potential to cause cardiac arrhythmias. A variety of central nervous system effects including, anxiety, nervousness, tremor, irritability, insomnia and headache, are described with use of beta-adrenergic agents.
Technique of Aerosol Administration
Both pMDIs and nebulizers can be adapted for in-line use in the ventilator circuit during NIPPV (Fig. 4). The recommended techniques for using pMDIs and nebulizers during NIPPV are summarized in Tables 5 and 6. However, optimal settings for maximum drug delivery with a pMDI and spacer/facemask or nebulizer/facemask during NIPPV are not well established. For bronchodilator therapy in patients with airflow obstruction, low levels of CPAP ∼5 cm H2O are probably just as effective for drug delivery as higher levels (≥10 cm H2O). A moderate level of inspiratory pressure support (ΔIPAP-EPAP) of 10 to 15 cm H2O seems to give the best results without the risk of delivering higher than normal tidal volumes. Adequate humidification should be ensured to enhance patient comfort and to prevent an increase in nasal and airway resistance due to mucosal dryness and reflex changes caused by breathing cold dry air. Drug doses may need adjustment considering the known effects of humidity in reducing the efficiency of aerosol delivery (Fig. 3).
If a heat and moisture exchanger is employed, it should not be placed between the pMDI and the mouth because it will trap the drug particles.
Preferably a heat and moisture exchanger should not be employed during jet nebulization; if placed between the nebulizer and the mouth it traps the drug particles in the aerosol, and if placed between the ventilator and nebulizer, i.e., away from the mouth, there is the potential for the heat and moisture exchanger's filter to get clogged by water condensate and drug particles in the nebulized aerosol. Clogging of the filter increases its resistance and leads to a greater work of breathing for the patient.
Summary and Conclusion
Delivery of aerosols to patients receiving NIPPV is extraordinarily complex. Several factors related to the type of ventilator employed, the type of aerosol generator and its position in the ventilator circuit, mode of ventilation, circuit humidity, density of the gas in the circuit, type of mask interface, mask size, breathing parameters, inspiratory and expiratory pressure settings, and patient characteristics all influence the efficiency of aerosol delivery in this setting. Moreover, the single circuit design, continuous air flow, and the presence of a leak port further complicate aerosol delivery. Despite all these obstacles, aerosols can be delivered to patients receiving NIPPV with remarkable efficiency provided meticulous attention is given to the technique of administration. Future scientific and technological advancements will surely further improve upon the ability to safely and predictably administer inhaled therapies to patients receiving noninvasive ventilation.
Footnotes
Acknowledgments
The author thanks Diane E. Johnson, Assistant Director, Information Services and Resources in the J Otto Lottes Health Science Library for her assistance in the preparation of this paper.
Author Disclosure Statement
The author declares that no competing financial interests exist.
