Abstract
Abstract
Background:
Delivery of powder formulations to the nose is an attractive alternative for many drugs and vaccines. This study compared the regional nasal deposition and clearance patterns of lactose powder delivered by the OptiNose powder device (Opt-Powder; OptiNose US Inc., Yardley, PA, USA) to that of liquid aerosol administered via a traditional hand-actuated liquid spray pump (Rexam SP270, Rexam Pharma, France).
Methods:
The study was an open-label, crossover design in seven healthy subjects (five females, two males). The regional nasal deposition and clearance patterns of the Opt-Powder device were compared to a traditional liquid spray pump by dynamic gamma camera imaging after administration of either 99mTc-labeled lactose powder or liquid 99mTc- diethelyne triamine pentaacetic acid-aerosol. The gamma camera images were scaled and aligned with sagittal magnetic resonance images to identify nasal regions. Possible deposition of radiolabeled material in the lungs following both methods of delivery was also evaluated.
Results:
Both powder and spray were distributed to all of the nasal regions. The Opt-Powder device, however, achieved significantly larger initial deposition in the upper and middle posterior regions of the nose than spray (upper posterior region; Opt-Powder 18.3%±11.5 vs. Spray 2.4%±1.8, p<0.02; sum of upper and middle posterior regions; Opt-Powder 53.5%±18.5 vs. Spray 15.7%±13.8, p<0.02). The summed initial deposition to the lower anterior and posterior regions for spray was three times higher compared to Opt-Powder (Opt-Powder 17.4%±24.5 vs. Spray 59.4%±18.2, p<0.04). OptiNose powder delivery resulted in more rapid overall nasal clearance. No lung deposition was observed.
Conclusions:
The initial deposition following powder delivery was significantly larger in the ciliated mucosa of the upper and posterior nasal regions, whereas less was deposited in the lower regions. Overall nasal clearance of powder was slower initially, but due to retention in anterior nonciliated regions the overall nasal clearance after spray was slower.
Introduction
Powder formulations are common in pulmonary drug delivery, but are so far rare in nasal products. However, powder formulations may offer a number of advantages over liquid formulations. Powder formulations may be more stable, the need for excipients may be reduced, and clearance from the ciliated regions of the mucosa may be slower.(1) It was against this background that a powder formulation of sumatriptan was developed and tested in placebo-controlled clinical Phase I and II trials using a bidirectional powder device for the treatment of acute mild to moderate migraine.(6,7) This device has shown compelling results in these studies. Scintigraphic techniques have been used to assess nasal deposition and clearance of radiolabeled liquid or powder formulations delivered with a variety of devices.(8–12) However, these techniques were relatively crude and did not allow for reliable absolute or relative quantification of deposition in well-defined nasal regions. An improved system for regional quantification of deposition and clearance was introduced some years ago,(3) and recently this method was further improved by applying magnetic resonance images (MRI) overlay and by correcting for attenuation of photons as they travel through tissues of different thicknesses and densities from the radioactive source in the nose to the gamma camera.(13)
Previous studies comparing a traditional spray pump and a breath actuated bidirectional liquid device incorporating the same spray pump showed significantly improved nasal deposition beyond the nasal valve.(3) The objective of the present study was to compare nasal deposition patterns achieved with a traditional hand actuated spray pump and the breath powered powder device (Opt-Powder) developed by OptiNose (Yardley, PA, USA).
Materials and Methods
The present study was conducted according to the principles of the Declaration of Helsinki and approved by an independent ethics committee, the Regional Committee for Medical Research Ethics, Eastern Norway. Written, informed consent was obtained from subjects before inclusion.
Clinical study design
This was an open-label, crossover study in seven healthy subjects (five females, two males) aged between 20 and 28 years. Only subjects in good health with clear nasal airways, normal nasal examinations, normal nasal dimensions measured using acoustic rhinometry (AR), and a history of good lung function (free of major disease) were included. Subjects with any of the following conditions were excluded: previous nasal surgery causing severe deviation of the septum or blockage of the nasal airways; severe nasal septum deviation; severe nasal or sinus problems within the 2 months prior to the study; inability to use the device effectively; inability to maintain posture due to back or other problems; lactose intolerance; allergy to Technetium (Tc); female subjects who were either pregnant or lactating.
Acoustic rhinometry
Before each study session AR was performed with the subject in the seated position using the anatomic nose adaptors (Rhin200, RhinoMetrics, Lynge, Denmark) to verify normal nasal dimensions and assess potential differences in nasal dimensions between study arms.(14)
Liquid spray
The solution for the spray bottle was made by adding 120 to 150 MBq 99mTcO4 (IFETEC generator, Isopharma, Kjeller, Norway) in 6 mL saline–water to a vial containing freeze-dried diethelyne triamine pentaacetic acid (DTPA). The 99mTc-DTPA solution was then diluted to a concentration suitable to provide a delivered dose of 1 MBq to the last of the subjects studied on the same day. The time span between studies of the first and the last subject was approximately 6 h, which equals the half-life of 99mTc. The range of radioactivity delivered was thus approximately 1.0–2.0 MBq.
Lactose powder
Each capsule was filled with approximately 16 mg of lactose with a mean particle size (MPS=d50) of 15 μm (d10=3 μm, d90=38 μm) according to the data sheet on the lactose used (LH 200, Friesland Foods, Meppels, The Netherlands). The radiolabeling of the lactose was performed at the Isotope laboratories at the Institute for Energy Technology (Kjeller, Norway) and based on the validated procedure described by Karhu et al.(15) for labeling similar lactose carriers with pertechnetate (99mTcO4−). The powder was radiolabeled with 99mTc (99mTcO4−) and dispensed in a lead box with HEPA-filtered air stream using aseptic technique. The “cold” lactose powder emitted from the capsule and device 2–3 weeks after labelling had a d50 of 11 μm (d10=3 μm, d90=30 μm) when measured by laser diffraction (Malvern Spraytec system, Malvern Instruments Ltd., Worcestershire, UK). The estimated amount of radioactivity actually administered from the device into the subject's nose varied between 0.5 and 1.1 MBq.
Administrations
Traditional liquid spray
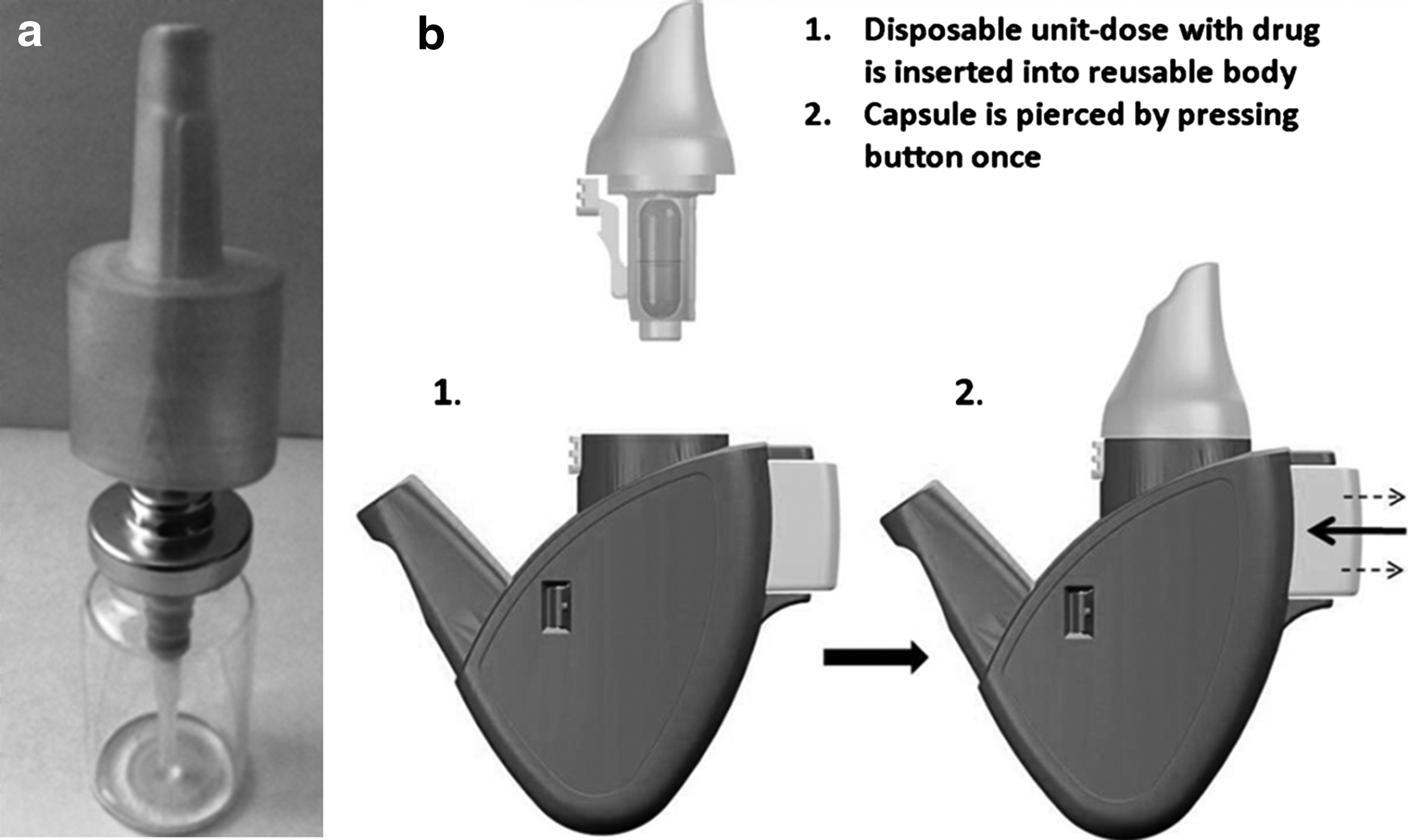
Approximately 3 mL of the 99mTc-DTPA solution was filled into a glass bottle and a traditional multidose nasal spray pump (Rexam SP270, Rexam Pharma, France) delivering 100 μL of 99mTc-labeled DTPA per spray was crimped on top. An applicator manufactured by Sovrin Plastics (Slough, UK) was attached to the pump. A nosepiece (Andersen Industrimodeller, Jessheim, Norway) was fitted outside the applicator to allow proper grip and actuation of the spray pump (Fig. 1a). The ≈3-mL fill would allow the required five to seven priming strokes and reproducible delivery of one spray of 100 μL (well within the maximum dose variability of 15%) to each of the seven subjects. The nosepiece/grip was changed between subjects. Subjects were instructed to hold the spray bottle between the thumb (bottom of bottle) and fingers 2 and 3 and to position the tip of the primed spray device in the vertical position perpendicular to the plane of the nostril some millimetres inside the nostril while tilting the head slightly forward according to written instructions and illustrations from package inserts for commonly used marketed nasal spray products in Norway like Otrivin® (Xylometazoline) and Nasonex® (Mometasone). The subject actuated the device once by pressing the bottle and subsequently slowly inhaled through the nose. The plume angle from this spray was approximately 58°. Laser diffraction analysis using a Malvern Spraytec system (Malvern Instruments Ltd.) showed that the plume mean particle size was 48.5 μm for distilled water.
(
Opt-Powder device
The powder was delivered with an Opt-Powder device (bidirectional delivery) that contained a capsule filled with 99mTc-labeled lactose powder (16±3 mg, range 13–21 mg). The fraction (mean±SD) emitted from the capsule was calculated to be 78±12% based on measurements of radioactivity in the device after delivery. This is similar to the emitted fraction 77±11% calculated from analysis of drug residuals in returned devices in a clinical Phase II study.(7) The device was identical to the one used in previous Phase I and II clinical trials (Fig. 1b) and the bidirectional delivery procedure has recently been described in detail elsewhere.(6,7) In brief, the dry powder was filled into a standard #3 inhalation grade capsule. To operate, the piercing button on the side of the device is pressed. Two steel pins connected to the button pierce the capsule and are retracted when the button is released. The user then inserts the device into his/her nose and mouth, takes a full breath with an open mouth, closes the lips around the mouthpiece, and blows into the device. Due to the sealing nosepiece, the drug-containing airflow expands the narrow nasal passages and penetrates deep into the nose, whereas the closed soft palate prevents lung inhalation. The dynamic pressure balances the pressure across the soft palate to optimize communication behind the septum and the airflow leaves through the other nostril via the small opening in the back of the nose connecting the two nasal passages. Powder delivery occurs within a fraction of a second. The oscillatory movements of the capsule creates the energy to disperse the powder into the flow and creates a characteristic rattling sound which also serves to indicate correct use.
Image acquisition and regional quantification
The MRIs were produced on the Siemens MAGNETOM Espree 1.5T (Siemens, Germany) using a three-dimensional (3D) pulse sequence (magnetization prepared rapid gradient echo imaging) with a voxel size of 1×1×1.3 mm3 and a field of view of 192×256×270 mm3. The acquisitions of images of radioactivity deposited in the nose were carried out on an Argus single-head Gamma-camera (Philips Healthcare, Best, The Netherlands) equipped with a general purpose low-energy collimator. All the static and dynamic images acquired in the study had identical image formats of 128×128 pixels, covering a field of 256×256 mm (pixel size 2.06 mm).
Acquisition of scintigraphic images and methods for regional quantification of the deposits have been described in detail elsewhere(13) and are therefore only outlined here. The subject was seated and positioned relative to the scintillation camera field of view by means of a fixation frame. After nasal administration, a dynamic series of images were acquired with a 2-min frame duration for 32 min (number of images in each series 16) viewing from a lateral direction. Assuming symmetry with respect to the sagittal midplane of the head, the images were corrected for photon attenuation by a pixel-by-pixel calculation of correction factors derived from a lateral transmission image acquired before the delivery of aerosol or powder. Marker images, obtained with a 99mTc line source fixed to a balloon and kept for a short while against the palate as well as with a 99mTc point source held on anatomical landmarks, were used to coregister the scintigraphic images to sagittal sections through the three-dimensional MRI series. The MRIs had been scaled to have a pixel size exactly equal to half the pixel size of the scintigraphic images, and coregistration was done with scintigraphic images rescaled (rebinned) to have 256×256 pixels. Sagittal slices through the MRI volume were used to define the nasal regions used for quantification. The inner nose contour was defined by marking upper and lower limits in a sequence of coronal slices.
Normalization procedure and presentation of the data
The gamma-images were superimposed on the MRI. In the sagittal image through the nasal turbinates, the anterior vertical line approximately at the head of the inferior turbinate and the posterior vertical line at the posterior end of the turbinate were then marked. These anatomical landmarks were chosen as they were easily identified on the sagittal MRIs. The upper and lower limitations of the inner nasal cavity were identified from the inner nasal contour derived from the coronal MRI sections. The region between the olfactory bulb region and nasal floor was then divided into three equal parts based on the measurement of the individual nasal height (Fig. 2) (see Skretting and Djupesland(13)).
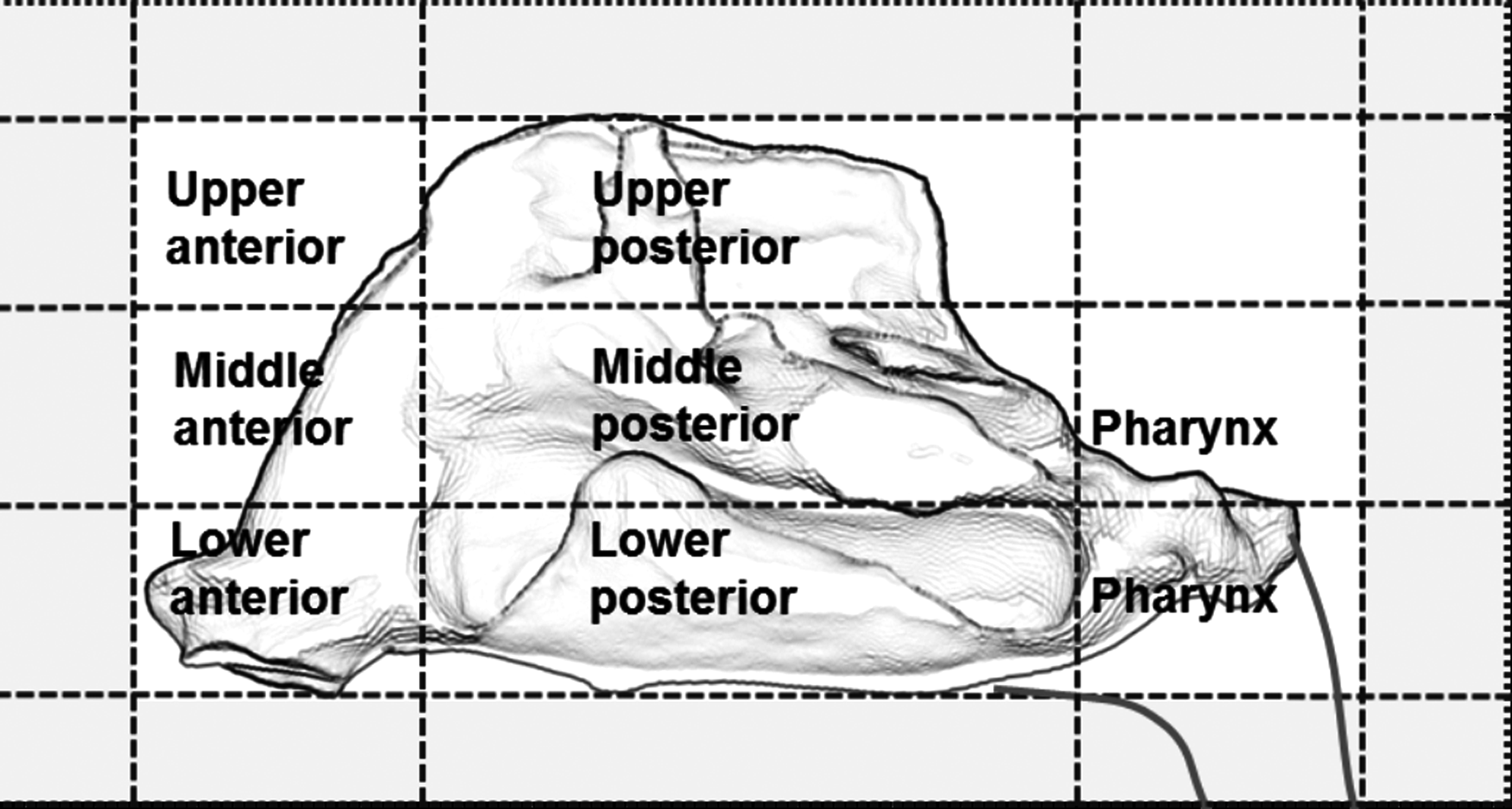
Schematic diagram of the nasal cavity showing the division into the six nasal regions. Counts above the background inside the gray zone are included to take into account scattered radiation from deposition inside the regions of interest.
Normalization to 100,000 counts for the first 120 sec was done for the entire camera field not just the six nasal regions. In cases where there is drip out of the front of the nose or the subject sniffs the liquid to the pharnyx or hypopharnyx during the first 120 sec more of the counts would fall outside the six regions (with the five pixel/10-mm zone to include scattered radiation) (Fig. 2). This results in the amount of normalized counts within the six regions to be below the 100,000 count. All subsequent images were normalized relative to this first image and all values in the analyses conducted reflect normalized values. This normalization significantly facilitated comparison between subjects and the two different devices and formulations. The data are presented as fractions (%) of the initial delivered dose (100,000 normalized counts correspond to 100%).(13) (For details, see Skretting and Djupesland(13).)
Lung images
Immediately after the dynamic study (with associated marker images) had been acquired, the subject was taken to a second scintillation camera (Philips, Argus equipped with a Low energy High sensitivity LEHS collimator for the spray administration and Philips Vertex plus equipped with a general-purpose low-energy collimator, VXGP for the powder administration) and a 5-min image with the subject in the supine position was obtained with a field of view that covered the body from the neck to the stomach. The image format was 256×256 pixels.
Regions of interest (ROI) were drawn on the scintigraphic image outlining approximately the anatomic extension of the lungs (Fig. 3). Lung regions were drawn on the basis of activity in the esophagus and stomach that could usually be seen in the images and knowledge of the normal size and location of the lungs. Since the aim of this part of the study was to detect any lung uptake above the normal background, the regions were in most cases deliberately drawn too large, while ensuring that the entire lung field was included. With the actual camera, collimator and acquisition time an activity of 1 MBq within the image field of view would contribute approximately 65,000 photon counts to the image.
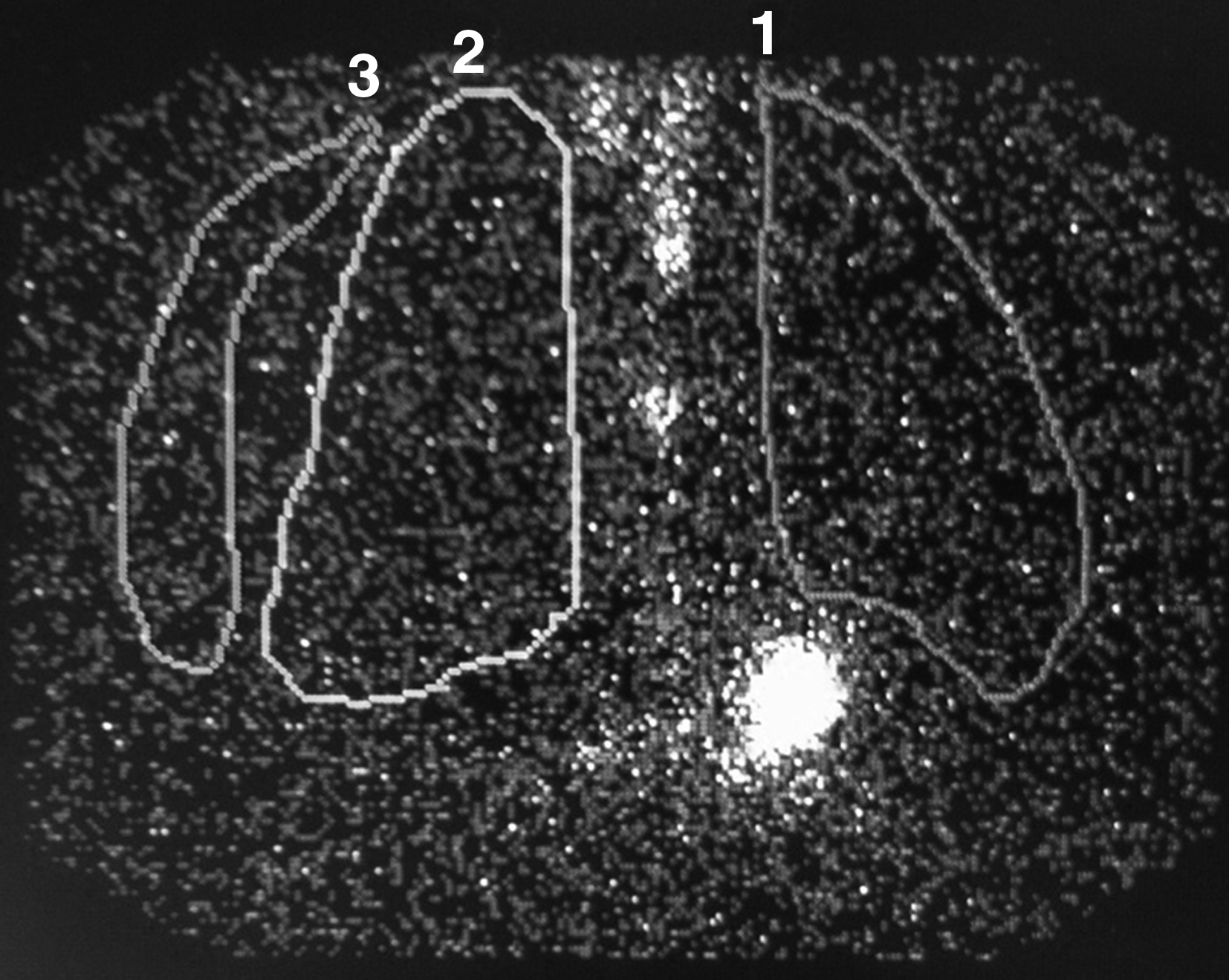
Scintigraphic image of the upper body with regions of interest drawn. 1=left lung; 2=right lung; 3=Area used to measure background radiation. The white area between the lungs is radiolabel in the esophagus and the intense white area just below the middle edge of the left lung is radiolabel in the stomach.
In addition, a background region was drawn outside the right side of the body contour of the subject. The number of counts in each region and the corresponding sizes of the ROIs were recorded. To allow comparison all data were normalized to a region of 1000 pixels.
Statistical analysis
Nonparametric testing with the Wilcoxon signed-rank test for matched pairs was used to compare regional deposition for spray and powder using the GraphPad PRISM 5.0 Software. Statistical significance was accepted for p<0.05.
Results
Both powder and spray were distributed to all of the nasal regions. The initial deposition in the six nasal regions between 0 and 2 min after administration for Opt-Powder and the traditional liquid spray is shown in Figure 4. The sums of deposition in various regions are shown in Table 1.
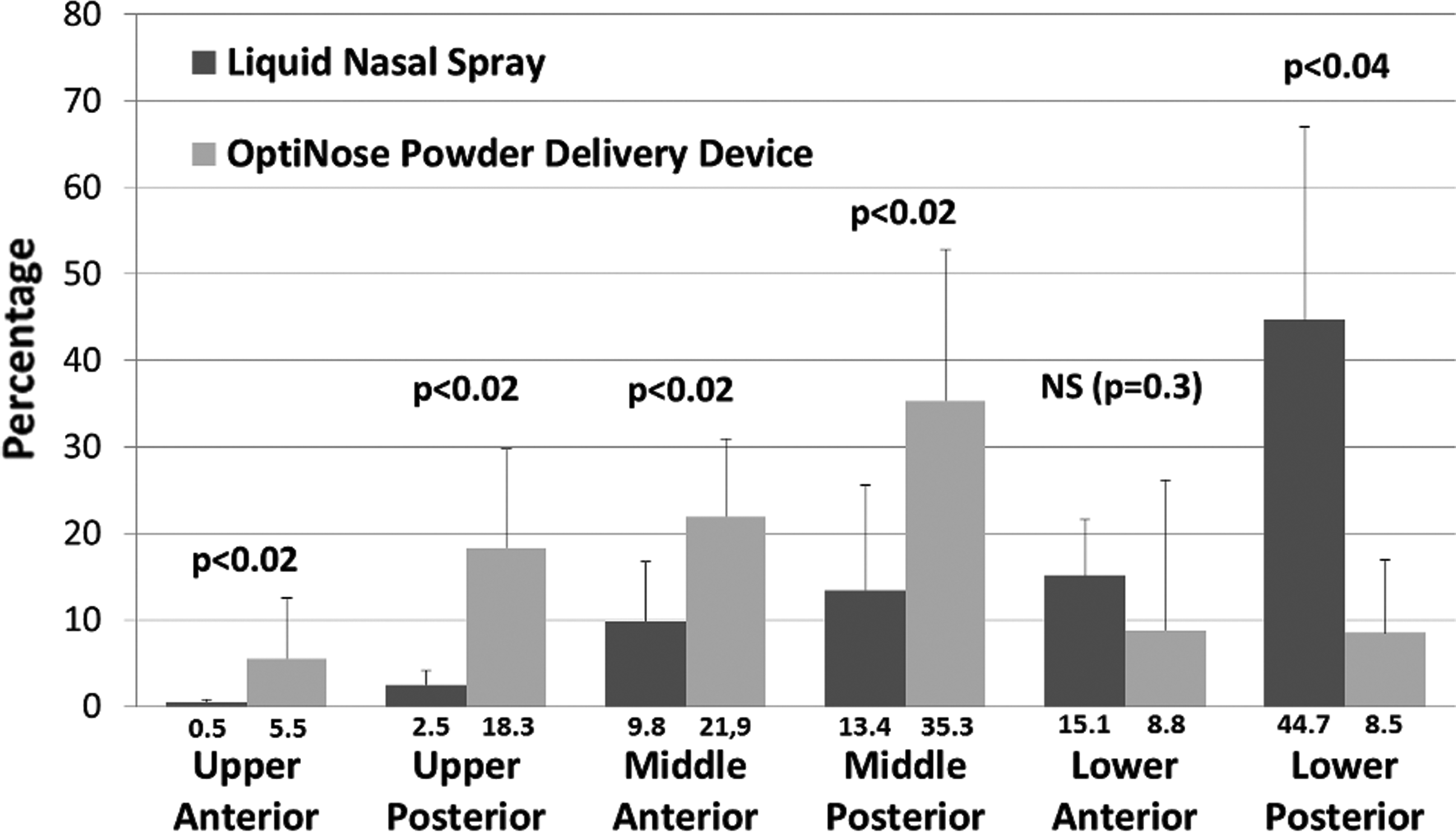
Percent distribution of Powder and Spray by nasal region 0–2 min after administration. Bars are mean±SD.
NS, not statistically significant.
Significantly more of the radioactivity was found in the six nasal regions 2 min after delivery of powder than for spray (Table 1). The main explanation for this difference was loss of drug (radioactivity) to anterior drip-out and to the pharynx within the first 2 min, probably due to spontaneous sniffing to prevent drip-out. With the anterior positioning of the vertical dividing line chosen in the present study (tip of the inferior turbinate), a higher fraction of the initial deposition was found in the summed anterior regions after Opt-Powder compared to the liquid spray (36 vs. 25%). However, this is mainly a result of a greater deposition in the middle and upper anterior regions of the nose after Opt-Powder delivery. In the lower anterior region deposition following spray was higher than after Opt-Powder (Fig. 4).
In the upper posterior region of the nose 18.3% of the initial deposition was observed for Opt-Powder compared with 2.4% for the traditional liquid spray (Fig. 4). A threefold larger deposition was observed when adding the deposition in the upper and middle regions of the nose (Table 1). In the lower regions of the nose, however, a reversed pattern was observed with much lower initial deposition for Opt-Powder compared to liquid spray, indicating that the dose from the liquid spray was delivered predominantly along the floor of the nasal cavity (Table 1 and Fig. 4). These different deposition patterns of the Opt-Powder and traditional liquid spray are illustrated in the example gamma camera images from one of the subjects (Fig. 5).
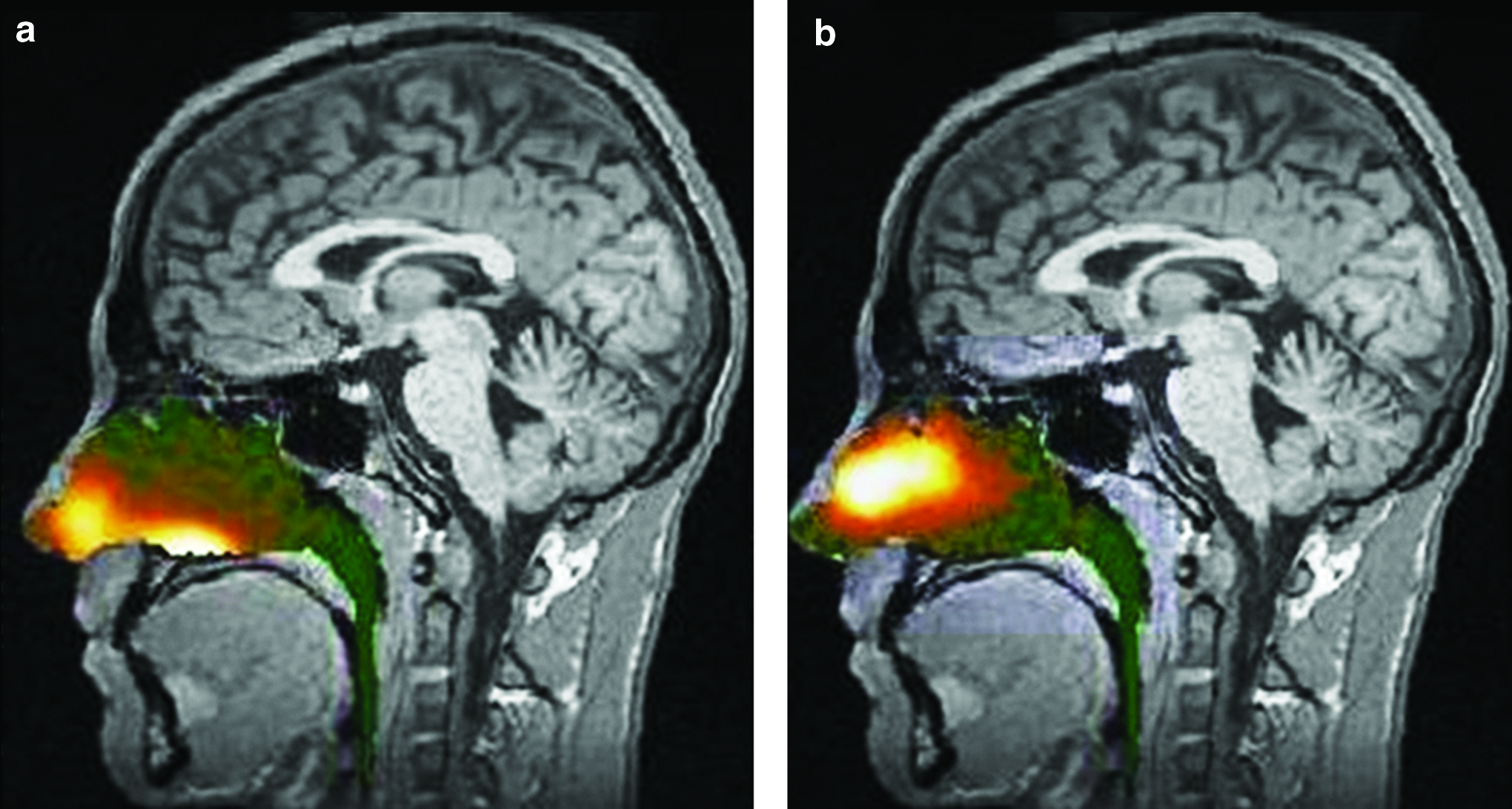
Gamma camera image information (logarithmic hotiron intensity scale) from the nasal cavity is superimposed on the corresponding sagittal MRI section. The image represents deposition 2 min after delivery using a traditional liquid spray (
Total clearance of powder from the nose was slightly slower than liquid for the first few minutes, probably due to the time required to dissolve the powder, but after this time the total clearance of powder was faster than liquid, such that when considering the entire observation period the powder was cleared at a faster overall rate (Fig. 6). The clearance of powder and liquid from the six nasal regions is presented in Figure 7. The patterns of clearance are distinctly different for powder and spray, reflecting the significant differences in the initial deposition. This was particularly notable in the posterior regions, but was also true for the anterior regions where the summed fraction was only reduced from ≈25% to ≈20% over 32 min following standard nasal spray delivery, whereas the fraction was reduced from ≈36% to ≈10% following Opt-Powder.
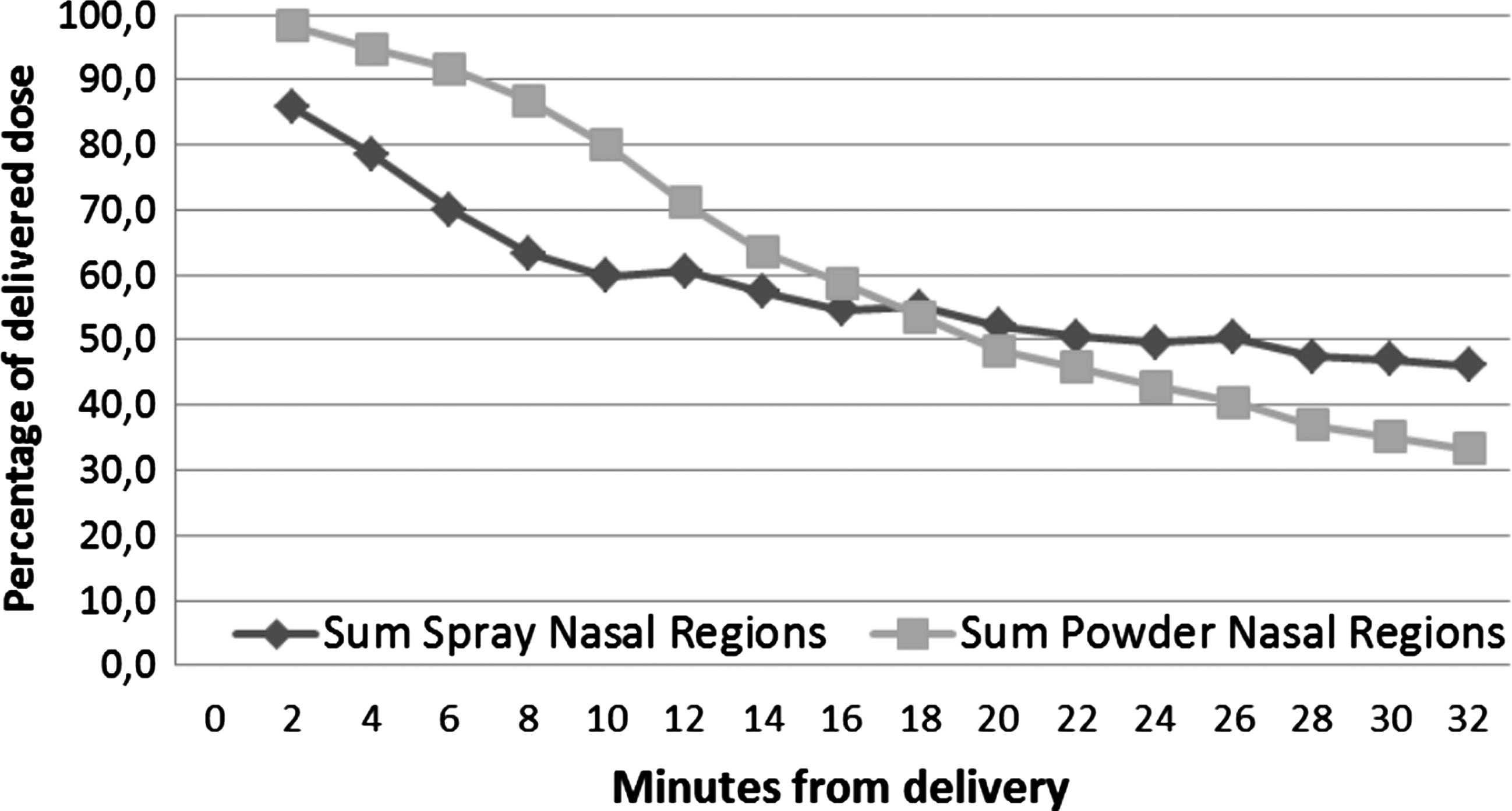
Cumulative clearance of powder and spray from all nasal regions.
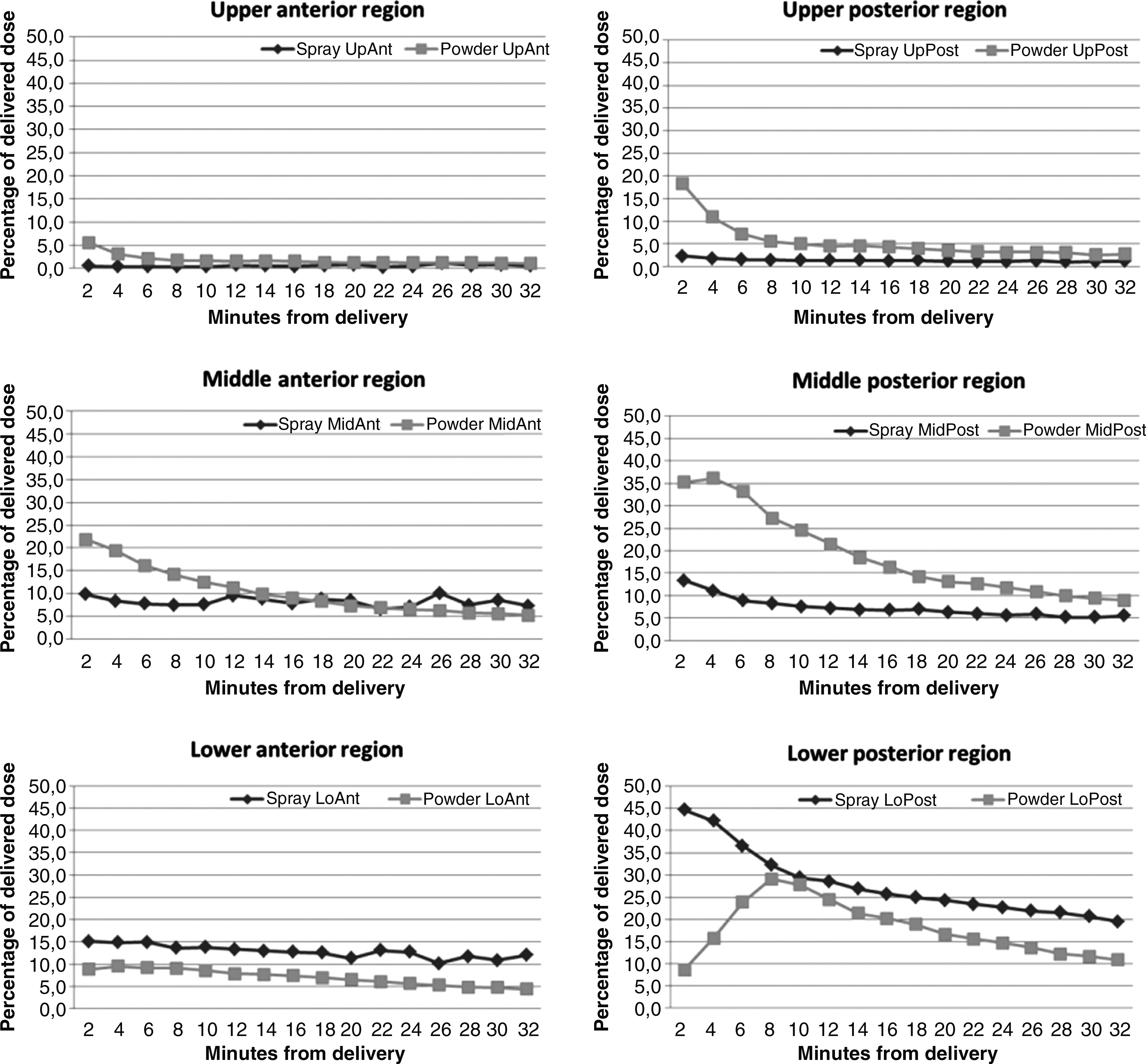
Clearance of powder and spray by region of nasal cavity.
The intersubject reproducibility of initial deposition was higher for Opt-Powder. The fraction and the reproducibility of the emitted dose found in the six nasal regions 2 min after delivery was higher for Opt-Powder than the traditional liquid spray (98.3±0.01%, CV=0.01 vs. 85.8±20.1%, CV=0.23, p<0.04). The reproducibility of delivery to the posterior regions of the nose was higher for Opt-Powder than for the liquid spray (CV=0.25 vs. 0.47). Similarly, the reproducibility of the delivery to the upper and middle posterior regions of the nose was higher for Opt-Powder compared to the liquid spray (CV=0.35 vs. 0.88).
The AR results confirmed that mean MCA and nasal volumes were similar for the subjects prior to administration of the spray and powder (Table 2). Differences in distribution of radioactivity observed in this study were therefore not due to changes in MCA or nasal volumes between the spray and powder study sessions.
Data presented are mean±SD.
MCA, mean cross-sectional area; Vol0–5 right=volume 0–5 cm into the right side of the nose; Vol0–5 left=volume 0–5 cm into the left side of the nose.
There were no major increases in the counts per pixel in the lung fields following either powder or spray administration compared with the very low background counts (Table 3). The counts over the lung regions are unavoidably always affected by scattered radiation due to radioactivity remaining in the nose and pharynx as well as swallowed in the esophagus and stomach. These contributions add to the counts over the lungs, but represent no true increase in lung deposition.
Data presented are mean±SD.
Regions representing the outlines of the right and left lungs (see also Fig. 3).
Discussion
Adequate positioning and operation of the bidirectional powder device was easily achieved by all subjects following the same brief instructions for the powder device as provided to the patients included in the Phase I and II trials where the same device was used.(6,7) The delivery was well tolerated and no adverse events were reported. As previously shown for a bidirectional liquid device,(3) this study shows that bidirectional nasal delivery of a powder offers larger and more reproducible initial deposition to the upper and middle target sites with ciliated nasal mucosa found predominantly beyond the nasal valve when compared directly to a traditional hand-actuated liquid spray pump. In agreement with previous studies,(3,5) there were no signs of lung deposition following bidirectional delivery or traditional spray delivery.
Methodological considerations and limitations
Labeling procedures
The labeling process used was identical to Karhu et al.(15) performed on similar lactose fractions. According to the validation, the particle size of the lactose carrier remained unchanged during the labeling process.(15) The Andersen cascade impactor fractionates only small particles (<10 μm) and is therefore of limited value for the powders used with the Opt-Powder devices. Still, it may be considered a weakness of our study that we did not perform analysis of the “hot” radiolabeled powder using a cascade impactor to validate the uniformity of the labeling process. However, analysis by laser diffraction of the “cold” labeled powder delivered from the device (d10=2 μm, d50=11 μm, d90=31 μm) was similar, but somewhat smaller compared to the data provided by the manufacturer (d10=3 μm, d50=15 μm, d90=38 μm), which suggest that particle agglomeration is not a major issue. Differences in the methods used to label particles and analyze the particle size distribution probably accounts for some of the observed differences, but a more likely explanation is that residuals in the capsule, chamber, and nozzle of the disposable nosepiece are dominated by larger particles. The mean emitted fraction from the powder device of 78±12% is almost identical to the 77±11% reported from chemical analysis of the residual sumatriptan in the returned device in the Phase II clinical trial.(7) offering some support that the lactose powder labeling is fairly uniform.
Dissociation and absorption
There is also uncertainty related to how labeled lactose particles will behave after administration. The differences in labeling procedures for the liquid (99mTc-DTPA) and powder (99mTc pertechnetate) could potentially have an impact on the particle clearance. However, there is no known chemical binding between 99mTc-O4−- or 99mTc- and a lactose particle. Because the particles were thoroughly mixed with the liquid before freeze drying, the result is to cover the surfaces of the particles with a uniform very thin layer of 99mTc. The powder will rapidly dissolve into a solution when it comes in contact with the fluids of the nasal mucosa. It is likely that the lactose and pertechnetate (99mTcO4−) will dissociate and the deposition and clearance patterns may represent a “ghost” image of the pertechnetate rather than the lactose particles. However, this is not a major concern, either for the initial deposition, or for the studies of clearance as long as the absorption of label is limited during the observation time. Any significant absorption to blood vessels would lead to rapid removal of radioactivity from the field of view, which was not observed. Furthermore, if 99mTc-O4−-(eluate) did dissociate from the lactose and was absorbed to any extent into the blood after deposition on the nasal mucosa, radioactivity would have been visible in the thyroid. Such uptake was never seen on the static study carried out just after the dynamic study.
The limited literature available on absorption of labeled 99mTc-DTPA suggests that 16±8% of the amount delivered to the lungs is absorbed within 20 min in healthy nonsmokers.(16) The nasal absorption is likely to be small compared to the pulmonary absorption. The only study on nasal and pulmonary absorption of 99mTc-DTPA we are aware of was carried out in rats and suggests that nasal absorption is three to four times lower than pulmonary absorption.(17) Thus, for all practical purposes it seems reasonable to assume that a maximum of 5–10% of the label is absorbed from the nose within 32 min. The clearance curves showing an increased deposition for Opt-Powder up to 4 min in the middle posterior region and up to 8 min in the lower posterior region, also suggest that drug is moved between regions rather than being absorbed in this time frame (Fig. 7). Finally, although differences in substances and procedures used to label powder (pertechnetate) and liquid (DTPA) could impact clearance and absorption, it is unlikely to influence the initial deposition data.
Alignment and segmentation
The use of MRI overlay, palate contour marking, and regional correction of tissue attenuation(13) offers more individualized, detailed, and accurate quantification of deposition and clearance than in a previous study with a liquid version of the bidirectional device.(3) However, even with the improved methodology for alignment and segmentation employed in this study, it is important to be aware that the quantification of radioactivity in adjacent regions of a gamma camera image is affected by the relative wide point spread function [full width at half maximum (FWHM), is typically 8 mm].(13) This means that a distinct pattern of deposition unavoidably will be smeared out, and a source in one region may thus affect (increase) the counts collected in the neighboring region. The numbers given for regional distribution in this work may thus contain additional small systematic errors that it has not been possible to correct for. Furthermore, the position of the vertical separation between the anterior and posterior regions was determined by the location of the head of the inferior turbinate on a sagittal MRI section,(13) whereas it was placed in a fixed position from the transition to the nasopharynx inferred from measurements by acoustic rhinometry in the previous study with the bidirectional liquid device resulting in a more posterior location in most subjects.(3) In addition, the horizontal plane of the nose was divided into three horizontal segments with a total of six nasal regions(13) compared to two horizontal segments with a total of four nasal regions in the former study.(3) These differences in the methodology and segmentation are less critical for comparison within subjects, but may be critical to the interpretation and comparison of data from different studies and methods. Although the vertical separation is useful in assessing differences in regional deposition and clearance, it does not necessarily describe well the functional transition between regions with nonciliated and ciliated epithelium. This is reflected by the differences in clearance from the middle anterior region following OptiNose powder delivery and liquid spray delivery suggesting that the middle anterior region is partly ciliated and nonciliated (Fig. 7).
Suman and coworkers(18,19) used ventilation of radioactive Xenon gas to delineate the contours of the nasal cavity and divided the nose into three sections in both the vertical and horizontal plane, which is similar but not identical to the segmentation used in our study, and compared ratios of deposition. They found that a nebulizer producing small particles with a MPS of 6 μm provided increased deposition in the upper region of the nose.(18) The downside was a substantial lung deposition of 33 and 56% in the two subjects assessed. Significant lung deposition was also observed in the only in vivo gamma study with a commercial nasal powder inhaler, the novel VibrENT Pari nasal nebulizer, and also reported to be the case with the ViaNase handheld nebulizer.(5,9,20,21)
The key role of the nasal valve
The nasal valve represents a triangular shaped region extending anterior and posterior to the head of the inferior turbinate.(22,23) The region anterior to the valve is lined by nonciliated squamous epithelium that in the valve region gradually transitions into ciliated epithelium typical of the respiratory epithelium posterior to the valve region.(22,23) This narrow slit located 2–3 cm from the nostril opening acts as a dynamic valve to modify the rate and direction of the airflow during respiration.(24,25) The nasal valve represents a substantial challenge for efficient drug delivery to more posterior regions for both traditional spray pumps and nasal inhalers.(2,18) Recently, Suman and coworkers(19) showed that differences between nasal spray pumps based on in vitro tests (recommendations in the U.S. FDA guidance Nasal Spray and Inhalation Solution, Suspension, and Spray Drug Products—Chemistry, Manufacturing, and Controls Documentation) do not lead to differences in the in vivo site of droplet deposition within the nose, or to differences in the rate or extent of drug absorption, or the physiologic response. These results suggest that current measures of in vitro spray pump performance, particularly spray angle and spray pattern for a traditional hand-actuated spray pump, may not be clinically relevant.(19) Clearly, smaller particles have a better chance of bypassing the nasal valve to reach higher up in the nose.(18) However, due to the anterior narrow valve, the plume for a spray pump does not develop properly before the particles impinge on the lateral walls of the anterior nonciliated mucosa or on the head of the inferior turbinate.(11,18,19) Consequently, the mean particle size of a spray plume from a conventional hand actuated spray pump (recorded 2.5 cm from the applicator) seems to have very limited impact on the deposition pattern and physiological responses.(19) This anterior impingement is accentuated during nasal inhalation with powders and liquids creating an anterior “hotspot.”(5,9,11) Sniffing is often performed when spray pumps are used and will further narrow the nasal valve, whereas bidirectional delivery is characterized by both the mechanical expansion of the nasal valve by the anatomical nosepiece and the positive expanding pressure produced by blowing through the device.(3)
Regional deposition and clearance
The bidirectional powder device provides a more even distribution and proportionally larger deposition to the middle and upper parts of the nose, rather than the lower regions. In agreement with the previously published study with a bidirectional liquid device, bidirectional delivery appears to offer significantly improved delivery to the upper posterior nasal regions while reducing exposure to the nonciliated lower anterior region and the lower posterior region compared to traditional spray pumps.(3) In the present study with a traditional liquid nasal spray approximately 50% of the delivered dose was left in the nose after 30 min, whereas only 35% was left following bidirectional delivery of the powder. After delivery with the bidirectional liquid device only 25% was left after 30 min, but 50% was left following traditional spray pump delivery.(3) In Suman's study,(18) approximately 65% of the radioactivity was left in the nose 30 min after the spray pump delivery and 70% for the nebulizer, suggesting that a very large fraction was deposited on the nonciliated mucosa with both devices.
The clearance following powder delivery shows an interesting pattern (Fig. 6) where clearance is slightly slower than for the spray the first 6–8 min, but then speeds up probably reflecting dissolution of the powder into the mucosal layer. The radioactivity in the lower posterior region following powder delivery actually increases substantially during the first 8 min after delivery, mainly due to clearance from the regions above (see Fig. 7). Deposition in the lower anterior region remains stationary following both spray pump and powder delivery. However, in the middle anterior region some degree of clearance is observed following bidirectional powder delivery (see Fig. 7). A similar pattern was observed in the previous study with a bidirectional liquid device,(3) suggesting that the upper anterior region(s) of the nose is partly ciliated and that bidirectional delivery to a larger extent reached these ciliated areas. The exposure following spray delivery is higher and much greater than after powder in the lower anterior and posterior regions throughout the observation period of 32 min (see Fig. 7). This is explained by the substantially greater retention in nonciliated anterior regions and the foremost part of the lower posterior region with nonciliated or transitional mucosa.(22) This difference is likely to increase with time as illustrated by the regional and total clearance curves (see Figs. 6 and 7).
Although rapid clearance could represent a disadvantage for drug absorption, the mucosa of the posterior regions of the nose is more vascularized, and therefore offers a better chance of greater absorption. Bidirectional devices deliver more drug to this highly vascularized respiratory mucosa in the posterior part of the nose. Provided the drug powder dissolves readily it is likely to have a rapid and more extensive absorption than spray that concentrates in the anterior part of the nose. This was observed in a pharmacokinetic study with sumatriptan and OptiNose device where the bioavailability observed was higher than previously reported with similar doses given as a liquid spray.(6) Furthermore, the onset and degree of pain relief during treatment of migraine headaches appears to be enhanced for the sumatriptan nasal powder when compared to published data for nasal sumatriptan liquid spray.(7) In addition, bidirectional delivery of a topically acting steroid with a bidirectional delivery device with a similar deposition and clearance pattern also suggests superior clinical efficacy.(3,26,27)
Conclusions
All six regions of the nose are reached with both devices. The initial deposition following powder delivery was significantly larger in the ciliated upper and middle regions beyond the nasal valve.
Clearance of powder was slightly slower initially, but due to retention in the lower anterior nonciliated regions after spray delivery a considerably larger fraction of the deposited aerosol was still present in the nose at 32 min. The impact of the differences in deposition on absorption and clinical efficacy or safety of different drugs should be assessed in future clinical trials evaluating different delivery devices.
Footnotes
Acknowledgments
This work has been conducted at The Department of Nuclear Medicine, Oslo University Hospital, Oslo, Norway. This article was presented in part at the International Headache Society meeting in Philadelphia, USA, September 2009, and at the European Migraine Trust meeting in Nice October 2010. The study has been supported by the Norwegian Regional Industrial Development Fund, The Norwegian Research Council, and OptiNose AS, a commercial company developing nasal delivery devices. The authors gratefully acknowledge the contributions of Henrik Bruun Andersen, Trond Holand for the conduct of the study, and Graeme Hewson for his valuable assistance in preparing the manuscript.
Author Disclosure Statement
P.G. Djupesland is a founder and shareholder of OptiNose AS, a commercial company developing nasal delivery devices. A. Skretting has no commercial interests in OptiNose AS.
