Abstract
Abstract
Background:
Inhaler technique and spray characteristics are critical for adequate management of asthma symptoms with pressurized metered-dose inhalers (pMDIs). A lower spray force has been directly associated with a decrease in throat deposition of asthma medication, and a higher spray temperature may alleviate the “cold Freon effect” associated with pMDIs. The objective of the study was to characterize and compare the temperature, maximum spray force, and duration of the emitted plume from two pMDIs: ProAir® hydrofluoroalkane (HFA) and Ventolin® HFA.
Methods:
A spray force tester model SFT1000 and thermocouple were used to test 10 units from three separate lots (total of 30 units) of each inhaler type. Three consecutive actuations were tested at a spray distance of 40 mm from the edge of the mouthpiece. Room temperature, humidity, and initial weight of the pMDI were recorded. Final weight of each pMDI was recorded to determine the spray weight of individual actuations. pMDIs were primed and operated according to instructions provided in the package insert. Aerodynamic particle size distribution (APSD) was also assessed using a next-generation impactor at a flow rate of 28.3 L/min.
Results:
Measurements were obtained from three consecutive actuations for each of 30 units of ProAir® HFA and Ventolin® HFA (10 units from three separate lots), resulting in a total of 90 actuations tested for each pMDI. Minimum plume temperatures recorded were 7.2±0.7°C and −35.9±12.7°C, respectively, for ProAir® HFA and Ventolin® HFA. ProAir® HFA produced more than a twofold greater plume duration (385±46 ms vs. 156±58 ms; p<0.001) and a significantly lower mean maximum spray force (33.6±11.4 mN vs. 75.9±12.0 mN; p<0.0001) compared with Ventolin® HFA. APSD analysis demonstrated that ProAir® HFA produced almost twice as much fine particle (<5 μm) dose with lower geometric standard deviation, compared with Ventolin® HFA. Two inhalers produced similar mass median aerodynamic diameters, ranging from 2.3 to 2.4 μm.
Conclusions:
The ProAir® HFA delivers a warmer, lower-impact, and longer-lasting plume compared with Ventolin® HFA, which may provide a more consistent, comfortable experience for patients using a pMDI. ProAir® HFA produces higher fine particle dose than Ventolin® HFA.
Introduction
Because the inhalation route delivers the active drug directly to the site of action (i.e., the lung) in patients with asthma and may minimize the side effects associated with systemic exposure,(4) inhaled antiasthma agents are a crucial component of asthma therapy. However, consideration of inhaler technique and spray characteristics of the delivery system [such as a pressurized metered dose inhaler (pMDI)] is critical for optimizing the delivery of drug to the lungs and thereby providing adequate control of asthma symptoms.(1,5)
Product characteristics are also important for maximizing patient satisfaction.(6) This has implications for compliance with inhaler medications because there are differences in spray characteristics that may influence patients' attitude toward the use of the medication. A higher spray force has been directly associated with an increase in throat deposition of asthma medication, and the lower spray temperature (the “cold Freon effect”) associated with some pMDIs may not be tolerated by some patients.(4,7,8) Furthermore, patients may stop inhaling prematurely due to the cold Freon effect, which may result in reduced deposition of medication within the lungs. Such characteristics may decrease patient satisfaction, which is an important determining factor in whether a patient persists with a medication.(9) Thus, a pMDI that delivers a warmer, longer lasting plume with a lower impact on the throat may improve the patient experience in using pMDIs, thereby improving compliance. The objective of this study was to characterize the temperature of the emitted plumes and maximum spray force from ProAir® hydrofluoroalkane (HFA) (Teva Specialty Pharmaceuticals LLC, Horsham, PA, USA) and Ventolin® HFA (GlaxoSmithKline, Research Triangle Park, NC, USA).
Methods
Apparatus
A schematic diagram of the experimental model is presented in Figure 1. Equipment included a spray force tester (model SFT1000, Mecmesin Advanced Gauge-AFG2.5N with accompanying printer), a data acquisition system (Omega OMB-DAQ-3005; compressive force measurement only), and calibrated CO1 Chromel probes (compressive force measurement only). The test format for the temperature and compressive force measurements is summarized in Table 1. The equipment and apparatus used to perform the force and temperature measurements were calibrated per relevant standard operating procedures to ensure consistent results were obtained.
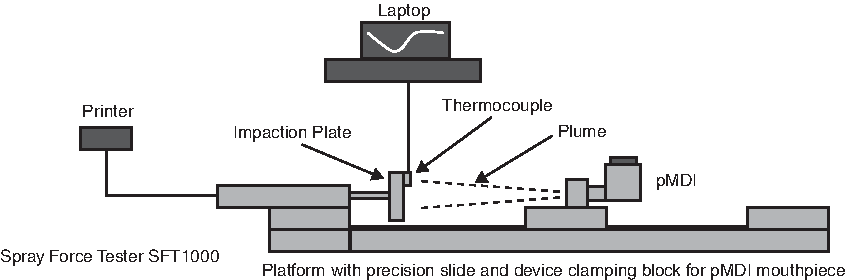
Schematic diagram of experimental setup.
Ten units from three lots for a total of 30 units were tested.
Temperature measurement of emitted plume
Plume temperature measurements were performed over 3 days. The temperature range was 16.8 to 18.8°C. The relative humidity range was 19.3 to 25.2%. Each pMDI was weighed, shaken, and properly placed in a clean, dry actuator, and primed according to package insert instructions. The actuator was then placed in the correct base of the spray force tester at a distance of 40 mm from the transducer panel. The software program was started and allowed to run for 30 s. The pMDI was actuated once and the temperature was recorded as a function of time and printed. The software program was allowed to run for 1 min before stopping for the probe to return to room temperature. Data were saved and the pMDI was weighed for calculation of the weight of the individual actuation. After each actuation, the transducer panel and thermocouple were wiped with an appropriate solvent and 1 min was again allowed to pass for the temperature probe to return to room temperature. These steps were repeated three times for each pMDI.
Maximum compressive force measurement
Each pMDI was weighed, shaken, and properly placed in a clean, dry actuator, and primed according to package insert instructions. The actuator was then placed in the correct base of the spray force tester at a distance of 40 mm from the transducer panel. The software program was started and allowed to run for 30 s. The pMDI was actuated once and the maximum compressive force was recorded and printed. Data were saved and the pMDI was weighed for calculation of the weight of the individual actuation. After each actuation, the transducer panel was wiped with an appropriate solvent and at least 1 min was allowed to pass for canister temperature equilibration. These steps were repeated three times for each pMDI.
Aerodynamic particle size distribution
All measurements to determine aerodynamic particle size distribution (APSD) were conducted using a next-generation impactor (NGI). Ten actuations were fired into the NGI at a flow rate of 28.3 L/min and flow volume of 4 L. Drug deposited on stages 1 to 7 and on the microorifice collector was recovered individually by adding 20 mL 0.01 N hydrochloric acid to each sample cup; the cups were then shaken on a gentle rocker for 10 min. Drug deposited on the induction port and mouthpiece adaptor was also recovered using 20 mL 0.01 N hydrochloric acid. The samples were finally analyzed using a validated high-performance liquid chromatography (HPLC) method. The HPLC column was Spherisorb ODS 2.5 μm, 250 mm long with a 4.6-mm internal diameter. The mobile phase was a mixture of 200 mL acetonitrile (HPLC grade) and 2 mL orthophosphoric acid (85%) and 1 mL diethylamine, diluted to 2 L with Milli-Q water. The sample injection volume was 50 μL; mobile phase flow rate was 1.5 mL/min; and ultraviolet detection wavelength was 225 nm.
Statistical analyses
Mean (±SD) values for minimum and maximum temperatures, plume duration, and spray force were calculated. p-Values were calculated using a Student two-tailed t-test at 95% confidence interval.
Results
Measurements were obtained from three consecutive actuations for each of 30 units of ProAir® HFA and Ventolin® HFA (10 units from three separate lots), resulting in a total of 90 actuations tested for each pMDI. The weight corresponding to each actuation is summarized in Table 2.
Results of the temperature studies are summarized in Figure 2. Figure 2A presents a comparison of representative plume temperature profiles for ProAir HFA® and Ventolin HFA. Mean minimum plume temperatures recorded were significantly higher for ProAir® HFA than for Ventolin® HFA (7.2±0.7°C vs. −35.9±12.7°C, respectively; p<0.001) (Fig. 2B). Mean maximum plume temperatures for the two products were 18.23±0.67°C and 19.02±0.58°C, respectively, with the difference reaching statistical significance (p<0.001).
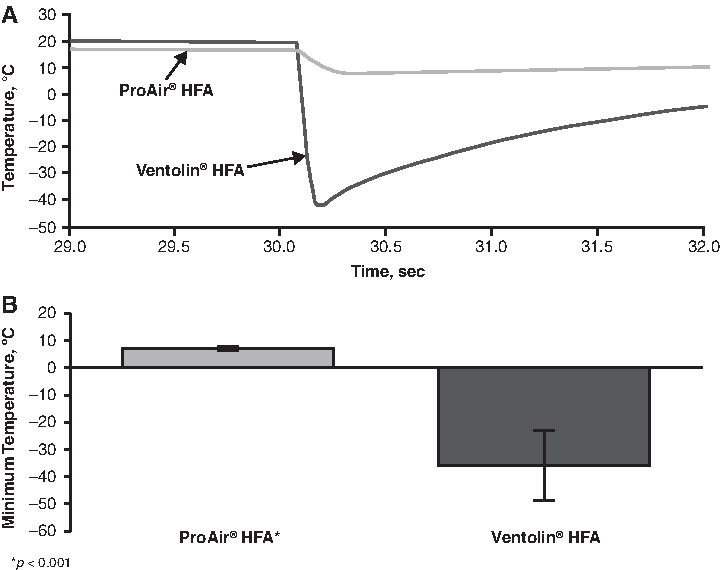
(
Compared with Ventolin® HFA, ProAir® HFA produced 2.5 times the plume duration (385±46 ms vs. 156±58 ms; p<0.001) (Fig. 3). In the spray force studies, ProAir® HFA produced a mean maximum spray force of 33.6±11.4 mN, which was significantly lower (55%) than that of Ventolin® HFA (75.9±12.0 mN; p<0.0001) (Fig. 4).
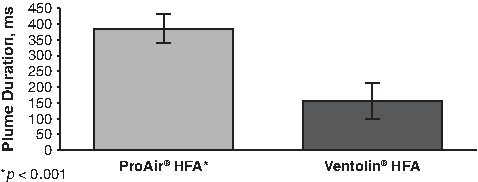
Comparison of plume durations.
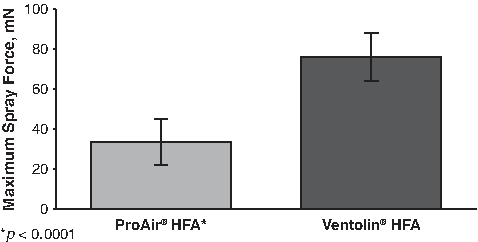
Comparison of maximum compression force measurements for ProAir® HFA and Ventolin® HFA.
To assess whether these plume data correlate with aerodynamic properties of drug particles, APSDs of albuterol from two inhalers were measured using a standard method. As shown in Table 3, ProAir® HFA produced almost twice as much fine particle dose (FPD) as Ventolin® HFA. Mass median aerodynamic diameter (MMAD) was similar between the two products, whereas geometric standard deviation (GSD) was smaller for ProAir® HFA than for Ventolin® HFA.
FPD, fine particle dose (<5 μm); FPF, fine particle fraction (FPD expressed as percentage of total exactuator dose); GSD, geometric standard deviation; MMAD, mass median aerodynamic diameter.
Discussion
Before the introduction of HFAs in the mid-1990s, chlorofluorocarbons (such as Freon®) were used as the propellants for pMDIs. However, chlorofluorocarbons are not considered environmentally friendly because of their potential effect on ozone depletion.(4) Another disadvantage of chlorofluorocarbon pMDI sprays is that they were associated with a high impaction force and a very cold plume temperature (<0°C).(10) The environmental concerns led to the replacement of chlorofluorocarbons by HFAs, which have similar chemical and physical properties.(4) However, because they lack a chlorine molecule, HFAs are more readily broken down in the environment and thus have no potential to deplete the ozone layer.(11) Toxicology studies have also demonstrated that HFAs have no discernible acute or long-term toxicity.(12)
Results from this study demonstrate that ProAir® HFA has a less forceful spray impact (by 55%), a warmer plume temperature (by >43°C), and longer plume duration (by 2.5 times) compared with Ventolin® HFA. There are differences between the two pMDIs evaluated in this study that may explain these results. Although ProAir® HFA and Ventolin® HFA both contain microcrystalline suspensions of albuterol and the propellant HFA-134a, ProAir® HFA contains ethanol, whereas Ventolin® HFA does not.(13,14) There are also differences in the characteristics of the actuators in these products. These factors may contribute to the softer, warmer, and longer lasting plume of ProAir® HFA compared with Ventolin® HFA. In fact, the plume duration for ProAir® HFA is longer than any of the pMDI products (press-and-breathe products bronchodilators) presented in the manuscript by Gabrio and colleagues.(7)
In addition to the softer, warmer, and longer lasting plume, ProAir® HFA also produced higher FPD than Ventolin® HFA, suggesting that ProAir® HFA is more efficient in generating respirable dose than Ventolin® HFA. The fact that ProAir® HFA and Ventolin® HFA produced similar MMAD values, ranging from 2.3 to 2.4 μm, suggests that two inhalers might deliver the drug particles predominantly to bronchi and bronchioles to exert bronchodilatory effect. ProAir® HFA was associated with a lower GSD than Ventolin® HFA, suggesting that aerosolized albuterol particles from ProAir® HFA had a narrower size distribution that was more focused on its MMAD, compared with those of Ventolin® HFA. Therefore, aerosolized particles from ProAir® HFA might be more likely to be delivered to bronchi and bronchioles because more ProAir® particles have aerodynamic diameters between 2 and 5 μm, compared with Ventolin® HFA particles.
Optimally, the majority of an administered aerosolized dose is delivered to the lungs while minimizing deposition in the mouth and throat. Several factors contribute to variability in dosing with inhaled products, with the pattern of deposition determined by the interrelationship between the patient, the formulation, and the delivery device.(15) Patient-related factors include mouth/throat geometry (wide variances are reported) and the patient's inhalation technique.(16) Each of the available types of inhalers requires specific inhalation technique for optimal drug delivery. A less than optimal technique can result in decreased drug delivery and potentially reduced efficacy.(17) Physicochemical properties of the formulation and design of the actuator also influence performance. In particular, large droplet size and high spray velocity are associated with most of the dose being deposited in the oropharynx.(10) Some devices use a solution rather than a suspension, resulting in the emission of ultrafine particles, which may allow the drug dose to be halved.(18) The use of HFA as a propellant has also been shown to decrease the aerosol particle size.(11,19) Spray patterns produced by pMDIs are also significantly influenced by the physical design of the device. Spray pattern analysis has demonstrated that orifice size, orifice length, and expansion chamber (sump) depth have a significant influence on spray pattern measurements.(20)
Given that spray characteristics are a crucial determinant of optimal dose delivery, there has been an effort toward designing efficient pMDI systems, including efforts to produce softer and warmer sprays. In vitro studies suggest that pMDI systems can be designed to minimize spray force and maximize plume temperature.(7) In these studies, spray force was found to be highly sensitive to orifice diameter; spray force increased significantly with increasing actuator orifice diameter.(7) Throat deposition of drug also increased significantly as the orifice diameter increased from 0.29 to 0.40 mm, whereas above 0.48 mm, throat deposition decreased slightly.(7) These studies also found a correlation between spray force and throat deposition; deposition was lowest for pMDIs with the lowest spray force. Throat deposition reached a plateau at a spray force around 60 mN and may decrease for pMDIs with higher spray forces.(7) Valve size was not found to impact spray force.
Increasing the generation time of spray aerosol has also been shown to reduce throat deposition.(16) This is based on the principle that spray turbulence (which is associated with throat deposition) is greatest during the early stages of aerosol generation.(16) Increasing the generation time is associated with temporal dissipation and a minimization of the initial “burst effect,” which arises from the initial entrance of the spray aerosol into the mouth and is associated with high momentum and turbulence and increased mouth/throat deposition.(16) These data suggest that newer pMDIs with actuators that deliver a lower impact and longer lasting plume have the potential to limit oropharyngeal deposition.
The development of new pMDI systems that deliver warmer, lower-impact sprays also has the potential to improve patient acceptability and treatment compliance by providing a more comfortable experience. The high-velocity, cold, rapidly moving, and short-duration spray associated with CFC pMDIs appears to contribute to errors in inhaler technique and poor patient acceptance.(7,10) In contrast, softer and warmer sprays (influenced by lower impaction forces and higher plume temperatures to minimize the cold Freon effect) may result in better patient experience so that they may be less likely to discontinue treatment.(10)
The major weakness of the current study is that it is not a “real world” design. The in vitro design of this study does not account for differences in patient technique or handling—potential issues to consider when utilizing an actuator. Additionally, the devices studied were not exposed to typical patient handling, storage, use, and washing. Thus, this study represents a “best case scenario” for patients' technique and inhaler use.
In summary, results from this study demonstrate that ProAir® HFA delivers a warmer, lower-impact, and longer-lasting plume containing a higher FPD compared with Ventolin® HFA. These characteristics suggest that the product may provide a more consistent, comfortable experience for patients using a pMDI, particularly for patients who do not tolerate the cold, forceful blast associated with some formulations. In addition, compared with Ventolin® HFA, ProAir® HFA more efficiently generates a respirable dose.
Footnotes
Acknowledgments
The authors thank Next Breath, LLC, for their assistance. Editorial assistance was provided by ApotheCom.
Author Disclosure Statement
All authors are employees of Teva Branded Pharmaceutical Products R&D, Inc., and have no other financial interests or conflicts of interest.
