Abstract
Abstract
Background:
To investigate the pharmacokinetics of single dose morphine inhaled by modified ultrasonic nebulizer versus intravenous administered in ventilated dogs.
Methods:
Six healthy dogs were randomly assigned to receive nebulized or intravenous morphine and crossed over to the alternative medication 1 week after. Morphine was nebulized by modified ultrasonic nebulizer (YuYue 402A, Jiangsu, China). Arterial blood was sampled every minute during the 10 min of administration and at 2, 5, 7, 10, 15, 20, 45, 60, 90, 120, 150, 180, 240, and 360 min after the administration for the determination of morphine concentration by RP-HPLC.
Results:
The main pharmacokinetic parameters of morphine by inhaled and intravenous administration were: MRT 59±14 min versus 19±4 min, T1/2 21.9±5.1 min versus 3.3±1.0 min, Tmax 23.0±2.7 min versus 8.8±2.4 min, Cmax 0.245±0.09 mg·L−1 versus 1.09±0.32 mg·L−1, AUC0−∞ 9.7±1.1 mg·min·L−1 versus 15.2±7.2 mg·min·L−1, CL 0.069±0.019 L·min−1·kg−1 versus 0.063±0.028 L·min−1·kg−1, and the absolute bioavailability of inhaled morphine was 35.5±10%. There were no significant differences (p<0.05) between inhaled and intravenous morphine in AUC and CL. As expected, the T1/2 and MRT of inhaled morphine were significantly greater than those of intravenous morphine.
Conclusions:
Morphine nebulized by ultrasonic nebulizer can be rapidly and extensively absorbed by lungs in ventilated dogs.
Introduction
Materials and Methods
Modification of an ultrasonic nebulizer
The original ultrasonic nebulizer (YuYue 402A, Jiangsu, China) (Fig. 1) was an open system designed for patients with spontaneous inspiration. We had the ultrasonic nebulizer modified in three steps. First, cancel the original inlet and rebuild the connection of the upper part and lower part of nebulization chamber into a screw to contain the whole system. Second, modify the two gas vents to the middle top of the nebulization chamber, one as a fresh air entrance to the ventilator and the other as a mixed air exit to the therapeutic subjects. Third, separate the upper nebulization chamber along anteroposterior axes to isolate fresh air and mixed air. As a result, when fresh air came into the upper nebulization chamber, it would go to the lower part and carry the aerosol nebulized by the nebulizing film at the bottom to the other part of the upper nebulization chamber and then exit to the therapeutic subjects (Fig. 2).
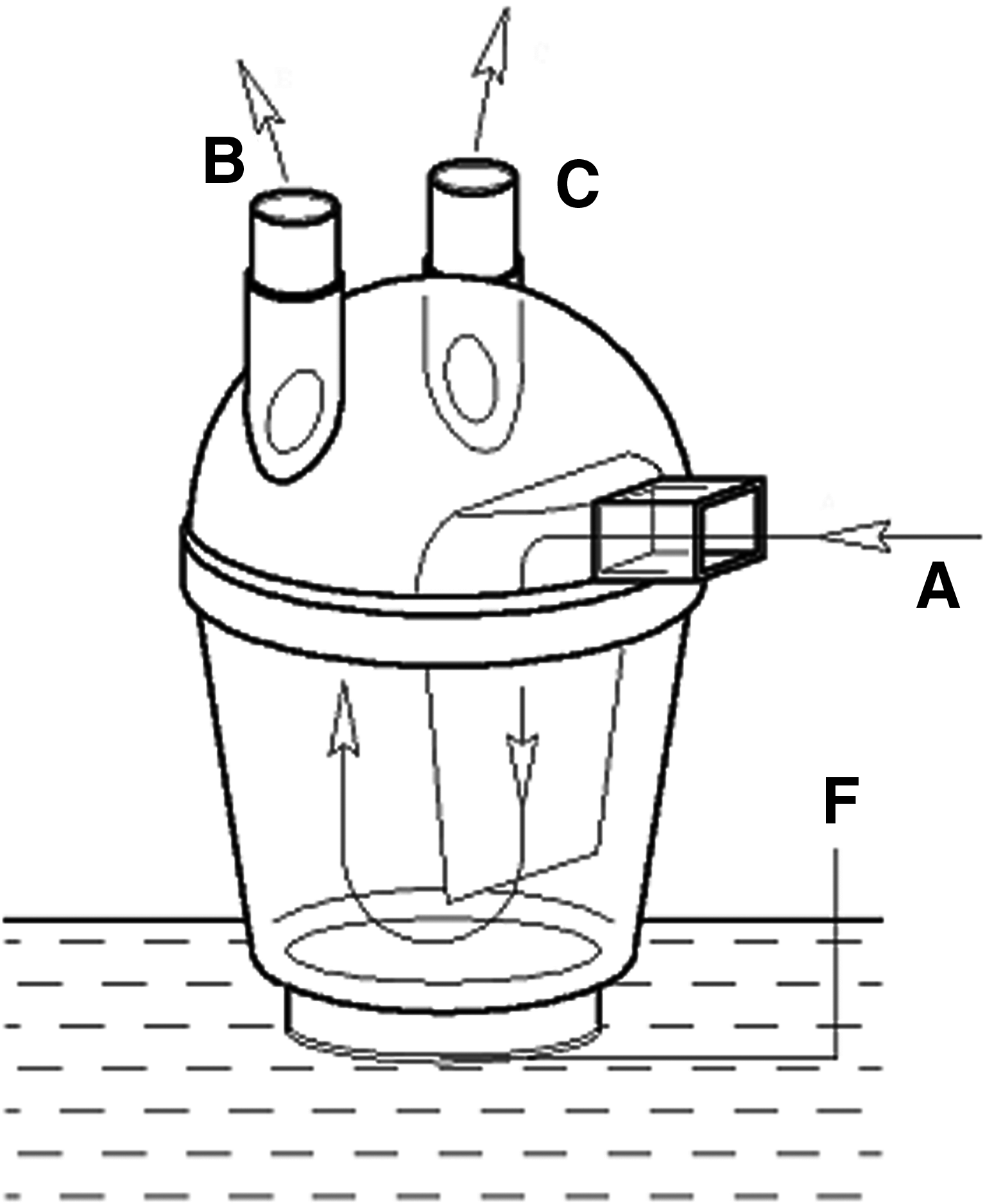
The original nebulization cup (A, inlet; B, C, air vent; F, nebulizing film).
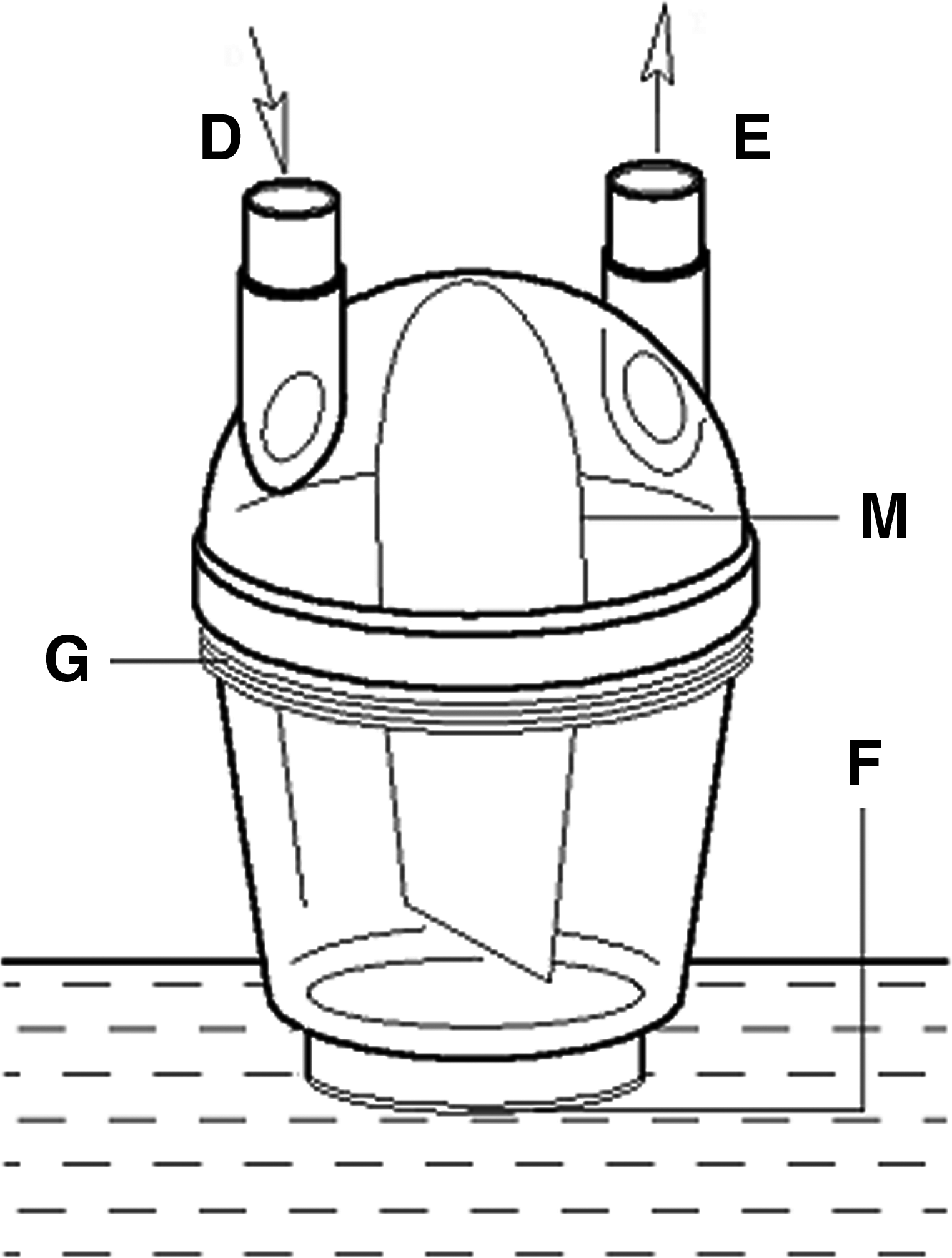
The reformed nebulization cup (D, fresh air entrance; E, mixed air exit; F, nebulizing film; G, screw; M, intermediate partition).
Subject selection and grouping
The study protocol was approved by the institutional ethics committee boards of our hospital. Six healthy dogs (three female, three male, age 2–3 years, weight 10.1±1.7 kg) were recruited and randomly assigned to receive either a single dose of 1.32 mg/kg morphine within 10 min by pulmonary delivery using the modified ultrasonic nebulizer (the inhalation group) or a single dose of 0.66 mg/kg by intravenous administration using the microinfusion pump (the intravenous group) during mechanical ventilation. After a 1- week washout period, a crossover medication was made by the alternative administration to each dog.
The dose was doubled in the inhalation group because in our previous study we found that there was always 3 mL residual after the 10 min of nebulization, and in this study morphine was diluted into 6 mL to load to the nebulizer.
Anesthesia and administration
After fasting 12 h, the subjects were anesthetized with 10–15 mg·kg−1 of ketamine, 0.5 mg of atropine, and 10 mg of droperidol intramuscularly, and intravenous transfusion was then started at a rate of 200–250 mL/h from left forearm vein. Mechanical ventilation was applied after tracheal intubation (Drager Ventilator, Savina, Germany). The ventilation parameters were set as follows: tidal volume 13–15 mL·kg−1, respiratory rate 18·min−1 with an inspiratory time 1.1 sec, 29% FiO2 at a flow rate of 2 L·min−1. The anesthesia was maintained with intravenous administration of propofol at 200–300 μg·kg−1·min−1 and intermittent i.v. of ketamine 3∼4 mg·kg−1 when needed. The right femoral artery was cannulated with a 20-gauge catheter connected to a three-way stopcock for serial blood sampling and blood pressure monitoring. Half an hour after the induction of anesthesia, when arterial blood pressure got steady, all subjects in the intravenous group received a single dose of 0.66 mg/kg morphine (morphine solution diluted to 6 mL with 0.9% NaCl solution) administered by microinfusion pump (Graseby 3100 pump) within 10 min, while a single dose of 1.32 mg/kg in 6 mL was loaded to the ultrasonic nebulizer to deliver intratracheally in the inhalation group instead.
Meanwhile, one simulation test was carried out where one group of 10 samples of morphine solution was nebulized under the same condition as in the inhalation group, and the morphine concentration was detected before and after the 10 min of nebulization to determine the real dose that nebulized and delivered.
Samples collection and HPLC analysis
Arterial blood of 1 mL was sampled every minute during the 10 min of administration of morphine, and at 2, 5, 7, 10, 15, 20, 45, 60, 90, 120, 150, 180, 240, and 360 min after the administration in heparined tubes. All blood samples were centrifugated at 3500 rpm−1 for 10 min to get plasma, which were then stored at −20°C until analysis.
The plasma samples of 100 μL were first mixed well with 5 μL of internal standard solution (ferulic acid 2 mg/L in methanol), then 100 μL of 50 mmol/L phosphate buffer and 700 μL of ethyl acetate (EtOAc) were added. After vortex-mixing and centrifugation, the supernatant was dried under nitrogen evaporation at 40°C. The residue was dissolved in HPLC mobile phase, of which 20 μL was analyzed by reverse-phase high performance liquid chromatography (RP-HPLC). The RP-HPLC system consisted of an Agilent 1100 binary pump system equipped with a G1314 UV-detector. The HPLC mobile phase was 0.2% triethylamine solution (pH 6.89), methanol (78:22, v/v) with the velocity of 1.0 mL·min−1. Chromatographic separation was achieved on a Kromasil C18 column (150 mm×4.6 mm, 5 μm) (Hanbang, Jiangsu) with the detection wavelength at 215 nm. A complete separation was obtained under these chromatographic condition with retention times for morphine and internal standard of 11.890 min and 3.67 min (Fig. 3).
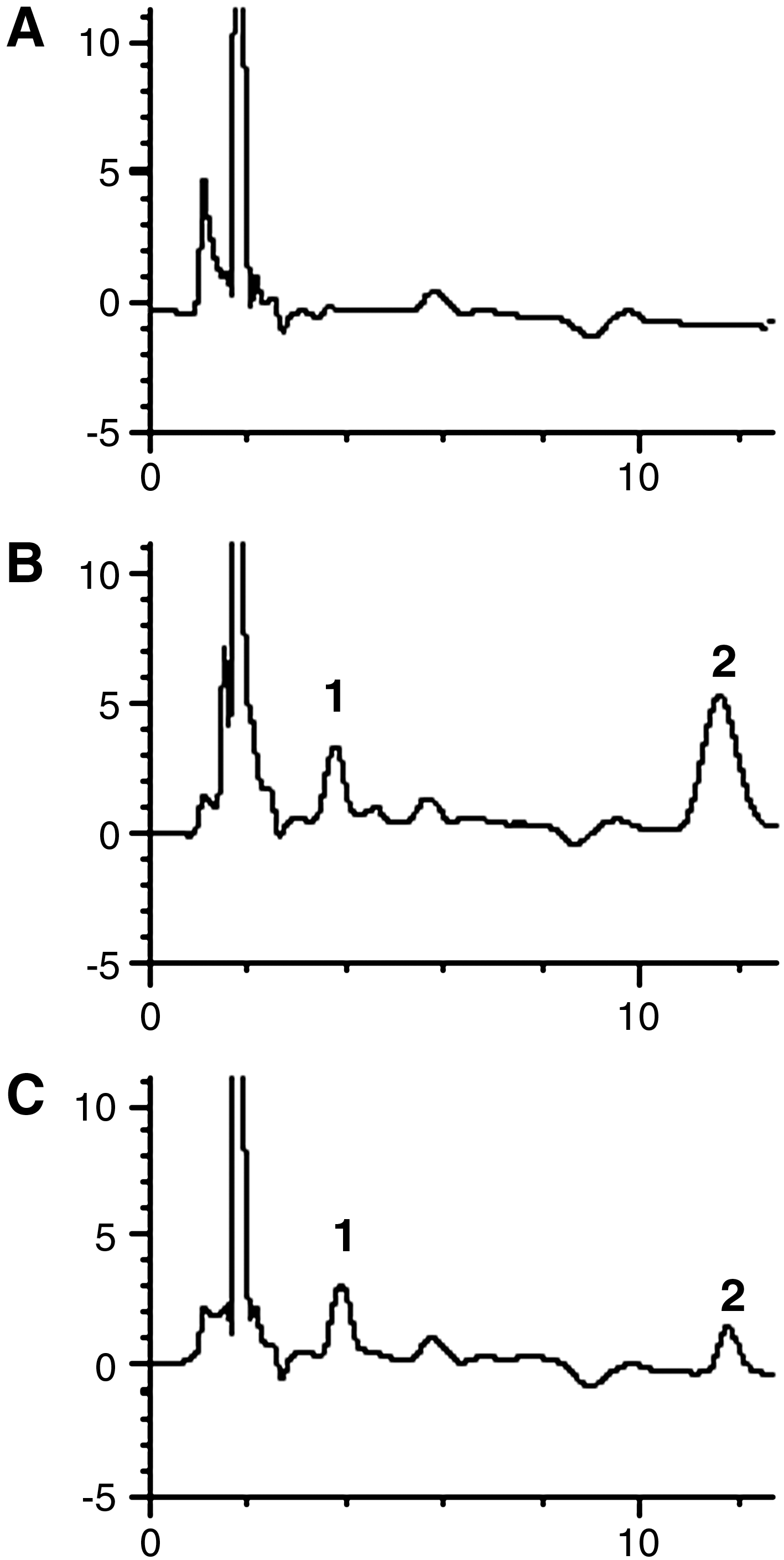
Chromatogram. (
The standard curves were prepared by plotting peak area ratios of morphine against the morphine concentration and analyzed by the linear least-squares regression analysis. The standard curve for morphine was linear over the concentration range of 5–2000 ng/mL (y=1.4368x+0.0201, r 2 =0.9994). For the validation of the HPLC method, precision and accuracy were determined by repeated analysis of three concentration levels of quality control samples (25, 100, and 1000 ng·mL−1, n=5) on 5 separate days. The minimum detectable concentration was 2 ng·mL−1 (S/N ratio, 3:1). The recoveries of internal standard were 84.29% to 106.45%. The innerday and interday precisions (as the percentage relative standard deviation) ranged from 5.98 to 8.86% and 7.82 to 11.40%, respectively.
Statistics
All data were presented as mean±standard deviation, and all statistical tests were two sided with significance level of 0.05. The pharmacokinetic parameters of morphine were got from Drug and Statistics Ver1.0, including the area under the concentration–time curve extrapolated to infinity (AUC(0–∞)), peak plasma concentration (Cmax,), the time to reach Cmax (Tmax), and the mean residence time (MRT (0–t)). Meanwhile, the terminal half-life (T1/2) and the clearance rate (CL) were obtained according to compartment model analysis. The absolute bioavailability was calculated by F = (AUC inhalation×Dose i.v.)/(AUC i.v.×dose inhalation)×100%, where the dose inhalation is the nebulizer loaded dose of 1.32 mg/kg. SPSS for Windows statistical package was used. Correlations were determined with simple linear regression models. Values of pharmacokinetic parameters for the inhaled and intravenous morphine were compared by paired Student's t- test.
Results
There was a rapid appearance of morphine in arterial blood after pulmonary administration, and the plasma concentration of morphine peaked about 23 min after the administration. The variation of plasma morphine concentrations at the selected time in two groups is presented in Table 1. The curve of morphine plasma concentration–time is displayed in Figure 4. There were no significant differences (p<0.05) on AUC and CL between these two groups. However, T1/2 was significantly longer in the inhalation group than that in the intravenous group (21.9±5.1 min vs. 3.3±1.0 min, p=0.003), and MRT in the inhalation group was also significantly prolonged compared with the intravenous group
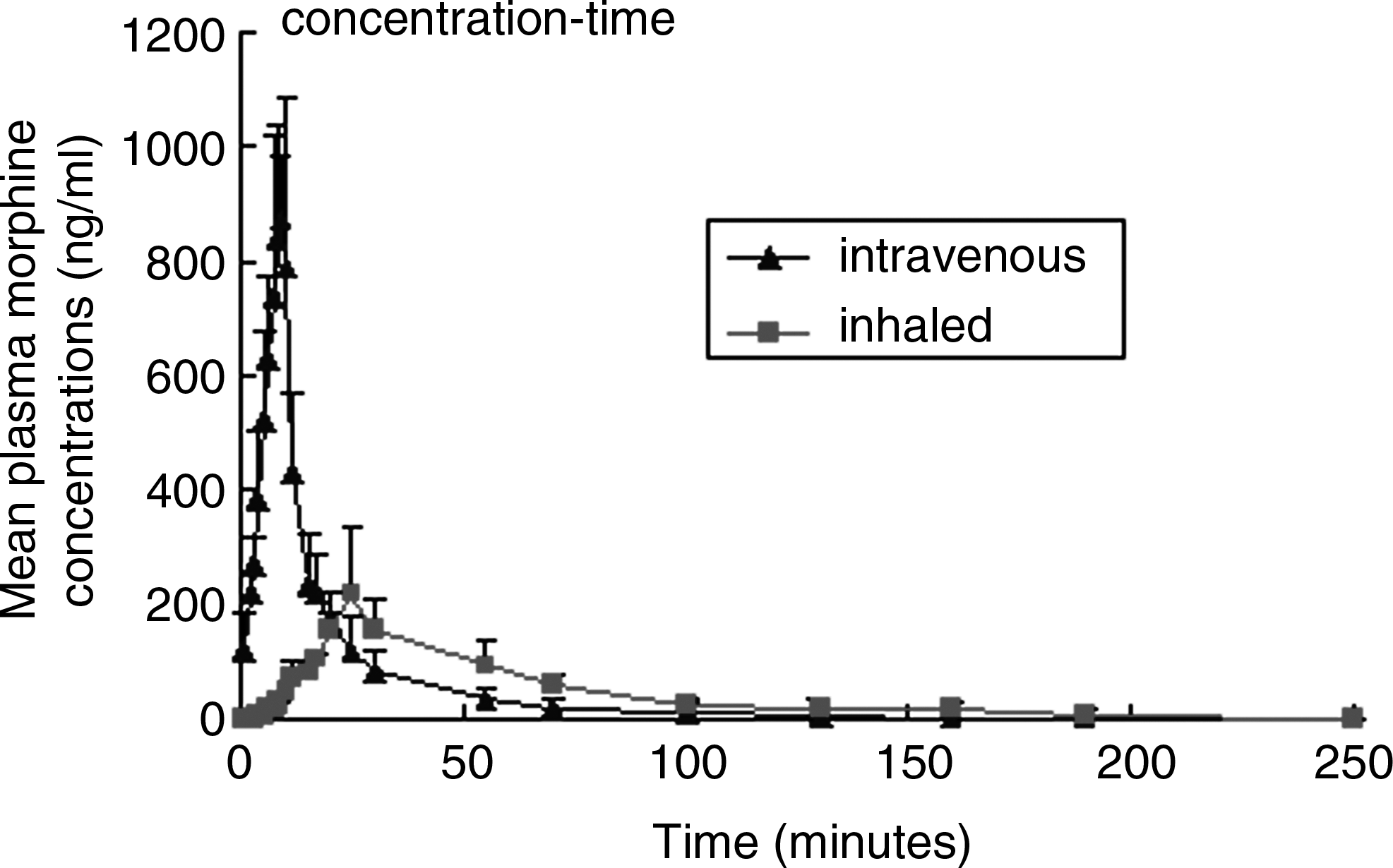
Plasma morphine concentrations by intravenous and inhaled administration in six dogs in the randomized crossover study.
p<0.05 means the difference between two groups was significant at the time.
Cmax, peak plasma concentration; Tmax, time to reach Cmax, AUC0–∞, area under the blood morphine concentration-time curve extrapolated to infinity; T1/2, terminal half-life; MRT(0–t)
The simulate test showed that there was no significant change on morphine concentration before and after the 10 min of nebulization (Table 3).
Discussion
Recently, close attention has beens paid to the pulmonary delivery systems, including the air-jet nebulizer, ultrasonic nebulizer, and the unit-dose inhaler of the AERx pulmonary delivery system. The air-jet nebulizer has been applied successfully for the pulmonary administration of morphine in clinic. But all of them are open systems, which usually are well suited for the patients with spontaneous breath, while unable to be applied during mechanical ventilation. As a result, there is lack of the pharmacokinetics data on inhaled morphine using ultrasonic nebulizer under mechanical ventilation. We modified the ultrasonic nebulizer and had it connected closely to the ventilator for the pulmonary administration of morphine during general anesthesia and mechanical ventilation. The previous simulate experiments showed that the ventilator could work normally when it was connected with the modified ultrasonic nebulizer.(15) The present study confirmed that the modified ultrasonic nebulizer and ventilator were all doing well.
The pharmacokinetic data showed that morphine could be detected quickly in arterial blood after pulmonary delivered by the modified ultrasonic nebulizer. The results of longer Tmax and lower Cmax in the inhalation group than those in the intravenous group indicate that the administration of morphine via intratracheal tube could produce a longer duration of action. In our previous study, we also found the inhalation of morphine nebulized by this modified ultrasonic atomizer via an intratracheal tube before the end of operation could produce longer and satisfying postoperative pain relieving in obstetric and gynecologic patients. Obviously the pharmacokinetic results of Tmax and lower Cmax in the inhalation animal is in conformity with the pharmacodynamics feature in the clinic.(16) We think that the longer and satisfying analgesic effect in postoperative patients is based on the slowly absorption of morphine from lungs. It is well known that the inhaled morphine must travel through the pneumocytes and the vessel walls into blood circulation, while intravenous morphine was directly infused into circulatory system. As a result, the T1/2 and the MRT of inhaled morphine were significantly prolonged than those of intravenous morphine. Compared with intravenous morphine, the curve of blood morphine concentration–time showed that plasma morphine concentrations from 10 min to 60 min after the administration of inhaled morphine were significantly higher. These data suggested that nebulized morphine has a better pharmacokinetics profile in slow release and lower toxicity than the intravenous morphine.
The absolute bioavailability of inhaled morphine in this study was (30.5±10)%, which was lower than the 59% reported in Dershwitz's(9) study using the AERX system and higher than the 17% reported in Chrubasik's study(6) using the jet nebulizer. In fact, in our study there was always 3 mL residual fluid in the nebulizer, so the real dose delivered to the dog was only 3 mL of the 6 mL loaded to the nebulizer. Meanwhile, as shown in Table 3, morphine concentration in the residual remained unchanged; thus, the real nebulized and delivered dose should be 0.66 mg/kg in stead of 1.32 mg/mg, and the absolute bioavailability might get doubled accordingly. Therefore, it suggests that morphine could have a high bioavailability when it is nebulized by the ultrasonic nebulizer during mechanical ventilation.
The bioavailability of inhaled drug is closely related to the aerosol efficiency of drugs. The mechanism of the ultrasonic nebulizer is that the ultrasonic generator inputs high-frequency power to make high-frequency oscillation of the nebulizer film to atomize the solution into droplets with 1–5 μm in diameter, which is close to the alveoli of lung in size. In addition, a normal ultrasonic nebulizer has its own air driving system, but after being connected to a ventilator, the air supply of the modified nebulizer will be replaced by the fresh air of the ventilator. Obviously, the driving power of the ventilator is stronger than the air driving system of the nebulizer. We surmise that the higher bioavailability of inhaled morphine in this study may be attributed to both the strong driving system of the ventilator and the matching size of the aerosol. Clinically, we can adjust the diameter of the aerosol, the inspiration velocity, and the inspiration time to facilitate the distribution of the drug to the alveoli of lung in the further study.
In the present study, that the dose of thev aerosol drug was twice that of the intravenous dose was established according to approximately 3 mL solutions remained unnebulized in this type of ultrasonic nebulizer. The residual liquor varies with different types of nebulizers; therefore, the dose of loaded drug should vary according to the type of the nebulizer.
In summary, our pharmacokinetic data shows that morphine administered by the ultrasonic nebulizer can be rapidly absorbed by the peripheral lung during mechanical ventilation, and has a better pharmacokinetics profile in slow release and lower toxicity than a typical bolus intravenous injection.
Footnotes
Acknowledgments
This research was funded by Nanjing Health Bureau.
Author Disclosure Statement
The authors declare that no conflicts of interest exist.
