Abstract
There is a growing interest internationally to use more biomass for power generation, given the potential for significant environmental benefits and long-term fuel sustainability. However, the use of biomass alone for power generation is subject to serious challenges, such as feedstock supply reliability, quality, and stability, as well as comparative cost, except in situations in which biomass is locally sourced. In most countries, only a limited biomass supply infrastructure exists. Alternatively, co-firing biomass along with coal offers several advantages; these include reducing challenges related to biomass quality, buffering the system against insufficient feedstock quantity, and mitigating the costs of adapting existing coal power plants to feed biomass exclusively. There are some technical constraints, such as low heating values, low bulk density, and grindability or size-reduction challenges, as well as higher moisture, volatiles, and ash content, which limit the co-firing ratios in direct and indirect co-firing. To achieve successful co-firing of biomass with coal, biomass feedstock specifications must be established to direct pretreatment options in order to modify biomass materials into a format that is more compatible with coal co-firing. The impacts on particle transport systems, flame stability, pollutant formation, and boiler-tube fouling/corrosion must also be minimized by setting feedstock specifications, which may include developing new feedstock composition by formulation or blending. Some of the issues, like feeding, co-milling, and fouling, can be overcome by pretreatment methods including washing/leaching, steam explosion, hydrothermal carbonization, and torrefaction, and densification methods such as pelletizing and briquetting. Integrating formulation, pretreatment, and densification will help to overcome issues related to physical and chemical composition, storage, and logistics to successfully co-fire higher percentages of biomass (>40%) with coal.
Introduction
Presently, biomass is gaining importance worldwide for power generation along with coal. Because biomass is considered carbon neutral, it can significantly reduce net carbon emissions and negative environmental impacts when it replaces coal or other fossil fuels. Thus, it is a clean development mechanism (CDM) for reducing GHG emissions and other pollutants. 3 Despite the dominance of fossil fuels for heat and electricity production in many developed countries, sources of biomass exist and continue to be exploited. Such sources include forest and wood products, residues, yard clippings, construction and demolition debris, and municipal solid waste (Fig. 1). According to the Billion-Ton Update report released by the US Departments of Energy (DOE) and Agriculture (USDA) in August 2011, there are more than a billion tons of woody and herbaceous biomass available annually, which can potentially replace about 30% of petroleum usage. 4
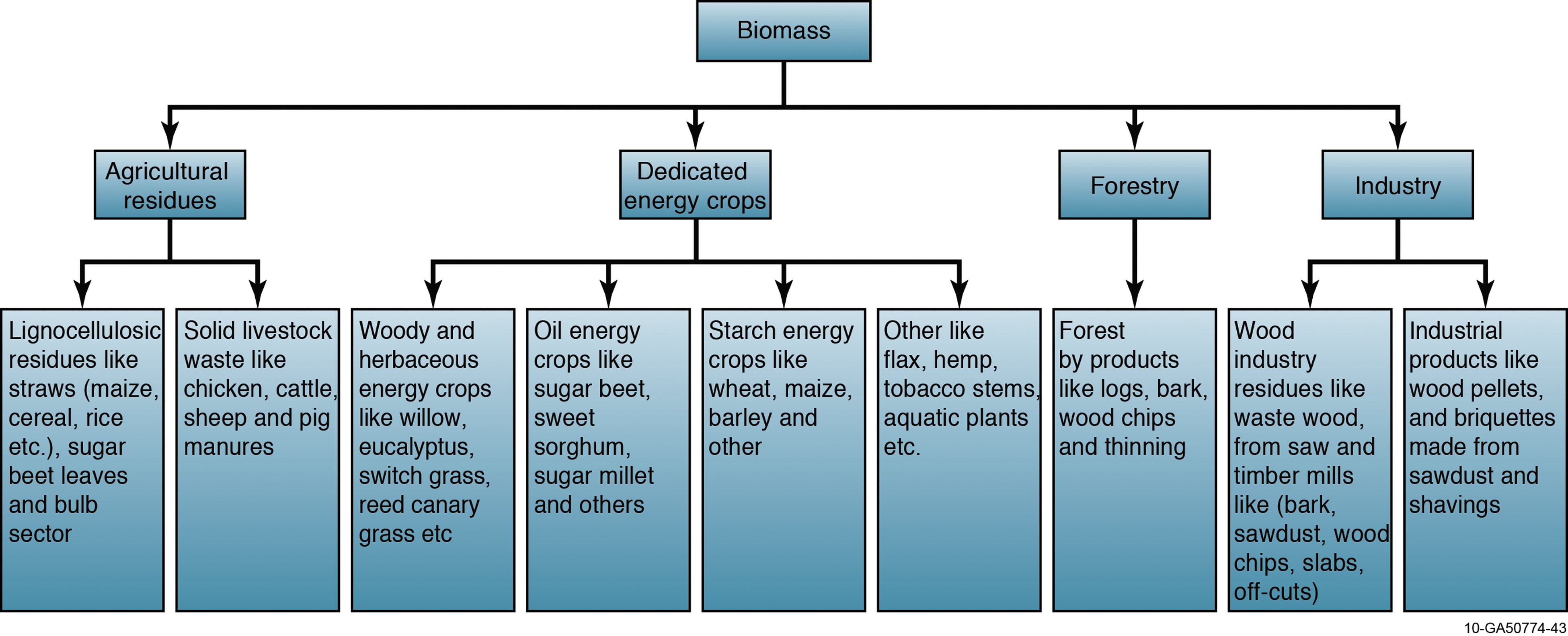
Biomass feedstocks useful for co-firing applications. Color images available online at
There are challenges to increasing the use of biomass for energy purposes. The diversity of biomass resource types and production conditions causes variability in biomass composition and physical properties. Reliability of the feedstock supply and accessibility to distributed resources are also important issues because in many countries biomass resources are dispersed and the infrastructure for supply is not well-established. The collection and transport of dispersed biomass feedstocks are too expensive for biofuels production. 5 Technical limitations to using biomass as a sole feedstock for bioenergy production pose further challenges to widespread adoption; the inherent low heating values and low bulk densities of biomass relative to coal necessitate transporting larger units of biomass. 6 All of these factors underscore the importance of developing consistent feedstock specifications that will allow biomass parameters and variability to be handled predictably and systematically.
One of the foremost approaches to overcoming these challenges is to co-fire biomass with coal. Co-firing represents the lowest-risk, least expensive, most efficient, and shortest-term option for renewable-based electrical power generation. 7 In general, the term co-firing refers to the simultaneous combustion of two different types of material or use of a supplemental fuel in a boiler in addition to the primary fuel the boiler was originally designed to use. In conventional power plants, co-firing typically involves modification of the existing equipment, which is only designed to handle coal. Furthermore, because of the inherent differences between coal and biomass, co-firing biomass is typically done on a limited basis, with biomass providing only 5–15% of the total heat input to the boiler. 8,9 Due to the relatively small biomass-to-coal ratio, this method typically does not require substantial modifications to the boiler(s). 10,11 Minor modifications to the burners are still needed, however, to prevent early ignition and fuel staging within the flame zone, which arises due to high volatile content in biomass. 12 If higher ratios of biomass to coal are desired, separate handling systems and specially dedicated biomass burners should be used to avoid negatively impacting the coal delivery system. 11 Traditionally, co-firing biomass with coal requires both materials to be added to the combustor in separate streams. However, advanced systems have been developed that allow biomass and coal to be mixed prior to insertion into the combustor.
Facilities that support co-firing of biomass with coal include large coal-fired power and heating plants, cement kilns, and industrial boilers. Co-firing provides important industry-enabling advantages: • Issues related to biomass quality can be overcome. • Variations in feedstock supply can be moderated with increased coal. • Costs of adapting the existing coal power plants are lower than for building new systems dedicated to biomass.
6
• SOx emissions will be reduced. • Types of fuel used can be diversified to create markets for a variety of locally grown, indigenous, and renewable fuels. • Industries and other facilities can meet or exceed goals for environmental stewardship. For example, carbon dioxide, sulfurous gases, and ozone emissions can be reduced; ozone is particularly impacted by reducing nitrogen oxides by about 30%.
13
A major limitation of co-firing biomass with coal is loss in boiler efficiency, which is mainly due to higher moisture and lower energy content of the biomass fuels compared to coal. 14 A second limitation to co-firing is the ash that is produced by biomass; the American Society for Testing and Materials Standard C618 requires that only coal ash be used in the mixture in Portland cement. Presently, US Department of Energy (DOE) and other organizations are working on changing this standard to allow co-fired ash to be used in manufacturing Portland cement. Utilities' resistance to biomass may be reduced if they are able to sell the ash from co-firing.
Nevertheless, the majority of available literature indicate that there are major physical and chemical composition issues that limit the amount of biomass that can be co-fired with coal. 15 –26 Considerably less attention seems to have been given to strategies for overcoming these limitations. Some reports identify co-firing issues and mitigating steps, including pretreatment methods such as washing and torrefaction, and densification methods such as pelleting and briquetting. 27 Research by Mohammad et al. and Hughes and Tillman explored the effect of biomass physical properties and chemical composition on co-firing efficiencies, ash properties, fly ash sales, boiler slagging, and fouling. 28,29 They also studied the influence of fuel blending on pulverizer performance.
With a limited amount of available information, there is a need for a comprehensive assessment of the physical and chemical property challenges and available remedies to co-fire higher percentages of biomass with coal. This review aims to identify the status of co-firing around the world, with particular emphasis on the US; compare coal and biomass composition and identify issues related to both physical properties and chemical composition; evaluate available remedies, including formulation, pretreatment methods (such as washing/leaching, steam explosion, and hydrothermal torrefaction) and densification methods (such as pelleting, briquetting, screw press, and agglomeration), and evaluate the integration of these technologies to develop a uniform feedstock commodity for co-firing applications; compare the formulated, pretreated, and densified material properties with those of coal; and compare different co-firing approaches. The findings will promote the development of specifications for the reliable and efficient processing of biomass, which will in turn serve to channel future research to effective paths for co-firing higher percentages of biomass with coal.
Biomass Co-Firing Status in IEA Member Countries
Biomass co-firing has been demonstrated in over 234 installations worldwide for most combinations of fuels and boiler types in the range of 50–700 megawatt (MW). Approximately 170 of these demonstrations have been in Europe, while the US and Canada account for about 47. In Australia and Southeast Asia, there have been 8 and 4, respectively. 30 Figure 2 shows the number of power plants in International Energy Agency (IEA) countries that have tested or documented co-firing of biomass or are currently co-firing on a commercial basis. A broad combination of fuels, such as residues, energy crops, and herbaceous and woody biomass has been co-fired in pulverized-coal combustion (PCC), stoker, and cyclone boilers. Some studies on co-firing of biomass with coal have indicated that the boiler efficiency and boiler capacity are reduced. 31,32 Co-firing percentages have ranged from 1–20% biomass, by mass. Reduced emissions of sulfur dioxide (SO2), nitrogen oxides (NOx), and mercury can be attained by substituting biomass for coal; however, herbaceous biomass in particular often contains detrimental semi-volatile alkali mineral matter, which can result in increased slagging and fouling as discussed by Al-Mansour and Zuwala. 33,34
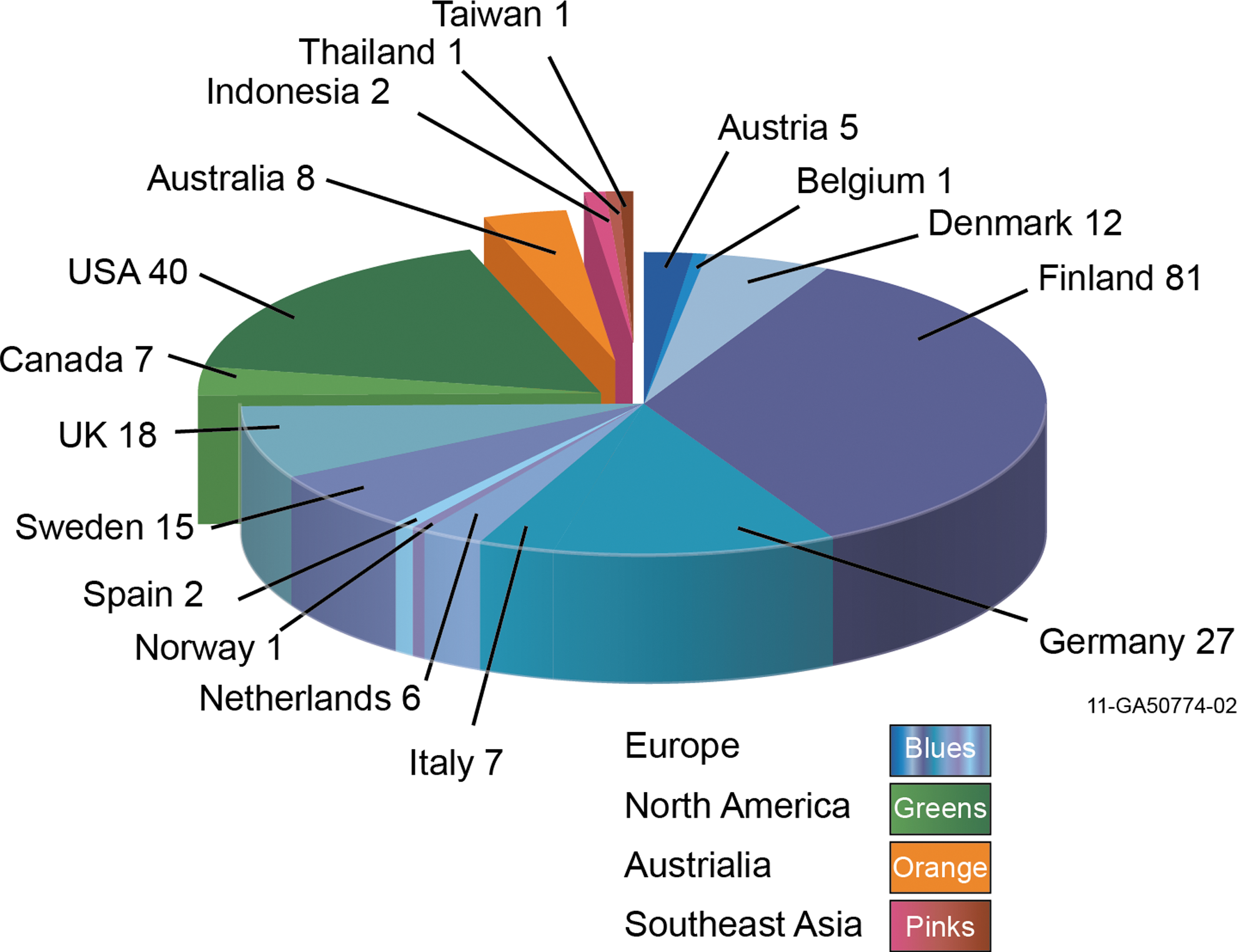
Number of power plants demonstrating co-firing capabilities in IEA member countries. 30
Biomass co-firing has been used in Europe and the Netherlands for more than a decade. Full-scale commercial co-firing of at least 10% biomass (based on heat input) is a common practice using a variety of biofuels and co-firing configurations. The technology of co-firing has been demonstrated in many boiler types in Europe.
In the US, power generation and co-generation from biomass, waste, and recovered fuels are now receiving attention in the power sector as a result of new environmental policies and regulations and societal values. The Electric Power Research Institute (EPRI)—working in cooperation with the Tennessee Valley Authority (TVA) and supported partially by DOE—initiated a co-firing program in 1992. Beginning with engineering studies and expanding through tests and demonstrations, this program was designed to evaluate, demonstrate, and commercialize co-firing as a strategy for immediately increasing the use of biomass in utilities.
The USDA and DOE began to support biopower development under their respective fossil energy and renewable energy programs. In December 2009, the DOE's Office of Energy Efficiency & Renewable Energy (DOE–EERE) Biomass Program held a Biopower Technical Strategy Workshop to explore biopower opportunities in the US. Workshop participants explored the challenges and potential solutions to expand the use of biopower, including technology research, development, and demonstration; policies; and other market transformation mechanisms. 35
Chariton Valley Biomass Project is an example of a successful commercial co-firing project. The project was a cooperative effort between Chariton Valley Resource Conservation and Development Inc., Alliant Energy, Prairie Lands Biomass LLC, DOE, and USDA to study co-firing switchgrass with coal, with 3% of the heat input from biomass in a 725 MW power plant. 36 A long-term test burn was conducted between February and May of 2006, with the facility operating continuously, to gain a better understanding of the operation of commercial power plants. The project set a world record for power generated from switchgrass during the test burn and collected valuable performance data and experience for evaluating the commercial viability of the project. This co-firing test was considered highly successful because no negative environmental emissions or loss in electricity output were reported. 37
Over the past 15 years, Keuring Elektrotechnische Materialen Arnhem (KEMA), a Dutch company, has performed co-firing with both direct and indirect systems using different biomass materials. It has attempted co-firing coal and biomass mixtures up to 25% and has been involved with more than 50 small- and full-scale biomass trials. In North America, KEMA has performed extensive feasibility studies on co-firing for six large utilities. 38 These co-firing demonstrations have helped identify major technical barriers, which include fuel feeding, boiler chemistry, and ash deposition and disposal, and have resulted in an emerging consensus that co-firing is feasible at most coal-fired power plants, especially if the challenges are addressed by biomass pretreatment. 39
Biomass and Coal Composition
Biomass solid fuels include woody and herbaceous materials. Processed biomass is typically referred to as feedstock. Various biomass resources available for energy purposes include oil seeds, grains, sugar crops, agricultural residues, trees, grasses, and algae. 40 Table 1 indicates the proximate and elemental composition of woody and herbaceous biomass. Note that herbaceous biomass has higher volatiles and less elemental carbon. 41
Typical Wood and Herbaceous Biomass Composition
Source: Tillman et al. 41
Coals are classified based on heating value and the ratios of volatile hydrocarbon material to fixed carbon. Anthracite coal is low in volatile material and high in fixed carbon. Soft coals are low in carbon and high in volatile matter and moisture content. Anthracite contains the highest proportion of pure carbon (about 86-98%); hence, it has the highest heat values of 30-36 MJ/kg.
42
Typical coal composition for Central Appalachian (anthracite), Illinois Basin (bituminous), Powder River Basin (sub-bituminous), and North Dakota lignite are given in Table 2.
41
Compared to coal, biomass feedstocks have the following characteristics
18
: • Higher organic volatile matter; • Lower energy density, resulting in lower heating value; • Lower bulk density; • Lower concentration of sulfur; • Higher concentrations of alkali and alkaline-earth elements (potassium, sodium, calcium, and magnesium); • Higher concentrations of phosphorous, chlorine, and water vapor; • Lower ash fusion temperatures; • More fibrous composition, which impedes grindability in existing coal pulverizers and results in much larger and more irregularly shaped particles.
Typical Coal Composition
Source: Tillman et al. 41
Biomass Physical Properties and Compositional Issues for Combustion
Biomass is a good candidate for co-firing with coal because the equipment designed to burn coal can be used to co-feed biomass with little or no modification when feeding a composition of less than 5% biomass (by mass). However, when feeding higher percentages, the physical and chemical properties of biomass can impose some challenges. Several variables need to be considered, including fuel preparation, handling, storage issues, milling, feeding, differing combustion behavior, possible changes in overall efficiency, deposit formation (slagging and fouling), agglomeration and sintering, corrosion and/or erosion and consequent changes in lifetime of equipment, ash utilization issues, and overall economics, as discussed in Baxter, European Bioenergy Networks (EUBION), and Jarvinen and Alakangas.
7,43
–45
Sami et al. and Demirbas reported that biomass chemical composition significantly affects the combustion characteristics when co-fired with coal.
23,46
–48
These authors have also identified several differences between coal and biomass combustion: • Early pyrolysis for biomass fuels compared to coals. • Fractional heat contribution by volatiles in biomass is 70% compared to 36% in coal due to higher volatile content in the biomass. • Oxygen content of the biomass chars is higher compared to coal. • Gases released by pyrolysis of biomass char mostly contain CO, CO2, and H2O.
As biomass has higher volatiles, biomass undergoes early pyrolysis compared to coal, and the ash in biomass is more alkaline in nature, resulting in greater fouling and corrosion. Biomass char has more oxygen content and is more porous and reactive. Belosevic indicated that the varying physical and chemical properties of biomass fuels have a significant impact on the characteristics of feeding and combustion, particularly the flame front and brightness. 11 These limitations play a major role when co-firing high percentages of biomass with coal. The same authors have also indicated that co-firing high percentages of biomass requires separate feeding lines and injection ports.
Chemical Composition
Typically, woody biomass contains less ash than coal; its ash composition is based on the chemical components required for plant growth, whereas coal ash reflects mineralogical composition. In both coal and biomass, ash-forming matter can be present in four general forms: easily leachable salts, inorganic elements associated with the organic matter of the biomass, minerals included in the fuel structure, and loosely bound inorganic material—typically sand, salt, or clay. Alkaline metals that are usually responsible for fouling of heat transfer surfaces are well represented in biomass ash and are released during combustion. These inorganic compounds are either in the form of salts or bound in the organic matter. In peat, for example, inorganic matter is bound mostly in silicates, which are more stable at high temperature. The elemental composition of ash (alkali metals, phosphorus, chlorine, silicon, and calcium) affects ash-melting behavior. Chlorine, a minor constituent in virtually all coals and biofuels, contributes to a number of combustion phenomena, depending upon concentration. Even a small concentration of chlorine in the fuel can result in deposition of harmful alkaline and chlorine compounds on boiler heat-transfer surfaces. Numerous corrosion mechanisms exist with chlorine in solid fuels, and these mechanisms have been elucidated for biomass co-firing with coal. However, higher chlorine content in coal can help reduce mercury emissions when coal is burned because it combines to form mercuric particulate that is more easily filtered or scrubbed than elemental mercury.
Moisture and Volatiles Content
The properties of biomass, like high moisture and volatile content, affect grinding, feeding, and combustion behavior during co-firing. The higher moisture content of biomass can limit the capacity of grinders when biomass is co-milled with coal for co-firing. Also, coal mills or pulverizers cannot process certain types of biomass due to their fibrous nature. These problems are not encountered in indirect, or parallel co-firing configurations, as independent lines are available for biomass milling or grinding. However, these scenarios require separate grinding equipment and handling systems, which increases production costs. In addition, co-milling wet biomass material with coal will have a significant impact on the mill heat balance. 49 Backreedy et al. indicated that the kinetics of biomass combustion are not thoroughly understood, and that modeling of coal/biomass blends is more complex due to gas and two particulate phases, as well as chemical reactions. 50 According to Dai et al., as biomass and coal are two chemically different fuels—with biomass being more reactive and having higher levels of volatiles and moisture than coal—biomass has a lower calorific value. 31 Fresh wood typically contains 50% water by weight, whereas bituminous coals hold approximately 5%. Higher moisture content reduces the maximum combustion temperature and increases the necessary residence time of feedstock in a combustion chamber. This results in incomplete combustion and increased emissions (volume of flue gas produced per energy unit), as per European Committee for Standardization (CEN) CEN–335, 2003. 51 Chao et al. studied thermo-gravimetric analysis (TGA) and differential thermal analysis (DTA) of coal, rice husk, and bamboo, and concluded that high volatile matter (VM) concentrations and low activation energies of rice husk and bamboo resulted in early pyrolysis and volatile oxidation. 52 They also inferred that higher VM content of biomass results in two distinct stages of weight loss, where the first is due to initial gas-phase oxidation, and the second to char oxidation at the latter stage of combustion. Their results indicated that most of the energy from the biomass combustion came from the VM reaction, whereas, in the case of coal, the energy was from char oxidation. The different combustion phenomena observed during co-firing can affect the sizing and design of the combustion chamber and burner registers. Backreedy et al. also explained that biomass components like hemicellulose, cellulose, and lignin influence combustion rates, reasoning that biomass with high lignin results in less reactivity. 50
ASH Composition
Issues related to ash become increasingly important when biomass is considered for combustion along with coal. Ash contents of different biomass fuels can vary significantly, and this influences their deposition rate, as shown in Table 3 and Figure 3. Straw and other herbaceous fuels like miscanthus or grass have higher ash contents than wood because they uptake more nutrients during growth. In the case of wood fuels, the bark content in the fuel has an influence on the ash content, as bark has higher ash content and higher levels of mineral impurities such as sand and soil. 44,53 –56
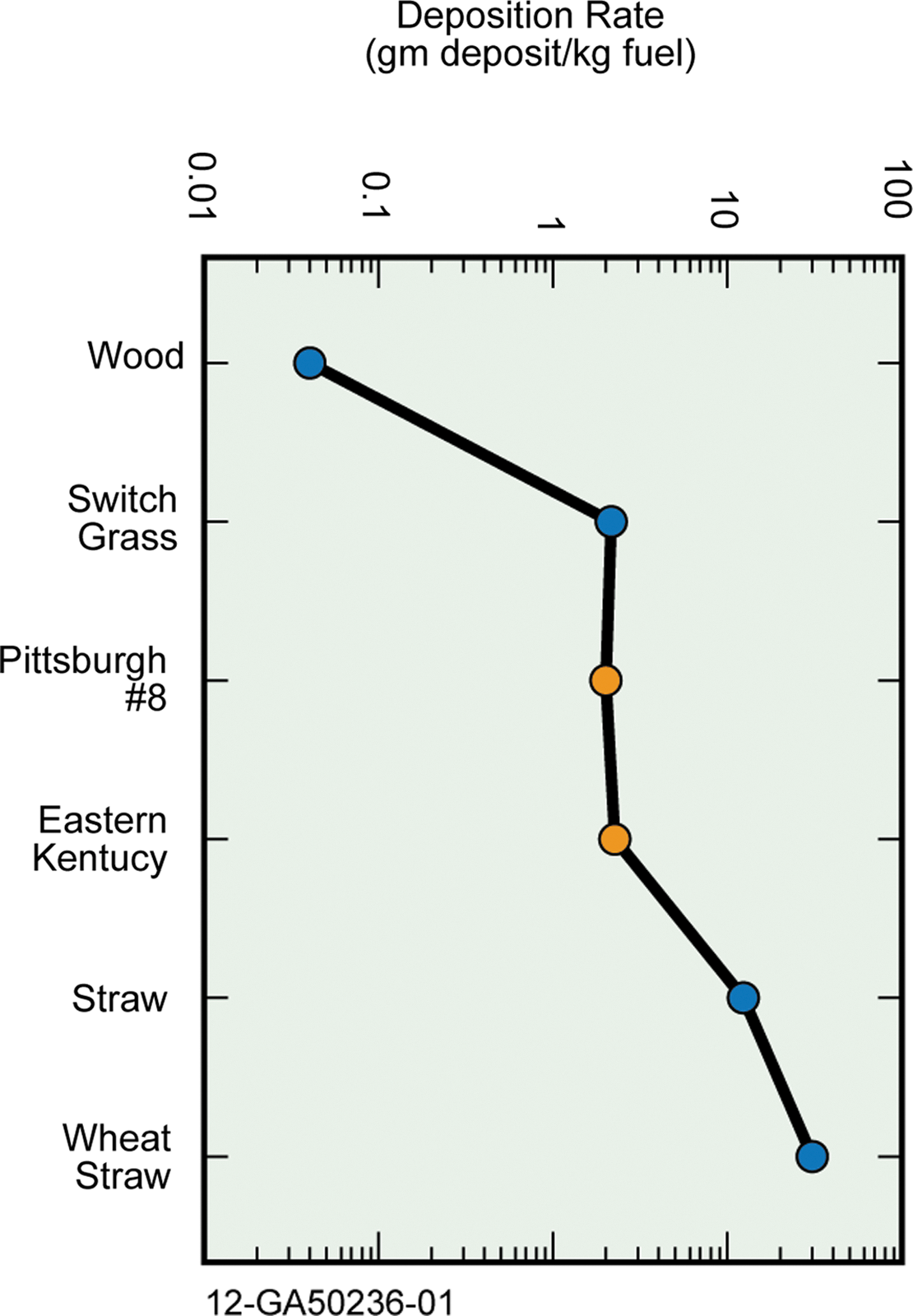
Ash deposition rate for woody and herbaceous biomass and coals. Color images available online at
Ash Content of Different Biomass Fuels and Coal
Direct co-firing of biomass and coal produces mixed biomass and coal ash, whereas parallel co-firing and indirect co-firing can produce separate biomass and coal ashes. The commercial uses for the ash produced (single coal, single biomass, and mixed biomass with coal) are important from environmental and performance perspectives. The ash produced during firing of coal (boiler slag, fly ash, and bottom ash) is used in construction, underground mining, and restoration of open cast mines, pits, and quarries. Presently, several research activities are characterizing biomass ash and assessing the possibilities for its utilization. Potentially, the ash originating from combustion or gasification of biomass can be used as fertilizers or in fertilizer production, as it is rich in Mg and Ca; however, the lack of nitrogen and soluble phosphorous may limit its use in many fertilizer applications. Direct utilization of biomass ash in building applications might be possible for some types of bottom ash, or it might be used as raw material in the production of building materials. Large amounts of unburned carbon in biomass fly ash from fluidized-bed gasification can be used directly in a PF boiler as fuel for power generation because of its high calorific value, and the hydrophobicity of this material allows it be stored for long time periods without loss in quality. Densifying ash in the form of pellets, briquettes, or granules, has many advantages: volume is reduced by a factor of 4–6, storage in ambient air is possible, and the health and safety risks (e.g., dust explosion) are reduced. Successful re-use of ash as a fuel will require consistently high-quality ash in sufficiently large quantities to boost interest and development of ash utilization options. 57
Nox and Sox Emissions
Emissions of oxides of nitrogen and sulfur during coal and biomass combustion arise predominantly from nitrogen and sulfur in the fuel. In general, NOx formation is highly dependent on residence time, temperature profile, and the air-to-fuel stoichiometry of the combustion process. The lower NOx values reported when biomass is co-fired with coal may be due to higher release of volatiles and fuel-N in the flame, which suppresses the formation of NO. 58 Most commercial biomass combustors operate at sufficiently low temperatures so that thermal NOx contributes only a small fraction of the total. Nevertheless, the formation of thermal NOx is directly related to operational parameters. Once released into the atmosphere, NOx, in combination with hydrocarbons and photochemicals, can lead to the formation of ozone and photochemical smog, which are lung and eye irritants and create a major problem in urban environments, as pointed out by Robinson et al. and Boardman and Smoot. 22,59 NOx and SOx emitted during coal combustion can form nitrate and sulfate particulates, which are known to be acid-rain precursors because of their high settling velocity. Replacing coal with biomass feedstocks, which are low in sulfur and nitrogen, can reduce NOX and SOx emissions and minimize environmental damage. 60 Corroborating data indicate that co-firing biomass usually reduces NOx, even when it may have higher nitrogen. 24 This is attributed to the form of nitrogen in biomass, mainly amine or amine-like function groups. Under reducing conditions or at moderate flame temperature, gaseous amine molecules will react with the NOx that has already formed. This benefit may, however, be contradicted by the formation of higher amounts of thermal NOx when the high percentage of oxygen in the biomass leads to significantly higher peak flame temperatures in the near burner region. Hence, no a priori credit can be taken for NOx reduction, and each co-firing case needs to be evaluated individually. On the other hand, SOx emissions are reduced by the total reduction of sulfur when compared to 100% coal. Additionally, the higher content of calcium and alkali oxides of the flue gas can serve as a sorbent of sulfate as well as chlorides.
Mercury Emissions
According to the State of North Carolina Department of Environmental and Natural Resources (NCDENR), mercury exists in elemental, inorganic, and organic forms. Co-firing biomass with coal should reduce toxic mercury emissions by converting the elemental mercury into inorganic mercury. 61 A large amount of work was done in the late 1990s and into the current decade to quantify mercury disposition during conventional coal burning of all ranks and boiler packages. 62,63 Some coals are higher in mercury, while others are higher in chlorides. 59 High chloride concentrations are attributed to the formation of mercuric chloride particulate that is readily captured in the bag house (or electrostatic precipitator), and in all scrubbing units. 61 Many strategies have been developed for mercury removal, and many of these involve the addition of bromine or chlorine, or co-firing with feeds that are high in these elements. Hence, biomass with a high chlorine content may reduce mercury emission when blended with coals—especially coals that are low in halogens. 59 Unfortunately, the relative benefits of higher chlorine contents in the feed are not likely to outweigh the detrimental impacts of chlorine on boiler-tube and catalytic flue-gas clean-up surfaces. 64
Chlorine Emissions
Corrosion of the boilers' surfaces is one of the major issues when co-firing biomass with coal. Chlorine, a minor constituent in virtually all coals and biofuels, contributes to corrosion of boiler surfaces depending upon concentration. Corrosion and deposition are primary concerns; the occurrence and extent of these phenomena depend significantly upon the concentration and form of chlorine in the fuel and other combustion considerations. 65 –69 Reactions of chlorine with alkali metals such as potassium and sodium are the most commonly identified corrosion mechanism when co-firing biomass alone or with coal. According to Tillman et al., potassium chloride is the most prevalent compound deposited on pendant tubes and other heat-transfer surfaces when firing biomass or co-firing biomass with coal. 41 Hydrogen chloride formed during coal and biomass co-firing can contribute significantly to low-temperature corrosion areas like flues, precipitators, baghouses, and other post-air heater equipment. 21
Physical Properties
Differences in the physical properties of feedstocks can have an impact on grinding and co-feeding with coal, as well as flame profile and maximum temperature during combustion. Studies conducted by Yin et al. on computational fluid dynamic (CFD) modeling of coal and biomass co-firing indicate that the physical properties of the solid fuels have a great influence on the predicted temperature profiles. 70 Biomass has low bulk energy density and hydrophilicity and a non-friable character. 51 These properties significantly influence handling operations and feeding systems. According to Moulton, the amount of biomass able to be co-fired with coal depends on physical properties such as bulk density, particle size distributions, moisture content, and unconfined yield (shear) strength. 71 The difference in densities of biomass and coal powders poses a bigger problem when biomass and coal are co-milled and fed together to the burners.
Bulk Density and Particle Size/Shape Distributions
Biomass particles are naturally large and non-uniform in size, shape, and density. Yin et al. reported that the complex physical nature of the biomass particles, including their shape and size, dramatically influence their trajectory path and conversion in a combustor. 72 Size and shape significantly influence the char burnout, which can be due to non-melting of biomass. Larger size particles of a given mass result in faster burnout due to non-sphericity. 31 Co-milling of biomass with coal tends to result in larger biomass particles because their low particle density renders the pulverization process less efficient.
According to Moulton the amount of biomass able to be co-fired with coal depends on physical properties like bulk density. 71 The difference in densities of biomass and coal powders poses a bigger problem when biomass and coal are co-milled and fed together to the burners. Furthermore, large particle sizes, high moisture content, irregular shapes, and low bulk density tend to promote feed-rate irregularities.
Flowability
Bulk solids, such as ground biomass, flow primarily by shear deformation. This involves both plastic and elastic behavior and is much more complex than that of liquids or gases, resulting in additional challenges. Common problems encountered during biomass flow include plugging, segregation, obstructed or limited discharge, erratic flow, sudden uncontrollable flow, and sticking of material to container walls, which causes a loss of live storage and can lead to spoilage. In extreme cases, flow problems can even cause high transient stresses that result in structural failure. These difficulties routinely cause industrial plants that handle biomass like wood pellets to underperform compared to their counterparts that only handle liquids. 73 It has been observed that processes for liquids and gases typically run at close to 90% of their design capacity, while similar processes for bulk solids tend to operate at only 40-50% of their design specification. 74 The primary properties that impact flowability of biomass materials include particle size and shape distribution (particle lengths greater than 2 cm tend to be non-flowing), cohesive strength/unconfined yield strength, moisture, bulk density, compressibility, springback (elastic recovery), wall friction, and permeability for fine powders. Unfortunately, reliable measurements of these properties and demonstration of their impacts on flow performance are scarce in the literature, despite the challenges that have already been recognized. 75,76 Table 4 indicates the physical and chemical characteristics of biomass and their influence on co-firing. 53,77
Co-firing Issues in Terms of Biomass Chemical Composition and Physical Properties
SCR=selective catalytic reduction, HCN=hydrogen cyanide, HF=hydrogen fluoride
Formulation, Pretreatment, and Densification Methods
Formulation
Biomass feedstocks, both woody and herbaceous, have great variability in their chemical composition, as indicated in Table 1. These properties need to be uniform to successfully co-fire different biomass types at higher percentages without sacrificing boiler efficiency. Formulations of biomass can help to meet the specifications required for co-firing in terms of calorific value, volatiles, oxygen, hydrogen, nitrogen, chlorine, sulfur, and nitrogen contents. For example, feedstocks with low lignin or high ash contents, like straws and grasses, can be blended with woody biomass, which has higher lignin and lower ash contents. These types of formulations not only help to modify the chemical composition of biomass, but also often improve its densification characteristics. Formulated feedstocks can include both raw and pretreated biomass. The formulation concept has been used in different industries, such as food and feed, to develop high value and customized products. 78,79 In most coal power plants, coals of different grades are blended to reduce SOx and NOx emissions. 80,81 Sami et al. indicated that a mixture of high-ash biomass sources and low-ash coal can be used for power generation. 46 In general, formulation can include blending (biomass feedstocks from multiple sources of the same biomass resource to average out compositional and moisture variations), aggregation (combination of different raw or preprocessed biomass resources to produce a single, consistent feedstock with desirable properties), and amendment (combination of raw or preprocessed biomass resources with non-biomass additives to produce a consistent, on-spec feedstock).
Pretreatment
Efficient and cost-effective biomass pretreatment is very important for overcoming biomass limitations and hurdles in biofuels production, and can generally help improve the physical and chemical properties of biomass to allow higher percentages of biomass to be co-fired with coal. According to van Loo and Koppejan, pretreatment helps to overcome the limitations of biomass for fuel applications. 53 Common methods that help to improve the physical properties, chemical composition, and energy properties of biomass are washing/leaching, steam explosion, torrefaction, and hydrothermal carbonization. Pretreatment helps to improve densification behavior and makes many biomass materials suitable for different densification systems like pellet mill, briquette press, screw extruder, and agglomerator. 82 Certain pretreatment processes can greatly reduce energy consumption of energy-intensive operations such as milling and can produce high-quality (high durability, bulk, and energy density), densified uniform feedstocks for co-firing applications. 82 Such uniform feedstocks can be integrated into a commodity market to help reliably overcome problems related to co-firing, including the following: operational constraints, by allowing utilization of coal infrastructure for feeding, milling, etc. of biomass, without costly modifications or installation of separate processing lines for biomass; technical constraints, by, for example, reducing corrosion due to biomass washing; and problems associated with the biomass supply chain, because the cost of transportation for pelletized or briquetted biomass is lower than that of raw or pretreated biomass. 82
Washing/leaching
The potential problems that occur with co-firing biomass with coal include ash deposition, slag formation, corrosion, sintering and agglomeration, and problems related to chemical composition due to the presence of Cl, Na, and K. The alkali compounds in biomass can be reduced by washing or leaching steps that help remove troublesome elements. Additives such as dolomite and kaolin are capable of reducing sintering by raising the melting point of ash. Also, vaporization of alkali compounds can be reduced by a decrease in process temperature. 83 Washing herbaceous biomass is very important due to high ash content. 83 –85 The removal of alkali metals, chlorine, and sulfur can be beneficial in increasing the fusion temperatures of straw ash. 84 In general, washing and leaching at low temperatures with solvents or catalysts using water, dilute acid, dilute alkali, organic solvent, or supercritical fluids can be effective in removing salts, silica, low molecular organics, and polar or non-polar solvents. 83,86 –90 Arora et al. reported that ionic liquid solvents are particularly effective in reducing the recalcitrance in the biomass. 91 They also indicated increasing the severity of leaching makes biomass less suitable for biochemical conversion.
Approximately 90% of alkali in biomass is present in water-soluble or ion-exchangeable form and is susceptible to vaporization during heating. 83 Typical ash-forming components that are leached out by water include alkali sulfates, carbonates, and chlorides. 31 Elements leached out by ammonia are organically associated, such as Mg and Ca as well as K and Na. HCl leaches the carbonates and sulfates of alkaline earth and other metals. Silicates and other minerals remain in the insoluble residue. If biomass washing can effectively reduce problems caused by alkali components in biomass, large-scale washing of problematic fuels prior to combustion may be economically feasible in industrial conversion systems. 83 Reduction of alkali compounds in biomass feedstock before co-firing can save costs involved in maintenance (e.g., superheaters would become more durable). 92 Cerezo indicated that leaching with de-ionized water and organic solvents helps to modify ash composition, reducing the alkali index (K2O+Na2O) and the ash-melting temperatures and increasing the gross calorific value. 93
Steam explosion
Steam explosion was originally developed by Mason in 1925 and has been extensively used in the manufacture of hardboard. 94 The commercial process involves filling a vertical cylinder with wood chips and then sealing and pressurizing it with saturated steam at pressures up to 1,000 pound-force per square inch (psig). The chips are permeated by the saturated steam and develop high internal pressures. When the bottom of the cylinder is opened, the wood chips are defibrated by the sudden decompression. Steam explosion not only causes a physical change in the wood but also causes considerable chemical change. Marchessauk et al, DeLong, and Foody demonstrated that steam explosion induces a number of chemical compositional changes: lignin is broken down into low-molecular weight (Mw) products (Mw,400-8,000), retains its basic structure, and remains reactive due to extensive depolymerization; and hemicelluloses are partially broken down and become predominantly soluble in water. 95 –99 In addition, some degradation products are formed that apparently condense with lignin, increasing the lignin content. This helps produce high-density pellets at lower energies. In their studies on the pelleting of steam-exploded poplar wood, Shaw et al. found that density increased from 1,086 kg/m3 to 1,226 kg/m3. 100 In their studies on the pelleting characteristics of Douglas fir, Lam et al. showed that a maximum calorific value of about 21 MJ/kg is achievable in a product that has less moisture uptake during storage (<4%wb). 101 Bruno et al. found that steam-treated pine has a lower moisture uptake (approximately 5.4%wb at lower relative humidity of 35%). 102 Ding et al. reported that Mongolian pine lumber, treated in pressurized steam, shows significantly lower moisture sorption and improved dimensional stability than that treated in atmospheric steam, and concluded that increased lignin content in the steam-exploded wood reduces the moisture uptake during storage. 103 In general, the hygroscopicity of the wood components is mainly due to the presence of the hydroxyl groups in hemicellulose and cellulose, which provide active sites for water molecules through hydrogen bonding. Most of the hemicellulose is removed during high-pressure steam explosion, further reducing the hydroxyl groups and inducing hydrophobicity in the material. 104
Hydrothermal carbonization
Hydrothermal pretreatment involves treatment of biomass in a hot, pressurized, aqueous environment. This is a wet process in which hot compressed water is used as the driving force for carbonization. This process has many names, including hydrothermal pretreatment, wet torrefaction, coalification, hot compressed water (HCW) treatment, and hydrothermal carbonization (HTC). 105 Bobleter performed work on wet torrefaction, or hydrothermal pretreatment of polymers derived from plants. 106 The process results in three products: gases, aqueous chemicals, and solid fuels. The temperatures for hydrothermal processing are 200–260°C at pressures up to 700 psi, and the solid product contains about 55–90% of the mass and 80–95% of the fuel value of the original feedstock. 107 The gas product is about 10% by mass of the feedstock, and aqueous chemicals, primarily sugars, make up the balance. 106,108,109 In their wet torrefaction study of lignocellulosic biomass, Yan et al. indicated that at lower temperatures (260°C), the calorific value of the biomass was 25.55 MJ/kg, greater by about 2.99 MJ/kg than the biomass torrefied at 300°C using a dry torrefaction technique. 107 Their studies indicated that the wet torrefied biomass has moisture in the range of 1.3–3.6% and higher fixed carbon (12.3–26.3%) than that produced using the dry method. In addition, the resulting material acted as though it were hydrophobic. Equilibrium moisture content values observed at two different relative humidity values—11.3% and 83.6%—at 30°C revealed that wet torrefaction at 260°C resulted in a lower moisture uptake of 5.3%, compared to dry torrefaction at 300°C (which was about 8.7%). 107 In their studies on the pelletization of hydrothermal treated biomass, Reza et al. found that higher mass and energy densities of 1,400 kg/m3 and 38–40 GJ/m3, respectively, are achievable at lower temperatures of 260°C. 110 In their review on HTC of biomass residuals, Libra et al. stated that this process can be used to produce chars with a wide range of applications, including soil amelioration, nutrient conservation, and increasing carbon stock in degraded soils. 111
Torrefaction
Torrefaction is a mild pyrolysis process conducted in the absence of oxygen at temperatures between 200–300°C for residence times of 15–120 min. During this process, the biomass partially decomposes, giving off volatiles, smoke-producing compounds, and a small amount of liquid. 112 The resulting char has approximately 70% of the initial weight and 80–90% of the original energy content, resulting in increased energy density. Torrefaction is considered beneficial for combustion or gasification conversion pathways and is known to make biomass more similar to coal and, consequently, more suitable for co-firing applications. Torrefaction can achieve results similar to low-rank coals and contribute to achieving several goals: upgrading energy density; increasing combustion and/or gasification reactivity; improving grindability and handling properties; significantly reducing the biodegradability of the cellulose and hemicellulose structures; significantly decreasing hemicellulose/hygroscopic characteristics; and lowering the hydrogen/carbon ratio to make it more energy efficient during thermochemical conversion. 113 Tumuluru et al. reviewed torrefaction and densification of torrefied biomass for bioenergy applications and concluded that the torrefied material acts similarly to coal in terms of physical, grinding, chemical, and storage properties, but has higher volatiles content. 82,114
The studies of Couhert et al., Sadaka and Negi, Pimchuai et al., and Roussset et al. on agricultural and woody biomass indicate that the moisture content reduces to <2%wb, and heating values increase to >22 MJ/kg. 115 –118 According to Bergman, densification of hot torrefied material at 225°C, without further cooling, reduces the densification pressure by a factor of 2 and doubles the throughput. 112 The increase in pelletability of torrefied material can be due to an increase in available lignin sites, breakdown of the hemicellulose matrix, and formation of fatty unsaturated structures. 112,119 –121 Bridgeman et al.'s studies on torrefaction of reed canary grass, wheat straw, and willow at 290°C showed an increase in carbon and energy content and reduction of sulfur and nitrogen content. 122 The research carried out on co-firing of torrefied wood, up to 9% (energy basis) mixed with coal, has led to the conclusion that there is still room for increasing the co-firing ratio (pulverizer's limits were not reached), and co-firing of torrefied wood can be a viable feedstock option for direct co-firing of biomass with coal. 123 Torrefaction, like most of the pretreatments discussed above, increases the relative percentage of ash content in the biomass, which can be minimized by prior washing/leaching processes. A significant drawback of torrefied material is its low volumetric mass density (180-300 kg/m3), which can be overcome by densification. Phanphanich and Sudhagar demonstrated that torrefied biomass can be ground to particle size distributions, sphericity, and particle surface areas similar to coal, which is desirable for increased efficiency in combustion and co-firing applications. 124 Current studies at Idaho National Laboratory using a Schulze-type ring shear tester indicate that torrefaction significantly decreases unconfined yield (shear) stress to reduce bridging and other flow problems in handling systems.
Densification
The densification process is critical to producing a feedstock material that is suitable as a commodity product. Densification improves storage as well as handling and conveyance, resulting in increased efficiencies throughout the supply system and co-firing in-feed. The controlled particle size distribution of densified materials improves feedstock uniformity and density, as well as conformance to predetermined conversion and supply-system specifications. 114 Another advantage of densification of pretreated biomass is reduced specific-energy consumption. With hot torrefied biomass, not only is the specific energy reduced, but the throughput of the mill also increases. Integrating densification with pretreatment helps to overcome technical, storage, and logistics problems encountered with raw biomass. Eranki found that densification of pretreated materials minimizes storage, handling, and transportation/logistical challenges. 125 Densification of biomass is commonly achieved using pellet mills and briquette presses that can process both formulated/blended and pretreated biomass.
Pelletizing
Pellet fuel is the most popular product for both biochemical and thermochemical conversion applications. Pelletizing is a compacting process in which finely ground biomass is converted into a homogenous dense fuel in cylindrical shape with free-flowing properties. 53,114,126 During the pelletization process, the incoming biomass is conditioned by controlled addition of steam, which helps gelatinize the starch and form more durable pellets. A pellet has uniform product characteristics in terms of size (length and diameter of 13–19 mm and 6.3-6.4 mm, respectively), shape (cylindrical), and unit densities (1,125-1,190 kg/m3). 127 A pellet mill consists of a perforated hard-steel die with one or two rollers. As the die and rollers operate, the feedstock is extruded through the die due to both roller pressure and die temperature, forming densified pellets. 128
Currently, pellets are the most appropriate biomass-derived fuel to be utilized in coal-fired plants. 112 Pelletization of the biomass addresses the low bulk density problems associated with biomass, which significantly impacts transportation costs and can also restrict the co-firing ratio due to the limited capacity of boiler input systems. Pelletization can be applied to various biomasses, woody as well as herbaceous. Additionally, the properties of pellets make them easier than loose biomass materials to grind, handle, and feed into the boiler. The process of pelletization includes drying, milling, conditioning, extruding, cooling, and separation of fines. 53 Using pellets in co-firing plants presents some challenges: lower energy densities compared to coal; disintegration of the pellets during loading and unloading due to mechanical damage, causing dust and handling problems; and the hydrophilic nature of pellets, due to which they tend to absorb moisture from their surroundings, resulting in swelling and creating handling and storage problems.
Briquetting
Briquettes are usually produced using hydraulic and mechanical presses. The advantages of briquette presses are that they can handle larger particle sizes and wider moisture contents (as high as 22%wb). Briquettes offer a number of advantages: better feed handling characteristics; higher calorific value; improved combustion characteristics, including more uniform combustion and a combustion rate comparable to coal; reduced particulate emissions; and more uniform size and shape. 129, 130 In addition, briquettes can typically be used in furnaces in which other solid fuels such as wood pellets are used. Co-firing of coal with biomass briquettes is also an interesting option because the properties allow for the use of existing coal infrastructure (transport, storage, feeding) without modification of the system. 130
Briquettes can be made from biomass or blends of biomass and coal-dust. 131 The typical diameter of briquettes is 30-100 mm. 53 Briquettes have a density of 800–1200 kg/m3, compared to 60–180 kg/m3 for loose biomass. The major limitation of biomass briquettes is uptake of moisture during storage, which can lead to an increase in biological degradation and loss of dimensional stability. Compared to pellets, biomass briquettes can be made using larger particle sizes and higher moisture content, and at lower specific energy consumption. However, briquettes have lower mechanical strength. Other briquetting technologies, like roller press, tabletizer, and cuber, may be useful in densifying the biomass for co-firing applications. 114
Screw extruder
In an extruder, a rotating screw moves biomass from the feed port, through the barrel, and against a die, resulting in a significant pressure gradient, friction, and shear. The heat due to wall friction at the barrel, internal friction in the material, and high rotational speed of the screw (∼600 rpm) increases the temperature of the biomass. 129 The heated biomass is further forced through the extrusion die to form either briquettes or pellets. In an extruder, external heat is also provided using band or tape heaters in case the temperature of the biomass material is not sufficient to reach a pseudo-plastic state for uniform and smooth extrusion. Due to compression and shear and the high temperature encountered during extrusion, biomass particles become relatively soft and lose their elastic nature, resulting in the formation of local bridges and interlocking particles. In general, smaller particle sizes (2–4 mm, depending on die diameter) are normally preferred during extrusion as they lead to better binding of the materials. In the die-head section, where the final compression takes place, the moisture is evaporated due to the high temperatures encountered (about 280°C). These high temperatures encountered by the biomass during extrusion result in charring of the material. Screw-extruded briquettes are more suitable for co-firing applications because the biomass is carbonized as a result of the high temperatures encountered during densification. 114
Agglomeration
Agglomeration increases particle size by “gluing” powder particles together. This technique has been widely used for a variety of materials, including hydrated lime, pulverized coal, iron ores, fly ash and cement, although little has been done on agglomeration of biomass. The most commonly used method of agglomeration is tumbling with binders, using a disk or pan agglomorator. During agglomeration, the fines, wetted by binder, form small, seed-type particles by nucleation, and these grow by coalescence into larger particles until they discharge from the pan. Typically, agglomerated materials are spherical, with diameters ranging from 4–6 mm depending on the residence time of the material in the agglomerator. Because of their spherical shape, agglomerated materials tend to flow better than cylindrical pellets and briquettes. Tumuluru et al. discussed the quality of material produced by different densification technologies for bioenergy applications in a review article on densification systems for developing uniform feedstock commodities for bioenergy applications. 114
Table 5 compares property values of coal with raw, pretreated, formulated, and densified biomass. Data on formulated pellets was obtained from a blend of switchgrass, corn stover, pine, and eucalyptus, each in equal proportions on a mass basis. Studies at Idaho National Laboratory have shown that this formulated material is easily pelletized with less energy than the raw individual feedstocks, especially switchgrass and eucalyptus, which require considerable energy in addition to pretreatment, such as steam conditioning, to pelletize separately. Pretreatment and densification bring the physical and fuel properties of biomass closer to those of coal, making it more suitable for co-firing at high percentages (>40%) with coal, even using existing equipment that is set up for firing coal alone (Table 5). 82,91,110,132 –134 Importantly, most of the pretreated and densified pellets included in Table 5 have handling, milling, and transportation characteristics similar to coal, so that they may be introduced into coal feed systems with only minimal modifications. Perhaps the major difference between coal and pretreated, densified biomass is the volatiles content. Table 6 summarizes the corresponding hurdles, desired attributes, and proven and potential solutions.
Properties Indicative of Co-firing Performance for Different Pretreated and Densified Biomass and Coal-based Fuels
SEP=steam exploded pellets; HTTP=hydrothermal pellets; TOP=torrefied pellets; HPL=hydrophyllic; HPB=: hydrophobic.
equal proportions of corn stover, switchgrass, eucalyptus, and pine.
Biomass Co-firing Hurdles, Desired Attributes, and Remedies
Co-Firing Approaches
Four approaches to injecting biomass into the coal-fired power generation process, each targeting a different area of the process, are shown in Figure 4. 38 The first three are called direct co-firing, which involves mixing of the biomass before the pulverizers (“co-milling” or blending) or direct injection (separate feed system for the biomass fuel). The fourth approach is an indirect co-firing method, which consists of separate boilers for the primary fuel and for biomass linked by a common connection to the steam cycle (i.e., the two boilers may share the same steam generator and steam turbine). 135 In Approach 1 in Figure 4, the coal/biomass mixture is blended on a conveyor belt and then milled and combusted together using standard coal pulverizers and burners. The compelling advantage of this approach is that the existing infrastructure of coal is used throughout, reducing investments in separate dedicated biomass process lines. According to Livingston et al., co-firing by co-milling is the preferred approach for coal-power stations that are adopting co-firing for the first time. 136 They have reported that this approach permits co-firing at levels up to 5-10% on a heat-input basis, with the major limitation being the ability of coal mills to process biomass with coal. This limitation can be minimized by torrefying and densifying biomass so that it behaves more like coal in terms of energy content and grinding behavior.
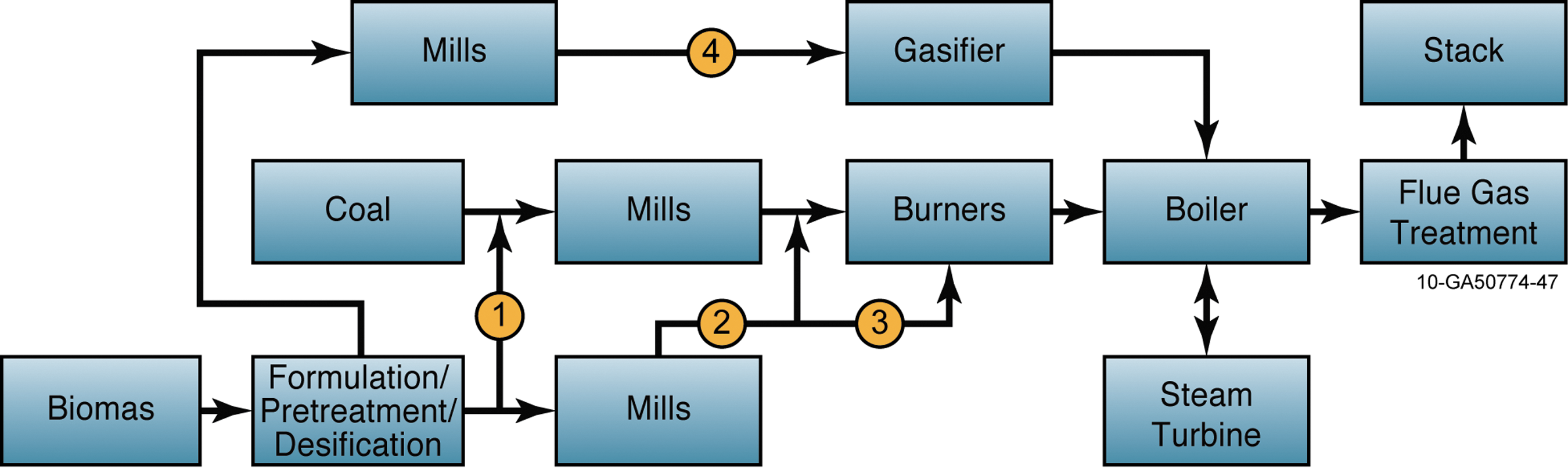
Alternative co-firing approaches (KEMA). Color images available online at
In Approach 2, biomass is dried and milled separately and then injected into the fuel lines carrying pulverized coal. By milling the biomass and coal separately, the throughput and reliability of the coal-pulverization process is maintained, while the biomass and coal are still combusted together, either in the original or modified coal burners. Alternatively, in Approach 3, biomass is preprocessed, fed, and combusted in separate, dedicated biomass burners. Finally, Approach 4 illustrates indirect co-firing of biomass (for example, through an upfront gasifier with co-combustion of the cleaned fuel gas in the main coal-fired boiler). The advantage of Approaches 3 and 4 is that the ash produced during combustion of coal and biomass can be kept apart—giving operators the flexibility to sell the ash separately to the construction and fertilizer industries. 27
Discussion
Biomass is a good candidate for co-firing with coal because both are solid fuels and the equipment designed for coal can be used for certain biomass feedstocks as well. However, significant differences exist in their chemical compositions; biomass has higher fractions of hydrogen, oxygen, and volatiles content and lower carbon fractions and calorific value compared to coal. These differences affect the combustion properties and limit the percentage of biomass that can be co-fired with coal. The methods described, including formulation, pretreatment, and densification, have been used to overcome the physical and chemical composition challenges of biomass. Many of these processes are expensive relative to the fairly low-value material that they produce, but great opportunities exist to combine different pretreatment and densification steps to save cost, minimize product losses, and reduce the number of separate operations.
The concentrations of inorganic elements in biomass, including silica, sulfur, and alkali metals, are an important specification because they form alkali silicates or alkali sulfates that melt or soften at temperatures as low as 700°C. 137 Formulation, or blending, can help modify the biomass composition such that different new products with specific end-use applications can be developed that have minimal slagging and fouling problems. In general, agricultural residues have high levels of silica and potassium, which can result in lower ash-fusion temperatures. Also, the presence of high levels of chlorine, calcium, and phosphorus in biomass can result in corrosion and reduced ash-fusion temperatures. Washing and leaching of raw biomass and formulated feedstocks help to minimize ash content, which further reduces corrosion, slagging, fouling (ash deposition), and sintering and agglomeration of the bed. Washing and leaching minimize the risk of system shut-down for ash-related maintenance by removing most of the alkalis, which is particularly problematic for herbaceous materials. Data describing the effect of washing and leaching on chemical compositional changes and the suitability of these processes for thermochemical conversion applications are scarce. EPRI recently experimented with washing and leaching herbaceous biomass and showed that this process effectively modifies the ash composition to produce clean biomass that could be used for firing/co-firing with coal.
Another major limitation of biomass co-firing is utilization of ash produced in Portland cement. American Society for Testing and Materials Standard (C618) has strictly specified that only coal ash be used in the mixture for Portland cement. Presently, DOE and other organizations are working to change this standard, which would allow the use of co-fired ash in Portland cement manufacturing. Success in this effort may encourage many utilities to use biomass because they would still be able to sell the ash that is produced. The research on chemical composition of ash produced due to formulation, washing, and leaching needs to be thoroughly explored and compared with standards.
Other major limitations of biomass compared to coal include higher moisture content and lower heating or calorific values, which limit biomass from co-firing applications. High moisture content in the biomass reduces storage stability, limits the percentage of biomass that can be co-milled with coal, and negatively affects combustion behavior. Thermal pretreatment like torrefaction reduces the moisture content in the biomass and makes the material more friable and easier to grind (grinding energy required is reduced by 70-80%). This improves particle size distribution, surface area, sphericity, and flow characteristics while increasing calorific value. The resulting hydrophobic nature makes the pretreated biomass storable for long periods without degradation and with reduced off-gas emissions during storage. 114,127 Torrefied biomass behaves much more like coal in handling and combustion systems than raw biomass; however, torrefied biomass still has a low bulk density, which is challenging in terms of transportation logistics. This limitation can be overcome by combining torrefaction with densification. The end product of other pretreatment methods, like steam explosion and HTC, are similar to that of torrrefaction, but must include a drying operation before densification and use in co-firing applications. 138,139 Acharjee et al. studied equilibrium moisture content of hydrothermal pretreatment of lignocellulosic biomass and found that most of the moisture resulting from the process is free water, which can be removed by simple filtration techniques. 140 The utility of these materials for co-firing applications is still in the research phase.
The physical property limitations of biomass for co-firing with coal include low bulk density and irregular particle size distribution. Biomass must be densified for co-firing applications. Densification of biomass, using pellet mills, briquette presses, or screw extruders helps to improve the rate and uniformity of combustion, reduces particulate emissions, decreases dry-matter losses during storage, produces better handling properties, and improves transportation and feeding characteristics. Densified biomass has been successfully used along with coal in power plants, although densification studies on blended or formulated feedstocks have not yet been thoroughly explored. Mixing of woody and herbaceous biomass can improve the densification characteristics of herbaceous biomass, which are difficult to densify due to low lignin content. This type of formulated feedstock can produce more durable pellets with less energy consumption. A primary limitation of densified biomass is moisture absorption during storage, which results in dry-matter losses and off-gas emissions. Densification of pretreated biomass can reduce these limitations.
Figure 5 portrays possible combinations of formulation, pretreatment, and densification methods that can jointly improve the physical and chemical properties of biomass. This integrated approach helps to develop uniform feedstocks with minimum physical and compositional issues for biochemical, thermochemical, and combustion applications, including co-firing with coal at high percentages. Future research should address how these individual operations impact one another and how they can be combined. In this effort, particular attention must be paid to specifications, such as low moisture and ash contents and high mass and energy densities. Often achieving one specification, such as low ash content through chemical pretreament, can negatively impact other important specifications, such as calorific value. Consequently, an integrated approach that weighs the relative importance of different feedstock specifications is necessary.
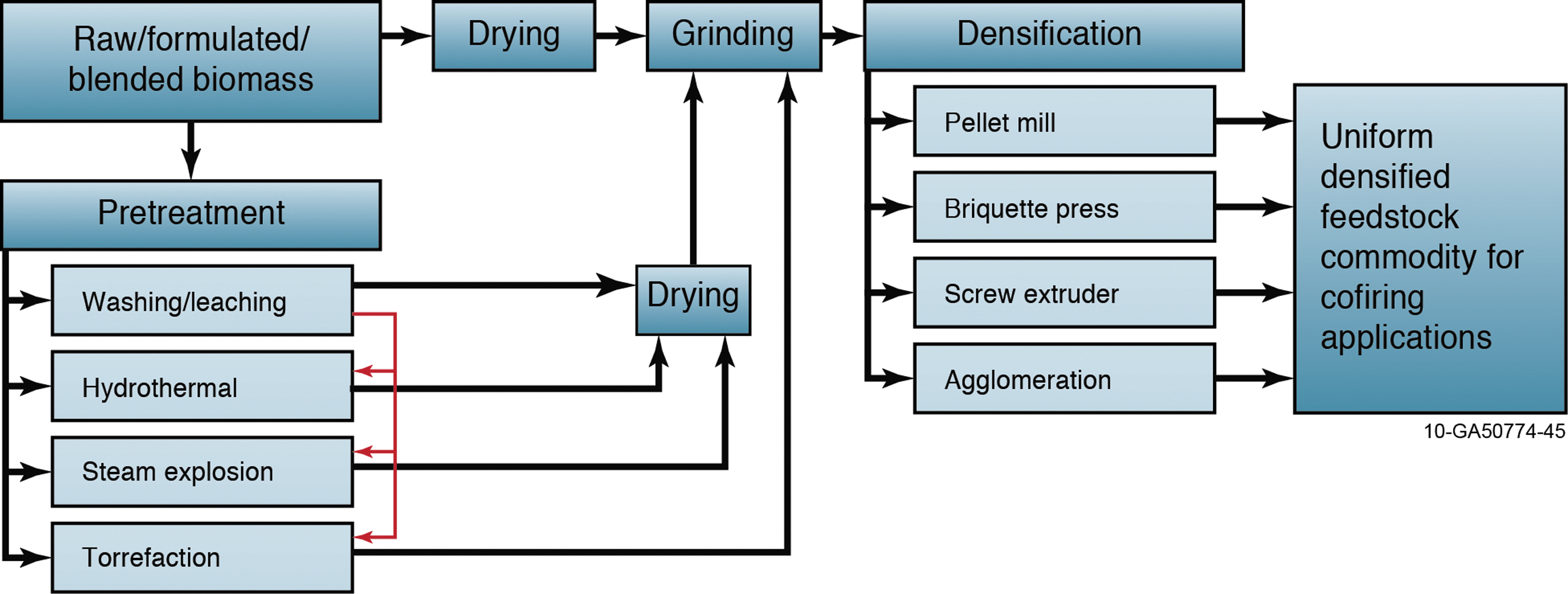
Flow diagram for producing uniform densified feedstock commodity for biochemical and thermochemical conversion or for combustion co-firing applications. Color images available online at
Hamalainen developed a figure for ranking different fuels available for combustion in a fluidized-bed gasifier. 141 He compared various plastic and biomass fuels with respect to standard fuels, like peat, brown coals, bituminous coal, and petroleum coke, that are used in standard boiler design. Figure 6 presents a modified version of the Hamalainen description, which ranks formulated, pretreated, and densified biomass on their compatibility with standard coal fuels in regards to ash and energy content. 141 It is clear that raw biomass is more challenging than pretreated biomass, primarily due to energy and mass density differences, ash-melting behavior, and fouling of the boiler, resulting in new requirements for boiler design and operation. 141 Pretreating biomass feedstocks, especially formulation, washing, and leaching, will minimize these operational constraints. Formulation/densification can address other challenges such as low bulk and energy density of biomass. Pretreated, formulated, and densified biomass can fit into the existing boiler design with little or no modifications.
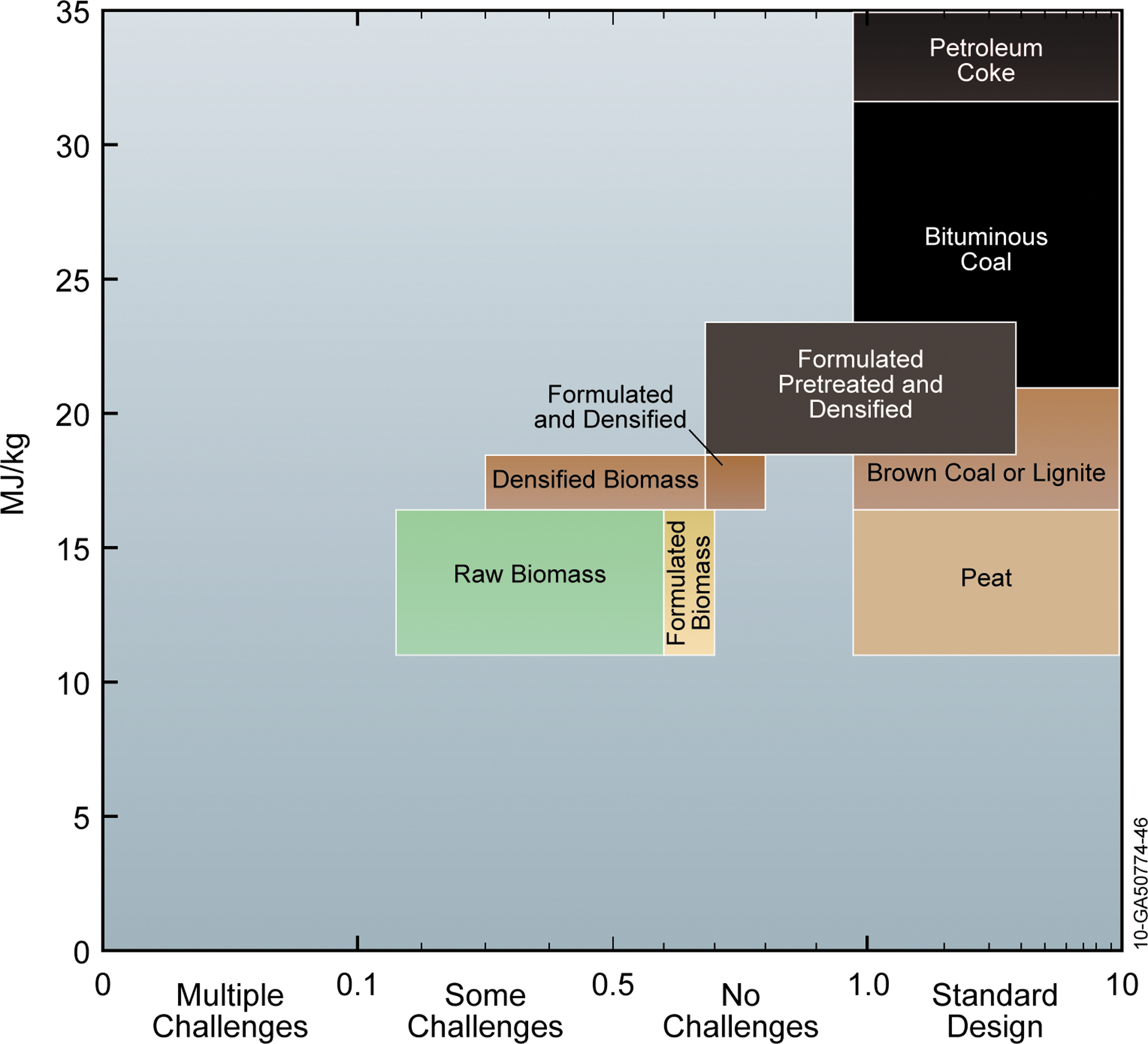
Modified Hamalainen plot showing progression of raw biomass from low energy density, challenging (in terms of physical and chemical properties) fuel into a formulated, pretreated, and densified feedstock that is compatible with coal in standard design coal power plants.
141
Color images available online at
Table 7 compares green woodchips and wood pellets, which are presently used for co-firing with coals like bituminous and Powder River Basin (PRB). In the Chariton Valley Biomass Project, feedstock specifications such as moisture, ash, volatiles, fixed carbon, heating values, and elemental composition were used as the basis for successful co-firing of switchgrass with PRB coal at the commercial scale.
42,114,127,142,143
–152
Using the Chariton Valley Biomass Project as a reference, the coal and biomass specifications in Table 7 are divided into Tier-1 and Tier-2 based on their impact on the efficiency of the boiler. Most of the Tier 1 specifications, such as moisture content, energy and bulk densities, ash composition, grindability, particle-size distributions, and hydrophobicity can be achieved by pretreatment and densification (Table 5). The table indicates that there are certain Tier-2 properties that are not available for raw and densified biomass. Establishing these values will aid in improved understanding of co-firing efficiencies so that the maximal percentage of biomass that can be co-fired with coal can be determined for particular situations. Data for raw, formulated, pretreated, and densified biomass in the following areas will be especially useful in understanding the co-firing characteristics of biomass with coal: • Pulverized geometric particle size; • Minimum slag/fouling limits (percent co-firing/temperature °C); • Particle burning times in premixed flame (sec); • Free swelling index; • Rheological properties such as unconfined yield strength, wall friction angle, angle of internal friction, and compressibility, elastic recovery (springback), angle of repose, and permeability; • Reactivity data in terms of spontaneous combustion and explosion index; • Storage properties such as auto-ignition, spontaneous combustion, explosion index, and biological degradation and hydrophobicity at different storage temperatures.
=Values to be established based on research.
Flowability is defined as the ratio of the major principal stress to the unconfined yield strength. 153
PRB=Powder River Basin coal, HHV=higher heating value, HGI=Hardgrove grindability index, HACCP=hazard analysis and critical control point.
Among the various approaches discussed, the challenges associated in co-firing raw biomass include the necessity to introduce a dedicated biomass infrastructure (e.g., feeding, milling, storage, conveying) into the existing coal system. A more expensive alternative (in terms of investment costs) is introducing more advanced co-firing modes, such as parallel co-firing or indirect co-firing, in which not only fuel preparation and feeding lines, but also conversion units for biomass and coal, are independent. Integrating formulation, pretreatment, and densification to develop a biomass feedstock with specifications closer to coal will help to increase the co-firing ratios with coal in standard coal handling and combustion systems with little or no modification.
Conclusions
Biomass co-firing has potential benefits, such as GHG reduction and distributed, sustainable power production. Utility-scale biopower co-firing can be a near-term renewable-energy option for improving the environmental footprint of existing coal plants. One primary requirement is that co-firing at existing plants be economically viable, although economic risks are tied to uncertainties, including technical challenges, environmental regulations, and feedstock availability. Enhancing biomass quality and using pretreatments combined with densification will dramatically improve supply, transport, storage, and combustion of biomass feedstock materials. These research areas have not been thoroughly explored as yet.
Coal and biomass are solid fuels that share many commonalities—both contain carbon, hydrogen, oxygen, water, ash-forming elements, nitrogen, and sulfur. But significant differences also exist between coal and biomass. For example, the concentration of oxygen in wood is 45% by weight, whereas it is only 2% in anthracite coal on a dry ash-free basis. In addition, fresh wood typically contains 50% of water by weight and has high volatile matter content, whereas the moisture content for bituminous coal is approximately 5% with a relatively low volatile matter content. Finally, wood-based fuels usually have low sulfur content, but can also have very high chlorine content.
The main constraints for co-firing biomass with coal, especially in the more common direct co-firing operation, are handling, storage, milling, and feeding problems, deposit formation (slagging and fouling), agglomeration, corrosion or erosion, and ash-utilization issues. Chemical composition issues of the biomass can be reduced by methods such as formulation, washing, and leaching, which can save costs involved in maintenance of co-firing systems and minimize ash-related issues such as slagging and fouling. The bulk density impediment can be overcome by densification using a pellet mill or briquette press. Pretreatment of the biomass using techniques such as torrefaction, steam explosion, and hydrothermal carbonization can help produce a friable product that can be ground easily with less grinding energy and better particle size distributions. Pretreatment also results in significant changes in biomass chemical composition, reducing hydrogen, oxygen, and volatile matter, while increasing carbon content and calorific values. Also, thermally or hydrothermally pretreated biomass is hydrophobic in nature, making it suitable for long-term storage without any degradation. Formulation and pretreatment followed by densification can help to modify the physical properties, chemical composition, and storage behavior of the biomass for improved co-firing with coal. Physical and chemical properties of pretreated and densified biomass are comparable with some coals, like North Dakota lignite and PRB coal (Black Thunder), available in the US. Pretreated and densified biomass can be co-fired with coal without separate feeding lines. Tier-1 and Tier-2 properties of formulated, pretreated, and densified biomass should be thoroughly evaluated for co-firing high percentages with coal in direct co-firing systems with little or no modifications to the mills, feeding lines and burners.
Footnotes
Acknowledgments
The authors would like to acknowledge Dr. David N. Thompson and Dr. Vicki S. Thompson of Idaho National Laboratory for discussions and information provided in the formulation section of the manuscript. The authors would also like to acknowledge Leslie Park Ovard for her valuable contribution to the manuscript and Gordon Holt, Allen Haroldsen, Barney Hadden, and Lisa Plaster from Idaho National Laboratory's R&D Publications Support Team for their editorial and graphics creation assistance. This work was supported by the DOE, Office of Energy Efficiency and Renewable Energy under DOE Idaho Operations Office Contract DE-AC07-05ID14517. Accordingly, the US government retains and the publisher, by accepting the article for publication, acknowledges that the US government retains a nonexclusive, paid-up, irrevocable, worldwide license to publish or reproduce the published form of this manuscript, or allow others to do so, for US government purposes.
Author Disclosure Statement
No competing financial interests exist. This information was prepared as an account of work sponsored by an agency of the US government. Neither the US government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product, or process disclosed, or represents that its use would not infringe privately owned rights. References herein to any specific commercial product, process, or service by trade name, trademark, manufacturer, or otherwise, do not necessarily constitute or imply its endorsement, recommendation, or favoring by the US government or any agency thereof. The views and opinions of the authors expressed herein do not necessarily state or reflect those of the US government or any agency thereof.
