Abstract
Background:
In 2003, patients in the Netherlands gained legal access to medicinal cannabis due to legislative measures by the Dutch government. Studies on patient experiences related to medicinal cannabis usage are necessary to complement the data obtained from randomized controlled trials.
Aim:
To gain insight into patient's experiences with the use of medicinal cannabis in the Netherlands.
Methods:
We conducted an observational longitudinal cohort event monitoring study. Data were collected through online questionnaires. From March 2021 to March 2022, patients were included. After registration, participants received four online questionnaires over a 12-month period. Descriptive statistics were used to present the included variables using MS Excel 2022.
Results:
A total of 251 participants were included in this study at baseline. The mean age of the participants was 55 ± 15 years. A total of 140 participants (56%) were female. Oil was the most commonly used dosage form (52%), followed by flos (35%) and granular form (13%). Medicinal cannabis was prescribed by a general practitioner (51%) or by a specialist doctor (47%). The most prevalent indications for use were chronic pain (n = 151, 60%), followed by ADHD (n = 18, 7%), cancer (n = 18, 7%), and multiple sclerosis (n = 17, 7%). A total of 53 participants (21%) reported one or more adverse drug reactions (ADRs), of which 19 participants were new users. In total, 85 ADRs were reported. The most commonly reported ADRs were dizziness (n = 10, 4%), somnolence (n = 5, 2%), dry mouth (n = 5, 2%), headache (n = 4, 2%), and increased appetite (n = 4, 2%). Most participants indicated that their quality of life had improved since the start of medicinal cannabis.
Conclusions:
Our research offers insights into patient experiences with the use of medicinal cannabis in daily practice. The participants experienced positive effects on their quality of life, as medicinal cannabis was well tolerated. This can help to inform clinical practice.
Background
Since 2003, the Dutch government has passed legislation enabling cannabis-based products to be prescribed as unregistered medicines. This provided access to medicinal cannabis for patients on medical grounds under the supervision of a doctor and pharmacist. In the Netherlands, all activities related to medicinal cannabis are strictly regulated, and the Dutch Office of Medicinal Cannabis (BMC) is responsible for this process. This means that authorizations have to be obtained for the cultivation, distribution, and sale of medicinal cannabis. To date, only one company (Bedrocan) has been authorized to cultivate cannabis for medicinal use.
The efficacy of cannabis in various indications is still a subject of debate. In general, limited evidence exists for an effect on pain and spasticity from multiple sclerosis and neuropathic pain, cancer-related pain, insomnia, and anxiety. 1 Other indications for which medicinal cannabis is used include HIV/AIDS, epilepsy, chemotherapy-related nausea and vomiting, posttraumatic stress disorder, Parkinson's disease, and treatment-resistant glaucoma. 2
The main biologically active constituents of cannabis are cannabinoids. The cannabis plant contains over 144 different cannabinoids. They exert their effect on the body by binding to cannabinoid receptors types 1 and 2. The two most prevalent cannabinoids are delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD). 2 In the Netherlands, cannabis can be prescribed in the form of oil, ointment, flos (dried flower), and granules. These different types of products differ in THC and CBD content.
Access to cannabis-based medicinal products has increased globally due to legislative changes. However, patient access still faces barriers such as a paucity of high-quality evidence surrounding the clinical effects of cannabis-based medicinal products. 3 Although randomized controlled trials are the gold standard for clinical evaluation, they are subject to limitations: cannabis-based medicinal products have a complex pharmacology and contain many different compounds (cannabinoids, flavonoids, and terpenes), which can result in heterogeneity between finished cannabis-based medicinal products. This makes it difficult to extrapolate the results of clinical trials. Furthermore, it is difficult to identify an appropriate placebo control, and cannabis-based medicinal products are expensive. 4 Therefore, other sources of information are needed in addition to the evidence provided by randomized controlled trials.5,6 Insight into patient experiences can be helpful to inform clinical practice and is an important component of pharmacovigilance.
The aim of this study is to gain insight into patient experiences with the use of medicinal cannabis in the Netherlands.
Methods
Study design and data collection
We conducted an observational longitudinal cohort event monitoring study. Data were collected through online questionnaires with the Lareb Intensive Monitoring system. 7 Patients who already used medicinal cannabis as well as new users of medicinal cannabis were included, regardless of age and indication. Recreational cannabis use and use of cannabis for medicinal purposes obtained at the coffee shop were excluded.
Patients were invited to participate in this study by means of a leaflet that was added to the packaging of the medicinal cannabis by the distributor Fagron NL BV or having received a leaflet from the pharmacist during the dispensing of the medicinal cannabis. After registration, participants received four online questionnaires over a 12-month period: at the time of enrollment and at 2 months, 6 months, and 1 year after enrollment. If there was no response, a maximum of two reminders was sent. If the questionnaire was not completed after 2 weeks, the questionnaire was closed, and the participant was considered lost to follow-up. At all times, participants were able to withdraw from the study. These participants were also considered lost to follow-up. Questionnaires that had already been completed were included in the analyses.
Questions related to patient characteristics (age, sex), the type of medicinal cannabis product used, indications for use, reasons for choosing a specific medicinal cannabis product, product satisfaction, perceived adverse drug reactions (ADRs), cost of medicinal cannabis, and quality of life. The indication for use and ADRs were coded using the Medical Dictionary for Regulatory Activities. 8 ADRs were classified as serious if they resulted in (prolonged) hospitalization, a life-threatening situation, death, permanent disability, or congenital abnormalities. 9 The data were stored in an Oracle database.
Population
Patients registered between March 1, 2021, and March 1, 2022, were included in this study. Current and new users of medicinal cannabis were included. Participants were considered long-term users if they had been using medicinal cannabis for more than 6 months. Participants were considered new users if they had started using medicinal cannabis <6 months ago or were about to start. If patients did not complete all four questionnaires, data for these patients were included for the available follow-up period.
Analyses
Descriptive statistics were used to present characteristics of the population, the type of medicinal cannabis product used, indications for use, the number of patients reporting an ADR, incidence of ADRs, and quality of life. Demographic data were analyzed using the mean (±standard deviation) or frequency (%) as appropriate. Data extraction and analysis were performed using Microsoft SQL Server Management Studio, v18.12.1, 2022 and Microsoft Excel 2022. The duration of use and sex were univariably tested for their association with the occurrence of ADRs using the χ 2 -test.
Results
Between March 1, 2021, and March 1, 2022, 251 patients were included in this study (Fig. 1). The mean age of the patients was 55 ± 15 years. A total of 140 patients (56%) were female. Oil is the most commonly used dosage form (52%), followed by flos (35%) and granular form (13%). Most of the patients had been using medicinal cannabis for more than 6 months (79%). The other patients (21%) started using medicinal cannabis <6 months ago. Full demographic details are presented in Table 1.
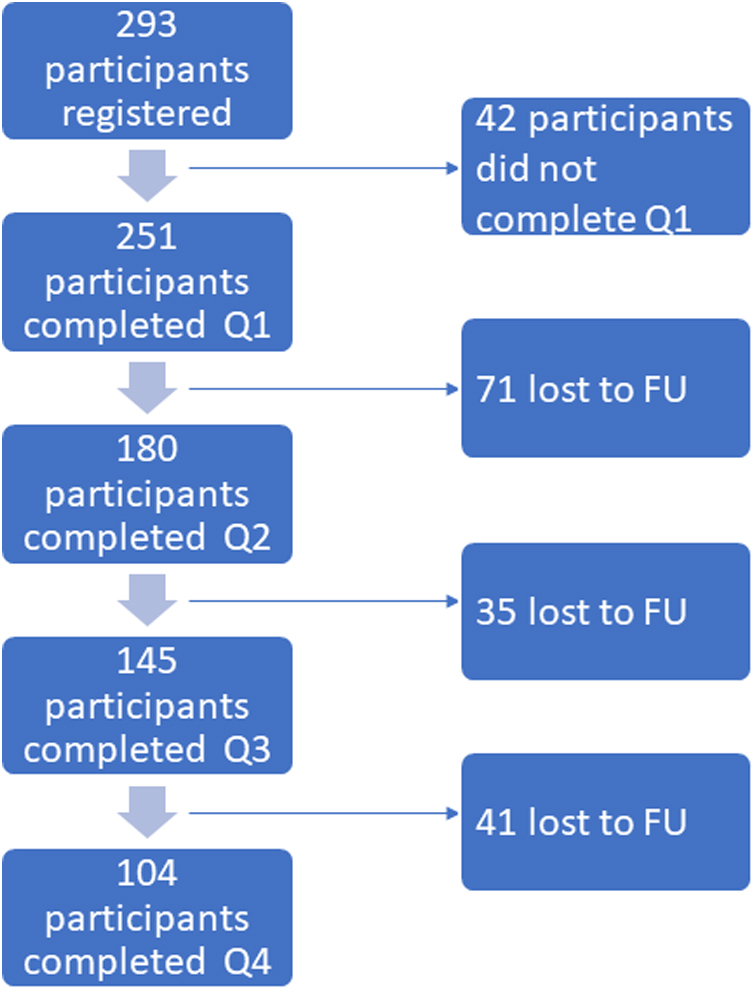
Flow diagram of patient inclusion.
Baseline Characteristics of the Participants
Sixty percent of male patients had used cannabis obtained in the coffeeshop in the past, and 15% still used cannabis obtained in the coffeeshop in addition to the medicinal cannabis obtained in the pharmacy. For female patients, these percentages were 32% and 9%, respectively (Table 1). The most common reason to start using medicinal cannabis obtained in the pharmacy was the improved quality of the medicinal cannabis obtained in the pharmacy. The most common reason to keep using cannabis obtained in the coffeeshop was the lower efficacy of medicinal cannabis obtained in the pharmacy.
The medicinal cannabis product that was used by most patients was cannabis flos with a content of THC 22% and CBD <1% (n = 85), followed by cannabis oil with a content of THC 1.3% and CBD 2.0% (n = 79) (Fig. 2). Eighty-nine percent of the patients who used cannabis oil were satisfied with its use, compared with 82% of the patients who used cannabis flos and 78% of the patients who used cannabis in granular form.

Number of participants who use a medicinal cannabis product and number of ADRs reported. ADRs, adverse drug reactions.
Medicinal cannabis was prescribed by a general practitioner (51%) or by a specialist doctor (47%). In 2%, the prescriber was unknown. Figure 3 details the top 10 indications for which medicinal cannabis was used. The most prevalent indications for use were chronic pain (n = 151, 60%), followed by ADHD (n = 18, 7%), cancer (n = 18, 7%) and multiple sclerosis (n = 17, 7%). Patients could select more than one indication.

Top 10 indications for which medicinal cannabis was used.
Adverse drug reactions
A total of 53 patients (21%) reported one or more ADRs, of whom 19 patients were new users. Sex was not significantly associated with the occurrence of ADRs (χ 2 = 2.87; p = 0.090). In total, 85 ADRs were reported. The most commonly reported ADRs were dizziness (n = 10, 4%), somnolence (n = 5, 2%), dry mouth (n = 5, 2%), headache (n = 4, 2%), and increased appetite (n = 4, 2%). No serious ADRs were reported. Long-term users (n = 33, 17%) as well as new users (n = 19, 37%) reported ADRs. More new users than long-term users reported ADRs (χ 2 = 9.87, p = 0.0017). New users mostly reported dizziness (n = 5) and headache (n = 4), whereas long-term users mostly reported dizziness (n = 5) and increased appetite (n = 4).
Seventy-eight percent of the ADRs occurred within 1 day or days of starting medicinal cannabis. Lung disorders, anxiety, gynecomastia, and mood disorders occurred months or years after the start of treatment. The most common impact of ADRs was “not burdensome” (n = 31, 39%), followed by “slightly burdensome” (n = 25, 32%). Most ADRs were reported when using Bedrocan flos (n = 28), followed by CBD/THC 2%/1.3% oil (n = 24). These products were also most often used (Fig. 2).
Most patients who use medicinal cannabis in flos form have experienced feeling “high” (65%) compared to the granular form (38%) and the oil form (18%). Additionally, tolerance and withdrawal effects occurred more in patients who used medicinal cannabis in flos form compared to the granular or oil form (Table 2).
Feeling High, Tolerance, and Withdrawal Effects
Concomitant medication
Sixty-eight percent of the patients reported that they were currently using other prescribed medications. The most commonly reported comedications were paracetamol, cholecalciferol, and omeprazole.
Quality of life
Most patients indicated that their quality of life had improved since the start of medicinal cannabis. More long-term users reported an improvement in their quality of life compared to new users, 93% versus 62%, respectively (χ 2 = 36.57; p < 0.00001) (Fig. 4). New users indicated that it was too soon to evaluate the impact on their quality of life since they had only recently initiated medicinal cannabis use. Therefore, they neither agreed nor disagreed (Fig. 4).

Patient-reported improvement in quality of life.
The cost of medicinal cannabis is a burden for the majority of patients (73%, Table 3).
Cost of Medicinal Cannabis
Discussion
This study provides insight into patient experiences with the use of medicinal cannabis in the Netherlands. Most patients in this study used medicinal cannabis for chronic pain. The reported ADRs were mostly mild and occurred within days of starting medicinal cannabis. No serious ADRs were reported. Patients were satisfied with the medicinal cannabis product they used and reported an improvement in their quality of life. The results showed that the cost of medicinal cannabis is a burden for the majority of patients.
The literature also shows that pain management is one of the most commonly reported reasons why people use medicinal cannabis.10–14 A systematic review and meta-analysis by Wang et al. 15 to determine the benefits and harms of medicinal cannabis for chronic pain concluded that noninhaled medicinal cannabis results in a small improvement in pain relief, physical functioning, and quality of sleep in patients with chronic pain compared with placebo. No improvement was seen in emotional or social functioning. A systematic review by Nugent et al. 16 that included 27 studies on the effects of cannabis on chronic pain concluded that cannabis may alleviate neuropathic pain in some patients, but insufficient evidence exists for other types of chronic pain.
Cannabinoids are thought to affect pain through various pathways, including the endocannabinoid system, which has receptors in the central nervous system, periphery, immune system, and hematologic system. The various analgesic and anti-inflammatory properties of cannabinoids may positively influence the perception of pain across different conditions. 17
In our study, the reported ADRs were mild and not very burdensome. Dizziness, somnolence, dry mouth, headache, and increased appetite were the most commonly reported ADRs. No serious ADRs were reported. Most ADRs occurred within 1 day or days of starting medicinal cannabis and usually did not require treatment.
The literature also shows that ADRs of cannabis products are generally mild in nature. In a retrospective study where medicinal cannabis users were asked to answer questionnaires, dry mouth, drowsiness, and dizziness were the most commonly reported ADRs. 18 A study by Ergisi et al., 19 who studied the changes in health-related quality of life and clinical safety in patients prescribed medicinal cannabis, found that 30.1% of patients reported adverse events, of which nausea, dry mouth, dizziness, and somnolence were the most common. A systematic review that included 32 studies concluded that medicinal cannabis resulted in a small increased risk of transient cognitive impairment, vomiting, drowsiness, impaired attention, nausea, and dizziness. 15 A meta-analysis by Zeraatkar et al. 20 showed that while adverse events were common in patients using medicinal cannabis, serious adverse events were uncommon. Few patients discontinued medicinal cannabis due to adverse events, suggesting that most adverse events were transient and/or outweighed by benefits.
Most patients in our study were very satisfied with the medicinal cannabis product they used. Our study included more long-term users than new users; however, both groups experienced a positive effect of medicinal cannabis on their symptoms. More long-term users reported that their quality of life had improved since the start of medicinal cannabis than new users. This is also to be expected if a product is used for a long time. New users often indicated that they had just started using medicinal cannabis and that the effect on quality of life had yet to be seen.
These positive effects of medicinal cannabis on quality of life and treatment are also found in the literature. A prospective study of 1186 elderly people showed that 59% found treatment with medicinal cannabis successful and 94% reported an improvement in their condition after 6 months of use. 21 Similar results were found in other studies: in an online survey study with a total of 1429 participants, an 86% reduction in symptoms as a result of cannabis use was reported. 13 Cahill et al. reported an improvement in well-being in 62% of the 214 participants. However, while their results showed that patients found cannabis treatment effective for a broad range of medical conditions, cannabis was not a remedy for all the conditions investigated. 22 In a prospective registry where patients were administered quality of life questionnaires, statistically significant improvements in health-related quality of life at 1, 3, and 6 months were demonstrated. 23 Meng et al. reported in a prospective longitudinal observational study of 757 patients an improvement in quality of life compared with baseline in patients who continued using cannabis for 6 months. 24
For a large proportion of patients, the cost of medicinal cannabis is a problem. The number of medicinal cannabis sales in the Netherlands has been declining since 2018, when insurance reimbursements were halted. 25 Because medicinal cannabis is not a registered medicine and it is not considered rational pharmacotherapy because of a lack of evidence of efficacy, it is not reimbursed. 26 Until a few years ago, there was still a leniency scheme with health insurers, but that scheme was discontinued. 27 Therefore, patients have to pay for medicinal cannabis themselves. A substantial part of the patients reported having a low income, resulting in medicinal cannabis taking up a large part of their budget. Patients report spending up to hundreds of euros per month on their medicinal cannabis.
This was also reported in a study by Garcia-Romeu et al. 28 where respondents also voiced concerns about the high cost and lack of insurance coverage resulting in barriers to implementing and maintaining treatment.
In July 2022, the National Health Care Institute in the Netherlands advised that the medicine CBD (Epidyolex®) be reimbursed from the basic health insurance package. This is the first time that reimbursement for a substance (CBD) found in medicinal cannabis has been advised. 29
Strengths and Limitations
Web-based intensive monitoring is an observational cohort study that is able to study the use and ADRs of medicinal cannabis in daily practice compared with clinical studies, which have strict inclusion and exclusion criteria. Furthermore, in our study, the patient is the source of information. An advantage of this is that ADRs are reported by the person who has actually experienced the reaction.
In our study, participants were invited to participate by means of a leaflet they received from their pharmacist during the dispensing of the medicinal cannabis. However, not all patients who were eligible participated in this study. There are ∼6700 patients who use medicinal cannabis under the supervision of a doctor and pharmacist in the Netherlands. 30 Of this total number of users of medicinal cannabis, 251 participated in our study (3.7%). This may lead to nonresponse bias. Information on patients who declined to participate in this study is not known. Whether the participants in this study are representative of all patients using medicinal cannabis is unclear.
The number of participants declined in each follow-up questionnaire (questionnaire 2: 72%, questionnaire 3: 58%, questionnaire 4: 41%). This loss to follow-up may introduce selection bias, leading to a selection of patients benefitting the most from medicinal cannabis or a selection of patients who do not experience ADRs leading to withdrawal of medicinal cannabis or a selection of patients who can afford to use medicinal cannabis for a long time. It is not known whether participants who no longer wanted to participate in this study did so because of withdrawal of medicinal cannabis due to insufficient effect, ADRs, or cost. This loss to follow-up can impair the generalizability of our results to all Dutch medicinal cannabis users.
Twenty-one percent of participants reported an ADR. This percentage is lower than that seen in other studies where 30%–40% of patients reported an ADR.23,31,32 It is possible that due to the large proportion of long-term users in our study, a selection of participants has occurred who benefit from medicinal cannabis and did not experience many ADRs. Furthermore, ADRs that occurred at the start of the medicinal cannabis are possibly less well remembered by long-term users.
Conclusions
Our study provides an overview of patient experiences with the use of medicinal cannabis in daily practice. The use of medicinal cannabis as reported by patients over a 12-month period was well tolerated and improved the quality of life of the participants. By conducting monitoring studies, we can gather valuable information about the safety and patient experiences of medicinal cannabis over the long term in the real world. These data can guide and shape clinical practice.
Footnotes
Acknowledgments
We would like to express our sincere gratitude to Thomas Lieber for the data extraction. We also wish to thank the Transvaal Apotheek, Clinical Cannabis Care BV, Verenigde Apotheken Limburg and Fagron NL BV for their help with the inclusion of participants. Finally, we are grateful to all of the participants who generously gave their time and effort to this research.
Authors' Contributions
All authors contributed to the design and implementation of the research. C.E. analyzed the data and took the lead in writing the article. All authors provided feedback and approved the final article.
Ethical Approval and Consent to Participate
All subjects gave their informed consent for inclusion before they participated in the study. This is an observational study. The Ethics Committee of Brabant has confirmed that no ethical approval is required.
Availability of Data and Materials
Due to the data protection policy of the Netherlands Pharmacovigilance Centre Lareb, the data cannot be publicly shared in a repository. However, the datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request and with permission of the Dutch Office of Medicinal Cannabis.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was financed by the Dutch Ministry of Health, Welfare and Sport. Grant Number - 201865006.026.201.
