Abstract
Delandistrogene moxeparvovec is a gene transfer therapy for Duchenne muscular dystrophy (DMD) that uses an adeno-associated viral vector to deliver a micro-dystrophin transgene to skeletal and cardiac muscle. This study evaluated the long-term survival and cardiac efficacy of delandistrogene moxeparvovec in a DMD-mutated (DMDMDX) rat model of DMD-related cardiomyopathy. DMDMDX male rats, aged 21–42 days, were injected with 1.33 × 1014 viral genomes/kilogram (vg/kg) delandistrogene moxeparvovec and followed for 12, 24, and 52 weeks. Ambulation was recorded via the Photobeam Activity System, whereas echocardiograms, cardiomyocyte contractility, calcium handling, and histological analysis of fibrosis were used to evaluate cardiac disease at 12-, 24-, and 52-weeks post-treatment. A separate cohort of rats was used to assess the impact of delandistrogene moxeparvovec on survival. Treatment with delandistrogene moxeparvovec extended median survival in DMDMDX rats to >25 months versus the 13-month median survival in saline-control-treated DMDMDX rats. Compared with saline control, delandistrogene moxeparvovec therapy elicited statistically significant improvements across cardiac parameters approaching wild-type values with additional benefits in mobility, histopathology, and fibrosis observed. Transgene expression was maintained up to >25 months and micro-dystrophin expression was broadly distributed across skeletal and cardiac muscle. Taken together, these findings demonstrate long-term cardiac efficacy and improved survival following delandistrogene moxeparvovec treatment in DMDMDX rats.
INTRODUCTION
Duchenne muscular dystrophy (DMD) is a rare, X-linked neuromuscular disease caused by mutations in the dystrophin (DMD) gene that disrupt the production of functional dystrophin protein. 1 The absence or insufficient production of dystrophin can lead to myofiber damage, muscle degeneration, and necrosis, which results in progressive muscle weakness and wasting. 2,3 DMD is associated with early mortality often from cardiac or respiratory failure, with a life expectancy of approximately 30 years. 4,5 Cardiomyopathy has become the most common cause of death in patients with DMD. 6,7 Dystrophin plays various roles in the cardiac myocyte, but the underlying DMD pathophysiology is in part linked to calcium dysregulation. 8 Calcium excess due to membrane instability is a primary mechanistic contributor to disease progression. 8
Delandistrogene moxeparvovec is an adeno-associated viral (AAV) vector-based gene therapy approved in the United States for the treatment of patients with DMD at least 4 years of age with a confirmed mutation in the DMD gene, regardless of ambulatory status. 9 As of May 2024, delandistrogene moxeparvovec is approved in the UAE, Qatar, Kuwait, Bahrain, and Oman for the treatment of ambulatory pediatric patients with DMD aged 4–5 years with a confirmed mutation in the DMD gene. 9 –14 Delandistrogene moxeparvovec has been designed to address DMD using a recombinant AAV serotype rh74 (rAAVrh74) vector for targeted production of delandistrogene moxeparvovec micro-dystrophin, an engineered dystrophin protein that retains key functional domains of the wild-type (WT) protein (Supplementary Fig. S1). 15
As clinical trial data primarily focus on the functional evaluation of ambulation in patients, there is a need to better understand cardiac outcomes following treatment with delandistrogene moxeparvovec. In pre-clinical studies using mdx mice, reductions in fibrosis and improvements in skeletal muscle function were observed following systemic delivery of delandistrogene moxeparvovec. 16,17 Although the mdx mouse is a valuable genetic and biochemical model, it manifests a mild cardiac phenotype relative to human disease. 17 –21 DMDMDX rats are a valuable alternative model as they demonstrate cardiac dysfunction that more closely resembles the cardiac phenotype seen in patients with DMD. 17,18
Here we report the long-term therapeutic benefit of delandistrogene moxeparvovec in DMDMDX rats, including improved functional and cardiac outcomes with prolonged survival.
MATERIALS AND METHODS
Ethics
All procedures were in compliance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and were approved by the local institution's animal care and use committee.
Animal model
Male Sprague-Dawley DMDMDX and WT rats, obtained from the University of Nantes (France) 17 and Taconic Biosciences, were intravenously administered saline or delandistrogene moxeparvovec at 21–42 days of age and euthanized (Euthasol 150–200 mg/kg; Supplementary Table S1) for tissue collection at predetermined time points. Operators of all functional outcomes were blinded to the treatment group (see Supplementary Table S2 for an overview of the study design and outcomes; see supplementary methods for further details).
Delandistrogene moxeparvovec expression, histology, and biochemical analyses
Details on the following methods can be found in Supplementary Appendix SA1: biodistribution analysis using droplet digital polymerase chain reaction, western blot analysis, histology and morphological analysis, immunofluorescence, and serum analysis.
Activity cage assessments
Activity cage assessments were performed at 10 and 50 weeks. Ambulation was measured by light beam breaks over a set period using the Photobeam Activity System-Open Field Activity Cage (San Diego Instruments). Animals were placed in the open field for monitoring of unprovoked movement.
Echocardiography
For information on how rats were anesthetized, see Supplementary Appendix SA1. For echocardiography, parasternal long and short axis views were obtained. Apical four-chamber views were obtained in a subset of animals. M-mode and apical four-chamber images were analyzed using Vevo LAB Systems software. Echocardiography personnel were blinded to the treatment group.
Cardiomyocyte isolation
Individual cardiomyocytes were isolated as previously described. 22 Briefly, rodent hearts were excised and cannulated through the aorta for perfusion with ice-cold calcium-free Tyrode’s solution. Cannulated hearts were transferred to a perfusion-flow Langendorff apparatus containing calcium-free Tyrode’s solution set to maintain a temperature of approximately 37°C. Hearts were perfused for 5–15 min before the solution was changed to Tyrode’s solution containing Liberase TH (Roche). Following enzymatic digestion, hearts were removed and minced in a dish containing the perfusion solution. The resultant cell solution was filtered and mixed with Tyrode’s solution containing bovine serum albumin to neutralize enzymatic activity.
Cardiomyocyte functional analysis
Freshly isolated cardiomyocytes were incubated with Fura-2 AM dye for 20–30 min. Following loading, myocytes were allowed 15–20 min for de-esterification. The cell solution was placed in a laminin-coated microwell dish, and intracellular calcium transient and sarcomere shortening measurements were obtained by electrical field stimulation of 0.5–2 Hz (Supplementary Table S3) with the IonOptix High Throughput System (HTS). For Fura dye measurements, data were captured using the IonOptix HTS with a microscope supplemented with a dual-excitation LED light source and photomultiplier detector for simultaneous ratio metric calcium measurements, as previously described. 23 Operators performing cardiomyocyte functional analysis were blinded to the treatment group.
Statistical analysis
Normality was determined by the Shapiro–Wilks test. If necessary to meet parametric assumptions, data were transformed using a log10 transformation. If the data set was unable to be transformed to fit a normal distribution, the raw data set was used for analysis. For parametric data sets, data were analyzed using a one- or two-way analysis of variance to determine the main effect of the independent variable(s) (e.g., group, time point). If a significant effect was observed for the independent variable(s), a post hoc Tukey test was performed to compare differences between groups. For non-parametric data sets, data were analyzed using a Kruskal–Wallis test to determine the main effect of the independent variable(s). If a significant effect was observed for the independent variable(s), a post hoc Dunn’s test was performed to compare differences between groups. Data comparisons were considered significantly different when p < 0.05.
RESULTS
Overview of study design and study outcomes
Supplementary Tables S1 and S2 provide a summary of dosing cohorts and an overview of the study design including the study outcomes measured. Briefly, Male Sprague-Dawley DMDMDX and WT rats from 21 to 42 days of age were intravenously administered saline or delandistrogene moxeparvovec and subsequently euthanized at predetermined time points for tissue collection (Supplementary Table S2). Activity cage assessments were performed to measure ambulation. Echocardiography was conducted to assess cardiac function, and isolated cardiomyocytes were used for functional analysis, including calcium transient and sarcomere shortening measurements (Supplementary Tables S3 and S4). A separate cohort of rats was used to assess the impact of delandistrogene moxeparvovec on survival (Fig. 1A).
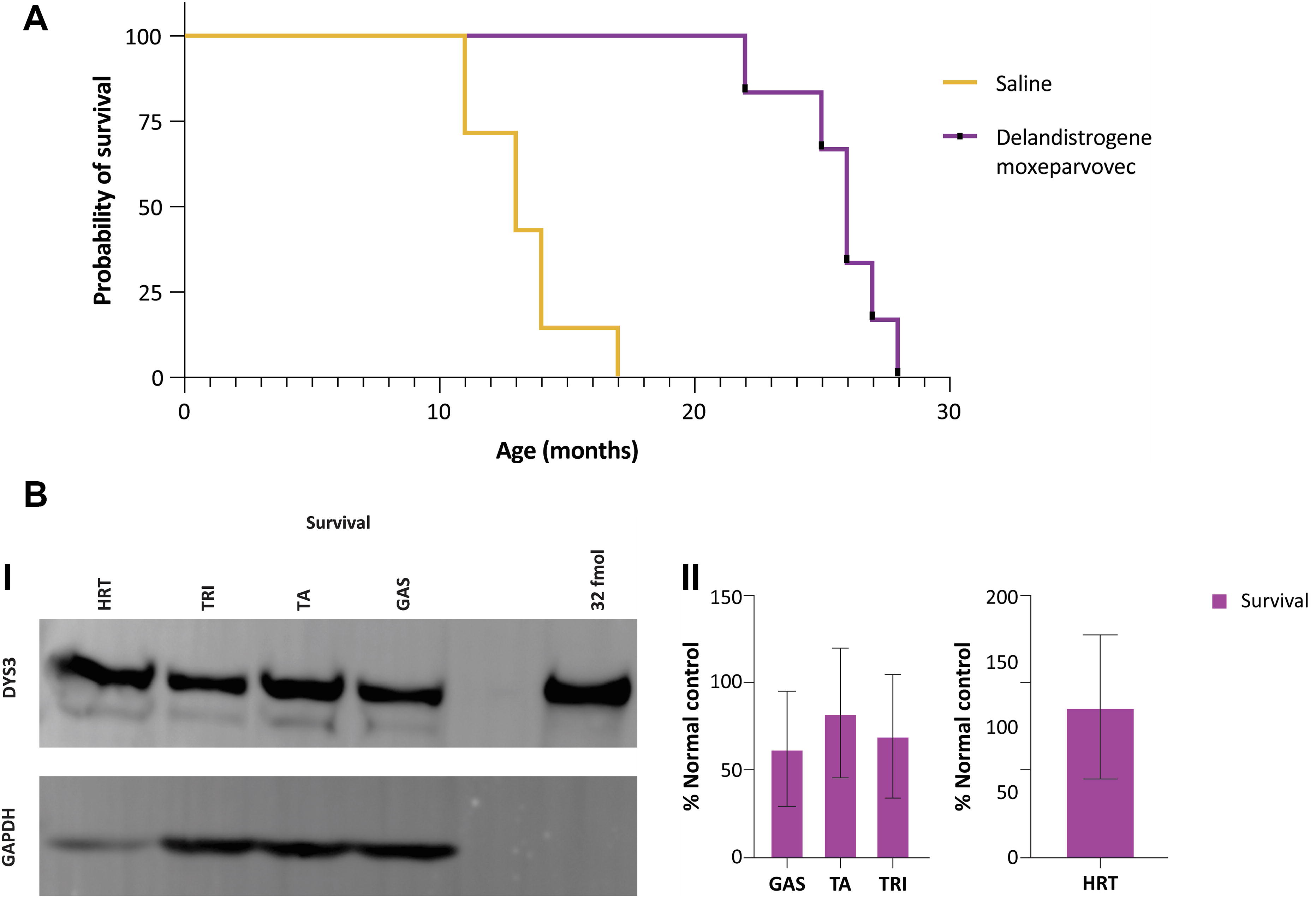
Survival and long-term durability analysis in DMDMDX rats following treatment with delandistrogene moxeparvovec.
Survival
The overall benefit of delandistrogene moxeparvovec treatment was examined over 2.5 years in DMDMDX rats. Without intervention, survival in DMDMDX rats was <17 months, with a median survival of 13 months (Fig. 1A). Treatment with delandistrogene moxeparvovec (1.33 × 1014 viral genomes/kilogram [vg/kg]) extended median survival in DMDMDX rats to >25 months. To examine the long-term treatment effects of delandistrogene moxeparvovec in the survival cohort of DMDMDX rats, vector biodistribution (Supplementary Fig. S2A) and western blot analysis (Fig. 1B) were performed in selected skeletal muscles (triceps, gastrocnemius, and tibialis anterior) and cardiac tissues. In the selected samples, delandistrogene moxeparvovec was detected at varying levels, with the highest levels found in the heart (Supplementary Fig. S2A). Western blot analysis showed robust delandistrogene moxeparvovec micro-dystrophin expression in the selected skeletal muscles and heart tissue examined (Fig. 1B). Of note, representative western blot images showed no dystrophin protein expression in livers from rats treated with delandistrogene moxeparvovec (Supplementary Fig. S2B). These data demonstrate that in DMDMDX rats, treatment with delandistrogene moxeparvovec provided sustained expression of the micro-dystrophin protein and extended survival to >25 months.
Delandistrogene moxeparvovec transgene and micro-dystrophin expression
Delandistrogene moxeparvovec biodistribution
The second arm of the study was limited to 1 year to evaluate the cardiac efficacy of delandistrogene moxeparvovec. Vector biodistribution analysis was performed at 12, 24, and 52 weeks, indicative of short-, intermediate-, and long-term delandistrogene moxeparvovec treatment, respectively. Delandistrogene moxeparvovec vector DNA was detected at varying levels in all collected tissue, with the highest levels in the liver, followed by the heart, and was observed broadly throughout skeletal muscle (triceps, gastrocnemius, and tibialis anterior) (Fig. 2A). Importantly, vector transduction was maintained over 52 weeks, with no significant differences in copies per nucleus between 12, 24, and 52 weeks. These results support the targeted expression and durability of delandistrogene moxeparvovec treatment with the use of a cardiac/muscle-specific promoter over 52 weeks.
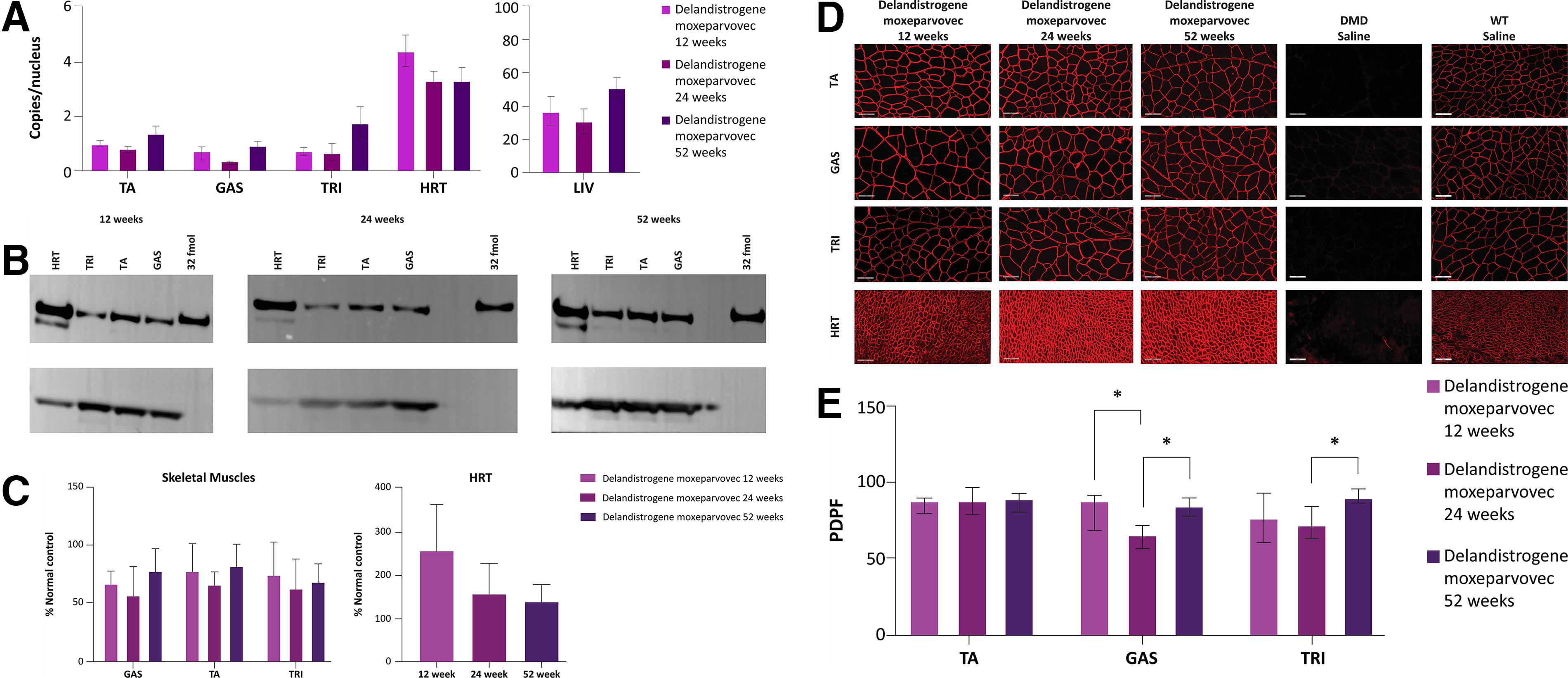
Delandistrogene moxeparvovec vector transduction and micro-dystrophin protein expression 12-, 24-, and 52-weeks following treatment in DMDMDX rats.
Delandistrogene moxeparvovec micro-dystrophin expression
Delandistrogene moxeparvovec micro-dystrophin levels were examined by western blot and quantified as percentage of normal control (Fig. 2B and C). Treatment led to muscle and cardiac micro-dystrophin expression at 12-, 24-, and 52-weeks post dosing (Fig. 2B, C, and D), with the highest level of expression achieved in cardiac tissue (Fig. 2B, C, and D). No significant changes in expression levels were observed over time in any tissue, suggesting sustained protein expression up to 52 weeks following treatment with delandistrogene moxeparvovec (Fig. 2B and C).
Delandistrogene moxeparvovec micro-dystrophin expression and localization in treated DMDMDX rats was examined by immunofluorescence at 12-, 24-, and 52-weeks post-treatment. Treatment led to substantial micro-dystrophin expression in skeletal muscles (tibialis anterior, triceps, and gastrocnemius) (Fig. 2D and E). A qualitative examination of cardiac tissue revealed membrane localized micro-dystrophin restoration following treatment at all time points, consistent with protein expression measured by western blot (Fig. 2C, D, and E). These data demonstrate sustained micro-dystrophin expression following delandistrogene moxeparvovec therapy in DMDMDX rats.
Fibrosis quantification
Untreated DMDMDX rats exhibited increased fibrosis in skeletal and cardiac muscle at 12 weeks versus WT rats, confirming that DMD-related remodeling occurs as early as 12 weeks (Fig. 3A). At all time points, delandistrogene moxeparvovec treatment led to a significant reduction in skeletal muscle (gastrocnemius) fibrosis in DMDMDX rats (Fig. 3A and B), indicating a profound benefit in preventing dystrophic remodeling in skeletal muscle.
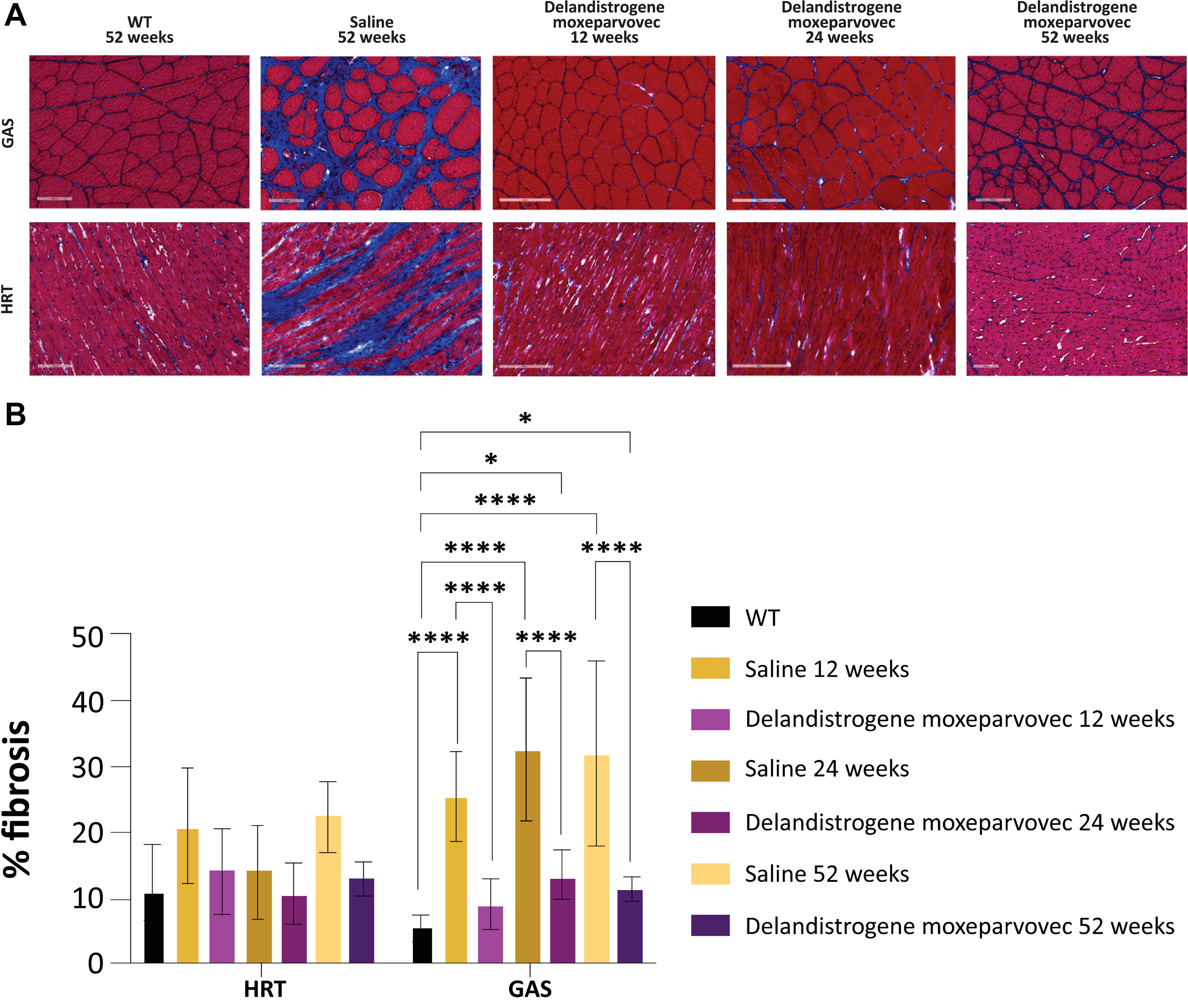
Fibrotic remodeling in skeletal muscle and heart of DMDMDX rats and impact of delandistrogene moxeparvovec treatment.
Cardiac tissue in saline-treated DMDMDX rats trended toward increased fibrosis versus WT at all time points (WT 12 weeks vs. 12-, 24-, and 52-weeks saline-treated rats). Delandistrogene moxeparvovec treatment led to a trend toward reduced cardiac fibrosis in DMDMDX rats at all time points evaluated. No adverse elevation in fibrosis levels or cardiac damage was observed in response to delandistrogene moxeparvovec treatment at all time points examined.
Histopathology
A reduction in skeletal muscle central nucleation (CN) was observed in delandistrogene moxeparvovec-treated DMDMDX rats, indicative of reduced muscle degeneration versus saline-treated animals (Supplementary Fig. S3A and B). Overall, the 12-, 24-, and 52-week cohorts of saline-treated DMDMDX rats exhibited a mean percent positive CN of >50%, whereas delandistrogene moxeparvovec-treated rats were <25% (Supplementary Fig. S3B). Furthermore, there were no signs of pathological effects related to delandistrogene moxeparvovec treatment in cardiac tissue.
Echocardiography
To avoid early mortality due to prolonged anesthesia exposure during echocardiography and facilitate a higher number of rats per experimental condition, echocardiography was limited to baseline and 52-week assessment. Untreated DMDMDX rats progressed toward cardiac dilation at 52 weeks, with a trend toward increased end-systolic volume (ESV) and significant reductions in left ventricular anterior wall (LVAW) and left ventricular posterior wall (LVPW) thickness (mm) systole compared with WT saline-treated rats (Fig. 4A, Supplementary Table S3). Delandistrogene moxeparvovec treatment attenuated dystrophic-related cardiac remodeling and restored LVAW systole and LVPW systole in DMDMDX rats (Fig. 4A, Supplementary Table S3). No significant differences in ESV, LVAW, and LVPW were observed between WT and delandistrogene moxeparvovec-treated cohorts (Fig. 4A I–III). Furthermore, no differences were observed in isovolumic relaxation time (IVRT), end-diastolic volume, or LVPW and LVAW diastolic wall thickness between delandistrogene moxeparvovec-treated DMDMDX and WT rats (Supplementary Fig. S4).
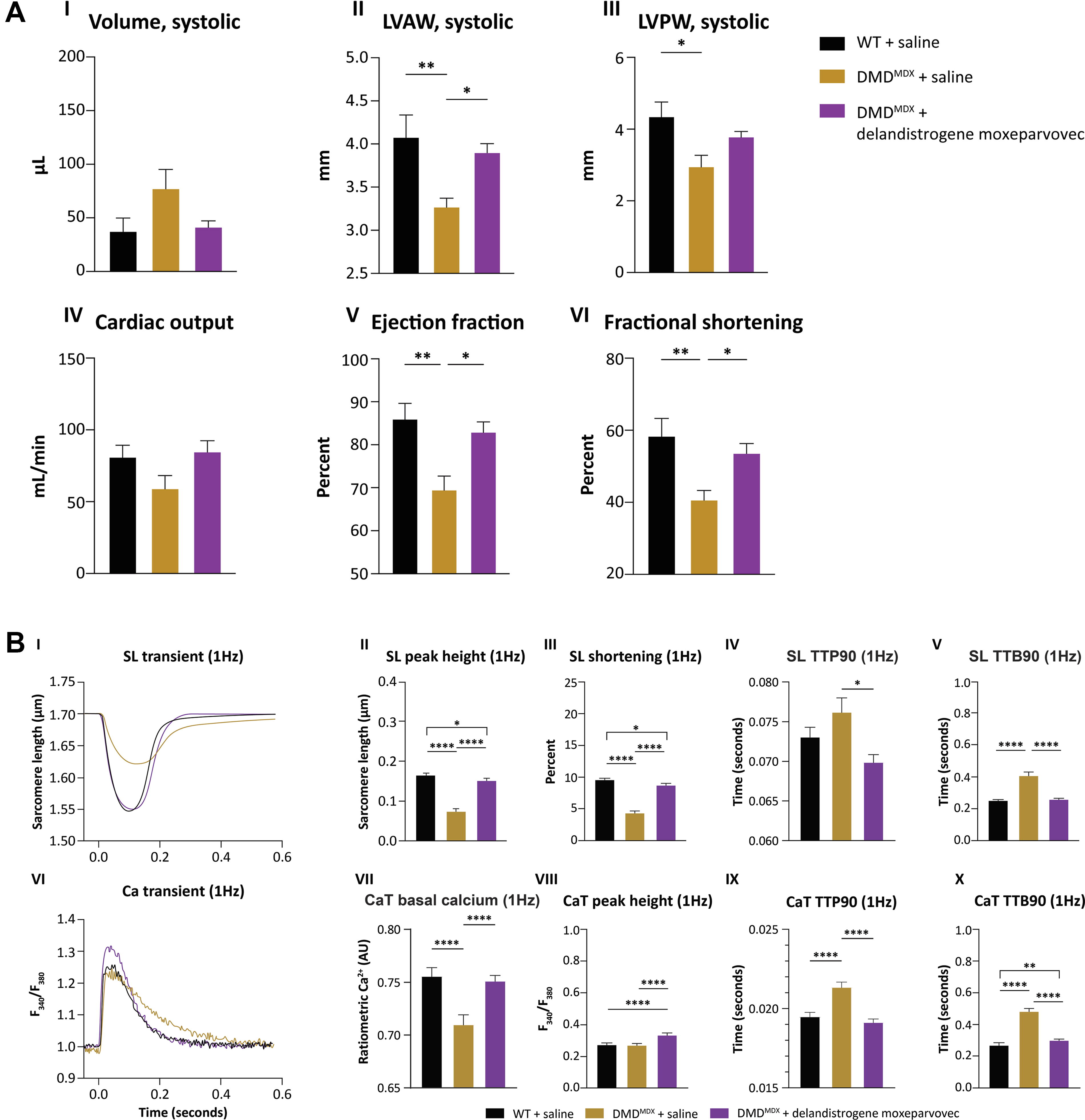
Cardiac echocardiography and cardiomyocyte functional measurements in DMDMDX rats treated with delandistrogene moxeparvovec.
In alignment with structural aberrations and progression toward dystrophic cardiomyopathy, DMDMDX rats had impaired cardiac function, demonstrated by significant reductions in ejection fraction and fractional shortening versus WT rats (Fig. 4A V–VI). Importantly, delandistrogene moxeparvovec led to a significant increase in ejection fraction and fractional shortening to WT levels in DMDMDX rats 52 weeks post-treatment (Fig. 4A V–VI). Taken together, these results highlight the cardiac efficacy of delandistrogene moxeparvovec in attenuating cardiomyopathy in DMDMDX rats.
Cardiomyocyte contractility and calcium handling
Cardiomyocytes from saline-control DMDMDX rats had impaired contractile function, characterized by a significant decrease in sarcomere length amplitude and percent sarcomere shortening versus WT myocytes (Fig. 4B, I–III, Supplementary Table S4). Additionally, DMDMDX cardiomyocyte contractile speed was impaired, as indicated by a trend toward an increase in time to peak 90% (TTP90) and a significant increase in sarcomere length time to return to baseline 90% (TTB90) (Fig. 4B, IV–V, Supplementary Table S4). Delandistrogene moxeparvovec significantly improved cardiomyocyte contractile response (to near WT levels) in DMDMDX myocytes, as demonstrated by a significant increase in sarcomere length peak height, percent sarcomere shortening, and a reduction in sarcomere length TTB90 (Fig. 4B, II–V, Supplementary Table S4). However, sarcomere contractility (sarcomere length peak height and percent shortening) was significantly increased in WT versus delandistrogene moxeparvovec-treated cardiomyocytes (Fig. 4B, II–III).
DMDMDX cardiomyocytes had impaired calcium handling, illustrated by a slower return to baseline 90% repolarization versus WT myocytes (Fig. 4B, VI–X, Supplementary Table S4). Significant decreases were observed in basal calcium levels in saline-treated DMDMDX myocytes compared with WT. Myocytes treated with delandistrogene moxeparvovec normalized resting calcium levels to WT levels in DMDMDX rats (Fig. 4B, VII). Delandistrogene moxeparvovec led to a significant increase in calcium transient peak height with a concomitant reduction in calcium transient time to baseline 90% versus DMDMDX myocytes (Fig. 4B, VIII–X). In addition, delandistrogene moxeparvovec normalized calcium time to 90% depolarization (TTP90) to WT levels in DMDMDX rat cardiomyocytes (Fig. 4B, IX–X).
Serum biomarkers
Serum cardiac troponin I (cTnI) was measured to evaluate cardiovascular pathology and response to delandistrogene moxeparvovec 12- and 52-weeks post-treatment (Supplementary Fig. S5A). At week 12, a significant elevation in cTnI levels was observed in saline-treated DMDMDX rats. Delandistrogene moxeparvovec treatment led to a significant reduction in cTnI in DMDMDX rats 12 weeks post-treatment (Supplementary Fig. S5A). At 52 weeks, delandistrogene moxeparvovec treatment showed a reduction in cTnI levels; however, the reduction was not statistically significant versus that in saline-treated DMDMDX rats (Supplementary Fig. S5A).
Elevated levels of creatine kinase (CK), aspartate aminotransferase, and alanine aminotransferase reflect muscle damage and are considered biomarkers of the muscular pathology in DMD. 2 Evaluation of these clinical biochemical parameters revealed reductions in CK at week 12, aspartate aminotransferase at weeks 12 and 52, and alanine aminotransferase at week 12 in delandistrogene moxeparvovec-treated DMDMDX rats versus saline-treated rats (Supplementary Fig. S5B–D). Due to varying levels of hemolysis across treatment groups, CK measurements at weeks 24 and 52 were not assessed.
Activity cage assessments
A statistically significant increase in horizontal ambulation and vertical activity was demonstrated at 12 weeks post-treatment in delandistrogene moxeparvovec-treated DMDMDX rats versus saline controls (Supplementary Fig. S6). Delandistrogene moxeparvovec treatment led to a trend toward improved ambulation at 52 weeks (Supplementary Fig. S6). These data demonstrate functional improvement in mobility in DMDMDX rats after delandistrogene moxeparvovec treatment. The noted decline in ambulatory performance across all experimental groups from 12 and 52 weeks could be due to the size constraints of the activity cage, sensitivity of the assay, and age of the animal.
Transduction of AAVrh74.MHCK7.GFP in cardiomyocyte cells
To further understand the effect of delandistrogene moxeparvovec treatment on cardiac function in human cells, transduction of a reporter construct AAVrh74.MHCK7.GFP was evaluated in human induced pluripotent stem cell cardiomyocytes (hiPSC). Expression of green fluorescent protein in hiPSC cardiomyocytes treated with AAVrh74.MHCK7.GFP indicated that the Myosin Heavy Chain Creatine Kinase promoter (MHCK7) promoter effectively transduced and expressed the GFP transgene in human cardiomyocytes. In congruence with MHCK7 derived in vivo data, an in vitro reporter-based assay with AAVrh74.MHCK7.eGFP led to increased GFP expression in hiPSC cardiomyocytes. These data confirm that MHCK7 can express a transgene payload in human cardiomyocytes (Supplementary Fig. S7A and B).
DISCUSSION
Cardiomyopathy has become the most common cause of death in patients with DMD. 6,7 This study was designed to evaluate the cardiovascular efficacy of delandistrogene moxeparvovec in a more suitable model of cardiomyopathy, the DMDMDX rat. Delandistrogene moxeparvovec contains the MHCK7 promoter, which includes an alpha-myosin heavy chain enhancer to drive a high level of expression in the heart. 16,24 This purposeful design was intended to provide protection against cardiomyopathy. This study provides the first direct evidence for the biological activity of delandistrogene moxeparvovec micro-dystrophin in the heart with a demonstration of robust expression and significant improvements in cardiac measures.
Importantly, treatment with delandistrogene moxeparvovec extended the life expectancy of DMDMDX rats, prolonging survival to that of WT Sprague-Dawley rats. 25 In addition, improved ambulation was observed in treated rats versus saline controls, which illustrates the skeletal muscle efficacy of delandistrogene moxeparvovec and supports the improved functional data seen in clinical trials.
Treatment with delandistrogene moxeparvovec demonstrated improvements in muscle integrity, with reductions in fibrosis seen at all time points across skeletal and cardiac muscle. Additionally, similar findings have been reported in a recent study, highlighting fibrosis as a consistent pathological hallmark of DMD. 26 The reduction in fibrosis suggests that delandistrogene moxeparvovec attenuates the dystrophic remodeling characteristic of DMD. Additionally, decreased CN with delandistrogene moxeparvovec treatment reflects improvement in muscle regeneration and reduction in muscle degeneration.
This study allowed for a comprehensive analysis of delandistrogene moxeparvovec micro-dystrophin protein expression and localization across all tissues including the heart. Delandistrogene moxeparvovec vector biodistribution and micro-dystrophin expression evaluated by droplet digital polymerase chain reaction and western blot, indicated biodistribution and robust expression across tissues and time points. Our findings are in line with a similar AAV-based gene therapy assessed in the same DMDMDX rat model. 26 Biodistribution, protein expression, and histological parameters were consistent across the 12-, 24-, and 52-week time points, indicating long-term durability of delandistrogene moxeparvovec treatment. The durability of cardiac micro-dystrophin expression at week 52 supports the efficacy of delandistrogene moxeparvovec in improving cardiac function and reversing adverse cardiac remodeling in DMDMDX rats.
Since cardiomyopathy is a significant contributor to morbidity and mortality in patients with DMD, 27 the efficacy of delandistrogene moxeparvovec in the heart was evaluated. A key characteristic of heart failure is abnormal calcium transients, which often have diminished amplitude and are prolonged in ventricular cardiomyocytes of patients. 18 These changes at the cardiomyocyte level are causative for impaired cardiac function. 18 Here we show that delandistrogene moxeparvovec treatment enhances cardiomyocyte contractility and normalizes calcium handling in DMDMDX rats as a result of membrane stabilization via delandistrogene moxeparvovec micro-dystrophin cardiac expression. The precise mechanism regarding calcium stabilization following delandistrogene moxeparvovec treatment is not completely understood. Based on our data from isolated ventricular cardiomyocytes, the delandistrogene moxeparvovec micro-dystrophin construct enhances the amplitude of calcium transients and reduces the transient return to baseline speed. The construct contains key regions of the full dystrophin protein (ABD-H1-R1-R3-H2-R24-H4-CR) that have been shown to reduce basal calcium levels in DMDMDX rats. 28 These data indicate that upstream micro-dystrophin construct elements are sufficient to improve calcium handling in DMDMDX rats.
Previous studies have characterized diastolic and systolic dysfunction in DMDMDX rats as early as 12 weeks, depending on time point and treatment, highlighting the progressive nature of cardiac impairment in this model. 17,18,26,28 Data from our echocardiography analysis revealed improved indicators of cardiac systolic function with delandistrogene moxeparvovec 52 weeks post-treatment versus saline controls. For diastolic dysfunction, data at 52 weeks indicated an improvement in isovolumetric relaxation time in DMDMDX rats treated with delandistrogene moxeparvovec.
This study demonstrated that delandistrogene moxeparvovec micro-dystrophin was well tolerated in rats with no evidence of toxicity. Our findings show that long-term treatment of DMDMDX rats with delandistrogene moxeparvovec micro-dystrophin at a clinical dose level (1.33 × 1014 vg/kg) did not lead to adverse cardiac events or dilated cardiomyopathy progression. Cardiac toxicity following gene therapy may be transgene expression dependent, 2,29 and the threshold of expression required to reach toxicity in the human heart versus a rodent model may differ. 30 Hart et al. (2022) observed cardiac toxicity in a DMD mouse model (D2.mdx mouse) due to a 55-fold increase in micro-dystrophin expression in the heart. 29 In contrast, our study achieved one-to-two-fold higher cardiac expression in DMDMDX rats compared with WT, resulting in functional improvements at the cardiomyocyte and whole-heart levels. Importantly, our findings indicate enhanced survival rates, with no reported cardiac events or fatalities in DMDMDX rats treated with delandistrogene moxeparvovec.
Limitations
There are certain limitations of this study that should be considered. The specific causes of death of study animals were not evaluated. However, the improvement in survival observed in delandistrogene moxeparvovec-treated DMDMDX rats is a clear demonstration of a treatment-related effect.
Functional analysis at intermediate time points (24, 36, and 48 weeks) could have offered a more expansive view of the trajectory of motor function decline within the DMDMDX rats. In addition, reduced animal viability over 52 weeks, particularly in untreated rats, limited the study to small cohorts (approximately six to eight animals). Consequently, the variability in echocardiography and the small sample size may have contributed to the reduced significance observed in several measures.
CONCLUSIONS
This study provides evidence for the biological activity of delandistrogene moxeparvovec micro-dystrophin in the heart and skeletal muscles of DMDMDX rats. These data demonstrate a previously undescribed role for this therapy in improving cardiac function and support delandistrogene moxeparvovec treatment for DMD.
Footnotes
AUTHORS’ CONTRIBUTIONS
Conceptualization: R.A.P. Methodology: S.B., C.W., L.L., G.C.-O., A.K., A.H., J.E., A.F., E.N., R.A.P. Formal analysis: S.B., C.W., L.L., G.C.-O., A.K., A.H., J.E. Investigation: S.B., C.W., L.L., G.C.-O., A.K., A.H., J.E., A.F. Resources: L.R.R.-K., R.A.P. Writing—original draft: S.B., C.W., G.C.-O., R.A.P. Writing—review and editing: S.B., C.W., L.L., G.C.-O., A.K., A.H., J.E., A.F., E.N., L.R.R.-K., R.A.P. Visualization: S.B, C.W., G.C.-O., R.A.P. Supervision: L.R.R.-K., R.A.P. Project administration: E.N., R.A.P.
AUTHOR DISCLOSURE
S.B., C.W., L.L., G.C.-O., A.K., A.H., J.E., A.F., E.N., L.R.R.-K., and R.A.P. are employees of Sarepta Therapeutics and may have stock options. L.R.R.-K. is a co-inventor of AAVrh74.MHCK7.micro-dys technology.
FUNDING INFORMATION
SUPPLEMENTARY MATERIAL
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Figure S7
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Appendix SA1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
