Abstract
Adeno-associated virus (AAV) vectors are used to deliver therapeutic transgenes, but host immune responses may interfere with transduction and transgene expression. We evaluated prophylactic corticosteroid treatment on AAV5-mediated expression in liver tissue. Wild-type C57BL/6 mice received 6 × 1013 vg/kg AAV5-HLP-hA1AT, an AAV5 vector carrying a human α1-antitrypsin (hA1AT) gene with a hepatocyte-specific promoter. Mice received 4 weeks of daily 2 mg/kg prednisolone or water starting day −1 or 0 before vector dosing. Mice that received prophylactic corticosteroids had significantly higher serum hA1AT protein than mice that did not, starting at 6 weeks and persisting to the study end at 12 weeks, potentially through a decrease in the number of low responders. RNAseq and proteomic analyses investigating mechanisms mediating the improvement of transgene expression found that prophylactic corticosteroid treatment upregulated the AAV5 coreceptor platelet-derived growth factor receptor alpha (PDGFRα) on hepatocytes and downregulated its competitive ligand PDGFα, thus increasing the uptake of AAV5 vectors. Evidently, prophylactic corticosteroid treatment also suppressed acute immune responses to AAV. Together, these mechanisms resulted in increased uptake and preservation of the transgene, allowing more vector genomes to be available to assemble into stable, full-length structures mediating long-term transgene expression. Prophylactic corticosteroids represent a potential actionable strategy to improve AAV5-mediated transgene expression and decrease intersubject variability.
INTRODUCTION
Adeno-associated virus (AAV) vectors efficiently transduce human cells but also stimulate innate and adaptive immune responses against the AAV capsid, transgene product, or AAV-transduced cells. 1 –4 Host immune responses may hamper transduction and contribute to interparticipant variability in clinical trials. 1,2,5 –11 Host cell pattern recognition receptors induce proinflammatory cytokines, and antiviral responses lead to vector deoxyribonucleic acid (DNA) degradation. 5,12 Adaptive immune responses, including expansion of CD8+ T cells directed against transgene-expressing target cells, cause inflammation and damage. 7,13,14
Corticosteroids inhibit immune responses by suppressing transcription of proinflammatory cytokines and chemokines. 15,16 Corticosteroids are frequently used with AAV gene therapies to treat alanine aminotransferase (ALT) elevations in liver-targeted gene therapies, and preserve transgene expression in some cases. 8,10 Differential timing of corticosteroid use across trials of the gene therapy valoctocogene roxaparvovec (AAV5-hFVIII-SQ) and differences in responses suggest that earlier corticosteroid use may promote transgene expression. 8,9,17 –19
Prophylactic corticosteroids administered before AAV dosing may promote transgene expression. In mice, prophylactic dexamethasone administered 2 h before AAV9 dosing increased hepatic levels of vector genomes. 20 Nonhuman primates (NHPs) treated with prophylactic prednisolone before AAV dosing had less CD8+ T cell infiltration and apoptosis than those who did not. 21 However, prednisolone treatment in mice 1 week after AAV5-hFVIII-SQ dosing did not affect transgene expression. 22 Similarly, dexamethasone treatment initiated 1 year after AAV9 did not change transgene expression levels in dogs. 20
In this study, we assessed transgene expression in mice after prophylactic prednisolone before AAV5-mediated gene therapy over 12 weeks. After confirming prophylactic prednisolone increased transgene expression, we investigated potential mechanisms of action at 2 and 24 h post-AAV dosing using RNAseq, proteomics, and targeted molecular analyses. Not only did prophylactic prednisolone suppress innate immune responses, it also upregulated the expression of the AAV5 coreceptor platelet-derived growth factor receptor alpha (PDGFRα).
MATERIALS AND METHODS
Vector constructs
Valoctocogene roxaparvovec (AAV5-hFVIII-SQ) gene therapy for severe hemophilia A is a recombinant AAV5 vector containing a human B-domain–deleted FVIII cDNA (hFVIII-SQ) controlled by a hepatocyte-selective promoter. 8,23,24 Mouse studies with serial blood sampling used the reporter vector AAV5-HLP-hA1AT, which delivers a human α1-antitrypsin (hA1AT) gene, instead of AAV5-hFVIII-SQ because hA1AT protein is nonimmunogenic in mice, and serial blood draws activate the clotting cascade and consume factor VIII (FVIII), introducing variability. 25,26
Study design
We assessed the effects of prophylactic steroids over 12 weeks with wild-type C57BL/6 mice (Jackson Laboratory, Bar Harbor, ME) that received either water or 2 mg/kg prednisolone in water vehicle by oral gavage beginning on day −1 or 0 before AAV dosing and continuing for 4 weeks (Fig. 1A). Blood was drawn at baseline and weeks 1, 4, 6, 8, and 12 postdose for serum hA1AT measurement; takedown cohorts were collected at weeks 4 (n = 3) and 12 (n = 7) and livers were collected for vector genome quantification. Mice were dosed with 6 × 1013 vg/kg AAV5-HLP-hA1AT. The impact of prophylactic methylprednisolone and rituximab before dosing with 6 × 1013 vg/kg AAV5-hFVIII-SQ was assessed in NHPs (Supplementary Methods section in Supplementary Data).
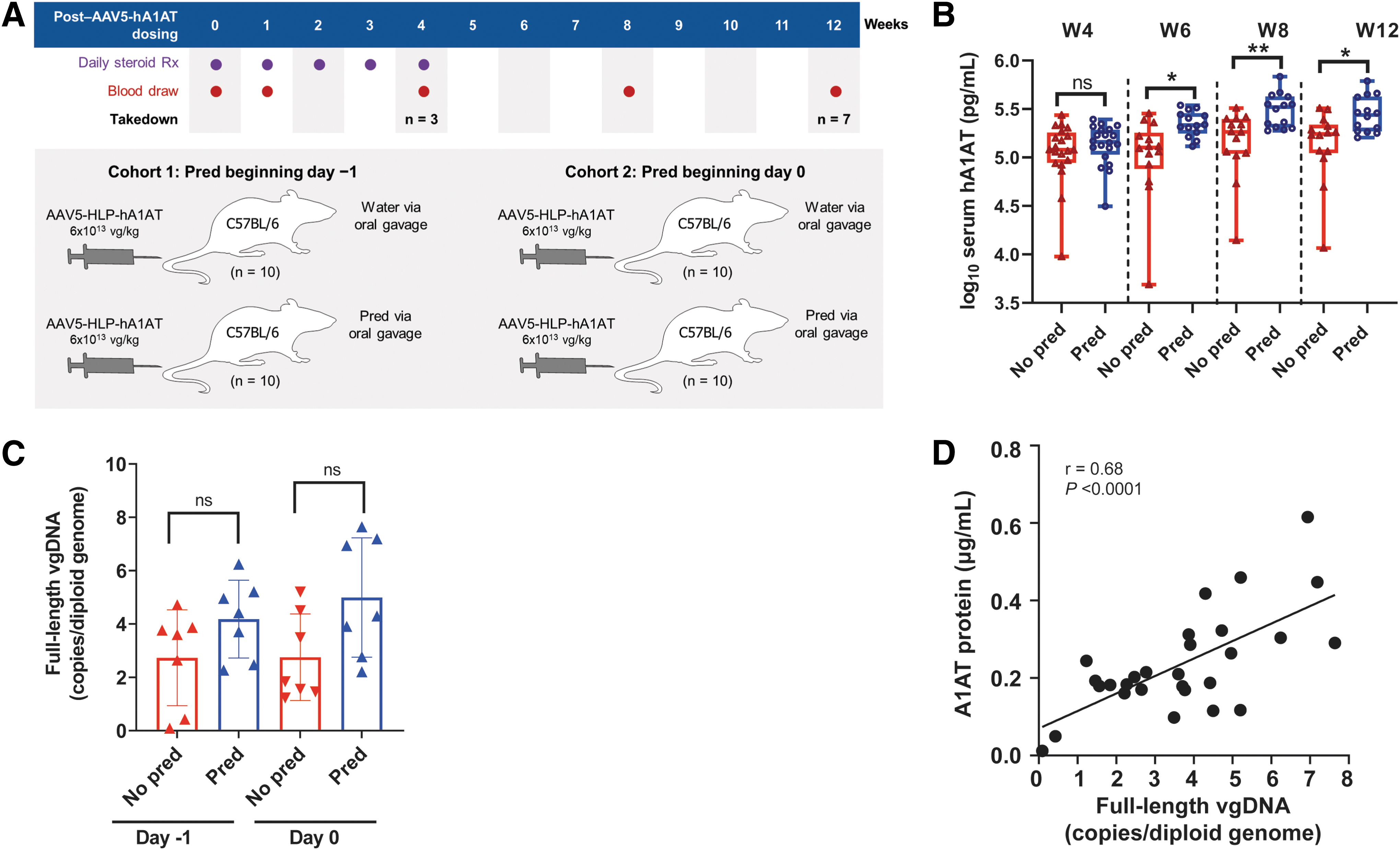
Effect of prophylactic prednisolone treatment over 12 weeks.
We also assessed the effects of prophylactic steroids on events immediately after AAV dosing with wild-type C57BL/6 mice who received one of four treatments (prophylactic prednisolone or water and 6 × 1013 vg/kg AAV5-HLP-hA1AT or vehicle) before euthanization at 2 or 24 h post-AAV dose (Fig. 2A). Blood samples were taken predose and terminally for hA1AT and other protein assays. Livers were collected for RNAseq, protein biomarker detection using an Olink assay, N-glycome analyses, and immunohistochemistry.
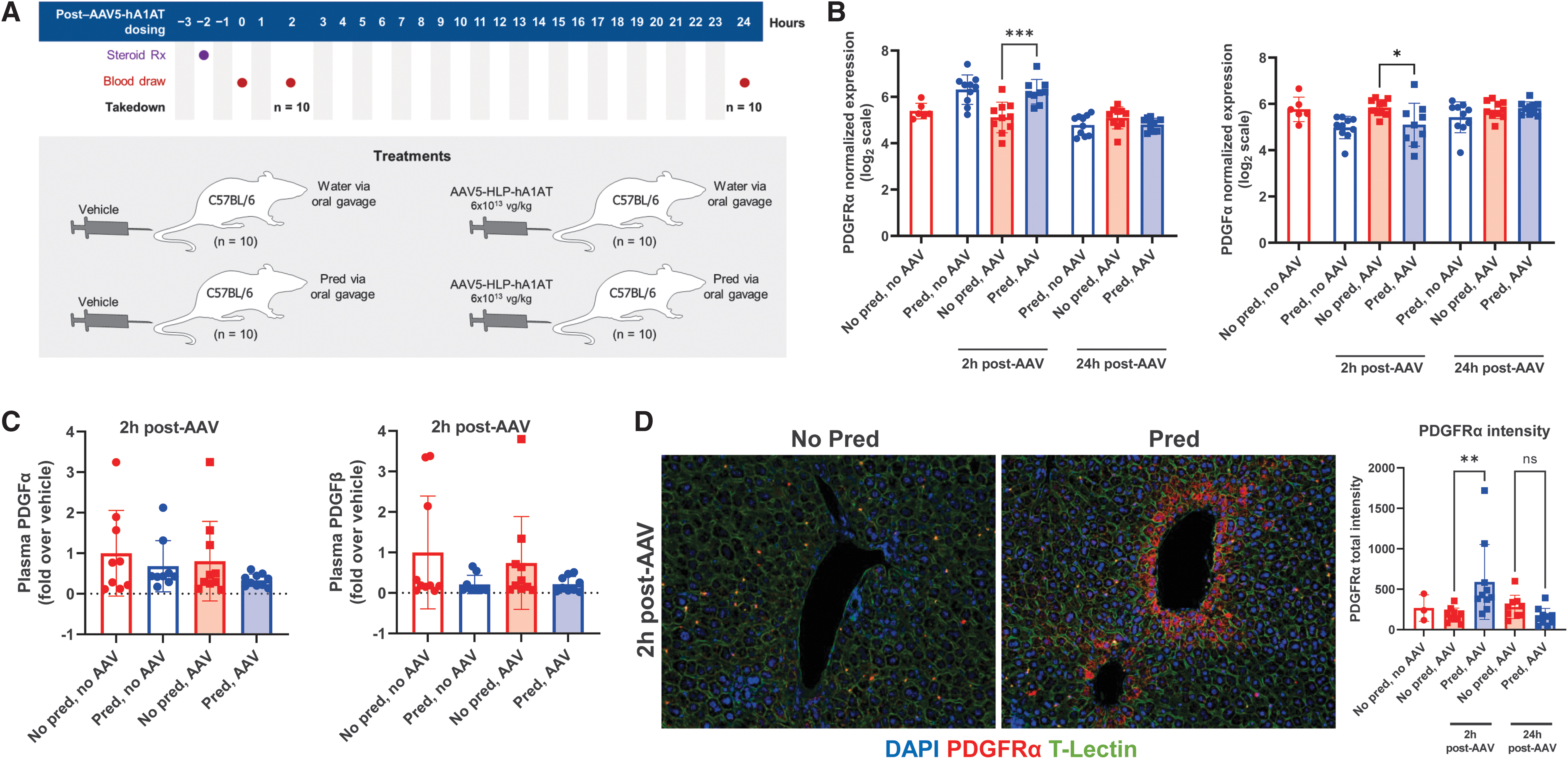
Effect of prophylactic prednisolone treatment on PDGFRα receptor and ligands at 2 and 24 h after vector dosing.
In addition, we assessed how natural PDGFRα variation correlates with transgene expression by dosing wild-type C57BL/6 and BalbC (Jackson Laboratory) mice with 6 × 1013 AAV5-hFVIII-SQ. Mice were euthanized at 5 weeks postdose, and blood and liver samples were collected for assessment of FVIII-SQ protein, transgene DNA, and PDGFRα protein.
Ethics statement
Mouse protocols were approved by the Animal Resource Committee of BioMarin Pharmaceutical, Inc. and the Institutional Animal Care and Use Committee of the Buck Institute. NHP protocols were approved by the Institutional Animal Care and Use Committee at the Charles River Laboratory (San Francisco, CA).
RNAseq and pathway enrichment analyses
mRNA samples were prepared using Illumina Stranded mRNA kits (Illumina, San Diego, CA) and sequenced on a NovaSeq 6000 v1.5 using single-end 100-bp reads. Reads were analyzed and mapped to the GRCm38/mm10 mouse genome assembly. 27 Differential expression was determined using the edgeR package in R (R Foundation, Vienna, Austria). Gene set enrichment analysis was performed using hallmark gene sets from MSigDb in R, using normalized enrichment score (NES) and Benjamini–Hochberg-adjusted p value. 28,29
Protein assays
hA1AT protein in mice was measured in serum (12-week study) or plasma (24-h study) using an enzyme-linked immunosorbent assay (ELISA) detecting hA1AT without mouse cross-reactivity (Human Serpin A1 DuoSet ELISA, DY1268; R&D Systems, Minneapolis, MN; Supplementary Methods section in Supplementary Data).
hFVIII-SQ protein was measured in plasma samples using a sandwich ELISA utilizing human-specific anti-FVIII capture (Green Mountain Antibodies, Burlington, VT) and detection (F8C-EIA; Affinity Biologics, Ancaster, ON, Canada) antibody pairs, as previously described. 23
Complement system activity was assessed with plasma C3b (Supplementary Methods section in Supplementary Data). An Olink proteomic assay was used to explore protein expression (Supplementary Methods section in Supplementary Data). Plasma PDGFα/α was measured using a Mice PDGFα/α ELISA kit (Abcam, Cambridge, United Kingdom).
Plasma ALT
Mouse plasma ALT levels were assessed at 2 and 24 h post-AAV dose and over 12 weeks (Supplementary Methods section in Supplementary Data).
DNA in situ hybridization
Hepatocytes staining positive for vector genome and in situ hybridization (ISH) area per cell were quantified in formalin-fixed paraffin-embedded (FFPE) liver sections of 5 μm prepared as described. 23 One whole liver section was acquired per animal, and two regions were randomly selected for analysis using Visiopharm (Hørsholm, Denmark).
Droplet digital polymerase chain reaction
Vector genome quantification, including full-length genomes and overall total genomes, was performed with drop-phase droplet digital polymerase chain reaction (ddPCR) analyses targeting the AAV5-hA1AT-HLP transgene sequence, as described 23 (Supplementary Methods section in Supplementary Data), following DNA and ribonucleic acid (RNA) extraction using the AllPrep DNA/RNA Mini kit (Qiagen, Hilden, Germany).
N-glycome analysis
Changes in N-glycosylation in mouse livers at 2 and 24 h post-AAV dose were assessed (Supplementary Methods section in Supplementary Data).
Immunohistochemistry
Hepatic expression and distribution of PDGFRα and viral protein 3 (VP3) were measured by immunohistochemistry. FFPE livers sectioned at 5-μm thickness were collected on Leica Microsystems Plus Slides (Leica Biosystems, Buffalo Grove, IL). Immunostaining was performed using a Leica BOND RX Autostainer (Supplementary Methods section in Supplementary Data).
hFVIII-SQ vector genome quantification
AAV5-hFVIII-SQ vector genome levels were measured with a quantitative real-time PCR assay using a TaqMan DNA probe and primers specific to hFVIII-SQ after genomic liver DNA was extracted using the Quick DNA/RNA MagBead kit (Zymo Research, Irvine, CA; Supplementary Methods section in Supplementary Data).
PDGFRα in vitro analyses
PDGFRα expression upregulation by prophylactic prednisolone was assessed in human and mouse primary hepatocytes in vitro (Supplementary Methods section in Supplementary Data). The effect of PDGFRα on AAV transduction was confirmed by knockdown in HepG2 cells with a short hairpin RNA (shRNA; Supplementary Methods section in Supplementary Data).
Statistical methods
Serum hA1AT was analyzed using a Student's t-test at each time point after log10 transformation. Benjamini–Hochberg-adjusted p values were calculated for differential expression and gene set enrichment analyses in RNAseq experiments and Olink proteomic analysis using p < 0.05. Plasma FVIII protein and liver hFVIII-SQ vector genome copies were analyzed by a Student's t-test. Plasma PDGFα/α, complement system activation, and immunohistochemistry staining intensity were analyzed using one-way analysis of variance (ANOVA) and a Tukey's multiple comparison test. PDGFRα and PDGFα normalized expression were compared using a Welch's unpaired t-test.
RESULTS
Prophylactic prednisolone treatment increases transgene expression and vector DNA
In the 12-week study, mice who received prophylactic prednisolone on either day −1 or 0 had significantly higher serum hA1AT protein than nonprednisolone-treated mice from week 6 onward (Supplementary Fig. S1A). Serum protein was ∼1.5- to 2-fold higher for mice receiving prednisolone at either time point in weeks 6 through 12 than for nontreated mice; prophylactic prednisolone appeared to lower interindividual variability by reducing the number of mice with low expression (Fig. 1B).
More hepatocytes stained positive for vector DNA in mice that received prophylactic prednisolone versus water on either day −1 or 0 at weeks 4 and 12 (Supplementary Fig. S1B), although significance was not reached. A clear trend for more overall and full-length vector genomes was also detected in prednisolone-treated mice compared with nonprednisolone-treated mice (Fig. 1C and Supplementary Fig. S1C). Similarly, after dosing with AAV5-hFVIII-SQ, significantly more full-length vector DNA (p < 0.05) was present in the livers of NHPs that received prophylactic methylprednisolone and rituximab than in those that did not (Supplementary Fig. S2).
In mice, serum hA1AT protein levels correlated significantly (r = 0.68, p < 0.0001) with full-length vector genomes in the liver at week 12 (Fig. 1D). Transgene RNA levels also correlated with vector DNA (r = 0.49, p < 0.01) and transgene protein (r = 0.59, p < 0.001) at 12 weeks (Supplementary Fig. S1D, E). Overall, prophylactic corticosteroids improved AAV5-mediated transgene expression by increasing levels of functional full-length vector DNA in hepatocytes, giving rise to more RNA and protein.
Prophylactic prednisolone promotes transduction within 24 h of AAV dosing
Previously, initiation of prednisolone 1 week after AAV5-hFVIII-SQ dosing did not modulate transgene expression, 22 while here, initiating prednisolone before AAV dosing resulted in improved transgene expression. Hence, we hypothesized that the mechanism promoting AAV-mediated expression in response to prophylactic prednisolone occurs early and investigated potential mechanisms active immediately after AAV and steroid dosing. 22 Mice received a single treatment of prednisolone or water 2 h before dosing with AAV or vehicle (Fig. 2A), and blood and livers were collected at 2 and 24 h.
First, we identified pathways impacted by AAV transduction or prednisolone treatment using RNAseq. Differential expression analyses were structured as treatment versus control, and differentially expressed genes (adjusted p < 0.05 and log2-fold change >1.5) identified (Supplementary Table S1). In mice treated with AAV5-HLP-hA1AT versus control, 115 and 231 genes were upregulated and downregulated, respectively, at 2 h postdose; by 24 h postdose, 36 and 160 genes were upregulated and downregulated, respectively. Significantly upregulated pathways, as indicated by NES, included mTORC1 signaling, complement pathway, interleukin (IL)-6, JAK-STAT3 signaling, and DNA repair pathways (Table 1).
Enrichment of hallmark gene sets for the effect of adeno-associated virus at 2 and 24 hours postdose
NES determines whether a gene set is moving up (positively regulated) or down (negatively regulated) the gene rankings when comparing mice treated with prednisolone+AAV5-HLP-hA1AT versus mice treated with prednisolone+vehicle (n = 10/group). These curated data were sorted by NES.
AAV, adeno-associated virus; AAV5-HLP-hA1AT, AAV serotype 5 hybrid liver promoter human α1-antitrypsin; DNA, deoxyribonucleic acid; E, exponential; E2F, E2 factor; IL-6, interleukin-6; JAK STAT3, JAK STAT pathway 3; JAK, Janus kinase; mTORC1, mammalian target of rapamycin complex 1; NES, normalized enrichment score; PMID, PubMed ID; STAT, signal transducer and activator of transcription; UV, ultraviolet; v, vector.
Prednisolone treatment suppresses innate immune responses
For mice treated with prophylactic prednisolone versus control before AAV dosing, 863 and 1561 genes were significantly upregulated and downregulated, respectively, at 2 h postdose. By 24 h, only 87 and 173 genes were upregulated and downregulated, respectively. Inflammatory response pathways, such as interferon-α and tumor necrosis factor-α/nuclear factor-κB signaling, were downregulated by prednisolone as indicated by NES (Table 2). We specifically examined IL-1β expression as a marker of the inflammasome and found it was significantly downregulated by prednisolone (Supplementary Fig. S3A).
Enrichment of hallmark gene sets for the effect of prophylactic prednisolone at 2 and 24 hours postdosing with adeno-associated virus
NES determines whether a gene set is moving up (positively regulated) or down (negatively regulated) the gene rankings when comparing mice treated with prednisolone+AAV5-HLP-hA1AT versus mice treated with water+AAV5-HLP-hA1AT (n = 10/group). These curated data were sorted by NES.
IFN-α, interferon-alpha; IFN-γ, interferon-gamma; KRAS, Kirsten rat sarcoma virus; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; TNF-α, tumor necrosis factor-alpha.
We used an exploratory Olink mouse panel (Uppsala, Sweden) to measure the effect of prophylactic prednisolone on plasma proteins at 2 and 24 h post-AAV dose. Normalized protein expression of most inflammatory response proteins was significantly suppressed by prophylactic prednisolone at both time points (p < 0.05; Supplementary Tables S2 and S3), but cytokines were upregulated by AAV treatment (p < 0.05; Supplementary Tables S4 and S5). However, serum ALT levels indicating liver damage were not significantly elevated with or without prednisolone (Supplementary Fig. S4A, B).
In addition, despite RNAseq data showing complement activation by AAV in the liver (Supplementary Table S6), there were no significant differences in plasma C3b levels between treatments (Supplementary Fig. S3B).
Prednisolone treatment upregulates the AAV5 coreceptor PDGFRα
Because prophylactic prednisolone increased liver vector DNA, we hypothesized it enhances initial transduction efficacy. We thus investigated mechanisms involved in the initial uptake of AAV vectors. AAV5 vectors interact with extracellular N-glycans before binding to the essential multiserotype AAV receptor (AAVR) and the AAV5 coreceptors PDGFRα and PDGFRβ on the cell surface. 30 –38
Liver levels of N-glycans in mice at 2 and 24 h were not affected by prophylactic prednisolone (Supplementary Fig. S5). In our RNAseq data set, PDGFRα expression at 2 h post-AAV dose was significantly higher in livers of mice that received prednisolone versus water before AAV treatment, and PDGFα, a ligand for PDGFR, was significantly lower in mice that received prednisolone versus water before AAV dosing (Fig. 2B). However, differences did not persist to 24 h. AAVR or PDGFRβ expression did not significantly differ (data not shown).
Secreted PDGFα forms homodimer (PDGFα/α) or PDGFβ heterodimer complexes (PDGFα/β), and so, to confirm RNAseq data, circulating ligands were assessed using an ELISA for PDGFα/α and an Olink assay for PDGFβ/β (both also detect PDGFα/β). At 2 h post-AAV, plasma levels of PDGF ligands were lower, although not significant, in mice that received prophylactic prednisolone versus vehicle (Fig. 2C). Mass spectrometry of total liver homogenate samples found no significant difference between groups in PDGFRα (data not shown). As liver homogenate may include lysates from multiple cell types that could dilute signals, we also used immunohistochemistry to detect PDGFRα expression on hepatocytes. Analysis confirmed a significantly higher PDGFRα protein in a subset of hepatocytes at 2 h post-AAV dosing in mice treated with prophylactic prednisolone versus vehicle (Fig. 2D).
Prophylactic prednisolone increases AAV5 uptake
At 2 h postdose, using immunohistochemistry, intracellular VP3—an AAV5 viral capsid protein—was significantly higher with prednisolone treatment (Fig. 3A), consistent with increased uptake of AAV by cells. 39,40 Furthermore, microscopy of VP3 and PDGFRα together showed that both were found in the same hepatocytes following AAV5 administration (Fig. 3B). PDGFRα staining intensity was significantly correlated (r = 0.7479, p < 0.0005) with VP3 intensity within hepatocytes at 2 h postdose (Fig. 3C).
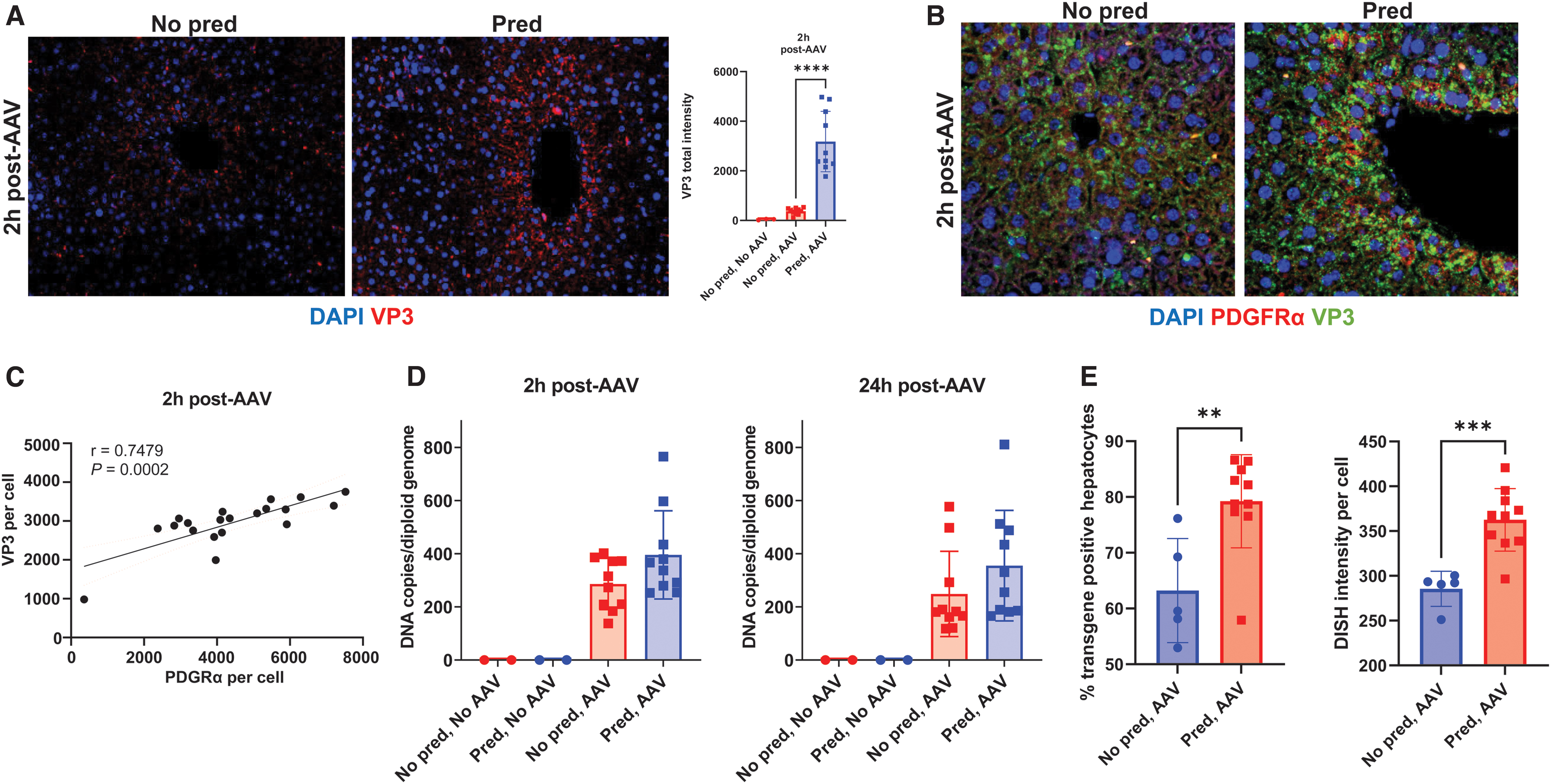
Upregulation of PDGFRα expression in hepatocytes correlates with increased AAV5 transduction.
In addition, prophylactic prednisolone in mice increased transgene DNA per cell as measured by ddPCR in the liver homogenate (Fig. 3D); however, these differences did not reach statistical significance. At the 2- and 24-h time points, the ISH signals for vector DNA were saturating, rendering quantitative analyses unfeasible. Therefore, one additional cohort was generated and taken down 1 week post-AAV dosing for ISH. The mean percentage of hepatocytes stained positive for vector DNA was significantly higher in mice treated with prophylactic prednisolone (79.2%) versus control (63.2%; Fig. 3E). Increased liver vector genomes resulted from both a higher percentage of hepatocytes taking up vector DNA and more uptake per hepatocyte (Fig. 3E). These results suggest that acute upregulation of PDGRFα proximal to AAV dosing increases levels of vector DNA within hepatocytes.
PDGFRα expression variation may contribute to intersubject variability
We assessed potential contributions of individual natural variation in PDGFRα expression to variable transgene expression by comparing outcomes after AAV5-hFVIII-SQ dosing in C5BL/6 and BalbC mice. In C57BL/6 mice naive to AAV or corticosteroid treatment, endogenous PDGFRα liver expression is highly variable (Fig. 4A); after AAV5-hFVIII-SQ dosing, the percentage of hepatocytes stained positive for PDGFRα significantly correlated with those positive for vector genome DNA (Fig. 4B).
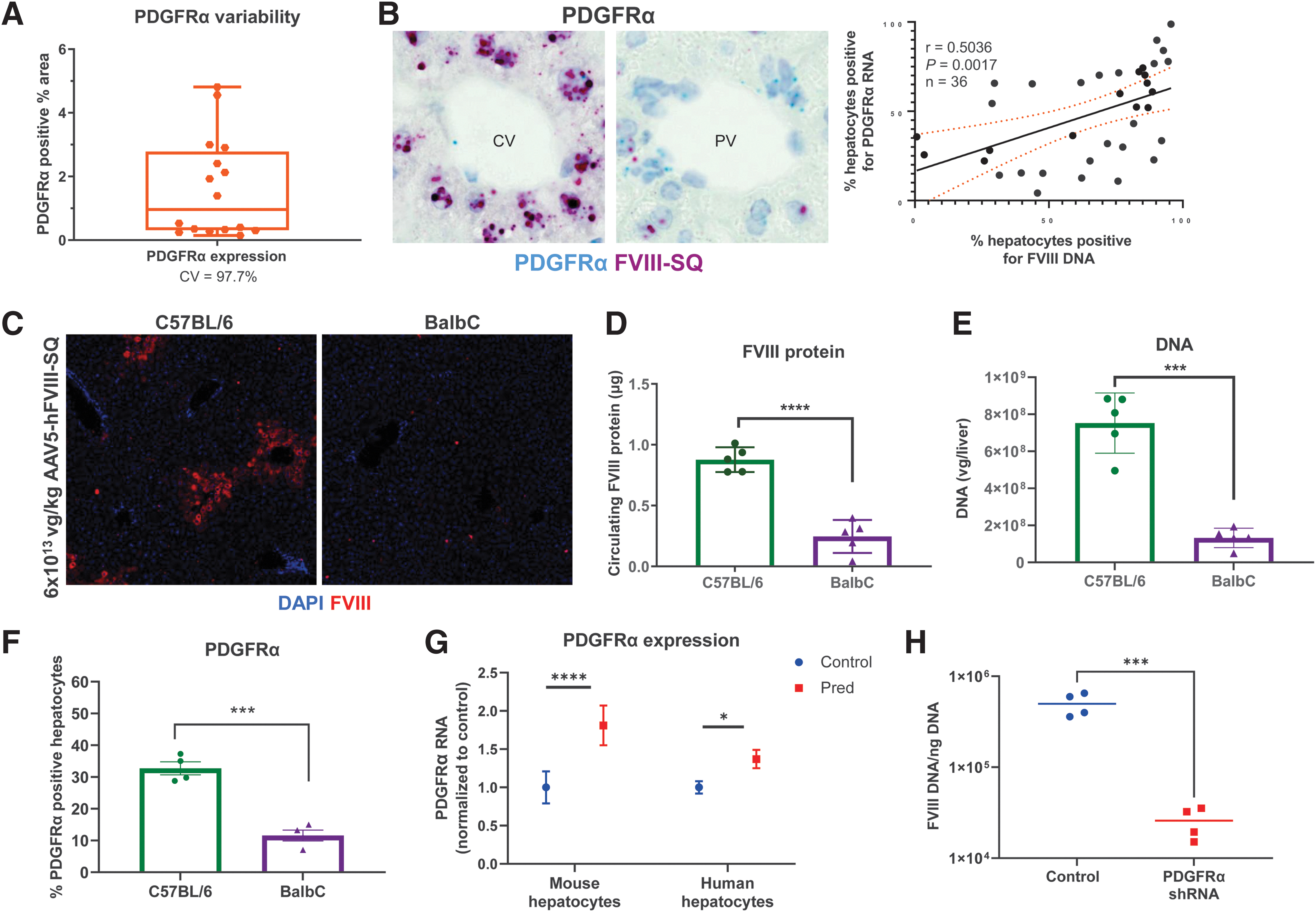
Relationship between PDGFRα and AAV5 transduction.
When comparing outcomes among laboratory mouse strains after AAV5-hFVIII-SQ treatment, BalbC mice had fewer hepatocytes expressing transgene protein (Fig. 4C), lower circulating transgene protein product (Fig. 4D), and lower levels of vector DNA (Fig. 4E) than C57BL/6 mice. Interestingly, a significantly lower percentage of hepatocytes expressed PDGFRα (but not PDGFRβ; data not shown) in BalbC mice versus C57BL/6 mice (Fig. 4F).
In vitro results confirm prednisolone increases PDGFRα expression, affecting AAV5 transduction
To assess if prednisolone affects PDGFRα expression in human hepatocytes, we treated primary hepatocytes with prednisolone. PDGRFα transcripts significantly increased in both human and murine hepatocytes (Fig. 4G), as well as monkey and dog hepatocytes (data not shown). Furthermore, shRNA knockdown of PDGFRα in the human hepatocyte HepG2 cell line significantly decreased AAV5 transduction (Fig. 4H). Altogether, prophylactic prednisolone increases PDGFRα expression, resulting in increased transduction of AAV5 vectors, leading to more vector DNA in hepatocytes, and improved transgene expression.
DISCUSSION
We investigated whether prophylactic prednisolone before AAV administration affected transgene expression. In mice, prophylactic prednisolone before AAV vector dosing increased steady-state transgene serum protein levels over 12 weeks and appeared to reduce interindividual variability in transgene protein expression. In mice and NHPs, prophylactic corticosteroids increased vector genome copy numbers in the liver. We then investigated mechanisms of improved transgene expression focusing on time points proximal to AAV dosing, since AAV uptake and acute innate immune responses occur rapidly following AAV administration. 41 Overall, prophylactic prednisolone increased the uptake of AAV5 vectors by suppressing innate immune responses and upregulating the expression of the AAV5 receptor PDGFRα. Together, these mechanisms increased vector genomes available to assemble into stable, full-length structures mediating long-term transgene expression.
Our RNAseq analyses reiterate that AAV transduction activates innate immune responses and prednisolone treatment modulates those responses through known pathways. 22,42 –61 By decreasing acute anti-AAV innate immunity, prophylactic steroids may reduce vector genome degradation and thus preserve more genomes postdosing, increasing available genomes to form full-length genome structures for transgene expression.
Surprisingly, prophylactic corticosteroids also modulated cellular mechanisms involved in receptor-mediated endocytosis of AAV5; this may explain why corticosteroids administered 1 week post-AAV have little effect. 22 Consistent with the role of PDGFRα in AAV5 transduction, 30,36,38 PDGFRα levels correlated with AAV5 transduction in laboratory mice strains, although differential immune responses may also have contributed. 62 –64 Knockdown of PDGFRα expression resulted in lower vector DNA levels in transduced hepatocytes in vitro. This suggests that prophylactic prednisolone increases the expression of the AAV5 coreceptor PDGFRα and decreases the expression of the competitive PDGF ligand complexes, resulting in an overall higher AAV5 uptake by hepatocytes. A significantly higher PDGFRα protein expression was present in hepatocytes 2 h after AAV dosing in mice treated with prophylactic prednisolone, coinciding with rapid cellular uptake of AAV vectors postdosing. 39,40
Collectively, these data indicate that variation in AAV5 receptors, particularly PDGFRα, may be a host factor contributing to the variable response seen in gene therapy trials. Although PDGFRα expression is variable in human livers (Supplementary Fig. S6), its contribution to response differences in clinical trials is unknown. 65 Insights will be provided by the ongoing phase 3 trial evaluating valoctocogene roxaparvovec with prophylactic corticosteroids in participants with hemophilia A (NCT04323098).
A limitation of this research is the use of a mouse model for human disease, given immune system differences. 66 We also did not consider the effect of vector dose, adaptive immunity, or mechanisms after capsid uptake.
Overall, prophylactic corticosteroids before dosing with AAV5-based gene therapy increase transgene expression in mice through multiple complementary mechanisms, including innate immune suppression and increased AAV5 uptake by hepatocytes. Therefore, prophylactic corticosteroids may represent an actionable strategy for increasing transgene expression and reducing interindividual variability in response to AAV5-mediated gene therapy.
Footnotes
ACKNOWLEDGMENTS
Project management support was provided by Micah Robinson, PhD, of BioMarin Pharmaceutical, Inc. Medical writing support was provided by Kathleen Pieper, PhD, of AlphaBioCom, a Red Nucleus company, and funded by BioMarin Pharmaceutical, Inc. We thank Katina Ngo of BioMarin Pharmaceutical, Inc. and Peter Colosi and Roger Lawrence, formerly of BioMarin Pharmaceutical, Inc. for their contributions to this research.
DATA AND MATERIALS AVAILABILITY
Materials and protocols will be distributed to qualified scientific researchers for noncommercial, academic purposes. The AAV5-hFVIII-SQ vector and sequence are part of an ongoing development program, and they will not be shared.
AUTHORs' CONTRIBUTIONS
Methodology: B.H., L.Z., L.X., R.M., B.B., SH.B., C.R.S., A.M.I., T.B., L.M., J.T., B.Y., S.S., W.M., W.T.K., and I.N.
Statistical analysis: C.S.
Supervision: S.F., St.B., and A.M.I. (Olink and RNAseq analyses).
Writing—review and editing: All authors.
AUTHOR DISCLOSURE
B.H., L.Z., B.Y., L.X., R.M., B.B., C.S., Sh.B., A.M.I., St.B., L.M., J.T., S.S., I.N., C.-R.S., T.B., and S.F. are employees and stockholders of BioMarin Pharmaceutical, Inc. W.T.K. and W.C.M. are former employees of BioMarin Pharmaceutical, Inc. and may hold stock.
FUNDING INFORMATION
Funding for this research was provided by BioMarin Pharmaceutical, Inc.
SUPPLEMENTARY MATERIAL
Supplementary Data
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
