Abstract
The hospital exemption (HE) allows for the use of advanced therapy medicinal products (ATMPs) next to marketing authorization (MA), but under special conditions. The HE is only applicable to individual patients treated in the hospital setting and it is limited to member states of the European Union (EU); HE is mainly conceded to the academic centers that developed the ATMP, being granted by the national competent authority (NCA), which, in the case of Spain, is the Spanish Agency of Medicines and Medical Devices (AEMPS). The HE follows strict standards of traceability, pharmacovigilance, and quality. In February 2021, our ATMP ARI-0001, a new autologous chimeric antigen receptor (CAR) targeting CD19, was approved by AEMPS under HE for patients >25 years with relapsed or refractory CD19+ acute lymphoblastic leukemia. This authorization was a first step in the development of, and access to, academic CAR T cell products in the EU. The fact that HE is limited to a specific country and hospital, the need of continuous evaluation by the NCA, and the potential future overlap with other centrally approved ATMPs, suggest that the HE could be used as an intermediate step before obtaining a centralized MA by the European Medicines Agency.
In 2011,
This achievement was the result of a long process of documentation of the efficacy and safety of ARI-0001 in the CART19-BE-01 clinical trial, designed to evaluate ARI-0001 in the most common CD19+ B cell malignancies (acute lymphoblastic leukemia [ALL], chronic lymphocytic leukemia, and non-Hodgkin's lymphoma [NHL]). 1 The trial was approved by Hospital Clinic's Review Board in February 2017, and by the AEMPS in May 2017. The AEMPS considered that the results were most convincing in patients >25 years with relapsed or refractory (R/R) ALL, thus becoming the indication approved as HE. 2
In contrast to marketing authorizations (MAs), HEs are awarded by national competent authorities (NCAs), allowing for the provision of the ATMP under special conditions: (1) it is limited to each member state of the European Union (EU) under specific regulations; (2) it is limited to the authorized hospital, developing mainly the ATMP; and (3) it is used under the responsibility of that hospital's staff members. This approval pathway, established by European Regulation [EC] 1394/2007, the amending Directive 2001/83/EC and the EC No 726/2004, was complemented by Spanish legislation on ATMPs (RD 477/2014), clearly requiring clinical data on efficacy and safety before any HE authorization. Next to Spain a total of nine countries in EU have developed HE rules until now, although there are some specific aspects that introduce some differences between these “national implementations.” 3,4 Following this pathway, ARI-0001 becomes the first treatment based on genetically modified cells developed in the EU, and the first CAR T cell therapy authorized by an NCA, although outside MA pathway, a fact that can be considered a milestone for human cell and gene therapy in the EU. Developed as a nonindustrial way with the same quality and safety standards of the European ATMP regulation, ARI-0001 was conditionally authorized because it addressed an unmet medical need: the treatment of “adults” (>25 years old) with R/R ALL. Although Kymriah® (tisagenlecleucel, developed by Novartis) was authorized by the FDA and EMA for R/R ALL patients <25 years of age, adult with R/R ALL also have a dismal prognosis, and ARI-0001 offers a hope for them.
The HE authorization might also mean a step forward for approvals of ATMPs for specific medical needs not covered by drugs that are awaiting authorization by the EMA, or for orphan indications that, due to a potential small number of patients' tributary of that ATMP, no pharmaceutical company is interested in investing the huge resources needed for applying to the EMA. Thus, ARI-0001 can become not only a model closer to real clinical practice than industrial proposals, but also a key tool for EU national health systems to increase patient access to ATMPs. 2
ALL is a rare disease (estimated overall annual incidence in Europe is just 1.28 per 100,000 inhabitants), especially in the adult population. ALL prognosis in adults is poor, with cure rates of only 40%, having even much poorer outcomes in R/R patients. 5 CART19 therapy revolutionized the management of CD19+ disorders, but only one was centrally approved for ALL and this approval was restricted to patients <25 years. Thus, among the current CART19 product authorized by the FDA and/or EMA, 5,6 Kymriah was approved for ALL <25 years old and also for diffuse large B cell lymphoma, the most frequent form of NHL. Other CART19 products (Yescarta®, Tecartus®, and Breyanzi®) are also indicated for diverse types of NHL, whereas the fifth approved CAR-T cell product, named Abecma®, targets B-cell maturation antigen and is intended for patients with multiple myeloma (Table 1). Currently, ARI-0001 is the only CART19 products authorized for patients >25 years with R/R ALL.
CD19-CART products approved for the indicated clinical indications comparing pharmaceutical versus academic CART
ALL, acute lymphoblastic lymphoma; CART, chimeric antigen receptor T cell; DLBCL, diffuse large B cell lymphoma; MCL, mantle cell lymphoma; mAb scFv, monoclonal antibody-derived single chain variable fragment; Pharma, pharmaceutical; Acad, academic; OS, overall survival.
We initiated the development of ARI-0001 in 2012 (Fig. 1; chronology of events for ARI-0001). This is a new construct based on A3B1, a monoclonal antibody generated >30 years ago at the Immunology Department, which was linked to structural and stimulatory sequences of CD8, CD137, and CD3ζ. After preclinical experiments showing the efficacy and specificity of ARI-0001 against CD19+ cell lines (benign and malignant) and demonstrating robust and reproducible lentivirus and ARI-0001 cell production, its safety and efficacy in patients was assessed in CART19-BE-01, the clinical trial conducted at HCB and the pediatric Hospital Sant Joan de Déu. Forty-seven patients with R/R CD19+ malignancies were included, with a median age of 47.5 years (range 3–67); ALL accounted for 80% of these patients. Overall, safety and efficacy outcomes achieved were similar data to those of other academic and commercial products. 2 ARI-0001 is currently being further investigated in a second clinical trial for adult (≥18 years) patients with R/R CD19+ ALL (NCT04778579) for providing additional continuous data to AEMPS. Looking globally at ATMP developments in Spain, ARI-0001 was the second ATMP approved as HE by the AEMPS, the first being NC1 (a cell product of autologous mesenchymal stromal cells developed by Hospital Puerta de Hierro in Madrid for patients with chronic traumatic spinal cord aftermath) 2 years before.
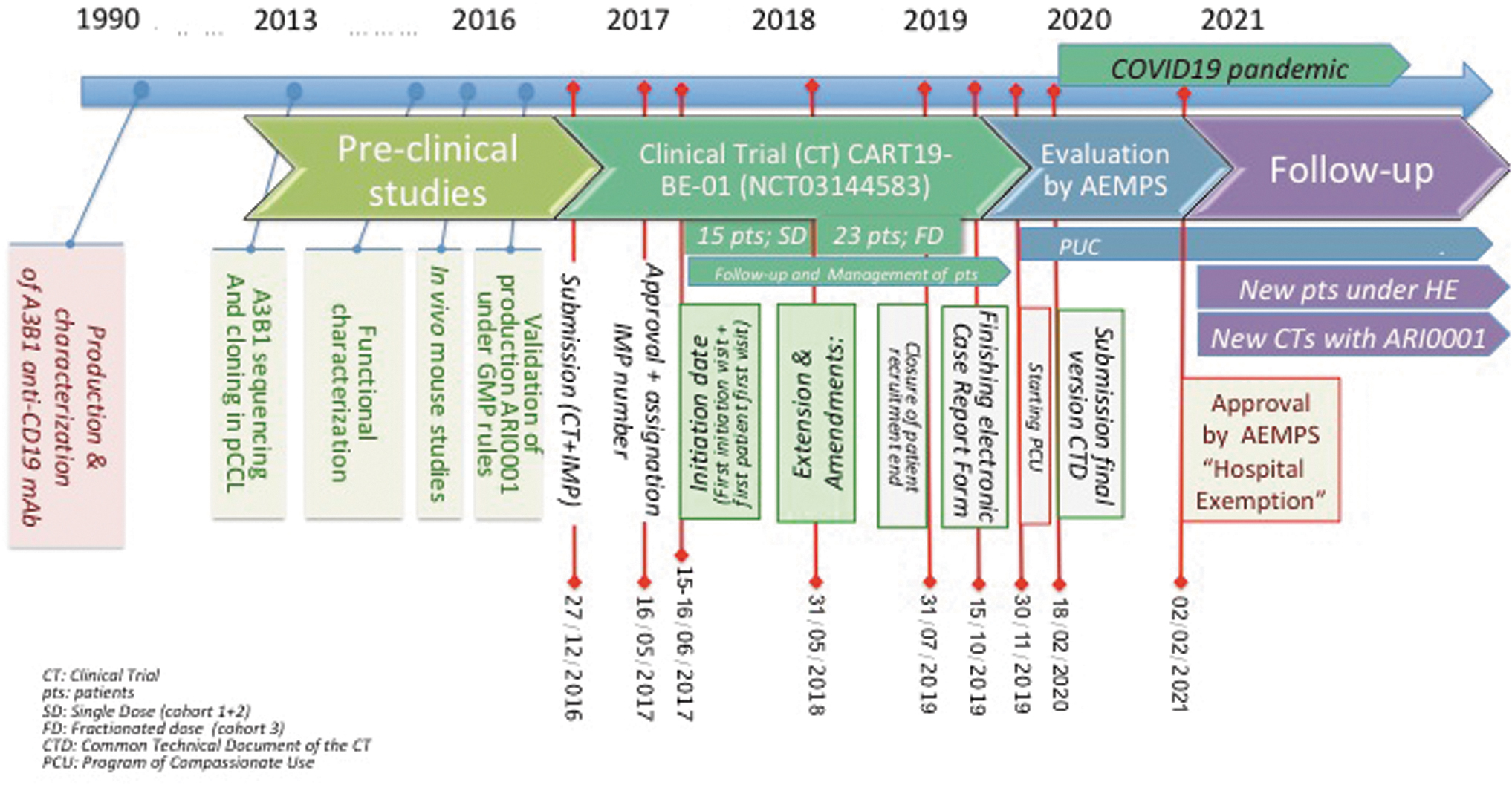
Chronology of events developed until final approval of ARI-001 under HE by AEMPS. Milestones are pointed out by ended-with-dots lines and a specific date. AEMPS, Spanish Agency of Medicines and Medical Devices; CT, clinical trial; CTD, common technical document; FD, fractionated dose (cohort 3); HE, hospital exemption; pts, patients; SD, single dose (cohort 1 + 2). Color images are available online.
In this study, we would like to emphasize that HE might be considered as a great opportunity for the development of ATMP in Europe (as for those countries that endorse and embrace this option), because it is close to the real clinical practice and providing rapid access to treatments, as it happens with R/R patients with hematologic malignancies. However, some barriers have still to be overcome to get the best out of it, especially the harmonization of HE rules across EU countries. 7,8 Sharing common standards of quality, safety, and efficacy would provide consistency and increase confidence in HE-ATMP. 7 It is not just academia and industry that demand harmonization of HE rules, 8 but also national health care systems that must develop the best way to manage these options, when they demonstrate to be useful for patients. Some therapies (mainly those for diseases with low incidence, such as most of the pediatric cancers) will only be accessible to patients in the form of academic proposals. Academic centers could develop ATMPs for orphan diseases and ask for HE authorization, allowing for the use of nonprofit products in different countries. In fact, it is important to have in mind that many, if not all, of the available commercial products were acquired from academic institutions once the pharmaceutical companies were aware that they could be profitable. This aspect is relevant considering the high economic impact of the ATMPs, especially if they demonstrate efficacy for solid tumors. It is possible that the high prices for ATMPs might be untenable for the National Health Systems if only the commercial products are available. In this sense, the HE could provide a complementary opportunity and balance these costs. Although costs (public academic ATMPs) and prices (commercial ATMPs) are obviously never comparable (and even less when indications are different), the cost in Spain of ARI-0001 is <1/3 of the price of commercially available CART19 products. This difference is a direct aspect in favor of the sustainability of EU health care systems, but it is not the main reason for this development.
ARI-0001 was developed and brought to the patient bedside, following the HE route; now, the “conditional license,” granted for 3 years, is subjected to yearly follow-up reports and a re-evaluation of data for another 5-year renewal, 9 by adding essential further data on efficacy and safety. These aspects carry several issues, important for the implantation of the product and its later improvement. Some of these regulatory concepts have been previously broadly commented 10 ; in this article we want to spotlight the HE approval of ARI-0001 as a model for the future development of ATMPs in the EU. ARI-0001 was possible thanks to the work and expertise of a multidisciplinary team of almost 200 professionals, which participated in the successive steps of this process. The availability of logistic resources (clean room with CAR-T manufacturing facilities) was also a key pillar for this development that also reflects the Spanish commitment with R&D&I in general. ARI-0001 was financially possible thanks to public-civil cooperation, including the “ARI Project,” but also several public grants of the Instituto de Salud Carlos III.
Summarizing, the HE pathway allows for the treatment of patients with unmet medical needs, including a dose-customization according to the patients' specific characteristics. By reducing manufacturing and shipping costs, they can be more affordable for public and private payers. The comprehensive requirements of the Spanish ATMP legislation, in accordance with the EU Regulation, allowed developing a CAR-T cell therapy with safety and efficacy profiles similar to commercial CAR-Ts. Confidence in these products is a critical point if we want the HE to become an option for the future use of ATMPs. However, the fact that HE is limited to the specific country and hospital in which it was developed, the continuous scrutiny by the AEMPS and the potential overlap with other centralized authorization of ATMP suggests using HE as an intermediate step before obtaining a centralized MA by the EMA.
Conclusions and Future Remarks
The pathway for the approval of CAR-T ATMP as HE is well defined in several EU member states. The process is feasible and could become an intermediate step before requesting a centralized MA by the EMA. The EMA approval, if obtained, should finally allow the widespread use of academic ATMPs, thus gaining experience in the “real world.” The HE could help achieve a faster and more personalized use for ATMPs, improving the development of many other promising therapies in more integrated way across the EU. It is time for the “legislative power” in each country to decide if they want to bet on this alternative option or if they just wish to remain operating under the classical pharmaceutical-based rules. To have future, new challenges require new tools with new standards for a newer, more personalized, and more sustainable reality in our world, … at least at the health level.
Footnotes
Authors' Contributions
M.J. is responsible of the definition of the main structure and first redaction. J.D., G.C., and E.T. have the main knowledge about regulatory aspects defined in the article. J.D. is the PI of the clinical trial (CT) that allowed the HE; G.C. was the responsible of obtain and analyze data in CT, E.T. defined production of ATMPs. A.U.-I. was a key author for defining main aspects that were discussed.
Acknowledgments
Thanks to Dr. Michael Edel for the reading of this article. Thanks also for the support of CatSalut. We thank the patients who participated in the clinical trial and their families, friends, caregivers, and referring physicians.
Author Disclosures
The main conflict of interests derives of the direct involvement of the authors in the clinical trial CART19-BE-01, although any economical benefit was received for them.
Funding Information
Crow-funding (mainly ARI project), and grants cofinanced by Instituto de Salud Carlos III, Spanish Ministry of Health (Fondo de Investigaciones Sanitarias PI18/00775, PICI14/122 PICI18/00012, and complementary grant for CONCORD-023), Fondo Europeo de Desarrollo Regional (FEDER), 2017SGR00792 (Agència de Gestió d'Ajuts Universitaris i de Recerca; Generalitat de Catalunya), and “La Caixa” Foundation (CP042702/LCF/PR/GN18/50310007).
