Abstract
Adeno-associated viruses (AAVs) are nonenveloped viruses that have become popular gene transfer vectors to deliver DNA to target cells in clinical gene therapy. Iodixanol-based density gradient is one of the widely used purification methods for serotype-independent AAVs. However, residual iodixanol in AAV could be a safety concern, and further purification to remove this process-related impurity is typically needed. An analytical assay with high sensitivity is essential for the detection of residual iodixanol to ensure the safety of AAV products. We developed a liquid chromatography-mass spectrometry method with the limit of quantification of 0.01 μg/mL for residual iodixanol measurement in AAVs. The method also demonstrated linearity over four orders of magnitude that allows quantifying a high iodixanol concentration in in-process samples with excellent recovery and accuracy. In addition, we further explored a highly efficient purification method for removal of the residual iodixanol, to minimize the safety concern from iodixanol as a process impurity.
Introduction
Recombinant adeno-associated viruses (AAVs) have become widely used vectors in gene therapy due to their low toxicity and long-term expression in a variety of tissue types. There have been >150 ongoing clinical trials using AAVs for various generic diseases, such as muscular dystrophy, hemophilia, macular degeneration, Parkinson's disease, and so on. 1 –3 AAVs are small icosahedral nonenveloped viruses with single-stranded DNA genome encapsulated in a protein capsid shell. There have been 13 AAV serotypes reported. 4
Cesium chloride- and iodixanol-based density gradients are the core step among the most common purification methods for serotype-independent AAVs. 5 –7 Cesium chloride approach contains multiple steps and is typically considered to be time consuming, compared with the iodixanol method. It was also reported that iodixanol-purified AAV preparations showed higher vector purity. 5,7 For example, an iodixanol-based approach has been applied in separation of empty capsids and full capsids previously. 8 However, the residual iodixanol in the final AAV preparations may present a safety concern, and therefore, additional purification is warranted.
To ensure clearance of residual iodixanol in AAVs, an analytical method that can quantify iodixanol with high sensitivity is needed. Previously, multiple high-performance liquid chromatography (HPLC) 9 –13 methods for iodixanol quantification have been reported. With higher sensitivity and better selectivity, the combination of LC and mass spectrometry (MS) offered the ability to separate, detect, and quantify target molecules in complex matrices, such as plasma. 14
In this study, we developed and applied a reversed-phase liquid chromatography-mass spectrometry (LC-MS) method for quantification of residual iodixanol in AAV-based gene therapy development. Using iohexol, a structurally similar compound, as the internal standard (IS), we were able to quantify as low as 0.01 μg/mL iodixanol in AAV drug substance (DS). In addition, we further developed a highly efficient purification method for removal of iodixanol in AAVs that can reduce residual iodixanol over six orders of magnitude. We applied these methods in the characterization of the final AAV products and demonstrated that the high-sensitivity analytical assay and highly efficient purification approach are essential to control in-process impurities, and hence ensure drug safety.
Materials and Methods
Materials
Iodixanol (1343517) and iohexol (1344600) were purchased from United States Pharmacopeia (Rockville, MD). LC-MS grade water (W6), isopropanol (A461), water with 0.1% formic acid (FA) (LS118), and acetonitrile (ACN, A955) were from Fisher Scientific (Waltham, MA). Recombinant AAVs were produced by PackGene (Worcester, MA) or Biogen (Cambridge, MA).
Sample preparation
The iodixanol stock solution and iohexol stock solution were freshly prepared with AAV formulation buffer at concentration of 2 and 1 mg/mL, respectively. For working solutions, the stock solutions were further diluted to 100 μg/mL (iodixanol) and 10 μg/mL (iohexol), respectively, with formulation buffer.
Nine iodixanol calibration standards ranging from 0.01 to 100 μg/mL in AAV formulation buffer were prepared, that is, 0.01, 0.05, 0.1, 0.5, 1, 5, 10, 50, and 100 μg/mL.
The quality control (QC) samples were prepared with different iodixanol stock solutions, which was prepared freshly and separately. Five concentrations were prepared at 0.0167, 0.167, 1.67, 4.67, and 46.7 μg/mL with AAV formulation buffer, representing low to high QC samples.
All samples except AAV-A, including AAV-B, iodixanol calibrants, and iodixanol QC samples were spiked with the IS, iohexol working solution, at a ratio of 9:1 (v:v), before MS analysis.
LC-MS separation and quantification of iodixanol
All samples were analyzed by LC-MS on an Exactive™ Plus Orbitrap Mass Spectrometer (Thermo Fisher Scientific) coupled with an Agilent 1290 UHPLC system (Agilent Technologies). Ten microliters of samples was injected and separated using a ZORBAX Eclipse XDB-C18 column (2.1 × 150 mm, 5 μm; Agilent Technologies) at 40°C. Mobile phase A consisted of water with 0.1% FA, and mobile phase B was ACN. The gradient started at 0% B for 4 min, and then increased to 40% B over 9 min, and to 80% B in an additional 2 min, followed by 1 min at 80% B and re-equilibration at 0% B for 10 min. The flow rate was 0.2 mL/min. MS data were collected by Orbitrap from 150 to 2,250 m/z with resolving power at 70,000. The MS settings included advanced gaining control target at 3e6 and maximum injection time 200 ms. The mass spectra were processed using Xcalibur 3.1 QuanBrowser (Thermo Fisher Scientific). Calibrants were run from low to high concentration in duplicate.
LC-MS method qualification
Specificity and selectivity
The extracted ion chromatograms (EICs) of injections from iodixanol, iohexol, formulation buffer blank, AAV-A DS (free of iodixanol), and AAV-B DS (with IS added) were evaluated, to confirm there was no chromatographic interference from AAV sample matrices.
Accuracy, precision, and limit of quantification
Three replicates of QC samples at five concentrations of iodixanol were analyzed by LC-MS to evaluate method repeatability and reproducibility. Spike recovery was calculated at the same five levels for accuracy assessment. Limit of quantification (LOQ) was determined as the lowest concentration of calibrant that can be detected with an acceptable level of repeatability, accuracy, and precision.
Purification to remove residual iodixanol
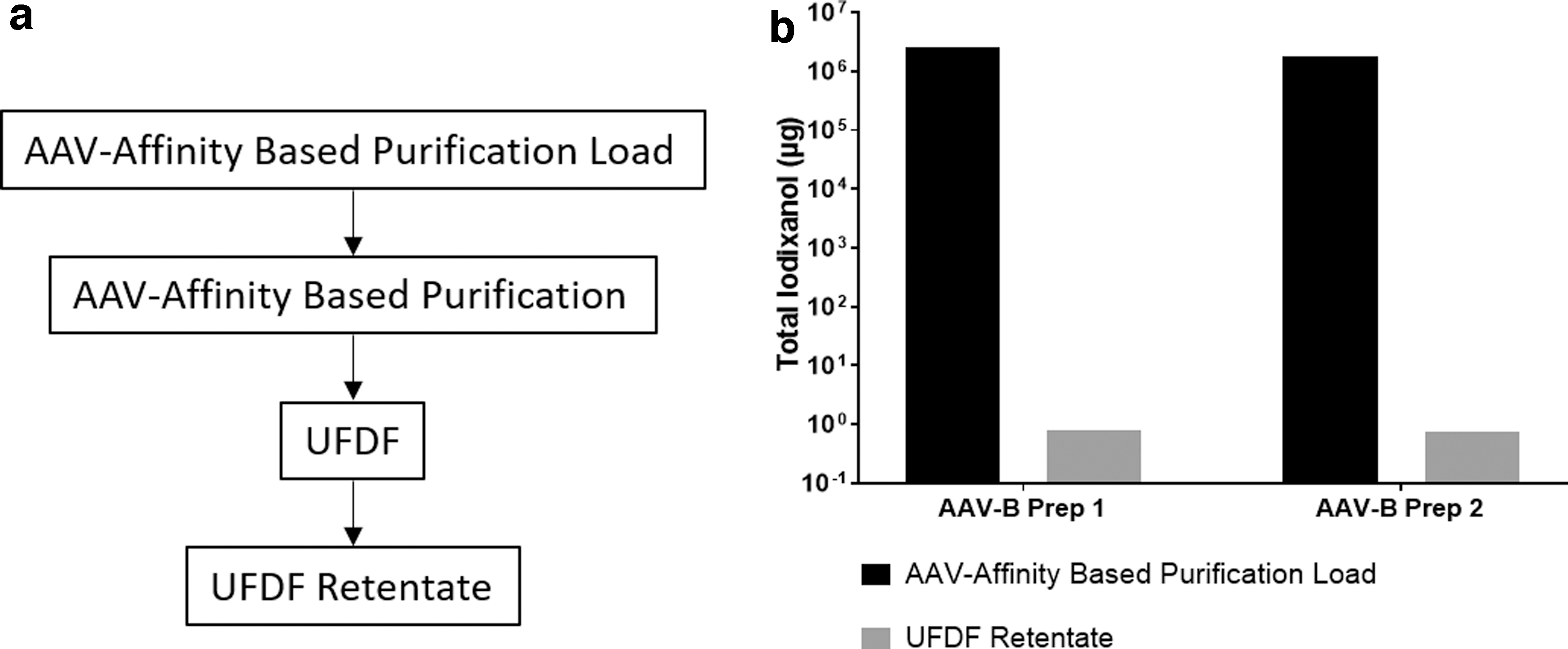
HiScale 26/40 column hardware (Cytiva) was packed with POROS™ CaptureSelect™ AAV affinity resin (Thermo Fisher Scientific) to a bed height of 7.5 cm for a final column volume of 40 mL. The column was loaded with 400 mL of equilibration buffer at pH 7.5 for each experiment containing a combination of iodixanol and AAV-B vector as per Table 1. Two different AAV-B preparations were used for this study as a demonstration. The column was then eluted via low pH buffer and the eluate fraction was collected into a vessel containing buffer for adjustment to a neutral pH. All steps were performed at a flow rate of 150 cm/hr. Each experiment used distinct but identical POROS CaptureSelect AAV affinity columns from the same lot. An AKTA Pure 25 system with UNICORN software (Cytiva) was used for each purification run.
Experiment design for iodixanol purification
Total mass of iodixanol measured to be 8.2E+05 μg.
AAV, adeno-associated virus.
The pH-adjusted AAV affinity-based purification eluate fractions for experiments containing AAV vector were concentrated and buffer exchanged into formulation buffer via ultrafiltration and diafiltration (UFDF) using hollow fiber filters. The levels of AAVs were estimated via genome titer quantification using digital droplet polymerase chain reaction (ddPCR), and residual iodixanol levels were determined by the LC-MS method described previously.
ddPCR genome titer
AAV samples were treated with DNase I, diluted, and combined with ddPCR™ Supermix for Probes (no dUTP; Bio-Rad) and a custom transgene-specific primer/probe set. Droplets were produced and analyzed using the QX200 with AutoDG system and the QuantaSoft 1.7 software from Bio-Rad.
Results and Discussion
LC-MS separation of iodixanol and IS
Iodixanol is a dimeric, nonionic, and hydrophilic compound with its structure shown in Fig. 1a. It was reported that iodixanol presents as a mixture of three geometric isomers at equilibrium, exo-exo, exo-endo, and endo-endo forms. 15 With reversed-phase LC, two, or, in some conditions, three peaks could be observed due to exo-endo isomerism. 9 –11,16 To keep all isomers as close as possible for consistent integration while maintaining full separation from the IS and AAV, we employed a C18 column for the optimal separation, as shown by the EICs in Fig. 1b. Iodixanol eluted as two partially resolved peaks at ∼9.2 min, whereas the IS was present as a single peak at 8.0 min. Both iodixanol peaks were integrated and sum of peak area was used in all data analysis. Representative total ion chromatogram and corresponding mass spectra are shown in Supplementary Fig. S1. No deiodinated species was observed for both iodixanol and iohexol within the conditions evaluated in this study. 17
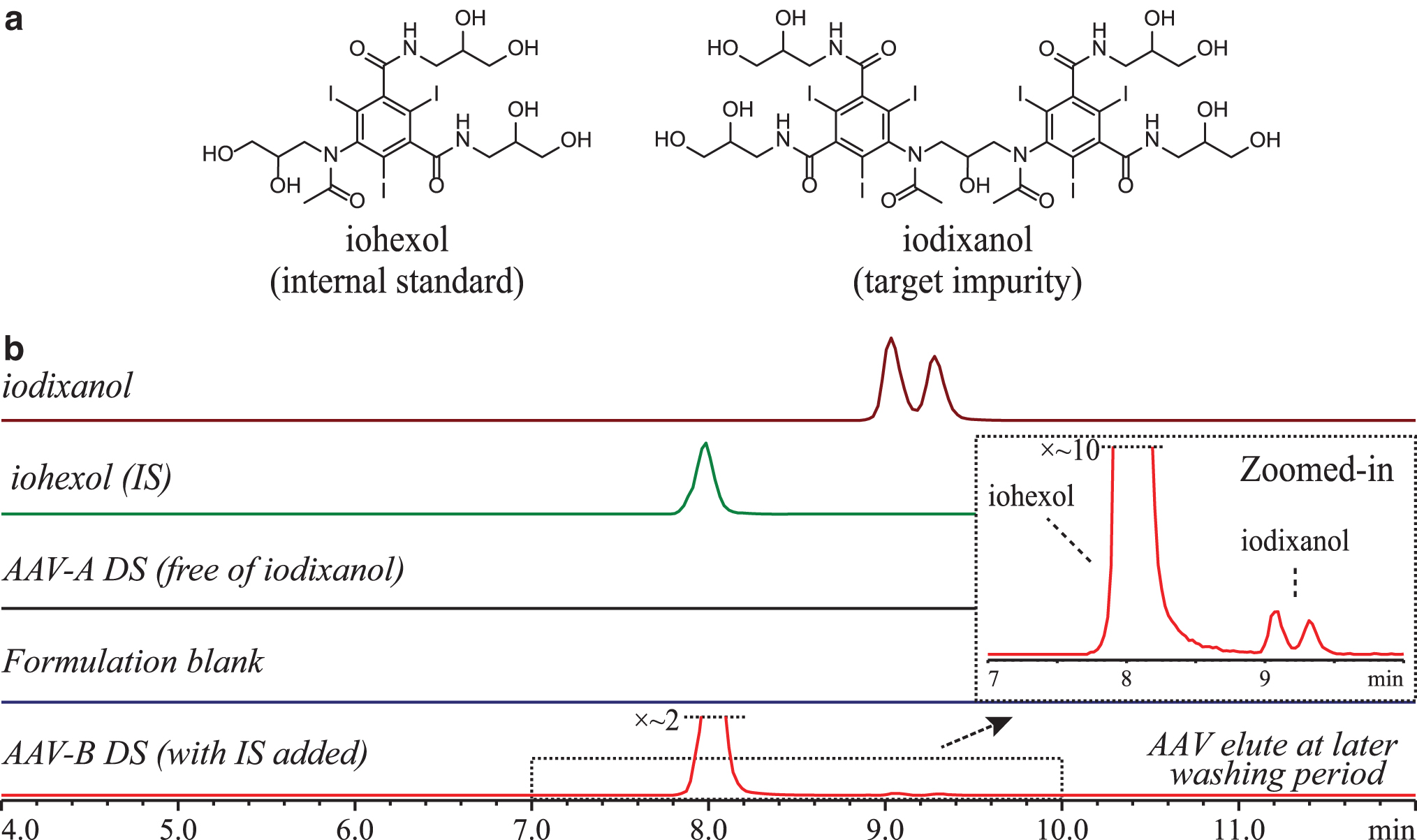
LC-MS method qualification
As shown in Fig. 1b, there was no chromatographic interference peak(s) observed by comparing EICs of iodixanol, IS, pure AAV DS, and formulation blank. AAVs eluted late during chromatographic washing step with high percentage of mobile phase B.
The calibration was performed with the area ratio defined by the equation below, against iodixanol concentration. Peak areas of iodixanol and iohexol were measured by EICs. The ratio of these peak areas was calculated. Linearity of the peak area ratio as a function of iodixanol concentration was then assessed. A good linear response was observed for iodixanol calibration with concentrations ranging from 0.01 to 100 μg/mL, with typical R
2 > 0.99.
The EICs of calibrants, including lower LOQ at 0.01 μg/mL, are shown in Fig. 2. Precision and accuracy assessment of the LOQ are summarized in Supplementary Table S1, and a representative linear standard curve is shown in Supplementary Fig. S2. Accuracy of data points at LOQ ranged from 88.9% to 103.8%, and with 30 as the lowest signal-to-noise ratio observed.
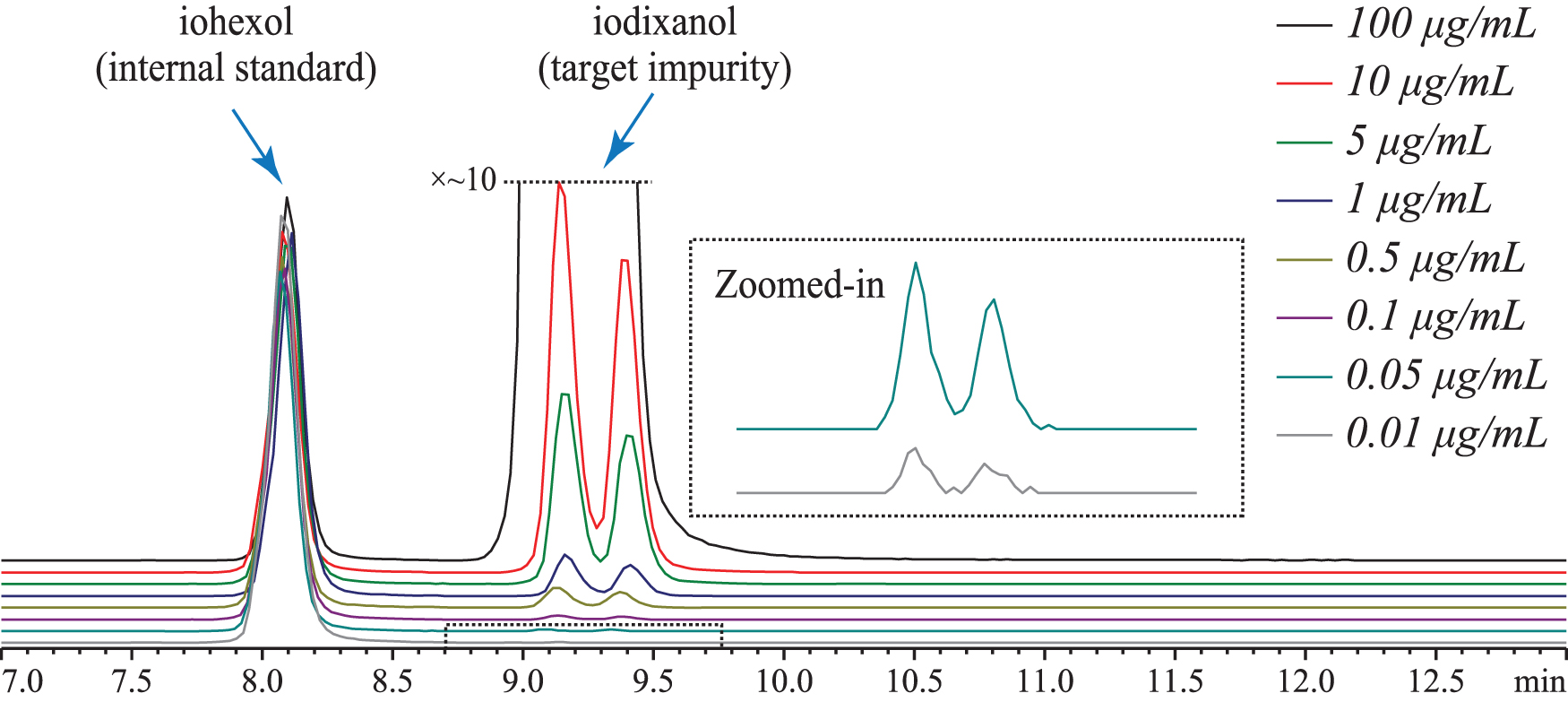
LC-EICs of iohexol (IS) and iodixanol at different calibration concentrations (trace for 50 μg/mL not shown), acquired during LC-MS analyses with a reversed phase column. The inset shows zoomed-in views for the calibrants at 0.05 and 0.01 μg/mL. Extracted masses monitored: m/z 1550.72 (iodixanol) and m/z 821.89 (iohexol) with ±5 ppm mass tolerance.
Results for precision and accuracy of this method are summarized in Table 2, with raw data listed in Supplementary Table S2. Recovery of iodixanol ranged from 93.7% to 106.2%.
Accuracy and precision for iodixanol quantification
RSD, relative standard deviation.
Purification to remove residual iodixanol
For experiments given in Table 1 that included iodixanol in the load, such as AAV-B preps, the load phase of the AAV affinity-based purification runs displayed considerable absorbance at 260 nm, as detailed in Supplementary Fig. S3. Since iodixanol absorbs strongly at 260 nm, 18 this elevated load absorbance suggests that iodixanol partitions in the flow-through fraction (FTF) for those experiments. This is supported by the detection of iodixanol in the FTF using the LC-MS assay described above. For samples that included AAV vector, a sharp absorbance peak at 260 nm during the elution phase is observed in the chromatograms, corresponding to detection of AAV vector in the eluate. There was no absorbance at 260 nm observed in the chromatogram during the experiment that included neither iodixanol nor AAV vector. The combination of online absorbance and residual iodixanol quantification data supports the conclusions that iodixanol does not bind to the chromatography resin and that the lack of interaction between the resin and iodixanol is not impacted by the presence of AAV vector under the conditions evaluated in this study.
For the two experiments that included AAV vector in the load, the AAV affinity-based purification and UFDF step recoveries by AAV genome titer were 86% ± 3% and 98% ± 4%, respectively. The residual iodixanol levels in the AAV affinity-based purification load for those experiments were reduced by greater than six orders of magnitude to final mass of 0.80 μg (0.09 μg/mL) and 0.74 μg (0.12 μg/mL), respectively, in the UFDF retentate for Prep 1 and Prep 2 (Fig. 3). Residual iodixanol levels in the iodixanol-spiked blank load sample were reduced from a total mass of 8.2E+05 μg to 6 μg in the AAV affinity-based purification eluate (Supplementary Fig. S4). The data indicate that the iodixanol removal capability is primarily due to an efficient AAV affinity-based purification step.
Safety assessment of iodixanol
The safety of the residual iodixanol is supported by its clinical application as a radiographic contrast agent indicated for the intra-arterial and intravenous (IV) procedures. 19 The intra-arterial or IV doses administered depend on the specifics of the procedure and the patient's characteristics, and vary from 640 mg iodine up to 16,000 mg iodine per injection volume. 19 Intrathecally (IT) injected iodixanol at 810 mg iodine/dose was also described. 20 However, in the case when iodixanol is an impurity with no benefit to the patient, the risk assessment is based on establishing the permitted daily exposure (PDE) values. PDE represents a dose (oral, IV, IT, or ocular) that is unlikely to cause an adverse effect by that route if an individual is exposed at or below this dose every day for a lifetime. 21,22 PDE is derived from the no-observed effect level, or the lowest observed effect level in the most relevant animal study divided by appropriate adjustment factors that describe the robustness of the data. The detailed process of establishing the PDEs for iodixanol is outside of the scope of this article. However, hypersensitivity reactions including anaphylaxis are the major safety concern for small quantities of iodixanol. 19,23 Considering that there is no specific threshold for triggering these types of reactions, every effort should be made to limit the amount of residual iodixanol in the drug product. For example, our purification method brought down the residual iodixanol level to 0.12 μg/mL reportable level, which would result in the delivery of only 12 ng of iodixanol in 100 μL intraretinal injection volume, thus minimizing a likelihood of an adverse reaction from iodixanol as the process impurity.
Conclusions
An LC-MS method was developed that can quantify as low as 0.01 μg/mL iodixanol in AAVs. The method also demonstrated linearity over four orders of magnitude that allows quantifying high iodixanol concentration in in-process samples with excellent recovery, accuracy, and precision. The purification method discussed demonstrated ability to significantly remove residual iodixanol from AAVs. Overall, these combined approaches enable safe, definitive, and efficient process development for AAV-based gene therapy products.
Footnotes
Authors’ Contributions
Y.P. designed the study, performed LC-MS experiments, analyzed data, and wrote the article; R.K. designed the study, performed purification experiments, and wrote the article; Y.C. designed the study and analyzed data; V.K. performed safety assessment and wrote the article; P.C. conducted ddPCR analysis; S.L., Z.S., and B.Y. designed the study and supervised the project.
Acknowledgment
The authors thank Chris Argento for his helpful discussions.
Author Disclosure
No competing financial interests exist.
Funding Information
No funding was received for this article.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Table S1
Supplementary Table S2
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
