Abstract
The administration of adeno-associated virus (AAV) vectors to nonhuman primates (NHP) via the blood or cerebrospinal fluid (CSF) can lead to dorsal root ganglion (DRG) pathology. The pathology is minimal to moderate in most cases; clinically silent in affected animals; and characterized by mononuclear cell infiltrates, neuronal degeneration, and secondary axonopathy of central and peripheral axons on histopathological analysis. We aggregated data from 33 nonclinical studies in 256 NHP and performed a meta-analysis of the severity of DRG pathology to compare different routes of administration, dose, time course, study conduct, age of the animals, sex, capsid, promoter, capsid purification method, and transgene. DRG pathology was observed in 83% of NHP that were administered AAV through the CSF, and 32% of NHP that received an intravenous (IV) injection. We show that dose and age at injection significantly affected the severity whereas sex had no impact. DRG pathology was minimal at acute time points (i.e., <14 days), similar from one to 5 months post-injection, and was less severe after 6 months. Vector purification method had no impact, and all capsids and promoters that we tested resulted in some DRG pathology. The data presented here from five different capsids, five different promoters, and 20 different transgenes suggest that DRG pathology is almost universal after AAV gene therapy in nonclinical studies using NHP. None of the animals receiving a therapeutic transgene displayed any clinical signs. Incorporation of sensitive techniques such as nerve-conduction velocity testing can show alterations in a minority of animals that correlate with the severity of peripheral nerve axonopathy. Monitoring sensory neuropathies in human central nervous system and high-dose IV clinical studies seems prudent to determine the functional consequences of DRG pathology.
Introduction
Gene therapy using recombinant adeno-associated virus (AAV) has been linked with histopathological findings in dorsal root ganglion (DRG) sensory neurons in preclinical studies using nonhuman primates 1 and pigs. 2 The pathology manifests as mononuclear cell infiltrates and sensory neuron degeneration within the DRG in addition to secondary axonopathy, which affects both the central axon of dorsal spinal cord (SC) tracts and the peripheral axon of peripheral nerves (Fig. 1).
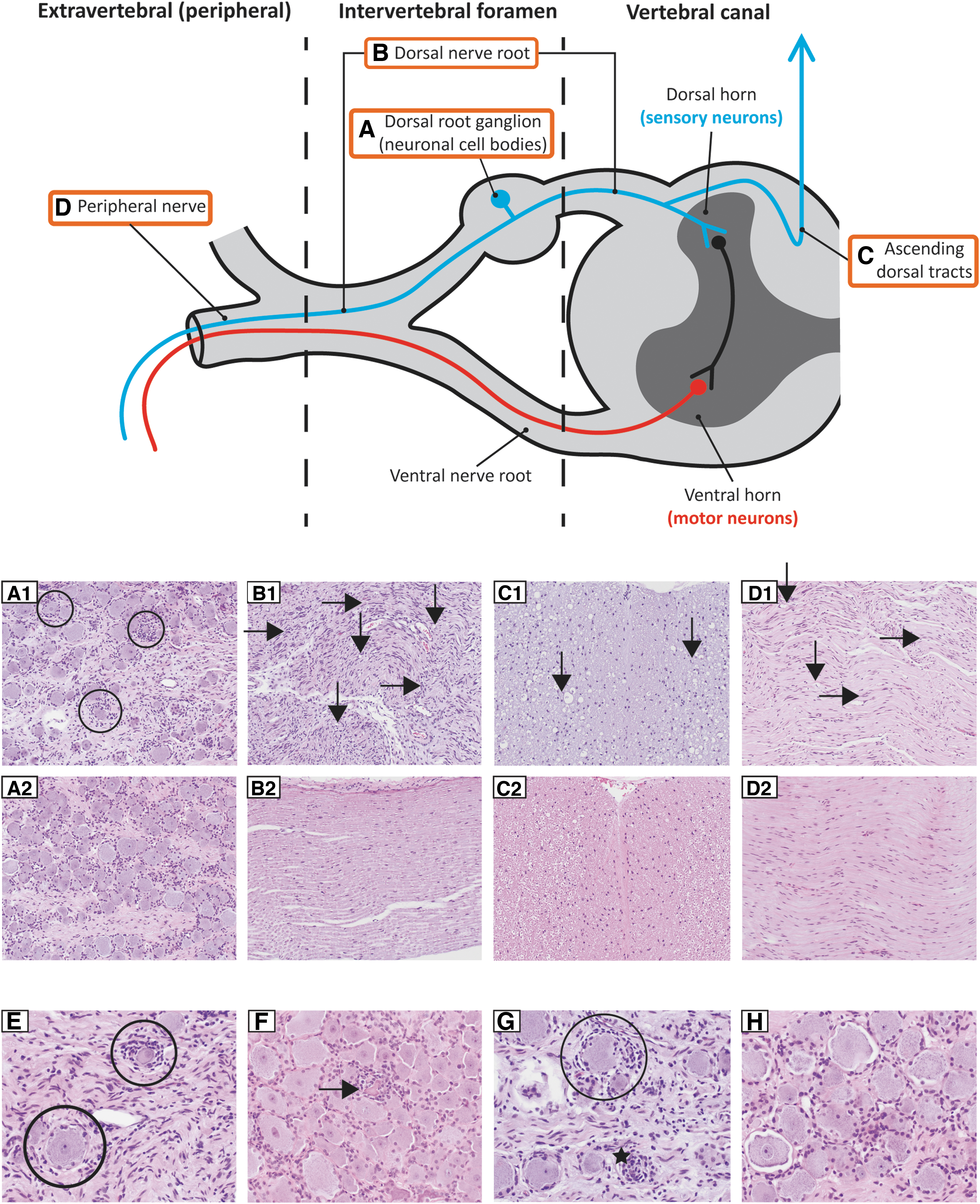
Neuroanatomy and microscopic findings. Neuronal cell bodies of the DRG
DRG pathology or toxicity has been reported in nonclinical studies using AAV administration into the cerebrospinal fluid (CSF) 1,3 –5 or systemic high-dose administration to target the central nervous system (CNS). 2 Animals remained asymptomatic in most of the studies in which pathology was minimal to moderate. More severe pathology and overt toxicity involving progressive proprioceptive deficits and ataxia were observed in a study of piglets within 14 days of high-dose intravenous (IV) vector injection. 2 Immune suppression using a combination of mycophenolate mofetil and rapamycin did not eliminate the histopathological findings in nonhuman primates (NHP). 1 The significance of DRG pathology in human clinical trials is unknown.
Because most of the NHP studies have a low number of animals and limited opportunity for statistical analysis, we aggregated data from 33 studies and conducted a meta-analysis on a total of 256 macaques to look for the effect of route of administration (ROA), dose, time course, study conduct, age of the animals, sex, capsid, promoter, capsid-purification method, and transgene.
Materials and Methods
Data availability statement
We have presented aggregated data alongside experimental details with the exception of specific transgenes, which are proprietary to the sponsors who funded the work.
Animals
This meta-analysis includes a total of 237 rhesus macaques and 19 cynomolgus macaques from 33 studies. The Institutional Animal Care and Use Committee of the University of Pennsylvania or of the Children's Hospital of Philadelphia approved all animal procedures. Rhesus macaques (Macaca mulatta) or Cynomolgus macaques (Macaca fascicularis) were procured from Covance Research Products, Inc. (Alice, TX), Primgen/Prelabs Primates (Hines, IL), or MD Anderson (Bastrop, TX), or they were donated. Animals were housed in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International-accredited Nonhuman Primate Research Program facility at the University of Pennsylvania or at the Children's Hospital of Philadelphia. Animals received varied enrichments such as food treats, visual and auditory stimuli, manipulatives, and social interactions.
Test or control article administration
For CSF administration, NHP received vectors diluted in sterile artificial CSF (vehicle) injected into the cisterna magna under fluoroscopic guidance as previously described. 6 We performed lumbar puncture under fluoroscopic guidance in anesthetized animals. After inserting a spinal needle into the L4–5 or L5–6 space, we confirmed placement by CSF return and/or by injecting up to 1 mL of contrast material (Iohexol 180). For IV administration, we placed a catheter in the saphenous vein and administered the vector diluted in sterile 1x Dulbecco's phosphate-buffered saline.
Nerve conduction velocity testing
Animals were sedated with a combination of ketamine/dexmedetomidine and placed in lateral or dorsal recumbency on a procedure table, with heat packs to maintain body temperature. The stimulator probe was positioned over the median nerve with the cathode closest to the recording site. Two needle electrodes were inserted subcutaneously on digit II at the level of the distal phalanx (reference electrode) and proximal phalanx (recording electrode). Meanwhile, the ground electrode was placed proximal to the stimulating probe (cathode).
A pediatric stimulator delivered the stimulus, which we increased in a step-wise fashion until we reached the peak amplitude response. Up to 10 supramaximal stimuli were averaged and reported for the median nerve. We measured the distance (cm) from the recording electrode to the stimulation cathode and used this distance to calculate the conduction velocity. We measured both the conduction velocity and the average of the sensory nerve action potential amplitude.
Vectors
For research studies, AAV vectors were produced and titrated by the Penn Vector Core as previously described. 7,8 Briefly, HEK293 cells were triple-transfected and the culture supernatant was harvested, concentrated, and purified with an iodixanol gradient. For Good Laboratory Practice (GLP)-compliant toxicology studies, vector was also produced by triple-transfection of HEK293 cells and purified by affinity chromatography by using a POROS™ CaptureSelect™ AAV9 resin (Thermo Fisher Scientific, Waltham, MA) as previously described. 1
Histopathology
For the majority of studies, a board-certified veterinary pathologist—initially blinded to test article/treatment groups—established severity scores defined as 0 for absence of lesion, 1 for minimal (<10%), 2 for mild (10–25%), 3 for moderate (25–50%), 4 for marked (50–95%), and 5 for severe (>95%). These scores were based on microscopic evaluation of hematoxylin and eosin-stained tissues in which % represents the proportion of tissue affected by the lesion in an average high-power field. For all GLP and some non-GLP studies, an external board-certified veterinary pathologist completed the peer review.
Severity scores from DRG degeneration and dorsal axonopathy of the SC were established from cervical, thoracic, and lumbar segments; however, the number of sections evaluated varied across studies. In some studies, scores were assigned to individual sections of DRG and SC when multiple tissue sections were present on a slide for a given segment; these scores were then averaged to obtain a single representative score. We consider SC axonopathy a better indicator of DRG pathology, as it represents the collation of axons coming from all the DRG. We define DRG pathology as histopathological findings within the DRG cell bodies and SC or SC alone throughout this article.
Peripheral nerve axonopathy grades were established based on evaluation of the median (proximal and/or distal), radial, ulnar, sciatic (proximal and/or distal), peroneal, tibial, and/or sural nerves. When we evaluated the proximal and distal median nerve, the proximal segment corresponded to the portion of nerve from the brachial plexus to the elbow and the distal segment corresponded to the portion of nerve from the elbow to the palm of the hand. When present, a severity score was given for periaxonal (i.e., endoneurial) fibrosis in peripheral nerves. For studies when peripheral nerves were evaluated bilaterally, axonopathy and periaxonal scores were averaged for each nerve.
Data extraction
We extracted the raw data, including pathology scores and all pertinent study information from study files, and aggregated them into a single Excel spreadsheet. Two persons independently extracted and sorted the scores based on predetermined search criteria to generate graphs and perform statistics. In case of discrepancy between the extracted outputs, consensus was reached via collegial quality control.
Statistics
For each parameter (i.e., age at injection, capsid, ROA, time course, promoter, sex, vector purification method, and dose), we compared the pathology score between each pair of groups for each DRG or SC segment (i.e., cervical, thoracic, and lumbar) by using a Wilcoxon rank-sum test with function “wilcox.test” within the R Program (version 4.0.0;
Results
DRG pathology assessment
We developed a method to accurately evaluate and score lesions to DRG neurons based on neuroanatomy and systematic evaluation of neurons and their corresponding axons. Neuronal cell bodies of primary sensory neurons located in DRG appear as ovoid swellings at the base of each spinal dorsal root. DRG are within the subarachnoid space and are bathed in CSF. They contain fenestrated capillaries and, thus, lack a blood–brain barrier. 9,10 DRG neurons are pseudo-unipolar, with one peripheral branch extending into the peripheral nerve and one central branch ascending dorsally in the SC white matter tracts (Fig. 1).
It is our experience that neuronal degeneration does not affect DRG uniformly, meaning multiple DRG from cervical, thoracic, and lumbar regions need to be collected to provide a representative sample. Pathology in the DRG manifests as infiltration involving mononuclear inflammatory cells and proliferating resident satellite cells, with neuronal degeneration becoming visible at a later stage (Fig. 1, A1 circles). Secondary to neuronal cell body injury is axonal degeneration (i.e., axonopathy) along DRG axonal projections in the nerve root (Fig. 1, B1), ascending dorsal tracts of the SC (Fig. 1, C1), and peripheral nerves (Fig. 1, D1). Typical histopathological findings with the normal counterparts are pictured in Fig. 1 A2–D2 and H; we have also shown high-magnification images of varying stages of DRG pathology (Fig. 1E–G).
Early in the degenerative process, the neuronal cell bodies appear relatively normal with only proliferating satellite cells in addition to microglial cells and infiltrating mononuclear cells (neuronophagia; Fig. 1E/G, circles). As the lesions progress, the neuronal cell bodies exhibit evidence of degeneration (Fig. 1F, horizontal arrow) characterized by small, irregular- or angular-shaped cells with fading or absent nuclei and cytoplasmic hypereosinophilia. End-stage neuronal cell-body degeneration involves their complete obliteration (Fig. 1G, star) by satellite cells, microglial cells, and mononuclear cells.
The severity of the histological findings in DRG and corresponding axons is graded based on the percentage of neurons or axons that are affected on an average high-power field (irrespective of the stage of neuronal degeneration): 0 as absence of lesion, 1 as minimal (<10%), 2 mild (10–25%), 3 moderate (25–50%), 4 marked (50–95%), and 5 severe (>95%). DRG represents a mosaic, with an abundance of neurons being normal and only a minority of neurons showing degeneration on a given section. We consider SC axonopathy to be a better indicator of DRG pathology as it represents the collation of axons coming from all the DRG. Throughout this article, we define DRG pathology as histopathological findings within the DRG cell bodies and SC or within the SC alone.
Studies and populations characteristics
We aggregated data from 33 studies, including 256 animals injected with AAV vectors or vehicle controls at the Gene Therapy Program at the University of Pennsylvania from 2013 to 2020. A summary of these studies is provided in Table 1.
Nonhuman primate population and study characteristics
Un-injected n = 2 (no pathology).
GLP, Good Laboratory Practice; ICM, intra-cisterna magna; LP, lumbar puncture.
Effect of study characteristics on severity of DRG pathology
We observed DRG pathology in 83% of NHP that received AAV intra-cisterna magna (ICM) or lumbar puncture (LP) (170/205 animals), 32% of NHP for the IV route (8/25 animals), 100% for the combination of ICM + IV (4/4 animals), and 0% for intramuscular (IM, 0/4 animals). Pathologists graded the DRG lesions based on the severity score in DRG and their corresponding axons in the SC and peripheral nerves. We obtained scores for each DRG and spinal region (i.e., cervical, thoracic, and lumbar). Average scores are depicted in Figs. 2–4; the data are split by region of the SC (i.e., cervical, thoracic, and lumbar) in Supplementary Figs. S1–S4. Severity in DRG was lower than in SC because each SC region groups the totality of axons coming from DRG, thus collating pathology scores from several DRG (Fig. 1).
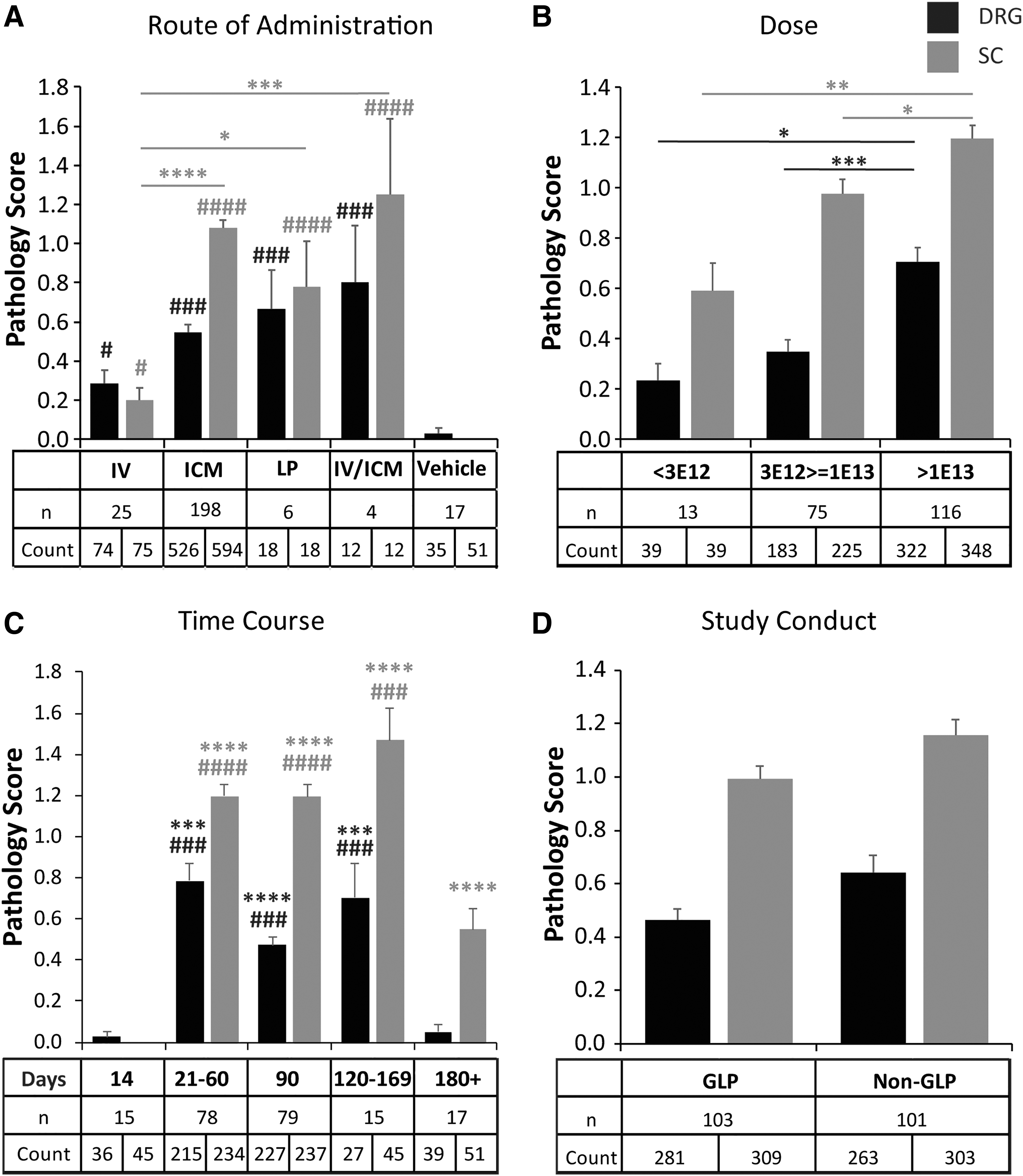
Effect of study characteristics on severity of DRG pathology. Average pathology scores in DRG (black) and dorsal SC axons (gray) with different
The study design parameters that significantly impacted the severity of the pathology were the ROA, dose, and necropsy time point (Fig. 2A–C). The compliance with GLP practice for nonclinical laboratory studies (as set in title 21 of the Code of Federal Regulations, part 58) did not impact the severity of the pathology (Fig. 2D). All the ROAs except IM led to significant pathology in both DRG and SC when compared with vehicle controls (p = 0.04 DRG and SC, IV versus vehicle; p < 0.001 DRG and p < 0.0001 SC, all other routes vs. vehicle). ICM, LP, and ICM/IV were all similar and significantly worse than IV in SC (Fig. 2A: IV vs. ICM, p < 0.0001; IV vs. LP, p = 0.02; IV vs. ICM/IV, p = 0.0006). IM (not shown) did not lead to pathology (all score 0) and was similar to vehicle controls. For all the following analyses, we only considered animals with intra-CSF administration (i.e., ICM or LP).
The two lower dose ranges (<3E+12 GC and 3E+12–1E+13 GC) were similar, whereas the maximal dose range (>1E+13 GC) led to significantly worse pathology scores than both the lowest (p = 0.04 in DRG; p = 0.001 in SC) and the middle dose range (Fig. 2B: p = 0.0009 in DRG, p = 0.04 in SC). The post-injection time point (i.e., when the necropsy was performed after vector administration and tissues were analyzed) showed similar pathology severity between 21–60, 90, and 120–169 days. Pathology was rarely seen and minimal at the early (i.e., day 14) time point, and the longer follow-ups greater than or equal to 180 days showed a significant reduction of severity compared with all other time points (Fig. 2C: p < 0.001 in DRG; p < 0.001 to p < 0.0001 in SC compared with ≥180 days).
Effect of animal characteristics on severity of DRG pathology
Age at vector administration had a significant effect on pathology severity when measured at later time points. Juvenile animals had less severe DRG degeneration when compared with adults (p = 0.003) but showed similar SC axonopathy (Fig. 3A). The four animals treated as infants showed no signs of DRG or SC pathology as previously reported. 11 This result needs to be interpreted with caution due to the small sample size and a possible effect of the study endpoint (4 years post-injection was much later than any time point involving juvenile or adult animals). In addition, and importantly, sex had no effect on SC or DRG pathology (Fig. 3B).
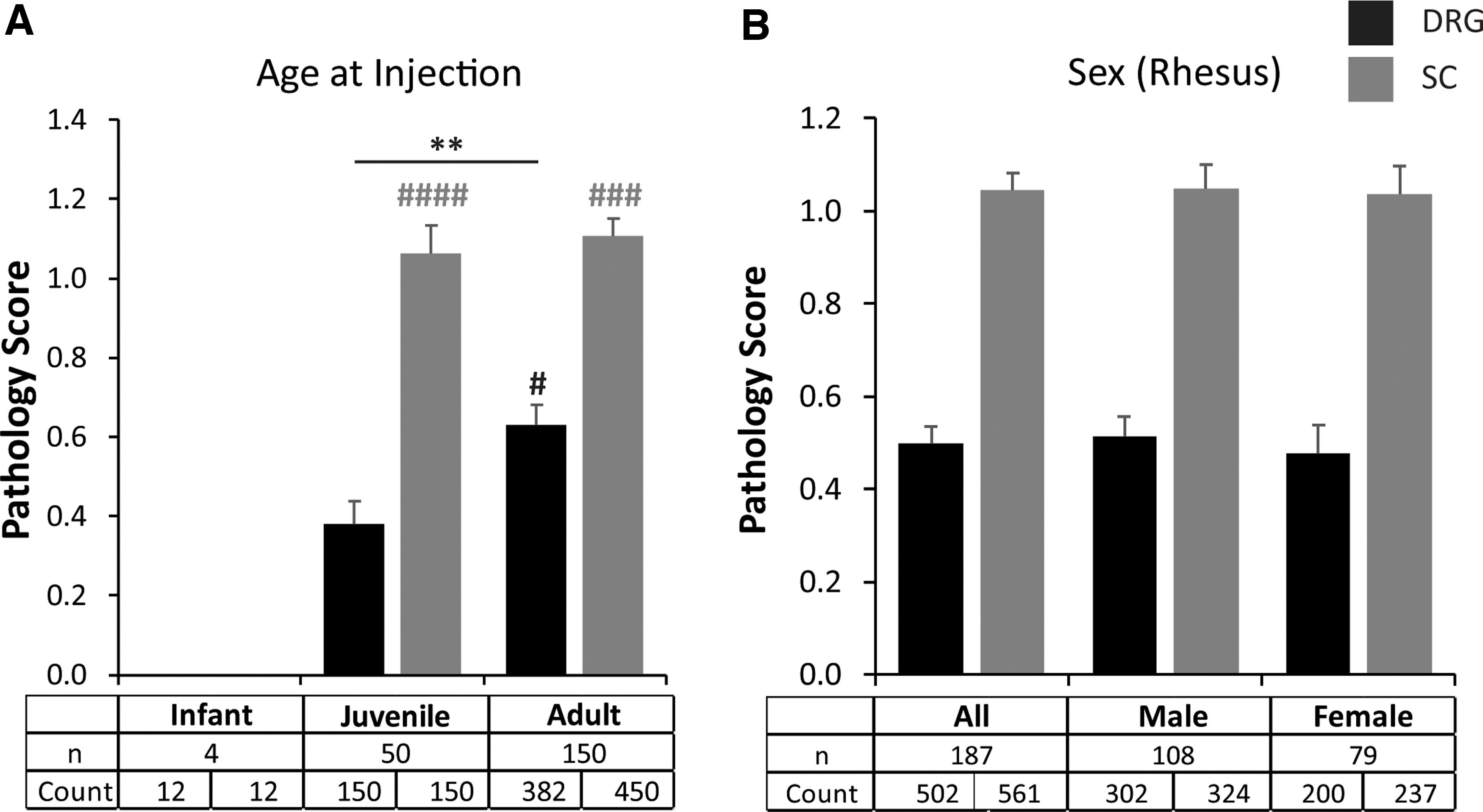
Effect of animal characteristics on severity of DRG pathology. Average pathology scores in DRG (black) and dorsal spinal cord axons (gray) with different
Effect of vector characteristics on severity of DRG pathology
DRG neurodegeneration was present with all capsids, although there were some differences in severity among serotypes (Fig. 4A). When such variations are restricted to DRG and are absent in SC scores, they may not be meaningful since the DRG represents a mosaic more susceptible to sampling artifact than the SC cross-sections. Both axonopathy and DRG scores (Fig. 4A) were significantly worse with AAV1 than AAVhu68 (p = 0.01 SC; p = 0.0004 DRG) and with AAV1 than AAV9 (p = 0.007 SC and DRG). These quantitative differences in severity may not necessarily represent capsid-specific properties because the collage of data from individual studies utilizing only one capsid and different transgenes made direct comparisons difficult.
Promoters CB7, UbC, and hSyn were all similar to each other. SC axonopathy was worse for promoter CAG than hSyn (p = 0.017), and for MeP426 compared with CB7 (p = 0.001), UbC (p = 0.002), and hSyn (p = 0.0003; Fig. 4B). We tested 20 different transgenes and all but one caused DRG pathology (Fig. 4C). Pathology severity varied greatly between transgenes (from 0 to 2.7 average SC axonopathy score); pathology severity was 20–25% less for non-secreted transgenes compared with secreted transgenes (Fig. 4D; for DRG, secreted mean = 0.61, non-secreted mean = 0.47, p = 0.05; for SC, secreted mean = 1.17, non-secreted mean = 0.94, p = 0.02). Moreover, the purification method (i.e., iodixanol in non-GLP studies and column chromatography in GLP studies) did not impact the presence or severity of DRG pathology (Fig. 2D).
Regional severity, and clinical manifestations of DRG pathology
We assessed regional differences in pathology with respect to the cervical, thoracic, and lumbar spine. We also analyzed the trigeminal nerve ganglion (TRG), as it represents a sensory ganglion with similar characteristics to the DRG located at the base of the skull inside the subarachnoid space. Figure 5 shows a distribution of the actual pathology scores in each region as well as the average. TRG pathology was similar to thoracic DRG (nonsignificant), whereas higher scores were reported in cervical DRG (p = 0.005) and lumbar DRG (p = 0.04). SC regional scores were all significantly worse than their corresponding DRG scores (p < 0.0001), which is consistent with SC collating axons from several DRG, meaning it comprises more lesions. The vast majority of sections had normal or low (grade 1) severity scores, with a few grade 4 and almost no grade 5 scores reported (grade 5 corresponds to 95% or more of the tissue surface being affected by the lesion on an average high-power field; Fig. 5).
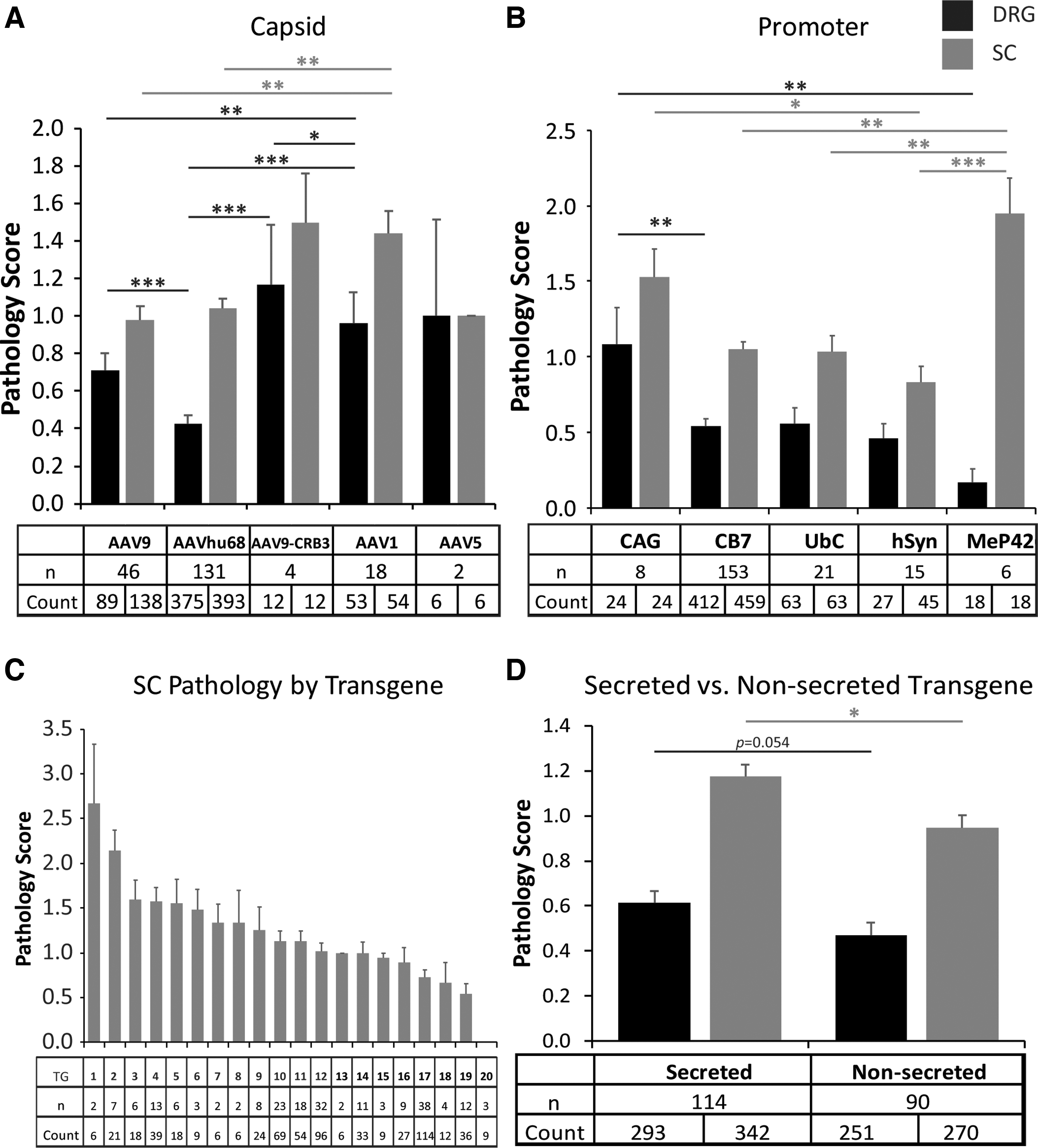
Effect of vector characteristics on severity of DRG pathology. Average pathology scores in DRG (black) and dorsal spinal cord axons (gray) with different
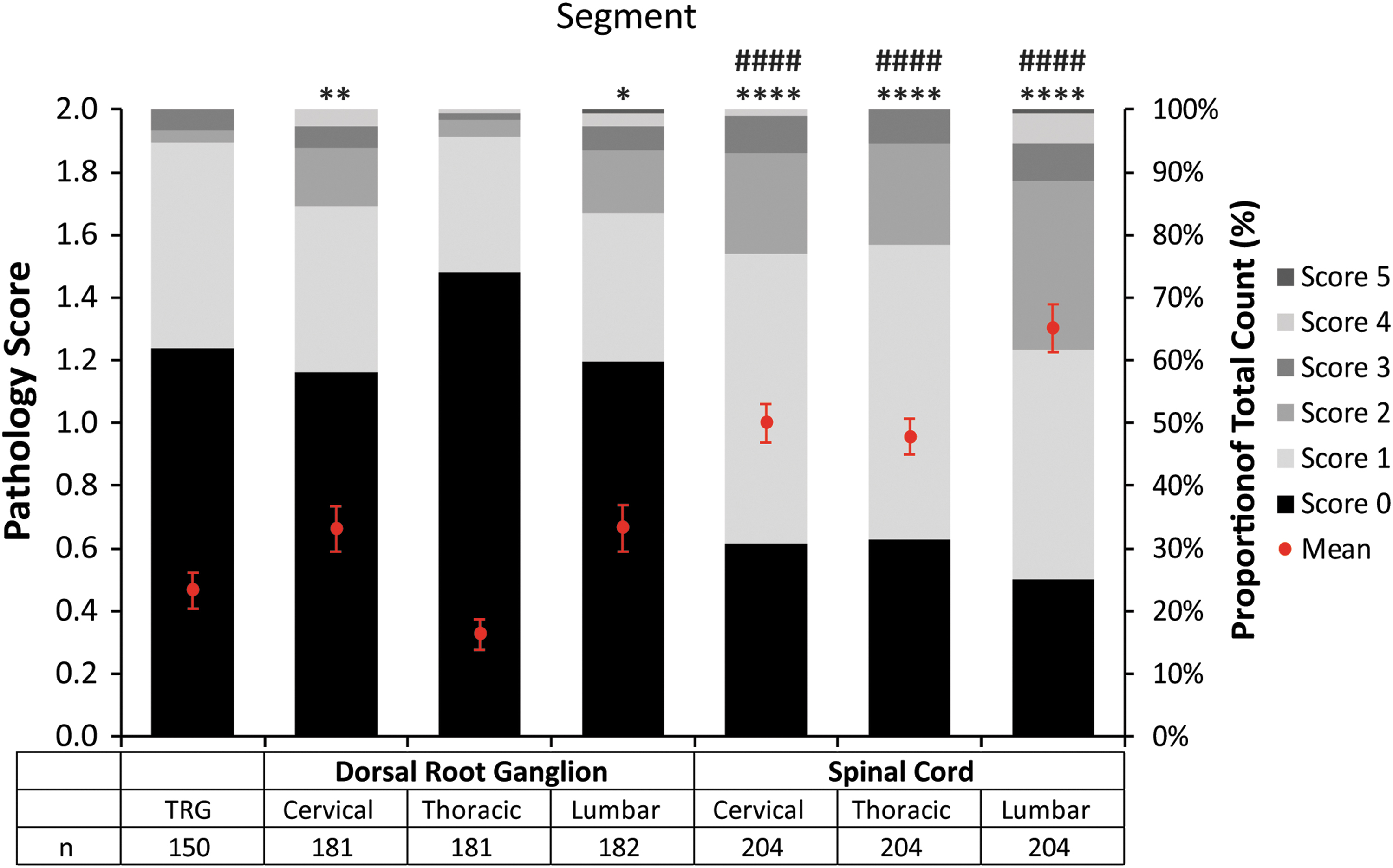
Regional pathology scores with distribution of severity grades. Mean percentage proportion of pathology scores with standard error of mean (red dots and bars) and distribution of severity grades by region (stacked columns). Tables indicate number of animals (n) in each group. We compared means by using Wilcoxon signed-rank test between TRG and DRG and between DRG and spinal cord respective regions (i.e., cervical, thoracic, and lumbar). Statistical significance was assessed at the 0.05 level. *Indicates significance for TRG to DRG/SC comparisons; #indicates significance for DRG to SC regional comparisons. **p < 0.01; ****,#### p < 0.0001. TRG, trigeminal nerve ganglion.
We performed a neurological examination on most animals involving cage-side evaluation of mentation, posture, proprioception, and gait, as well as a restrained evaluation of cranial nerves, motor strength, sensory function, and reflexes. Of the 204 animals administered AAV ICM or LP, only three developed clinical signs of ataxia and/or tremors. All three animals received vectors encoding GFP at doses >1E+13 GC, and the pathology appeared 21 days post-injection. Nerve-conduction velocities of the median nerve were recorded in 56 animals. Two animals developed a marked bilateral sensory amplitude reduction at 28 days post-injection that persisted until necropsy. This correlated with marked (grade 4 severity) axonopathy and periaxonal fibrosis in the median nerve but no obvious clinical sequelae (data not shown). Most animals had low severity grades of axonopathy and fibrosis in peripheral nerves (Fig. 6).
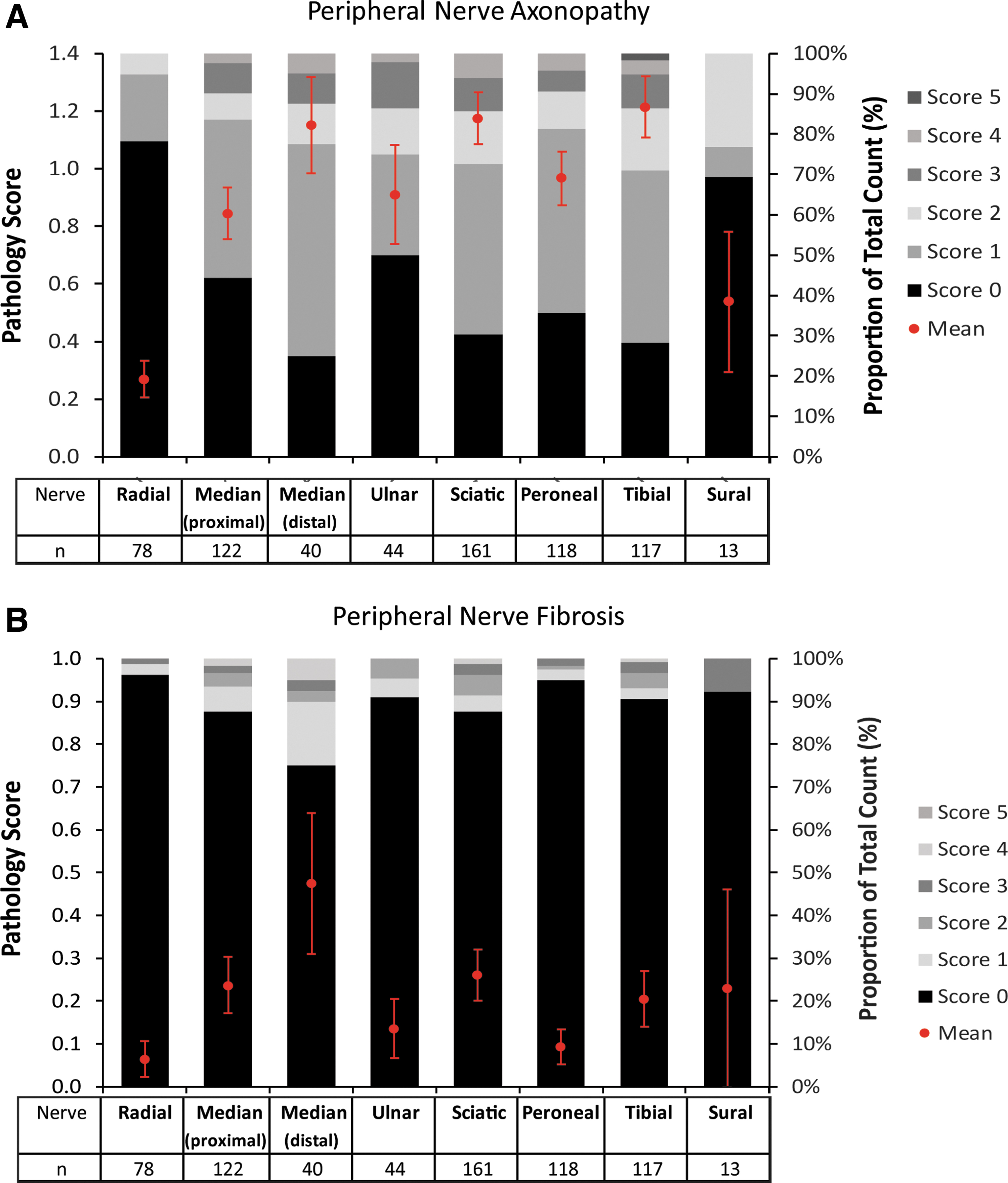
Peripheral nerve pathology. Mean percentage proportion of pathology scores with standard error of mean (red dots and bars) and distribution of severity grades by peripheral nerve (stacked columns). Tables indicate the number of animals (n) in each group. We did not perform statistical analysis, because the list of peripheral nerves collected varied greatly between studies.
Discussion
DRG pathology and secondary axonopathy is minimal in the vast majority of our NHP studies and can be challenging to detect with an untrained eye. In our first GLP toxicology study evaluating an ICM AAV administration, 1 the contract research organization (CRO) who performed the initial pathology assessment missed the lesion, which was only caught by a peer-review pathologist experienced in neuropathology. Because neuronal degeneration is sparse and DRG are a mosaic of mostly normal neurons with a few degenerative events on a given section, we found that multiple DRG needed to be collected for a robust histological analysis (we recommend at least three DRG per spinal region). An easier way to detect and quantify DRG neuronal damage involves evaluating the secondary consequences of pathology in the cell body by assessing axon degeneration in the SC; this technique facilitates detection and represents a collation of ascending fibers coming from multiple DRG.
When collecting and carefully analyzing the relevant tissues, we found some evidence of DRG pathology in 83% of NHP that received AAV ICM or LP and 32% of NHP that received AAV IV. The IV doses showing pathology were as low as 1E+13 GC/kg, a dose currently evaluated in the clinics for several hemophilia trials. 12 The manufacturing purification method had no impact on pathology. All of the capsids and promoters that we tested demonstrated some level of DRG pathology, which suggests that changing capsid or promoter is not a viable solution. Of relevance to nonclinical study design and clinical translation, we found that dose and age at injection significantly affected the severity whereas sex had no impact. The aspect of our studies with the greatest impact on severity of pathology was the transgene, which is consistent with our hypothesis that transgene overexpression drives the early events that lead to degeneration. For most transgenes, it was not possible to identify a no-observable adverse effect level above the minimum efficacy dose.
Time course is important to consider for study design, as acute time points (i.e., day 14 or below) do not consistently show histopathology whereas longer studies (i.e., >180 days) tend to demonstrate less severe pathology, which suggests a lack of progression and possible partial remission over time. Our experience with health authorities has involved incorporating two necropsy time points—one after the onset of pathology (i.e., around 1 month) and another to show the pathology is not getting worse (i.e., 4–6 months). The four NHP infants that were dosed at 1 month of age included in our meta-analysis were remarkable given the absence of DRG and SC axonopathy. This result occurred despite good transgene expression levels when the animals were necropsied almost 4 years post-injection. 11 This observation may suggest a more favorable safety profile when dosing infants or that acute pathology does not progress and in fact resolves; we did not have any early time-point necropsies in this study.
None of the animals that received a therapeutic transgene (i.e., not a reporter gene such as GFP) demonstrated any clinical findings. In later studies, we incorporated routine monitoring of sensory neuron pathology through the use of nerve conduction velocity measurements. We found NCV abnormalities in two animals associated with more severe peripheral nerves axonopathy and fibrosis (i.e., grade 4 severity) without evidence of clinical sequelae.
In summary, we show that pathology in DRG is a consistent finding in virtually all NHP studies when the AAV vector is delivered into the subarachnoid space and—in many studies—when higher doses are administered systemically. Our meta-analysis is remarkable for a notable absence of any clinical sequelae while using therapeutic transgenes. Careful analysis of other nonclinical studies in other species failed to show any evidence of DRG pathology, except in newborn pigs, suggesting that NHPs are the best model for evaluating this potential pathology. It seems prudent to monitor sensory neuropathies in human central nervous system and high-dose IV clinical studies to detect the presence of clinically meaningful DRG pathology if it occurs.
Footnotes
Acknowledgments
The authors thank the Pathology Core; Program for Comparative Medicine; and Vector Core of the Gene Therapy Program at the University of Pennsylvania for study support. All vectors were produced by the Penn Vector Core.
Authors' Contribution
J.H. and E.L.B.: investigation, supervision, and writing—original draft preparation; C.D., T.G., T.W.M., L.R., C.H., N.K., R.S., R.M., G.R.C., M.H., and K.N.: investigation; N.D.: data curation and visualization; H.Y. and M.L.: formal analysis; J.M.W.: conceptualization, funding acquisition, and writing—review and editing.
Author Disclosure
J.M.W. is a paid advisor to and holds equity in Scout Bio and Passage Bio; he holds equity in Surmount Bio; he also has a sponsored research agreement with Ultragenyx, Biogen, Janssen, Precision Biosciences, Moderna, Inc., Scout Bio, Passage Bio, Amicus Therapeutics, and Surmount Bio, which are licensees of Penn technology. J.M.W., J.H., C.H., and N.K. are inventors on patents that have been licensed to various biopharmaceutical companies and for which they may receive payments.
Funding Information
Individual studies utilized for data extraction were supported by REGENXBIO (all studies previously published 1,11 ), Biogen (some studies previously published 2 ), Passage Bio, Amicus Therapeutics, ODC MPS I pilot grant MPS-18-D010–01 and MPS-19-001-0, Janssen, Cure FA, Rett Syndrome Research Trust, and Elaaj Bio.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
