Abstract
Neointima formation and vascular remodeling through vascular smooth muscle cell migration and proliferation can limit the long-term success of coronary interventions, for example, in coronary artery bypass grafting (CABG). Ex vivo gene therapy has the potential to reduce unnecessary cell proliferation and limit neointima formation in vascular pathologies. To date, the species C adenovirus serotype 5 has been commonly used for preclinical gene therapy; however, its suitability is potentially limited by relatively poor tropism for vascular cells and high levels of preexisting immunity in the population. To avoid these limitations, novel species of adenovirus are being tested; here we investigate the potential of adenovirus 49 (Ad49) for use in gene therapy. Transduction of primary human vascular cells by a range of adenovirus serotypes was assessed; Ad49 demonstrated highest transduction of both vascular smooth muscle and endothelial cells. Gene transfer with Ad49 in vascular smooth muscle and endothelial cells was possible following short exposure times (<1 hr) and with low MOI, which is clinically relevant. Ex vivo delivery to surplus CABG tissue showed efficient gene transfer with Ad49, consistent with the in vitro findings. Luminal infusion of Ad49GFP into intact CABG samples ex vivo resulted in efficient vessel transduction. In addition, no seroprevalence rates to Ad49 were observed in a Scottish cohort of patients from cardiovascular clinics, thus circumventing issues with preexisting immunity. Our results show that Ad49 has tropism for vascular cells in vitro and ex vivo and demonstrate that Ad49 may be an improved vector for local vascular gene therapy compared with current alternatives.
Introduction
C
Reduction of neointima formation by gene delivery to the vasculature is an attractive proposition to improve long-term patency of CABG procedures and reduce the need for repeat surgery. Unlike cardiomyocytes, only low levels of transduction are possible in VSMCs or endothelial cells (ECs) using AAVs. 5,6 However, preclinical studies using adenovirus vectors have shown that gene delivery to the vasculature can prevent neointima formation. 7,8 Expression of therapeutic proteins, including endothelial nitric oxide synthase (eNOS), 9 inducible nitric oxide synthase (iNOS), 10 tissue inhibitors of metalloproteinases 1–3 (TIMPs 1–3), 7,11,12 p53, 13 and Nogo-B, 14 has been shown to enhance EC regeneration and/or reduce VSMC proliferation and migration, in turn reducing neointima formation, in animal models of CABG. In fact, CABG surgery may be particularly suited to gene therapy as the clinical setting facilitates ex vivo gene transfer to the vein before implantation. To date, the majority of gene therapy strategies have used adenovirus serotype 5 (Ad5) as the gene delivery vector. However, Ad5 transduces vascular cells relatively poorly and high multiplicities of infection are required to achieve therapeutic levels of transgene expression. 15,16
Previous analysis of receptor expression on VSMCs and ECs has shown only low levels of coxsackie and adenovirus receptor (CAR), 17 the primary Ad5 receptor, and no detectable expression in intact human vessels. 18 Additionally, in CABG, the time available for ex vivo gene transfer before grafting is short, potentially less than 30 min, further limiting the gene delivery possible with Ad5. An added clinical consideration of Ad5 use is the common prevalence of preexisting neutralizing antibodies in the population, 19 up to 80% seroprevalence in some studies, 20 which may reduce the efficacy of transgene expression, 21 although the effect of preexisting neutralizing antibodies on adenovirus transduction in the CABG setting is unknown as gene transfer occurs ex vivo. Together, these limitations of Ad5 suggest that, while the vector may be suitable for human CABG gene therapy, alternative vectors might provide improved opportunities.
To increase transgene expression in the vasculature, pseudotyping of Ad5 has been shown to be effective. 22 Several subgroup B adenoviruses, including Ad35, use CD46 as a receptor, 23 which is relatively highly expressed on VSMCs, and chimeric Ad5/Ad35 viruses show more efficient gene transfer to the vasculature than Ad5. 17 An alternative strategy is exploitation of the natural tropism of novel Ad serotypes. Subgroup D adenoviruses vary in receptor use and tropism; some infect cells by binding CAR, while others use CD46 24 or sialic acid. 25 However, the receptors for many subgroup D adenoviruses have not been identified, and therefore they may provide new vectors for gene therapy. An additional clinical benefit is that species D adenoviruses are reported to have low seroprevalence rates. 24 Here we evaluated a panel of adenoviruses and highlight the efficacy of Ad49 as a vector for vascular gene therapy.
Materials and Methods
Cells and tissue
PER.C6 cells 26 (kind gift from Jerome Custers, Crucell) and HEK293 cells (human embryonic kidney: ATCC CRL-1573) were used for viral propagation and cultured in Dulbecco's modified Eagle's medium (DMEM; Invitrogen) with 2 mM L-glutamine (Invitrogen) and 10% fetal calf serum (FCS; PAA Laboratories) with the addition of 10 mM MgCl2 (Sigma-Aldrich) for PER.C6 cells. Hep G2 (hepatocellular carcinoma: ATCC HB-8065), A549 (human lung carcinoma: ATCC CCL-185), and SKOV3 (human ovarian carcinoma: ATCC HTB-77) cells were cultured in minimal essential medium (MEM) or RPMI-1640 medium (Invitrogen), with 2 mM L-glutamine, 10% FCS, and 1 mM sodium pyruvate (Sigma-Aldrich). Cells were maintained at 37°C and 5% CO2 with the exception of PER.C6 maintained in 10% CO2. Human saphenous vein segments were obtained from patients, who gave informed consent, undergoing CABG. Ethics permission was obtained from the West of Scotland Research Ethics Committee 4 (reference number 10/S0704/60). Serum samples were obtained, with consent, from patients attending cardiovascular disease clinics with ethics permission from the West Glasgow Ethics Committee 1 (reference number S/0703/110).
Smooth muscle and EC isolation and culturing
VSMCs were isolated from human saphenous vein segments using the explant technique as previously described 27 and cultured in SMC growth media 2 (PromoCell) with addition of provided supplement mix, 10% FCS, and 2 mM L-glutamine. ECs were isolated from human saphenous vein segments by collagenase digestion as previously described and cultured in Large Vessel Endothelial Cell Basal Medium (TCS CellWorks) with provided supplements and 20% FCS. Primary cell lines were used between passage 3 and 6.
Recombinant adenoviral vectors
All recombinant adenoviruses described in this article were kind gifts from Jerome Custers (Crucell); Ad5, Ad26, Ad35, Ad48, and Ad49 are replication-incompetent E1/E3 deleted vectors constructed as described previously. 28,29 Viruses were propagated in HEK293 or PER.C6, E1-complementing cell lines and purified using CsCl gradients. Viral recovery was quantified by microBCA assay (ThermoFisher) assuming that 1 μg protein=4×109 viral particles (vp) and confirmed by nanosight measurement (Nanosight). Infectious units (pfu) were quantified by end-point dilution plaque assay. 30
Transduction assays and neutralization assays
Cells were plated in 96-well format, 1×104 cells/well, and incubated overnight. Cells were infected with 1000, 2500, 5000, or 10,000 vp/cell in 50μl serum-free media, ±hFX (8 μg/ml) where stated. Cells were incubated with virus for 3 hr at 37°C, unless otherwise stated; washed with PBS; and maintained in standard medium until harvesting with reporter lysis buffer (RLB; Promega) 48 hr postinfection. Luciferase activity was measured in cell lysates using a luciferase assay (Promega) and protein concentrations were calculated by BCA (ThermoFisher) assays were measured using a Wallac Victor2 plate reader (PerkinElmer) and values expressed as relative light units (RLU)/mg of protein. For serum neutralization studies, transduction of Hep G2 cells was carried out as above in the presence of 2.5% serum as previously been shown to be optimum. 31 Samples were considered neutralizing if greater than 90% reduction in transduction was seen in comparison to no serum control. To determine GFP expression, cells were detached from the culture vessel with 1xTE, washed, and quantified using a flow-assisted cell sorter (FACS) Canto II flow cytometer (Becton Dickinson) and FACS DIVA software. Viable cells were gated by their FSC/SSC profiles, with a minimum of 10,000 gated events analyzed per sample. Results are expressed as percentage of positively stained cells per sample, from three independent samples analyzed in triplicate.
Ex vivo transduction of CABG tissue
Human saphenous vein tissue was trimmed to remove excess periadventitial fat and divided into equal (∼5 mm) sections before incubation with adenovirus: 1×109 vp or PBS in 500 μl serum-free media for 1 hr at 37°C. Tissue sections were washed and cultured for a further 48 hr in SMC media. Luciferase activity was analyzed using bioluminescence quantification imaging (IVIS Spectrum; Caliper Life Sciences) quantified as average radiance (photons per second per centimeter squared per steradian). Human saphenous veins were transduced by luminal delivery of adenoviruses as previously described. 11 Four- to six-centimeter segments of vein were cannulated, and the luminal surface was exposed to adenoviral vectors at 1×1010 pfu/ml at physiological pressure for 1 hr. Segments of vein were cultured for 3 days in DMEM containing 30% FCS and antibiotics.
Immunohistochemistry
Immunohistochemistry was performed on 4 μm paraformaldehyde fixed sections. Following rehydration, heat-induced antigen retrieval was performed with 10 mM sodium citrate buffer (pH 6) and sections were blocked in 15% goat serum before incubation with primary antibodies: chicken anti-gfp (Abcam), mouse anti-smooth muscle actin (Dako), and rabbit anti-von Willebrand factor (Dako). Fluorescent secondary antibodies were used for detection, goat anti-chicken 488, anti-rabbit 546, and anti-mouse 647 (Invitrogen), and slides were mounted with ProLong Gold Antifade Reagent with DAPI (Invitrogen). Images were acquired using a Zeiss LSM510 confocal imaging system (Carl Zeiss).
Statistics
In vitro data presented are representative from a minimum of three separate experiments with at least three experimental replicates per group unless otherwise stated. Transduction graphs are displayed with a log scale as appropriate, and for these experiments data were log transformed before analysis. Statistical significance was calculated using ANOVA or Student's t-test; p<0.05 was considered statistically significant.
Results
Ad49 tropism for human vascular cells in vitro
To initially evaluate vector tropism for vascular cells, the transduction efficiency of four serotypes of adenovirus was evaluated in primary VSMCs and ECs, in comparison to the commonly used vector Ad5. Previous reports have demonstrated relatively high transduction of vascular cells by Ad35, 17 therefore acting as a positive control for efficient transduction in this study. The other three Ad serotypes chosen belonged to subgroup D and have not been investigated in the terms of vascular cell transduction. Additionally, low seroprevalence to these Ad serotypes has been found 24,32 in serum samples from sub-Saharan Africa.
First, transduction efficiency was assessed using vectors expressing luciferase. In comparison to Ad5, VSMCs transduced with Ad48 and Ad49 had significantly higher luciferase activity (Fig. 1A). Similarly, Ad49 also had the highest luciferase activity following infection of ECs (Fig. 1B). To enable visualization of transgene expression in vascular cells, an Ad49 vector expressing GFP was generated. VSMCs and ECs were transduced with a range of viral titers and fluorescence assessed microscopically, and representative images of VSMCs are shown in Fig. 1C. GFP expression was further analyzed using FACS, and VSMCs (Fig. 1D) and ECs (Fig. 1E) transduced with Ad49 had higher GFP expression compared with Ad5. Collectively, these data demonstrate that Ad49 can efficiently transduce primary human vascular cells.
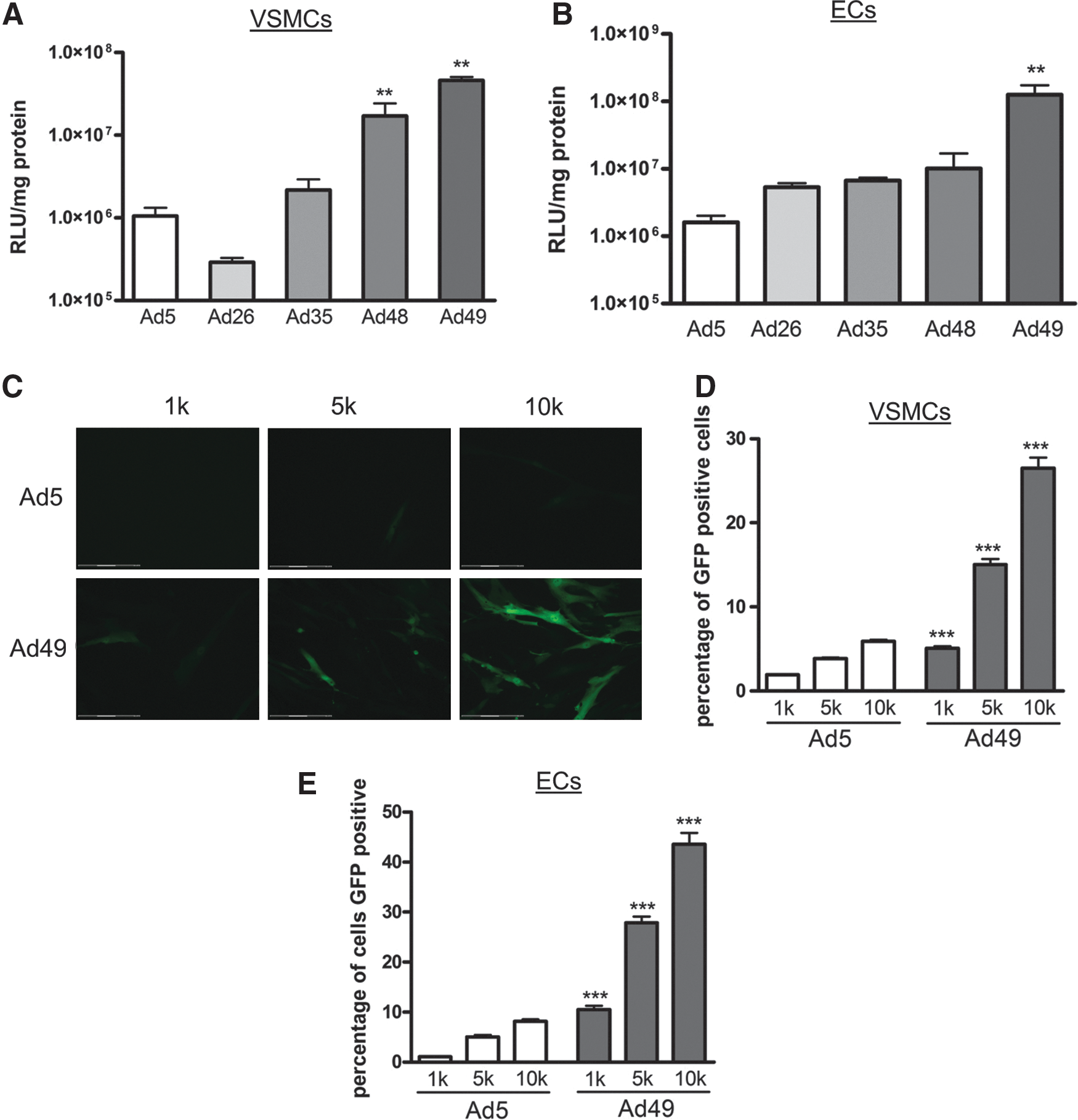
Transduction of primary vascular cells. VSMCs
To investigate more general selectivity, Ad49 gene transfer was assessed in other cell lines with different receptor profiles (Supplementary Fig. S1; Supplementary Data are available online at
Transduction of VSMC and ECs in short time periods
In the context of CABG surgery, the time available for gene transfer is limited, and therefore rapid and efficient transduction is important. To investigate the potential for Ad49 to transduce cells following minimal, clinically relevant contact times, exposure of Ad5 or Ad49 to VSMCs and ECs was reduced to between 10 and 60 min. In comparison to Ad5, higher luciferase activity was detected in VSMCs (Fig. 2A) and ECs (Fig. 2B) transduced with Ad49 at all time points tested, down to just 10 min exposure. Furthermore, efficient gene transfer was possible when both low titers of 1,000 vp/cell and high titers of 10,000 vp/cell were used for short incubation times (Fig. 2A and B).
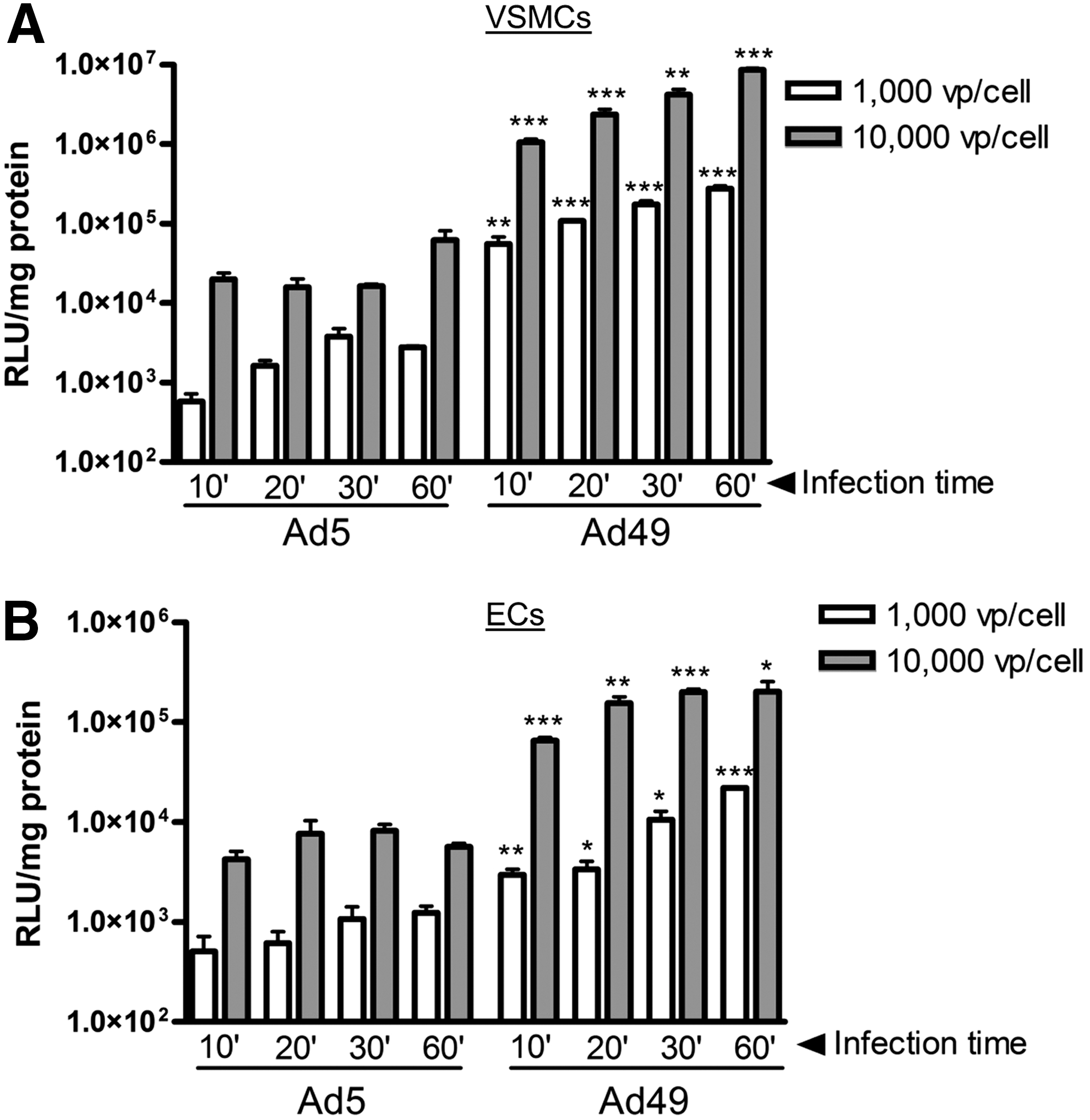
Transduction of cells with short exposure times. VSMCs
Ex vivo gene transfer to intact saphenous vein
The above data demonstrate Ad49 as an efficient vector for gene transfer to vascular cells in vitro. To further investigate the potential for clinical use, transduction of surplus CABG tissue was undertaken. First, whole vessels were incubated with Ad vectors and luciferase activity was quantified using an IVIS imaging system. Consistent with the in vitro data, luciferase activity was detected in tissue exposed to Ad49 at significantly increased levels to control (Fig. 3A). Follow-up studies exposing only the luminal surface of the tissue to Ad49GFP were performed in CABG samples from three patients. Cells positive for GFP co-localized with those stained for von Willebrand factor, a marker of ECs (Fig. 3B). Sections were stained throughout the length of vein exposed to Ad49GFP, and positive GFP staining was detected in ECs throughout the vessel.
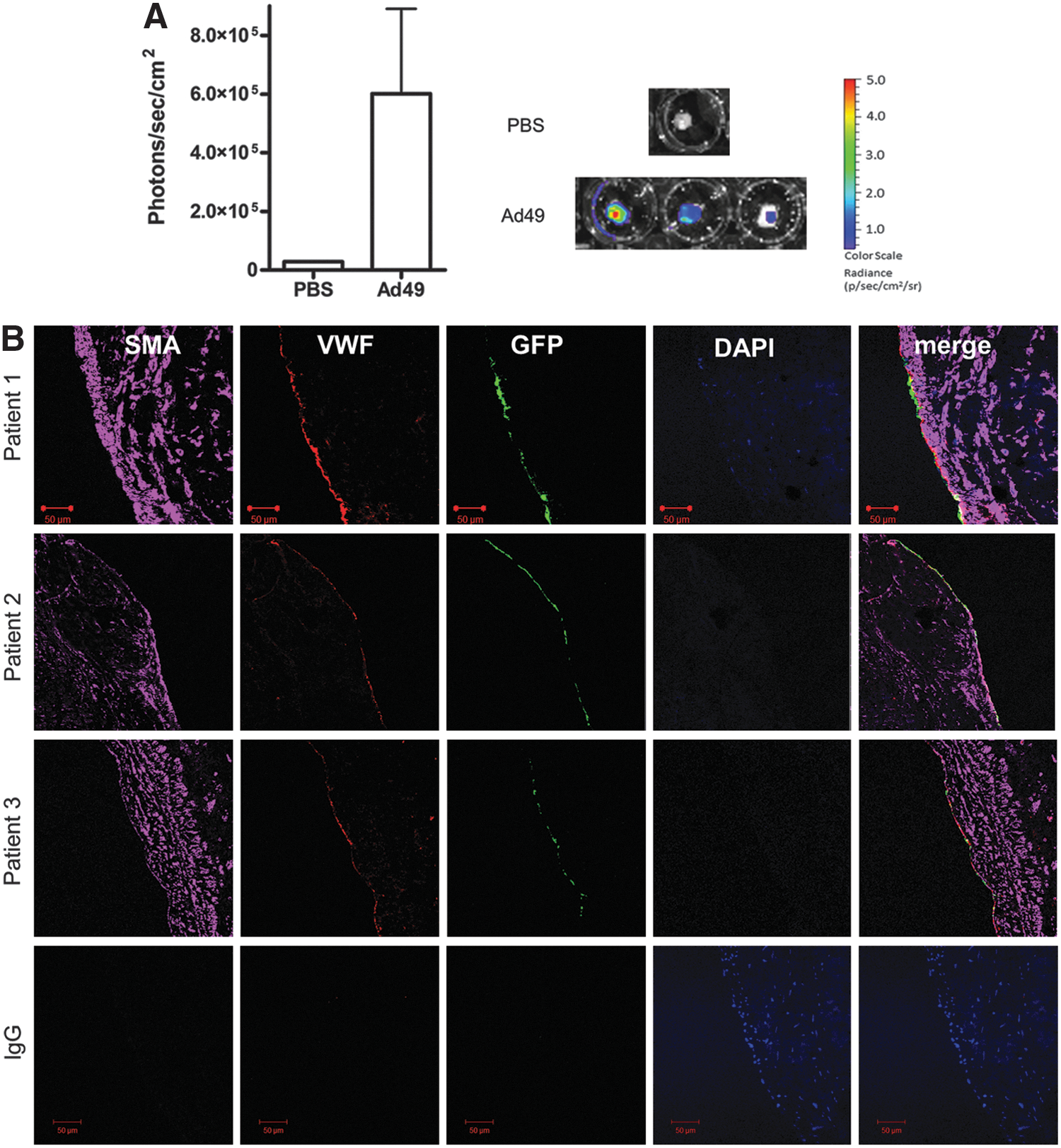
Ex vivo transduction of human coronary artery bypass graft tissue. Surplus coronary artery bypass grafting (CABG) tissue was cut into equal sections and incubated with 1×109 vp Ad49LUC or PBS for 1 hr at 37°C. Media was replaced with 20% and samples were cultured for a further 48 hr. Luciferase activity was analyzed using the IVIS system and quantified as average radiance
Preexisting immunity to Ad49
The use of adenovirus vectors in a clinical setting may be limited by preexisting immunity, 33 depending on the route of administration and target tissue, although this may not be problematic where ex vivo gene transfer is being utilized. To determine the seroprevalence of Ad49 in a clinically relevant population, serum samples were collected from 103 patients in Glasgow cardiovascular clinics. Presence of neutralizing antibodies was determined by preincubating Ad5 or Ad49 with 2.5% serum, recognized as an optimal dilution, 24,31 before transduction of HepG2 cells (Fig. 4A). Samples were considered neutralizing if transduction was reduced by 90% compared with virus alone, previously shown to be ideal for studying neutralization patterns of Ads. 22,34 Ad5 was neutralized by 31% (32/103) of serum samples, while none of the 103 serum samples reduced Ad49 cell transduction by 90% (Fig. 4B). Further analysis found that 1 serum sample could reduce Ad49 transduction by 50%, while an additional 16 samples could reduce Ad5 transduction to between 10% and 50% of control.
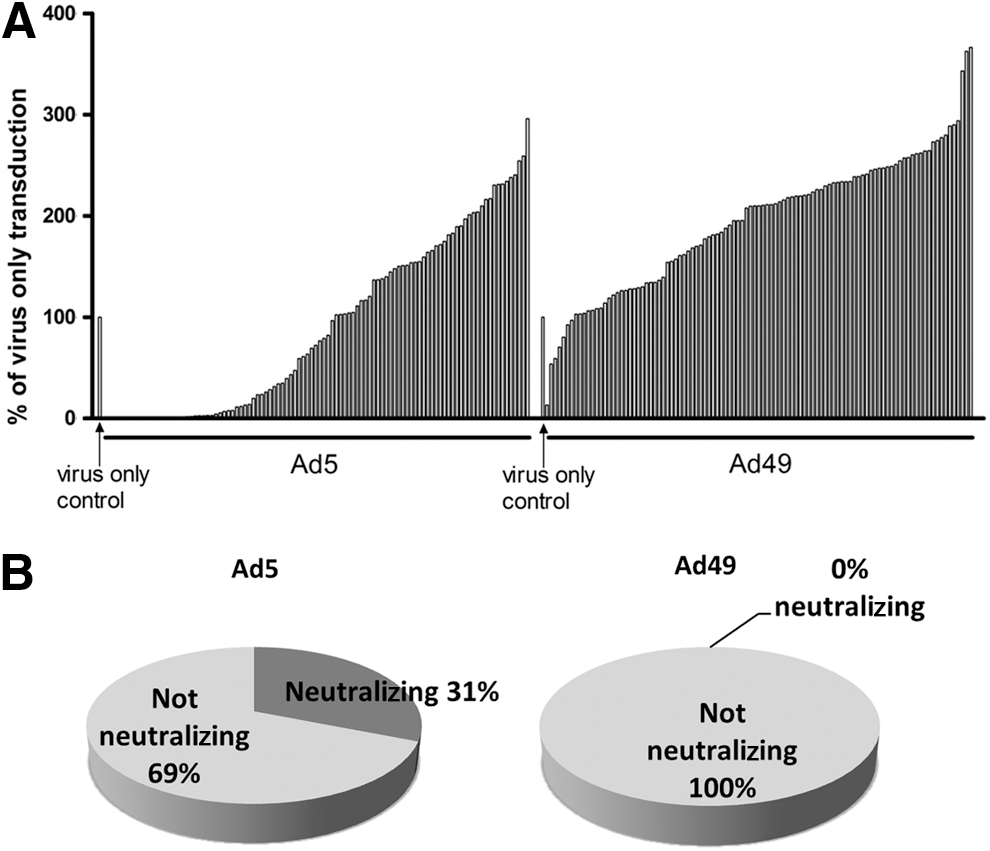
Preexisting immunity to Ad49 in patients undergoing CABG. Transduction assays were carried out in the presence of serum samples from 103 patients from Glasgow Cardiovascular Clinics, and each bar represents 1 patient
Discussion
The aim of this study was to identify an Ad serotype suitable for use as a vascular gene therapy vector: assessing efficacy in vitro and ex vivo and determining any seroprevalence in the population that may limit clinical use. For the first time we report the natural tropism of Ad49, a species D adenovirus, for vascular cells demonstrating efficient gene transfer in primary vascular cells and human CABG tissues.
Previous vascular gene therapy studies have used high concentrations of Ad5 for gene transfer. Ad5 transduces cells through engagement with CAR and further internalization using integrins. 35,36 The limited expression of CAR on vascular cells presumably leads to the requirement for high concentrations of Ad5. The in vitro results presented here demonstrate that Ad49 is able to transduce both VSMCs and ECs, resulting in higher reporter gene activity than Ad5. We demonstrated that gene transfer with an alternative reporter gene, GFP, was more efficient with Ad49 than Ad5 over a range of viral concentrations. Ad35 and pseudotyping of Ad5 with Ad35 fiber have previously been proposed as efficient vascular gene therapy vectors because of high expression of their receptor CD46 on vascular cells. 17 Interestingly, these results suggest that Ad49 can transduce vascular cells with greater efficiency than Ad35 and that lower concentrations of vector than have been previously used may be sufficient in vivo.
Furthermore, we tested the efficacy of Ad49 following short exposure times on cells because of the limited clinical time available for gene transfer. In comparison to Ad5, luciferase activity was 100 times higher following Ad49 transduction of VSMC and ECs and still efficient with low multiplicity of infection and only 10 min incubation with cells. The transduction efficiency of Ad49 in the cell lines, A549, SKOV3, and Hep G2, was similar or less than that of Ad5. This further supports the superior efficiency of the vector in vascular cells compared with Ad5, which is not evident in all cell types. It will be important to determine the primary receptor used by Ad49. The in vitro data led us to speculate that Ad49 could be a useful vector for vascular gene transfer, using lower concentrations and incubation times than is optimal with Ad5.
To confirm the clinical relevance of the Ad49 vector, further gene transfer experiments were carried out ex vivo in CABG tissue. While primary cells are a good tool for modeling in vitro, their phenotype can alter in culture in isolation from normal neighboring cell types. 37 First, whole sections of CABG tissue were incubated with Ad49, confirming that efficient vascular transduction of intact tissues was possible. To recreate the clinical situation and determine exactly which cell types were being transduced ex vivo, we delivered Ad49 to the lumen of saphenous vein. The luminal surface of the vessel will experience the dramatic changes in flow and pressure following grafting into the arterial circulation and as such these cells may be the optimal clinical target for preventing neointima formation. Efficient gene transfer of GFP was detected in samples from three different patients. Interestingly, transgene expression co-localized with positive staining for the EC marker von Willebrand factor. This is consistent with the transduction data in primary cultured ECs from similar samples; however, no gene transfer was detected in the medial smooth muscle cell layer. Previous reports have shown Ad5 gene transfer to VSMC. 12 The reason for the differences in cell specificity ex vivo is currently unclear but may be because of receptor expression distribution and levels and/or differences in surgical approaches to graft harvest and treatment. It has been previously suggested that Ad49 can bind to CD46, 28 which is highly expressed on ECs. 17 Additionally, over the last decade surgeons have adopted a no-touch technique for harvest of saphenous veins that has been shown to reduce SMC activation and damage to ECs. 38 The change in harvest method, maintaining a complete endothelial layer, could contribute to the distribution of infected cells.
Gene transfer to ECs is clinically relevant as, in addition to control of VSMC proliferation, ECs often get damaged during vascular procedures and re-establishing a healthy endothelium can reduce neointima formation. 39 –41 Despite the new surgical techniques designed to preserve the endothelium, CABG failure is still a clinical problem. Delivery of eNOS 42 and vascular endothelial growth factor (VEGF) 43 to ECs has been shown to reduce neointima formation in animal models. Furthermore, delivery of Ad49 vectors encoding secreted transgenes such as p53 13 or A20 44 could be used to reduce the VSMC proliferation associated with neointima formation. EC gene therapy is also beneficial in other disease settings: to promote revascularization of ischemic tissue and reduced angiogenesis in cancer. Efficient gene transfer has thus far been limited in angiogenic gene therapy (reviewed in ref. 45 ), Ad49 may also be a useful vector in this setting, and further research is required.
Clinical use of viral vectors can be limited by preexisting immunity. In the CUPID clinical trial, almost 50% of patients had to be excluded because of AAV-neutralizing antibody titers of >1/2. 46 Preexisting immunity to Ad5 has been demonstrated in the general population. 47 As such to be considered for clinical use, new vectors must have a more favorable neutralization profile. This does not diminish the use of Ad5 in a vein graft gene therapy setting, but these findings may impact gene transfer settings where contact to the blood may be relevant, such as intravascular administration. The sera from 31% of patients tested in this study could neutralize Ad5, while none were able to reduce transduction with Ad49. Here we specifically used serum from a potential target population for vascular gene therapy, patients of a cardiovascular clinic. One previous study has also reported low levels of preexisting immunity to Ad49 in sera taken from four continents. 28 Consistent with a previous study, 28 there was no cross-reactive Ad5/Ad49-specific immunity observed in the serum cohort in this study.
Taken together, the data demonstrate that Ad49-based vectors have great potential for CABG gene therapy application in the clinic. Further investigation using Ad49 in preclinical CABG models is warranted.
Footnotes
Acknowledgments
This work was funded by a British Heart Foundation Project Grant (PG/12/19/29455). AHB is supported by a Royal Society Wolfson Merit Award. We thank Nicola Britton, Gregor Aitchison, and Zaki Rabhan for their technical assistance. We would also like to thank Jerome Custers and Taco Uil (Crucell, Leiden, The Netherlands) for supplying the PER.C6 cells and adenovirus strains used in this study.
Author Disclosure Statement
No competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
