Abstract
Liver gene transfer with adeno-associated viral (AAV) 2/8 vectors is being considered for therapy of systemic diseases like mucopolysaccharidosis type VI (MPS VI), a lysosomal storage disease due to deficiency of arylsulfatase B (ARSB). We have previously reported that liver gene transfer with AAV2/8 results in sustained yet variable expression of ARSB. We hypothesized that the variability we observed could be due to pre-existing immunity to wild-type AAV8. To test this, we compared the levels of AAV2/8-mediated transduction in MPS VI cats with and without pre-existing immunity to AAV8. In addition, since levels of lysosomal enzymes as low as 5% of normal are expected to be therapeutic, we evaluated the impact of pre-existing immunity on MPS VI phenotypic rescue. AAV2/8 administration to MPS VI cats without pre-existing neutralizing antibodies to AAV8 resulted in consistent and dose-dependent expression of ARSB, urinary glycosaminoglycan (GAG) reduction, and femur length amelioration. Conversely, animals with pre-existing immunity to AAV8 showed low levels of ARSB expression and limited phenotypic improvement. Our data support the use of AAV2/8-mediated gene transfer for MPS VI and other systemic diseases, and highlight that pre-existing immunity to AAV8 should be considered in determining subject eligibility for therapy.
Introduction
Therapies for MPS VI and other LSDs rely on physiological secretion and uptake of lysosomal enzymes by most cells via the mannose 6-phosphate receptor pathway (Sands and Davidson, 2006). Enzyme replacement therapy (ERT) is the current treatment for MPS VI. Clinical evidence showed several limitations of ERT. First, despite reduction of visceromegaly and improvement of endurance, ERT failed to ameliorate cardiac and visual function and bone abnormalities, likely because of the limited biodistribution of recombinant human ARSB (rhARSB) (Harmatz et al., 2004; Harmatz et al., 2005a; Harmatz et al., 2006; Harmatz et al., 2008). Second, the requirement of weekly intravenous infusions as a result of the rhARSB short half-life (Crawley et al., 1996; Harmatz et al., 2005b) carries the risk of anaphylactic reactions and results in limited compliance to the therapy. Third, ERT has extremely high costs preventing access to therapy for patients in less-developed countries. Thus, alternative strategies with a similar or better efficacy and without inconvenient requirement of multiple infusions are required.
We have recently demonstrated that gene therapy has the potential to provide a more effective and long-term treatment for MPS VI. In our previous studies (Tessitore et al., 2008; Tessitore et al., 2009; Cotugno et al., 2010; Cotugno et al., 2011), performed on rat and cat models of the disease (Yoshida et al., 1993; Yogalingam et al., 1996), we used a gene therapy approach based on intravenous administration of adeno-associated viral vectors based on serotype 8 (AAV2/8), which efficiently transduce the liver (Thomas et al., 2004; Gao et al., 2006; Wang et al., 2010b; Nathwani et al., 2011a). Single systemic administration of AAV2/8 vectors encoding ARSB resulted in long-term production and systemic secretion of therapeutic ARSB from transduced hepatocytes (Cotugno et al., 2012). This resulted in amelioration of the MPS VI phenotype, including reduction of GAG storage and heart valve abnormalities and improvement of long bone length and mobility in both MPS VI rats and cats (Tessitore et al., 2008; Tessitore et al., 2009; Cotugno et al., 2010; Cotugno et al., 2011). These data bode well for clinical translation of AAV2/8-mediated liver gene therapy for MPS VI. Importantly, the initial results of a clinical trial that tests the efficacy and safety of intravenous administration of AAV2/8 in hemophilia B patients are extremely encouraging, with factor IX (FIX) expression stable for over a year after vector administration (Nathwani et al., 2011b).
One factor that may influence the outcome of in vivo gene transfer is the presence of pre-existing immunity due to previous natural infections with wild-type AAV8 (referred to in this manuscript as AAV8 and different from the recombinant vectors we call AAV2/8), which has been isolated from nonhuman primates (Gao et al., 2002) and is able to infect humans (Calcedo et al., 2009; Boutin et al., 2010). Indeed, the prevalence of serum antibodies to AAV8 varies between 15 and 25% in the human healthy population (Calcedo et al., 2009; Boutin et al., 2010). Recent studies have pointed out that very low levels of neutralizing antibodies (Nab) to AAV8 are sufficient to prevent liver transduction by the vector (Wang et al., 2010a; Nathwani et al., 2011b; Wang et al., 2011). However, to our knowledge no studies yet have characterized if these lower transduction levels may still be therapeutic. This information may be crucial in order to decide whether individuals with low levels of Nab to AAVB may still be eligible for treatment. LSDs, including MPS VI, are ideal to answer this question since lysosomal enzyme levels as low as 5 to 10% of normal are expected to be therapeutic (Neufeld and Muenzer, 2001).
In our previous study on MPS VI cats (Cotugno et al., 2011), we observed variability in liver transduction efficiency, which was independent of vector production, presence/absence of the WPRE (woodchuck hepatitis virus post-transcriptional regulatory element), and sex of the cats injected. Exposure to AAV8 before vector administration was hypothesized in the majority of animals showing low levels of ARSB expression, although pre-existing levels of Nab to AAV8 were not assessed.
In the present study, MPS VI cats were screened for the presence of Nab to AAV8 before treatment with AAV2/8-TBG-fARSB (a vector which encodes feline ARSB under the transcriptional control of the thyroxine-binding globulin promoter-TBG). We evaluated liver transduction and therapeutic efficacy of AAV2/8-mediated liver gene transfer in MPS VI cats both pre-exposed and not to AAV8.
Materials and Methods
Animal colony
The feline MPS VI colony was maintained at the University of Pennsylvania, School of Veterinary Medicine. Animals were raised under National Institutes of Health (NIH) and U.S. Department of Agriculture (USDA) guidelines for the care and use of animals in research. The animals' genotype was determined by polymerase chain reaction (PCR)-based analysis.
AAV vector production and administration
Therapeutic AAV2/8-TBG-fARSB and control AAV2/8-TBG-eGFP (enhanced green fluorescent protein) vectors were produced by the AAV Vector Core of the Telethon Institute of Genetics and Medicine (TIGEM, Naples, Italy), as previously described (Cotugno et al., 2011). MPS VI cats were injected by catheter with various doses (reported in the Results section and in Table 1) of AAV2/8-TBG-fARSB vector in a cephalic vein at p50-p63. As control, one normal and two MPS VI cats received 6×1012 genome copies (gc)/kg of AAV2/8-TBG-eGFP at p50. Pre-existing immunity to AAV8 capsid was induced in one MPS VI cat by subcutaneous injection of 1×1011 gc/kg of AAV2/8-TBG-eGFP at p26 before treatment with the therapeutic vector at p50. One cat in the group receiving 2×1012 gc/kg of AAV2/8-TBG-fARSB and one in the group with pre-existing immunity injected with the same vector dose was sacrificed at six months post-injection. For one cat receiving 2×1011 gc/kg, sacrifice was necessary due to development of feline infectious peritonitis (FIP). Thus, data at 12 months post-treatment are not available for these animals. The experimental groups are described in Table 1.
NB: NR, normal cats; AF, MPS VI affected cats; p, postnatal day; gc/kg, genome copies/kg.
Nab to AAV assay
MPS VI cats were screened for the presence of pre-existing neutralizing antibodies to AAV8 using the in vitro transduction inhibition assay. The AAV8 neutralizing antibody assay was performed on Huh7 cells as previously described (Calcedo et al., 2009). The limit of detection for the assay is 1:5 serum dilutions.
Blood and urine collection
Blood and urine were collected at different times during the period of analysis from treated and control cats. Blood samples were centrifuged at 3,000 g in a Hermle Z 200A centrifuge (Labnet, Woodbridge, NJ) for 10 minutes at room temperature to obtain the serum. Urine was collected by cystocentesis and frozen at −80°C until the analysis was performed.
ARSB activity assay
ARSB activity in serum samples was measured as previously described (Cotugno et al., 2011). Briefly, 15 microliters (μl) serum was diluted with 285 μl H2O, and 100 μl of diluted serum was incubated in the following solution: 5 mmol/l 4-methylumbelliferyl sulfate, 0.05 mol/l sodium acetate pH 5.6, 0.3 mmol/l silver nitrate, 3 mmol/l lead acetate (Sigma-Aldrich, St. Louis, MO). Samples were incubated for 2 hours at 37°C, and the reaction was stopped by the addition of 2 ml of 0.32 M glycine and 0.2 M carbonate (pH 10.5), which contained 1 mM ethylenediaminetetraacetic acid (EDTA). Fluorescence was then measured with a VersaFluor fluorometer (BioRad, Hercules, CA). Serum ARSB activity is expressed as nmol/mL/h. For comparison of ARSB activity among experimental groups, as reported in the Results section, the serum enzyme activity measured over time was averaged for each cat, and the resulting value was then averaged for each group.
Transaminases analysis
Serum transaminases were measured in MPS VI cats receiving AAV2/8-TBG-fARSB, at different times following vector administration, and in untreated controls. The analysis was run on a Vitros 350 chemistry analyzer (Ortho-Clinical Diagnositics, Johnson & Johnson, Inc, New Brunswick, New Jersey).
Quantitative analysis of GAG accumulation in urine
Urine samples were clarified by centrifugation and GAG extraction was performed using a Glycosaminoglycan Isolation & Concentration Kit (Biocolor Ltd., Carrickfergus, Antrim, UK), following the manufactory's protocol. Creatinine concentration was determined by the Jaffe' colorimetric method (Cook, 1975).
Radiograph analyses
Radiographs were taken 6 and 12 months after treatment with the therapeutic vector using a General Electric MPH high-frequency multipulse generator with automatic exposure control radiology machine. The length of the femur and other long bones (data not shown) was measured on digital radiographs with the iSite Enterprise 3.5 software (Stentor; Philips Medical System, Foster City, CA) and reported as % of the length of each bone in sex- and age-matched normal cats. All but one normal cat and all but two affected, untreated cats used for femur length analysis were historical controls.
Results
Serum ARSB levels in MPS VI cats injected with various doses of AAV2/8-TBG-fARSB and the impact of pre-existing immunity to AAV8 on liver transduction
MPS VI cats were screened for the presence of Nab to AAV8 by an in vitro transduction inhibition assay prior to AAV2/8-TBG-fARSB vector administration. MPS VI cats without detectable pre-existing Nab to AAV8 were injected at postnatal day 50 to 63 (p50–63) with various doses of AAV2/8-TBG-fARSB vector which encodes feline ARSB (fARSB) under the control of the liver-specific thyroxine-binding globulin (TBG) promoter (Table 1). As control, one normal and two MPS VI cats received 6×1012 gc/kg of AAV2/8-TBG-eGFP at p50 (Table 1). As expected, MPS VI cats that received AAV-eGFP vectors showed serum ARSB activity similar to untreated affected animals while normal, stable levels of serum ARSB were measured in the wild-type cat receiving AAV-eGFP (data not shown). Normalized serum ARSB activity was measured in all MPS VI cats injected with 2×1012 gc/kg of AAV2/8-TBG-fARSB, whereas cats injected with a ten-fold lower dose (2×1011 gc/kg) did not show detectable increases in the levels of serum ARSB. Indeed these levels remained in the range of MPS VI untreated cats. The mean ARSB activity was 13±3.6 and 2.4±0.8 nmol/mL/h in cats receiving 2×1012 and 2×1011 gc/kg, respectively (Fig. 1). In cats treated with the intermediate dose (6×1011 gc/kg) of vector, ARSB circulating levels were in the normal range in two out of three cats, and were above affected but below normal in one out of three (average: 8.7±4.7 nmol/mL/h) (Fig. 1). Even though a significant correlation between circulating ARSB levels and the vector dose administered could not be observed, serum ARSB levels were more consistent within the same dose administered than what we observed previously (Cotugno et al., 2011), suggesting that the variability reported could indeed be ascribed to the presence of pre-existing Nab to AAV8.
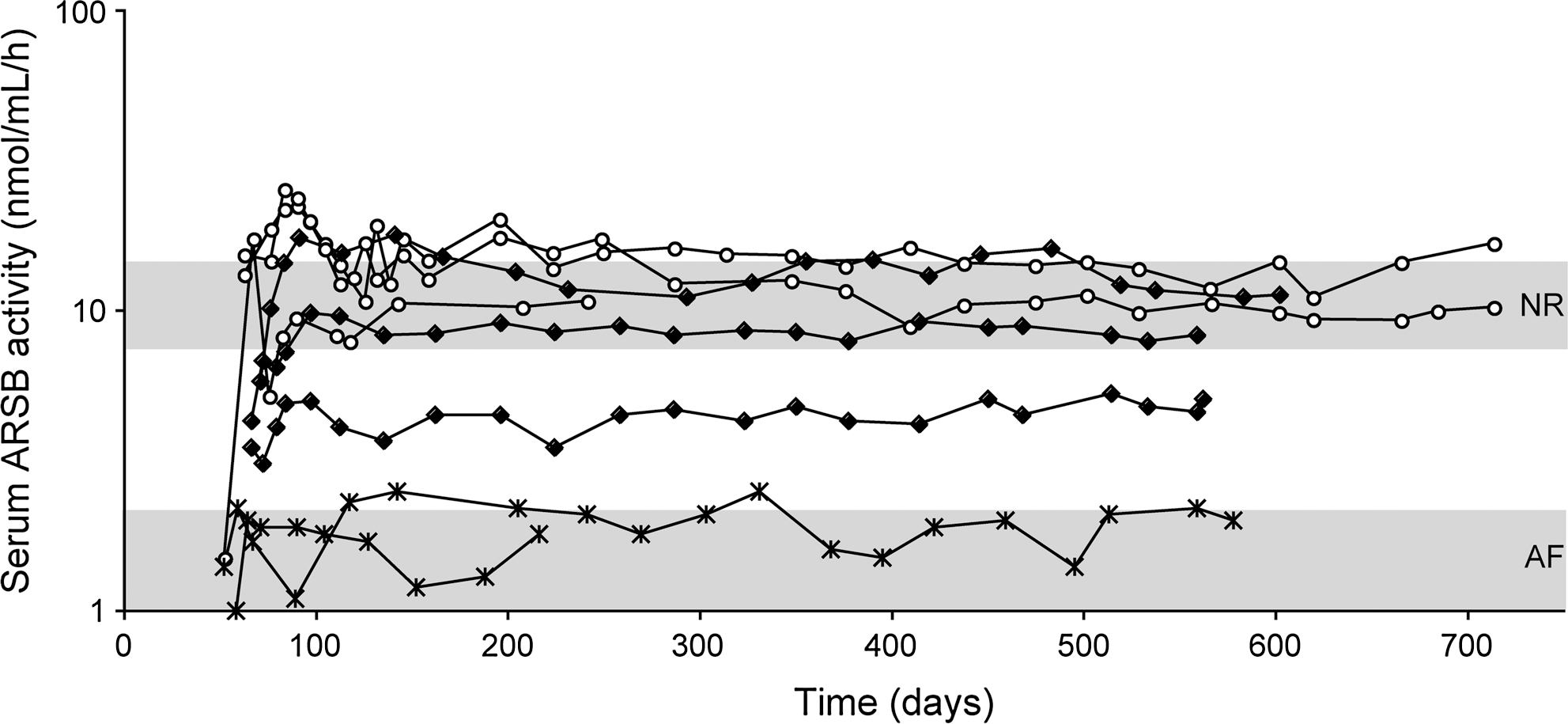
Serum arylsulfatase B (ARSB) activity in mucopolysaccharidosis type VI (MPS VI) cats treated with AAV2/8 in the absence of Nab to AAV8. MPS VI cats were screened for the presence of Nab to AAV8 before the treatment and those that were negative were injected at postnatal days 50–63 with 2×1011, 6×1011, or 2×1012 gc/kg of AAV2/8-TBG-fARSB. Serum ARSB activity was monitored over time after vector administration. Each line represents ARSB values over time from a single animal. The upper and lower gray areas represent the range of values of normal (NR) and affected (AF) cats, respectively. The lines with×represent AF animals (n=2) treated with 2×1011 gc/kg. The lines with full symbols represent AF animals (n=3) treated with 6×1011 gc/kg. The lines with empty symbols represent AF animals (n=3) treated with 2×1012 gc/kg. AAV2/8, adeno-associated viral vectors based on serotype 8; TBG, thyroxine-binding globulin fARSB, vector-encoding feline ARSB; Nab, neutralizing antibodies.
To further support this hypothesis, two cats with pre-existing immunity to AAV8 at the time of injection were treated with 2×1012 gc/kg of AAV2/8-TBG-fARSB (Table 1), and their serum ARSB activity was compared to that of cats with no Nab to AAV8 that were injected with the same vector dose (Fig. 2). One cat with a Nab to AAV8 titer of 1:5/1:10 showed slightly increased levels of circulating ARSB above the range of untreated MPS VI cats. In the other cat, pre-existing immunity to AAV8 was induced by a subcutaneous injection of 1×1011 gc/kg of AAV2/8-TBG-eGFP 24 days before treatment with the therapeutic vector. Although the Nab titer measured 21 days after subcutaneous injection of AAV2/8 remained under the detection limit of the assay (1:5), the serum ARSB levels of this animal were similar to those of MPS VI untreated cats (Fig. 2). These results suggest that pre-existing immunity had a negative impact on AAV-mediated liver gene transfer in MPS VI cats that are immune-tolerant to feline ARSB (Tessitore et al., 2008; Cotugno et al., 2011).
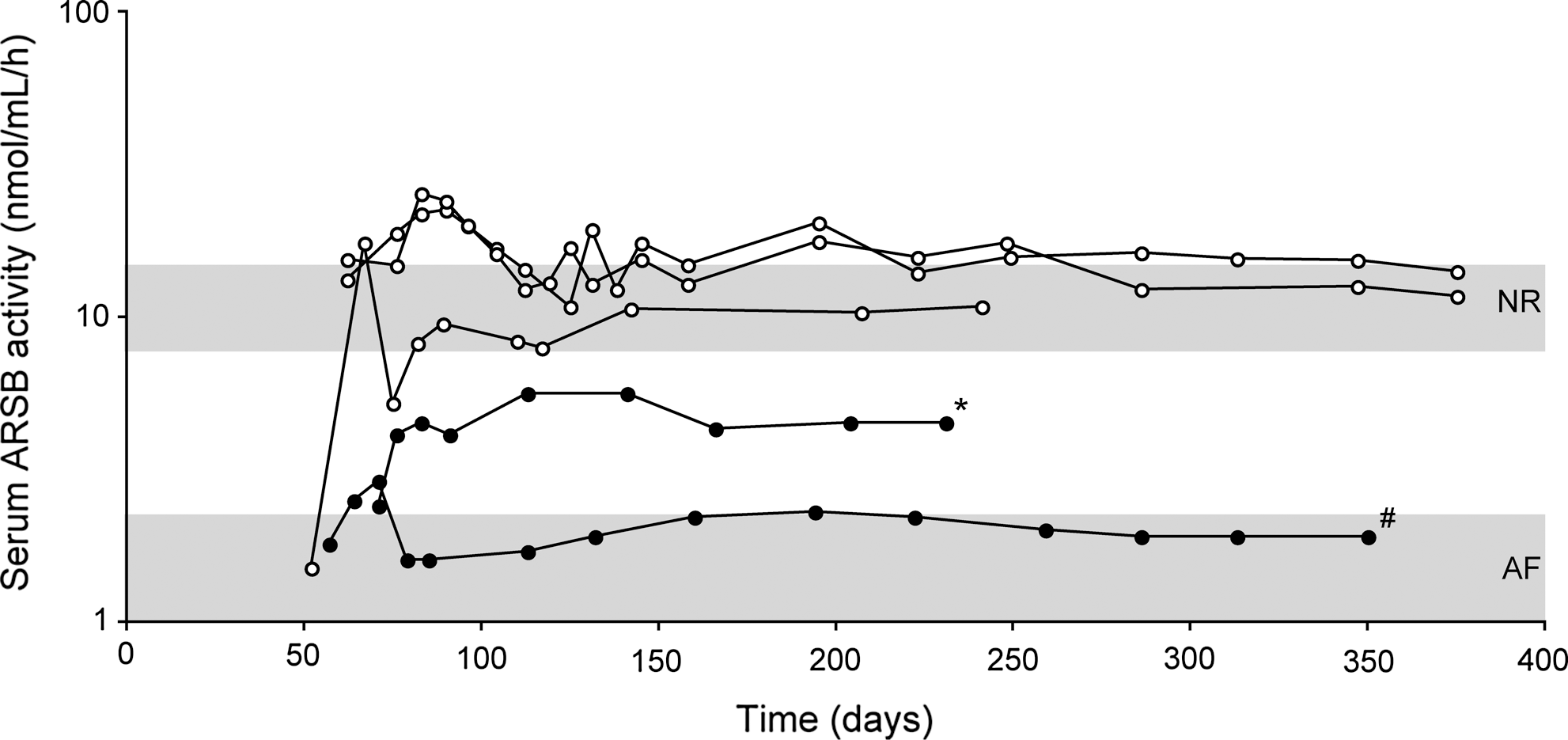
Impact of pre-existing immunity to AAV8 on liver transduction in MPS VI cats treated with AAV2/8. MPS VI cats with (solid circle) and without (empty circle; these are the same animals as in Fig. 1) pre-existing immunity to AAV8 were injected with 2×1012 gc/kg of AAV2/8-TBG-fARSB. The line marked with * is relative to a cat with a pre-existing Nab titer to AAV8 of 1:5/1:10. The line marked with # is relative to a cat in which pre-existing immunity to AAV8 was induced by subcutaneous injection of 1×1011 gc/kg of AAV2/8-TBG-eGFP vector before treatment with the therapeutic vector. Serum ARSB activity was monitored over time after vector administration. The upper and lower gray areas represent the range of values of normal (NR) and affected (AF) cats, respectively.
In addition, we have measured serum transaminases in some cats receiving AAV2/8-TBG-fARSB, and we did not observe significant increases in alanine transaminase (ALT) and aspartate transaminase (AST) levels (Table 2). This indirectly suggests the absence of major inflammatory and T-cell-mediated responses to AAV at the time of the analysis.
NB: SE, standard error; U/L, units/L; normal values: ALT=33–152 U/L, AST=1–37 U/L. Please note that untreated cats age-matched with AAV-treated cats analyzed at 5–7 days post-injection have an AST mean value of 41.3±8.9 U/L.
Urinary GAGs in MPS-VI cats treated with AAV2/8
Reduction of urinary GAGs is a sensitive marker of lysosomal storage clearance and therapeutic efficacy in LSDs. Urinary GAGs were significantly increased in MPS VI cats, which received AAV-eGFP, compared to normal cats (Fig. 3). A slight urinary GAG reduction was observed in the group (n=2) of MPS VI cats treated with the lower dose of AAV2/8-TBG-fARSB vector (2×1011 gc/kg). Significant reduction of urinary GAGs was observed in MPS VI cats receiving either 6×1011 or 2×1012 gc/kg of AAV2/8-TBG-fARSB. On the other hand, administration of the highest dose of AAV2/8-TBG-fARSB (2×1012gc/kg) did not result in any significant decrease in urinary GAGs in MPS VI cats with pre-existing immunity to AAV8 (Fig. 3). Indeed, a modest reduction of urinary GAGs was present only in the animal with levels of circulating ARSB above affected.
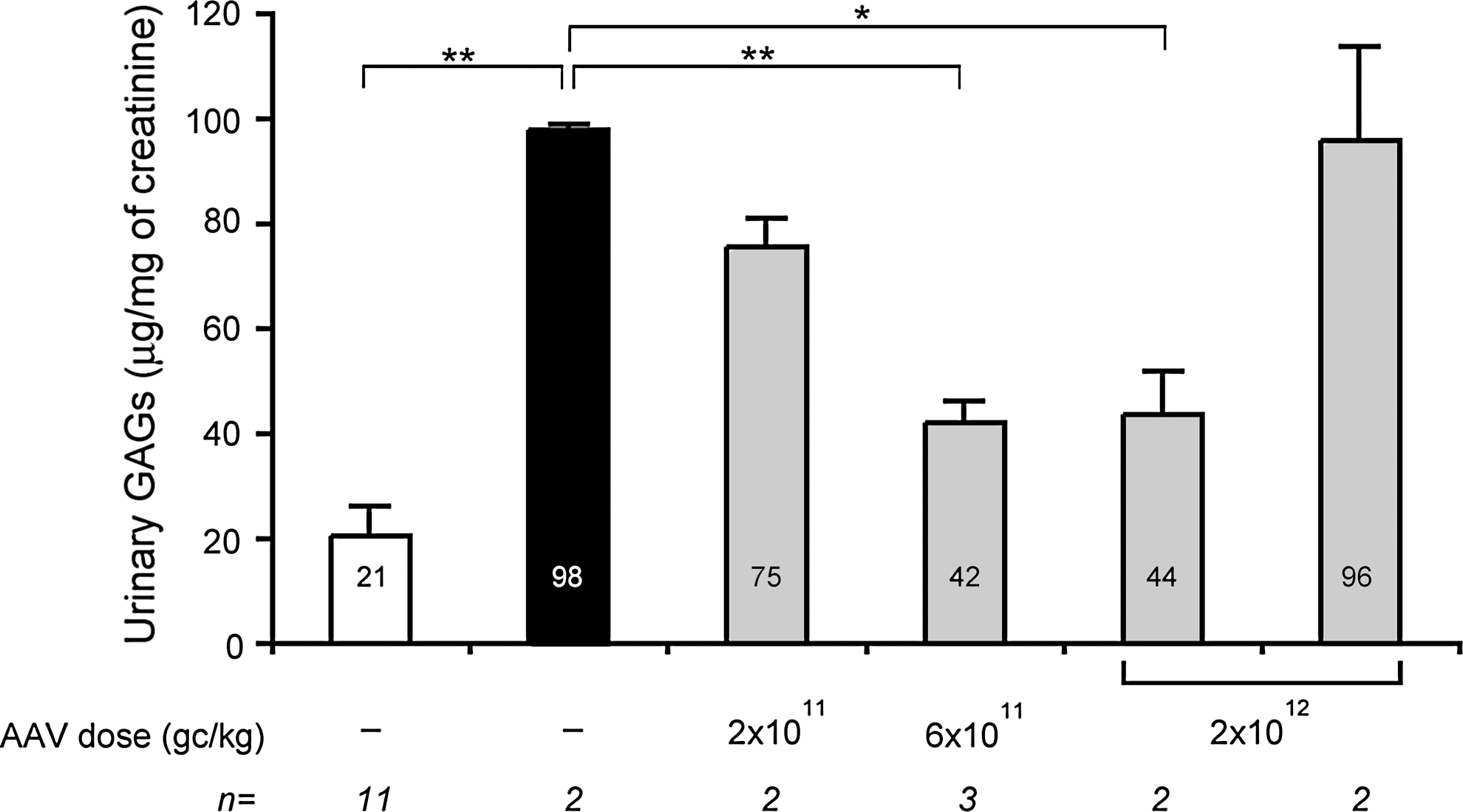
Urinary GAGs levels in MPS VI cats treated with AAV2/8. Urinary GAGs were measured in MPS VI cats with (last gray bar on the right) or without pre-existing immunity to AAV8 (gray bars) and in normal (NR, white bar) and affected (AF, black bar) controls. Cats without pre-existing immunity to AAV8 were injected at postnatal day 50–63 with three different doses (indicated under each bar) of AAV2/8-TBG-fARSB. Cats with pre-existing immunity to AAV8 were injected with the highest vector dose (2×1012 gc/kg). The urinary GAG levels measured over time were averaged for each cat, and the resulting value was then averaged for each group and is indicated inside each bar. Values are represented as mean±SE. The p value vs. AF: * ≤0.05, ** ≤0.01. The number (n) of animals in each group is indicated under each bar. GAG, glycosaminoglycan.
Femur length in MPS VI cats treated with AAV2/8
MPS VI is characterized by severe skeletal dysplasia, which is one of the major challenges of treating MPS VI. In our previous study (Cotugno et al., 2011), we reported that AAV2/8-mediated ARSB liver gene transfer improved long bone length of treated animals. To confirm this and to assess the impact of pre-existing immunity to AAV8, we performed radiographic analyses on AAV-treated MPS VI cats at 6 and 12 months post-injection as well as on untreated normal and affected age-matched controls.
As shown in Figure 4, femur length was significantly reduced in MPS VI cats compared to age- and sex-matched normal controls. Six months after vector administration, we observed increased femur length in all AAV2/8-TBG-fARSB-treated groups without pre-existing immunity (Fig. 4). In particular, the improvement was more consistent in the group treated with the highest dose of AAV vector (2×1012 gc/kg). Indeed, all cats within this dose cohort showed improved femur length, whereas more variable rescue was observed in cats injected with 2×1011 and 6×1011 gc/kg (Fig. 4). On the other hand, no amelioration occurred in the group of cats showing pre-existing immunity to AAV8 injected even with the highest dose (2×1012gc/kg) of vector (Fig. 4), despite the presence of one animal with circulating ARSB levels slightly above the range of affected cats. Further amelioration of femur length occurred in all AAV-treated cats negative for Nab to AAV8 that were followed up to 12 months post-treatment (Fig. S1; Supplementary Material available online at
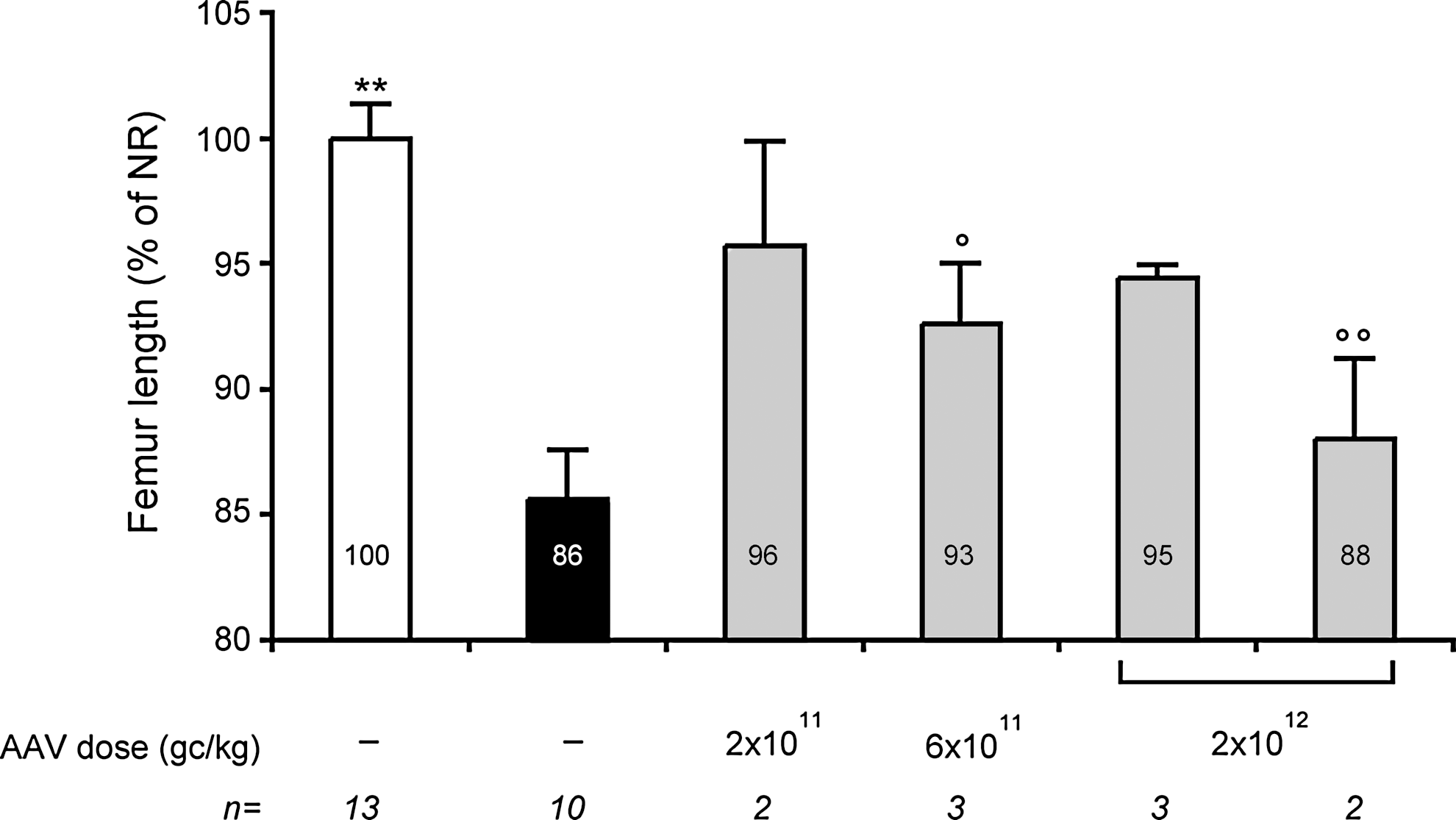
Femur length in MPS VI cats treated with AAV2/8. Femur length was measured in MPS VI cats with and without pre-existing immunity to AAV8. Cats without pre-existing immunity to AAV8 (gray bars) were injected at postnatal day 50–63 with three different doses (indicated under each bar) of AAV2/8-TBG-fARSB. Cats with pre-existing immunity to AAV8 (last gray bar on the right) were injected with the highest vector dose (2×1012 gc/kg). Radiographs were performed at 6 months post-injection and in age-matched affected (AF, black bar) and normal (NR, white bar) controls. Femur length is reported as a percentage of age- and sex-matched NR cats and indicated inside each bar. Results are reported as mean±SE. The number (n) of animals analyzed in each group and the vector doses used are reported under each bar. The p value vs. NR: ° ≤0.05, °° ≤0.01; p value vs. AF: ** ≤0.01.
Discussion
Liver-directed gene transfer using vectors based on AAV2/8 is being considered for long-term treatment of both liver-specific and systemic diseases (Brunetti-Pierri and Auricchio, 2010). The results from a recent clinical trial testing the safety and efficacy of intravascular administration of AAV2/8 in hemophilia B patients are extremely encouraging (Nathwani et al., 2011b). In fact, long-term expression of therapeutic levels of FIX has been achieved in hemophilia B patients treated with AAV2/8 (Nathwani et al., 2011b).
MPS VI is an ideal LSD for testing the efficacy of a similar strategy based on AAV2/8-mediated liver gene transfer because (1) MPS VI presents systemic life-threatening symptoms without central nervous system (CNS) involvement, thus therapy is not limited by the blood brain barrier; and (2) the current treatment of weekly lifelong ERT infusions could be replaced by gene therapy that provides stable ARSB expression following a single intravascular vector administration.
Indeed, we have shown that AAV2/8-mediated liver ARSB gene transfer can provide therapeutic levels of circulating enzyme long-term after a single systemic vector administration in MPS VI cats, resulting in significant phenotype improvement (Cotugno et al., 2011). In the previous study, an unexpected finding was the high variability in liver transduction efficiency (measured as serum ARSB activity) in AAV-treated cats receiving the same vector dose (Cotugno et al., 2011). We hypothesized that this could be due to the presence of pre-existing immunity to AAV8. Indeed, several preclinical and clinical studies showed that pre-existing immunity to AAV is a factor that may limit effective gene transfer by antibody-mediated neutralization of vector particles in a serotype-specific manner (Gao et al., 2006; Manno et al., 2006; Hurlbut et al., 2009; Wang et al., 2010a; Nathwani et al., 2011b; Wang et al., 2011).
Here we show that animals without detectable Nab to AAV8 present similar levels of AAV2/8-mediated liver transduction. This is especially true for those treated with the highest and lowest vector doses. Systemic administration of intermediate vector doses (6×1011 gc/kg of AAV2/8-TBG-fARSB) resulted in variable levels of circulating ARSB activity. A possible explanation for this variability is that this dose may be at the threshold required to achieve a detectable increase in transgene expression. Thus, expression could be more susceptible to the effects of differences due to the mixed genetic background of MPS VI cats (Haskins et al., 1979) and/or to the gender of the animals injected. Some studies suggest that AAV vector-mediated liver gene transfer is more efficient in males than females (Davidoff et al., 2003; Nathwani et al., 2009; Paneda et al., 2009), and in this dose cohort, cats showing the lower levels of serum ARSB were both females. However, we can not exclude that the variable levels of circulating ARSB activity observed in this cohort of cats could be alternatively due to the presence of Nab to AAV8 below the sensitivity of our assay, similarly to the cat without detectable levels of anti-AAV8 Nab despite the previous administration of AAV2/8-TBG-eGFP.
Our study shows that pre-existing Nab to AAV8, even at low levels (1:5/1:10 to undetectable), negatively impacts transgene expression levels. These results are in agreement with other studies, showing that even low titers of pre-existing Nab strongly limit the levels of AAV2/8-mediated liver transduction (Wang et al., 2010a; Wang et al., 2011).
While previous studies analyzed the impact of pre-existing immunity to AAV8 by measuring levels of transgene expression following AAV2/8 delivery (Gao et al., 2006; Hurlbut et al., 2009; Wang et al., 2010a; Wang et al., 2011), our study additionally tested if this significantly limits therapeutic efficacy in an animal model of human disease. For this purpose, we have selected a model of lysosomal storage disorder in which even low levels of transgene expression are expected to provide phenotypic improvement. Although in animals without pre-existing Nab to AAV8 low to undetectable levels of circulating ARSB were still associated with phenotypic improvement, no significant amelioration of the MPS VI phenotype was observed in animals with pre-existing immunity to AAV8, suggesting that low levels of Nab to AAV8 severely limit therapeutic efficacy.
We additionally observed that in animals without detectable pre-existing immunity to AAV8, vector doses as low as 6×1011 gc/kg resulted in significant reduction of urinary GAGs and improvement in femur length. Interestingly, in MPS VI cats receiving 2×1011 gc/kg, despite serum ARSB levels similar to untreated MPS VI cats, minor reductions in urinary GAGs were observed. In particular, one cat showed more evident reduction of urinary GAGs and also femur length normalization at 12 months post-injection. These data indicate that administration of 2×1011 gc/kg of AAV2/8-TBG-fARSB resulted in therapeutic efficacy even in the absence of detectable increases in circulating ARSB levels, similar to what we have observed previously (Cotugno et al., 2011). These findings have two important implications. First, the doses of AAV2/8-TBG-ARSB vector to be tested in a clinical trial for MPS VI should range between 2×1011 and 2×1012 gc/kg. Second, urinary GAGs are a more sensitive endpoint than serum ARSB activity to evaluate gene transfer efficacy.
These data also highlight differences in response to ARSB gene transfer between MPS VI rats and cats. While in MPS VI rats close-to-normal levels of circulating ARSB are required to improve long bone length (Cotugno et al., 2010), in the feline model of MPS VI improvement of long bones can be achieved at lower levels of circulating ARSB obtained with low or intermediate doses of AAV2/8-TBG-fARSB. This could be explained by (1) the different vascularization of long bones or the different levels of mannose 6-phosphate receptor expression between the two species; and (2) the ARSB transgene type, in that we used the species-specific feline transgene in cats, whereas the human transgene was used in MPS VI rats (Cotugno et al. 2010).
In conclusion, we confirm that AAV2/8-mediated ARSB gene transfer to liver resulted in a vector-dose-related improvement of both biochemical and skeletal characteristics of feline MPS VI. We also showed that pre-existing immunity to AAV8 negatively impacted liver transduction and, more importantly, the therapeutic efficacy of AAV2/8-mediated gene transfer in MPS VI cats. Our data support the exclusion of patients with detectable Nab to AAV8 from gene therapy clinical trials for MPS VI. In this context, the lower seroprevalence of Nab to AAV8 than AAV2 in humans (Calcedo et al., 2009; Boutin et al., 2010) is advantageous when considered for the clinical application of vectors based on AAV8.
Footnotes
Acknowledgments
This work was supported by funds from the Italian Telethon Foundation (grant TGM 11 MT6); NIH grants RR02512, DK25759, and P30-DK047757; the U.S. National MPS VI Society; and the Isaac Foundation. We thank Graciana Diez-Roux and Ellen Abrams (Telethon Institute of Genetics and Medicine, TIGEM) for the critical reading and proofreading of the manuscript.
Author Disclosure Statement
J.M.W. is a consultant to ReGenX Holdings, and is a founder of, holds equity in, and receives a grant from affiliates of ReGenX Holdings; in addition, he is an inventor on patents licensed to various biopharmaceutical companies, including affiliates of ReGenX Holdings. Other authors declare no conflicts of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
