Abstract
In these studies we delivered by hydrodynamic limb vein (HLV) injection plasmid DNA (pDNA) expressing the full-length mouse dystrophin gene to skeletal muscles throughout the hind limbs of the mdx mouse model for Duchenne muscular dystrophy (DMD). We evaluated the levels and stability of dystrophin expression and measured the resulting muscle protection, using Evans blue dye (EBD) to mark the damaged myofibers. Plasmid delivery was as efficient in the dystrophic mice as in wild-type mice and equally efficient in young adult and old mice, as long as the dose of pDNA was adjusted for the target muscle weight. The HLV gene delivery procedure was tolerated well by the dystrophic mice and repeat injections could be performed over an extended period of time. Multiple gene deliveries additively increased the amount of dystrophin protein and also increased the percentages of dystrophin-expressing myofibers. Plasmids expressing dystrophin from a cytomegalovirus (CMV) promoter construct containing the HMG1 intron provided stable dystrophin expression for the life of the mouse and provided significant benefit to the limbs. EBD staining showed that dystrophin gene delivery preserved myofibers in the CMV-HMGi-mDys-injected leg by 2.5- to 5-fold in large groups of muscles and by 2.5-fold throughout the injected legs, compared with the contralateral control legs injected with a nonexpressing plasmid. A similar degree of protection was measured in young adult mice evaluated soon after the last gene delivery and in aged mice injected over an extended period of time. This degree of protection resulted from 18 to 20% of the normal level of dystrophin protein, with 11–16% dystrophin-expressing myofibers. These studies show promise for the use of HLV injections to deliver therapeutic doses of full-length dystrophin-expressing plasmids for long-lasting protection of skeletal muscles in patients with DMD.
Introduction
There are no cures for DMD at present in the clinic, but there has been a flowering of new approaches for treatment. The treatments can be broadly categorized as gene therapy, cell-based approaches, and drug therapies (Allamand and Campbell, 2000; Voisin and De la Porte, 2004; Cossu and Sampaolesi, 2007; Braun, 2008). Gene therapies involve nonviral delivery of the dystrophin gene in a plasmid or viral delivery of the internally deleted minidystrophin or a U1 or U7 small nuclear RNA expression vector to skip exons containing mutations (Goyenvalle et al., 2004; Romero et al., 2004; Zhang et al., 2004; Denti et al., 2006; Odom et al., 2007). Cell-based therapies involve myogenic precursor cells or stem cells (Sampaolesi et al., 2006; Kimura et al., 2008). Some drug therapies may be palliative to reduce muscle inflammation and degeneration (Matsumura et al., 2009). Others are designed to replace dystrophin by expression of an internally modified endogenous dystrophin gene via antisense oligonucleotides to induce exon skipping or read-through of nonsense mutations (Yokota et al., 2007) or by upregulating endogenous utrophin as a replacement for dystrophin (Miura and Jasmin, 2006). Each of these approaches has advantages and disadvantages.
Nonviral delivery of naked plasmid DNA (pDNA) to express therapeutic genes is an attractive approach to treat muscle diseases and has a good safety record (Braun, 2008; Duan, 2008). Plasmid DNA is a stable, biologically active molecule that is relatively inexpensive to produce in large quantities. Unlike viral vectors, pDNA does not induce an immune response toward the vector and thus repeat deliveries for chronic diseases are possible (Jiao et al., 1992; Le et al., 2000; Zhang et al., 2001; Hagstrom et al., 2004; Romero et al., 2004). The full-length dystrophin gene can be encoded in a plasmid and delivered to skeletal muscles (Acsadi et al., 1991; Danko et al., 1993; Hagstrom et al., 2004; Romero et al., 2004; Zhang et al., 2004). Plasmid DNA delivered to skeletal muscle of rodents or primates is retained in muscle fibers and expresses the encoded gene product for many months, likely because the differentiated skeletal myofibers have a low rate of turnover (Wolff et al., 1992; Danko et al., 1993, 1997; Zhang et al., 2004; Sebestyen et al., 2007). Such vectors have been delivered locally to skeletal muscles in mice by intramuscular injection and electroporation (Wolff et al., 1992; Danko et al., 1993, 1997; Hartikka et al., 2001). More recently, vascular delivery procedures have been used to deliver pDNA to an entire limb by intraarterial injection (Zhang et al., 2001, 2004) or by hydrodynamic limb vein (HLV) injection (Hagstrom et al., 2004). In the HLV injection procedure, a tourniquet is used to limit the delivery area to one limb per injection. Naked pDNA is rapidly injected into the vein in the anterograde direction. A sufficient volume of saline is used to facilitate extravasation of the pDNA from the vasculature and into the muscle tissue. The transcutaneous injection is a nonsurgical procedure in large animals, and multiple injections can be performed to increase expression levels. This procedure has a good safety profile (Hagstrom et al., 2004; Toumi et al., 2006; Vigen et al., 2007) and is a compelling candidate for the treatment of neuromuscular diseases such as DMD.
The mdx mouse is the most widely used animal model for DMD (for reviews see Allamand and Campbell, 2000; Grounds et al., 2008). There are several strains of mdx mice that all have a mutated dystrophin gene and the genetics are well defined. The Dmdmdx strain (mdx-10ScSn) arose spontaneously from the C57BL/10ScSn strain. These mice have a premature termination codon in exon 23. Approximately 1% of the myofibers are revertants that express a truncated dystrophin protein. The Dmdmdx-4Cv (mdx4Cv) strain was developed by chemical mutagenesis and backcrossed into the C57BL/6 strain. mdx4Cv mice have a premature termination codon in exon 53 of the dystrophin gene. This strain has 10 times fewer revertant myofibers than the mdx-10ScSn strain (Danko et al., 1992). The DMD disease phenotype appears less severe in mdx mice than in humans. Compensatory mechanisms that decrease the severity of DMD in mdx mice include upregulation of utrophin and integrin, and enhanced muscle regeneration (Bulfield et al., 1984; Coulton et al., 1988). Exercise regimens have been used to challenge mdx muscles and exacerbate the disease in order to determine whether a therapy is effective (Brussee et al., 1997; Vandebrouck et al., 2002; De Luca et al., 2003; Okano et al., 2005; Burdi et al., 2006; Hodgetts et al., 2006). Aged mdx mice have a decreased capacity for regeneration and a concomitant increase in severity of the disease. Therefore, the muscular dystrophy exhibited by old mdx mice more closely resembles the myopathy of DMD (Lefaucheur et al., 1995).
In this paper, we delivered to mdx mice by HLV injection pDNA expressing the full-length mouse dystrophin gene. First we evaluated approaches for increasing dystrophin gene delivery and expression in the limb and then we analyzed the efficacy of the gene therapy. Most studies that evaluate muscle strength in mdx mice use the small muscles of the anterior lower leg, the extensor digitorum longus or the tibialis anterior, for contraction studies (Petrof et al., 1993; Dellorusso et al., 2001). However, these small muscles were not transfected well under the conditions used in most of the studies reported here. The large muscles of the leg that transfect best by HLV injection can be tested for an improvement in strength by measuring the whole body tension, also called an escape test, as mice attempt to escape from a stimulus.
Protocols employing Evans blue dye (EBD) are commonly used to characterize muscular dystrophy in mouse models and also as an end point in therapeutic trials conducted in these mice (Matsuda et al., 1995; Straub et al., 1997; Grounds et al., 2008). EBD staining is a direct measure of myofiber membrane integrity. In this paper, we observed by EBD analysis in four independent studies direct functional benefits from the plasmid-delivered dystrophin gene in mdx mice.
Materials and Methods
Animals
Breeding pairs of B6Ros.Cg-Dmdmdx-4Cv /J (mdx4Cv) and C57BL/10ScSn-Dmdmdx /J (mdx-10ScSn) mice were obtained from Jackson Laboratories (Bar Harbor, ME) and bred at the University of Wisconsin-Madison (Madison, WI). Male and female mdx4Cv mice were used except where otherwise indicated. Animals had access ad libitum to water and Mouse Diet 5015 chow (PMI Nutrition International, Brentwood, MO) or Teklad Rodent Diet (Harlan Laboratories, Indianapolis, IN). Outbred ICR mice and inbred C57BL/6 mice were obtained from Harlan. Studies were conducted in accordance with the guidelines set forth by the animal care and use committees of the University of Wisconsin and Mirus Bio (Madison, WI) and by the National Institutes of Health (Bethesda, MD) Guidelines for the Care and Use of Laboratory Animals.
Hydrodynamic limb vein injection procedure
Mice were anesthetized with 1–2% isoflurane (Abbott Laboratories, North Chicago, IL). They were shaved and prepared for surgery and a small latex tourniquet was wrapped tightly around the upper hind limb (above the quadriceps) and secured in place with a hemostat. A small incision was made in the skin between the ankle and the knee to expose a segment of the distal great saphenous vein. A 30-gauge butterfly needle (made by our laboratory as described in Zhang et al., 2004) was then inserted into the distal great saphenous vein, advanced about 0.5 cm, and held in place during the injection. The catheter was connected to a Harvard PHD 2000 syringe pump (Harvard Apparatus, Holliston, MA) and the pDNA–saline solution was injected at a rate of 8 to 15 ml/min. To keep the injection time similar in animals, smaller mice (those weighing approximately 25 g or less) were injected at a rate of 8 ml/min and larger mice were generally injected at 12 ml/min. The tourniquet was removed about 2 min after injection and the skin was closed with 4-0 absorbable suture. The total tourniquet time was about 3 min and each procedure took 10 min to complete. When multiple injections were performed in the same leg, injections were done at least 2 weeks apart.
Injection solution volume
The HLV injection volume for wild-type mice was optimized in 25-g animals. To adjust for larger animals, the following formula was used: 1 ml + {[(body weight − 25 g)/25 g] × 0.5} ml, based roughly on the muscle weight increase relative to the body weight increase. The mdx mice were injected with 1 ml or with 1 ml + {[(body weight − 20 g)/20 g] × 2/3} ml. This formula takes into account the larger muscles of mdx relative to wild-type mice and the volume required to obtain similar amounts of muscle swelling. The hind leg muscle mass of a 25-g C57BL/6 mouse is similar to that of a 20-g mdx mouse. As was shown in nonhuman primates, the efficiency of pDNA delivery requires adequate muscle swelling (Vigen et al., 2007). These volume formulas are equivalent to approximately 1.5 ml/g muscle in the legs of wild-type and mdx mice. To avoid having an excess of injection solution that can leak out of the injection site, this formula was modified somewhat for mice weighing more than 40 g to 1 ml + {[(body weight − 20 g)/20 g] × 0.5} ml because the excess weight in large animals is often fat and not muscle.
Plasmid DNA
CMV-HMGi-mDys contains the human major immediate-early cytomegalovirus (CMV) enhancer/promoter, murine 3-hydroxy-3-methylglutaryl-Coenzyme A reductase (HMG) intron 1, mouse dystrophin gene coding sequence, late simian virus 40 (SV40) polyadenylation signal, and kanamycin resistance gene; it is 21,813 base pairs (bp) in size. CMV-SV40i-mDys contains the same CMV enhancer/promoter, along with the 16S/19S SV40 intron, mouse dystrophin coding sequence, late SV40 polyadenylation signal; and kanamycin resistance gene; it is 17,878 bp in size. Control plasmids contained no mammalian promoter and were ampicillin-resistant pUC19 or kanamycin-resistant plasmid pMIR266 that contained a 535-bp stuffer fragment of mouse genomic sequence from the intergenic region of the albumin gene family to enlarge the minimal size plasmid to 2257 bp. Luciferase-expressing pCMV-Luc contained the luciferase-encoding gene in the pCI mammalian expression vector (Promega, Madison, WI). All of these plasmids contained the pUC-based ColE1 origin of replication. Preparations of pDNA were purchased from commercial sources. All batches of pDNA passed quality control specifications for less than 100 EU of endotoxin per milligram, and for several preparations we confirmed independently that the levels were at least one magnitude less than this.
Testing pDNA preparations for muscle toxicity
DNA preparations were tested for toxicity by HLV injection of 500 μg of pDNA into the hind leg of an ICR mouse or 2.7 mg into a Sprague-Dawley rat and a group size of six animals. The contralateral legs received HLV injection of saline as a control. Three days later, rodents were anesthetized with 3 to 5% isoflurane and killed. The skin was then removed from the hind limb and the gastrocnemius muscle (posterior lower leg) was harvested and fixed in 10% buffered formalin. Specimens were taken from the mid-gastrocnemius muscle and then further processed, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E). H&E sections were evaluated by a pathologist. Morphometry (Sigma-Pro imaging program; Systat Software, Chicago, IL) was conducted to quantitate the area of damage.
Rotarod exercise
Animals were individually exercised on a rotarod (Economex; Columbus Instruments, Columbus, OH). Initially the speed of the rotarod was set at 12 rpm. If the animal was not able to maintain this speed and fell off the rotarod right away, the rotarod speed was immediately reduced to 8 rpm or further reduced to 6 rpm to determine the highest of these speeds at which the mouse could remain on the rotarod. Once an appropriate speed was found, animals were allowed to run continuously until they fell off. The run time and speed for each animal were recorded.
Treadmill exercise
Mice were exercised at 8–12 m/min for 10–30 min by running uphill at a 5° angle on a treadmill (IITC model 800; IITC Life Science, Woodland Hills, CA). An electric shock bar grid at the end of the tread delivered a mild shock to mice if they stopped running. The shock bar was customized to reduce the intensity of the electrical stimulus (maximum, 200 V) by adding a 100-Ω resistor to limit the current on the grid to a maximum of 2 mA.
Whole body tension
Muscle forces were measured as the amount of whole body tension force (WBT) produced by the mice when they tried to escape from a stimulation. The tail of the mouse was connected to a BIOPAC Systems (Goleta, CA) transducer with 4-0 silk thread (one end of the thread was tied to the tail and the other end was tied to the transducer). Each mouse was placed into a small tube constructed of a metal screen with a grid spacing of 2 mm. The length of the tube was 16 cm and the diameter was 3.5–4.5 cm. The tube was taped to a table. The mice entered the apparatus and exerted a small resting tension on the transducer. A forward movement could be reproducibly evoked by pinching the tail with a forceps. Each mouse remained in the tube for 5 min, during which 15–20 forward pulling tensions were evoked (with 15- to 20-sec intervals). The WBT was determined from the average of the five highest forward pulling tensions divided by body weight (millinewtons per gram). The WBT test was performed three times, once per week, before the HLV injections and three times afterward. For each mouse, the average of the three tests before gene delivery was compared with the average of the three tests afterward.
EBD injection procedure
A 5-μg/μl concentration of Evans blue dye (cat. no. E2129; Sigma-Aldrich, St. Louis, MO) prepared in physiological saline was injected intravenously through the tail vein by means a 1-cm3 syringe with a 30-gauge needle. Injections were performed 1 hr before or 20–30 min after exercising the mice. Injection after exercise was preferable because some mice were unable to run or died when injected before exercise. The volume to inject into each mouse was determined by the weight of the animal: 50 μl/10-g body weight. Mice were killed 24 hr after EBD injection except where otherwise indicated.
Creatine kinase measurement
Blood was collected for creatine kinase (CK) measurements by retro-orbital bleed after anesthetizing mice briefly with 1–2% isoflurane. CK levels were monitored with an Ektachem DT60 analyzer (Eastman Kodak, Rochester, NY).
Animal sacrifice and tissue preparation
Mice were anesthetized with 3 to 5% isoflurane and killed by cervical dislocation. The skin was then removed from the hind limb to expose the muscles. All muscles of the hind leg from above the quadriceps down to the ankle were divided into five groups: (1) the anterior group of muscles of the upper leg (includes the quadriceps, on average 20% of the leg muscle mass), (2) the posterior group of muscles of the upper leg (includes the biceps femoris, 26% of leg), (3) the medial group of muscles of the upper leg (includes the gracilis and adductor muscles, 24%), (4) the posterior group of muscles of the lower leg (includes the triceps surae: gastrocnemius, soleus, and plantaris, 20%), and (5) the anterior group of muscles of the lower leg (primarily the tibialis anterior and extensor digitorum longus, 9%). Muscle for cell counting was immediately frozen in Tissue-Tek O.C.T. compound (cat. no. 4583; Sakura Finetek, Torrance, CA) on liquid nitrogen and then stored at −80°C.
Immunohistochemistry and cell counting
Cryostat sections (8–10 μm) were cut and fixed in 2–4% formalin for 3–5 min. Dystrophin in the tissue sections was detected with a 1:100 dilution of rat anti-dystrophin polyclonal antibodies (generated by HLV injection of dystrophin-expressing pDNA) as the primary antibody and a 1:100 dilution of fluorescein isothiocyanate (FITC)-conjugated goat anti-rat IgG (cat. no. F6258; Sigma-Aldrich) for the secondary antibody. Some muscle sections were stained with a 1:100 dilution of mouse polyclonal antibodies against mouse dystrophin for 60 min and a 1:100 dilution of FITC-conjugated goat anti-mouse IgG as the secondary antibody (Fab specific, cat. no. F8771; Sigma-Aldrich) and incubated for 40 min. A Nikon Diaphot-TMD inverted microscope with an FITC filter was used (excitation, 480 ± 20 nm; emission, 535 ± 25 nm) to evaluate sections for dystrophin staining and to obtain electronic images, and a Texas Red filter was used for EBD-positive cell counting. Myofibers were counted in 10 or 11 microscope views across the diameter of the mid-section of each of the five groups of muscles in microscope views at × 160 magnification.
Immunoblotting
After tissue sections were taken for immunohistochemistry, the remaining muscle was evaluated by immunoblotting. Protein was extracted from mdx muscle of the hind limb by grinding the tissue with a mortar and pestle chilled with liquid nitrogen. Extraction buffer (7.5 mM Tris, 20% glycerol, 15% sodium dodecyl sulfate [SDS], 0.1 M dithiothreitol [DTT], and Complete protease inhibitor [cat. no. 11-697 498 001; Roche Diagnostics, Indianapolis, IN]) was added to the powdered muscle (20 μl/mg tissue) and incubated at ambient temperature for 10–15 min while inverting several times to mix, and then heated at 100°C for 3 min. This solution was passed 20 times through a 20-gauge needle to shear the DNA, and then centrifuged at 10,000 × g for 5 min. The supernatant was assayed for protein concentration, using an RC DC protein assay kit (Bio-Rad, Hercules, CA). Protein extracts for dystrophin standard were prepared from normal C57BL/6 and mdx4Cv or mdx-10ScSn mouse muscle. Various percentages of normal mouse protein (1–25%) were then combined with the appropriate amount of mdx mouse protein. The samples shown in some of the blots had an insufficient amount of SDS and DTT, resulting in some smearing of the dystrophin protein at the top of the gel. This problem was corrected by diluting samples in extraction buffer. Samples were electrophoresed in a 3–8% Tris-acetate SDS–polyacrylamide gel (Invitrogen, Carlsbad, CA), with 25 μg of total protein loaded per well. Proteins were transferred onto polyvinylidene difluoride (PVDF) at 40 V for 2 hr in NuPage transfer buffer (Invitrogen) with 0.1% SDS added. Primary antibody NCL-DYS1 (Novocastra Laboratories, Newcastle upon Tyne, UK) was diluted 100-fold. The affinity-purified, peroxidase-conjugated, goat anti-mouse IgG secondary antibody (Sigma-Aldrich) was diluted 1000-fold. Dystrophin was detected by enhanced chemiluminescence (ECL Plus Western blotting detection reagents; GE Healthcare Life Sciences, Piscataway, NJ) using Kodak BioMax MS film (Carestream Health, Rochester, NY). Subsequently, the membrane was stained with Ponceau S red (Fluka; Sigma-Aldrich) and the gel with Brilliant Blue G stain (Sigma-Aldrich) to verify that the proteins transferred evenly. For quantitation, Carestream Molecular Imaging software (Carestream Molecular Imaging, Woodbridge, CT) was used to generate a standard curve for each blot.
Calculations and statistical analysis
Weighted averages for EBD- or dystrophin-positive myofibers in the hind leg muscles were determined as follows: (∑A 1 M 1 + A 2 M 2 + A 3 M 3 + A 4 M 4 + A 5 M 5)/M t, where Ai is the average number of positive myofibers per microscope view in muscle group i and Mi is the average weight of that muscle group, and M t is the total weight of all limb muscles. Average values in the text and figures are given ± standard error. Protection from muscle damage was defined as the percentage of EBD+ myofibers in a muscle group from the control leg divided by the percentage of EBD+ myofibers in the contralateral muscle group of the dystrophin plasmid-injected leg. The decrease in EBD+ myofibers in each group of muscles from the dystrophin plasmid-injected leg relative to the control leg was determined as a percentage reduction, using the following formula: (% EBD+ myofibers in control muscle group − % EBD+ myofibers in dystrophin plasmid-injected contralateral muscle group)/% EBD+ myofibers in control muscle group. Statistical analyses were performed with Prism (GraphPad, San Diego, CA). The p values and correlations were determined with a two-tailed t test, unless otherwise indicated. To accommodate high variability between animals and the non-Gaussian distribution of EBD staining, the Wilcoxon signed rank t test was used to compare contralateral pairs of muscles for statistical analysis.
Results
The HLV delivery procedure was optimized for wild-type mice by using an injection volume that resulted in high levels of expression from an injected reporter gene plasmid, indicative of the efficiency of pDNA delivery, and low levels of serum creatine kinase (CK), an indicator of muscle damage. Optimal injection conditions are described in Materials and Methods. Data from 156 mice were combined to compare preinjection CK levels with those 1 to 5 days after the HLV injection (see Supplementary Fig. 1a at
mdx mice respond to their muscular dystrophy by developing hypertrophic muscles that are larger, relative to wild-type mice. The delivery procedure was then adapted to mdx mice by increasing the injection volume in proportion to the muscle weight and adjusting the injection rate so that the injection delivery procedure would take a similar amount of time in large and small mice (see Materials and Methods). Muscles of dystrophic animals are expected to be more fragile than normal muscle. Therefore, we examined the histology of mdx leg muscles 1 or 3 days after HLV injection of saline into one leg of each mouse. The injection caused no measurable increase in muscle damage (see Supplementary Fig. 1c). Consistent with the results of others, we measured areas of necrotic and regenerating myofibers comprising 2–6% of the muscle in the uninjected leg. These areas in the contralateral saline-injected legs were indistinguishable from the control legs.
Because of the muscular dystrophy, CK levels in mdx mice are higher than in normal mice. Instead of increasing after an HLV injection, however, CK levels in the mdx mice were lower 1 and 3 days postinjection (p < 0.0001, one-way ANOVA with Bonferroni multiple comparison test). CK levels decreased from an average of 5566 ± 344 U/liter before the injection to 4085 ± 292 U/liter 1 day later and to 3041 ± 302 U/liter on the third day (n = 31–81). By the fourth day, CK levels were statistically the same as they were before the injection. The HLV injection procedure in rodents requires minor surgery to expose the great saphenous vein of the hind limb in order to visualize insertion of the needle. The transient reduction in CK levels was likely the result of decreased activity as mice were recovering from the surgical procedure. Mice are typically active as soon as the anesthesia wears off and a possible reduction in activity was not actually measured.
Another factor that could potentially affect gene expression after pDNA delivery would be a difference in plasmid uptake by fast-twitch compared with slow-twitch myofibers. Dystrophin-expressing pDNA was delivered by HLV to mdx mice to determine whether fast and slow myofibers take up and express pDNA differentially after HLV injection (see Supplementary Materials and Methods at
Experimental details for animal studies are shown in Table 1 and are referred to by study number throughout this paper.
Abbreviations: EBD, Evans blue dye; N/A, not applicable.
Group size indicates number of animals except in study 1. Both legs from three mice per group were injected in study 1; for this study, n = number of legs.
Weight of animals is at time of injection.
When the right leg was given a different treatment than the left leg, the second plasmid is indicated.
The injected dose of pDNA is shown in micrograms when all animals received the same amount of pDNA. The injection volume for study 1 was adjusted for animal weight. The injection volume for studies 8, 9, and 10 was 1 ml. When the dose was scaled to animal weight, it is shown as the concentration of pDNA in the injection solution (μg/ml).
Gene delivery in young and old mdx mice
As with humans, the DMD disease phenotype becomes more severe in aged mdx mice. To determine whether animal age affects the efficiency of gene delivery to skeletal muscles, two strains of normal mice and two strains of mdx mice were each injected with the same dose of luciferase-expressing plasmid (Fig. 1a; Table 1, study 1). The mice varied in age from 1.5 to 14 months. The total amount of luciferase protein produced in the leg was similar in all strains and all ages (p = 0.37; Fig. 1a).
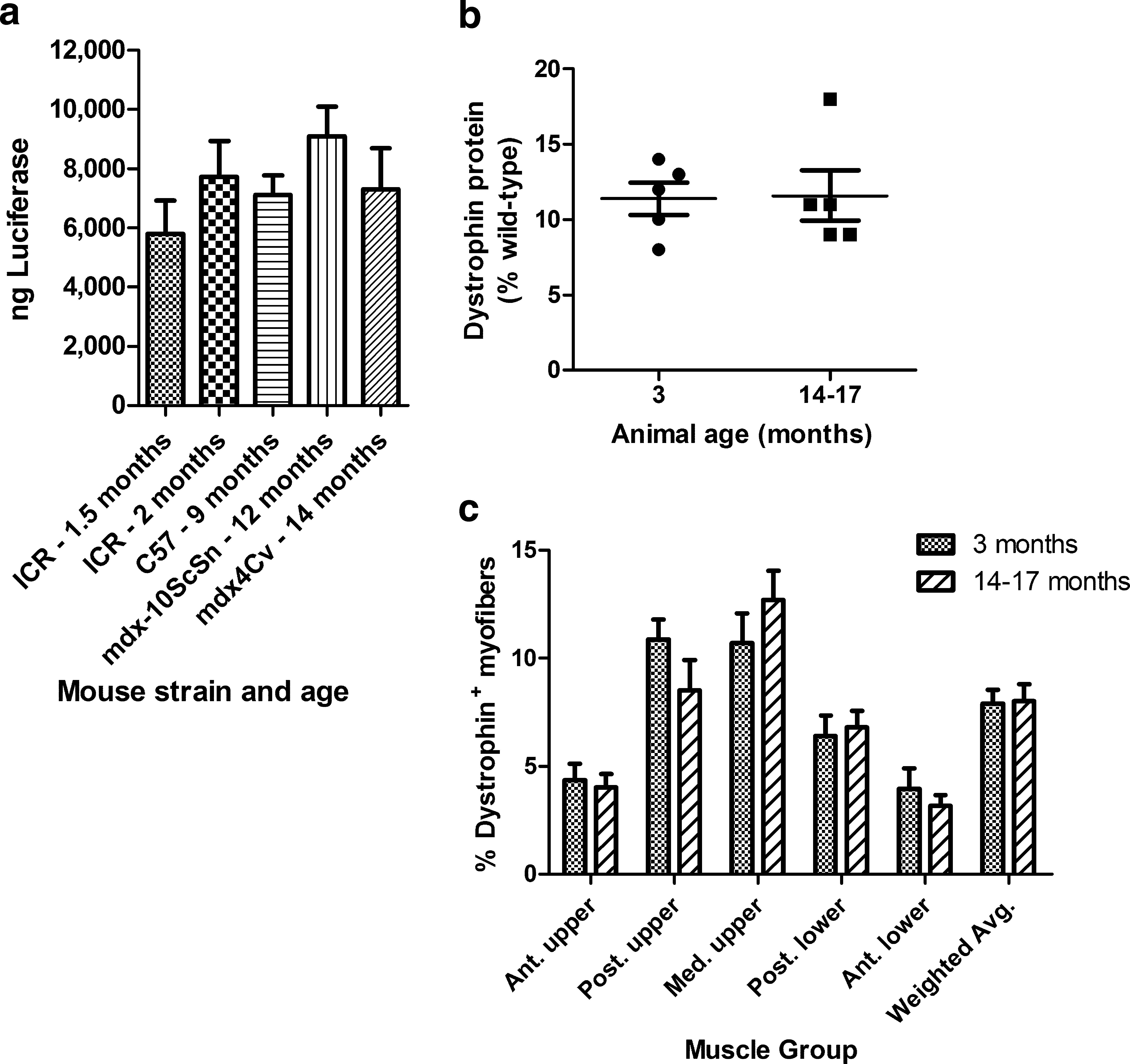
Gene delivery compared in strains of mice of varying ages. (
Dystrophin gene delivery was then compared in young adult and old mdx mice (Fig. 1b and c; Table 1, study 2). In this study all mice received two HLV injections in one hind leg with the same 300-μg/ml concentration of CMV promoter-driven mouse dystrophin expression plasmid CMV-HMGi-mDys. Injection volumes were adjusted to accommodate the larger size of the old mice, which weighed approximately twice as much as the young adults. The pDNA per target volume was fixed, so larger animals with a larger target muscle mass received more pDNA than did smaller animals. The younger mice produced 11.4 ± 1.1% dystrophin protein, relative to a wild-type mouse, and the aged mice produced 11.6 ± 1.7% dystrophin (p = 0.92; Fig. 1b). The percentages of myofibers that expressed dystrophin, an average of 8% in the whole leg, and their distribution within the legs were similar for both age groups (Fig. 1c). Gene delivery was equally effective in the aged and young adult mdx mice when the pDNA dose was adjusted for the increased muscle mass in the old mice. The 300-μg/ml concentration of pDNA was less than the optimal dose of approximately 500 μg/ml. A larger dose results in higher percentages of transfected myofibers in the anterior thigh and anterior lower leg muscles (our unpublished observation).
In conclusion, age does not affect the efficiency of pDNA gene delivery in the mdx mouse model.
Dystrophin expression from multiple gene deliveries
Using CMV-SV40i-mDys, we compared dystrophin expression in the hind leg muscles of mdx mice that received one or three HLV injections of this pDNA (Fig. 2a–c; Table 1, study 3). The three large groups of muscles that transfect best in rodent hind legs are the posterior and medial thigh muscles and the posterior lower leg muscles, which together comprise 71% of the leg muscles. For convenience, we refer to these as muscle groups 2, 3, and 4 (see Materials and Methods for details). The average percentages of dystrophin-expressing myofibers in these muscles increased from 5.7 ± 0.6% after one injection to 11.8 ± 0.9% after three injections. The amount of dystrophin protein produced in these muscles increased from 4 ± 1% after one injection, relative to a normal mouse, to 14 ± 1%.
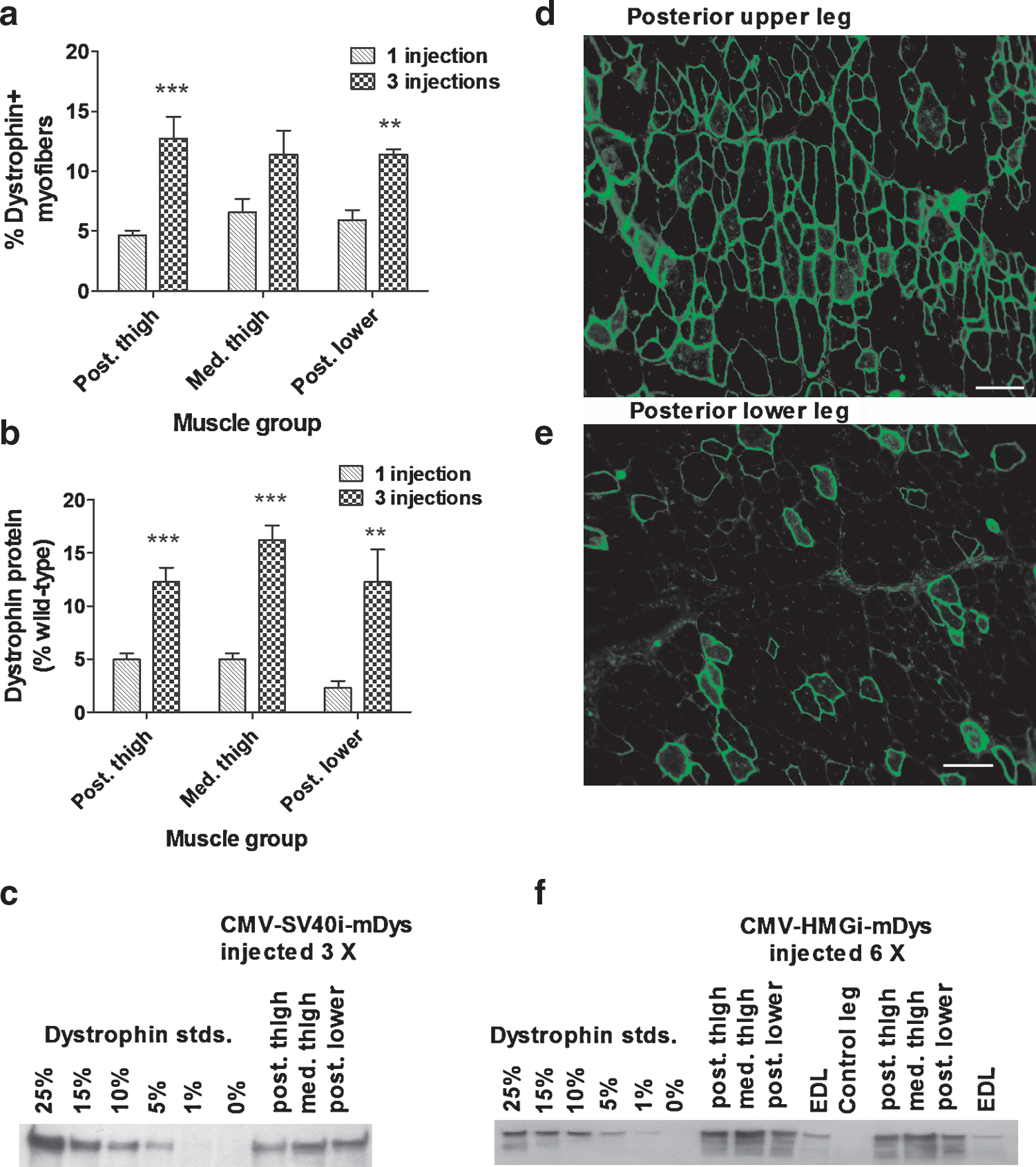
Dystrophin expression. (
In mdx mice that were injected six times with dystrophin-expressing pDNA, this time with the larger CMV-HMGi-mDys (Fig. 2d–f; Table 1, study 4), up to 51% of myofibers expressed dystrophin in some of the large muscles (Fig. 2d), although other muscles expressed less (Fig. 2e). The average percentages of expressing myofibers in muscle groups 2–4 was 13.3 ± 1.9%. More than one-third of these muscles produced at least 20% of the wild-type amount of dystrophin protein (n = 11 mice, 33 groups of muscles). These experiments show that the mice tolerate repeated injections well, and that multiple gene deliveries increase dystrophin expression.
Comparing introns in the plasmid constructs
The plasmids CMV-SV40i-mDys and CMV-HMGi-mDys differ only in the intron that they contain, which in both cases is positioned just upstream of the dystrophin cDNA. CMV-SV40i-mDys contains a 96-bp hybrid intron from simian virus 40 (SV40i). CMV-HMGi-mDys contains the 4141-bp human HMG1 intron (HMGi) and, as a result, is larger than the SV40i construct. The two CMV-mouse dystrophin plasmids were compared by using equal molar concentrations, one injection per mouse, and expression was evaluated 2–6 weeks after injection (Fig. 3; Table 1, study 5). Expression from both plasmids is stable during this period of time (data not shown). Delivery is less efficient with larger plasmids (Zhang et al., 2004). Despite this, the mice that were injected with CMV-HMGi-mDys had on average nearly 50% more Dys+ myofibers than those injected with CMV-SV40i-mDys (p = 0.0011). In addition to having more Dys+ myofibers, the mice injected with CMV-HMGi-mDys produced 2.3 times more dystrophin protein than those injected with the construct containing the smaller SV40i (p = 0.0003). The HMG1 intron was more effective for gene expression than the SV40 intron. The percentages of Dys+ myofibers increased from 4 to 6% after one injection when a higher, more optimal dose of CMV-SV40i-mDys (500 μg/ml) was used (compare Fig. 3 with Fig. 2a). However, even with the optimized dose of CMV-SV40i-mDys, this plasmid produced approximately half as much dystrophin protein as the HMGi-containing plasmid (p = 0.03, n = 6–12; compare Fig. 2b with Fig. 3).
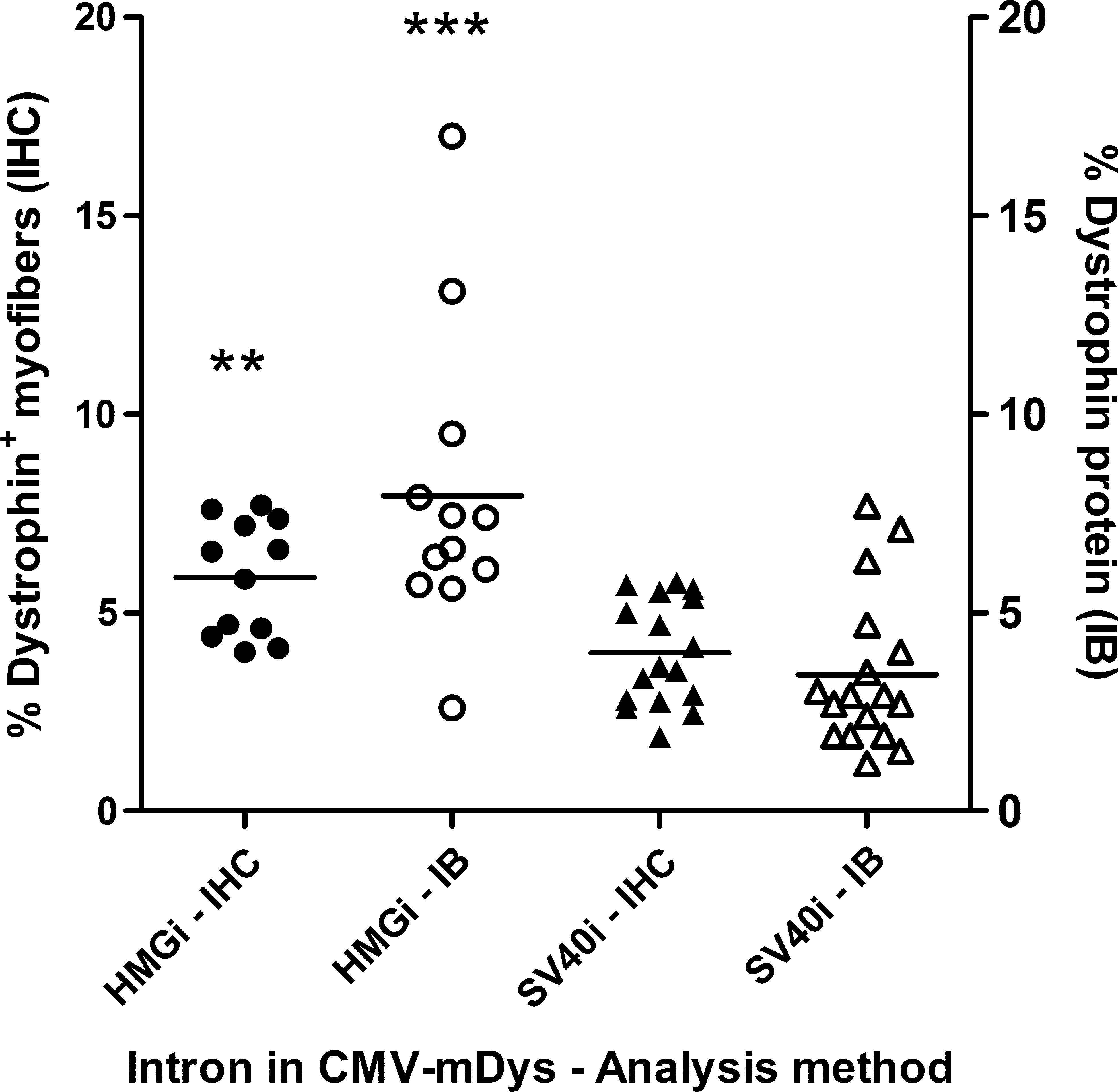
Comparing introns in CMV-mDys expression plasmids. mdx mice received one HLV injection of CMV-HMGi-mDys at 491 μg/ml (HMGi, n = 12; Table 1, studies 5-1 to 5-3) or of CMV-SV40i-mDys at 402 μg/ml (SV40i, n = 17; Table 1, studies 5-4 to 5-6), which were equal molar concentrations. The mice were killed 2–6 weeks after the injection and dystrophin expression was evaluated by immunohistochemistry (IHC) or immunoblotting (IB). Each data point represents the weighted average expression of dystrophin per mouse in muscle groups 2–4. Statistical analysis between groups was performed with a two-tailed t test (**p < 0.01, ***p < 0.001).
Stability of plasmid-driven dystrophin expression
The longevity of dystrophin expression from the CMV-HMGi-mDys plasmid was evaluated by injecting mdx mice six times with this pDNA and then killing them from 0.5 to 15 months after the last injection (Fig. 4). One group of mice was injected at the usual 2-week intervals and killed 2 weeks after the last gene transfer, which was 3 months after the first injection (Table 1, study 4). These mice were compared with groups that were injected with the pDNA over the course of 5 months and then killed 5 months (Table 1, study 6) or 15 months (Table 1, study 7) after the last injection. Those evaluated 5 months after the last injection were 14 months old at sacrifice and those evaluated 15 months after the last injection were 23 months old. The latter time point was nearly the end of the mouse's life span. The average percentages of Dys+ myofibers in the muscle groups that transfect well was the same in mice evaluated at the earliest time point and in those that had been injected over the course of 5 months and evaluated 5 months after the last injection (Fig. 4a). Both groups had an average of 13% Dys+ myofibers (p = 0.93) and 15–17% dystrophin protein in these muscles (p = 0.32).
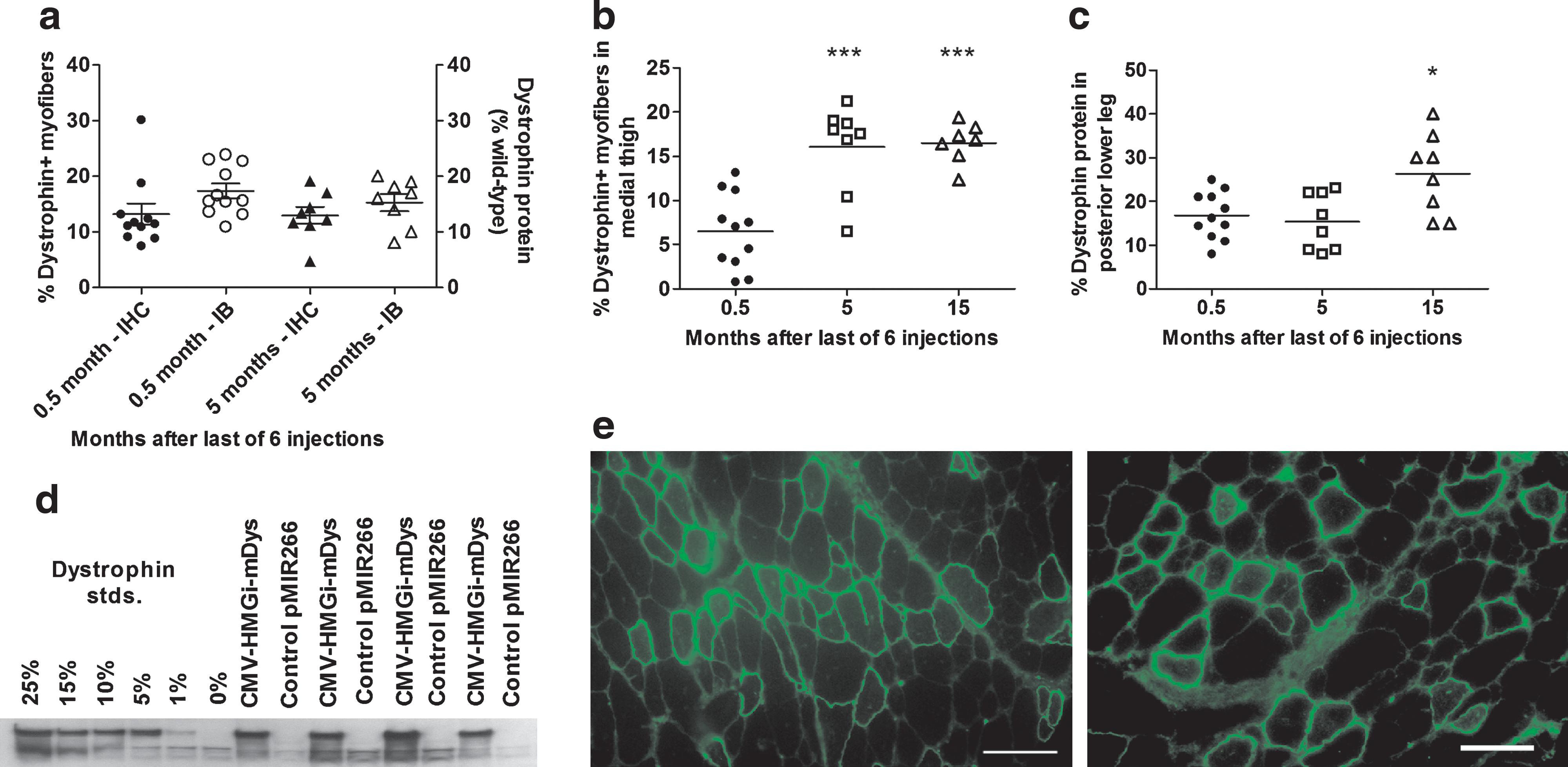
Longevity of dystrophin expression from plasmid constructs. mdx mice received six HLV injections of CMV-HMGi-mDys (491 μg/ml) and were evaluated for dystrophin expression at 0.5 months (Table 1, study 4), 5 months (Table 1, study 6), or 15 months (Table 1, study 7) after the last injection. (
The oldest mice, evaluated when they were 23 months old (Table 1, study 7), had an average of 16.5% Dys+ myofibers in their medial upper leg muscles, with higher percentages in some areas (Fig. 4b and e). This was a similar percentage as in the mice evaluated 5 months after the last gene transfer and 2.5 times more than in the mice killed at the earliest time point (p < 0.0001; Fig. 4b). The oldest group of mice produced an average of 26% of the normal amount of dystrophin protein in their posterior lower leg muscles at sacrifice, nearly 60% more than the mice that were evaluated at either the early or the intermediate time point (p = 0.008; Fig. 4c and d). Thus, dystrophin expression after six injections of CMV-HMGi-mDys in at least some groups of muscles was not only stable for the life of the animals, but even appeared to increase in the oldest animals.
Immune response to dystrophin
Most mdx mice injected with dystrophin-expressing pDNA begin to generate antibodies to the dystrophin 2 weeks after gene transfer, whether the plasmid is injected by intraarterial injection (Zhang et al., 2004) or by HLV injection (see Supplementary Table 1 at
To address the possibility of a myositis response to expression of a foreign dystrophin gene, mdx and wild-type mice and rats were injected with plasmids expressing human or mouse dystrophin or a reporter gene, or with nonexpressing control plasmids. We observed that some batches of pDNA caused muscle damage that was evident by histology as areas of regenerating myofibers 3–4 days after a large dose of pDNA was delivered by HLV injection (0.5 mg in a mouse or 2.7 mg in a rat), but this was not myositis and did not depend on expression of any gene product (C.I. Wooddell, V. Subbotin, M. Sebestyen, G. Zhang, J.B. Griffin, T. Huss, and J.A. Wolff, unpublished data). The apparent pDNA-associated toxicity was completely batch dependent and not due to expression of dystrophin or to the pDNA itself. The toxicity was unlikely to be due to endotoxin because some preparations containing as little as 0.2 EU of endotoxin per milligram caused muscle damage whereas other batches with comparable amounts or more caused no damage.
Muscle function studies
Four independent studies were performed to assess muscle function in mdx mice injected with a pDNA encoding murine dystrophin (Table 1, studies 6, 8, 10, and 11). A fifth study (Table 1, study 9) was performed with the same mice used for study 8. The whole body tension test was performed first to evaluate leg muscle strength in young adult mdx mice that were naive or that received three HLV injections in both hind legs with 491 μg of either dystrophin-expressing CMV-HMGi-mDys or control pUC19 (Table 1, study 8; Fig. 5). These were compared with wild-type mice. In this kind of evaluation, both hind legs are injected with the same pDNA. Animal-to-animal variation is high, so the mice were tested before and after the treatment (Fig. 5a). Overall, mdx mice and wild-type mice had increased pulling strength at the later time point (after HLV injections in the mice that were injected), likely because both strains grew larger during the 6 weeks in which injections were performed. The pulling force was normalized to animal weight to allow comparison of animals that differed in size. mdx mice were randomly divided into treatment groups. Although the control group of mice that later received pUC19 exerted more pulling force before the injections than the other groups of mdx mice, their normalized strength did not increase significantly after the injections (Fig. 5b; p = 0.844; Table 1, study 8-2), nor did that of the uninjected mdx mice (p = 0.998; Table 1, study 8-3). The dystrophin plasmid (Dys)-injected mdx mice, however, pulled away with 27% more force after gene delivery, compared with the force measured before gene delivery (Fig. 5b; p = 0.0129). Nine of the 10 mice had greater pulling strength after gene delivery. Before injections, the pulling force of the mdx mice that later received the Dys pDNA was 66% that of the wild-type mice. After three Dys injections, their pulling force was 88% that of the wild-type mice (Fig. 5a). The pulling force of the uninjected mdx mice was initially 63% that of wild-type mice and at the later time point was nearly the same, 68% (Fig. 5a). The normalized strength of the wild-type mice decreased slightly at the later time point, but the difference was not significant (p = 0.34) and was mostly due to an increase in weight.
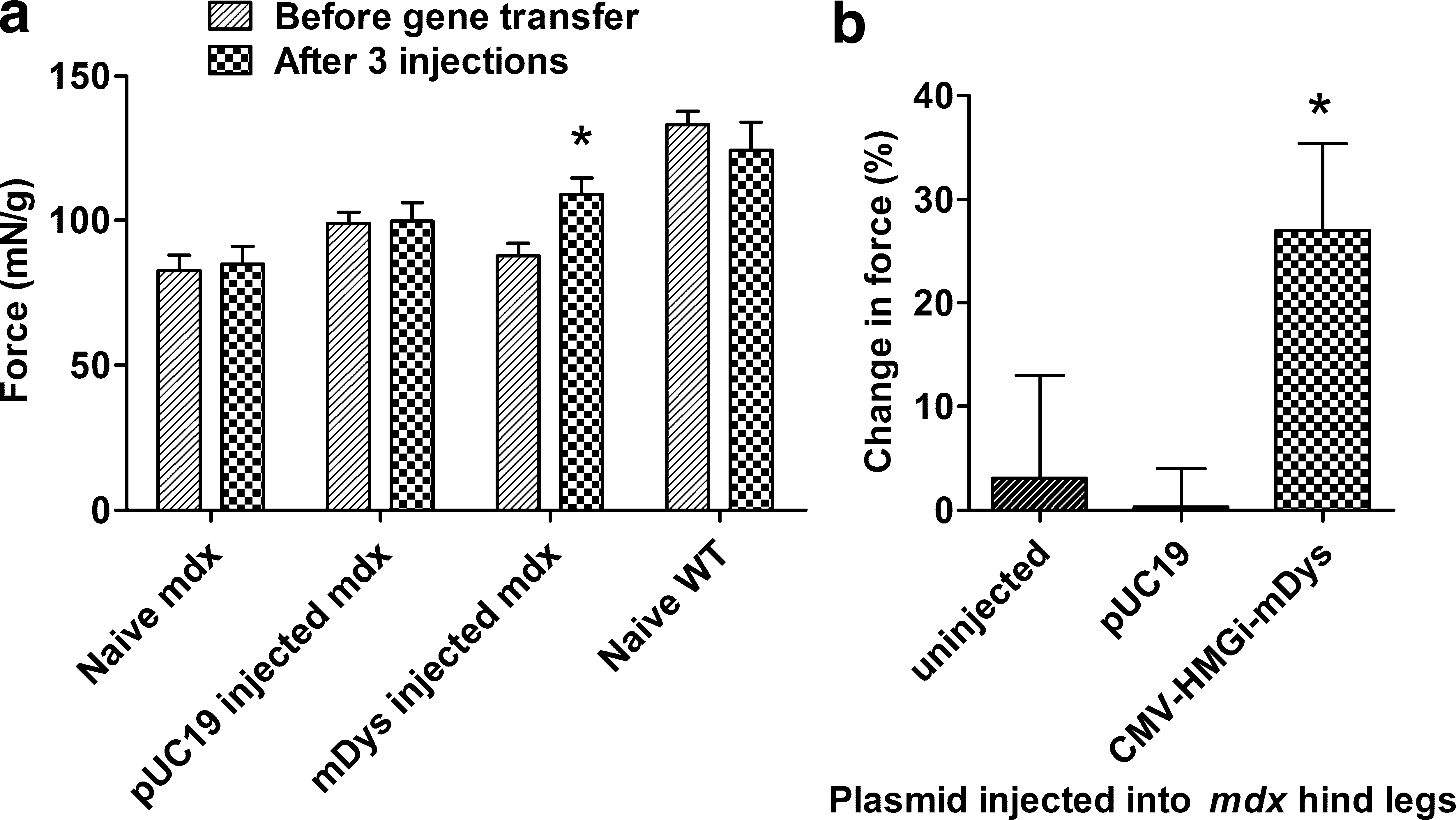
Muscle strength testing. Groups of mdx mice were uninjected or injected three times in each leg with 491 μg of empty vector (pUC19) or CMV-HMGi-mDys (Table 1, study 8). Wild-type (WT) C57BL/6 mice were uninjected. Before gene delivery and again beginning 2 weeks after the last delivery, the mice were tested for whole body pulling force, shown as (
Evans blue dye (EBD) was used to measure the degree of protection afforded the dystrophic leg muscles that received dystrophin-expressing pDNA (Figs. 6 and 7). Exercise protocols were used to challenge the hind leg muscles in these studies and cause damage in myofibers that were not protected by adequate dystrophin expression. Damaged myofibers are permeable to the EBD–albumin conjugate as well as to serum proteins (McGeachie et al., 1993; Straub et al., 1997; Wooddell et al., 2009).
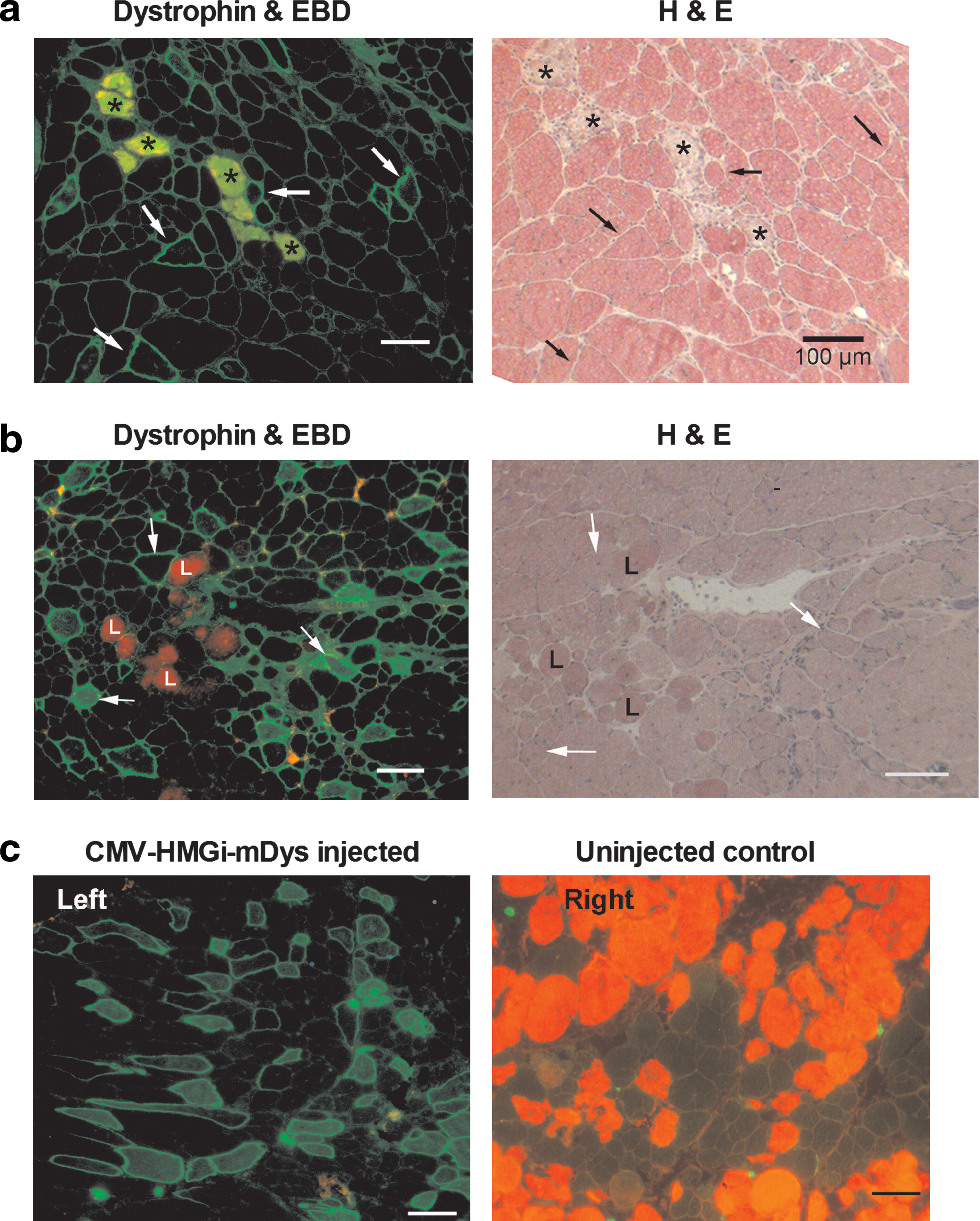
Evans blue studies in mdx mice. (
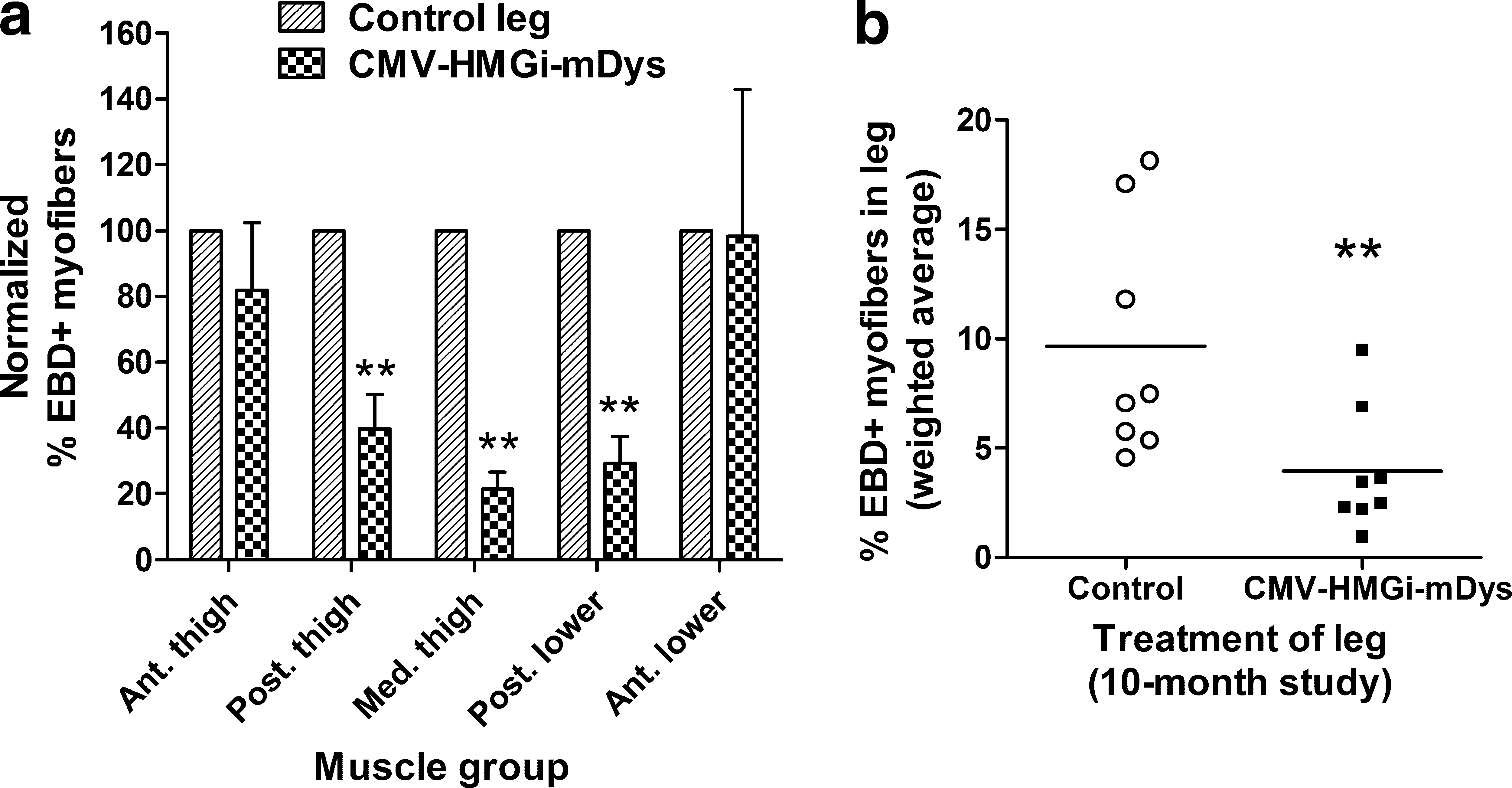
Reduction in Evans blue staining. (
The same mice that were previously used to perform the whole body tension study thereafter received three additional HLV injections of the same plasmids (Table 1, studies 9-1 and 9-2). When these mice were 18 months old, they were exercised on a treadmill, injected with EBD, and subsequently evaluated for muscle damage by counting EBD-positive myofibers in the hind legs. The average number of myofibers expressing dystrophin in large muscle groups 2–4 was only 4.7 ± 0.5%. The pDNA dose in these animals had not been adjusted for their increasing weight as they grew older. Even so, the Dys-injected mice (Table 1, study 9-1) had 73% fewer EBD+ myofibers in their medial thigh muscles than the age-matched pUC19-injected mdx mice (p = 0.0015; Table 1, study 9-2) and 60% fewer than age-matched uninjected mdx mice (p = 0.0026; Table 1, study 9-3). As shown in Fig. 6, dystrophin-expressing myofibers excluded EBD. Dystrophin expression was not evident in either the myofibers that were darkly stained with EBD or in those that stained only lightly (Fig. 6a and b). The darkly stained EBD+ myofibers were clearly damaged, as can be seen by histological staining. Serum proteins including IgG antibodies leaked into these myofibers. These endogenous IgG antibodies were detected by immunohistochemistry with an FITC-conjugated anti-mouse IgG secondary antibody. EBD–albumin also leaked into the damaged myofibers. Autofluorescing EBD appears red. Overlapping FITC and EBD signals appear yellow-green in the myofibers that stained darkly for EBD. The secondary antibody also labeled the anti-dystrophin mouse polyclonal antibodies, seen as staining around the periphery of the myofibers. Lightly EBD-stained myofibers, although leaky, appeared to be morphologically more normal (Fig. 6b). We characterized dark and light EBD staining in mdx mice and found that darkly stained myofibers increased after a vigorous exercise regimen, but not the approximately 1% of myofibers that stained lightly with EBD (Wooddell et al., 2009).
EBD staining is highly variable between animals and even in the anatomical location within the legs of individual mdx mice (McGeachie et al., 1993; Straub et al., 1997; Wooddell et al., 2009). In subsequent short-term and long-term gene therapy studies, therefore, we used one hind leg as an internal control for the dystrophin plasmid therapeutic treatment given to the other leg (Table 1, studies 6, 10, and 11). The mdx mice in these experiments were injected six times in one leg with CMV-HMGi-mDys or CMV-SV40i-mDys.
The first of the EBD studies that used one leg as an internal control for dystrophin pDNA injections in the other was a relatively short-term evaluation in which young adult mdx-10ScSn mice received six HLV injections of CMV-HMGi-mDys in the left leg, 2 weeks between injections (Table 1, study 10). Three to 4 months after the first gene transfer, the mice were exercised on a rotarod, injected with EBD, and killed to quantitate dystrophin expression and the percentages of EBD-stained myofibers in muscles throughout both legs. The average percentage of dystrophin-expressing myofibers in muscle groups 2–4 was 12.4 ± 1.6% and the average dystrophin protein expression in these muscles was 20 ± 3%. Dystrophin expression in each group of muscles is shown in Supplementary Fig. 3a and b at
The rotarod exercise regimen was effective for challenging only some of the muscles of the mice in this short-term study, at least in part because the mice exercised inconsistently and their serum CK levels increased on average only 5-fold. The degree of protection that is possible depends not only on the reduction in EBD staining of the Dys-injected leg, but also on sufficient levels of EBD staining in the control samples. When the number of EBD+ myofibers in the control leg is low, such as when the muscles are not sufficiently challenged by the exercise regimen, it is difficult to detect any potential protective effect from dystrophin expression in the other leg. Microscopy images of the posterior thigh muscles from both legs of one of the mice that exercised well show that dystrophin-expressing myofibers excluded EBD, and EBD staining throughout this group of muscles was reduced by 71% in the Dys-injected leg, compared with the same group of muscles in the contralateral control leg (Fig. 6c and data not shown). The whole Dys-injected limbs in this group of mice had on average 50% fewer EBD+ myofibers than their contralateral control legs (data not shown).
The second study using an internal control leg was a long-term study. Young adult mdx4Cv mice were given six HLV injections of CMV-HMGi-mDys over a 5-month period and the doses were adjusted for the increasing weight of the animals (Fig. 7; Table 1, study 6). The 491-μg/ml dose was equal to 2 × 1013 plasmids/ml for CMV-HMGi-mDys and 2 × 1014 plasmids/ml for the smaller control plasmid. The mice in this long-term study were evaluated when they were 14–15 months old, which was 10 months after the first gene delivery and 5 months after the last. Mice were exercised on a treadmill, resulting in a 48 ± 8-fold increase in serum CK levels that confirmed muscles had been challenged in all animals. The Dys-injected legs had levels of dystrophin expression similar to those in the short-term study, an average of 12.9 ± 1.5% dystrophin-expressing myofibers and 18 ± 2% dystrophin protein in muscle groups 2–4 (see Supplementary Fig. 3c and d for dystrophin expression data). The control legs had an average of 0.06% revertant myofibers.
For each animal and each group of muscles, the percentages of EBD+ myofibers in the Dys-injected legs were compared with their contralateral control muscles (Fig. 7). Compared with their controls, the Dys-injected legs had 4.9-fold fewer damaged (dark EBD+) myofibers in the medial thigh muscles, 4.0-fold fewer in the posterior lower leg, and 2.5-fold fewer in the posterior thigh (Fig. 7a). The two groups of anterior leg muscles that were transfected least well showed no significant improvement. In the whole Dys-injected limbs, all groups of muscle included, there were an average of 2.5-fold fewer EBD+ myofibers than in the control legs, a 58% reduction in the Dys-injected compared with the control legs (Fig. 7b).
A third independent long-term study using an internal control leg confirmed that the reduction in EBD staining depended on the level of dystrophin expression (Table 1, study 11). A group of mdx mice was injected six times in one leg with a suboptimal dose of CMV-SV40i-mDys, which expresses less dystrophin than CMV-HMGi-mDys. In addition, this pDNA preparation caused some batch-dependent muscle toxicity. The injections were performed over a period of 5 months and the mice were evaluated 5 months after the last gene transfer. Only 4.4% of the myofibers in muscle groups 2–4 expressed dystrophin, and these muscles contained 5% of the normal amount of dystrophin protein (data not shown). Although this amount of dystrophin expression protected myofibers less than in the previous studies, there was a 46% reduction in EBD+ myofibers in the dystrophin pDNA-injected legs compared with their contralateral controls (p = 0.016).
In summary, four independent studies indicated that pDNA encoding the full-length dystrophin gene delivered by HLV injections protected leg muscles from exercise-induced muscle damage. The protection was as high as nearly 5-fold in muscles that expressed the most dystrophin, an average of 16% dystrophin-positive myofibers and an average of 20% of the wild-type amount of dystrophin protein. Muscles that had as few as 4% dystrophin-expressing myofibers were afforded some protection, but much less than in the muscles with higher expression.
Discussion
Studies with transgenic mdx mice show substantial reduction in the dystrophic pathology of mice expressing 20% of the normal level of full-length dystrophin (Phelps et al., 1995; Wells et al., 1995). Although not fully reversing the dystrophic phenotype, lower levels of expression also reduced the dystrophic pathology. Partial improvement in the dystrophic phenotype due to low levels of dystrophin expression was validated in the mdx3Cv mouse strain, which produces approximately 5% of the normal level of almost full-length dystrophin (Li et al., 2008). In this case the mice produce a uniform low level of expression. They showed improved muscle strength, compared with the mdx4Cv strain, but similar muscle pathology such as central nuclei, variable myofiber size, macrophage infiltration, and sarcolemmal damage, the latter being measured by EBD uptake. Thus, protection of the myofibers from exercise-induced damage appears to require more than 5% dystrophin per myofiber.
Viral vectors encoding mini- or microdystrophin genes are under consideration for the treatment of muscular dystrophy (Rodino-Klapac et al., 2007), but pDNA therapy has the advantage of being amenable to repeated treatments that provide additive amounts of dystrophin expression. The ability to perform repeated gene deliveries seems especially important for a chronic disease. It may also be a consideration that while truncated forms of dystrophin improve function in mdx mice, they may restore only partial function in humans. Adeno-associated viral vectors have the capacity for truncated forms of the dystrophin gene, but not the full-length gene. Full-length dystrophin protein alleviates dystrophic symptoms at a lower dose than is needed for alleviation of symptoms by minidystrophin expression. Another advantage of naked pDNA is that it is easily produced and relatively inexpensive, whereas large-scale production of viral vectors is much more challenging and costly.
Although mdx mice have been reported to generate humoral and cytotoxic immune responses to human dystrophin expressed from intramuscularly injected pDNA (Braun et al., 2000), we and others have not noted any cytotoxic response to the expression of full-length mouse dystrophin (Ferrer et al., 2000; Zhang et al., 2004). We previously observed an antibody response in most mdx mice injected with full-length mouse dystrophin plasmid, whether expressed from the viral CMV promoter or the muscle-specific desmin promoter (Zhang et al., 2004). Such a response was not previously observed after intramuscular injection of a much smaller dose of mouse dystrophin pDNA (Ferrer et al., 2000). Even though anti-dystrophin antibodies were produced after whole limb hydrodynamic injection, we show in this study that dystrophin was stably expressed for the lifetime of the mouse. Any immune response that occurred in the mice did not prevent long-term expression of dystrophin at the doses that were used.
The full-length dystrophin gene was delivered by intramuscular injection into DMD and Becker patients in a phase 1 trial and was shown to have an excellent safety profile with no adverse effects (Romero et al., 2004). The plasmid TG5001 used in that trial was the human equivalent of the CMV-HMGi-mDys that gave substantial protection from myofiber damage to mdx mice in our studies. Intramuscular delivery provided only modest dystrophin expression. Our studies suggest that dystrophin expression could be substantially increased by HLV delivery of the dystrophin-expressing pDNA.
Hydrodynamic limb vein injection is a robust procedure for delivering a therapeutic gene to the skeletal muscles of the limbs. In primates the delivery procedure requires no surgery, as injections are transcutaneous, and in rodents only minor surgery is required to access a surface vein. Gene delivery was as efficient in dystrophic mice as in wild-type mice, and as effective in aged mice as in young ones, as long as the dose of pDNA was adjusted for the increased leg mass of the older animals. The HLV gene delivery procedure is tolerated well by the dystrophic mice and repeat injections can be performed over an extended period of time to increase the number of expressing myofibers, as well as the overall amount of gene product.
Large plasmids like those expressing the full-length dystrophin gene transfect myofibers less efficiently than smaller plasmids delivered by HLV injection (Zhang et al., 2004). In wild-type C57BL/6 mice, an average of 24–27% of myofibers in the three large groups of hind leg muscles expressed β-galactosidase after just one HLV injection of 100 μg of CMV-LacZ (data not shown). A similar experiment in rats showed that up to 40% of the myofibers in some large muscle groups were transfected by a single HLV injection (Hagstrom et al., 2004; and our unpublished data). A single HLV injection of an optimal dose of the large dystrophin plasmids transfected 4–8% of the myofibers throughout the injected limb, and multiple injections increased the amount of expression. HLV injection results in a fairly even distribution of dystrophin-expressing myofibers scattered throughout most muscles and high levels of expression in the myofibers that are visibly transfected. Dystrophin protein levels increased additively with multiple injections. The percentages of transfected myofibers increased with multiple injections in a manner that suggested at least some of the myofibers transfected by the initial injection were also transfected by subsequent injections.
Our previous study demonstrated that full-length dystrophin expressed from pDNA prevents myofiber turnover (Danko et al., 1993). In the current study, dystrophin expression from the CMV-driven dystrophin plasmid containing the HMG1 intron was stable for the life of the mice and provided significant benefit to the limbs. An improvement in muscle strength was measured by whole body tension, also called the escape test, in mdx mice that received the dystrophin-expressing pDNA. Using Evans blue dye, we then analyzed in detail the protective effect of dystrophin plasmid delivery throughout the hind legs. Large groups of muscles in the dystrophin plasmid-injected legs had 4- to 5-fold less damaged muscle than the contralateral muscles of the control legs, and whereas the vast majority of muscles in the control legs clearly had damaged muscles, most of the muscles in the dystrophin plasmid-injected legs had only low amounts of myofiber damage. This amount of protection resulted from dystrophin expression in just 11–16% of the myofibers. Significant muscle protection from less than 20% dystrophin-expressing myofibers suggests that there is a strong bystander effect. Myofibers that were transfected appear to have taken in sufficient pDNA to express physiological amounts of dystrophin, and these dystrophin-positive myofibers were distributed fairly evenly throughout a group of leg muscles. It appears that the high-expressing myofibers may serve as flexible anchors, so to speak, stabilizing much of the muscle.
The degree of protection measured in young adult mice evaluated soon after the last gene delivery was similar to that in aged mice injected over an extended period of time and evaluated when they were old. A significant protective effect could be measured in more areas of the legs of older mice compared with the younger ones in the short-term study, but this was likely due to the more consistent exercise regimen of treadmill running and the higher biological consistency between contralateral groups of muscles in old mice (Wooddell et al., 2009).
The amount of dystrophin expressed in aged mice appeared to be higher than that expressed in younger animals. Given that mdx mice have a decreased ability to regenerate muscle as they age, the higher dystrophin expression in old mice suggests that the myofibers that expressed dystrophin may have had a selective advantage. This phenomenon resembles the selective pressure that results in genetic normalization of dystrophin expression in DMD carriers with skewed X-inactivation (Pegoraro et al., 1995). In essence, the dystrophin-negative myofibers are lost by necrosis, resulting in a higher proportion of muscle that is positive for dystrophin expression.
There is some debate as to whether stable, long-term expression of pDNA in mammalian cells requires removal of bacterial sequences and CpG dinucleotides from the plasmid vector (McMahon et al., 1998; Wells et al., 1998; Darquet et al., 1999) or if a scaffold/matrix attachment region (S/MAR) and tissue-specific promoter are required to prevent transgene silencing (Argyros et al., 2008). We and others have demonstrated high and stable levels of long-term expression in the liver (Miao et al., 2000; Wooddell et al., 2005, 2008) and in muscle (Sebestyen et al., 2007) from plasmids containing a bacterial backbone including the ColE1 origin of replication and devoid of any S/MAR element. A tissue-specific promoter and enhancer/locus control element were key in some of these studies, but were not necessarily required (Yew et al., 2001). In most of these reports the intron contributed greatly to the level of expression and its stability, as the HMG1 intron appears to do in the dystrophin expression plasmid. Although the CMV-HMGi-mDys plasmid construct does contain a bacterial plasmid backbone, it is also composed of approximately 15.5 kilobase pairs from the dystrophin gene that have a low CpG dinucleotide content. We cannot rule out the possibility that the low CpG content contributed to the stability of expression, or that expression could be increased by removal of CpG motifs in the vector backbone.
An inflammatory response or muscle damage has been reported as being associated with pDNA delivery to muscle by either intramuscular injection or electroporation (McMahon et al., 1998; Wells et al., 1998; Hartikka et al., 2001; Durieux et al., 2004), and in some cases was attributed to unmethylated CpG motifs in the bacterially derived pDNA. We, too, have observed damaged muscle in wild-type rodents within a few days of pDNA delivery. However, this response was completely dependent on the pDNA preparation, not the sequence of the pDNA. Some pDNA preparations caused significant amounts of muscle damage, whereas other preparations of the same plasmids caused no detectable damage at even large doses (C.I. Wooddell, V. Subbotin, M. Sebestyen, G. Zhang, J.B. Griffin, T. Huss, and J.A. Wolff, unpublished data). Although further studies are needed to determine the toxic contaminant in some pDNA batches, preparations can be produced without this component.
Translation of full-length dystrophin gene therapy from mice to humans would likely benefit by improvements to the vector. The CMV promoter provides long-term expression in mice, but plasmid expression in primate muscle was more stable when the muscle creatine kinase promoter was used (Sebestyen et al., 2007). A muscle-specific promoter is also expected to be safer because it is less likely to express the transgene in antigen-presenting cells. The large HMG intron was more effective for expression than the small SV40 intron, but the larger plasmid size makes its delivery less efficient. Large introns can be internally deleted to reduce the size while maintaining the beneficial effects for expression (Miao et al., 2000; Wooddell et al., 2008). The size of the vector and the bacterial sequences could be reduced further by using a minicircle construct (Darquet et al., 1999; Chen et al., 2005). These changes are expected to improve the efficiency of pDNA delivery and expression in mammalian muscles, allowing for fewer injections to obtain the desired level of dystrophin expression.
In conclusion, our studies demonstrate improvement in muscle function after HLV-mediated dystrophin plasmid delivery in the mdx mouse model and provide support for the use of this approach in human clinical trials.
Footnotes
Acknowledgments
The authors thank Ying-hua Bian and Jessica Connor (University of Wisconsin-Madison) and Tracie Milarch, Mark Noble, and Mavis Eldridge (Roche Madison) for technical assistance with animals, Vladimir Subbotin for histological evaluations, Magdolna Sebestyén for critical discussions regarding plasmid DNA quality evaluation, and James Ludtke for assistance with images (all from Roche Madison); Tom Delahanty (IITC) for customization of the treadmill; and Jens Eickhoff and Michael Evans (University of Wisconsin-Madison) for statistical analysis. Funding was provided by the Association Française contre les Myopathies.
Author Disclosure Statement
G. Zhang, C.I. Wooddell, J.O. Hegge, J.B. Griffin, and J.A. Wolff are employed by Roche Madison, which owns patents for the hydrodynamic limb vein delivery procedure. T. Huss is employed by Transgene, which conducted a phase I clinical trial for MyoDys plasmid delivery by intramuscular injection.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
