Abstract
The COVID-19 pandemic has revealed the need for nations to prepare more effectively for emerging infectious diseases. Preparing for these threats requires a multifaceted approach that includes assessing pathogen threat, building flexible capabilities for rapid medical countermeasure (MCM) development, and exercising, maintaining, and improving those response capabilities. The Biomedical Advanced Research and Development Authority (BARDA) promotes the advanced development of MCMs in response to natural and manmade threats. In this article, the authors describe a BARDA threat assessment tool that, in conjunction with other factors, facilitates portfolio management and decisionmaking related to the advanced development of MCMs for emerging infectious diseases. The tool was designed to use readily accessible public data and information related to the threat posed by pathogens. It can be adjusted to modify the specific areas of interest (eg, removal/addition of parameters) to facilitate cross-pathogen analysis and can be updated to reflect changes (eg, new outbreak) that may impact the overall threat assessment. Finally, assessment of certain pathogens is included.
Introduction
T
EID pathogens evolve rapidly through mutations, genetic rearrangements, and adaptation to new hosts. They are often zoonotic in nature, spilling over from infected animals as humans encroach on their habitats.2,3 They can be transmitted from animal to human, human to human, and/or by vectors. Globalization, increased travel, urbanization, poverty, and population growth are all factors that can lead to the emergence of novel pathogens. These factors (and others) make it impossible to predict which known, or currently unknown, pathogen might cause the next outbreak or pandemic. Further, with over 300 novel pathogens having emerged in the last century alone, 4 developing MCMs for each is not feasible. Therefore, BARDA has pivoted from the “1 bug, 1 drug” paradigm to a more flexible and sustainable preparedness and response posture. 5 BARDA’s EID preparedness efforts have taken a more pathogen-agnostic approach, through investments in robust and rapid platforms instead of pathogen-specific MCMs. However, FDA approval of specific MCMs is a necessary and important validation step to ensure that an MCM is safe and effective for its intended use, can be marketed in the United States, and that previous findings can be leveraged in new or future FDA applications. 6 Further, the fastest response occurs when an already licensed product can be immediately distributed in response to an outbreak. 7 Therefore, identifying specific pathogens to target for MCM development is central to BARDA’s preparedness and response strategy.
Proactive investment in EID MCMs is essential to national security, as EIDs pose a threat not only in terms of their devastating effects on public health, economic development and output, and defense capability, but they can also be exploited in biowarfare scenarios. 8 For example, some pathogens possess characteristics that make them useful as bioweapons, such as stability in aerosols for easy dispersal and human-to-human transmission, low infective dose resulting in high morbidity and mortality rates, and ability to be produced easily and in large quantities. 9 By harmonizing EID preparedness with BARDA’s broader efforts to address CBRN threats, BARDA’s approach ensures that adaptable, scalable solutions are ready to protect public health across a spectrum of evolving dangers.
Several public health entities have taken on the challenge of prioritizing EIDs by assessing the threat they pose to public health, developing research and development roadmaps, and outlining developmental and regulatory pathways for the approval of MCMs.10-15 In each of the assessments, the priorities and goals of the respective organizations were considered. These efforts serve to not only assess risks to public health and raise public awareness but also to prioritize research agendas, establish global collaborations, and enhance surveillance, laboratory capacity, and public health infrastructure. Lastly, they inform public health policy and resource allocations throughout the world. However, many of the assessment tools consider attributes other than just pathogen threat. BARDA’s portfolio process relies on the ability to assess and understand both the pathogen threat independently and in the context of other criteria (eg, US government requirements, clinical/regulatory strategy, manufacturing platforms) to support decisionmaking, thus requiring development of a specific pathogen assessment tool.
This article summarizes the tool developed by BARDA to assess pathogen threat. The tool was designed to include objective metrics that are important to BARDA’s mission and to assess a defined list of pathogens, in alignment with the BARDA Strategic Plan: 2022-2026 16 and language included in the congressional appropriations law for fiscal year 2024 that called on BARDA to support “expeditious development of MCMs against priority viral families, including those effective against novel pathogens with pandemic potential.” 17 This tool, along with others in development that focus on MCM development feasibility, will serve as a roadmap for MCM developers to better understand how BARDA will prioritize future MCM investments in known and unknown (ie, Disease X) pathogens of interest. The tool can be modified, as needed, to (1) reassess pathogens based on incoming data from new cases or outbreaks, and/or (2) add or remove parameters to accommodate changes in the intent of the tool (eg, to include nonviral pathogens) or in BARDA’s strategy.
Methods
The threat assessment described here focuses on viral attributes that are central to BARDA’s mission to develop MCMs that address the public health and medical consequences of threats such as EIDs. The assessment incorporated 7 parameters: transmissibility, case fatality rate, global outbreak size, global outbreak frequency, US outbreak frequency, disease severity, and duration of disease. Figure 1 shows the rating scale that was developed to assess each virus. A scale of either 0 to 5 or 1 to 5 was used to rate the 7 parameters with respect to each virus, with a score of 5 indicating the greatest impact and a score of 1 or 0 indicating low or no impact, respectively. For the purposes of this analysis, each parameter was weighted equally. Furthermore, within each parameter, the ratings were normalized to a 5-point scale to avoid implicit weighting. Figure 1 also shows the heatmap that was developed to visually represent the scores determined for each parameter assessed.
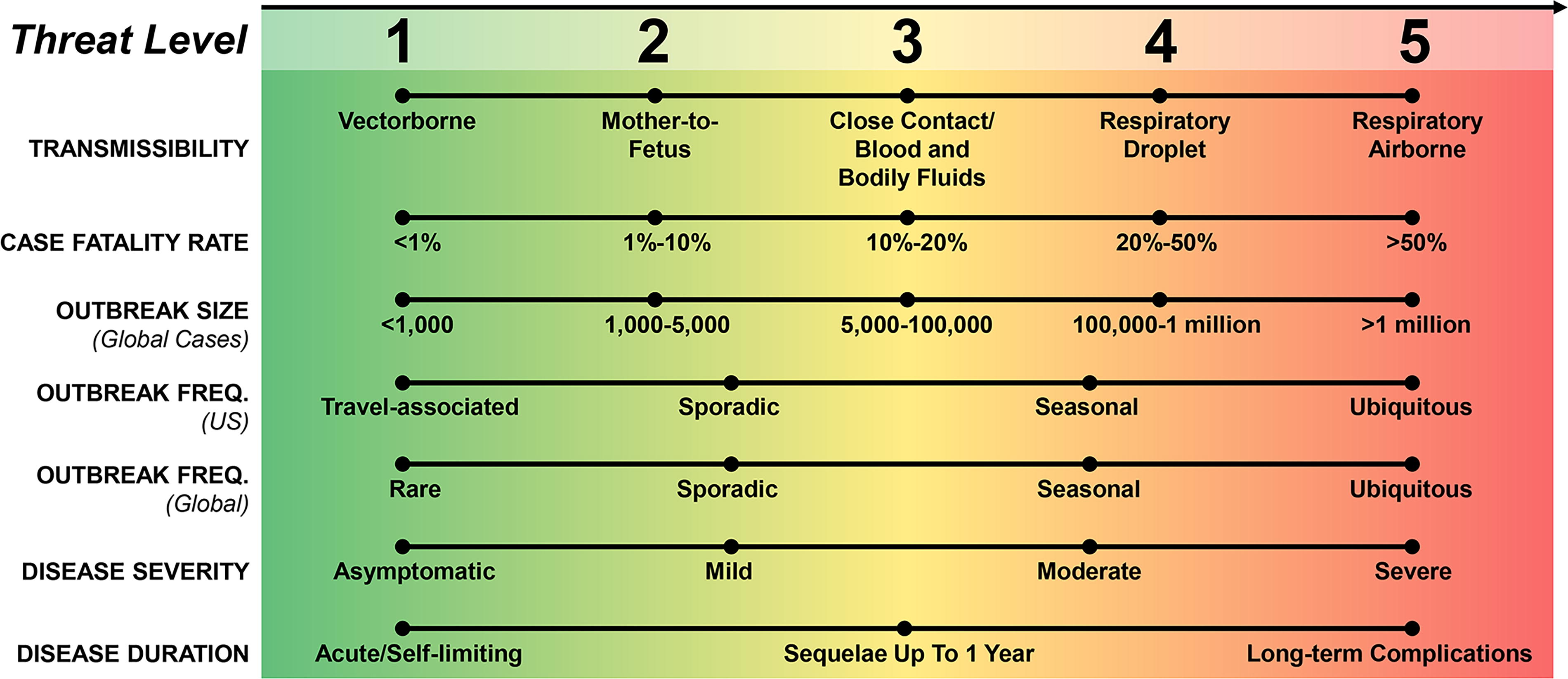
Parameters and rating scale used to create heatmap visualization for assessing viral pathogen threat. For the parameter “Outbreak Frequency (US),” a rating of 0 (not shown) indicates that no confirmed cases (imported or locally acquired) were reported in the past 10 years within the United States. Abbreviation: FREQ, frequency.
To identify viral pathogens for inclusion in this work and to evaluate the utility of this approach, we reviewed various priority pathogen lists that have been published by public health organizations, including the World Health Organization, 10 the National Institute of Allergy and Infectious Diseases,11,18 and the Coalition for Epidemic Preparedness Innovations. 12 We also included viral pathogens identified by the Department of Homeland Security as potential biological weapons (eg, Zaire ebolavirus, Sudan ebolavirus, Marburg virus) as well as pandemic influenza and SARS-CoV-2, because these viruses are designated high-priority biological threats by the Public Health Emergency Medical Countermeasures Enterprise, 19 are already in BARDA’s threat space, and BARDA receives (or has received) substantial funding to develop MCMs for them. Finally, to understand the versatility of the assessment tool, 2 other globally endemic viruses, seasonal influenza and human parainfluenza viruses (HPIV-1 and HPIV-3), were assessed. In total, 27 viruses across 14 virus families were selected (Figure 2). Lastly, we also included Disease X, a concept that refers to a viral pathogen that has yet to be identified and could cause a severe public health emergency, such as a global pandemic. 10 SARS-CoV-2 is an archetype of a Disease X, a novel virus previously unknown to infect humans that led to a global public health emergency within a matter of months.
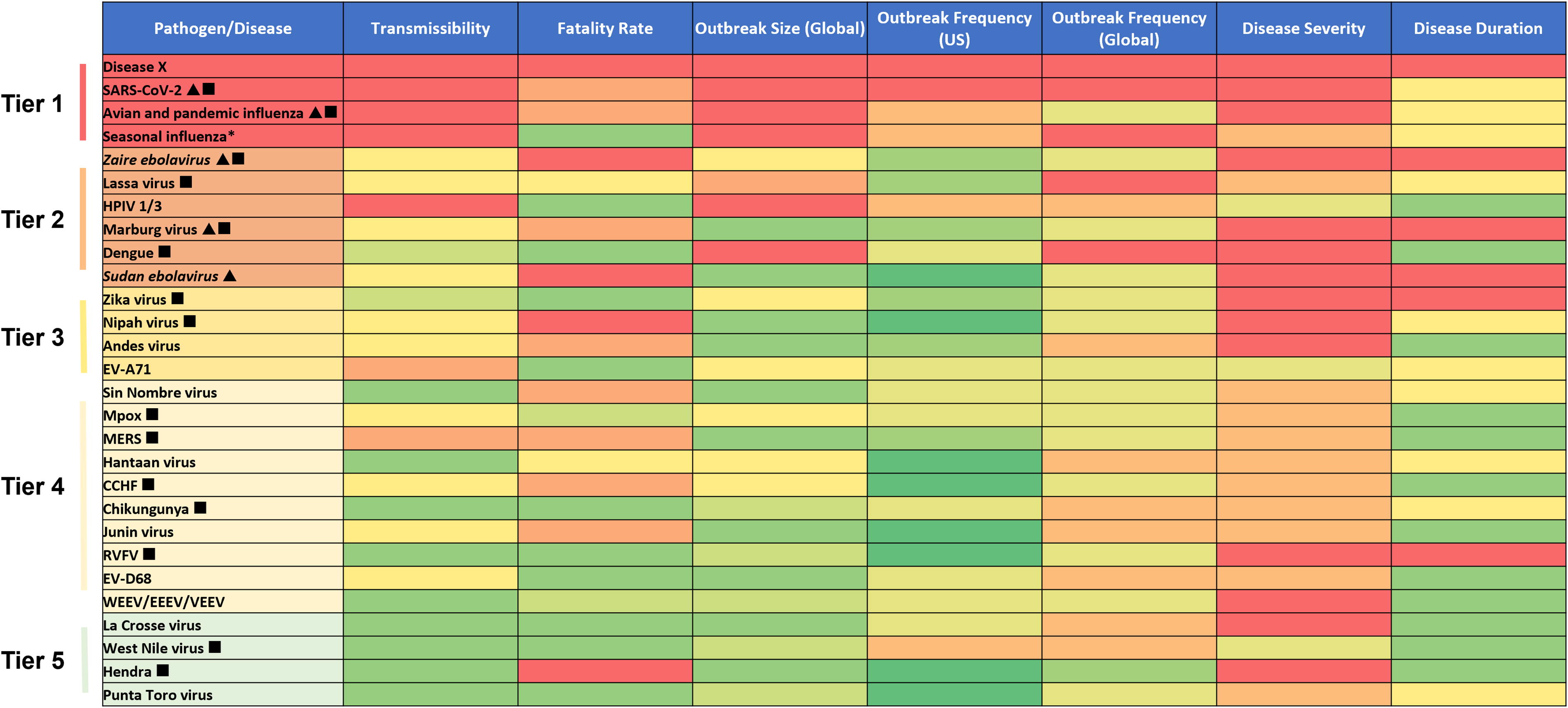
Heatmap denoting the threat level of each viral pathogen by tier. The triangle symbol denotes viral pathogens for which BARDA receives (or has received in the past 5 years) substantial funding to support medical countermeasure development. The square symbol denotes infections that have newly appeared in a population within the last 20 years or have existed but are rapidly increasing in incidence or geographic range. Abbreviations: BARDA, Biomedical Advanced Research and Development Authority; CCHF, Crimean-Congo hemorrhagic fever; EEEV, Eastern equine encephalitis virus; EV-A71, enterovirus A71; EV-D68, enterovirus D68; HPIV-1 and HPIV-3, human parainfluenzavirus types 1 and 3; MERS, Middle East respiratory syndrome; RVFV, Rift Valley fever virus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; VEEV, Venezuelan equine encephalitis virus; WEEV, Western equine encephalitis virus. aWhile BARDA does not receive funding specifically for seasonal influenza medical countermeasures, it does receive and has leveraged funding for pandemic influenza to support new vaccine technologies, therapeutics, and diagnostics that have a positive impact on seasonal influenza.
Based on publicly available information collected for the 7 parameters, values were assigned for each virus according to the scale in Figure 1 and a heatmap was created (Figure 2). The heatmap was further subdivided into 5 tiers, proportional to the overall threat level, and includes all pathogens that were assessed. BARDA’s tiered list of viral pathogens was then compared with those lists identified by other institutions that have conducted similar prioritization exercises.
Results
This study was undertaken to develop a tool to better understand pathogen threat, a key component for identifying targets for MCM development. Seven parameters were selected to objectively assess the threat of each virus (Figure 1). The list included several pathogens for which BARDA has developed a robust portfolio of MCMs, including Zaire ebolavirus, Sudan ebolavirus, Marburg virus, pandemic influenza viruses, and SARS-CoV-2, to determine where they would rank relative to other viral pathogens of interest. We also included Disease X given the potential devastating impact of a novel pandemic threat and because BARDA is actively developing preparedness and rapid response capabilities that are pathogen agnostic and could be used to develop MCMs for presently unknown pathogens. The list did not include bacterial, fungal, or protozoan pathogens, although the tool can be adapted to assess these pathogens in the future. Each virus was subsequently rated according to each parameter on a scale of 0 to 5 or 1 to 5, and a heatmap was generated to visualize each virus relative to one another (Figure 2). Five threat tiers were identified, with the top 2 tiers containing Disease X, SARS-CoV-2, avian/pandemic influenza, Ebola, and Marburg—all viral agents for which BARDA has active MCM portfolios. Viruses for which BARDA receives (or has received in the past 5 years) substantial funding for MCM development are denoted by a triangle. Viruses that cause infections that have newly appeared in a population within the last 20 years, or have existed but are rapidly increasing in incidence or geographic range (ie, EIDs), are denoted by a square.
Discussion
As described in BARDA’s strategic plan, 16 one of its primary missions is to support the advanced development and eventual FDA approval of MCMs for a myriad of known and unknown threats. Preparing for, and potentially responding to, EIDs requires careful planning and strategic thinking, which is rooted in extensive knowledge of the threat. Since BARDA’s yearly congressional appropriations are insufficient to support a pipeline of products to address all potential emerging pathogens, it must make thoughtful portfolio decisions based on the best available knowledge. 5 This article describes one of the many tools BARDA is using in assessing viral threat pathogens with respect to EID preparedness.
Portfolio management plays a crucial role in the formation and sustainment of a robust pipeline of MCMs. It is imperative that BARDA uses a rigorous process in which its strategic goals are aligned with MCM development. Figure 3 illustrates some of the major criteria that come into play, including pathogen threat, when BARDA evaluates new technologies and MCMs. Many of the criteria are based on the readiness level, such as the landscape of candidates available for development and their maturity levels, the existence of qualified nonclinical models to assess safety and efficacy, the clinical feasibility of conducting efficacy trials and the appropriate regulatory strategy for approval, alignment with target product profiles, feasibility of employing rapid manufacturing platforms, and the ability to sustain the availability of the MCM once it is FDA approved. Other criteria that inform the decisionmaking process include: the availability of funds for a specific threat; mandates and requirements in the form of supportive legislation; partnerships with developers and potential funding agencies (including coordinating research and development agendas with nongovernmental partners and with interagency partners through the Public Health Emergency Medical Countermeasures Enterprise); and the priority pathogen itself and how it ranks relative to the myriad of other pathogens for which BARDA is called upon to develop MCMs.
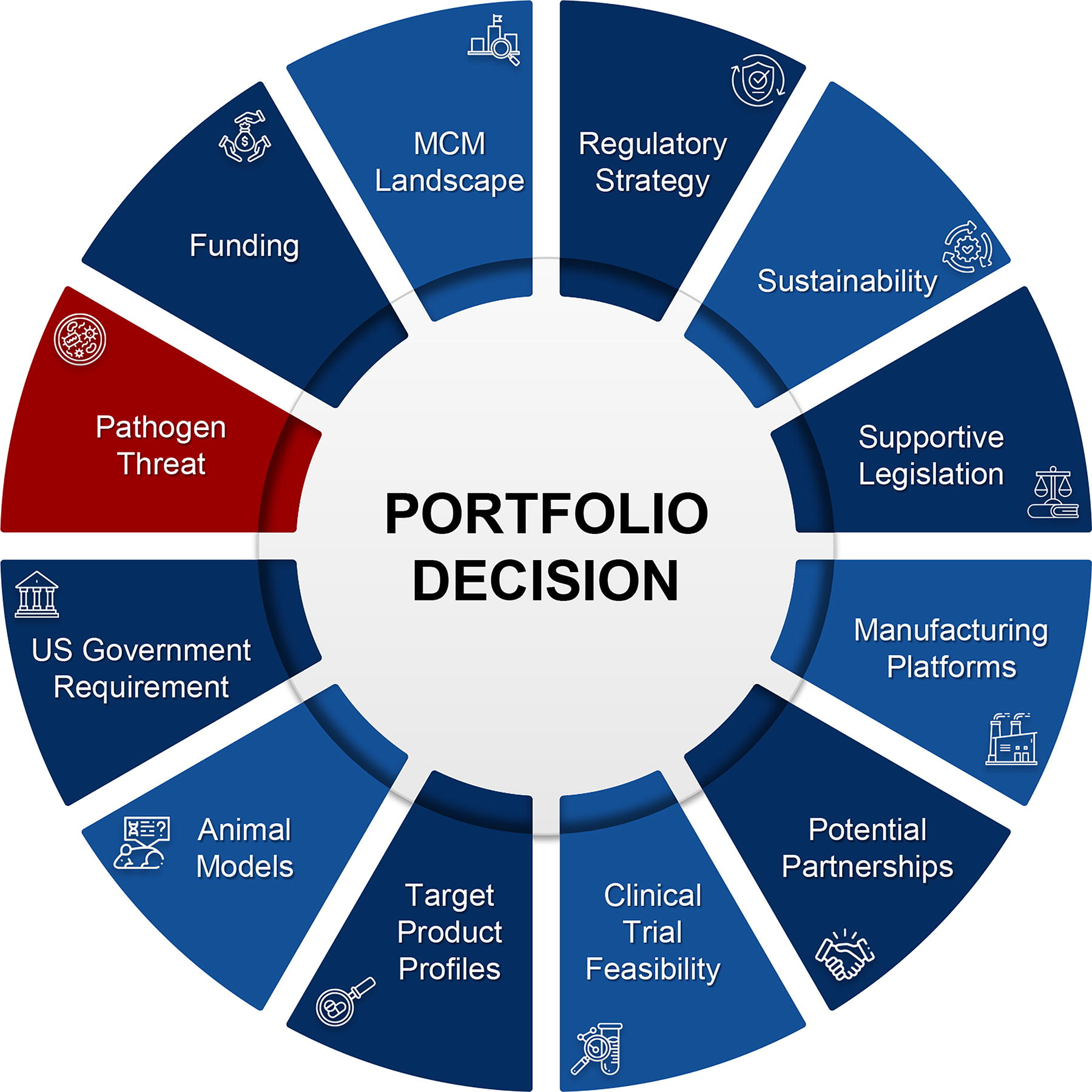
BARDA’s portfolio management and decision analysis criteria. Abbreviation: MCM, medical countermeasure.
As of December 2024, BARDA has supported 95 FDA licensures, approvals, or clearances of MCMs. 20 These include vaccines, therapeutics, diagnostics, and devices for use against agents identified by the Department of Homeland Security as “material threats” (eg, Ebola, anthrax, nuclear and chemical threats), pandemic influenza, and EIDs (eg, SARS-CoV-2, Zika). In addition to addressing known pathogens of interest, BARDA is developing rapid response platforms and partnership networks (eg, Rapid Response Partnership Vehicle Consortium) that would accelerate a response to an unknown pathogen, such as Disease X. Rapid response platforms are essential for responding to Disease X-type pathogens as well as developing MCMs for known pathogens for which MCMs are lacking or no longer effective. The tool described herein will also facilitate the prioritization of MCMs for those types of novel pathogens.
When selecting parameters for inclusion in the tool, we prioritized those that could be assessed using more objective measures (eg, transmission mode, fatality rate) based on publicly available data. This was intended to reduce bias introduced during the consultative process (eg, use of expert opinion surveys). One limitation of this approach, however, is the number of pathogens that can be included in the tool due to lack of available data. To facilitate visual comparisons across pathogens in the tool, the ratings were limited to no more than 5 per parameter. In some cases, this resulted in large ranges spanning an order of magnitude or greater (eg, case fatality rate: 1% to 10%, global outbreak size: 5,000 to 100,000). Constrained by the limited number of parameters that could be included in the assessment tool (based on data availability), BARDA assigned pathogens to tiers instead of using absolute ranking, which avoids prioritizing one pathogen over another with nearly identical scores. Of note, other widely referenced priority pathogen lists—including those that informed the selection of viruses for inclusion in this assessment—are not ranked (ie, the shortlisted viruses are presented in no particular order), such as lists from the World Health Organization, Coalition for Epidemic Preparedness Innovations, and National Institute of Allergy and Infectious Diseases.
Although the parameters included in the tool were assigned equal weight (as was done in a study by the Africa Centres for Disease Control and Prevention [Africa CDC] 14 and several other published studies 21 ), the tool can be easily modified to accommodate criteria weighting, which may be of interest to other institutions that apply different weights to adapt the prioritization scheme to their individual mission or in response to evolving policies and directives. For example, a national security organization may weight US outbreak frequency (Figure 1) more heavily than global outbreak size and frequency. Conversely, a global health organization may consider reducing the weight of outbreak frequency in the United States.
BARDA’s focus on development of the assessment tool was to evaluate the pathogen threat. Perhaps not surprisingly, Disease X is among BARDA’s top-tier threat, which supports its focus on threat-agnostic rapid response capabilities. Further, many of the pathogens already in BARDA’s development portfolio, such as SARS-CoV-2 and pandemic influenza, appear in the top tier (Figure 2).
A comparison of BARDA’s tiered list of viral pathogens with those published by other organizations shows general alignment (Table), with some notable exceptions driven at least in part by differences in the goals of the assessment. The VACCELERATE Site Network, for example, undertook a pathogen prioritization exercise based on the perceived likelihood of triggering a pandemic, 15 while Africa CDC conducted a more continent-level prioritization exercise to evaluate the risks of epidemic-prone diseases to inform its emergency preparedness planning. 14
Prioritization of Pathogens Across Selected Public Health Agencies and Institutions
Abbreviations: CCHF, Crimean-Congo hemorrhagic fever; EEEV, Eastern equine encephalitis virus; EV-A71, enterovirus A71; EV-D68, enterovirus D68; HPIV-1 and HPIV-3, human parainfluenzavirus types 1 and 3; MERS, Middle East respiratory syndrome; RVFV, Rift Valley fever virus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; VEEV, Venezuelan equine encephalitis virus; WEEV, Western equine encephalitis virus.
It is notable, however, that some viruses (eg, influenza, dengue, mpox) included in and ranked among the top-tier pathogens on BARDA’s and others’ lists (Table) are absent from other prominent pathogen lists.10-12 This is in part due to differences in approach that may include parameters such as robust research and development pipelines, established regulatory pathways, and stable funding streams. 22 The BARDA assessment tool does not discount the importance of MCMs in the pathogen prioritization; in fact, the data reflected in the heatmap analysis take into account mitigation by approved MCMs, indicating there is still substantial opportunity and positive impact to be had on global public health for such pathogens. For example, despite the availability of multiple FDA-approved influenza countermeasures, the US Centers for Disease Control and Prevention estimates that in the 2023-2024 influenza season there have been up to an estimated 900,000 hospitalizations and 100,000 deaths in the United States alone. 23 Thus, our assessment of the pathogen threat in the context of other decisionmaking tools can support portfolio management by prioritizing development of new (or improvements to existing) MCMs for the greatest public health impact.
Conclusion
The BARDA tool is intended to inform EID preparedness and response strategies, whether it is in target discovery, early proof-of-concept studies, platform development/refinement, or advanced development efforts. Our intent is to provide existing and future partners with a clearly defined roadmap of our priorities and goals. Different institutions prioritize pathogens for various reasons (and with unique parameters and decisionmaking approaches) and understanding these intents and methods is essential when interpreting what the lists indicate about the pathogens included. This analysis articulates the fundamental parameters that BARDA uses to assess pathogens of interest with respect to their threat. BARDA’s threat list reflects a representative sample of pathogens and captures a snapshot in time, and the list is expected to be updated with additional pathogens and new information, as necessary. The resultant list in Figure 2 is not meant to describe the order in which BARDA will make portfolio decisions. This analysis is only one part of an algorithm that BARDA uses to address the complex ecosystem of EID preparedness within the framework of budgetary constraints. Future analyses will include MCM development feasibility assessments and development of target product profiles.
Footnotes
Acknowledgments
The authors appreciate the input of Gary Disbrow in the review of this manuscript and Amy Savoy for providing graphics support. The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of BARDA or the US Department of Health and Human Services.
