Abstract
From influenza to COVID-19, emerging infectious diseases have taken a heavy toll on lives and resources. Emerging infectious diseases represent one of the largest threats to national security. The primary mission of the Center for Biomedical Advanced Research and Development Authority (BARDA), within the US Administration for Strategic Preparedness and Response, is to support the advanced development of medical countermeasures (MCMs) for public health security threats, including select infectious diseases. Given the number of potential emerging infectious diseases, it is not feasible to develop a suite of MCMs necessary for a full response, including vaccines, therapeutics, and diagnostics. In this article, the authors describe BARDA’s 3-step strategy to address emerging infectious diseases: (1) prioritize the development of MCMs for BARDA’s priority pathogens with an increased focus on “platform technologies” with rapid development capabilities; (2) develop response capabilities including specific licensed medical countermeasures and flexible, rapid MCM development infrastructure; and (3) improve those response capabilities, so they are finely tuned and ready when needed.
Introduction
T
Development of BARDA’s EIDreparedness Strategy
BARDA’s longstanding and highly effective preparedness strategy has been to create public–private partnerships for advanced research and development to deliver clinically proven MCMs for each threat. 5 Because the availability of products that have been approved, licensed, or cleared * by the US Food and Drug Administration (FDA) allows for the quickest response to these threats, BARDA invests in product development through to FDA approval and readiness for stockpiling. Once procured into the Strategic National Stockpile, these products can be onsite anywhere in the United States, many within 12 hours. 6 BARDA has successfully supported more than 90 MCMs through FDA approval. 7
A strategy requiring development through successful Phase 3 efficacy trials, development and maintenance of manufacturing capability, FDA approval, and acquisition of large quantities of MCMs is very costly and time consuming. Unfortunately, the potential for EID threats is enormous. Approximately 335 EIDs were identified between 1940 and 2004. 8 Newly identified pathogens, such as SARS-CoV-2, and new variants of fast-evolving pathogens, such as HIV and influenza, make it impossible to predict the next pathogen or disease-causing variant. Approval is hampered when disease outbreaks are too brief or small to support efficacy trials. Finally, a previously unidentified threat that emerges and spreads rapidly cannot be addressed in time with traditional development timelines. These issues make the traditional preparedness strategy (eg, large, ready stockpiles of FDA-approved MCMs) scientifically and financially untenable for EIDs, so we turn to learnings from influenza and COVID-19 for a different strategy.
Although pandemic influenza viruses are not novel pathogens, their rapid evolution makes them unpredictable, and their potential rate of human transmission makes response speed critical. Influenza is therefore a good EID model, and BARDA’s pandemic influenza strategy, outlined below, is the starting point for our EID strategy.
Lessons From Influenza
To respond to pandemic influenza,9,10 BARDA looks for long-term partners that can leverage established domestic seasonal vaccine manufacturing and partners with threat agnostic capabilities. Threat agnostic examples include platform technologies for vaccines and monoclonal antibodies that can rapidly pivot to make MCMs for new influenza strains, host-directed therapeutics that can treat conditions caused by more than 1 infectious agent (eg, treatment of acute respiratory distress syndrome), broad-spectrum antivirals likely to be effective against most influenza strains (eg, oseltamivir), and next-generation sequencing-based diagnostics that are equally effective across all current and future influenza strains. BARDA and its partners develop MCMs through clinical trials and manufacturing scale-up to FDA approval. A portion of these products are stockpiled, but due to expiry and the unpredictability of the next strain of influenza, stockpiling alone is insufficient for a full and rapid response. To address the gap, BARDA and its partners also build flexible response capabilities in 3 areas: manufacturing and supply chain, advanced research and development, and contracting and other partnerships. BARDA and its partners invest to sustain and improve manufacturing capability, which includes increasing yields and reducing overall manufacturing time, cost, and supply chain risk. To respond to a pandemic influenza requiring new MCMs, BARDA and its partners invest in research and development efforts to validate their technologies in other influenza strains. Using animal models and early clinical trials, BARDA and its partners gain a better understanding of the limits and applicability of technologies, manufacturing capabilities, and research efforts needed for the fastest and most effective response to an emerging pandemic. Finally, BARDA continually works to improve contracting and other partnering mechanisms, so that roles and requirements are agreed to up front and work can start immediately in case of an outbreak. These capabilities help ensure effective MCMs are available to respond as additional supplemental funding becomes available.
To date, pandemic influenza preparedness has been unique from other threats in 2 important ways. First, BARDA’s annual pandemic influenza funding enables end-to-end investment from partner identification through FDA approval and sustainment. Second, seasonal influenza creates a commercial market, defraying the costs of infrastructure, warm basing, and research and development needed to pivot to pandemic strains when required. While the funding and commercial market aspects of influenza are unlikely to be replicated for other EIDs, the technologies, capabilities, and strategies developed for pandemic influenza are applicable and were used in the COVID-19 response.
Lessons From COVID-19
Although SARS-CoV-2 was a novel pathogen, never experienced by humans, BARDA was able to leverage prior work conducted on spike antigen stabilization 11 as well as novel platform technologies (eg, messenger ribonucleic acid [mRNA]) it had previously funded. Leveraging the influenza infrastructure and response strategy, BARDA immediately initiated efforts with existing partners to develop therapeutics, vaccines, and diagnostics against SARS-CoV-2. BARDA repurposed an FDA-approved, host-directed therapeutic developed to treat rheumatoid arthritis (eg, tocilizumab, an anti-interleukin-6 monoclonal), and initiated development of novel therapeutics by leveraging a monoclonal antibody platform used in a product licensed for Ebola. BARDA pivoted vaccine technologies that were under development or had been developed and licensed in the context of other EIDs, including Zika (mRNA), Ebola and influenza (vector-based), and influenza (recombinant) to begin development of MCMs against SARS-CoV-2. In diagnostics, BARDA and its partners quickly evaluated the many existing molecular-based platforms to determine which might be suitable for fast development, manufacture, and distribution of MCMs to detect infection. Repurposed and newly developing MCMs were then tested in clinical studies and successful candidates were cleared by the FDA.
The COVID-19 response demonstrated the value of the pandemic influenza strategy. Experience developing, licensing MCMs on, and validating technologies already supported by BARDA greatly accelerated COVID-19 MCM development and FDA approval. Where preexisting contracts and funding mechanisms existed, MCM development could begin immediately. MCM availability was also accelerated where large-scale commercial manufacturing existed (eg, monoclonal antibodies).
The COVID-19 response also illuminated opportunities to improve BARDA’s strategy and capabilities, including increasing emphasis on threat agnostic MCMs and capabilities, developing technologies that improve access and move response capabilities closer to the point of need, increasing manufacturing capacity, and further refining contracting and partnership mechanisms. 2
3-Step Strategy to Prepare for Emerging Infectious Outbreaks
Reflecting on the COVID-19 experience, and building on the influenza preparedness model, BARDA is implementing an explicit 3-step strategy to (1) prioritize and partner, (2) develop and sustain, and (3) validate capabilities for the most rapid response to the widest variety of EIDs (Figure 1).

BARDA strategy to build an MCM rapid response capability for emerging infectious diseases. Abbreviations: BARDA, Biomedical Advanced Research and Development Authority; EID, emerging infectious disease; FDA, Food and Drug Administration; MCM, medical countermeasure; MTD, material threat determination.
Prioritize and Partner
Through annual appropriations, BARDA will continue to prioritize chemical, biological, radiological, and nuclear threats identified as national security concerns, pandemic influenza, and EIDs with supplemental funding (eg, COVID-19). 5 Prioritization enables BARDA to better support development of MCMs and their technologies through to approval and long-term manufacturing sustainment.
Within these efforts, BARDA will explicitly prioritize products, capabilities, and technologies that also support rapid MCM development and response capabilities for EIDs. This includes MCMs built on existing or new platform technologies and threat agnostic capabilities, such as host-directed and broad-spectrum therapeutics and next-generation sequencing diagnostics. BARDA will also prioritize partners willing to commit in the long term and structure partnership deals with explicit commitments to deliver as-needed support for response, including for EIDs.
Develop and Sustain
BARDA will continue to develop products through the process to FDA approval. For MCMs, such as those based on next-generation sequencing or host-directed therapeutics, approval of broad indications (eg, treatment of acute respiratory distress syndrome) allows for immediate availability of the product, whether for direct patient care or evaluation in studies for a more specific indication. For platform technologies, the required product licensure supports a faster pivot to development of new threat-specific MCMs (Figure 2). Licensure establishes critical safety, immunogenicity, and manufacturing and control data, while establishing potential regulatory pathway(s) for accelerated development of new EID-targeted MCMs. Licensure establishes design, manufacturing, and execution parameters, as well as baselines for storage, distribution, dosing, and other critical supply chain needs. Licensure requires building and qualifying commercial-scale manufacturing that will be needed in response. Finally, licensure and use of platform technologies builds physician, laboratory, and public confidence12-15 necessary for rapid, mass distribution and use of other products based on the same technology.
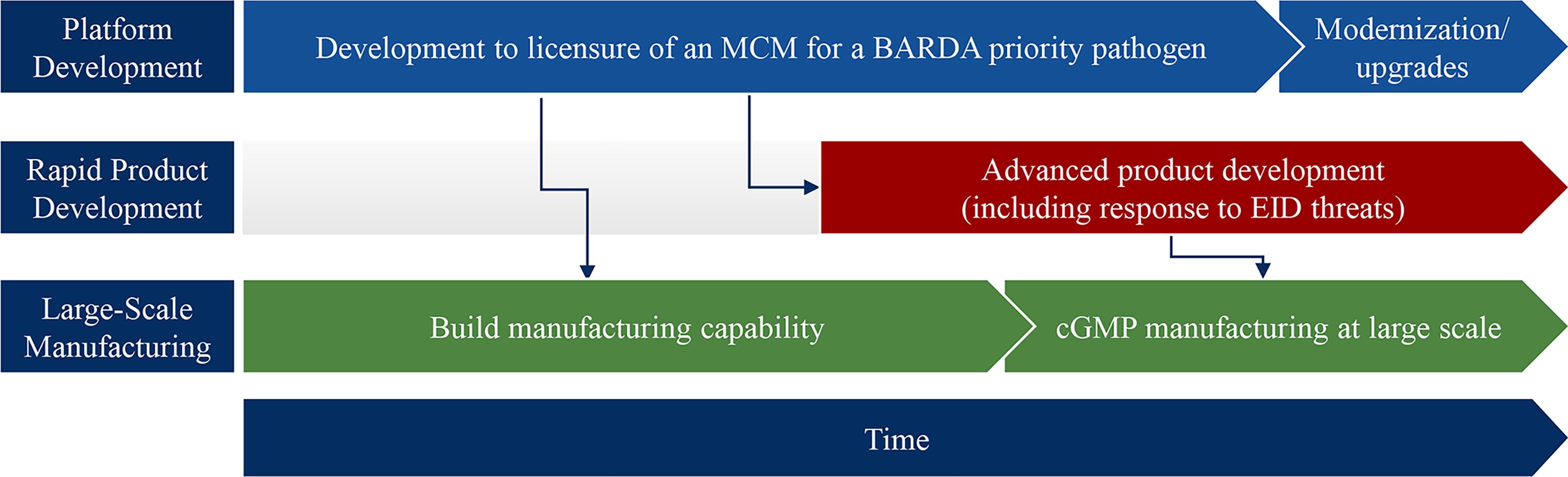
Acceleration of MCM development from “pivoting” an existing licensed platform or technology. In this approach, development of an MCM on a platform technology enables concurrent development of manufacturing and clinical capability that can be used in rapid development and scale up of MCMs for emerging infectious diseases. Abbreviations: BARDA, Biomedical Advanced Research and Development Authority; cGMP, Current Good Manufacturing Practice; EID, emerging infectious disease; MCM, medical countermeasure.
BARDA and its partners will invest to sustain their technology platforms. Example investment areas will include manufacturing capabilities, technologies, and methods to increase product efficacy, reduce product cost, and reduce time to develop new MCMs from these platforms. Where applicable, BARDA and its partners will also leverage existing and new commercial markets to contribute to sustainment.
Validate
Given the vast, unpredictable nature of EIDs, BARDA cannot develop MCMs for every threat. At the same time, BARDA and its partners must understand the capabilities and limitations of supported technologies to respond most effectively during an outbreak. To achieve this understanding, and to maintain platform development capabilities, BARDA and its partners will selectively invest in preclinical and early clinical testing to further understand the range of EIDs for which each technology may be applicable. This investment will also help define the limits, capacities, and capabilities of the technology’s manufacturing platforms, and ultimately support the most rapid and effective EID response.
Challenges
The challenges in preparing for and developing MCMs in response to emerging threats are well established.2,16,17 The breadth of EIDs, combined with the late-stage development and preparation focus of BARDA, add 2 less obvious challenges with large strategic implications.
The first of these challenges is the high cost of sustaining sufficient domestic manufacturing capacity and technology 18 to address an unknown threat that could require rapid production of up to 660 million doses (for a vaccine) in as little as 130 days. 19 A biopharmaceutical plant might cost tens or even hundreds of millions of dollars per year to sustain infrastructure and expert personnel. Biopharmaceutical companies and contract manufacturing organizations offset these costs by producing at large scale for a robust commercial market or by producing multiple products using the same manufacturing facilities and personnel. For BARDA, the same must be true. It is not financially tenable to sustain large numbers of facilities that each support only 1 MCM with a limited commercial market.
As a result, each platform that BARDA selects must be able to support rapid development and scale-up of MCMs for many, if not all, emerging EID threats. BARDA must partner with companies that are committed to long-term sustainment of their MCMs and technology platforms and are committed to using these platforms in the rapid development of new MCMs to address emerging threats. To keep costs within reach, BARDA must also explore new approaches, such as shared use of domestic manufacturing facilities for multiple platforms, and where possible, leverage commercial market/capabilities as is currently done for influenza.
The strength of this approach is the promise of fast-development capabilities to address new threats and MCM manufacturing capacity to respond in the event of an outbreak. The downside of this approach is 2-fold: First, it will require an evolution of the “prototype pathogen” development approach. 20 Vaccines and therapies targeting prototype pathogens will have to be built on, or compatible with, BARDA-selected platforms, rather than bespoke technologies. Second, once BARDA platform technologies are selected, there is a risk of slowing the development of promising alternative technologies. To address this, BARDA will continue market research and low-level investment in early platform development to ensure that the platform technology pipeline remains robust into the future.
A second set of challenges exists for EID therapeutics. For host-directed therapeutics, development of predictive animal models to support down-selection of candidates has been difficult; the regulatory path to approval is often uncertain, and late-stage clinical trials are expensive. Broad-spectrum antivirals that could work against an entire virus family, or multiple virus families, are limited by an extremely thin pipeline. The promise of monoclonal antibodies for rapid response is offset by their high specificity (eg, rapid obsolescence due to viral evolution), high costs, and manufacturing constraints. Finally, with few opportunities to generate clinical efficacy data from randomized controlled trials, preoutbreak regulatory approval for EID therapeutics is challenging.
BARDA is working to address these challenges using Project NextGen 21 and other COVID-19 supplemental funding, plus annual appropriations from pandemic influenza and advanced research and development. These efforts include: (1) developing therapeutic platforms (eg, the Flexible and Strategic Therapeutics, or FASTx, Program) that have the potential to rapidly produce MCMs at lower costs than current capabilities, (2) “tissue on a chip” and other tools that can facilitate preclinical candidate screening and selection for host-directed therapeutics, (3) platform clinical trial approaches to test efficacy of multiple products efficiently and quickly, and (4) past and future awards to developers with platform technologies including the ability to support rapid MCM development and domestic production in response to an EID outbreak. In the near future, BARDA will look to finalize its pathogen severity assessment and corresponding candidate selection criteria, and will initiate support for additional EID threats beyond its current portfolio.
Conclusion
BARDA’s strategic approach to preparing for the next emerging infectious disease outbreak balances the competing priorities of known and unknown threats with technological, organizational, and budget limitations. At its core, this strategy maintains BARDA’s focus on prioritizing, developing, and using FDA-approved products to support a response to currently prioritized threats or unknown threats. Approved disease-specific or host-directed products can be used immediately. Flexible platform technologies used to create licensed products can accelerate the development of new MCMs during a response. Such proven technology platforms provide baseline safety and effectiveness data on a large sample size and provide starting points for key product attributes, such as storage temperature and product presentation that can increase regulatory, physician, and patient confidence, resulting in increased uptake and patient access. BARDA’s mission to create MCMs for its priority pathogens remains. By developing those MCMs on flexible response platforms wherever possible, BARDA addresses the uncertain nature of EIDs and maximizes value and speed while minimizing costs to address current and future EID threats.
BARDA’s strategy provides a clear roadmap for potential partners and those funding early development of MCMs (ie, through Phase 2 clinical trials) to understand how BARDA will prioritize investments in late-stage development and manufacturing capability to support a rapid response MCM capability in alignment with its mission and congressional appropriations language. 3 As mentioned earlier, no matter how we name the threat (eg emerging infectious disease, threat X, pandemic potential, or other), our approach is to have access to capabilities and technologies to rapidly develop, gain approval for, and manufacture sufficient MCMs to address an outbreak of a known or new infectious disease. Efforts to prioritize viruses are ongoing and will be shared when complete, with an expected emphasis on outbreak frequency, transmission rates, and disease severity. A common understanding of BARDA’s EID strategy provides a level playing field for all interested parties and a starting point for further discussions on different approaches that could be taken to meet the requirements.
Footnotes
Acknowledgments
The authors would like to thank the many BARDA team members that contributed to shaping this commentary, in particular Dr. Gerald Kovacs, as well as Drs. Chris White, Chris Houchens, Kimberly Armstrong, Rodney Wallace, Daniel Wolfe, and Karen Martins for their careful review of the draft manuscript. The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of BARDA or the US Department of Health and Human Services.
