Abstract
In Japan, the Infectious Disease Control Law designates certain institutions across the country as medical institutions for infectious diseases, with the role to respond to and prepare for epidemic or pandemic infections. Since the early stages of the COVID-19 pandemic, these designated medical institutions have provided clinical care to patients with COVID-19. While these institutions primarily handle clinical care, they are also well poised to conduct rigorous clinical research that is needed to address future health emergencies. The COVID-19 pandemic highlighted the importance of clinical research as a medical countermeasure through its role in the development of effective novel vaccines and therapeutics. Under the Japanese system, designated medical institutions that cared for patients with COVID-19 had the privilege to access the earliest cases and were uniquely positioned to contribute to scientific evidence. Based on this understanding, we conducted a nationwide survey and analyzed data from 100 designated medical institutions to better understand their experiences and involvement in clinical research during the COVID-19 pandemic and their readiness and willingness to conduct clinical research in a future health emergency. While quite a few institutions showed willingness to participate in infectious disease research in the event of a future health emergency, it was evident that many would require additional expertise and financial support to facilitate such research. Our analysis suggests that further capacity development, empowerment for clinical research, and a strong collaborative network across stakeholders are required to improve pandemic response and preparedness in Japan.
Introduction
C
One of the major hurdles in conducting meaningful clinical research during the COVID-19 response was the divide between patient care and clinical research. 11 This was also the case in Japan where harmonization and collaboration among frontline hospitals and clinics, academic institutions, local governments, and public health centers responsible for patient triage and transfer were not systematically designed to conduct clinical trials for purposes such as identification of effective therapeutics. 12 Furthermore, understanding the opinions and experiences of frontline providers and hospitals/clinics is crucial for improving clinical research practices in a health emergency; existing literature has noted challenges such as unpredictability and urgency required in navigating and working during a pandemic, resource constraints, and some ethical concerns.13,14
When emerging and reemerging infectious diseases are on the rise, designated medical institutions provide clinical care to those patients, as stipulated by Japan’s Act on the Prevention of Infectious Diseases and Medical Care for Patients with Infectious Diseases (hereinafter referred to as the Infectious Disease Control Law). 15 In accordance with the law, during the early phase of the pandemic, COVID-19 patients sought care from the designated medical institutions. 16 While many saw the tireless efforts provided by these institutions, not much attention was given to the roles they could play in clinical research given their position in early access to and experiences with infectious diseases. This unique positioning enables these institutions to function as a cornerstone of clinical research to better respond to a pandemic.
Understanding the readiness and willingness of designated medical institutions to conduct clinical research during a future health emergency is the first step toward planning and better preparedness. We therefore conducted a nationwide survey among the designated medical institutions in Japan to understand their experiences with clinical research during the COVID-19 pandemic and to explore their willingness to contribute to clinical research in future infectious disease health emergencies.
Methods
Definition
According to the Infectious Disease Control Law, designated medical institutions are categorized as Specified, Class 1, and Class 2 hospitals by the Japanese
We use the term clinical research to encompass both noninterventional/observational studies that investigate the clinical characteristics and pathogenicity of emerging and reemerging infectious disease (eg first few hundred studies) as well as clinical trials involving interventions. 22 We also define clinical trials as (1) investigator-initiated clinical trials including both investigator-initiated registration-directed trials under the Pharmaceuticals and Medical Devices Act 23 and specified clinical trials under the Clinical Trials Act,24–26 and (2) industry-sponsored clinical trials.
Survey Respondents and Procedure
A survey was mailed on February 1, 2023, to all 376 designated medical institutions in Japan listed on the MHLW website, 27 which are composed of 4 Specified, 54 Class 1, and 318 Class 2 hospitals across all 47 prefectures. Of note, Class 2 hospitals include both hospitals with notifiable infectious disease beds and those with beds specifically dedicated to tuberculosis (operating beds), and we opted to include the former only. Attached to each survey was a request letter from MHLW directed to medical directors of each designated hospital, noting that the survey was a form of healthcare service evaluation and would be used anonymously for future policy development and research. A link was included to download the survey and upload the responses in a Microsoft Excel format. The survey was accessible for response through March 15, 2023. During this response period, a reminder letter was mailed once. Additional reminders were sent through personal networks of the authors of this article.
Survey Design
The survey was created after consultations with designated medical institutions’ infectious disease physicians and MHLW’s medical officers. It was composed of 26 questions. The questions were developed for clarity and brevity to avoid confusion by the respondents. The survey included a combination of closed, multiple-response, and open-ended free text response questions. We requested medical directors to fill out the survey with support from other technical and relevant staff (such as the infectious diseases department, clinical research department, or management) to respond to the survey.
The survey consisted of 3 parts: (1) characteristics of the hospital, (2) experiences in clinical research before and during the COVID-19 pandemic, and (3) willingness to conduct clinical research in future infectious disease health emergencies and participate in relevant clinical trial networks. First, survey respondents were asked how they provided clinical care for COVID-19 patients at the time of the survey. They were asked if they have a clinical research department and to describe how the department operates. We also specifically asked if they have an academic research organization (ARO) in place, whose function is primarily focused on promoting clinical and nonclinical research including drug development. 28 Typically, an ARO is a large organization or department specifically dedicated to research and can be located at various academic facilities including core clinical research hospitals as determined by the Medical Care Act. 29 Second, respondents were asked about their clinical research experience before the COVID-19 pandemic. The survey asked about details of the clinical research, such as whether the past research experience consisted of observational studies or clinical trials, and whether they have participated in global or domestic multicenter clinical research (as a leading facility or a collaborative facility). Specifically, for COVID-19, they were asked if they had participated in clinical trials for medical countermeasures (ie, diagnostics, therapeutics including repurposing drugs and development of novel drugs, or vaccines) or observational studies. Third, respondents were asked if they would be willing to participate in clinical research during future infectious disease health emergencies. Furthermore, they were asked if they would join clinical research networks for health emergencies, if any, and share their concerns for joining such networks. The full set of survey questions is available in the Supplemental Table 1 (Supplemental Materials are available at www.liebertpub.com/doi/suppl/10.1089/hs.2024.0044).
Statistical Analysis
We descriptively analyzed the responses obtained from the survey. We combined the responses from the Specified/Class 1 hospitals and compared the categorical variables between Specified/Class 1 hospitals and Class 2 hospitals using Pearson chi-square statistics. For questions that allowed participants to submit multiple responses, Bonferroni-corrected P values were reported when comparing responses between Specified/Class 1 and Class 2 hospitals. A P value of <.05 was considered significant. All statistical analyses were conducted using STATA version 16.1 (StataCorp LLC, College Station, TX).
Results
Among all 376 designated hospitals, we received 100 responses, which were from 3 of the 4 Specified hospitals (75%), 28 of the 54 Class 1 hospitals (52%), and 69 of the 318 Class 2 hospitals (22%).
Hospital Characteristics
The characteristics of hospitals are summarized in Table 1. Twenty-nine (94%) of Specified/Class 1 hospitals and 27 (39%) of Class 2 hospitals reported having a clinical infectious disease department. The majority of hospitals (94% of Specified/Class 1 hospitals, 97% of Class 2 hospitals) provided inpatient care for patients with mild to moderate COVID-19 symptoms. All Specified/Class 1 hospitals also admitted patients with severe COVID-19 symptoms, whereas 68% of Class 2 hospitals provided care to those patients. More than two-thirds of both Specified/Class 1 and Class 2 hospitals provided outpatient services for acute COVID-19 or for those with suspected COVID-19 (74% and 93%, respectively). Specified/Class 1 hospitals were more likely to be equipped with a support department for clinical research compared with Class 2 hospitals (90% and 38%, respectively). Support from an ARO was uncommon among all categories of hospitals (25% and 8%, respectively). Most of the hospitals (100% of Specified/Class 1 hospitals, 88% of Class 2 hospitals) had an ethics review board for in-house clinical research.
Characteristics of Hospitals for Clinical Research During the Study Period
Infectious disease department was defined as a clinical department specializing in treating infectious diseases.
We asked if they had established a clinic that offered care after the acute phase of COVID-19.
Academic research organization refers to a large organization and department specifically dedicated to research and can be located at various academic facilities including core clinical research hospitals that have been designated by the Medical Care Act.
Clinical Research Experience Before the COVID-19 Pandemic
All Specified/Class 1 hospitals 31 (100%) and 52 (75%) of Class 2 hospitals had some experience in clinical research before the COVID-19 pandemic (chi-square test for difference: P=.01); and 28 (90%) of Specified/Class 1 hospitals and 40 (58%) of Class 2 hospital conducted clinical research related to infectious diseases (chi-square test for difference: P=.006). We found significant differences between Specified/Class 1 and Class 2 hospitals in their prior experience of investigator-initiated trials, industry-sponsored clinical trials, observational study (Bonferroni-corrected P values of .001, .025, and <.001, respectively), and participation in multicenter clinical research as a leading facility (Bonferroni-corrected P value of .006) (Table 2).
Type of Clinical Research Experience and Participation in Multicenter Clinical Research Before the COVID-19 Pandemic
Note: Multiple answers were allowed.
We found significant differences using Bonferroni-corrected P values between Specified/Class 1 and Class 2 hospitals: investigator-initiated trials (P=.001), industry-sponsored clinical trials (P=.025), observational studies (P<.001), and participation in multicenter clinical research as a leading facility (P=.006).
Among the hospitals with previous experience in conducting clinical trials or infectious disease–related clinical research, frontline clinical providers—such as physicians (90% in Specific/Class 1 hospitals, 70% in Class 2 hospitals), nurses (74%, 33%), pharmacists (87%, 41%) and clinical laboratory technicians (48%, 20%)—were the major players involved in clinical research. Fewer other supporting staff—such as health information managers (13%, 2%), medical clerks (10%, 4%), and data analysts (26%, 9%)—had previously been involved in clinical research activities (Supplemental Table 2).
Experience and Challenges in COVID-19-Related Clinical Research
Twenty-six (84%) of the Specified/Class 1 hospitals and 38 (55%) of the Class 2 hospitals participated in COVID-19-related clinical research, either observational studies or clinical trials (chi-square test for difference: P=.02). Of those who participated in COVID-19-related clinical research, there were no significant differences as to types of research; 10 (38%) of the Specified/Class 1 hospitals and 2 (5%) of the Class 2 hospitals led multicenter clinical research for COVID-19 as a leading facility (Bonferroni-corrected P value of .003), whereas 20 (77%) of the Specified/Class 1 infectious hospitals and 38 (100%) of the Class 2 hospitals joined COVID-19-related multicenter clinical research as a collaborative facility (Bonferroni-corrected P value of .007) (Table 3).
Type of Research Among Hospitals That Conducted COVID-19-Related Clinical Research
Note: Multiple answers were allowed.
We found significant differences using Bonferroni-corrected P values between Specified/Class 1 and Class 2 hospitals: experience in multicenter clinical research as a leading facility (P=.003) and experience in multicenter clinical research as a collaborative facility (P=.007).
Challenges identified by survey respondents in infectious disease–related clinical research in a health emergency based on experiences from the COVID-19 pandemic are summarized in Figure 1; no differences between Specified/Class 1 and Class 2 hospitals were identified. Many respondents reported that clinical care provision was prioritized over clinical research, which reduced their capacity to be involved in clinical research. Human and financial resources, sufficient clinical research infrastructure, prior experience, and clinical research expertise were frequently reported as lacking. Furthermore, open-ended free text responses revealed the need for Japan to further develop organizational expertise in clinical research. Some respondents also emphasized the importance of coordination and communication, both internally with different clinical specialists/stakeholders and externally with governments and other core academic facilities (Supplemental Box).
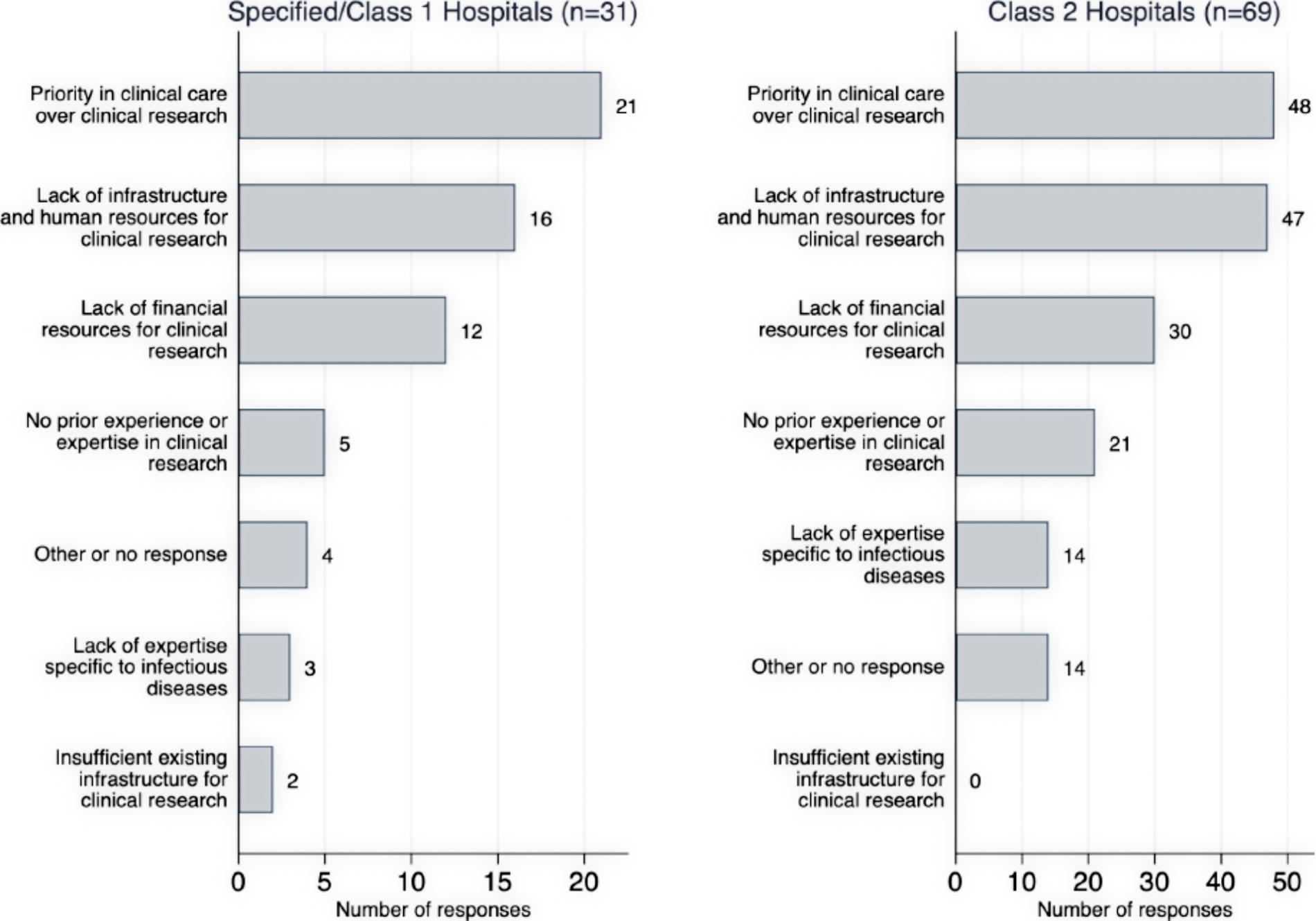
Survey responses to “Select the most important issues in conducting clinical research on infectious diseases at your institution,” stratified by hospital type (n=100). Multiple answers were allowed. No significant difference between Specified/Class 1 and Class 2 hospitals was present.
Willingness to Conduct Clinical Research and Join Clinical Trial Networks
Twenty-three (74%) of the Specified/Class 1 and 36 (52%) of the Class 2 hospitals were willing to conduct clinical research in future health emergencies (chi-square test for difference: P=.14) (Table 4). All of the Specified/Class 1 hospitals and 62 (90%) of the Class 2 hospitals wished to join multicenter research as a collaborative facility (Bonferroni-corrected P value of .264), whereas 10 (32%) of the Specified/Class 1 hospitals and 9 (13%) of the Class 2 hospitals wished to join multicenter research as a leading facility (Bonferroni-corrected P value of .094). Moreover, 11 (35%) of the Specified/Class 1 hospital and 18 (26%) of the Class 2 hospitals were willing to participate in clinical research networks; 15 (48%) of the Specified/Class 1 hospitals and 41 (59%) Class 2 hospitals reported that they would consider joining such networks if the circumstances permitted (chi-square test for difference: P=.50). Among the 59 hospitals willing to conduct clinical research in future health emergencies, observational studies were the most preferable type of study design for both Specified/Class1 (23; 100%) and Class 2 (34; 94%) hospitals, followed by clinical trials (Supplemental Table 3).
Willingness to Conduct Clinical Research in Future Health Emergency and Motivation to Join Clinical Research Networks
Note: No significant difference between Specified/Class 1 and Class 2 hospitals was present.
Support for Clinical Research
Of the 69 hospitals that said they could participate in clinical research networks depending on conditions, or that participation would be difficult, the following were reported as key factors that would support their ability to join clinical research networks: human and financial resources for clinical research; training programs for clinical research; collaboration with AROs, other research-supporting organizations, and local government; and training systems for clinical research (Figure 2).
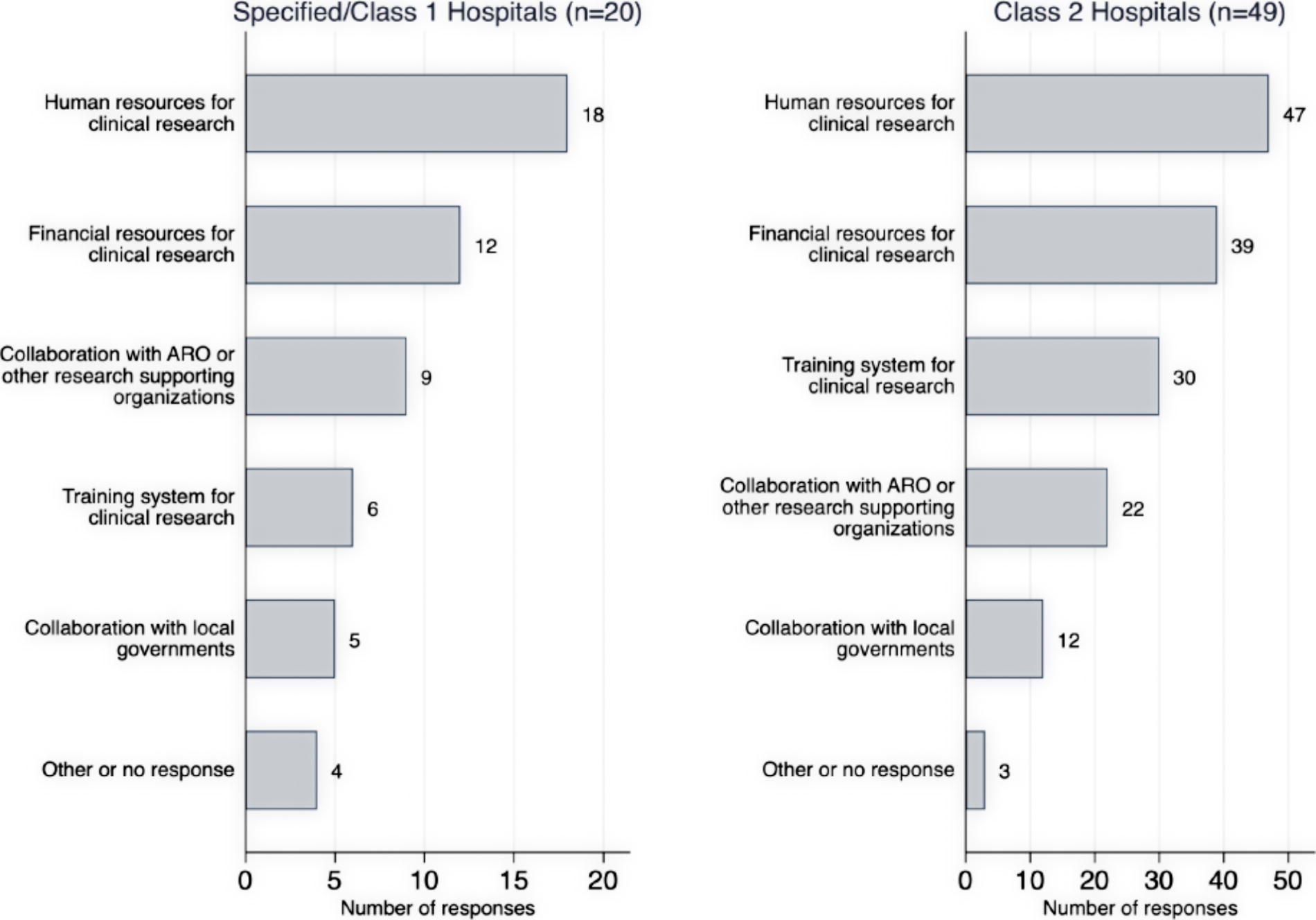
Survey responses to “What are the necessary factors to enable participation in clinical research on infectious diseases?” among the hospitals that showed conditional or no willingness to join clinical research networks, stratified by hospital type (n=69). Multiple answers were allowed. No significant difference between Specified/Class 1 and Class 2 hospitals was present. Abbreviation: ARO, academic research organization.
Likewise, of those 69 hospitals, 50 (72%) requested support from an external ARO (Supplemental Figure 1). They were looking to an ARO to provide a wide range of support including deploying clinical research experts, administrative and technical support such as ethics approval, and developing infrastructure such as training systems for clinical research.
Respondents were asked what sort of support they would request to more proactively promote clinical research in their institutions (Figure 3). The most common request from both Specified/Class1 and Class 2 hospitals was development of clinical research infrastructure and funds for human resource. This request was followed by securing general funding, operational support for administrative work, training, and access to experts in clinical research (including collaboration with academia). The requests were not significantly different between Specified/Class1 and Class 2 hospitals.
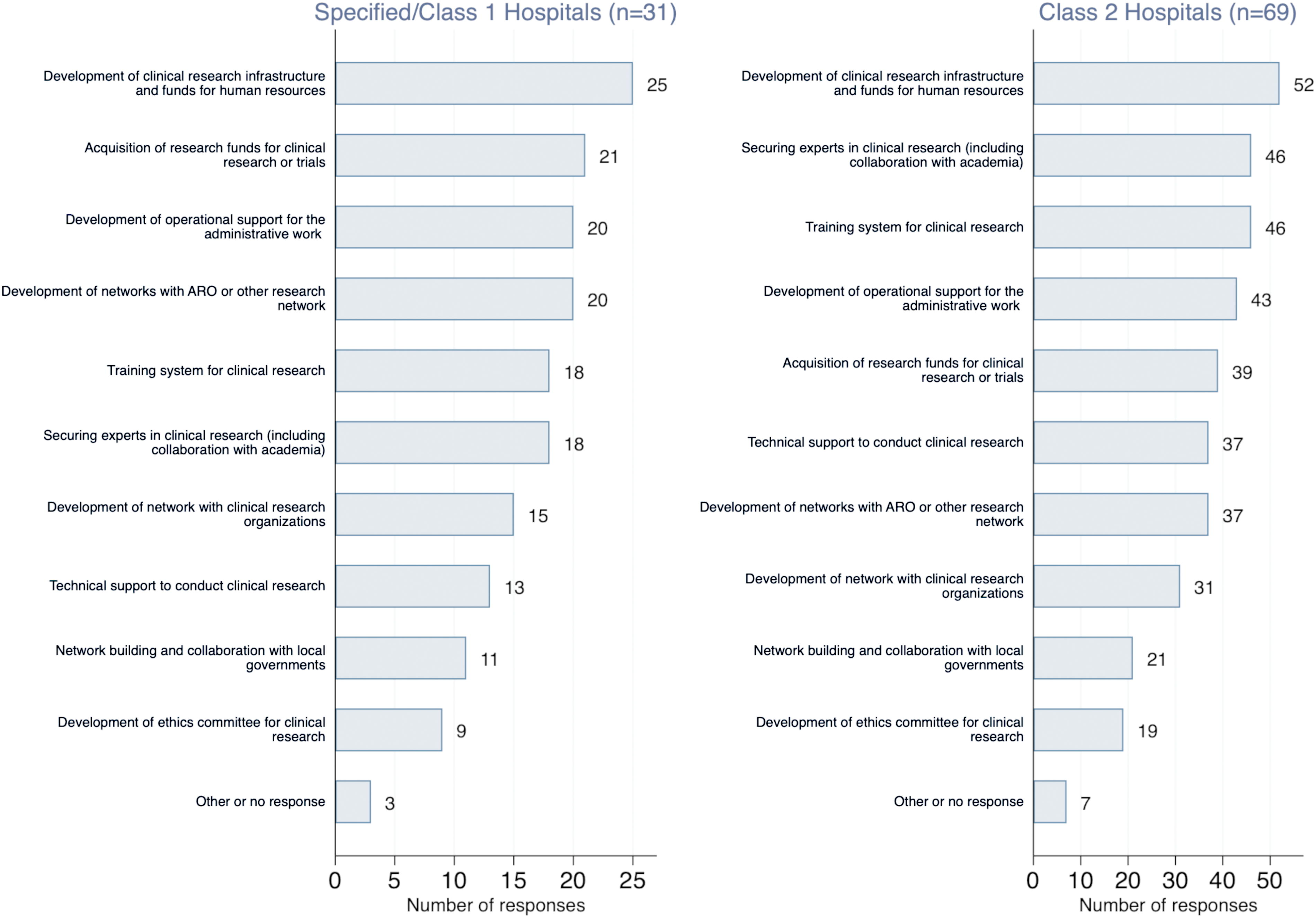
Survey responses to “Select what kind of support you would like to receive in order to promote clinical research on infectious diseases at your institution,” stratified by hospital type (n=100). Multiple answers were allowed. No significant difference between Specified/Class 1 and Class 2 hospitals was present. Abbreviation: ARO, academic research organization.
Discussion
Clinical research to better understand the pathophysiology of an infectious disease of concern and to identify medical countermeasures during infectious disease health emergencies is an essential component of public health to mitigate morbidity and mortality.30,31 The importance of a clinical research mindset, which is “bench to bedside” and “bedside to bench,” to advance science and improve clinical care was highlighted during the COVID-19 pandemic; however, it may have previously been overlooked from a public health standpoint. Challenges in conducting clinical research during a health emergency, particularly in the context of an infectious disease pandemic, are multifaceted. 32 A wide spectrum of issues11,14,32 has been pointed out including patient enrollment, ethical and regulatory hurdles, logistical and resource limitations, and coordination across multiple studies that were often small in size and overlapping in topics and subjects. For instance, in Japan, at least 4 different clinical research studies33-36 and 1 registration research study supported by the government 37 were conducted for favipiravir in Japan in the early stages of the COVID-19 pandemic. A global strategy on pandemic prevention, preparedness, and response during an interpandemic period (ie, beween pandemics) is currently being discussed among countries, and its active implementation is anticipated to better prepare countries for future health emergencies.9,38-40
The survey results presented in this article provide a snapshot of clinical research practices across designated medical institutions in Japan during the COVID-19 pandemic and their willingness to conduct clinical research in future health emergencies, as well as associated challenges. Because these designated medical institutions are uniquely positioned to be the first provider of clinical care during the initial phases of a pandemic, they are well suited to function as the foundation of clinical research; however, designated medical institutions have not previously had the expectation to take on clinical research functions during a pandemic. Although the expectation has not existed, there have been successful examples of designated medical institutions playing a role in clinical research.16,41,42 Our study expanded the understanding of the current state of designated medical institutions in Japan and identified opportunities for better pandemic response and preparedness.
During the COVID-19 pandemic, over half of the hospitals that participated in the survey were involved in some form of clinical research, although they also reported that resources for clinical research were not sufficiently available. For more medical institutions to be able to participate and succeed in both clinical care and research, it is critical to ensure that the deterrents are understood, and needs are met. Major hurdles identified in the survey included difficulties balancing clinical care in addition to clinical research, insufficient financial and human resources for clinical research, and poor research infrastructure specifically for health emergencies, all of which were further exacerbated by the lack of prior experience in clinical research. Seventy-four percent of Specified/Class 1 hospitals and 52% of Class 2 hospitals showed willingness to conduct infectious disease-related clinical research and 83% of Specified/Class 1 hospitals and 85% of Class 2 hospitals showed a desire to participate in a clinical research network (including those who responded affirmatively if they had more support). To ensure that all types of designated medical institutions maintain their motivation for clinical research, top priorities are financial and human resources support, talent development, and networking and establishment both inside and outside of the hospitals.
Due to the nature of a pandemic, the locations where patients are hospitalized cannot be changed to accommodate clinical research needs. In other medical disciplines, such as cardiovascular diseases and oncology, patients can be referred to more research-oriented hospitals and institutes, if appropriate. Infectious diseases, on the other hand, are often acute-onset and contagious by nature, and thus, it would not be easy to transfer patients to a different facility where there may be ongoing clinical research unless there is a clinical need. In practice, those with COVID-19 were triaged and hospitalized at designated medical institutions according to the Infectious Disease Control Law and transferring patients to other facilities with more clinical research resources was logistically difficult. 21 Therefore, a more sustainable and efficient clinical research infrastructure and ecosystem encompassing all elements of conducting clinical research8-10 needs to be established at designated medical institutions, including in remote geographic regions where patients with infectious diseases of concern first seek clinical care.
There is also room for improvement in the research environment in Japan for healthcare in general. A report from the Japanese Ministry of Education, Culture, Sports, Science and Technology in 2020 showed that the proportion of time dedicated to research among Japanese researchers decreased from 47% in 2002 to 33% in 2018. 43 Securing research hours has been problematic especially among physician scientists in Japan.44,45 For instance, clinical service is generally prioritized to compensate for low hospital revenues, and our study revealed that the higher priority on clinical care likely compromised potential clinical research opportunities. Furthermore, a restriction on physician working hours became effective in the spring of 2024, which will also likely reduce the time available for clinical research. 45
It should be noted that off-label and compassionate use therapies for pandemic infections, outside of the clinical research arena, can cause harm 46 ; therefore, fostering an environment for clinical research at health facilities is a prudent strategy for patient safety.
One of the key lessons from COVID-19 is that a pandemic can occur at any time; therefore, every health facility needs to be prepared, with policies in place to engage facilities in clinical research. The United Kingdom’s success in engaging frontline health facilities according to the national health policy serves as a good example for Japan where universal health coverage exists. 47 Clinical research should be embedded into universal health coverage as a public good. While the importance of clinical research in infectious diseases was not well recognized in Japan before the COVID-19 pandemic, it should be considered with utmost importance in future policymaking processes for better pandemic preparedness and response, as highlighted by the political commitment of the 100 Days Mission, in which medical countermeasures are to be made available within the first 100 days after a pandemic threat is identified.38,48 The Japanese government, with some hindsight and learnings from COVID-19, will establish the Japan Institute for Health Security in Spring 2025. This institution will combine 2 existing institutions: the National Institute of Infectious Diseases and the National Center for Global Health and Medicine (also a designated medical institution). One of the key aims is to promote clinical research during a health emergency. 49 It is safe to assume that the government also sees this potential for designated medical institutions given their unique responsibility as first-responding institutions in a pandemic.
Of the hospitals who reported that participation in clinical trial network would either be difficult or conditional, 72% requested support from AROs, which often work closely with core clinical research hospitals. As of February 2024, 15 core clinical research hospitals across Japan were registered with the MHLW and are expected to play a leading role in conducting clinical research and investigator-initiated clinical trials. 28 They also have a mission to support neighboring hospitals; however, even before the COVID-19 pandemic, supporting activities for clinical research by core clinical research hospitals was deemed not adequate. 50 Effective collaboration among the core clinical research hospitals and designated medical institutions during health emergencies or events is an important public health strategy and must be strengthened in the future.
The survey had some limitations. First, the survey response rate varied across the different categories of designated hospitals. In addition to the official request, reminders were sent through the networks of the authors, which improved the response rate to more than 50% among Specified and Class 1 hospitals but only 20% among Class 2 hospitals; thus, caution is warranted when interpreting data, as our results may be biased to the higher-tier Specific/Class 1 hospitals and may not capture the full viewpoints of Class 2 hospitals. 51 The low response rate among Class 2 hospitals may represent their lack of willingness to join clinical research during a health emergency. Conversely, many of the Class 2 hospitals that responded to our survey showed their willingness to engage in clinical research during health emergencies. It is worthwhile to consider specific needs facing Class 2 hospitals to increase the national competency and capacity for clinical research. Second, we presented the data stratified by the type of designated hospitals (ie, Specific, Class 1, and Class 2). Further qualitative and quantitative assessment with more granular hospital data may provide valuable insights about their role in conducting clinical research for health emergencies. Third, this survey was conducted in Japan only. Although the study findings may not necessarily be applicable in the context of other countries with different health systems and cultures, a collaborative comparative study with other countries may be helpful to learn from one another and ensure that, in a world where diseases see no boundaries, we are all better prepared for the next health emergency. 40
Conclusion
Swift identification and development of medical countermeasures, including therapeutics, is critical during a health emergency. Designated medical institutions in Japan are well poised to conduct clinical research but will need additional supports to do so. Improvements in existing systems, policy, and regulation, as well as reevaluation of resource allocation priorities, are imperative to facilitate these institutions to conduct clinical research to be better prepared for future pandemics.
Footnotes
Acknowledgments
We appreciate all the designated medical institutions for infectious diseases who participated in the survey. We also appreciate Ms. Mieko Kikuchi-Conbere, MSPH, for supporting Japanese–English translation of the survey and proofreading the manuscript. We thank INTAGE Healthcare Inc. for their logistical support and advice in creating the survey form. The views represented in this article are those of the authors alone, and they do not necessarily represent the views, decisions, or policies of the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
