Abstract
This case study describes a feasibility assessment of a novel isolation care tent used in health facilities in Uganda during the 2022 Sudan ebolavirus outbreak. The Isolation System for Treatment and Agile Response to High-Risk Infections Model 1B (ISTARI 1B) is a single-occupancy, portable, negative-pressure isolation tent designed for the safe delivery of standard care to patients with a communicable disease, including Ebola disease (Sudan). At the request of the Uganda Ministry of Health, the Makerere University Infectious Diseases Institute and University of Nebraska Medical Center partnered to evaluate 7 health facilities across 4 districts in Uganda for infrastructure, case management, and infection prevention and control (IPC) capacity relevant to isolation care and ISTARI 1B use. A 3-day workshop was held with IPC leaders to provide familiarization and hands-on experience with the ISTARI 1B, delineate appropriate use scenarios in Ugandan healthcare settings, contextualize ISTARI 1B use in case management and IPC workflows, develop a framework for site assessment and implementation readiness, and consider ongoing monitoring, assessment, and intervention tools. Workshop participants performed a comprehensive site assessment and mock deployment of the ISTARI 1B. In this case study, we describe lessons learned from health facility assessments and workshop outcomes and offer recommendations to support successful ISTARI 1B implementation. Use scenarios and implementation strategies were identified across facility levels, including tools for site assessment, training, risk communication, and ongoing quality and safety monitoring.
Introduction
O
At the request of the Uganda Ministry of Health, the Makerere University Infectious Diseases Institute and the University of Nebraska Medical Center (UNMC) partnered to evaluate the Isolation System for Treatment and Agile Response to High-Risk Infections Model 1B (ISTARI 1B) in the context of the evolving Ugandan healthcare system response to the SUDV outbreak.
The ISTARI 1B is a single-occupancy, portable, negative-pressure isolation tent designed to safely deliver care to patients with communicable diseases, including Ebola disease (Sudan). The ISTARI 1B was developed and evaluated by UNMC in partnership with Otherlab (San Francisco, CA) to address the target product profile elements for the intended use. 2 The ISTARI 1B device is composed of a durable frame of metal poles, a transparent plastic canopy, an external fan (FG 6M; Fantech, Lenexa, KS), and a manometer for monitoring pressure inside the tent (Figure 1A and B). Canopy features include bedside lean-in stations with arm and glove ports, closed-end trunks for the use of a stethoscope and ultrasound probe without contamination, in-wall airlock pass-throughs to move material in and out of the tent, and conduit panels for the passage of wires and tubes from externally placed equipment. Two trained staff can reliably assemble the tent within a healthcare facility in less than 15 minutes. It requires approximately 100 watts to operate a negative-pressure, high-efficiency particulate air (HEPA) filtered circulation system with more than 20 air exchanges per hour. The device enables continuous patient monitoring and interaction, enhances patient observation and communication, reduces total staff requirements because of less strain on work-rest cycles and number of individuals involved in routine care, and decreases the overall consumption of personal protective equipment. The current design parameters of the ISTARI 1B account for up to 10 reuses with cleaning and decontamination between patients. Lifecycle management includes the maintenance of durable components (ie, fan, manometer, external frame), reprocessing of the canopy for reuse, replacement of single-use parts (ie, arm ports and trunks), and decontamination and disposal processes. Performance testing of the ISTARI 1B has been completed according to US and international regulatory standards, to include packaging and distribution, shelf life, biocompatibility, electrical and fire safety, airflow and pressure maintenance, airborne particle containment, virus permeability, noise levels, durability to repeated use, resiliency to cleaning agents, human factors, and patient comfort and safety. The commercialization partner for ISTARI products, Carecubes (Carecubes Inc., Monroeville, PA), received 510(k) clearance for the ISTARI 1B from the US Food and Drug Administration in January 2024. 3
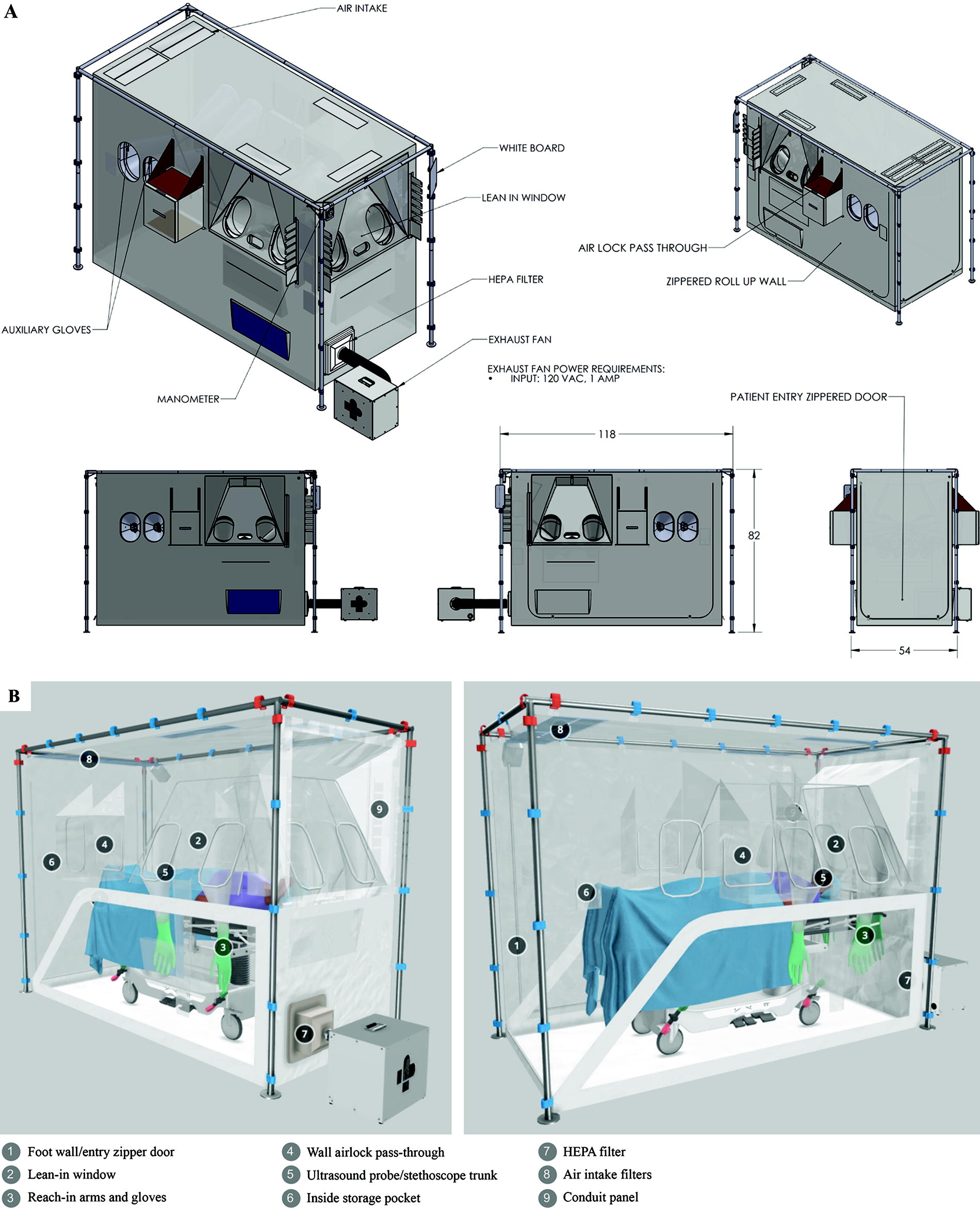
(A) Isolation System for Treatment and Agile Response to High-Risk Infections Model 1B (ISTARI 1B). (B) ISTARI 1B design features. Measurements denoted in inches. Abbreviations: amp, amperes; HEPA, high efficiency particulate air; VAC, volts alternating current.
The innovative ISTARI device blended well with the Ugandan national response strategy, which leveraged community-based and primary healthcare surveillance to support grassroots identification of patients, early initiation of barrier care, and timely referral to treatment centers. In this case study, we describe the evaluation of the ISTARI 1B across health facility levels in Uganda and share lessons learned to inform future implementation efforts in support of safe and effective patient care and integrated case management for highly infectious diseases.
Approach
Setting
At the time of the study, which took place from November to December 2022, Uganda had experienced 142 confirmed cases of SUDV with 55 deaths. 1 Nineteen healthcare worker infections resulted in 7 deaths. Emergency medical services providers performed over 1,500 Ebola-associated evacuations to 5 established Ebola treatment units (ETU) located at Mubende Regional Referral Hospital, Madudu Health Center IV, Entebbe Regional Referral Hospital, Fort Portal Regional Referral Hospital, and Mulago National Referral Hospital.
While the Mubende ETU was the most heavily utilized, each treatment unit required a substantial investment of personnel, equipment, and supplies and relied upon the redeployment of healthcare personnel from districts outside the affected areas. As the SUDV emergency waned and the national response shifted to a phase of heightened active surveillance, these large-scale, low-utilization, isolation-tailored clinical services were disassembled, putting more emphasis on the role of primary healthcare facilities.
In November 2022, a team from UNMC was invited by the Uganda Ministry of Health to bring 2 ISTARI 1B units to Uganda for familiarization and demonstration. During that visit, UNMC introduced the device to IPC mentors convened by the Infectious Diseases Institute while also reinforcing established IPC practices relevant to the emergency. A team returned in December 2022 and conducted an ISTARI 1B superuser workshop incorporating diverse IPC stakeholders. The Uganda Ministry of Health Case Management Committee, through the Infectious Diseases Institute, employed the UNMC team during the 2 visits to assess the potential appropriateness of using the ISTARI 1B device in representative settings.
Health Facility Assessment for Appropriateness of ISTARI 1B Use
Public sector health facilities in Uganda include health centers II to IV, general hospitals, regional referral hospitals, and national referral hospitals (Figure 2A). 4 In addition to public facilities, private hospitals also operate in the Ugandan healthcare system. Representative facilities in the region affected by the SUDV outbreak were selected for assessment of infrastructure, IPC capacity, and scope of case management relevant to ISTARI 1B use (Figure 2B). Facilities were selected based on their location within the districts that were actively engaged in heightened surveillance in consultation with the Ebola Response Case Management team and district officials, with the goal of representing a range of health facility levels. Assessments were performed by the UNMC team. IPC and clinical leads at each health facility provided information on policies and practices regarding management of patients with suspected or confirmed VHF, as well as IPC workflows and capabilities including staff training and experience, infrastructure for isolation care, access to personal protective equipment, and waste management. Facilities were further examined for appropriate covered space and utilities to support ISTARI 1B implementation. Continuous voltage output at a representative urban facility was measured with a multimeter (FLK-289/FVF/IR3000; Fluke Corporation, Everett, WA) to evaluate the need for ancillary battery power to operate the ISTARI 1B safely.
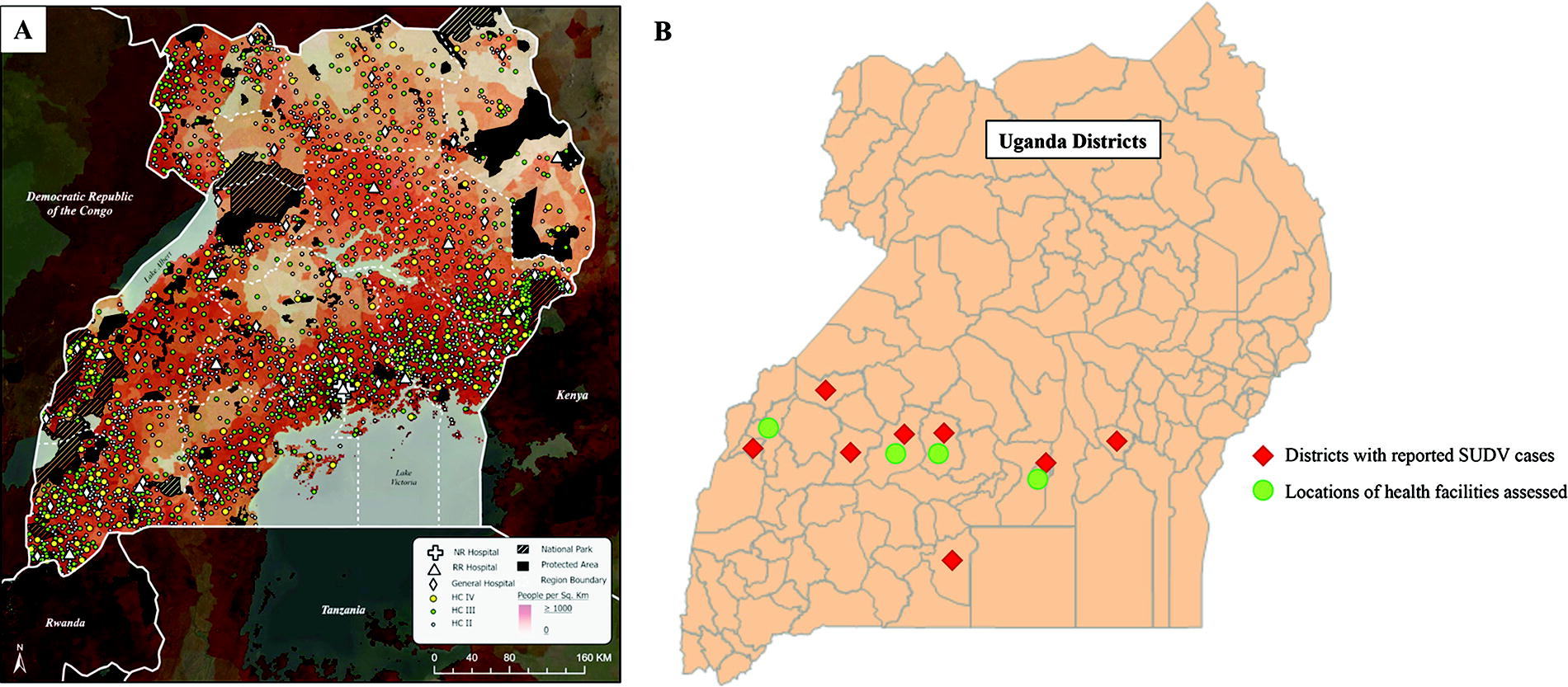
Distribution of health facilities and reported cases of Sudan ebolavirus infection during the 2022 outbreak. (A) Locations of health facilities including health centers II to IV and general, regional referral, and national referral hospitals. Reproduced from Dowhaniuk without modification. 4 (B) Districts with reported cases of Sudan ebolavirus infection during the 2022 outbreak and locations of health facilities assessed for appropriateness of ISTARI use from November to December 2022. Three health facilities were assessed in Kampala District, 2 in Mubende District, and 1 each in Kassanda and Kabarole districts. Abbreviations: HC, health center; ISTARI, Isolation System for Treatment and Agile Response to High-Risk Infections; NR, national referral; RR, regional referral; SUDV, Sudan ebolavirus.
ISTARI 1B Superuser Workshop
In December 2022, a 3-day superuser workshop with IPC leaders was held in Kampala, Uganda, at the Makerere University Infectious Diseases Institute to provide familiarization and hands-on experience with the ISTARI 1B. Participants included nurses, physicians, emergency medical service providers, and program managers serving as IPC focal points for health facilities. The workshop comprised classroom-based discussion, clinical and IPC simulation activities, and a mock site assessment with ISTARI 1B deployment in a hospital setting.
The UNMC Export Control Office, through the US Department of Commerce, determined that ISTARI 1B tents can be exported to Uganda without a commerce license.
No clinical use of the ISTARI 1B tent has been pursued, pending local medical device authorization. No human subjects research was pursued as part of the activities described in this study.
Results
Health Facility Assessment for Appropriateness of ISTARI 1B Use
Seven public and private health facilities were assessed across 4 districts in the Central and Western regions of Uganda (Figure 2B), ranging from rural health centers to a national referral hospital in Kampala. A team from UNMC assessed aspects of health facility infrastructure, scope of VHF case management, and isolation care capacity relevant for the use of the ISTARI 1B (Table 1). Designated or proposed spaces for isolating patients suspected or confirmed of Ebola disease (Sudan) or other highly communicable infectious diseases at different health facility levels are shown in Figure 3. These spaces included outdoor waiting areas (health centers III and IV), single-occupancy rooms (private hospitals), and multipatient wards (regional and national referral hospitals). Facilities also varied widely in the availability of trained staff and IPC resources to facilitate isolation care and management of infectious waste. Potential spaces for ISTARI 1B implementation were identified at each facility level (Table 1). Reliability of electrical power was a key consideration for the safe use of ISTARI tents in addition to the need for ancillary battery power. Voltage output measurements over 24 hours from an urban facility demonstrated several periods of unstable or no power up to 2.5 minutes (Figure 4).
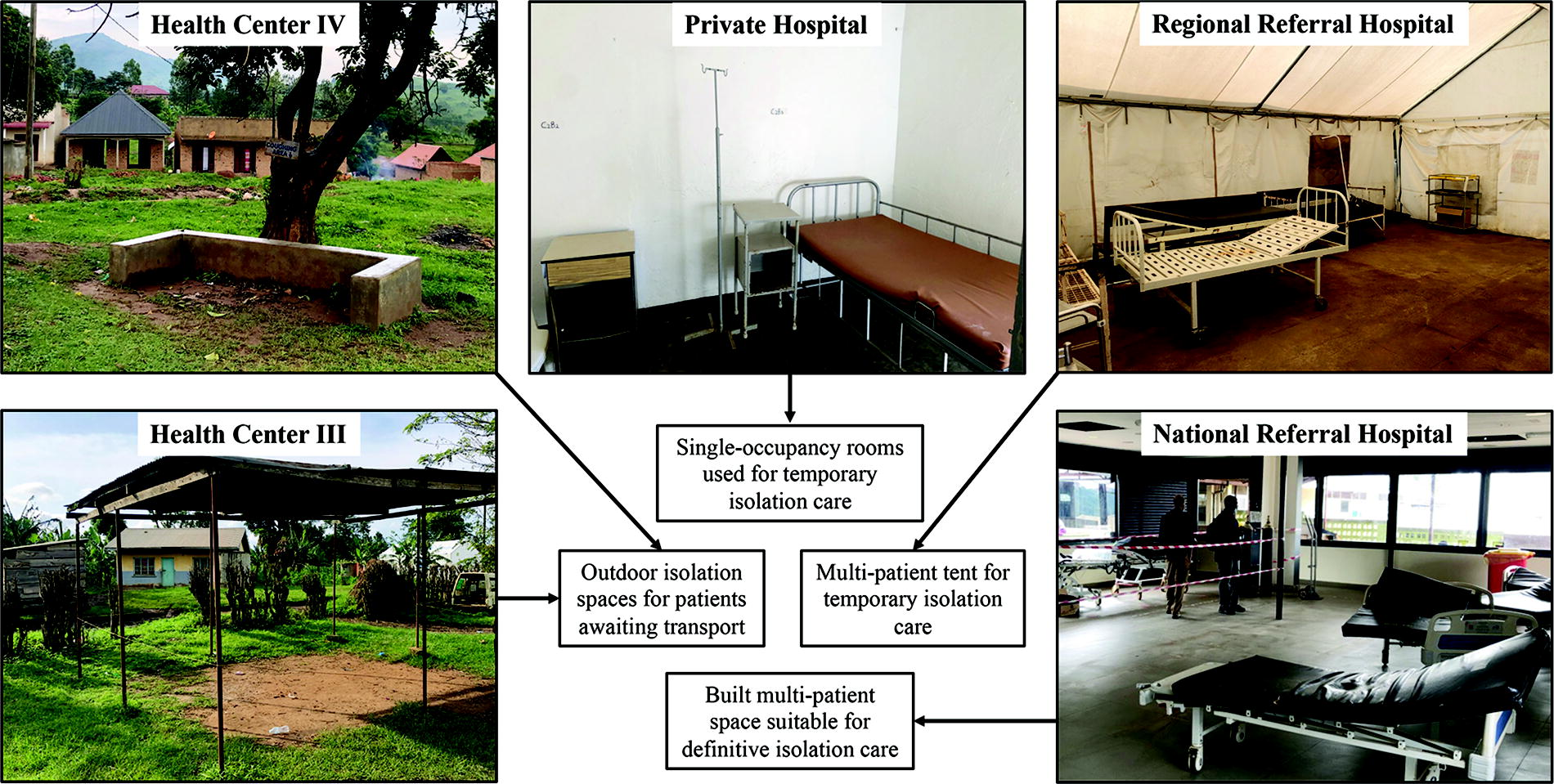
Isolation spaces across health facility levels for patients with suspected or confirmed Sudan ebolavirus infection during the 2022 outbreak.
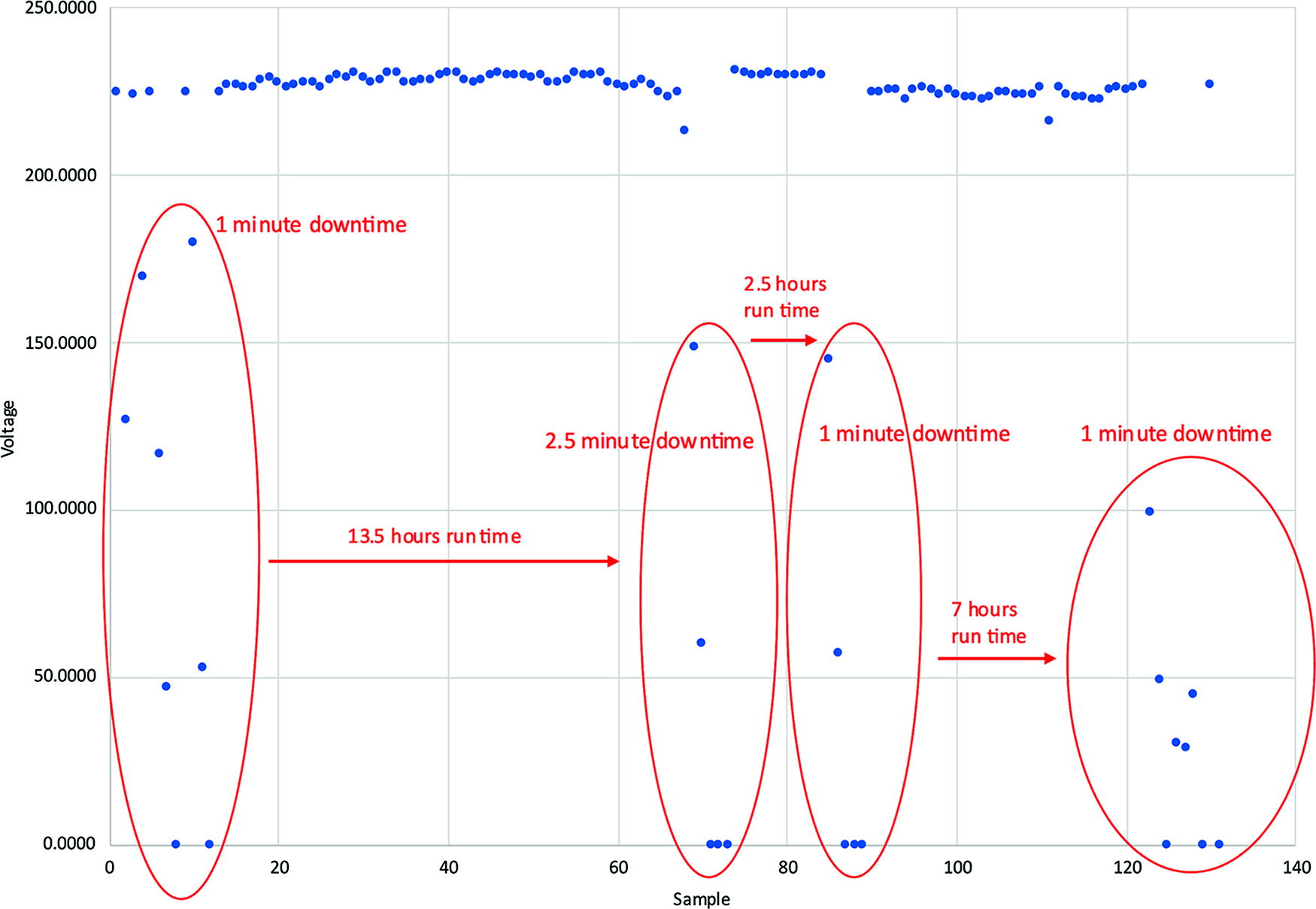
Voltage output over 24 hours from a typical wall outlet representative of a referral hospital in an urban area of Uganda. Nominal power output in Uganda is 240 V. “Downtime” denotes unstable or no power available at the outlet.
Isolation Care and Considerations for ISTARI 1B Use Across Health Facility Levels
Abbreviations: A&E, accident and emergency; IPC, infection prevention and control; ISTARI, Isolation System for Treatment and Agile Response to High-Risk Infections; OPD, outpatient department; PPE, personal protective equipment; VHF, viral hemorrhagic fever.
ISTARI 1B Superuser Workshop
The 8 objectives of the ISTARI 1B superuser workshop were to: (1) reinforce core IPC principles and practice, (2) explore ISTARI 1B use scenarios in the Ugandan context, (3) contextualize ISTARI 1B use in case management and IPC workflows, (4) become familiar with ISTARI 1B device features for clinical and IPC practice, (5) practice with IPC monitoring during simulated ISTARI 1B patient care activities, (6) develop ISTARI 1B site assessment elements, (7) gain practical experience with ISTARI 1B site assessment and initial quality check, and (8) consider ongoing monitoring, assessment, and intervention tools. The discussion topics, activities, and outputs are provided in Table 2. Key outputs included delineating appropriate ISTARI 1B use scenarios in Ugandan health facilities and developing a framework for site assessment and implementation, including ongoing monitoring, assessment, and intervention tools. Participants engaged in hands-on clinical and IPC simulation activities with the ISTARI 1B (Figure 5). Finally, recommendations regarding site assessment were incorporated into a checklist based on the use scenario of temporary holding and management of a patient with suspected VHF requiring standard care (Table 3). This checklist was used for a mock hospital site assessment in Kampala with ISTARI 1B setup and an initial quality check.
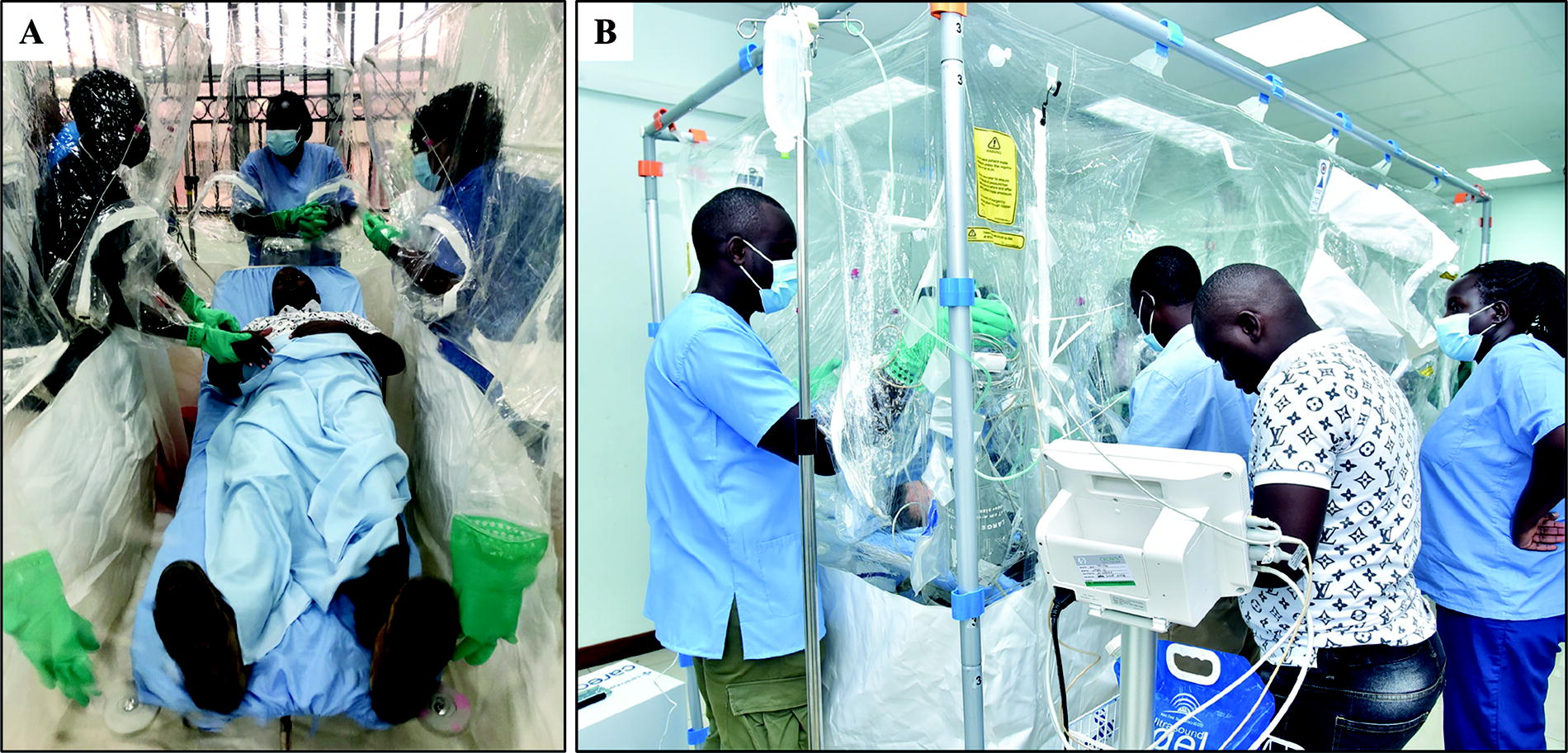
Clinical simulation activities during the ISTARI superuser training workshop. (A) A simulated patient is examined by providers in 3 lean-in stations. (B) Use of the conduit panel facilitates intravenous fluid administration and vital signs monitoring from outside of the ISTARI device. Abbreviation: ISTARI, Isolation System for Treatment and Agile Response to High-Risk Infections.
ISTARI 1B Superuser Training Content and Outputs, December 2022, Kampala, Uganda
Abbreviations: IPC, infection prevention and control; ISTARI 1B, Isolation System for Treatment and Agile Response to High-Risk Infections Model 1B; PPE, personal protective equipment; PUI, person under investigation.
ISTARI 1B Site Assessment Guide for Use Scenario: Temporary Management of Patients With Suspected VHF Requiring Standard Care
Abbreviations: IPC, infection prevention and control; ISTARI, Isolation System for Treatment and Agile Response to High-Risk Infections; PPE, personal protective equipment; VHF, viral hemorrhagic fever.
Discussion
Through the assessment of a range of health facilities and engagement with local and national IPC leaders, we identified multiple use scenarios for the ISTARI 1B tent in the Ugandan healthcare context relevant to each stage of management for highly communicable infectious diseases. As Uganda entered a late phase of enhanced surveillance in the 2022 SUDV outbreak, requiring intensive monitoring in 15 districts, suspect cases continued to be identified. A higher threshold for transport to centralized isolation facilities increased both the IPC and clinical care demands on community healthcare facilities. The ISTARI 1B tent is designed to support the safe care of patients with highly communicable diseases in nonspecialized healthcare settings, by reducing the infrastructural, material, and human resource burden associated with isolation care. The ISTARI 1B is differentiated from other isolation care systems, including the Trexler isolator 5 and the CUBE (Chambre d’Urgence Biosécurisée pour Epidémies) system introduced by the Alliance for International Medical Action (ALIMA), 6 by its portability, light footprint, ease of use, and manufacturing, distribution, and deployment scalability. The tent is 4.5 ft (1.4 m) wide, 9.8 ft (3.0 m) long, and 6.8 ft (2.1 m) tall, accommodating a standard size cot or hospital bed and a bedside commode with limited space for ambulation. This footprint makes the ISTARI 1B usable within most health facilities and amenable to arranging an array of tents in an isolation ward. The tent canopy may be decontaminated and reused for up to 10 patients; durable components (ie, tent frame, fan, manometer) may be maintained over multiple canopy replacements. Waste may be packaged, surface decontaminated, and removed through the airlock passthrough; however, the ISTARI 1B is not intended for extended length of care and waste may be packaged and stored in the tent until patient turnover occurs. In a large outbreak setting, a treatment unit may be scaled to care for dozens of patients, including temporary isolation care of patients with suspected infections pending ruleout or referral to definitive care. The ISTARI 1B offers critical advantages in this setting including direct patient observation and provision of standard care with minimal personal protective equipment, thereby reducing staffing needs and increasing the frequency of patient interactions. Key operational aspects of successful use of the ISTARI 1B in a sustained outbreak setting include the continuous monitoring of device function, the ability to repair or replace damaged components, and a reliable supply of tent canopies and replacement components.
Successful evaluation of innovative IPC tools during health emergencies requires careful consideration of underlying IPC and case management workflows. In this collaboration, the realities of Ebola virus disease (Sudan) case management, including operational challenges at facility and health systems levels, were balanced with the feasibility and potential value of ISTARI 1B tent usage. Capacity related to staff, procedures, materials, space, and utilities were incorporated into site assessments. Bringing an innovation into an emergency is a demanding task. In a 2022 analysis of time to first use of emergency vaccines in various crises, the time to launch a vaccination campaign lagged several weeks after peak case counts were experienced. 7 Challenges that any deployment of innovation may share with the vaccine experience include underlying clinical and logistical operating environments, time to meet regulatory and contracting requirements and adjust to local contexts, production limitations, export control restrictions (in some instances), storage and distribution options, and achieving equitable access and uptake. The challenges and demands required to deploy an innovation requires taking steps during the preparedness and recovery stages of health emergencies so that healthcare systems are resilient and capable of responding effectively.
The ISTARI 1B evaluation and initial training activities described here yielded the following recommendations to support its successful implementation in Uganda: (1) conduct local safety and human factors studies approved by institutional review boards; (2) implement a pilot program to assess local healthcare worker acceptability and usability across facility levels; (3) develop program tools for initial site assessment, onboarding, training, and ongoing safety and quality monitoring; (4) integrate familiarization and hands-on experience with the ISTARI 1B into training and exercises for outbreak response, and (5) accommodate for the Ugandan electrical infrastructure (240 VAC, 50 Hz) to ensure reliable operation including power downtimes. The Fantech FG 6M fan used with the ISTARI 1B tent is a UL-listed fan designed to be powered by standard US mains voltage (110 VAC, 60 Hz). A standard power inverter can step down the voltage from 240 V to 110 V (though the frequency is unchanged), and the fan has demonstrated comparable performance under these conditions without overheating. In facilities lacking backup generator power, an ancillary battery may be used to prevent loss of negative pressure in the tent during intermittent power loss. A backup 12 V lead acid battery must have a capacity of 6.5 amp hours to power the fan.
Conclusion
The processes and findings described in this case study are generalizable to the assessment of novel IPC tools for implementation in diverse healthcare settings to support preparedness and response to high-consequence infection disease events. The ISTARI 1B is a robust and highly adaptable tool with utility across multiple use scenarios to improve healthcare worker safety and the quality of patient care and minimize interruption of essential services during a health emergency response.
Footnotes
Acknowledgments
We are grateful for the guidance, collaboration, and support from the Uganda Ministry of Health Ebola Response Case Management Pillar (including Dr. Wilberforce Kabweru, Dr. John Baptist Waniaye, and Dr. Ronnie Bahatunguire), the Makerere University Infectious Diseases Institute leadership (Andrew Kambugu) and training department, and the dedicated IPC and medical professionals who facilitated and participated in health facility assessments (Maureen Kesande, Bernadatte Namugema, Micheal Lukoma, Grace Nsiimire, Prudence Nakitenda Asobola). Development of the ISTARI device family was funded by a US Centers for Disease Control and Prevention award (CK19-1905, National Infection Control Strengthening).
