Abstract
The New South Wales Biocontainment Centre is a statewide referral facility for patients with high-consequence infectious disease (HCID). The facility collaborates with researchers to adapt existing HCID procedures such as donning and doffing of personal protective equipment (PPE). However, information on how to respond safely to collapse of a healthcare provider in full PPE within a contaminated zone is scarce. To address this gap, we adapted Nebraska Medicine’s “provider down” protocol on paper and then simulated and video recorded the process, iteratively, in the facility. Clinicians analyzed the recordings collaboratively in researcher-facilitated reflexive discussions. Our primary aim was to ascertain how to maintain optimal infection prevention and control while providing urgent care for the healthcare provider. We tested participants’ suggested modifications, in repeated video recorded simulations, until consensus on optimal practice was achieved. Our secondary aim was to assess the utility of video-reflexive methods to enhance clinicians’ awareness and understanding of infection prevention and control in a rare and complex scenario. Six adaptations and simulations were discussed in video-reflexive sessions before consensus was reached; the final version of the protocol differed considerably from the first. Viewing footage of simulations in situ enabled participants to (1) identify infection and occupational risks not identified on paper or during verbal postsimulation debriefs and (2) test alternative perspectives on safe procedure. Video-reflexivity enables context-sensitive and consensus-building codesign of policies and procedures, critical to protocol development in a new unit. It contributes to a culture of teamwork, preparedness, and confidence before, rather than in the heat of, a crisis.
Introduction
R
The New South Wales Biocontainment Centre (NBC) is the statewide referral service for HCID that was purpose-built in 2021. NBC clinicians prepared for commissioning by reviewing existing local and international policies and protocols for safe work practices in the care of patients with viral hemorrhagic fever (VHF) and other HCIDs. Together with researchers, they embarked on an ongoing research project that aimed to develop context-specific policies and protocols for optimal safety, biopreparedness, and biocontainment in the new space. These protocols are taught in advance to prepare a deployment workforce for a patient who might present with an HCID. Priority was given to protocols and training for activities and incidents, for which there would be little time to work out optimal steps in the heat of a crisis. Examples include donning and doffing of HCID personal protective equipment (PPE), PPE breaches, patient transfers, and diagnostic specimen collection and transport.
Published evidence is available for some of these activities, such as safe donning and doffing of HCID PPE.9,10 However, other relevant activities are sparsely described in the literature. One of these is a “provider down” (PD) protocol, which “directs the plan to get an ill or incapacitated healthcare worker out of the patient care room, out of PPE, and provide them with the required medical care.” 11
The challenge in this situation is to maintain optimal infection prevention and biocontainment while providing urgent care for the PD in a timely manner. Although healthcare worker collapse is uncommon, it could be a high-impact event for both the PD and responders 12 and was identified as a priority for protocol development.
A literature search found no published research on this topic, but an existing protocol from Nebraska Medicine 13 was located via a Google search. In the absence of available published evidence regarding what to do in such an event, we used an “evidence-making intervention” approach to develop a protocol that is “made to work and evidenced in” the specific local setting in which it will be implemented. 14 In other words, we developed a safe and feasible PD protocol that was appropriate for the new NBC facility, using Nebraska Medicine’s provider down protocol as a starting point.
The aim of this article is 2-fold: first, to report the use of simulation and video-reflexive methods to develop, test, and evaluate a protocol for a PD scenario acceptable to all stakeholders; and second, to assess the utility of video-reflexive methods for enhancing clinicians’ awareness and understanding of infection prevention and control (IPC) in a rare and complex scenario.
Methods
Research Approach
Video-reflexive ethnography (VRE) is used globally as a means of involving healthcare workers and patients in collective sense-making and improvement of healthcare practices.
15
By videorecording real-time healthcare practices and showing the footage to participants and their colleagues, they become more aware of their ways of working that are often taken for granted. They are then able to optimize existing safe practices and devise new ways to approach suboptimal practices. VRE is guided by 4 principles
15
Exnovation – “innovation from within,” foregrounding the inherent capability and creativity of frontline actors to manage the complexity of everyday practices Collaboration – actively involving participants in data collection, analysis, and practice improvement Reflexivity – articulating and questioning taken-for-granted practices and mental models of how work is done Care – ensuring that participants’ psychological safety is a priority, to support their participation in the research.
The study design drew on our considerable, published experience with VRE focused on IPC, guided by these underlying methodological principles.16-21
VRE examines in situ hospital life as it actually takes place; but, in this case, recording a real-time healthcare worker collapse was not possible. Instead, we arranged simulations. We, therefore, describe our research approach as using video-reflexive “methods” rather than “ethnography.”
Study Setting and Participants
The study took place between April 2021 and November 2022 at Westmead Hospital, Sydney, prior to the commissioning of the hospital’s newly built biocontainment center. A nurse researcher, who is also a biocontainment nurse at the site, invited 15 clinicians who regularly attend biocontainment operational meetings and are involved in developing standard operating procedures to participate. All those invited agreed to participate and were provided with a written consent form via email or in person before the simulation. The participants included 3 infection control professionals/nurses (ICP-RNs); 3 biocontainment nurse educators (NEs); 4 infectious disease ward registered nurses (ID-RNs); and 5 infectious disease medical officers (ID-MOs). Verbal consent was obtained before any video or audio recording of simulations or reflexive sessions. Ethics approval for this study was granted by the Western Sydney Local Health District Human Research Ethics Committee (2020/ETH00553). No patients were involved in the study.
Study Design
Overview
We adapted Nebraska Medicine’s provider down protocol 13 and drafted a paper version for the NBC biocontainment unit’s quarantine rooms. We then designed a simulation scenario in which a healthcare worker collapses while caring for a patient with VHF. The assumption was that the provider’s PPE had been contaminated during patient care. This simulation was video recorded. The clinicians involved in the recording, as well as other stakeholders, then watched the footage and participated in researcher-facilitated reflexive discussions. We used their suggestions to develop an improved procedure, which was then tested in another video recorded simulation. This cycle was repeated 6 times before consensus was reached on a final protocol (see “Final provider down protocol” checklist in Supplemental Data S1, www.liebertpub.com/doi/suppl/10.1089/hs.2023.0165).
Simulated Scenarios
The simulations took place in one of the biocontainment quarantine rooms. Each simulation involved an unresponsive but breathing PD—someone who did not require CPR (cardiopulmonary resuscitation) or advanced life support. The early versions of the protocol were written as a bulleted list. Later versions were adapted for checklist instructions that could be read by an observer at the quarantine room observation desk to guide the responders. In the simulations, either a volunteer played, or a mannequin was used in, the role of PD. Roles were allocated prior to commencement of each simulation. For the video recording, a researcher used a handheld digital camera to collect roving footage, while wall-mounted GoPro cameras were used to collect wide shots. For the final version of the protocol, we used the internal room cameras to record wide shots.
Video-Reflexive Sessions
Video-reflexive researchers use the VRE guiding principles to plan and conduct reflexive sessions. 15 At the heart of the process is exnovation, whereby healthcare workers—and in some studies, patients—become more aware of the complexity of practices and are enabled to make sense of these practices collaboratively and reflexively through facilitated guidance. This process of learning through reviewing one’s own and others’ practices requires some vulnerability on the part of participants. Care must be taken by the facilitator to provide a safe environment for staff to confront and negotiate the way they work. This can be achieved in many ways. 22 In our study, we started with an overview of the structure of the reflexive session, a recap of its purpose, and an explanation that it would be participant-led with researcher facilitation. We thanked the participants who were willing to be video recorded so that improvements could be made, and we framed the session as an opportunity to learn rather than to criticize. A nurse researcher edited the video footage to convert them to clips for presentation at the video-reflexive debrief, using iMovie version 10.4 (Apple Inc., Cupertino, CA). This free application was available on the onsite MacBook Pro laptop computer and was easy and intuitive to use. It provided all the editing tools we needed to create nonprofessional clips. We also found online support for troubleshooting the software.
Video-reflexive methodology recognizes the selection of footage for clips as an act of analysis. As such, a careful process of deciding what to include and exclude, who will make these decisions, and how much footage to use, is required. 15 In many VRE studies, participants are involved in selecting the footage to be shown. Clip length depends on the time allocated to the reflexive session and the need to allow enough time for reflexive discussion. In this study, we wanted to show as much of the full process as possible at reflexive sessions, for each iteration of the procedure. Therefore, we did not include participants in selecting footage. Rather, we used editing tools to trim sections of the video where no action occurred (eg, when waiting for responders to arrive) or where known activity was occurring (eg, responders donning PPE). Cross-fades were inserted to indicate these edits to the viewer, with time elapsed indicated in subtitles where appropriate. Wide and close-up footage was used depending on which provided a clearer view of the action. The clips used in earlier reflexive sessions were shorter, for example 3.5 minutes. In later iterations, the PD procedure became more complex, and the clips shown at reflexive sessions might last as long as 15 minutes. We showed these versions to staff in 2- to 3-minute segments to allow reflexive discussion on each part of the process. We also used double speed for some sections of footage where an understanding of action would not be lost. In simulations where action was taking place in 2 places at a time, we used a split screen or a picture-in-picture edit effect to represent this to viewers.
The video-reflexive sessions were held face-to-face and online via Zoom, to allow a greater number of people to attend during COVID-19 restrictions, and were audio recorded. Debriefs took place as soon as possible after recording each simulation. On 1 occasion, an unplanned debrief was held immediately after the simulation but without video; that debrief was also audio recorded. COVID-19 restrictions in 2021 and 2022 prevented some of the face-to-face reflexive sessions, and we strove to overcome the technological, logistical, and methodological challenges involved in conducting these sessions via videoconferencing.23,24
Analysis
Data were analyzed and are presented here in response to each study aim. The first aim was to use the simulation and video-reflexive session transcriptions to provide feedback that could be used to improve the PD protocol. The feedback was derived through verbatim transcriptions, via a confidential transcription service, which were then entered in NVivo version 20.6.2 (QSR International, Burlington, MA), to analyze the unstructured text. We extracted major discussion points and presented them to the biocontainment unit staff responsible for the protocol, who then incorporated the feedback into the next iteration of the protocol.
The second aim was to assess the utility of video-reflexive methods to enhance clinicians’ awareness and understanding of IPC. To do so, we compared 2 reflexive debrief discussions focusing on the simulation recorded on March 14, 2022. The first debrief was held immediately after the simulation, but no video footage was shown at that time. The second debrief was a video-reflexive discussion held 4 months later. The 2 audio recorded discussions were transcribed and entered in NVivo to organize the data and identify instances where contamination or IPC were discussed. Excerpts of these instances were coded and then compared to identify similarities and differences between the 2 discussions.
Findings
Overview of Simulations and Video-Reflexive Sessions
[The procedure on paper] looked a lot neater before we saw the videos and I started thinking about things. (ID-MO#30; Video-reflexive 3)
A total of 6 different versions of the PD procedure were simulated and video recorded (Table). We held 5 video-reflexive sessions and 1 debrief (with no video shown) before final consensus was reached between stakeholders. There was a 7-month delay between the first and second video-reflexive sessions due to the need to acquire a scoop stretcher, and another 7-month delay between the fourth and fifth sessions due to stakeholder unavailability and admissions of mpox patients to the unit in May and June 2022. These delays reflect the stop — start nature of practice improvement in healthcare and the messy, but unavoidable, reality of research conducted in situ with busy clinicians. 14
Overview of Simulations and Video-Reflexive Sessions
Note: The list of topic discussed, decisions made, and points of procedure does not cover all that was discussed. It is beyond the scope of this article to present everything discussed at the reflexive sessions.
Number of staff required for the simulation scenario. aLength of the reflexive session in minutes and seconds. cNumber of staff at the reflexive session. dFor simulations 3 and 4, a mannequin was used as PD. For all other simulations, a clinician volunteered to play the role of PD.
Abbreviations: PD, provider down; PPE, personal protective equipment; sim, simulation.
The first simulation used a version of Nebraska Medicine’s protocol 13 adapted for the local context. The first 2 video-reflexive sessions generated discussion around what was safe and unsafe, and many people suggested adaptations. This resulted in more complex procedures, requiring many more responders, being trialed during simulations 3 and 4. These simulations led to recognition that the procedure had become too complex and therefore unsafe. Participants then explored a return to the initial PD response protocol, taking into consideration all that had been learned along the way. We found that the response worked best with a lead instructor guiding the actions using a checklist; that instructions in the checklist must be carefully constructed and clear to avoid misinterpretation by responders; and that the participants had identified the most suitable PPE for use by clinicians in different roles.
One of the key learnings was that strategies implemented to reduce risk in one area could increase risk in another. For example, introducing a scoop stretcher reduced the chance of ergonomic injury for responders, but also increased the time it took to remove the PD from the patient room, remove their PPE, and provide treatment. Using a scoop stretcher also required more responders than would be available on any shift, and therefore was not feasible. A compromise was made by returning to dragging the PD out of the room, but using an approved evacuation sheet rather than a bedsheet.
As part of the learning process, participants reflected on the value of watching the procedure in the videos, rather than reviewing on paper, and this reflection prompted our secondary analysis. We took advantage of the occasion, following simulation 4, when an immediate debrief that did not include review of the footage was followed by a video-reflexive discussion 4 months later.
Comparison of 2 Reflexive Discussion Sessions
To better assess how simulation debriefs help participants come to better understandings of the complexity of IPC in a PD scenario collaboratively, we compared the 2 debriefs held for our March 14, 2022, simulation (simulation 4). That simulation included an immediate debrief without video feedback with a debrief 4 months later that included video feedback to reflect on the value of video feedback to the content of the discussion and the learnings derived.
Eleven people were required to perform this iteration of the protocol: (1) 1 observer at the observation desk outside the quarantine room, who led the procedure using a checklist (see the “March 14 provider down protocol” checklist in Supplemental Data S2), which was read out over the intercom system; (2) 2 responders to enter the room and assess the PD (Figure 1A); (3) 2 more responders to collect the transport stretcher (Figure 1B) and transport the PD to a clean receiving room (Figure 1C); (4) 2 “clean team” staff, who prepared the receiving room (Figure 1D) and conducted parts of the procedure that required touching the PD’s skin (Figure 1F); (5) an extra nurse to don HCID PPE and take over care of the VHF patient; (6) an extra observer to take over the observer role for the VHF patient room; (7) an extra nurse to act as a runner for equipment and to direct the medical emergency team; (8) the PD, who, for this simulation, was replaced by a mannequin dressed in HCID PPE (Figure 1A).
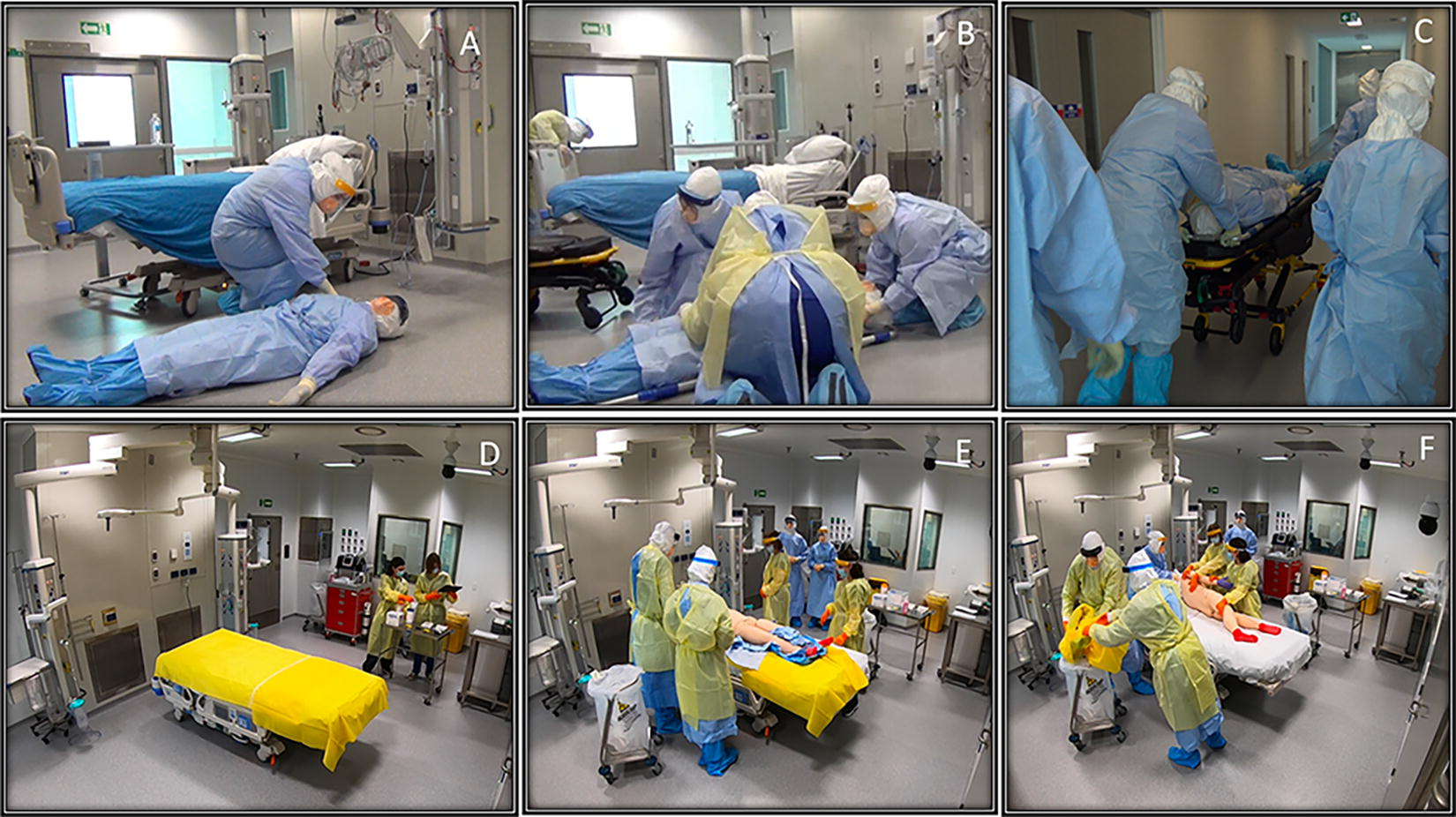
Still shots from video footage of simulation 4, using scoop stretcher. (A) First responder assesses PD who has collapsed in the patient room. (B) Three other responders arrive with scoop stretcher. (C) PD is transferred to a clean receiving room. (D) “Clean team” prepares the receiving room. (E) PPE is cut off and forms a clean layer beneath PD. (F) PD is rolled off the contaminated PPE and impervious drape and is now on a clean sheet ready for treatment. Abbreviations: PD, provider down; PPE, personal protective equipment.
In the debrief immediately following the simulation without video feedback, IPC was discussed in brief conversations about which vital signs could be taken while the PD was still in PPE, without contaminating the clinician. We also noted an instance where a contaminated gloved hand was used to open a door that was malfunctioning. The most time spent discussing IPC was in discussing contamination of the receiving room while removing PPE from the PD.
ID-MO#1: So, I guess what we are talking about is a [PD] that has been doffed and cleaned, is in an otherwise clean space?
NE#1: But it is not a clean space, you guys have been touching everything, like with the dirty PPE.
However, what items or spaces might have been contaminated was not specifically mentioned, except for the crash cart. The main concern was whether the room was clean enough for the medical emergency team to enter and provide care for the PD. However, removing the PD from the now-contaminated room also posed problems. Would the bed be clean enough for transfer out of the room?
ID-MO#5: How do they get to ED [emergency department]?
NE#1: So, we’re hoping, that bed is clean enough, and then it can go [down lift to ED].
Logistically, not all members of the medical emergency team can be trained in HCID PPE in advance (due to the high numbers and rotation of staff), and there is no time in this situation for just-in-time training. So, important IPC risks were raised in this debrief, but few mitigating strategies were identified.
The video-reflexive session for this footage was delayed 4 months due to a combination of participant unavailability and activation of the unit. The third ICP-RN, who did not attend the first debrief (without video), was present at this discussion. While watching the footage, participants noted several PPE breaches, including when a responder’s potentially contaminated booties touched his clean scrubs as he crouched down to place the PD on the scoop stretcher (see Figure 1B).
NE#3: Look at [ICP-RN#1’s] untied gown. You can see where he crouches down and he essentially sits on his booties with no protection between his scrubs underneath.
NE#1: It’s not often we’re crouching down on the ground like this, and it does all sorts of things to our PPE.
Subsequently, it was decided that coveralls should be worn by responders, to avoid contamination of scrubs during this procedure.
Approximately 20 minutes of the 50-minute video-reflexive session was spent considering cutting techniques for removing the PD’s contaminated PPE and then rolling the PD onto a clean sheet, ready for treatment. Figure 2 shows screenshots from this section of the procedure. The participants had differing views on potential contamination risks. While watching this section of footage, some noticed the potential for the PD to become contaminated by the cut PPE, which had been folded so the contaminated side was not in contact with the PD’s skin. Others in the group did not immediately comprehend the risk.
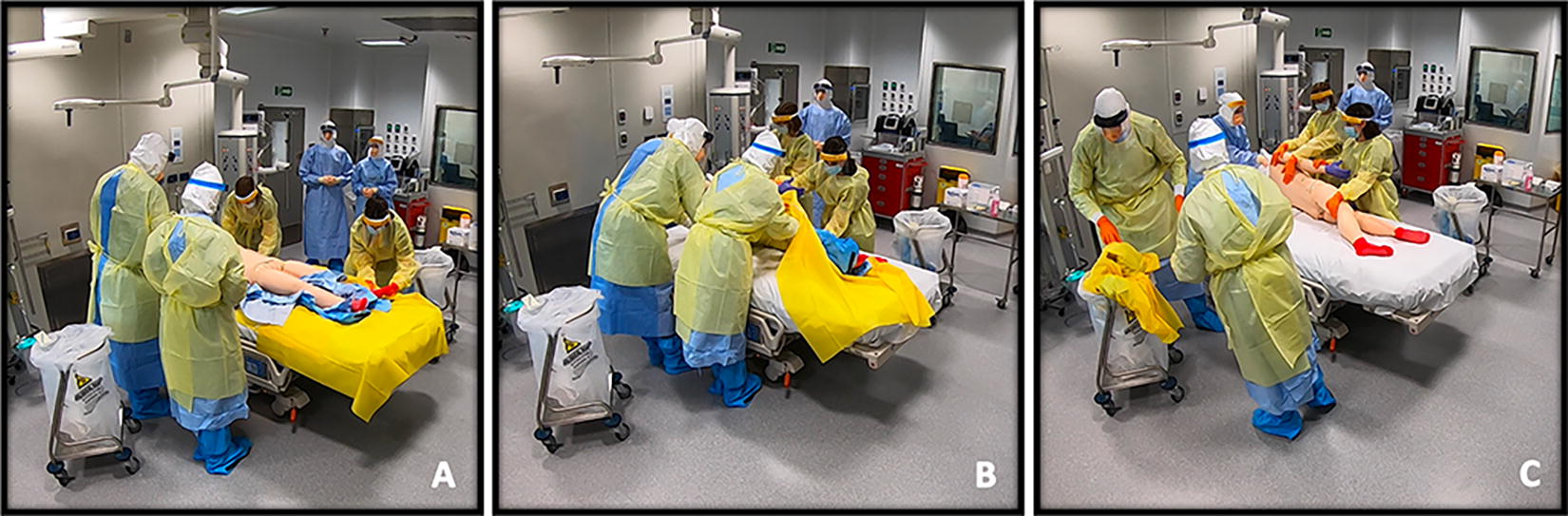
Still shots from video footage of simulation 4 showing removal of PPE. (A) PD is lying on top of an impervious drape. The cut PPE has been laid out with contaminated side facing the bed and clean side facing up to form a clean layer under PD. “Clean team” (on the right of the bed) tucks clean underside of the drape under PD, but over the clean PPE layer, in preparation for removal. (B) Contaminated team (on the left side of the bed), careful not to touch the clean layers of drape or bed, rolls PD toward the clean team, who can touch PD with their clean gloves. (C) Extra member of contaminated team supports PD’s head, holding it away from dirty side of the hood. Contaminated team pulls contaminated PPE and drape away from PD. Person supporting PD head slides PD’s hood off before laying PD head on the clean bedsheet. Abbreviations: PD, provider down; PPE, personal protective equipment.
ID-RN#3: But just looking at [the footage]…the way [the contaminated responders] pulled [the yellow sheet] out from the other side…[the PD’s contaminated PPE] is going to touch the [PD’s] skin.
NE#1: They’re lying on a clean bed of PPE and then you’re putting a clean layer of the yellow sheet onto that clean layer of PPE. So, I’m not sure where the dirty parts are hitting the [PD]?
ID-RN#3: If you watch it back, [the contaminated responders] are pulling it out…
ID-RN#2: …so that [the folded PPE] unrolls [and the contaminated side touches the PD].
The participants were then able to rewatch the footage and come to a shared understanding of the risk.
Other concerns expressed included that the PD might become contaminated by the contaminated responder who was stabilizing the PD’s head during the roll and that the contaminated team’s gowns were in contact with the sides of the bed, which would a problem if the bed were to be moved to the ED. Through repeated viewings of this section of footage, they were able to reach consensus and devise solutions to mitigate these risks. Solutions included ensuring that PPE is flattened, clean side up, to ensure that the contaminated side does not touch the PD; and using a longer drape to protect the bed. They also realized that the PD’s head was not contaminated during the procedure.
Discussion
The core principles of IPC are applied in all aspects of healthcare to curb the transmission of pathogens between patients, staff, and visitors. 25 Our study investigated how clinicians negotiate the application and enactment of IPC principles when applying them to a new and unusual context where the stakes are very high for all concerned.
An important component of effective learning through simulation exercises is a facilitated debrief.26,27 Methods such as scripted debrief, reflective discussion, and performance critique (to name a few) have been used to guide debriefs, so that participants gain a better shared understanding of their actions and thought processes.28,29 However, no gold standard for debriefing has been identified. 26 Using video feedback in simulation debriefs has been described as an adjunct technique that has not yet shown enough evidence of benefit in enhancing debrief outcomes. 26 Our findings show that simulation debriefs with and without video feedback contribute significantly, but differently, to enhancing participants’ awareness and understanding of IPC.
The debrief without video created an awareness of several potential problems in the PD protocol being developed. In particular, the multidirectional discussions between the different members of the multidisciplinary team promoted a shared understanding of the receiving room becoming increasingly contaminated.30,31 This had not been recognized in previous simulations, where people had concentrated more on how to safely cut the PPE off the PD. However, by the end of the debrief in question, participants were still not clear on exactly what became contaminated and how this could be addressed. It was understood that the crash cart, the bed, and the PD might become contaminated, but when, and by what, was not fully identified.
The video-reflexive debrief created a more specific and detailed awareness of contamination risk. Participants were able to examine the minutiae of the protocol and recognize that transmission prevention practices are not always discrete and easily defined sets of actions. Rather, they involve what has been described as infection control “boundary work,” 32 where clinicians are required to move “bodies and objects across multiple, often invisible boundaries demarcating potentially or actually contaminated, from clean or protected spaces.” 17 During the nonvideo debrief, participants started to gain insight into these boundaries in broad terms—including contaminated PPE touching things in the clean receiving room, and a potentially contaminated bed being taken down to the ED. While watching the video footage, however, participants responded by identifying and focusing on particular moments of transmission risk and then exploring specific causes and solutions. Moreover, when participants diverged in their interpretations of potential transmission risks, the footage could be paused and replayed again and again until understood by all parties. 33 For example, realizing that the PPE, which had been cut between the PD’s legs, was not arranged well enough before rolling the PD, the participants watched the footage repeatedly and then reached consensus on a solution: to flatten the contaminated side of the PPE so it would not touch the PD.
Review of video footage following a simulation has previously been acknowledged as an effective, if underutilized, method to identify insights into potential safety issues that are not recognized in conventional debriefing.27,34,35 Our approach to video debriefs was based on the 4 video-reflexive methodological principles.15,36 The principles of exnovation and reflexivity required participants to deliberate and gain new insights into their existing IPC practices, observing them in real time and explaining to their peers why they do things in certain ways. Participants were able to examine what was working well, allowing them to optimize these practices if possible or devise change if required. 36 Collaboration foregrounded the expertise of these biocontainment staff (rather than the researchers or simulation experts) to identify infection transmission risks and to suggest solutions most appropriate for their working life. Care was taken to ensure participants felt safe and respected while confronting visual evidence of their ways of working. 37 Our findings provide many examples where various viewpoints were raised and negotiated between members of the multidisciplinary team, ultimately eliciting learning about the complexity of the procedure and its effect on the accomplishment of effective IPC. This finding is in keeping with other video-reflexive studies on this topic.15,17,18,20
Video is critical to stimulating reflexivity, as it reproduces the dynamics and complexity of the scenario for all involved, making it tangible and discussable. In a realistic simulation, it is difficult for participants to be aware of, or remember, all that was done or said, especially if the task is complex. 31 By using video feedback, participants do not have to rely on memory or recall. 15 Our PD scenario was complicated in that several locations were involved: the VHF patient room, the transfer corridor, and the receiving room where the PD was taken. In the simulation we examined in depth, the staff in the receiving room did not experience what happened in the room where the PD collapsed, and the first responder did not experience what it was like setting up or waiting in the receiving room. By referring to video in the debriefs, all stakeholders, including those who were unable to attend the simulation at all, were given an opportunity to understand all aspects of the simulation.
It is worth noting that the process of developing the PD protocol commenced in April 2021 and was not completed until November 2022. This extended time period was due in part to the challenges of getting busy clinicians together for simulation and debrief meetings, particularly as simulations to develop other protocols were being run during the same period. But the delay was also a consequence of designing a new protocol and analyzing it so closely using video footage. As can be seen in the Table, the early simulations, based on Nebraska Medicine’s protocol, required only 6 to 7 staff on duty to respond to a PD event. As participants identified transmission risks and designed solutions, each iteration of the protocol became more complex and required more people to respond. In trying to accommodate all suggestions and ameliorate all transmission risks, the procedure became too complicated, required too many staff to respond, and was, therefore, considered unsafe by stakeholders.
We must note some limitations. The long time period needed for this process may hinder the promotion of simulation and video-reflexive methods for protocol development. However, the iterative protocol versions and discussions greatly increased the team’s situational awareness of cross-contamination risks and of ways to mitigate these should a colleague collapse when caring for a patient with a VHF. Considering the difficulty, in high-risk/low-frequency emergency events, of recalling and adhering to protocol, 38 it is likely that the learning that occurred over this protracted period of reflection will contribute to better outcomes. While this does not form part of the research data, we continue to test the PD protocol in simulations carried out during deployment team training sessions, and to make modifications based on participant feedback. Including the use of fluorescent powders or gels in future sessions, to test for contamination, could help validate the protocol.
Additionally, with regard to our second aim, we had only 1 simulation that could provide a comparison of debriefs with and without video feedback. Although we assume that, with the same set up, other simulations would provide similar feedback, we do not have the data to support that assumption. Simulations with other set ups (eg, fewer rooms or personnel involved) would likely provide additional or differing feedback.
Conclusion
Our study demonstrated the utility and advantages of simulation combined with video-reflexive methods to develop procedures for dealing with a complex, uncommon, and high-stakes situation in a biocontainment facility. The reflexive and collaborative components of the video-reflexive methods stimulated stakeholders’ awareness of, and development of practical responses to, infection transmission risks in the PD protocol. In this way, video-reflexive methodology contributes to a culture of teamwork, preparedness, and confidence before, rather than in the heat of, a crisis.
Footnotes
Acknowledgments
The authors thank the healthcare workers who participated in this study for providing insights that contributed to protocol development. This work was supported by the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
