Abstract
A high-level isolation unit (HLIU) is a specially designed biocontainment unit for suspected or confirmed high-consequence infectious diseases. For most HLIUs, maintaining readiness during times of inactivity is a challenge. In this case study, we describe a checklist approach to assess HLIU readiness to rapidly operate upon activation. This checklist includes readiness criteria in several domains, such as infrastructure, human resources, and material supplies, that are required to safely activate the unit at any time. The checklist audit tool was derived from a novel activation readiness checklist published by the biocontainment unit at The Johns Hopkins Hospital in Baltimore, Maryland. It was then adapted for the Irish healthcare setting and implemented at the Mater Misericordiae University Hospital, Ireland’s current isolation facility. Results from the audit were also used to inform recommendations for the construction of a new HLIU to open in 2025. The audit tool is user friendly, practical, and focuses on the essential elements of readiness to ensure a successful rapid operation.
Background and Aim
T
HCID cases are generally rare in Western Europe, with the majority of patients infected while abroad; although, some locally acquired cases of Crimean-Congo hemorrhagic fever have been reported in Spain.4,5 These cases have been cared for in specially designed high-level isolation units (HLIUs). The HLIUs are equipped with advanced engineering and administrative controls, personal protective equipment (PPE), and trained staff to mitigate the transmission risks HCIDs pose within healthcare settings, as demonstrated by nosocomial transmission of MERS in South Korea in 2015 and healthcare worker infections of Ebola virus in a non-HLIU setting in the United States during the 2014-2016 West Africa Ebola virus disease (EVD) epidemic.6,7
The Mater Misericordiae University Hospital (MMUH) is the home of Ireland’s only HLIU. The current isolation facility for HCIDs was established in 2008 in an adult infectious diseases ward located on the ground floor of the hospital. The HCID isolation rooms have interlocking doors, a designated room for PPE donning and doffing, and an air handling system that supports the negative pressure isolation rooms (categorized as Class Q rooms according to international health facility guidelines). 8 During times of inactivity, these rooms are used for regular inpatient care, particularly for patients who require airborne and droplet precautions. HCID diagnostics are processed in the National Viral Reference Laboratory located in Dublin, approximately 8 kilometers from MMUH.
The 2014-2016 West Africa EVD epidemic and resulting cases of EVD cared for in other European HLIUs reinforced the value of having a specialized HCID facility in Ireland. However, based on international guidelines for HLIUs published by US and European consensus groups,9,10 the MMUH isolation facility had known structural deficiencies, including the absence of an onsite biosafety level 3 (BSL-3) laboratory capacity and the fact that the isolation facility did not meet ICU specifications. In 2019, we sought an external review of the MMUH isolation facility by the clinical lead of the HLIU at the Royal Free Hospital in London. 11 Based on benchmarking of the MMUH facility against published HLIU guidelines10,–14 and European Centre for Disease Prevention and Control HLIU standards, 15 several critical shortcomings were identified in the unit’s ability to safely manage patients with HCIDs. As a result, a newly constructed HLIU—reconfigured with a dedicated ICU, onsite BSL-3 capacity, onsite waste autoclaves, and other recommended HILU capabilities—was commissioned and is set to open in 2025.10,12–15
Until construction of the new HLIU is completed, the current isolation facility at MMUH remains the primary referral center for HCID cases in Ireland. In 2022, 5 patients with suspected HCIDs were cared for in the existing isolation facility. These included 2 suspected cases of Lassa fever, both negative on polymerase chain reaction tests, and 3 confirmed mpox infections diagnosed early in the outbreak when mpox was still classified as a Category A pathogen (Figure). Lessons learned from these activations included the need for improved processes for packaging and transfer of Category A samples for HCID diagnostics, the importance of clear internal and external communication pathways, enhanced training on PPE donning and doffing, and having a cohort of trained staff prepared for the possibility of multiple sequential activations.
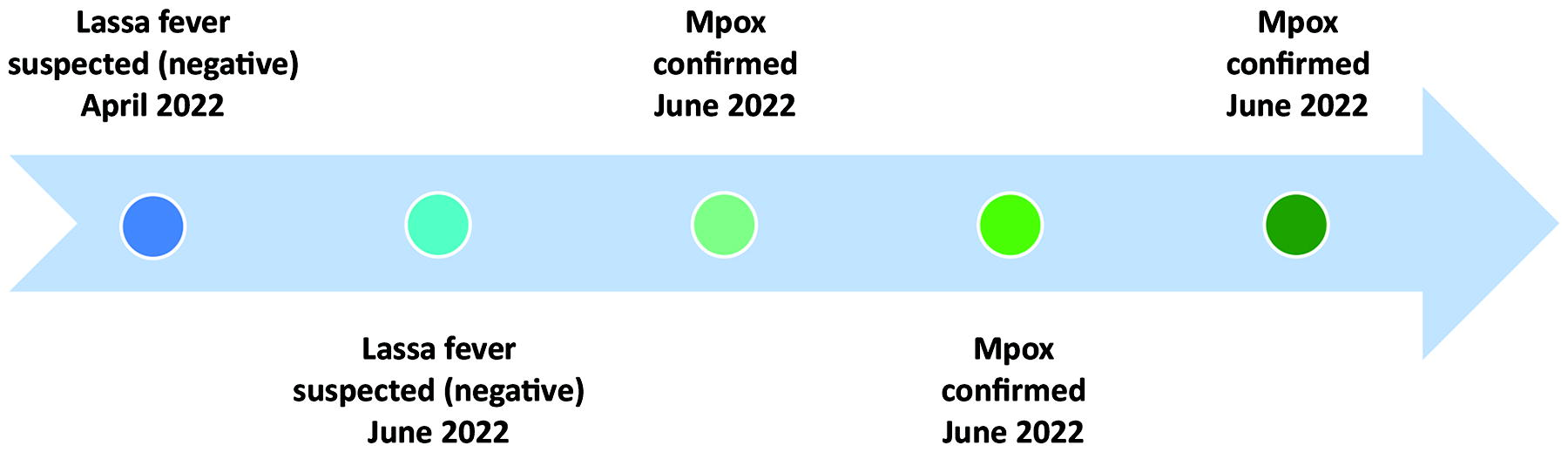
Timeline of activation for the high-level isolation unit at Mater Misericordiae University Hospital, April to June 2022. Mpox was a Category A pathogen during this timeframe.
The occurrence of suspected and confirmed HCID cases reinforced the importance of using a systematic approach to maintaining a state of readiness during standby periods. As a result, our team sought to develop an audit tool to maintain readiness of the current isolation facility during times of inactivity, with plans to adapt and continue to use the process when the new HLIU opens in 2025. After a review of various checklists and standards developed by the European Network for Infectious Diseases Physicians (EUNID), 12 the US National Emerging Special Pathogens Training and Education Center (NETEC), 14 and the Permanent Working Group of Competence and Treatment Centers for High Consequences Infectious Diseases (STAKOB) in Germany, 13 among others, we decided to adopt the checklist approach used by the biocontainment unit (BCU) at The Johns Hopkins Hospital in the United States due to its simplicity and easy application in clinical practice.16,17 We aim to describe our process of adapting and implementing a checklist to assess unit readiness for activation for an HCID case, as well as lessons learned. Because this was the first formal tool we developed to assess the readiness of our unit, we believe our experience could be useful for other units seeking to develop an audit process.
Audit Process
The audit process was conducted in 3 stages: (1) adapting the Johns Hopkins BCU readiness checklist to the MMUH facility and defining unit “readiness”; (2) conducting the first audit of the MMUH isolation facility in November 2022; (3) completing the second audit of the MMUH isolation facility in May 2023.
Adapting the Checklist to an Irish Healthcare Setting
The Johns Hopkins BCU readiness checklist consisted of 4 domains of readiness: waste handling, facilities (air handling system, biosafety cabinet, lights, running water, unit entry security), PPE, and staffing. 16 Each domain was assigned a color depicting the scale of readiness. The Johns Hopkins BCU team assessed their facility using the checklist in 2 stages. The first stage occurred daily for 50 days, while the second stage (reassessment) took place 7 months after the initial assessment.
Modification Process and Key Changes Made
Our first step was modifying the checklist to fit the operational plan of the MMUH isolation facility and the context of the Irish healthcare system. A team of 3, which included the MMUH isolation facility clinical nurse specialist, clinical lead, and infectious disease fellow, worked on adapting the checklist. The team first reviewed the Johns Hopkins checklist and decided to keep the overarching approach of the scale of readiness and domains. The Johns Hopkins audit tool was designed to assess activation readiness for 1 suspected or confirmed HCID case, either a viral hemorrhagic fever or highly pathogenic airborne infection. The period from notification (accepting patient referral) to “activation” (ready for patient arrival) of the unit was defined as 8 hours.
We made 3 key changes in adapting the Johns Hopkins checklist: expanded the domains, updated the terminology, and revised staffing numbers. The 4 domains used in the Johns Hopkins checklist were expanded to 6 domains, with the inclusion of training and air handling systems. These 2 new domains were incorporated into the Johns Hopkins checklist under the staffing and facilities domains, respectively. In the new audit checklist, we specifically created them as standalone domains because they were considered essential elements to our isolation facility’s readiness. In addition to expanding the domains, we modified a few terms to suit the Irish healthcare setting, including changing “healthcare provider” to “doctor,” “laboratory technician” to “laboratory medical scientist,” and “trained observer” to “runner.” In the staffing domain, respiratory therapists and health epidemiologists were omitted from the activation team, as these positions do not exist in the Irish isolation unit setting. Further, we defined staffing domain numbers according to our predefined rostering considerations. After making changes to the checklist, the tool was reviewed and approved by the isolation unit team and approved by the hospital audit ethics committee. The final audit checklist is shown in Table 1.
National High-Level Isolation Unit Audit Checklist a
Adapted from Johns Hopkins Biocontainment Unit, described in novel approach to maintaining unit readiness during inactivation times. 16
Abbreviations: CNM, clinical nurse manager (nurse coordinator); PPE, personal protective equipment; RN, registered nurse.
Determinants of HLIU Readiness
Our checklist provides metrics to assess the readiness of the HLIU to rapidly activate. The 6 domains were further categorized into 3 readiness capabilities, as used by the Johns Hopkins BCU: “Ready,” “Ready With Considerations,” or “Not Ready.” The MMUH isolation unit is considered ready to activate if all domains meet the criteria for being “Ready.” If 1 or more domains are deemed “Not Ready,” the unit is categorized as “Not Ready” to activate due to the risk to unit staff and other patients if the unit were to be activated. If 1 or more domains are determined to be “Ready With Considerations,” that implies that changes are required; however, those changes would need to be achievable within 8 hours of activation.
The isolation unit was considered safe to activate if the criteria in all the following domains were deemed “Ready” (see Table 1 for the scale of readiness):
Conducting the First Audit, November 2022
The adapted checklist was used to conduct an audit of the MMUH isolation facility readiness for activation from November 1 to 7, 2022. On November 1, the clinical nurse specialist and the infectious disease fellow leading the audit process conducted a walkthrough of the unit using the checklist to audit the domains of waste management, PPE, air handling system, and general facilities. They then worked with a clinical staff facilitator, who leads the unit staffing and training program, to audit the training and staffing domains, which were assessed retrospectively using available logbooks. The ensuing 6 days of the audit consisted of a series of meetings between the clinical nurse specialist, infectious disease fellow, clinical staff facilitator, and 2 infectious disease consultants, including the unit’s clinical lead. The meetings were used to establish consensus on the scale of readiness assigned to each domain, which was finalized on November 7. Strategies to enhance identified gaps were created and implemented in subsequent months.
Completing the Second Audit, May 2023
A second audit was conducted in May 2023 using the same process, team, and checklist tool as used in the November 2022 audit. The timing of the audit coincided with the MMUH audit cycle that allows for reauditing after 6 months. The second audit was again conducted on all 6 domains to assess readiness and whether the changes made following the first audit improved unit readiness.
Findings
The adapted audit checklist proved to be user friendly and applicable to the MMUH isolation facility. It was a practical tool for visualizing unit readiness and identifying immediate barriers to activation. Results from both audits are described in detail as follows.
First Audit, November 2022
The overall assessment of the current isolation facility after the first audit determined that the unit was not ready to activate, meaning that it did not meet the criteria for “Ready” in all 6 domains. One domain (general facilities) was deemed “Not Ready” and 2 domains (staffing and air handling system) were assessed as “Ready With Considerations.” Therefore, barriers were identified to safe activation of the facility.
Waste Management: Ready
While we consider onsite waste activation by autoclaves as the preferred strategy for Category A waste management, based on published HLIU guidelines, 11 the current isolation facility does not have an onsite autoclave. An alternative condition for “Ready,” a defined Category A waste management policy, was available to enable staff to package and transport waste to an offsite incineration facility safely (Supplementary Data S1, www.liebertpub.com/doi/suppl/10.1089/hs.2023.0148).
PPE: Ready
At the time of the audit, the isolation unit had an onsite supply of PPE for 72 hours and an offsite supply for 21 days for 2 patients with HCIDs. The offsite supply is located in a nearby storage facility and is easily accessible. The PPE supply for training is included in the stock and is sufficient for 3 years of training. PPE requirements were calculated using the US Centers for Disease Control and Prevention calculator tool to estimate PPE for healthcare facilities. 18 Current PPE is supplied by a German company that outsources production in Poland. For consistent quantities, production time ranges from 6 to 8 weeks plus 1 week for shipment.
Training: Ready
Although the staffing domain was deemed “Ready With Considerations” due to insufficient available staff during the first audit, over 75% of the available staff were trained. Based on the data for staff training garnered from logbooks, 37 nurses, 7 consultants, and 6 registrars had completed PPE training within the 3 months prior to the audit.
Staffing: Ready With Considerations
At the time of the first audit, staffing was deficient, as only 37 nurses were available to staff the unit for a week. A minimum of 40 nurses are required per week to be deemed “Ready.” We calculated the number of required nurses to activate the isolation facility for a week based on a calculation that is used by nursing managers at our hospital to develop nursing rosters. The criteria used for calculating the number of staff needed for an activation of the HLIU were:
Length of the day shift (hours) Number of nurses required for each day shift (depends on time allowed in PPE) Length of the night shift (hours) Number of nurses required for each night shift (depends on time allowed in PPE) Sickness and absence level (percentage)
The calculation was made by multiplying the length of the shifts by the number of nurses, and then multiplying that total by the absence level percentage. The sum gives the hours of nurses needed for 1 day, which is multiplied by 7 days. The result is then divided by the weekly contractual hours.
Of the 40 nurses per week, 23 are involved in direct patient care, 5 are clinical nurse managers, and 11 are nurse “runners.”
A restricted time of 3 hours in full PPE was adopted when calculating staffing requirements. 19 Nurses are divided into 2 teams, each consisting of 2 nurses who work inside the patient’s room, and they rotate every 3 hours. One nurse runner is positioned outside for assistance. Two nurse managers per 12.5-hour shift would also be rostered.
Staffing requirements would depend on the patient’s condition; more specialist ICU nursing and doctor staff would be required for a critically ill patient. In the first audit, 37 nurses were available; therefore, the domain was considered “Ready With Considerations.”
Risk mitigation: To overcome staffing challenges, a process of training newly hired staff and hospital-wide education sessions on HCIDs were implemented more regularly to improve awareness and participation in training and activation. Providing backfill for trained nurses was identified as a strategy to immediately staff the unit during activations. In addition, a proposal to provide an “activation allowance” or monetary uplift for staff during unit activation was considered. The MMUH isolation unit roster at the time of the audit included nurses from the infectious diseases department; therefore, recruiting nurses from other wards for regular training was proposed to maintain unit readiness in the staffing domains.
Air Handling System: Ready With Considerations
The existing isolation facility has 2 negative pressure isolation rooms with anterooms (Class Q). The minimum differential pressure between the isolation room and ambient pressure was appropriate at 10 Pa. The air handling system was functioning, meeting the standard of 12 air exchanges per hour. In the audit, we discovered that calibration was done annually.
Risk mitigation: We recommended that calibration checks occur every 6 months.
General Facilities
Water System: Ready
At time of the audit, there was a working policy for liquid waste, which included the process of solidification by adding Vernagel (Vernacare, Chorley, United Kingdom) and collecting waste in 60 L rigid bins. It was then packaged and transported with other Category A waste to the identified incineration site.
Security Measures: Ready With Considerations
Although entry to the unit was restricted by mandatory swipe access, the main entry door to the unit was shared with another ward.
Risk mitigation: During times of unit activation, inpatients of the other shared ward will be redistributed to other locations in the hospital. During unit activation, entry is permitted only to the unit staff by security personnel.
Onsite Laboratory: Not Ready
Diagnostic testing of suspected HCID cases is performed in the National Viral Reference Laboratory in Dublin. The isolation facility has an onsite arterial blood gas analysis machine (ABL90 FLEX, Radiometer, Dubai, United Arab Emirates), which was validated and functioning. However, routine laboratory tests in MMUH were not feasible due to national and hospital laboratory restrictions. To minimize staff exposure, a local agreement was in place to limit routine point-of-care testing to arterial blood gas machines.
Risk mitigation: The lack of ability to perform routine laboratory tests was a major gap in readiness. Although the HLIU that is scheduled to open in 2025 was already in the early design phases, results from the audit on gaps related to laboratory capabilities were escalated through appropriate hospital governance structures to reinforce the need for onsite BSL-3 laboratory capabilities within the new facility. Meanwhile, an agreement was established to transfer confirmed patients with HCIDs diagnosed in Ireland to an HLIU in Germany for medical management until construction of the new HLIU at MMUH is completed.
Key Recommendations
The key recommendations resulting from this audit are outlined in Table 2. Although the new HLIU was already well into the design phase during the first audit, results from the audit were escalated through appropriate governance structures within the hospital to recommend the new facility be equipped with 2 onsite autoclaves, an onsite BSL-3 laboratory, and a safe system for liquid waste management.
Recommendations for Maintaining Readiness to Activate MMUH HLIU
Indicates recommendations for the new HLIU, set to open in 2025. Other recommendations were implemented after the first audit in the existing isolation facility.
Abbreviations: BSL-3, biosafety level 3; HLIU, high-level isolation unit, MMUH, Mater Misericordiae University Hospital.
Second Audit, May 2023
The existing MMUH isolation facility was reaudited on May 2, 2023, 6 months after the initial audit. The waste management, PPE, and training domains that were assigned “Ready” in the November 2022 audit were reassessed as “Ready.” As a result of the gaps identified and changes implemented after the first audit, the air handling system and staffing domains improved from the “Ready With Considerations” category to “Ready.” The air handling system domain changed to the “Ready” category after the maintenance department scheduled calibration of the air handling system to occur every 6 months, rather than annually. During the second audit, 43 trained nurses were available to activate the unit for a week, compared with 37 nurses in the first audit. Staffing improved by having the clinical nurse facilitator, who also engaged in PPE training, actively recruit staff who were newly hired at the hospital. The general facilities domain remained “Not Ready” due to the lack of BSL-3 laboratory capabilities. As such, the unit continued to be assessed as “Not Ready,” and the agreement with Germany to transfer confirmed patients with HCIDs to a German HLIU remains in place until construction of the new HLIU is completed.
Discussion
We adapted the Johns Hopkins BCU activation readiness checklist to create an audit tool that fits the context of an Irish healthcare setting. By implementing our adapted readiness checklist, we were able to identify gaps and barriers that impede successful unit activation at MMUH. The adapted readiness checklist, the first formal tool used to assess readiness of the MMUH unit, was user friendly and easy to adapt. Implementing changes after the first audit led to improvements in the air handling system and staffing domains at the time of the second audit. Other domains that involve the infrastructure, specifically waste management and general facilities, will improve with the completion of the newly designed HLIU. Until the new HLIU is operational, the current plan is to transport a confirmed patient with an HCID diagnosed in Ireland to an HLIU in Germany; however, during diagnostic testing and until a confirmed patient with an HCID is transported, the MMUH isolation staff would need to care for the patient within its unit. Activations for suspected cases of Lassa fever and confirmed cases of mpox early in the weeks of the 2022 mpox outbreak reinforced the importance of maintaining an active state of readiness at the MMUH unit. This audit tool was helpful in identifying barriers to activation and areas for improvement, and it will continue to be used on a 6-month cycle in the existing isolation facility and when operations are transferred to the new HLIU.
Impediments to unit readiness in our audits included the lack of onsite autoclaves, onsite liquid waste treatment, and an onsite BSL-3 laboratory; however, protocols are in place for proper waste management that ensures activation readiness in the waste management domain until the new HLIU is operational. Processes for managing biohazard waste vary internationally. The HLIU in London uses an onsite passthrough autoclave, whereas the UK Newcastle HLIU manages the waste by transporting it to an approved waste contractor for incineration. 9 In the United States, three-quarters of units use autoclaves to inactivate Category A waste, with others exclusively incinerating waste. In some low- and middle-income countries, onsite burn pits are used for waste management. Liquid waste from patients’ body fluids and shower water can be problematic in HLIUs, and practices vary. The HLIU in Berlin uses heat treatment of the collected waste, while some HLIUs in Spain and the United States use chlorine treatment before releasing the waste into the normal waste stream. 9 We acknowledge that our current system of collecting and solidifying all liquid waste with gelling agents is somewhat impractical and carries risks; infrastructure for a liquid waste holding system will be incorporated into the new HLIU.
The most significant gap in unit readiness identified during both audits is in laboratory capabilities. The existing isolation facility had an onsite arterial blood gas machine that was validated and functioning; however, other onsite routine laboratory tests were not feasible due to hospital and laboratory concerns regarding staff safety. One of the key recommendations from the external 2019 audit of the isolation facility was to evolve an appropriate onsite laboratory. This is a key infrastructure and capability feature that will be added to the new HLIU.
Our team was able to improve gaps in the staffing domain in the 6 months between audits. Despite global shortages in healthcare worker staffing and high rates of staff turnover aggravated by the COVID-19 pandemic, 20 we found success in recruiting new nurses to join the unit. The clinical nurse facilitator targeted new hospital nurse hires, including those in departments outside of infectious diseases, and gave them an opportunity to be trained on PPE donning and doffing, waste management, and other unit processes. Education sessions for various MMUH departments were implemented more regularly to increase awareness of the unit and opportunities to be trained and join the team. Another strategy to retain staff included a proposed activation allowance for increased pay during unit activation and protected time for training and exercises. Staffing has consistently been cited as a challenge for HLIU teams and HLIU readiness; there is opportunity for HLIUs to continue sharing and learning from other teams’ successful strategies to recruit and retain staff. 21
The MMUH isolation facility is the referral center for patients with HCIDs in Ireland. It is imperative that the facility has high standards and maintains the ability to activate quickly without compromising patients’ care or staff safety. This audit tool assisted us to clearly visualize the gaps in our activation process and to address them more effectively. Construction of a new HLIU is underway to overcome key infrastructural deficiencies; in the meantime, several key risk mitigation strategies have been developed as outlined earlier. These strategies will assist in maintaining the basic principle of safely isolating a patient while providing optimal medical care during times of activation. That said, there remains a risk that a patient with a confirmed HCID could present and deteriorate to the point of requiring critical care support, which cannot be safely achieved in our isolation facility. For this reason, while awaiting the new Irish HLIU, a memorandum of understanding with EU partners was developed to transfer confirmed cases of HCID from Ireland to another European HLIU facility.
This study has a few limitations including the fact that the checklist was applicable to unit readiness for noncritically unwell adults only. It is important to consider readiness for others including pediatric, pregnant, and critically ill patients. Another limitation is that data were collected at 2 discrete points in time, whereas daily observations and assessments of readiness over several days to weeks would provide data on fluctuations and a more comprehensive perspective on readiness measures. The original study from Johns Hopkins assessed the facility over a more prolonged period (50 days), thereby improving the efficacy of the checklist; however, we were limited by staffing availability and other commitments. Since the audits were conducted, NETEC has developed the special pathogens operational readiness self-assessment (SPORSA), which is a more detailed readiness assessment. We plan to expand the audit process to include elements of SPORSA in an updated unit readiness assessment tool. 14
Future Direction
The new HLIU, expected to open in 2025, is designed to overcome infrastructure impediments in the current isolation facility, thereby enhancing readiness. The HLIU will be a standalone facility with ICU capability and a BSL-3 laboratory onsite. It will be a secure facility with onsite passthrough autoclaves to improve waste handling. The design of the new unit includes a separate wastewater holding system, allowing local treatment of liquid waste before its connection to the hospital waste. The processing of liquid waste will involve chemical treatment and separating hazardous wastewater from the unit from the rest of the ward block. The effluent is discharged to the holding tank for a minimum period of 24 hours prior to release. Moreover, during standby periods, the unit will serve as the center for HCID training and simulation in Ireland. It will form the hub of a national HCID network with other regional facilities, including pediatric and maternity hospitals, as well as transport agencies.
Conclusion
The audit process described in this case study demonstrates a successful strategy in adapting and applying an activation readiness checklist tool initially developed by the Johns Hopkins BCU. Our HLIU readiness checklist is a novel way to measure isolation unit readiness, was easy to use, and assisted in identifying (and therefore aided in improving) deficiencies that impacted the rapid activation of the unit. The checklist will be incorporated into the preparedness strategy for our new HLIU facility.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
