Abstract
The identification of a novel respiratory pathogen in late December 2019 and the escalation in the number of infections in January 2020 required healthcare facilities to rapidly assess their planning and preparations to identify and manage suspected or confirmed cases. As a Regional Emerging Special Pathogens Treatment Center, many of the policies, resources, and tools Massachusetts General Hospital had developed before the COVID-19 pandemic were based on the Identify-Isolate-Inform concept to enable rapid identification of persons under investigation; isolation from other patients, visitors, and staff; and appropriate information sharing with internal and external parties to ensure continued safety of the facility and community. Our team sought to leverage these existing resources to support other healthcare facilities and implemented a modified Plan-Do-Study-Act approach to develop, refine, and disseminate a novel coronavirus toolkit. The toolkit underwent 3 Plan-Do-Study-Act cycles resulting in revisions of specific products, and the addition of new products to the toolkit. The toolkit provided access to templated algorithms, policies and procedures, signage, and educational materials, which could be customized for local needs and implemented immediately. There was broad dissemination and use of the resources provided in the toolkit and response to end-user feedback was provided in subsequent revisions. This project demonstrates the role that Regional Emerging Special Pathogens Treatment Centers can play in supporting the sharing of resources and best practices, and the utility of a Plan-Do-Study-Act approach in meeting needs.
Introduction
The introduction of a novel respiratory pathogen on a global scale in late December 2019 and January 2020 required healthcare facilities to rapidly assess their planning and preparations to identify and manage suspected or confirmed cases of COVID-19. Creating a safe environment for healthcare personnel and patients meant that it was critical for healthcare facilities to have effective policies and practices in place to (1) rapidly identify patients meeting the person under investigation criteria, (2) isolate the patient, (3) notify the appropriate internal contacts and public health authorities, and (4) provide safe and effective care.
The Identify-Isolate-Inform concept has been applied to high-consequence infectious diseases, such as Ebola virus disease and other pathogens over the last decade, including communicable diseases not considered to be of high consequence, such as measles.1-4 As a Regional Emerging Special Pathogens Treatment Center (RESPTC),5,6 many of the policies, resources, and tools Massachusetts General Hospital had developed before the COVID-19 pandemic were based on the Identify-Isolate-Inform concept to enable rapid identification of persons under investigation; isolation from other patients, visitors and staff; and appropriate information sharing with internal and external parties to ensure continued safety of the facility and community. Additionally, the hospital had developed extensive tools to support healthcare personnel to safely don and doff personal protective equipment (PPE), and checklists to ensure safe delivery of care and appropriate management of the patient's environment.
The Plan-Do-Study-Act (PDSA) methodology has been widely used in quality improvement projects in healthcare and supported the timely approach to intervention needed in this situation. 7 In early 2020, the Massachusetts General Hospital RESPTC implemented a modified PDSA approach to develop, refine, and disseminate a novel coronavirus toolkit, incorporating feedback from end users. 8 In this article, we report on the PDSA process, outcomes, and lessons learned, with applications for ongoing pandemic preparedness as well as augmenting capacity for response to communicable diseases in healthcare facilities more broadly.
Methods
Using the PDSA approach (Figure 1), internal policies and procedures were inventoried and modified to meet the needs of healthcare facilities and were revised to be generalizable to a range of healthcare facilities. Local support services, including experts in graphic design and translator services, were engaged to enhance utility and versatility of various products, and the toolkit was made available online and free of charge.
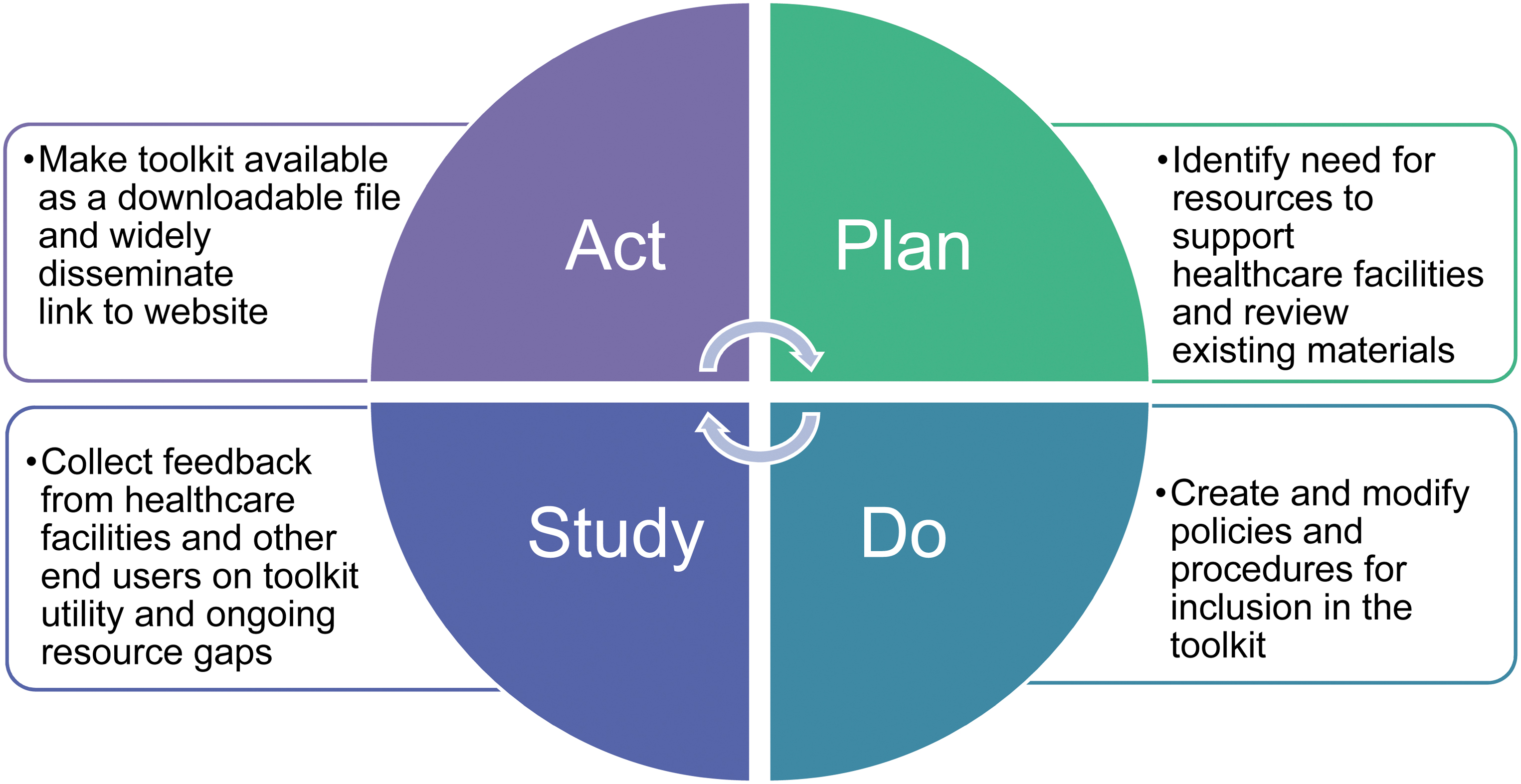
Plan-Do-Study-Act cycles for toolkit development and dissemination.
Plan
The first steps included a comprehensive review of existing RESPTC policies and procedures to determine which were in alignment with current public health guidance and appropriate for inclusion in the toolkit. The inventory covered the following domains: (1) evaluation tools that supported systemic and clinical determination of persons who might meet current case definition; (2) policies that establish systems and provide operational guidance and expectations, protocols, and procedures to support the safe implementation of transmission-based precautions; (3) signage, including materials translated into languages other than English, to support communication with patients and visitors; and (4) training materials for healthcare personnel, such as those to support PPE donning and doffing.
Do
Internal policies and procedures were modified before sharing externally to ensure that terminology and content was generic and adaptable. Many tools and resources had been previously developed for the care of patients with suspected or confirmed high-consequence infectious diseases requiring transmission-based precautions including airborne, contact, and eye protection, such as Middle East respiratory syndrome. These included an Identify-Isolate-Inform algorithm, internal policies for the management of patients with or suspected of having a high-consequence infectious disease, PPE donning and doffing checklists, and training videos. Graphic designers reviewed and modified the content to enhance utility and ease of customization for local needs. This included indicating where facilities could insert local contact numbers and facility-specific information, including links to other resources. Professional medical translators were engaged to support the creation of signage in languages commonly spoken by patient populations to support screening efforts.
Study
Requests for resources from within our organization initially helped guide what was placed within the toolkit. Our team also fielded requests for assistance and resources from outside entities, including a variety of healthcare facilities and public health authorities. As additional tools were developed, they were added to the toolkit. Specific requests for video content resulted in both the creation of new material, such as the appropriate procedure for obtaining a nasopharyngeal specimen, and the review and distribution of videos that had previously been created for internal purposes, such as the PPE donning and doffing videos (Figure 2).
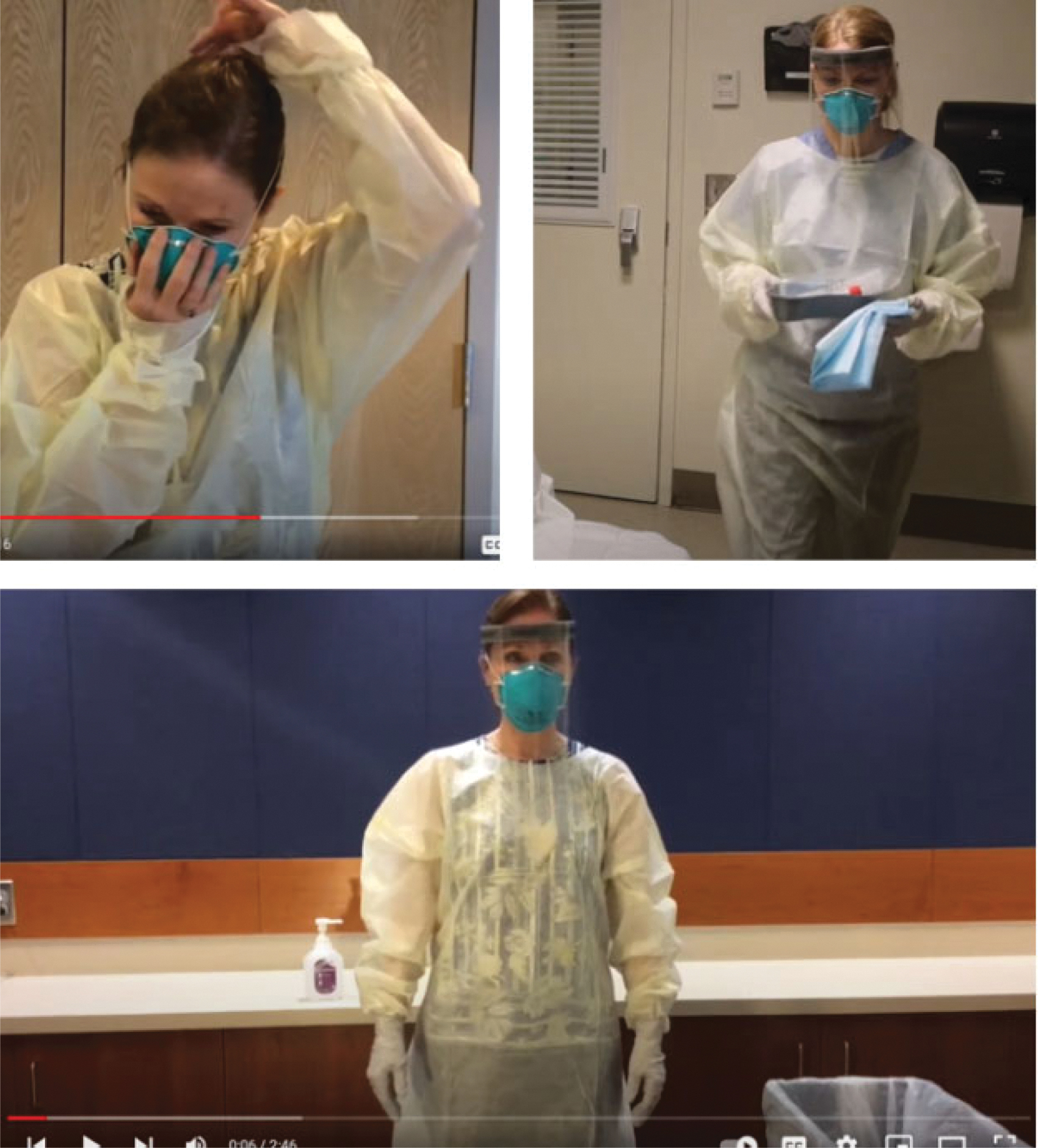
Training videos incorporated into the toolkit. Videos providing instructions on nasopharyngeal specimen collection, personal protective equipment donning, and personal protective equipment doffing were published online via an unlisted YouTube channel.
Act
The initial toolkit was posted as a downloadable PDF on the RESPTC's external website; the file and website link were also sent electronically to state and local public health leaders and posted to distribution lists including the Region 1 Regional Disaster Health Response System and other professional networks.
The PDSA cycles occurred between January 20 and February 28, 2020, and assessment of available utilization metrics was through June 30, 2020. The third version of the toolkit 9 is currently available online and as a Supplement (www.liebertpub.com/doi/suppl/10.1089/hs.2021.0184).
Results
Between January 20 and February 28, 2020, the toolkit underwent 3 PDSA cycles resulting in revisions of specific products and the addition of new products. The first toolkit was released on January 24, 2020. While a formal feedback mechanism was not used, end users were encouraged to provide feedback via email. Regional stakeholders received immediate feedback over the following several days, including detailed requests for training materials—specifically on the procedures to collect nasopharyngeal specimens for testing and on the donning and doffing of PPE. This feedback led to rapid cycle changes in video creation (eg, demonstrating nasopharyngeal specimen collection) and editing of internal training videos for external dissemination (eg, demonstrating proper usage of PPE), resulting in the release of the second version on January 29, 2020. Cycle 3 spanned January 30 through February 28, 2020, with release of the third version on February 28, 2020. This cycle included feedback from end users and updates related to evolving knowledge of virus epidemiology and transmission, infection prevention strategies, and management of anticipated and actual shortages of PPE.
The evolution of the toolkit and a description of the resources included are detailed in the Table. Examples of the types of changes incorporated into each cycle are provided by domain. Version 2 of the toolkit included 2 new evaluation tools specifically tailored to the COVID-19 conditions at that time. The purpose of these tools was to support the screening of patients to determine if they met person under investigation criteria, and to support the screening of visitors. Both of these tools also underwent updates in content between the second and third versions. A detailed resource was added to the policies and procedures in the PDSA Cycle 2 in response to requests for operational support regarding patient placement for suspected and confirmed patients, as well as employee tracking and symptom monitoring. The demand for educational materials was a primary component in PDSA Cycle 2 and Cycle 3 as healthcare facilities tried to ramp up healthcare personnel education and overall preparedness for the pandemic. The addition of video resources to support written checklists for PPE doffing and the collection of nasopharyngeal specimens were important components of the process.
Resources Included in the Toolkit Across PDSA Cycles
Abbreviations: CDC, US Centers for Disease Control and Prevention; HCF, healthcare facility; HCID, high-consequence infectious disease; PAPR, powered air-purifying respirator; PDSA, Plan-Do-Study-Act; PPE, personal protective equipment; PUI, persons under investigation.
Between January 20 and June 30, 2020, the primary web page that housed the toolkit received 1,448 views (Figure 3); duration of unique views averaged approximately 6 minutes. During the same period, users downloaded the full toolkit 469 times and viewed the nasopharyngeal specimen collection training video 4,326 times (Figure 4). Using regional email distribution lists, the toolkit reached an additional 79 individual subscribers, representing academic medical centers across New England, state health departments, and professional organizations. Additionally, the toolkit was hosted on the Region 1 Regional Disaster Health Response System site.
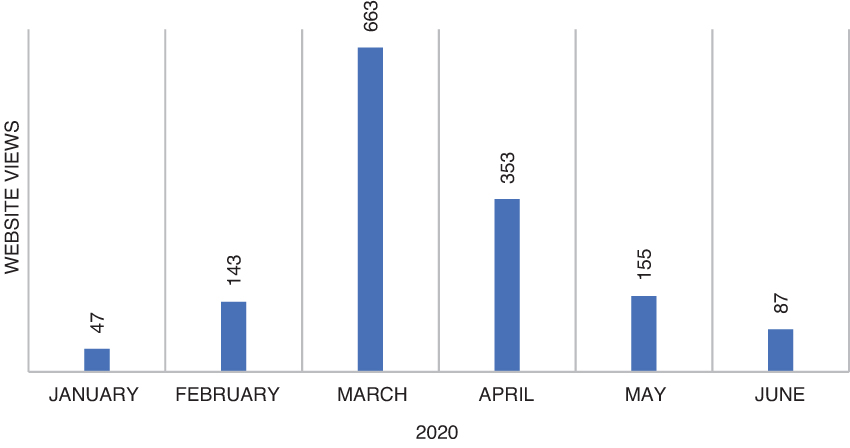
Web page views of the toolkit, January through June 2020.
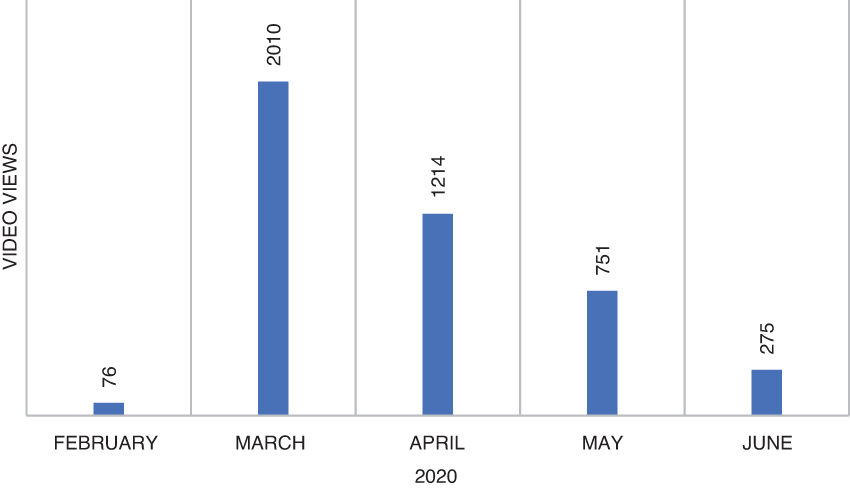
Views of the nasopharyngeal specimen collection video, February through June 2020.
Discussion
The PDSA approach was used in the early stages of the COVID-19 pandemic to leverage RESPTC resources with the intent of rapidly addressing the identified needs of healthcare facilities. Multiple PDSA cycles provided access to templated algorithms, policies and procedures, signage, and educational materials, which could be customized for local needs and implemented immediately. Similarly, responding to feedback from end users with enhancements to the product was achieved. Domains contained within the toolkit included evaluation tools, policies and procedures, signage, and educational materials; changes implemented during the cycle included the addition of tools not included in prior versions, and updates to materials to meet current public health guidance. There was a noted uptick in access to the toolkit and associated resources, including the videos, when the third version of the toolkit was released at the end of February 2020, and in early March 2020. When reviewing access to the training videos, the large number of views can be interpreted as ongoing access to the resources following toolkit download. By late spring 2020, additional resources developed by public health officials and healthcare facilities, many of which had activated their Hospital Incident Command Systems to respond to the pandemic, likely supplanted the need for the toolkit. However, in the initial stages of the pandemic, many hospitals used the toolkit as a resource springboard until additional resources were locally available. For example, the toolkit was quickly posted for reference by early March on the websites of the Massachusetts Hospital Association, the Massachusetts Medical Society, and others within the state, as well as the websites of the Homeland Security Digital Library, Tulane University, and others across the United States. As other national guidance emerged, portions of the toolkit were incorporated into updated protocols, as in the South Dakota COVID-19 Dental Office Protocol and Procedures. 10
The process of developing and disseminating the toolkit identified critical areas for future planning and response. First, the RESPTCs have invested substantial time, energy, and resources in developing response plans and protocols for the care of patients with confirmed or suspected high-consequence infectious diseases and are well positioned to share their expertise with other healthcare facilities. Despite this, modifying RESPTC tools in the setting of a pandemic—and other periods of surge in demand for resources—presents unique challenges that could be addressed in advance. Preplanning by creating generic versions of RESPTC algorithms, policies and procedures, screening tools, and educational materials that can be rapidly deployed is a potential solution. Additionally, having a preestablished structure for rapid sharing materials and incorporating feedback and revisions would improve the utility of materials and timeliness of updates. Moving forward, RESPTCs should engage in active needs assessments within their regions to better understand the needs and existing gaps in the system of care for high-consequence infectious diseases and should consider proactively developing tools, resources, and supportive educational materials to meet these needs.
The project experienced some limitations. Due to the rapid pace of the pandemic, our RESPTC had insufficient resources to assess the potential impact of the toolkit on pandemic response at healthcare facilities. Assessment of web page access and product downloads does not allow for a sufficiently detailed understanding of use or impact.
Conclusion
The benefit of early and frequent engagement with other healthcare facilities and entities in the region establishes RESPTC as a known and accessible expert and resource. As embedded regional hubs for special pathogen readiness, RESPTCs serve a critical function in nationwide preparedness and institution of a nationwide network for special pathogen response; as such, leveraging the previous and continued work of RESPTCs and other partners is likely to advance local, regional, and national capabilities to respond to emerging and reemerging infectious diseases.
The PDSA process, outcomes, and lessons learned from this experience can inform ongoing pandemic preparedness efforts and increase capacity for response to communicable diseases in healthcare facilities more broadly.
Footnotes
Acknowledgments
The authors thank all users of the toolkit for their feedback and their efforts in responding to the global pandemic of COVID-19. The authors additionally thank Paula Wright, RN, MSN, CIC for her substantial contributions to our RESPTC policies and procedures and Massachusetts General Hospital Translation Services for their support in translating materials for inclusion: Andrea Zhou (Chinese translation), Juhui Mo (Chinese translation), Hany Nakhla (Arabic translation), Omar Waid (Arabic translation), Paulo Chaves (Portuguese translation), Herve Dorsinville (Haitian Creole translation), Alejandro Cimadoro (Spanish translation), Anna Pandolfo (Spanish translation – coordinated and oversaw written translations for all languages). This work was supported by US Assistant Secretary for Preparedness and Response (6 U3REP150548-05-08 to EFS, SL, PDB, ESS).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
