Abstract
Research is foundational for evidence-based management of patients. Clinical research, however, takes time to plan, conduct, and disseminate—a luxury that is rarely available during a public health emergency. The University of Nebraska Medical Center (UNMC) developed a single institutional review board (IRB), with a vision to establish a rapid review resource for a network focused on clinical research of emerging pathogens in the United States. A core aspect of successful initiation of research during a pandemic or epidemic is the ability to operationalize an approach for rapid ethical review of human subject research and conduct those reviews at multiple sites—without losing any of the substantive aspects of ethics review. This process must be cultivated in anticipation of a public health emergency. US guidance for operationalizing IRB review for multisite research in a public health emergency is not well studied and processes are not well established. UNMC sought to address operational gaps and identify the unique procedural needs of rapid response single IRB (RR-sIRB) review of multisite research by conducting a series of preparedness exercises to develop and test the RR-sIRB model. For decades, emergency responder, healthcare, and public health organizations have conducted emergency preparedness exercises to test requirements for emergency response. In this article, we describe 2 types of simulation exercises conducted by UNMC: workshops and tabletops. This effort represents a unique use of emergency preparedness exercises to develop, refine, and test rapid review functions for an sIRB and to validate readiness of regulatory research processes. Such processes are crucial for conducting rapid, ethical, and sound clinical research in public health emergencies.
Background
Research is foundational for evidence-based management of patients, but it takes time to plan, conduct, and disseminate, which is rarely possible during an outbreak.1-5 Ethical and regulatory frameworks designed for nonacute epidemics do not lend themselves well to the purpose of acute epidemic research.1,3,6 Conducting clinical research during a public health emergency has long been identified as an area of need but mired with difficult operational challenges.7,8 Fulfilling regulatory requirements and the careful, methodical actions that define the path to approval are iterative processes. They involve the submission of a study for institutional review board (IRB) review, followed by instructions for further modification of study protocols, followed by resubmission, and so on. This iterative dimension can slow initiation of research, sometimes to the point that we miss the window of opportunity to conduct meaningful research to inform current and future outbreaks.3,4,9-11
In 2005, the University of Nebraska Medical Center (UNMC) implemented a rapid response IRB (RR-IRB) for local site studies. It serves to respond to urgent needs for review, including for research during a public health emergency. The RR-IRB complies with the membership requirements outlined in the US Food and Drug Administration (FDA) and the Federal Policy for the Protection of Human Subjects, also known as the “Common Rule.” 12 In addition, it is registered with the Office for Human Research Protections, composed of 8 members, including a nonscientist, a member not otherwise affiliated with the institution, and a prisoner representative. It also includes a membership of varying backgrounds and expertise representing a variety of colleges, departments, and medical disciplines, including public health, infectious disease, pharmacy, and ethics. The RR-IRB provided rapid review of multiple clinical research protocols during the 2014-2016 Ebola virus disease outbreak in West Africa, each with 24 hours or less from submission to approval.
Using an RR-IRB requires open communication and collaboration between the IRB and the investigator. At UNMC, this took place through extensive prereview of applications by IRB staff. Strong collaboration between the study team and the IRB staff was crucial, and flexibility and willingness to take a proactive role was needed from IRB staff to ensure efficiency of study approval. Through strong institutional support for RR-IRB studies, a lead IRB administrator could focus solely on the rapid response study, but often worked late into the night to ensure timeliness. The lead IRB administrator took an active role in ensuring that the investigator addressed the criteria for IRB approval of research. In addition, the lead IRB administrator supported development of the protocol by identifying changes and was in constant communication with the researcher to clarify these changes. Embedding the lead IRB administrator in the submission process enabled the IRB feedback to happen in real time. The investigator or a designee was also present at IRB meetings to answer questions and respond to IRB conditions for approval. Although the RR-IRB has a different set of standard operating procedures (SOPs) to compress the review timeline, the review occurs within UNMC's electronic IRB system and meets the review criteria, just like any other full board study reviewed by the UNMC IRB.
There have been multiple calls to create a centralized IRB model for research in disaster and public health emergency settings.13,14 In 2011, the National Preparedness and Research Science Board called for the creation of a specialized IRB that would be capable of rapidly convening to assess research protocols while maintaining robust protections for human subjects. 15 The Office of the Assistant Secretary for Health established a Public Health Emergency Research Review Board (PHERRB) that could review research conducted at multiple sites during public health emergencies. The mandate of the PHERRB is to provide investigators with expert IRB review of human subject protections within public health emergency research protocols. In the United States, regulatory requirements promulgated by revision of the Common Rule state that any institution engaged in federally funded cooperative research must be approved by an sIRB. 12 The PHERRB underscores the importance of having centralized IRB membership and staff experienced in clinical research in public health emergencies. Before June 2020, however, the PHERRB lacked accessible policies that could provide US IRBs with guidance or best practices, and it still lacks SOPs or operational guidance. 16 Beyond the PHERRB, national guidance for operationalizing single IRB (sIRB) review for multisite research in an emergency is not well studied and processes are not well established. 14
While a centralized approach would seem to promote greater efficiency, the operational complexities and challenges often expand when clinical research must be conducted at multiple sites. Multisite research introduces more bureaucratic processes (eg, reliance agreements, local context reviews, standard definitions) into the path toward study approval (Table 1). These processes are separate from IRB review but still intertwined in the pathway to sIRB approval, often increasing the average time to approval for a clinical trial using an sIRB.21-23 A core aspect of successfully initiating research during a pandemic or epidemic is the ability to operationalize an approach for rapid ethical review of human subject research and conduct those reviews at multiple sites—without losing any of the substantive aspects that ensure adequate protections, including the local context review.24-29 This core ability should be cultivated in anticipation of a pandemic or epidemic, rather than in response to them.30,31
Key Terms in Single Institutional Review Board Review
Abbreviations: CFR, Code of Federal Regulations; IRB, institutional review board; sIRB, single institutional review board.
To address these operational gaps and identify unique procedural needs of rapid multisite research review, UNMC led a series of emergency preparedness exercises to develop, refine, and test its rapid response sIRB (RR-sIRB) model. For decades, emergency responder, healthcare, and public health organizations have conducted emergency preparedness exercises to test requirements for emergency response. Hospital accreditation organizations and the Hospital Preparedness Program 32 promote the use of exercises to prepare organizations for potential emergencies by testing capabilities and identifying areas for improvement. Since 2001, substantial strides have been made in hospital disaster planning through implementation of training and exercise programs aimed at testing organizational readiness for emergency functions. 33 Preparedness activities before the COVID-19 pandemic were novel in the IRB sphere; although in late 2021, the Association for the Accreditation of Human Research Protection Programs, the accrediting body for US IRBs, announced it will start requiring institutions to have emergency preparedness plans to ensure Human Research Protection Programs (HRPPs) can continue to operate effectively to protect human subjects during a public health emergency. 34 A literature search on the topic revealed publications citing preparedness exercises as a potential solution to achieve research readiness for IRBs and SIRBs 14 and preparedness exercises including an IRB component 35 ; yet, to our knowledge, published results or evaluation from conducting preparedness exercises to refine IRB or sIRB SOPs, communication plans, or policies do not exist. 34
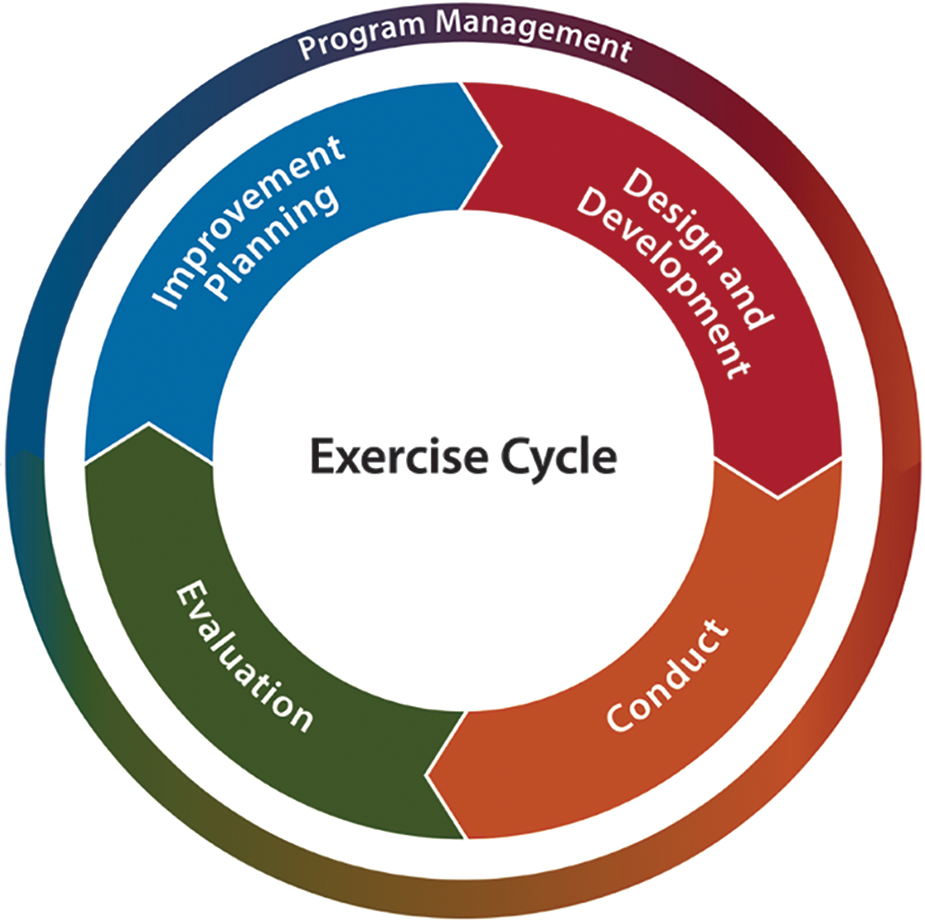
In this article, we describe a 9-month preparedness training and exercise effort to develop SOPs, assess SOPs, and advance capacity of the UNMC RR-IRB to act as an RR-sIRB for a network of institutions positioned to conduct clinical research in a public health emergency. We used 2 types of discussion-based exercises, workshops and tabletops, which have historically been used to develop emergency preparedness plans and test emergency response plans (Figure). This effort represents a unique use of emergency preparedness exercises to develop, refine, and test rapid response research functions and to validate readiness of regulatory research processes crucial for conducting multisite clinical research in emergencies.
Figure. The Homeland Security Exercise and Evaluation Program exercise cycle. 36
Methods
In April 2019, the Center for Preparedness Education (CPE) worked with the Special Pathogens Research Network (SPRN) and the UNMC IRB to develop a planning, training, and exercise calendar. The SPRN was established in the United States in November 2016 by the National Emerging Special Pathogen Training and Education Center to provide an organizational structure to leverage the expertise of the 10 Regional Emerging Special Pathogen Treatment Centers (RESPTCs); it was intended to develop and support infrastructure to improve readiness to conduct special pathogens clinical research in the United States.37,38 The SPRN has maintained a close collaboration with federal and other external partners as an extension of the relationships developed during the 2014-2016 Ebola virus disease outbreak in West Africa. The SPRN formed a partner working group with the goal of continuing collaborations to support better coordination of investigational medical countermeasures, which are medicines and medical technologies and supplies used to diagnose, prevent, or treat diseases resulting from chemical, biological, radiological, or nuclear incidents. 37 The partner working group includes the Biomedical Advanced Research and Development Authority (BARDA), the US Centers for Disease Control and Prevention (CDC), the FDA, the National Institute of Allergy and Infectious Diseases (NIAID), the US Department of Defense Infectious Disease Clinical Research Program, and the US Army Medical Research Institute of Infectious Diseases. Those who provide expertise for the selection of appropriate medical countermeasures and for the progression of medical countermeasures through the research and regulatory pipelines include BARDA, the CDC, the FDA, NIAID, and the Department of Defense. 38 Because UNMC's IRB is the sIRB for the SPRN, in the context of a public health emergency, the sIRB must work closely with federal entities who sponsor, request, or direct the study.
Beginning in April 2019, UNMC, CPE, and SPRN conducted a series of workshops, a planning conference, and tabletop exercises to develop, test, and use multisite, rapid response regulatory frameworks. Workshops were conducted in series to iteratively develop SOPs for the RR-sIRB. Two tabletop exercises were then conducted to test and refine RR-sIRB procedures with input from sites that agree to rely upon the reviewing IRB, otherwise known as relying sites. A functional exercise was planned in culmination, but a real-world scenario intervened—the COVID-19 pandemic. The RR-sIRB processes were then used for COVID-19 clinical research following the series of exercises. Each exercise conformed to the standards established by the Homeland Security Exercise and Evaluation Program. 36
Planning Team
A 4-member exercise planning team was convened to develop the exercise plan and to set overarching training and exercise program objectives. The planning team included 2 Center for Preparedness Education exercise experts and 2 UNMC IRB staff members with roles in the SPRN. The workshops focused on developing the RR-sIRB processes and the tabletop exercises aimed to test and refine the processes by incorporating research and clinical teams from the Nebraska biocontainment unit and RESPTCs within the SPRN. UNMC conducted 2 workshops focused on the developing RR-sIRB processes. Although there are standard models for sIRB processes, 39 no model processes for conducting rapid review of multisite research exist. The workshops focused on ensuring RR-sIRB processes were appropriate for public health emergency research teams.
The exercise planning team was expanded for developing and administering 2 tabletop exercises to include representatives from SPRN and relying sites. One tabletop exercise was conducted for the research community at the sIRB institution, UNMC tabletop, and the second SPRN tabletop exercise was conducted for both the sIRB institution and relying SPRN institutions: Emory University and Cedars-Sinai Medical Center. Each RESPTC has an IRB and an HRPP, which informed our planning efforts. The scenario developed for the UNMC and the SPRN tabletop exercises focused on testing and validating processes, communications, and information needs between the sIRB and relying institutions, investigators, and research coordinators to achieve IRB approval of an investigational product for multisite clinical research in less than 72 hours.
Training and Exercise Plan
In the first 6 months, 2 workshops and an initial planning conference for the tabletop exercises were completed. This time period facilitated the development of an RR-sIRB checklist (see Supplemental Materials 1, www.liebertpub.com/doi/suppl/10.1089/hs.2021.0181). In the second half of the year, 2 tabletop exercises were conducted by UNMC, CPE, and SPRN. Representatives from the Center for Preparedness Education, UNMC IRB, and the SPRN attended planning conferences. A series of training activities were also conducted by the UNMC team for reliant sites—Emory University and Cedars-Sinai Medical Center—in the lead-up to the SPRN tabletop exercise to ensure familiarity with the RR-sIRB processes.
Workshops are characterized as discussion-based exercises designed to develop policy, plans, or procedures (Figure). Workshops are generally structured to facilitate participant interaction aimed at building a product. This is done by delivering information through presentations and facilitated sessions where participants develop, review, and build consensus on plans and procedures. Workshop outcomes are generally defined products such as plans, SOPs, checklists, and communications tools. The primary materials used to create the draft plan for an RR-sIRB process were the UNMC RR-IRB policy 40 and the SMART IRB SOPs. 39 SMART IRB is a platform designed to provide sIRB SOPs, which ease common challenges (eg, communication, assigning study team roles, harmonization) associated with initiating multisite research. 41
During exercise planning, planning team members decide the type and number of planning activities needed to successfully develop a given exercise based on scope and complexity. To engage the leadership in reviewing and selecting the reliant sites, the initial planning conference convened key stakeholders from the IRB, UNMC research community, Center for Preparedness Education, and the SPRN sites. During the initial planning conference, stakeholders agreed upon the objectives and scope of the training and exercise plan.
The tabletop exercise is also a discussion-based exercise but designed to begin testing an emergency plan in an informal, low-stress environment. Tabletop exercises are used to clarify roles and responsibilities and to identify gaps in planning. 42 Tabletop exercises were planned as discussion-based simulations intended to generate a dialogue on prescribed issues to facilitate a conceptual understanding and to identify strengths and areas for improvement for emergency response plans, SOPs, and communication tools to achieve IRB review, approval, and initiation of an investigational study for multisite clinical research in less than 72 hours (see Supplemental Materials 2, 3, 4, and 5 at www.liebertpub.com/doi/suppl/10.1089/hs.2021.0181).
Results
Workshops 1 and 2
Two workshops, held 3 months apart, were conducted by UNMC, CPE, and SPRN to develop and refine RR-sIRB policies and procedures and to facilitate multisite review, approval, and initiation of clinical research in less than 72 hours. Workshops were attended by 3 IRB staff, 2 members of academic leadership, and 2 members of the Center for Preparedness Education team. Five objectives were achieved through the workshops (Table 2). Following the first workshop, we developed a draft workflow of the RR-sIRB review process. The primary outcomes centered on identifying the key gaps, which included: (1) a communication plan to support rapid review, (2) buy-in from the relying RESPTC institutions and the broader site HRPPs (eg, each RESPTC has an IRB and HRPP) to ensure local processes to support approval and release of the study at the site, and (3) an information technology (IT) platform that can support efficiency in review and communication.
Workshop 1 and 2 Outcomes a
The primary objective for both workshops was to develop a plan for rapid review of multisite protocol.
Abbreviations: HRPP, Human Research Protection Programs; IRB, institutional review board; IT, information technology; RR-sIRB, rapid response single institutional review board; sIRB, single institutional review board.
The second workshop included the draft workflow as a center point for discussion. Following the workshop, we refined the workflow by developing an RR-sIRB checklist for the sIRB (Table 2). This checklist covered the sIRB process from the initial requests to serve as the IRB of record for multisite all the way to study approval. A focus on this segment of the sIRB process was decided given that study approval was the central focus of the RR-sIRB process. A draft plan for communicating from the sIRB to study teams and site IRBs was also scoped out. Finally, recognizing the importance of a platform to support sIRB review—keeping review and communication organized—we began developing an IT system to support the entire process.
Tabletop Exercises
We conducted 2 tabletop exercises 3 months apart. The first tabletop exercise engaged stakeholders solely from the sIRB institution and the second tabletop exercise engaged stakeholders from both sIRB and relying research network institutions. Tabletop exercises utilized a facilitator-presented scenario and facilitation of a series of questions structured to drive problem-solving and in-depth discussion of procedures, plans, and communications required for an emergency response (Supplemental Materials 2).
Tabletop exercise participants were divided into groups to equally distribute each professional category. Moderators and evaluators were assigned to facilitate small-group discussion and report appropriate information back to the large group upon reconvening. After each small-group discussion, a large-group discussion was facilitated with all participants and moderators. The large-group discussion enabled participants to share ideas and consider UNMC preparedness resources that may not have seemed applicable for use by the IRB team (ie, Nebraska biocontainment unit communication plans, SOPs for Nebraska biocontainment unit clinical research). Following the tabletop exercise, participants completed feedback forms, moderators facilitated group debriefings and conducted a large-group discussion to review all concerns and ideas for inclusion in the after-action report. Outcomes from the tabletop exercise included:
Creating a communication plan that accounted for rapidly attending to a request to serve as the SIRB for multisite to study Creating a communication plan for study submission all the way to approval Identifying a need to develop educational material for the RR-sIRB process designed for different stakeholders (eg, principal investigators, study team, relying site IRBs) Enabling identification of key gaps; for instance, once developed, the communication plan should be tested biannually, at minimum, to ensure point of contacts at the sites (eg, principal investigators, study team, IRB contacts) are correct and familiar with the communication plan Identifying the need for a backup process with policies and procedures in the event the IT system is down
UNMC and SPRN Tabletop Objectives and Outcomes
Abbreviations: HRPP, Human Research Protection Programs; IRB, institutional review board; IT, information technology; PI, principal investigator; RR-sIRB, rapid response single institutional review board; sIRB, single institutional review board; SPRN, Special Pathogens Research Network; UNMC, University of Nebraska Medical Center.
Both tabletop exercises provided participants with scenarios that were modeled after challenges experienced conducting research during the 2014-2016 Ebola virus disease outbreak in West Africa. In the United States, investigational products during the outbreak occurred via uncontrolled single-patient-use emergency investigational new drug authorization from the FDA. 43 The scenario posed a generic investigational product that did not have existing clinical protocols for humans and had limited data available on pharmacokinetics, optimal dosing, and safety in humans. During the Ebola virus disease outbreak, investigational protocols had to be developed by local clinical investigators or the product sponsors in parallel with the emergency investigational new drug process for individual local IRB approval. The SPRN tabletop scenario recreated similar circumstances to understand whether the RR-sIRB would be able to efficiently approve a novel investigational antiviral product showing efficacy in preclinical studies, including nonhuman primate trials.
The tabletop exercise was structured to provide participating sites the opportunity to think through the RR-sIRB process and to be part of refining the process. Due to the complexity and idiosyncrasies of local IRBs, in the lead-up to the SPRN tabletop exercise, the UNMC IRB conducted training sessions on the RR-sIRB process with relying site IRB staff to orient them to the process before the exercise scenario. Through training exercises conducted before the tabletop exercise, the planning team was able to refine the educational materials for the RR-sIRB process, making it broadly applicable for principal investigators, study teams, and IRBs from relying institutions. In follow-up, the sIRB developed a draft RR-sIRB manual for principal investigators, study teams, and IRBs from relying institutions. To ensure strong communication with the IRBs from relying institutions, an SPRN workgroup with representatives from site IRBs was convened with a monthly meeting schedule.
Real-World Scenario: COVID-19
Following the SPRN tabletop exercise, the planning team scoped out a functional exercise enabling personnel to validate plans and readiness by performing their duties in a simulated operational environment. As the COVID-19 outbreak turned into a global pandemic in February 2020, the UNMC experts determined a need to evaluate potential medical countermeasures. This need was considered urgent and a request was submitted to the IRB for an adaptive platform clinical trial. As the study received IRB approval and evaluation of the first drug (ie, remdesivir) 44 progressed, it was determined that there was a need to activate the RR-sIRB, which enabled evaluation of the RR-sIRB process throughout SPRN (Table 4).
Real-World Scenario of Capabilities, Priorities, Objectives, and Evaluation of High-Priority Items
Abbreviations: DUA, data use agreement; HRPP, Human Research Protection Programs; IT, information technology; MTA, material transfer agreement; RR-sIRB, rapid response single institutional review board; sIRB, single institutional review board.
Discussion
The Homeland Security Exercise and Evaluation Program 36 provides a set of guiding principles for exercise programs and a common approach to program management, design and development, conduct, evaluation, and improvement planning. Exercises are a fundamental component of emergency preparedness and provide a diverse community with the opportunity to shape planning, assess and validate capabilities, and address areas for improvement. Priorities are developed through the results of other exercises, or in this case, real-world experiences with public health emergencies. The priorities guide the overall direction of a progressive exercise program in which individual exercises are anchored to a common set of priorities or objectives that build in a way that supports improvement. These priorities guide the design and development of individual exercises and enable planners to identify and align objectives to capabilities evaluated during the exercise. Exercise evaluation assesses the ability to meet the objectives and capabilities by documenting strengths, areas for improvement, capability performance, and corrective actions in an after-action report/improvement plan.
Common capabilities assessed by emergency responders in exercises are planning, communications, community preparedness and participation, risk management, intelligence and information sharing, and dissemination. Two target capabilities were identified as areas for improvement from the UNMC tabletop exercise: planning and communications (Supplemental Materials 4). Each of these capabilities had specific implications for participating sites. For example, communication improvements were identified by updating the SOP to include an IRB point of contact by position and name at each site during future network drills and tests of the RR-sIRB process. While all 10 RESPTCs have an IRB, not all institutions engaged in research do; in such cases, it would be important to identify a point of contact for negotiating the reliance agreement (a reliance point of contact) in planning efforts. In addition, a detailed communication guide, which included communication algorithms, was developed after the UNMC tabletop exercise.
sIRB models often rely on the research coordinator from the lead site study team to initiate and lead activity with the IRB and with relying sites. 41 IRB policies outline the responsibility of IRB staff in a reactive role—they respond when protocols are submitted, but processes described would not necessitate staff to proactively reach out to ensure submission of forms and other regulatory requirements. In early 2020, SMART IRB recommended that the responsibility for coordinating the study should rest with the principal investigator and the lead study team, and not with the sIRB.35,41
Based on the experience of conducting rapid review for a single-site study, the UNMC IRB recognized that greater efficiency could be attributed to (1) institutional support for the studies, (2) IRB staff willingness to lead the study team, and (3) principal investigators working with IRB staff throughout the process. With that in mind, to achieve rapid review, the sIRB would require procedures that would enable sIRB staff to lead the coordination of the study approval like the approach they had used for the RR-IRB, but to scale for a multisite study.
During study initiation, having clear points of contact at each site's IRB was particularly important to the UNMC IRB team (Table 4). While the IRB at the relying institution is not responsible for the IRB review, they often are required to coordinate review of institutional requirements and the local context considerations release the study before the SIRB can provide study approval. Bearing that in mind, it was also essential that sites had mapped out a process for rapid review with their own HRPPs to release the study. The reliance agreement often ensures that the institutions HRPP has the final authority to enable research to be conducted at their institution. As a result of the SPRN tabletop exercise, sites had the opportunity to coordinate, and in some cases create a process, with their own local ancillary committees (eg, pharmacy and therapeutics review, institutional biosafety committee review, grants and contracts) for rapid review. Sites participating in the tabletop exercise were able to identify those ancillary reviews that would block the approval of the RR-sIRB, and work with those committees to create a rapid review process for those ancillary reviews at their institution. Some of those ancillary review committees decided to increase protections (eg, require a full committee review) where institutional policies would not have required it. The discussion and decision to do so was based on the impact on potential study participants. The focused conversation led to scoping SOPs for relying site ancillary reviews that integrated with the RR-sIRB time frame.
The tabletop exercises stressed the need for continued preparedness exercises with SPRN sites to enhance readiness. Future work is recommended to carry out drills of RR-sIRB communication plans and formalize working groups among IRBs to strengthen the regulatory collaboration needed for rapid review of research. During the real-world scenario, the initial study was approved in February 2020 with 3 sites needing access to the study drug. The RR-sIRB was able to give IRB approval for the study at 3 of the SPRN sites by day 3 achieving the 72-hour time frame that was scoped at the early planning phases of the exercise activities.
Conclusion
Conducting research during a public health emergency takes planning and advanced preparation.24,45,46 A rapid response IRB can provide an effective approach to reviewing, such as the RR-sIRB model described in this article, using preparedness exercises to enable rapid review of clinical research. While the literature consistently points to the operational challenges, no operational guidelines advise an sIRB regarding rapid review of research in disaster, epidemic, or emergency conditions. More research is needed, but the exercises we described provide a model approach for developing processes to conduct research in acute settings such as epidemics, pandemics, or disasters. The decision to establish an sIRB capable of rapidly reviewing multisite research represents a complex operation involving stakeholder groups from multiple institutions; it also requires ensuring the appropriate safeguards have been attended to for the participating sites. The COVID-19 pandemic only underscores the importance of research capabilities during a public health emergency and the need to prioritize building the infrastructures that makes research possible.
Footnotes
Acknowledgments
We thank those who participated in the exercises. We would like to extend a sincere thanks to the University of Nebraska Medical Center IRB staff: Lindsay Hicks, Gail Kotulak, Gail Paulsen, Sue Logsdon, and Natasha Willms. We also owe gratitude to the UNMC IT staff of Teresa Pisale and Geoff Gloeb who created the electronic sIRB system. We would like to thank Sami Vasistha for her efforts in project management. The authors received support for the exercise activities from the National Emerging Special Pathogens Training and Education Center, which is funded by Assistant Secretary for Preparedness and Response.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
