Abstract
In West Africa, identification of nonmalarial acute febrile illness (AFI) etiologic pathogens is challenging, given limited epidemiologic surveillance and laboratory testing, including for AFI caused by arboviruses. Consequently, public health action to prevent, detect, and respond to outbreaks is constrained, as experienced during dengue outbreaks in several African countries. We describe the successful implementation of laboratory-based arbovirus sentinel surveillance during a dengue outbreak in Burkina Faso during fall 2017. We describe implementation, surveillance methods, and associated costs of enhanced surveillance during an outbreak response as an effort to build capacity to better understand the burden of disease caused by arboviruses in Burkina Faso. The system improved on existing routine surveillance through an improved case report form, systematic testing of specimens, and linking patient information with laboratory results through a data management system. Lessons learned will improve arbovirus surveillance in Burkina Faso and will contribute to enhancing global health security in the region. Elements critical to the success of this intervention include responding to a specific and urgent request by the government of Burkina Faso and building on existing systems and infrastructure already supported by CDC's global health security program.
A
Over one-third of the world lives in areas at risk of DENV transmission, including much of Africa. Although dengue epidemics have been reported in Africa since the late 19th century, the epidemiology and public health impact of dengue in Africa remains unclear due to limited surveillance and low awareness.
8
DENV transmission is expected throughout much of Africa given the wide distribution of
Burkina Faso is a sub-Saharan African country with an estimated population of 20 million people in 2018. 23 Burkina Faso has committed to the Global Health Security Agenda (GHSA), which was launched in 2014 to accelerate compliance with the 2005 International Health Regulations (IHR).24,25 As such, Burkina Faso receives technical and financial support from the US Centers for Disease Control and Prevention (CDC) and other global partners to strengthen capacity to prevent, detect, and respond to public health threats, including diseases that cause AFI. GHSA support has included establishing a Severe Acute Respiratory Illness (SARI) laboratory-based sentinel surveillance system, piloting an innovative system for transporting specimens using the National Society for Postal Services (SONAPOST) from health facilities to the reference laboratory, and enhancing diagnostic capacity at the National Reference Laboratory for Viral Hemorrhagic Fevers (NRL-VHF).
While there is historic evidence of dengue endemicity in Burkina Faso,8,11,13,26-33 recent case surveillance and laboratory testing confirmed dengue circulation.24-27 Although dengue reporting was not compulsory, during a dengue outbreak in 2016, a total of 1,947 suspected dengue cases were identified, of which 1,327 (78%) were positive by rapid diagnostic test (RDT), and additional laboratory testing of 35 patient specimens found 19 (54%) confirmed DENV infections using RT-PCR: 11 were DENV-2, 6 were DENV-3, 2 were nontypeable, and 1 was a DENV-2/DENV-3 co-infection. 34 Beginning in August 2017, dengue cases were again identified in Burkina Faso, and on September 28, the Burkina Faso Ministry of Health (MOH) declared a dengue epidemic. As part of the outbreak response, the Burkina Faso MOH, CDC, and DAVYCAS International collaborated with 2 healthcare centers in Ouagadougou to implement arbovirus sentinel surveillance.
In this report, we describe the surveillance system protocol, including prospective identification of arbovirus infections among outpatient and hospitalized AFI cases, laboratory testing, clinical chart abstraction, and data management. We discuss challenges and lessons learned, toward establishing evidence-based best practices to enhance global health security in the region and further afield.
Emergency Response Dengue Sentinel Surveillance
Objectives
The overall aim of the sentinel surveillance system was to enhance knowledge to guide the Burkina Faso MOH for targeted public health action regarding dengue and other arboviruses circulating in Burkina Faso. More specifically, there were 4 primary objectives: (1) describe dengue cases, including the clinical course of hospitalized patients; (2) provide descriptive epidemiologic analysis to inform public health action; (3) provide laboratory-based diagnostic testing to define the frequency of DENV infection among patients presenting with AFI at the 2 sentinel sites; and (4) describe the frequency of severe dengue among hospitalized patients who presented during the ongoing 2017 dengue outbreak. More broadly, the sentinel surveillance system aimed to systematically collect case-based laboratory data on DENV and other potentially circulating pathogens of importance to global health security.
AFI may be caused by a variety of pathogens, some of which are highlighted by GHSA, including arboviruses such as dengue, yellow fever, West Nile, and Zika viruses. Appropriate national, regional, and even global responses require thorough case investigations linked to laboratory detection. Given recent outbreaks of Zika virus in the Western Pacific and Americas 35 and yellow fever virus in Angola and the Democratic Republic of Congo 35 as well as Brazil, 36 during the 2017 outbreak in Burkina Faso case-based laboratory surveillance was crucial to confirm dengue and exclude other potentially circulating arboviruses.
Sentinel Site Selection and Description
The sentinel surveillance system was implemented in 2 sites in Ouagadougou, the capital of Burkina Faso and part of the Central Health Region. The first site was Nongr-Massom District Hospital, one of 4 district hospitals in the Central Health Region that comprises 16 units, including surgery, maternity, pediatrics, and medicine. It consists of 95 beds for hospitalization and 130 health workers. Nongr-Massom District Hospital was selected as a sentinel surveillance site due to its large population catchment (estimated at 365,133 persons), as well as the large burden of dengue cases among outpatients reported to the national syndromic surveillance system.
The second site was Yalgado Ouedraogo University Hospital Center (Yalgado CHU), a tertiary referral hospital located in Ouagadougou. 37 It comprises 10 departments divided into 47 clinical and medical technical services providing care and diagnostic services, as well as conducting clinical training and research activities. Yalgado CHU has 713 beds and 1,400 staff, with approximately 24,000 to 25,000 consultations per year. The infectious disease department has 25 beds and attends to an average of 250 patients per year. Patients generally are admitted to the department following consultation at Yalgado CHU emergency department, direct consultation of a febrile patient to the infectious disease department, or indirect referrals from outside public and private health facilities. Most outpatient consultations are for non-malaria AFI, including dengue. The Yalgado CHU infectious disease department has been identified by Burkina Faso MOH as a reference center for severe dengue cases from the Central and surrounding health regions. As such, clinicians have been deeply involved with the development of national dengue case management guidelines, using experience garnered during the 2016 dengue epidemic and the results of research activities conducted to better understand clinical aspects of dengue in Burkina Faso.
National Reference Laboratory for Viral Hemorrhagic Fevers
Laboratory testing was conducted at the NRL-VHF based at an MOH public health research center, Centre Muraz in Bobo-Dioulasso. 38 The laboratory was established in 2014 as part of the response to the Ebola outbreak in West Africa and has since been supported by CDC and other partners to enhance capacity to detect viral pathogens identified as priorities by the Burkina Faso MOH. In addition to Ebola virus, the laboratory can test for a range of arboviruses (eg, dengue, chikungunya, Zika, yellow fever), as well as Rift Valley fever, Crimea-Congo hemorrhagic fever, and Lassa fever viruses. In 2017, the NRL-VHF became the Regional Reference Laboratory for Viral Hemorrhagic Fevers for the Economic Community of West African States (ECOWAS). 39 The acquisition of a Biological Safety Level (BSL)-3 mobile laboratory increases the NRL-VHF's capacity to safely operate during potential future emergency responses to viral hemorrhagic fevers. In addition to research activities, the NRL-VHF has supported Burkina Faso MOH in the detection and response to disease outbreaks, including during the 2016 and 2017 dengue outbreaks.
Clinical Case Definitions and Case Reporting
A case report form (see Appendix 1, supplemental material, at https://www.liebertpub.com/doi/suppl/10.1089/hs.2018.0048) was developed using the 2017 Pan-American Health Organization/World Health Organization (PAHO/WHO) clinical case definition for dengue without warning signs (DNWS), dengue with warning signs (DWWS), and severe dengue (SD) (Figure 1). 40 Descriptive epidemiologic variables included age, sex, place of residence, and place of employment. Additional relevant variables included recent travel history, reporting recent contact with living or dead animals, and reporting recent mosquito bites.
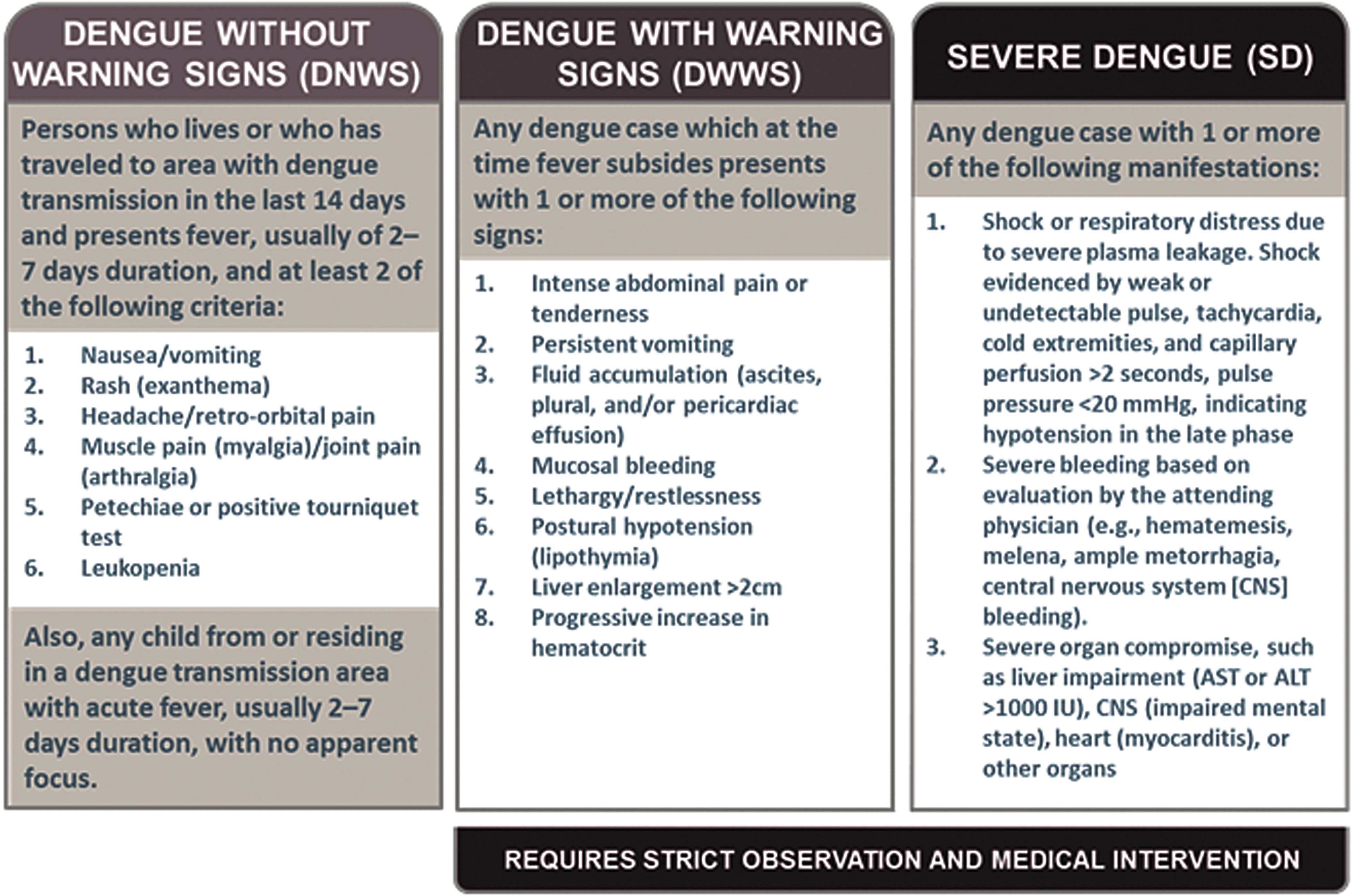
2017 PAHO/WHO Dengue Case Definitions by Clinical Severity
Case identification differed slightly between the 2 sentinel sites. At Nongr-Massom District Hospital, any patient who was seen at any department could be reported if they had a fever (subjective or objective) and at least 2 of the symptoms present in the 2017 PAHO/WHO clinical case definition (Figure 1). 40 Chief complaint of diarrhea or cough were used as exclusion criteria. At Yalgado CHU, outpatient and hospitalized cases in the infectious disease, emergency, nephrology, pediatric, gynecology, and obstetrical departments that met clinical case definitions were reported, and chart abstraction was conducted to collect data on all patients who were hospitalized in 2017 (Appendix 2, supplemental material, at https://www.liebertpub.com/doi/suppl/10.1089/hs.2018.0048).
Laboratory Testing and Laboratory-Based Case Definitions
At both sites, patient serum specimens were collected for laboratory testing. Site laboratory staff performed dengue RDT using the SD BIOLINE Dengue Duo, 41 as well as malaria RDT, as per routine testing for AFI. Results for NS1 antigen and anti-DENV immunogloblulin M (IgM) and IgG were recorded on the case report form. Using refrigerators provided, the remaining patient specimens were stored at 2 to 8°C and collected twice a week by SONAPOST for transport to NRL-VHF. From October 31, 2017, to April 4, 2018, SONAPOST transported 247 specimens from the sentinel sites to NRL-VHF.
At NRL-VHF, patient specimens were tested using the Trioplex real-time reverse transcriptase-polymerase chain reaction (rRT-PCR) assay to detect evidence of current infection with dengue, chikungunya, and Zika viruses.
42
Specimens were also tested by IgM enzyme-linked immunosorbent assay (ELISA) to detect anti-DENV, anti-CHIKV, and anti-ZIKV IgM antibodies. CDC recommended that specimens be tested by rRT-PCR and ELISA using the following diagnostic algorithm based on date of specimen collection post-illness onset:
0-4 days: rRT-PCR only 4-7 days: both rRT-PCR and IgM ELISA >7 days: IgM ELISA only
Patients with nucleic acids detected by rRT-PCR for any of the arboviruses tested were considered to have
Implementation of Activities
Implementation of activities took place primarily in November 2017, following a formal request for assistance from the Burkina Faso MOH to CDC. Coordination and technical meetings were organized among the Burkina Faso MOH, CDC, and DAVYCAS International. A surveillance protocol was developed by all stakeholders and was reviewed by the Burkina Faso MOH and CDC institutional review boards (IRB), which determined the protocol involved nonresearch emergency public health activities. Following a meeting with key high-level stakeholders from the Burkina MOH, CDC, DAVYCAS International, and leaders from both sites, half-day site-specific training for clinicians, laboratory technicians, and hospital administrators was conducted in late November. Additional standard operation procedures, such as specimen preparation procedures, were developed to guide in-field activities.
CDC technical assistance to NRL-VHF was provided through the CDC Burkina Faso Country Office as well as visits from laboratory experts in November and December 2017 and January 2018. Assistance focused on specimen management, testing quality control, and data management. Laboratory reagents and equipment were also donated by CDC to ensure timely specimen testing.
Data Management and Analysis
EpiInfo (version 7) databases were developed based on the case report form. In order to minimize the data management challenges, site-specific databases were developed for each of the sentinel sites and the NRL-VHF for entry and storage of relevant data; data from the sentinel sites and NRL-VHF were sent weekly to the MOH surveillance team. Unique case identification numbers were assigned via barcode stickers, which were developed by DAVYCAS International. Stickers with matching case numbers were placed on case report forms and specimen tubes; case numbers were entered into databases manually until barcode scanners could be implemented at sites. After case report forms were completed manually by clinicians, and copies of the case report forms were made prior to specimen shipment to NRL-VHF. Data on case demographics, clinical characteristics, dengue RDT results, and specimen collection were entered into the sentinel site databases. Case report form sections on specimen receipt and diagnostic testing were completed by NRL-VHF staff and entered into the laboratory database, along with case number and other critical case identification and definition variables. The Burkina Faso MOH surveillance team used the unique case numbers to merge datasets sent from the sentinel sites and NRL-VHF for data cleaning and analysis (Figure 2).
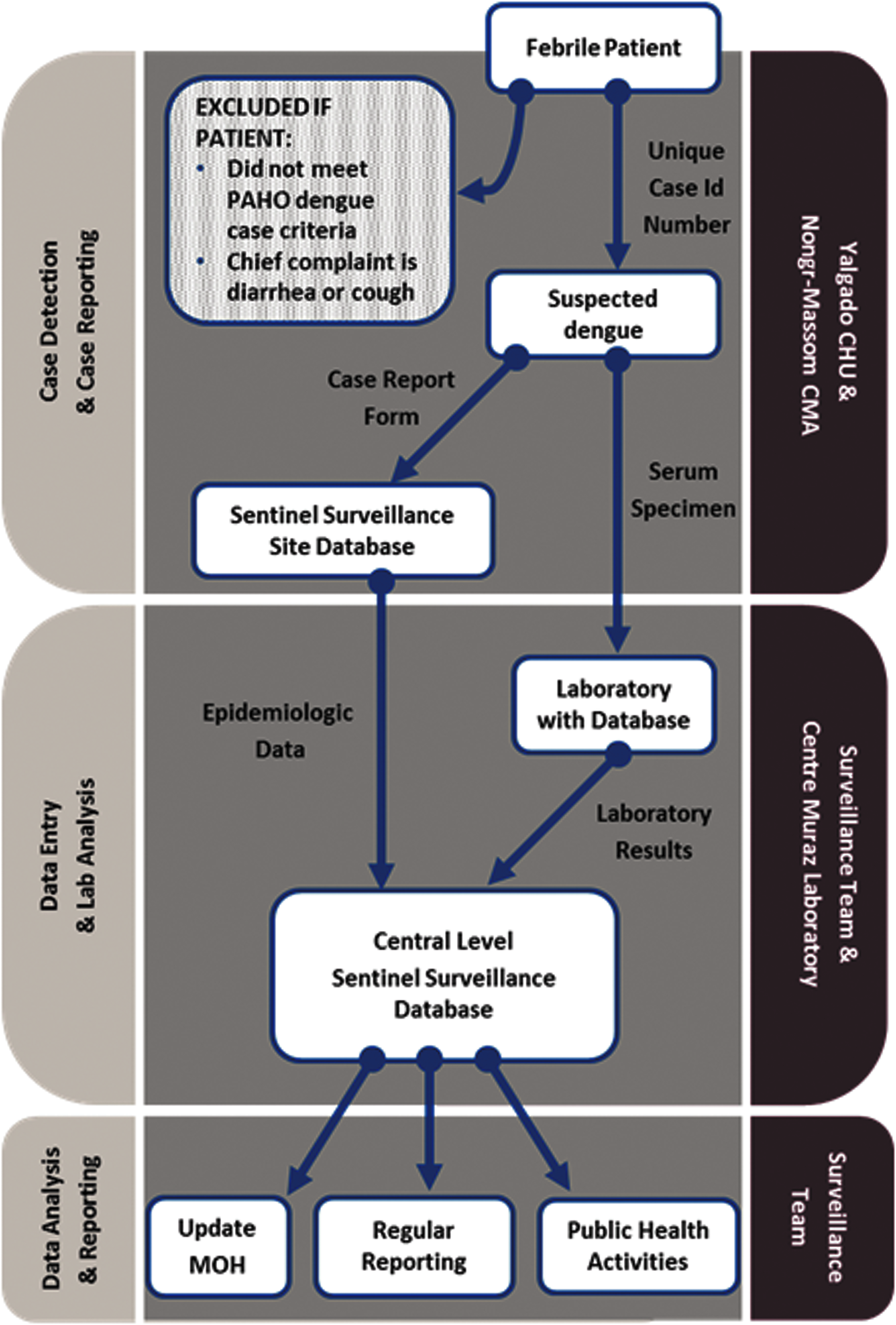
Case-Reporting Flow for Arbovirus Sentinel Surveillance in Yalgado University Hospital Center and Nongr-Massom District Hospital from October 31, 2017 to April 4, 2018
Leveraging Resources and Investment
Establishing a laboratory-based sentinel surveillance system was intended to be an effective tool to provide high-quality and timely data for the Burkina Faso MOH to guide public health action. To do this effectively during an outbreak requires investment of new resources but, more importantly, depends on the ability to leverage existing systems and investments. Since committing to the GHSA in 2015, the Burkina Faso MOH has put into place or reinforced routine surveillance for epidemic prone diseases, which the ministry was able to draw on as a part of the dengue outbreak response. Specific systems and developed resources include an innovative, effective, and rapid specimen transport system implemented by the Burkina Faso MOH and SONAPOST, enhanced diagnostic capacity including reagents and equipment, extensive experience with the internationally recognized meningitis surveillance system and associated data management system, and human resources trained via the Field Epidemiology Training Program (FETP) 43 with experience establishing a laboratory-based SARI sentinel surveillance system.
In addition to leveraging these existing resources, partners provided additional financial and technical support to the Burkina MOH to implement arbovirus sentinel surveillance during the dengue outbreak. Total direct financial support amounted to $16,631.49 (8,842,850 CFA francs) over 4 months to provide training, specimen transport, and monitoring of sites. Total training costs were $6,068.41 (36% of total costs), with $4,723.25 for on-site trainings and $1,345.16 for data management training sessions.
Specimen transport from Ouagadougou to the NRL-VHF in Bobo-Dioulasso cost $3,463.46 (21% of total costs). Support to ensure daily monitoring of sites and assure quality of data amounted to $3,715.13 (19% of total costs). The remaining costs were for material support, including 2 refrigerators for specimen storage ($2,165.72 for both) and 2 photocopiers ($1,412.42 for both) for sentinel sites, and other associated materials. In addition to this direct financial contribution, partners also donated diagnostic reagents and dengue RDTs and provided expert technical consultations, both onsite in Burkina Faso and remotely via teleconference or email.
Discussion
In November 2017, sentinel arbovirus surveillance was rapidly designed and implemented through a coordinated effort among the Burkina Faso MOH, CDC, and DAVYCAS International. The capacity built through this effort will contribute to understanding the characteristics and burden of arboviral disease, so that targeted public health interventions to prevent further disease can be defined and implemented. Moreover, by leveraging new and existing resources to establish this sentinel surveillance, Burkina Faso has efficiently acquired more knowledge and capacity to detect emerging pathogens that could be a public health threat to the region. The sentinel surveillance improved data collection through a case report form, systematic testing of specimens, and linking patient information with laboratory results through the data management system. Collected epidemiologic, clinical, and laboratory data are helping to better understand the arbovirus infection and AFI that caused the 2017 dengue outbreak and can be used to prevent future outbreaks or quickly detect and identify emerging outbreaks.
Mobilization of existing partnerships and systems strengthened or established under GHSA were also crucial in rapidly implementing the surveillance system. As mentioned, effective sentinel surveillance design drew on protocols and best practices for SARI sentinel surveillance and the SONAPOST specimen transport system. This included use of stickers to easily link case report forms and patient specimens, as well as previously developed specimen transport protocols. Furthermore, during nonepidemic times, partnerships have built local emergency public health capacity to strengthen outbreak response. Through the CDC-sponsored FETP training, 43 1 central level staff member had completed his advanced FETP program in July 2017, 2 staff members from Nongr-Massom District Hospital participated in the Frontline FETP in February 2017, and 1 staff member at Yalgado CHU completed Frontline FETP training in September 2016. Furthermore, a central level MOH staff received training through CDC's Atlanta-based public health emergency management (PHEM) fellowship. 44
Moreover, success was greatly aided by partners' support to prevent and detect dengue and other arboviruses provided during nonepidemic periods. Following the 2016 dengue epidemic, the Burkina Faso MOH worked closely with partners, including CDC and WHO, to better interpret public health data and improve outbreak response. This included high-level meetings held in February 2017 to distill key lessons learned. Finally, CDC has supported the NRL-VHF through technical training and provision of equipment and reagents to enhance viral diagnostics and improve biosafety environment and practices, including arbovirus testing using the Trioplex and DENV multiplex RT-PCR assays.
This outbreak response highlights the interrelation of activities focused on disease prevention, detection, and response. Through careful planning, dengue response activities in Burkina Faso drew on existing resources to improve the public health system's ability to detect dengue and other arboviruses. In March 2018, the experience of arbovirus sentinel surveillance was presented at a partner meeting led by WHO that brought together representatives from countries across the West Africa subregion and key partners such as the West Africa Health Organization. Lessons learned will be used to expand routine sentinel surveillance in 2018 in Burkina Faso and improve arbovirus case reporting nationally. While emergency public health response requires timely action, proper planning and review of experiences can help support prevention and detection activities during nonepidemic times.
Despite these achievements, several challenges and limitations bear mention. First, the surveillance system was unable to incorporate entomologic surveillance, which could have added an important component to guide vector control activities. Such activities are being considered as the sentinel surveillance system expands to new sites. Second, data management presented an ongoing challenge, especially given limited time for end-user testing in the field. Modifications to the data management system will improve data quality by refining internal quality control measures and the structure of the databases. Use of digital scanners to read stickers will improve entry of unique case numbers. Finally, the surveillance system was implemented after the peak of the outbreak, which will limit data analysis. Delays were attributed to required procedural steps and ethical reviews, as well as material preparations for implementation.
Overall, the Burkina Faso MOH was able to mobilize existing partnerships and resources toward the rapid implementation of arbovirus sentinel surveillance among AFI cases during a dengue outbreak. Information gathered will render useful information on the outbreak, including data on additional arboviruses potentially circulating and analysis of clinical features and case management of dengue in Burkina Faso focused on severe, hospitalized cases. The rich experience garnered will be used to implement sustainable routine AFI surveillance, given expected arbovirus endemicity, and has the opportunity to enhance Burkina Faso's ability to detect other viral pathogens of epidemic potential.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
