Abstract
Yunden Droma, Masao Ota, Nobumitsu Kobayashi, Michiko Ito, Toshio Kobayashi, and Masayuki Hanaoka. Genetic associations with the susceptibility to high-altitude Pulmonary Edema in the Japanese population. High Alt Med Biol. 26:215–226, 2025.—High-altitude pulmonary edema (HAPE) is a life-threatening, noncardiogenic pulmonary condition that may occur in individuals rapidly ascending to altitudes higher than 2,500 m above sea level. Exaggerated hypoxia-induced pulmonary hypertension plays a critical role in its pathophysiological mechanism. In addition to environmental factors such as hypoxia and hypobaria at high altitudes, individual genetic predisposition significantly influences HAPE occurrence. Several candidate genes have been proposed based on the pathophysiology of HAPE, particularly involving the hypoxia-induced factor pathway and vasodilators/vasoconstrictors. Over the past two decades, we have investigated the associations between susceptibility to HAPE and these candidate genes, including genes EPAS1 (endothelial Per-ARNT-Sim [PAS] domain protein 1), EGLN1 (egl-9 family hypoxia inducible factor 1), eNOS (endothelial nitric oxide synthase), ACE (angiotensin-converting enzyme), and TIMP3 (tissue inhibitor of metalloproteinase 3) in the Japanese population. This review summarizes the major findings of these studies, shedding light on genetic associations with HAPE in the Japanese population.
Introduction
High-altitude pulmonary edema (HAPE) is a form of noncardiogenic pulmonary edema that may occur in unacclimatized individuals rapidly ascending to altitudes above 2,500 m (Kobayashi et al., 1987). Symptoms typically include dyspnea, cough, fatigue, headache, nausea, and vomiting. Exaggerated hypoxia-induced pulmonary hypertension plays a critical role in the pathophysiological mechanism of HAPE, leading to overperfusion of the pulmonary circulation and stress failure of pulmonary capillaries. This results in the leakage of fluid, proteins, and cells into the alveoli (Hanaoka et al., 2000; Scherrer et al., 1999; Swenson et al., 2002; West and Mathieu-Costello, 1992). Continuous exposure to hypoxia triggers inflammatory reactions in the lungs, accelerating the formation of pulmonary edema (Droma et al., 2001; Kubo et al., 1998).
Japan is an island nation with approximately 73% of its land covered by mountains, including over 50 peaks exceeding 2,500 m, such as Mount Fuji at 3,776 m. The Japan Alps feature several summits over 3,000 m, attracting climbers year-round. Located at 610 m in Matsumoto City, Shinshu University Hospital serves as a central medical facility for climbers facing altitude-related diseases.
Until the first case of HAPE was identified in Japan in 1966, severe dyspnea or coma developing in climbers above 2,500 m was often misdiagnosed as pneumonia or congestive heart failure (Kobayashi et al., 1987). Since then, 67 patients have been treated at Shinshu University Hospital, utilizing management strategies such as rapid descent, bed rest, oxygen supplementation, and the administration of the calcium channel blocker nifedipine to reduce pulmonary hypertension and improve oxygenation. Owing to the efficient helicopter rescue system and the moderate altitude of the Japan Alps, few patients were admitted to the hospital in severe condition, and most recovered with bed rest and oxygen supplementation. Over the past 50 years, four severe cases of HAPE resulted in death, occurring in 1974, 1977, 1979, and 1991, with autopsies confirming the pathological diagnoses (Droma et al., 2001).
Based on the extensive clinical data collected from these patients, we conducted a series of studies focusing on the clinical features and pathophysiology of HAPE. A review summarizing the major achievements of the clinical studies was published recently (Hanaoka et al., 2024). Furthermore, we carried out a series of case-control genetic association studies at our institute to identify candidate genes relevant to HAPE pathogenesis and to enhance our understanding of the genetic factors associated with HAPE susceptibility in the Japanese population. This review will provide an overview of the significant results from these genetic association studies and outline the general genetic pathology of HAPE in the Japanese population.
Diagnostic criteria for HAPE in the Japanese population
While HAPE shares common clinical features worldwide, variations occur due to differences in ascent patterns, patient demographics, health care access, and environmental factors. At Shinshu University Hospital, HAPE diagnosis was based on criteria proposed by Hultgren and Marticorena (Hultgren and Marticorena, 1978), including the following: the onset of typical symptoms such as cough and dyspnea at rest after arriving at high altitude; absence of infection signs; presence of pulmonary rales and cyanosis; prompt symptom resolution with bed rest and supplemental oxygen after descent; and chest roentgenographical findings consistent with pulmonary edema. These criteria have been widely applied in global research and routinely used in our clinical and genetic studies on HAPE (Hanaoka et al., 2024).
Subjects of case-control genetic association studies
A series of case-control genetic association studies were conducted at our institute to identify genetic variants associated with susceptibility to HAPE. The case group included individuals who experienced at least one episode of HAPE in the Japanese Alps and were treated at Shinshu University Hospital. Their clinical features included dyspnea, dry cough, fatigue, headache, productive cough, nausea/vomiting, physical hypoxia with low arterial partial pressure of oxygen and oxygen saturation (SpO2), and pulmonary edematous shadows on chest roentgenography. These individuals were classified as HAPE-susceptible (HAPE-s) subjects. The control group (HAPE-c) consisted of Japanese mountaineers who frequently climbed mountains higher than 3,000 m in the Japanese Alps but had never experienced HAPE at high altitudes. All participants were healthy and free of pre-existing cardiopulmonary conditions. As of the latest case-control association study (Horiuchi et al., 2015), our database included 59 HAPE-s subjects (52 males, 7 females; average age 34.2 years) and 67 HAPE-c subjects (58 males, 9 females; average age 37.0 years). The exact numbers of HAPE-s and HAPE-c subjects in each study are listed in Table 1.
The Minor Allele Frequencies of Single Nucleotide Polymorphisms in the Candidate Genes in Japanese HAPE-s and HAPE-c Subjects, as well as in European and East Asian Populations
Allele frequencies are available in 1000 Genomes (https://asia.ensembl.org/Homo_sapiens/Info/Index). Ensembl release 113-October 2024.
P: Chi-square test (2 × 2 contingency table).
1EPAS1, endothelial Per-ARNT-Sim (PAS) domain protein 1; 2EGLN1, egl-9 family hypoxia inducible factor 1; 3eNOS, endothelial nitric oxide synthase; 4ACE, angiotensin-converting enzyme; 5TIMP3, tissue inhibitor of metalloproteinase 3; 6VEGFA, vascular endothelial growth factor-A; 7TH, tyrosine hydroxylase; 8F5, coagulation factor V.
A, adenine; C, cytosine; G, guanine; T, thymine; b/a, “b” represents a common large allele with 5 tandem 27-bp repeats in intron 4 of eNOS; “a” represents a rare small allele with 4 tandem 27-bp repeats in intron 4 of eNOS; I/D, 287-base pair sequence insertion (I) or deletion (D) in intron 16 of ACE; (TCAT)n, tetranucleotide microsatellite repeats; A, B, C, D, and E alleles, five alleles of the (TCAT)n tetranucleotide repeats identified in the TH gene; Met, methionine; Val, valine; R506Q, amino acid arginine (R) replaced by glutamine (Q) at position 506 of F5.
MAF, minor allele frequency; HAPE, high-altitude pulmonary edema; HAPE-s, HAPE susceptible subjects with history of HAPE; HAPE-c, HAPE control climbers without history of HAPE; SNP, single nucleotide polymorphism; PCR, polymerase chain reaction; RFLP, restriction fragment length polymorphism; No., number.
Susceptibility to HAPE in the Japanese population
Recurrence is one of the clinical phenotypes associated with susceptibility to HAPE. Individuals who have previously experienced HAPE are more likely to have future episodes at high altitudes. The first case of HAPE recurrence in Japan was reported in 1980 (Fukushima et al., 1980). Each episode of HAPE may affect different areas of the lungs on chest roentgenography in recurrent cases (Koyama et al., 1985). One recurrent case experienced his first episode of HAPE with edema predominantly affecting the left lung (Fig. 1A), while the second episode showed extensive infiltration of the right lung, in addition to the left lung that was involved in his first episode of HAPE (Fig. 1B).
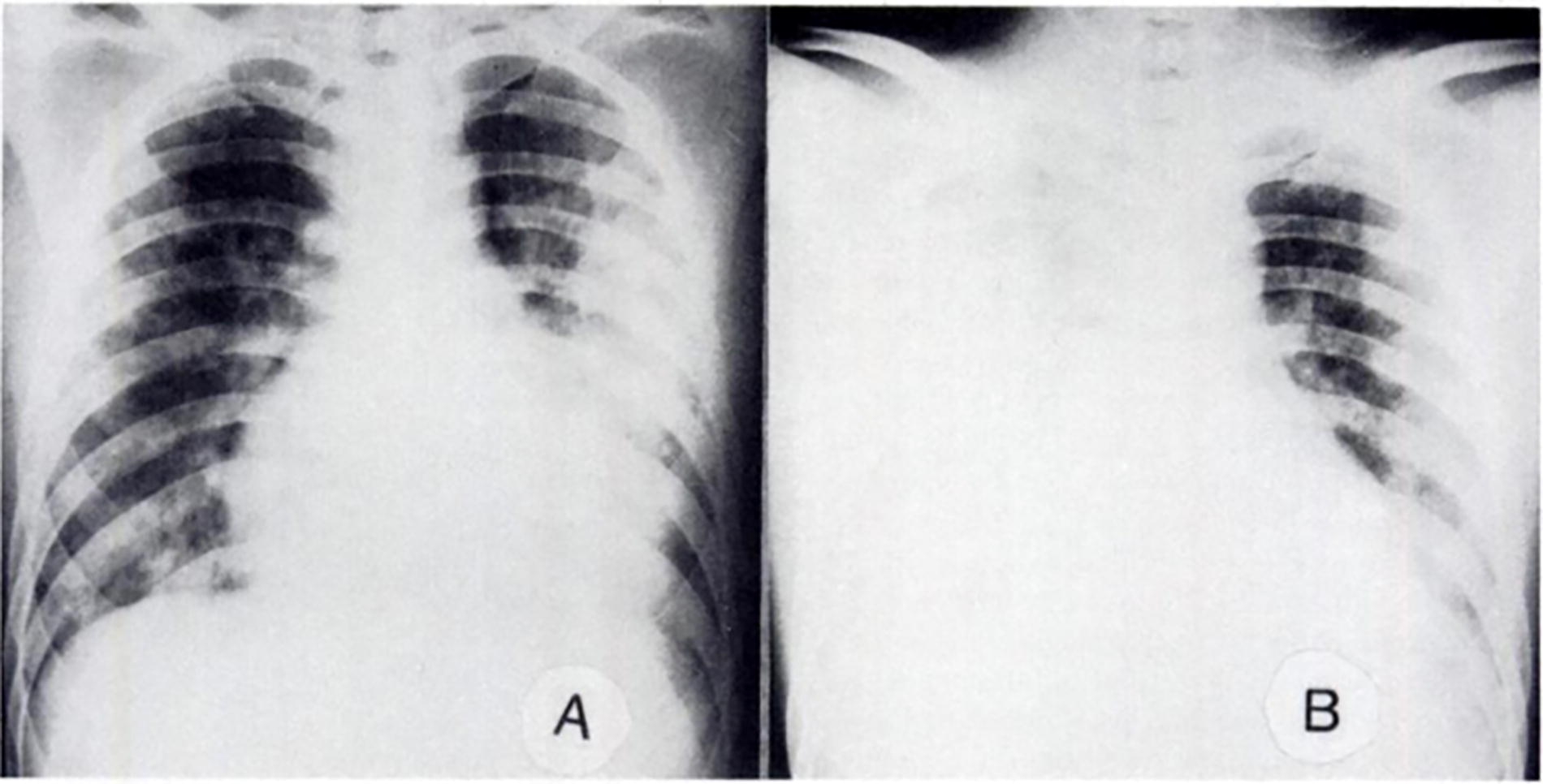
Chest roentgenograms of a recurrent case of HAPE.
The susceptibility to HAPE was further investigated through a prospective field study conducted at our institute in the 1980s, involving 12 Japanese subjects with a previous history of HAPE and 11 healthy climber controls with no history of HAPE (Seki, 1988). In this study, the acute mountain sickness (AMS) score was evaluated and SpO2 levels were measured. This study was conducted during a 4-day mountain journey in the Japan Alps, at altitudes ranging from 2,700 to 2,900 m. Chest roentgenography was performed both before ascent and after descent to diagnose HAPE. The results revealed that the AMS score was significantly higher (Fig. 2A) and SpO2 was significantly lower (Fig. 2B) in subjects with a history of HAPE compared to climbers without a history of HAPE at 2,850 m. On day 4, one subject with a history of HAPE was diagnosed with HAPE, as confirmed by a chest X-ray showing edematous infiltrations in the bilateral lower lung fields (Fig. 2C).
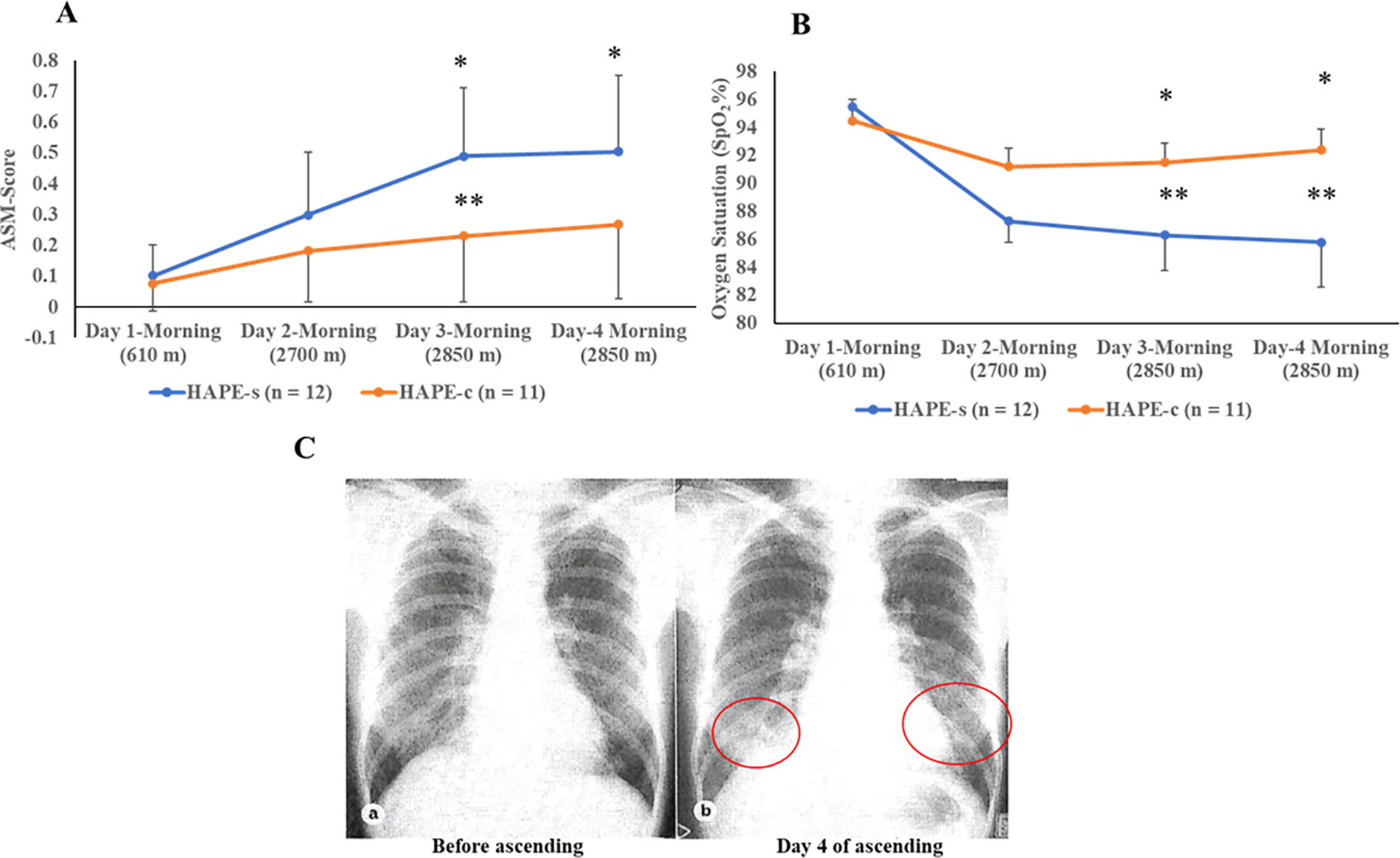
Field study on susceptibility to HAPE. (
Hanaoka et al. later reported a HAPE recurrence rate of 19.6% among Japanese patients (Hanaoka et al., 1998). Additionally, Vock et al. demonstrated a recurrence rate of 66% in lowlanders with a history of HAPE after ascending to 4,559 m within 42 hours, confirmed by radiographical evidence of HAPE (Vock et al., 1989). These findings strongly suggest that individuals with a history of HAPE are prone to recurrence, highlighting the potential role of genetic factors in HAPE development.
Case-control genetic association studies
Accordingly, we conducted a series of case-control association studies to identify specific genetic variants associated with HAPE susceptibility. All studies were approved by the Ethics Committee of Shinshu University School of Medicine (Matsumoto, Japan). Venous blood samples were collected from all subjects with written informed consent for genomic DNA extraction. Genetic variants were identified using polymerase chain reaction (PCR), restriction fragment length polymorphism (RFLP), or the TaqMan® single-nucleotide polymorphism (SNP) genotyping assay, as appropriate (Table 1).
Candidate genes involving hypoxia-induced factor pathway
Hypoxia is the initial trigger in the development of HAPE in lowlanders rapidly exposed to high altitudes. Hypoxia-inducible factors (HIFs) and their pathways activate downstream cascades of multiple molecular events associated with hypoxia (Semenza, 2001). The gene encoding endothelial Per-ARNT-Sim (PAS) domain protein 1 (EPAS1, also known as HIF2α) and the gene encoding egl-9 family hypoxia-inducible factor 1 (EGLN1, also known as prolyl hydroxylase domain protein 2) play key roles in the HIF pathway in humans (Bigham and Lee, 2014). These two genes have been demonstrated to be the most interesting genetic elements involving high-altitude hypoxia adaptation in humans (Bigham and Lee, 2014; Simonson et al., 2010). In addition, study demonstrated that EGLN1 genetic polymorphisms were associated with the SpO2 response in a Japanese cohort exposed to acute hypobaric hypoxia (Yasukochi et al., 2018). Moreover, Mishra et al. revealed that EGLN1 genetic polymorphisms were associated with HAPE susceptibility in the Indian population through upregulation of EGLN1 gene mRNA expression (Mishra et al., 2013). Thus, to elucidate the genetic susceptibility to HAPE associated with the HIF pathway, we investigated three SNPs in the EPAS1 (rs13419896, rs4953354, and rs495338) and three SNPs in the EGLN1 (rs1435166, rs7542797, and rs2153364) in 59 HAPE-s subjects and 67 HAPE-c subjects using the TaqMan SNP Genotyping Assay (Horiuchi et al., 2015). We found that the interaction between EPAS1 rs13419896 and EGLN1 rs2153364 was significantly associated with HAPE susceptibility in a pairwise model (p = 0.0049) (Fig. 3). This study suggests that genes within the HIF pathway may be associated with the susceptibility to HAPE, however, the underlying mechanisms are not yet fully understood.
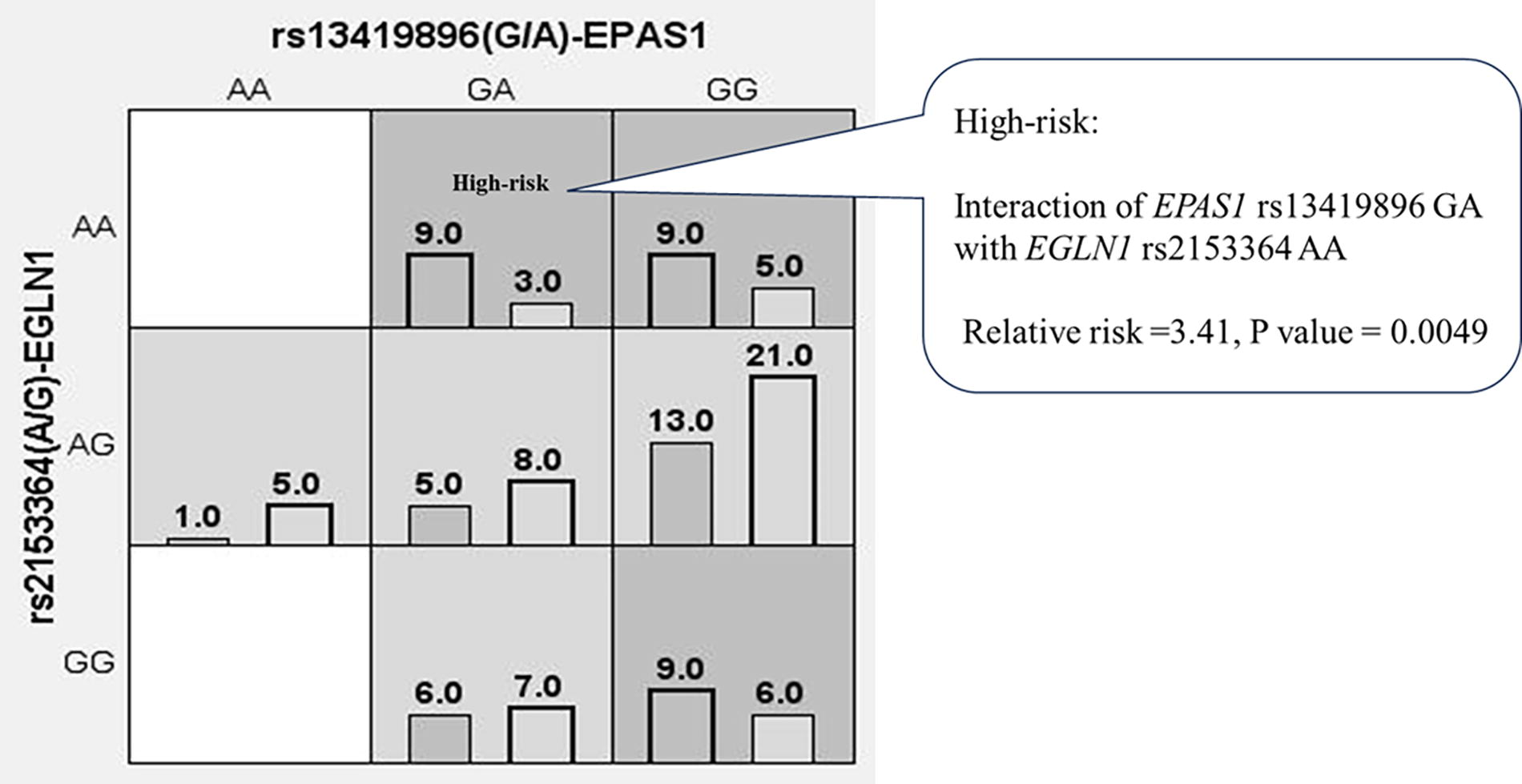
Interaction between the EGLN1 and EPAS1 genes in HAPE-susceptible subjects and HAPE controls. The interaction between EGLN1 rs2153364 AA and EPAS1 rs13419896 GA was significantly associated with susceptibility to HAPE (relative risk = 3.41; p = 0.0049). The areas shaded in dark grey represent a high risk of HAPE as determined by the pairwise model. EPAS1, endothelial PAS domain protein 1; EGLN1, egl-9 family hypoxia-inducible factor 1.
Candidate genes involving pulmonary hypertension
High-altitude hypoxia stimulates pulmonary artery contraction, leading to increased pulmonary artery pressure (PAP) that triggers stress failure in the pulmonary capillaries resulting in the movement of protein and fluid into the alveoli, causing pulmonary edema (Bärtsch et al., 2005; Maggiorini et al., 2001). To demonstrate the susceptibility of pulmonary hemodynamics to hypoxia, hypobaria, and exercise in subjects with HAPE, we measured the responses of PAP and pulmonary vascular resistance index (PVRI) to normobaric hypoxia (15% oxygen), hypobaric hypoxia (515 Torr), and exercise (supine bicycle ergometer) in five Japanese HAPE-s subjects and five HAPE-c subjects using right heart catheterization (Kawashima et al., 1989). The results indicated significantly greater responses of PAP and PVRI to normobaric hypoxia, hypobaric hypoxia, and exercise in the HAPE-s group compared with the HAPE-c group (Kawashima et al., 1989). These findings were replicated in another study that assessed hemodynamic responses to hypoxia in seven Japanese HAPE-s and six HAPE-c subjects under hypoxic conditions, utilizing both right heart catheterization and pulse Doppler echocardiography (Yagi et al., 1990). These significant results strongly suggest that pulmonary hemodynamics in HAPE-s subjects are instinctively susceptible to hypoxia, hypobaria, and exercise, greatly contributing to hypoxia-induced pulmonary hypertension in HAPE.
Based on these phenotypes, we identified potential candidate genes to investigate their associations with susceptibility to pulmonary hemodynamics in HAPE-s subjects, aiming to elucidate the genetic mechanisms underlying hypoxia-induced pulmonary hypertension.
Endothelial nitric oxide synthase gene
The endothelial nitric oxide (NO) synthase gene (eNOS or NOS3) encodes the enzyme responsible for synthesizing NO in endothelial cells (Andrew and Mayer, 1999). NO serves as a vital endogenous vasodilator, regulating pulmonary vascular tone and maintaining pulmonary vascular resistance (PVR) under vasodilatory conditions, thereby controlling vascular tone and PAP (Klinger and Kadowitz, 2017). Variants or alterations in the eNOS can disrupt NO production, leading to vascular dysfunction, which may contribute to pulmonary vascular constriction and pulmonary hypertension (Tran N et al., 2022). In the context of pulmonary hypertension associated with HAPE, NO plays a vasodilatory role in maintaining pulmonary vascular tone and keeping vascular resistance within normal ranges. Study has shown that the concentration of NO exhaled from the respiratory tract of HAPE-s subjects under hypoxic condition is reduced and showed a negative correlation with PAP (Busch et al., 2001). Moreover, inhalation of NO has been found to reduce pulmonary hypertension and improve arterial oxygenation in HAPE patients (Scherrer et al., 1996). These findings suggest that defective NO synthesis may contribute to elevated PAP in HAPE patients. Therefore, we investigated the eNOS variants in association with the increased PAP in HAPE-s subjects.
The missense variant rs1799983 (G894T) involves a nucleotide substitution from guanine (G) to thymine (T) at position 894, resulting in an amino acid change from glutamic acid (Glu) to aspartic acid (Asp) at position 298 in exon 7 of the eNOS (Lembo et al., 2001). A meta-analysis study concluded that the eNOS rs1799983 SNP is associated with an increased risk of hypertension (Shi et al., 2021). Additionally, the eNOS4b/4a is a variable number tandem repeat consisting of either 4 or 5 repeats of 27 base pairs in length located in intron 4 of the eNOS, and it has been linked to cardiovascular disease in Chinese patients (Tong et al., 2013). To explore the association of these variants with HAPE susceptibility, we investigated the genotypes of rs1799983 and eNOS4b/4a variants in 41 HAPE-s and 51 HAPE-c Japanese subjects using PCR followed by RFLP analysis (Droma et al., 2002). The results revealed that the thymine (T) allele in the rs1799983 variant and the eNOS4a allele were significantly more prevalent in the HAPE-s subjects compared to the HAPE-c controls. Notably, 11 out of 41 (26.8%) HAPE-s subjects carried both the T allele and the eNOS4a allele, whereas none of the 51 HAPE-c controls carried the two alleles simultaneously (p = 0.000059). These findings suggested a significant association between the susceptibility to HAPE and the eNOS genetic variants in the Japanese population. Meanwhile, the rs1799983 variant and the eNOS4a allele were also demonstrated to be associated with HAPE susceptibility in Chinese (Sun et al., 2010) and Indian HAPE-s subjects (Ahsan et al., 2004; Ahsan et al., 2006). Furthermore, a meta-analysis verified the significant association between HAPE susceptibility and these eNOS genetic variants in Asian HAPE-s subjects (Wang et al., 2013). However, such significant results were not replicated in Caucasian HAPE-s subjects (Smith et al., 2006; Weiss et al., 2003). These contrasting results may be attributed to genetic heterogeneity and/or ethnic-specific allele variations (Woodward et al., 2022).
The eNOS protein comprises a heme-binding region and a calcium/calmodulin-binding region. The exon 7 is located in the intermediate portion between the heme binding and calcium/calmodulin binding sites, while the intron 4 is situated next to the heme binding site (Wang et al., 1997). Molecular biological methods analyzed the functional changes of the rs1799983 variant, revealing that it causes a conformational change in the eNOS protein, transitioning it from a helix to a tight turn, thereby interfering with the function of the eNOS protein and reducing NO production (Joshi et al., 2007).
Angiotensin-converting enzyme gene
The angiotensin-converting enzyme (ACE) plays a crucial role in regulating vascular resistance and blood pressure, with its activity often elevated in patients with essential hypertension. ACE inhibitors are commonly prescribed to treat essential hypertension (Ahmad et al., 2023). The I/D variant in the ACE gene involves an insertion (I) or deletion (D) of an Alu repetitive element (287 base pairs) in an intron of the ACE gene (Rigat et al., 1990). The I/D variant (rs4646994) in the ACE gene is associated with essential hypertension and cardiovascular complications (Kumari et al., 2022; Li, 2012).
Hypoxia stimulates ACE gene expression and ACE synthesis (King et al., 1989; Nong et al., 1996). ACE activity is significantly heightened in patients with HAPE, contributing to hypoxia-induced pulmonary vasoconstriction (Charu et al., 2006). Studies have shown that the I allele is associated with higher SpO2 levels in elite mountain climbers at high altitudes, potentially enhancing physical performance (Woods et al., 2002).
We examined the I/D variant in the ACE in 49 Japanese HAPE-s and 55 HAPE-c subjects using PCR analysis (Hotta et al., 2004). The results indicated a significantly higher frequency of the ACE D allele in the HAPE-s group compared with the HAPE-c group. Moreover, the PVR and PVRI were significantly higher in HAPE-s subjects carrying the D allele than those without the D allele (PVR, p = 0.015; PVRI, p = 0.028). These findings suggest that the ACE genetic variant may be associated with hypoxia-induced pulmonary hypertension in the Japanese population (Hotta et al., 2004).
However, this statement remains contentious due to varying results observed across different ethnic populations. Association between HAPE susceptibility and ACE I/D variant has not been found in studies involving Caucasians (Dehnert et al., 2002). These discrepancies may be attributed to the diverse genotype or allele distributions of the ACE I/D variant across ethnic groups worldwide (Ahmad Yusof and Che Muhamed, 2021; Barley et al., 1996).
Nevertheless, the ACE I/D variant is associated with susceptibility to HAPE among Asians. Individuals with the ACE D allele exhibit elevated ACE levels (Rigat et al., 1990). Increased ACE activity promotes pulmonary vasoconstriction, contributing to hypoxia-induced pulmonary hypertension and potentially increasing the risk of developing HAPE.
Tissue inhibitor of metalloproteinase 3 gene
A genome-wide association study is commonly used to identify significant genetic variations across the entire genome that are associated with major human diseases (He and Gai, 2023a). This approach has been successfully employed to uncover genetic adaptations to high-altitude hypoxia in indigenous Tibetans, identifying numerous important genetic variants linked to high-altitude adaptation in Tibetan highlanders (Hu et al., 2017).
We conducted a study using 400 polymorphic microsatellite markers across the entire genome of 53 HAPE-s and 67 HAPE-c Japanese subjects to identify candidate genetic variants associated with HAPE susceptibility, without bias from evolutionary genomic changes (Kobayashi et al., 2013). The study identified nine significant microsatellite markers associated with HAPE susceptibility in the Japanese population. Notably, the gene encoding tissue inhibitor of metalloproteinase 3 (TIMP3) is located very close to one of these nine significant markers.
We then genotyped six SNPs of the TIMP3 in the same 53 HAPE-s and 67 HAPE-c Japanese subjects using a TaqMan® SNP genotyping assay. The analysis revealed a significant association of the rs130293 (A/G) with the HAPE-s subjects. The allele frequency of rs130293 was significantly different between the HAPE-s and HAPE-c groups (p = 0.00049), suggesting a strong association between this variant and HAPE susceptibility.
TIMP proteins play a crucial role in maintaining the structural integrity of the interstitial matrix and basement membrane during tissue repair, including lung tissue repair (Felbor et al., 1995). The interstitial matrix is vital for maintaining low pulmonary microvascular permeability, which protects against pulmonary edema (Beretta et al., 2021). Damage to the matrix integrity is implicated in the development of interstitial edema, a hallmark of the early stages of HAPE (Vock et al., 1989). The TIMP3 gene is located on chromosome 22 and spans approximately 55 kb of genomic DNA across five exons (Apte et al., 1994). The SNP rs130293, located in intron 1 of TIMP3, may influence mRNA stability and the efficiency of transcription and translation, potentially altering protein function and its biological properties (Lynch and Kewalramani, 2003). We propose that TIMP3 is associated with HAPE susceptibility, likely through its role in maintaining lung interstitial matrix integrity and preventing pulmonary edema.
Genetic variants negatively associated with HAPE susceptibility
Among the case-control studies at our institute on HAPE susceptibility associated with candidate genes selected based on well-known pathophysiological mechanisms, some studies yielded negative results.
Polymorphisms of tyrosine hydroxylase gene
In normal physiology, respiratory ventilation increases in response to hypoxia to counteract low oxygen levels in individuals exposed to hypoxic conditions. This phenomenon is known as the hypoxic ventilatory response (HVR) (Teppema and Dahan, 2010). Matsuzawa et al. measured the HVR in ten Japanese HAPE-s subjects and eight HAPE-c subjects and found that the HAPE-s subjects had a significantly lower HVR compared with the HAPE-c subjects (Matsuzawa et al., 1989). This study suggests that a blunted HVR in individuals susceptible to HAPE may be involved in the development of HAPE. Other studies have also consistently demonstrated that a reduced HVR is a major pathophysiological factor contributing to exaggerated hypoxemia in HAPE-s individuals exposed to hypoxia, playing a critical role in the development of HAPE (Hackett et al., 1988; Hohenhaus et al., 1995; Hoiland et al., 2018; Urushihata K et al., 2021).
Tyrosine hydroxylase (TH) is a key enzyme found in the oxygen-sensitive cells of the carotid body. These cells sense and respond to hypoxia, increasing respiratory ventilation (Daubner et al., 2011). In this context, we investigated the association between TH genetic variants and susceptibility to HAPE, specifically examining the tetranucleotide microsatellite repeat [(TCAT)n] in intron 1 and the Met81Val variant in exon 2 of TH. This analysis was performed in 43 HAPE-s subjects and 51 HAPE-c subjects using PCR followed by RFLP analysis (Hanaoka et al., 2003). However, our study did not find significant associations between these two polymorphisms and HAPE susceptibility.
Polymorphisms of the human vascular endothelial growth factor-A gene
Vascular endothelial growth factor-A (VEGF-A) is a potent mitogen and permeability factor that is specific to endothelial cells. It binds to the VEGF-A receptor and regulates endothelial cell proliferation, migration, vascular permeability, and other endothelial functions (Shibuya, 2013). Hypoxia significantly upregulates VEGF-A expression (Ramakrishnan et al., 2014). Overexpression of VEGF-A in the lung increases pulmonary vascular permeability, leading to pulmonary edema (Zhang et al., 2018).
However, in our genetic association study involving 53 HAPE-s and 69 HAPE-c Japanese subjects, we found no significant associations between HAPE susceptibility and five SNPs of the VEGFA gene—namely rs699947 (C-2578A), rs1570360 (G-1154A), rs833061 (T-46°C), rs2010963 (G + 405 C), and rs3025039 (C936T) (Hanaoka et al., 2009).
Polymorphism of the coagulation factor V gene
Laboratory studies have found reduced platelet counts in patients with HAPE (Hefti et al., 2010). A case report described pulmonary thromboembolism in a Japanese patient with HAPE (Nakagawa et al., 1993). In addition, histological examination of lung tissues from autopsied HAPE cases in Japan revealed numerous scattered microvascular thrombi and fibrin deposits within congested arterioles and capillaries (Droma et al., 2001).
The rs6025 SNP (R506Q) in the human coagulation factor V gene (F5) is a specific variant in which arginine (R) is replaced by glutamine (Q) at position 506 of the factor V protein. This substitution impairs the inactivation of coagulation factor V, leading to a hypercoagulable state and an increased risk of thrombosis (Dahlbck, 1997). We investigated the R506Q SNP in 44 HAPE-s and 51 HAPE-c subjects but did not find any significant associations between the F5 R506Q variant and susceptibility to HAPE in the Japanese population (Droma et al., 2003).
Genetic variations associated with HAPE susceptibility in Japanese and other populations
Table 1 summarizes the results of our experiments on HAPE-s and HAPE-c subjects, alongside the genetic variant data for European and East Asian populations, based on the genetic information from 1000 Genomes Project (https://asia.ensembl.org/Homo_sapiens/Info/Index). The differences in minor allele frequencies of genetic variants in the EPAS1 (rs13419896 and rs4953388), EGLN1 (rs7542797 and rs2153364), eNOS (rs1799983), ACE (rs4646994), and VEGFA (rs699947, rs1570360, and rs833061) genes between East Asians and Europeans suggest population-specific variations. These genetic predispositions may differ across populations due to distinct genetic backgrounds and environmental adaptations, potentially contributing to the observed variations in HAPE prevalence among different ethnic groups (Kobayashi et al., 1987; Li, 2012; Singh et al., 1965; Vock et al., 1989). Genetic variants associated with disease susceptibility have been identified at varying frequencies across populations (Serre et al., 2008). Further research is needed to clarify the characteristic genetic backgrounds and provide insights into the gene functions underlying HAPE susceptibility in different populations, as well as to explore the multiple mechanisms involved in HAPE pathogenesis.
Theoretically, both the presence of significant susceptibility to HAPE and the relative stability of this susceptibility serve as reliable phenotypes for identifying individuals genetically predisposed to HAPE (MacInnis and Koehle, 2016; Visscher et al., 2008). This suggests that those genetically susceptible will consistently develop HAPE when exposed to high altitudes. However, to our knowledge, no data are available regarding whether Japanese individuals who are susceptible to HAPE experience HAPE again in regions outside Japan. Furthermore, no studies have yet compared the occurrence of HAPE in Japanese climbers with that in other populations globally. Investigating the reasons behind the relatively high incidence of HAPE in the Japanese population at lower altitudes presents an intriguing opportunity to generate valuable hypotheses and stimulate future research.
Pathways of interaction among proteins encoded by associated genes
The genes EPAS1, EGLN1, eNOS, ACE, and TIMP3 are significantly associated with susceptibility to HAPE and play key roles in various biological processes. The network diagram (Fig. 4), generated using STRING (version 12.0) ([https://string-db.org/]), illustrates the pathways of interaction among the proteins encoded by these genes (Szklarczyk et al., 2023). The HIF pathway, involving EPAS1 and EGLN1, is central to the cellular response to hypoxic condition. Under low-oxygen condition, the activity of EGLN1 is reduced, allowing the accumulation of EPAS1, which in turn activates downstream genes (e.g., eNOS and ACE), altering their expressions and facilitating vascular adaptation (Bigham and Lee, 2014). Hypoxia also directly affects ACE activity, influencing HAPE susceptibility by modulating vascular tone and fluid balance (Ahmad et al., 2023). TIMP3 may interact with EPAS1 to maintain vascular integrity and prevent excessive permeability, which is crucial for reducing pulmonary edema under hypoxic condition (Beretta et al., 2021). Overall, this network highlights the interconnected pathways involving oxygen sensing, vascular regulation, and extracellular matrix integrity, which contribute to either adaptation or maladaptation to high-altitude environments.
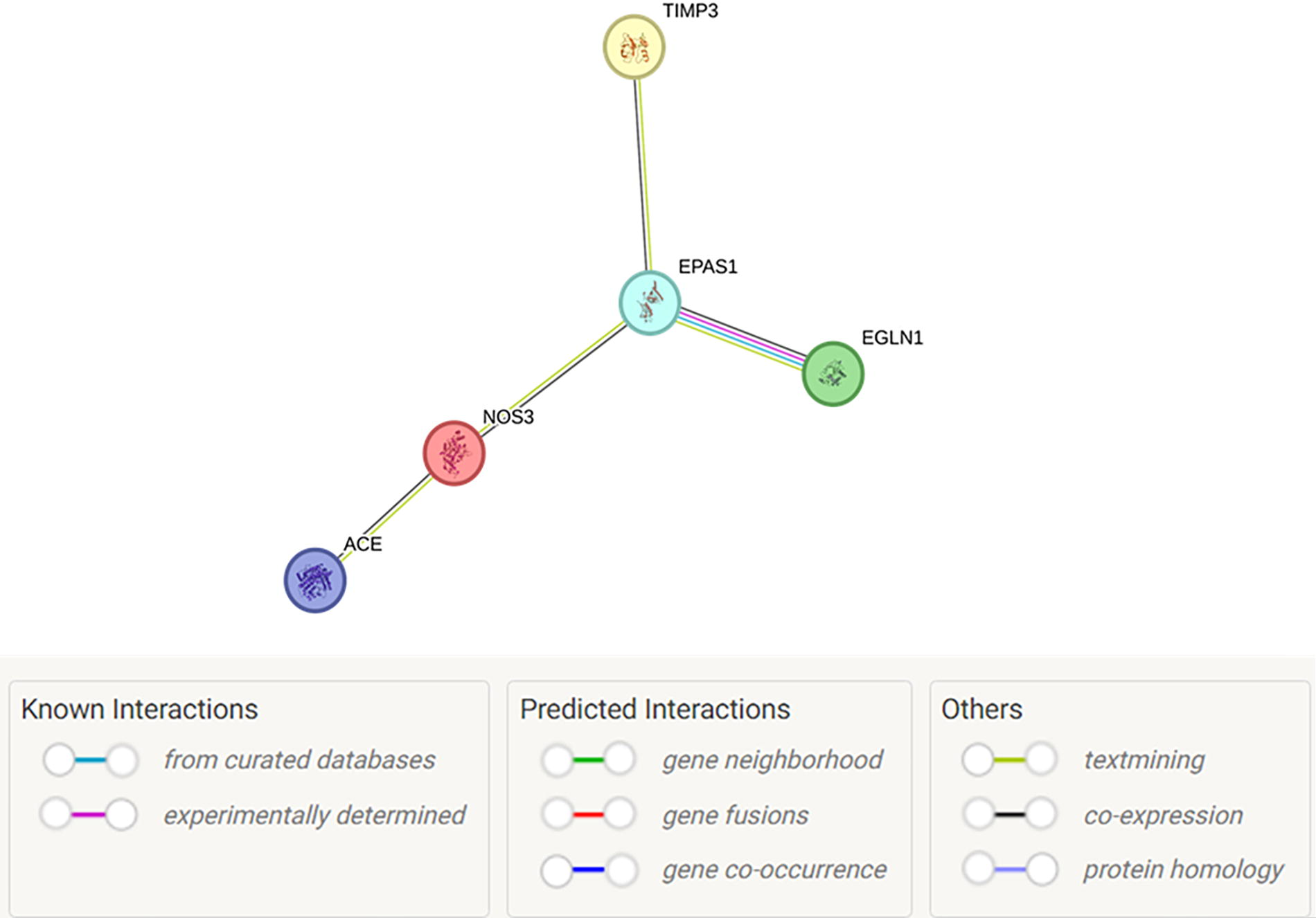
Pathways of interaction among proteins encoded by associated genes. This network illustrates the interactions among proteins EPAS1, EGLN1, eNOS, ACE, and TIMP3, which are encoded by their respective genes. It highlights key pathways involved in hypoxia sensing (EPAS1, EGLN1), regulation of vascular tone (eNOS/NOS3, ACE), and tissue integrity (TIMP3). The edges represent predicted or experimentally validated interactions, with colored links denoting the type of evidence supporting each interaction. The network diagram was generated using STRING (https://string-db.org/), version 12.0 (released July 26, 2023). ACE, angiotensin-converting enzyme; eNOS (NOS3), endothelial nitric oxide synthase; EGLN1, egl-9 family hypoxia-inducible factor 1; EPAS1, endothelial PAS domain protein 1; TIMP3, tissue inhibitor of metalloproteinase 3.
Future of the studies on genetic susceptibility to HAPE
With the advancement of genomic technologies, such as whole-genome sequencing, researchers are now able to explore the entire genetic landscape associated with HAPE susceptibility. By integrating genomic data with omics data, including transcriptomics, proteomics, and metabolomics, a more comprehensive understanding of the molecular mechanisms underlying HAPE can be achieved. Moreover, functional studies will help validate the significance of the identified genetic variants implicated in HAPE. These combined efforts have the potential to translate discoveries into improved prevention, diagnosis, and treatment strategies for diseases caused by high-altitude hypoxia or other hypoxia-related disorders, such as cardiovascular diseases and cancers.
Footnotes
Acknowledgment
The authors extend their sincere gratitude to all the patients with HAPE and the control participants for their invaluable contributions to the studies on the genetic susceptibility to HAPE. Without their cooperation, these achievements would not have been possible. The authors also gratefully acknowledge the assistance of the clinicians and hospital staff who contributed to their laboratory experiments and field studies.
Authors’ Contributions
The authors met the criteria for authorship as recommended by the International Committee of Medical Journal Editors in the Roles & Responsibilities guidelines. Y.D. and M.O. were responsible for conceiving this review article and interpreting the references included. N.K. and M.I. participated in discussions and provided constructive feedback on the final version of the article. T.K. and M.H. reviewed and substantially revised the review article for intellectual content and approved the final version for submission.
Author Disclosure Statement
The authors declare that this review was conducted without any financial interests. No personal interests to disclose.
Funding Information
No funding was received for this review.
