Abstract
Abstract
Wilson, Mark H., C.H.E. Imray, A.R. Hargens. The headache of high altitude and microgravity—similarities with clinical syndromes of cerebral venous hypertension. High Alt. Med. Biol. 12:379–386.—Syndromes thought to have cerebral venous hypertension as their core, such as idiopathic intracranial hypertension and jugular foramen outlet obstruction, classically result in headaches. Do they provide an insight into the cause of the headache that commonly occurs at altitude? The classic theory of the pathogenesis of high altitude headache has been that it results from increased intracranial pressure (ICP) secondary to hypoxemia in people who have less compliant intracranial volumes (Roach and Hackett, 2001). However, there does not appear to be a correlation between the headache of acute mountain sickness (AMS) and the presence of cerebral edema (Bailey et al, 2006; Wilson et al, 2009). Research has concentrated on arterial perfusion to the brain in hypoxia, but there has been little study of venous drainage. Hypoxia results in markedly increased cerebral blood flow; however, if it has been considered at all, venous outflow has to date been assumed to be of little consequence. Retinal venous distension and the increased venous blood demonstrated by near infra-red spectroscopy and more recently by MRI imply that, in hypoxia, a relative venous insufficiency may exist. Similarly, there is increasing evidence that manifestations of the fluid shift during microgravity is of similar nature to idiopathic intracranial hypertension, which is thought to be primarily a venous insufficiency condition. The unique anthropomorphic adaptations of large brained biped humans with cerebral venous systems that have to cope with large changes in hydrostatic pressure may predispose us to conditions of inflow/outflow mismatch. In addition, slight increases in central venous pressures (e.g., from hypoxia-induced pulmonary vasoconstriction) may further compromise venous outflow at altitude. A better understanding of cerebral venous physiology may enlighten us with regards the pathogenesis of headaches currently considered idiopathic. It may also enable us to trigger headaches for study and hence enable us to develop new treatment strategies.
Introduction
Background to a Venous Mechanism
At rest, the brain receives approximately 14% of the cardiac output, around 700 ml per minute (McArdle et al, 2006). The jugular veins therefore have to drain 700 ml per minute, a factor often overlooked. This is a sizeable volume, considering the average adult male intracranial volume (including brain and cerebrospinal fluid) is only twice this (1473 ml) (Abbott et al, 2000). Unlike the dual arterial system (carotid and vertebral systems), there is only one significant venous exit from the cranium, the internal jugular veins. Any compromise to this outflow (the small amount of pressure from a cervical collar or the increased intrathoracic pressures from positive pressure ventilation being common clinical examples) results in greater cerebral venous pressures and intracranial pressures. This phenomenon was recognized in 1935 (Bedford, 1935) and, while it is routinely seen in the setting of mechanical ventilation, the venous contribution to ICP is often overlooked.
Clinical relevance
In adults, a number of headache syndromes are related to venous obstruction, and forms of headaches in children and adolescents have been associated with venous anomalies (Fenzi and Rizzuto, 2008) or “tight” posterior fossae (Nesterovskii Iu et al, 2007). If hypoxia-induced headache can be used as a model for these syndromes, it may encourage further investigation into venous causes of otherwise idiopathic headache and offer new ways of treatment. Table 1 outlines some of the relevant clinical syndromes.
Cerebral Venous Comparative Anatomy
As bipeds with large brains, humans have unique anatomical differences to other mammals. Neurosurgeons are distinctly aware that the sagittal sinus has a negative pressure when patients are in the sitting position, as this can result in (potentially fatal) air embolism if opened. When supine, bleeding from the sinuses can be torrential. Most other mammals do not have such pressure changes with which to contend.
Three main venous drainage systems converge to drain into the internal jugular veins (Fig. 1):
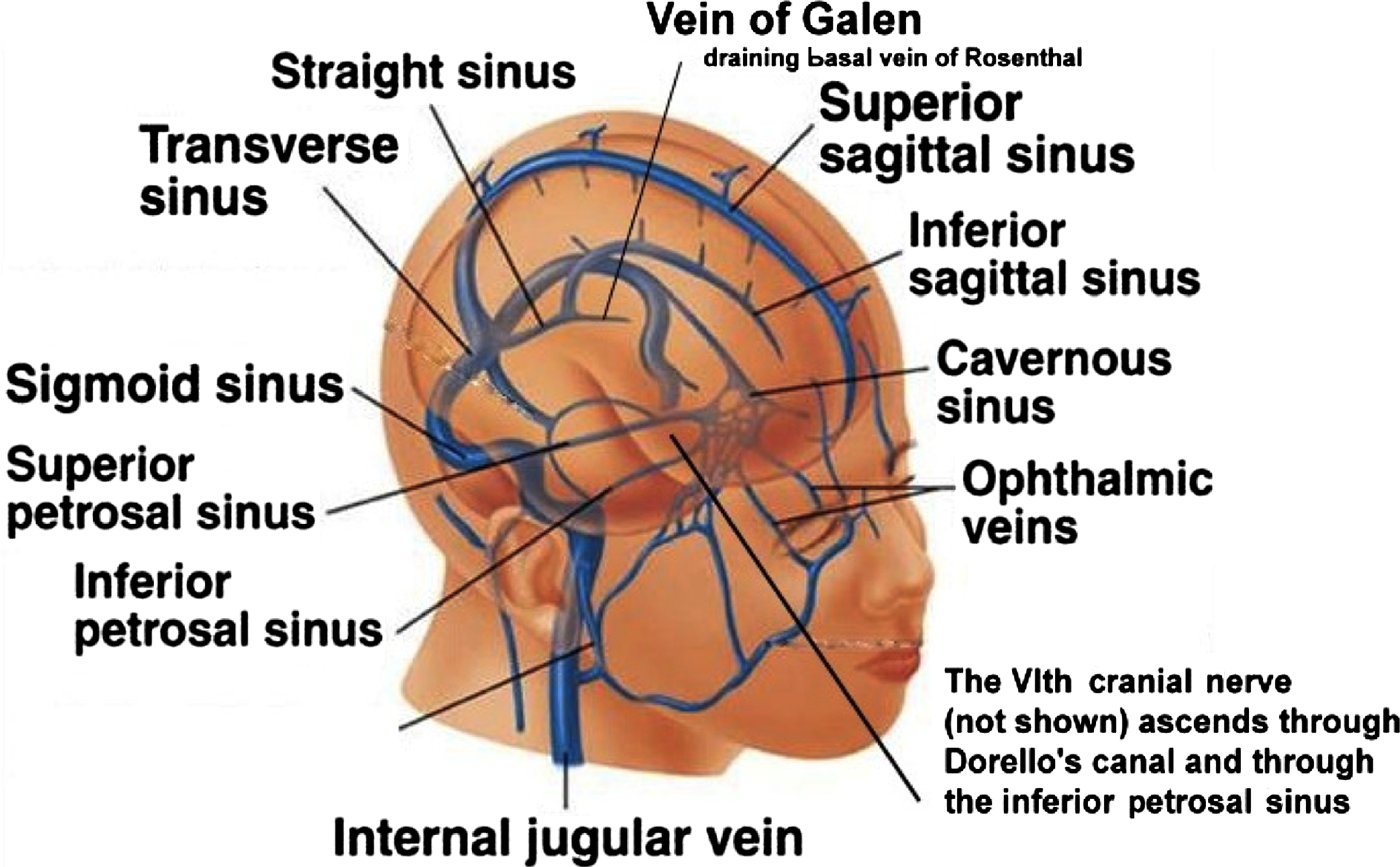
Schematic simplified diagram of cerebral venous drainage.
a. Cortical venous drainage occurs via bridging veins that cover the brain surface. These veins drain into the superior sagittal sinus which flows posteriorly to the torcula (confluence of sinuses) and then to the transverse sinus (in most people, to the right transverse sinus).
b. Deeper (anterior) venous drainage occurs into the cavernous sinuses anteriorly which in turn drain via superior and inferior petrosal sinuses into the jugular bulbs.
c. Central (thalamic) areas drain via a series of small veins into the internal cerebral veins superiorly and the basal veins of Rosenthal inferiorly. These unite (behind the splenium of the corpus callosum) to form the Great Vein of Galen which then drains via the straight sinus to join the torcula and then to the transverse sinus (in most people, to the left transverse sinus).
The final common venous outlets for all the above tributaries are the two internal jugular veins. Increases in right atrial or central venous pressures are therefore transmitted directly back to the brain. Very minor additional venous drainage is provided by orbital veins and vertebral venous plexi. Interestingly, as humans, we have largely lost the enlarged occipital-marginal sinus that provided significant drainage to the cerebral venous plexus which our australopithecine ancestors of 3 million years ago had (Falk, 1986). The vertebral venous plexi are still the main outflow for most supine mammals such as swine (Lavoie et al, 2008).
Unlike normal vessels, the dural venous sinuses are composed of dura mater lined with endothelium. They lack normal vessel layers (e.g., tunica media) and hence are more susceptible to external compression, and as a consequence they are therefore also more prone to distension. Certain sinuses (e.g., the cavernous sinus) have sympathetic innervation (van Overbeeke et al, 1995) and other venous structures have dense trigeminal innervation (Penfield and McNaughton, 1940; Strassman and Levy, 2006) that may relay distension as headache before any rise in ICP.
A Venous Hypertension Model of Hypoxia-Induced Headache
The matching of inflow and outflow volumes of blood within the cranium must be complex as so many factors (arterial blood pressure, vessel caliber, and central venous pressure) have an influence. A simplified venous hypertension model is outlined in Figure 2. An “unplugged running bath” with the taps representing arterial blood delivery, the plughole representing venous outflow, and the water level representing intracranial blood volume serves as a good, though simple analogy. In a steady state, flow in equals flow out. In clinical syndromes of outflow obstruction, flow in still equals flow out, but the pressure to drive the outflow is greater to overcome additional resistance.
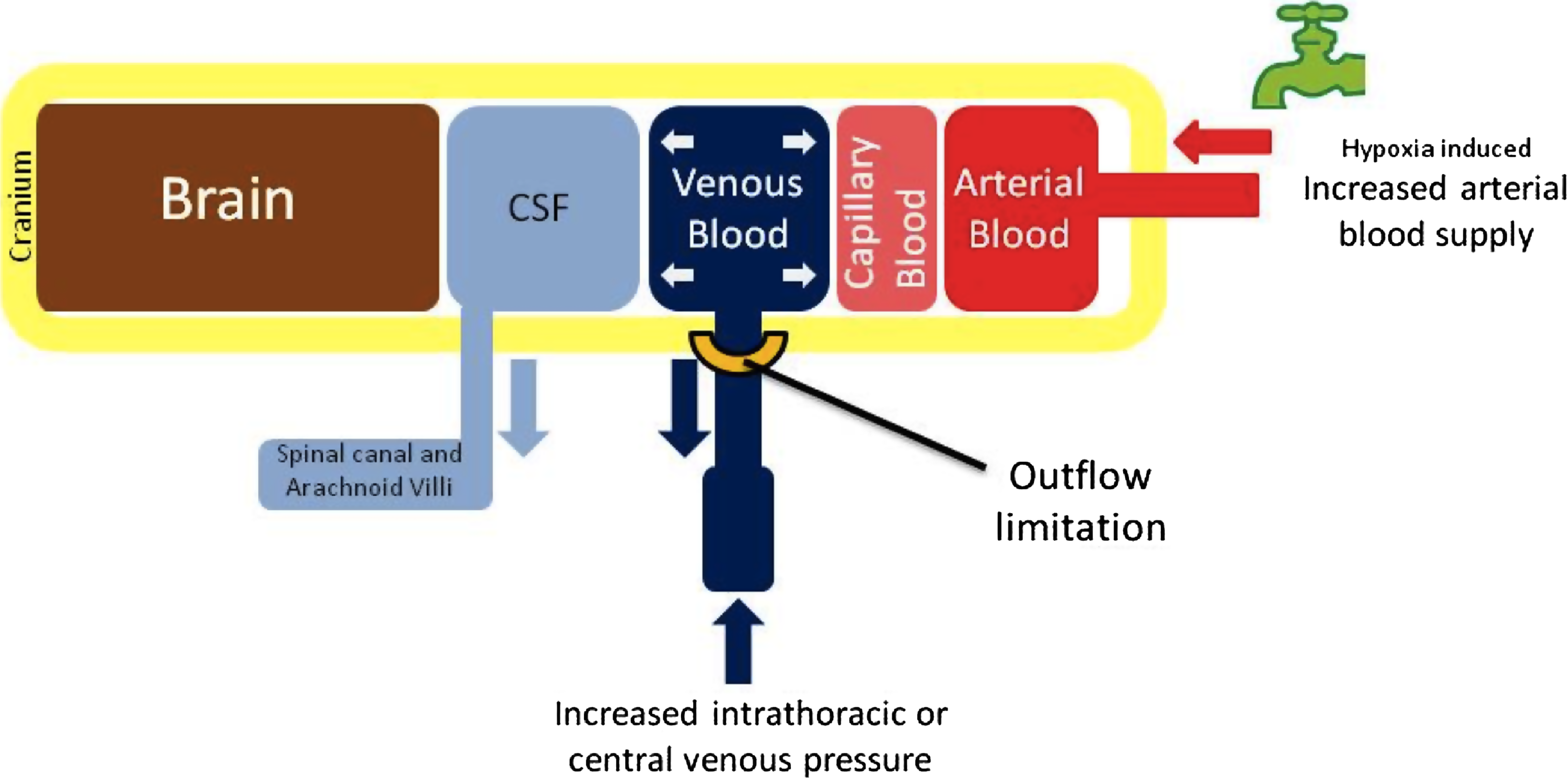
Diagram of venous hypertension mechanism. CSF=cerebrospinal fluid.
In hypoxia a number of mechanisms may contribute to increased intracranial blood (and in particular venous) volume, but all center on the limiting step of venous drainage. Hypoxia is a relatively unique cause of increased cerebral blood flow in the non-injured brain since autoregulation normally prevents increased flow (e.g., secondary to normoxic exercise-induced hypertension).
In hypoxia, arterial blood flow increases to maintain oxygen delivery (Brugniaux et al, 2007). To increase venous drainage to match this increased flow, a greater venous pressure is required (i.e., the water level rises to increase pressure driving blood out the plug hole and keep a steady state of inflow=outflow).
Any phenomenon that reduces venous outflow will increase intracerebral venous pressures. Two principal factors restrict flow: vessel diameter and distal pressure.
High Altitude Research Suggesting Venous Congestion
At altitude, there is marked dilatation of retinal veins (Fig. 3) and this dilatation correlates with the severity of headache (Bosch et al, 2009). This occurs without the high ICP indicator of papilledema. The central retinal veins drain to the cavernous sinuses, and hence this dilatation may well reflect gross intracerebral venous changes. Retinal hemorrhages are common, and in survivors of severe altitude sickness, intracerebral microhemorrhages have been observed (Kallenberg et al, 2008). Such microhemorrhages are pathognomonic of venous obstruction (Tsai et al, 1995). Similarly, the retinal hemorrhages of shaken baby syndrome are now thought to result from hypoxia, not direct trauma (Geddes et al, 2003).
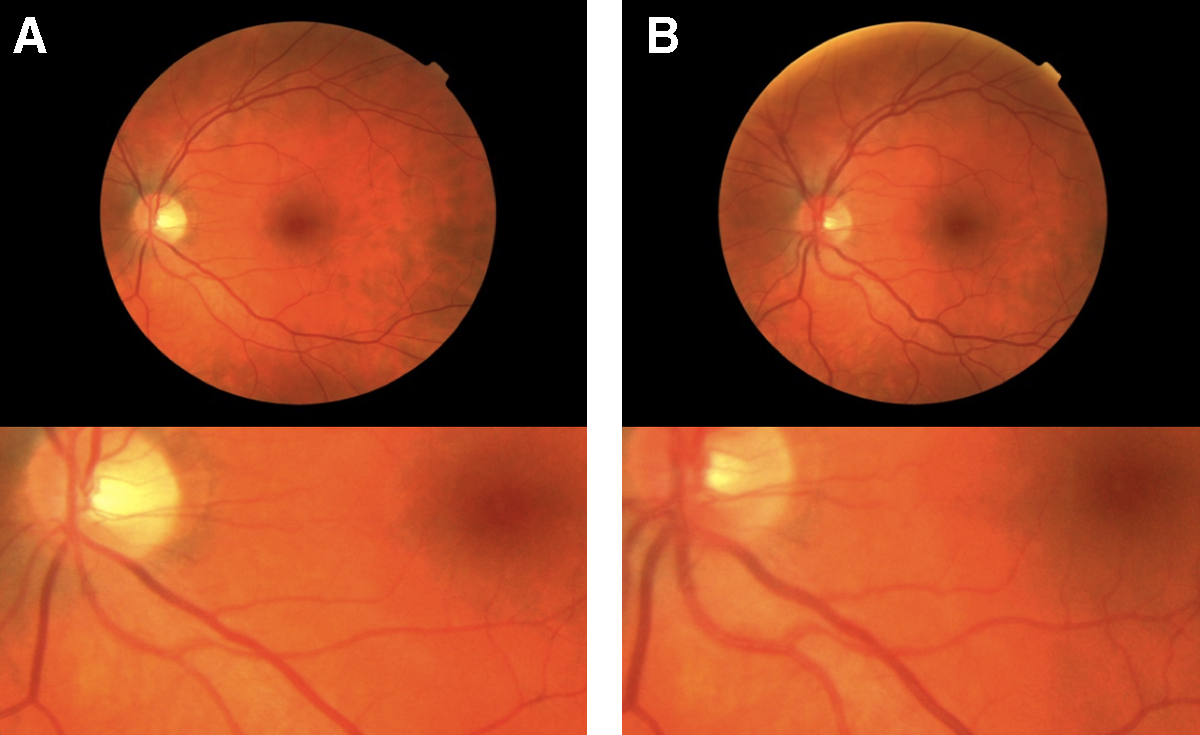
Retinal vasculature
Recent microcirculatory studies of sublingual circulation have, in a similar manner to retinal imaging, suggested that venous congestion occurs with hypobaric hypoxia (Martin et al, 2009). Stagnation is seen (despite the presumed rise in cardiac output) and, although technically difficult to confirm, similar stagnation in brain microcirculation probably occurs.
Near infra-red spectroscopy (NIRS) reflects regional brain oxygen saturation (rS
Dubowitz et al (2009) have recently demonstrated that acute hypoxia causes a significant reduction in cerebrospinal fluid (CSF) volume with small increases in estimated cerebral blood volume (+2.3 ml (SD 2.5) and estimated brain parenchymal volume (+8.2 ml (SD 6.4), respectively). Since the MRI techniques used do not distinguish venous and parenchymal structures, this increase could also reflect venous engorgement.
Clinical Syndromes Resulting from Restricted Cerebral Venous Outflow
Idiopathic intracranial hypertension (IIH) and transverse sinus stenosis
Idiopathic intracranial hypertension (also known as benign intracranial hypertension or pseudotumour cerebri) is a condition characterized by headache, nausea, and vomiting (Binder et al, 2004). Bilateral transverse sinus stenosis is found in 90% of sufferers (Bono et al, 2005; Higgins et al, 2005; Karahalios et al,1996; Pickard et al, 2008), and has been successfully treated with endoluminal stenting (Donnet et al, 2008b; Higgins et al, 2002; Higgins et al, 2003; Owler et al, 2005). Other studies have demonstrated internal jugular stenosis in 80% (Alperin et al, 2005). Nine percent of patients with chronic tension-type headache have bilateral transverse sinus stenosis (BTSS) and idiopathic intracranial hypertension without papilloedema (Bono et al, 2008), as have 6.7% of migraine sufferers (Bono et al, 2006) (the same researchers found that none of the 45 headache-free controls had BTSS). Both IIH and high altitude headache are commonly and successfully treated with acetazolomide (Bono et al, 2008). Exertional headache has been found to occur secondarily to bilateral transverse sinus stenosis (resolving with transverse sinus stenting)(Donnet et al, 2008a). Patients who suffer with headaches with bilateral transverse sinus stenosis have higher CSF opening pressures (Bono et al, 2009). Interestingly, this sinus stenosis is not due to transverse sinus compression from raised ICP as it persists even when ICP has been lowered (Bono et al, 2005).
Jugular syndromes, superior vena cava obstructions, and Chiari malformations
Jugular syndromes blocking outflow (Graus and Slatkin, 1983), superior vena cava obstruction (Al-Hilali et al, 2003), and Chiari 1 malformations (Cinalli et al, 2005) (which commonly present in early adulthood, the same age group that suffer more with AMS) all increase cerebral venous pressures, cause headaches, and ultimately raise ICP.
Obstructive sleep apnea
Obstructive sleep apnea (OSA) is commonly associated with morning headaches (Alberti et al, 2005) and Cheyne-Stoke breathing. Both of these are common at altitude. At altitude, hypobaric hypoxia can cause central sleep apnea which in turn can exacerbate OSA (Burgess et al, 2006). The larger intrathoracic pressures and neck sizes associated with obesity and OSA are also thought to increase venous pressures.
Possible Sites of Venous Obstruction in High Altitude Headache
Venous obstruction could occur at a cranial or thoracic level.
Cranial obstruction
Although typically the superior sagittal sinus drains to the right and the inferior sagittal sinus to the left, there are marked differences between individuals, with many having a dominant transverse sinus (the right in 65%) (Durgun et al, 1993). Limitations in outflow caliber (transverse/sigmoid sinus and jugular foramen) that under normal physiological conditions are adequate might be unmasked with the increased drainage required to remove the increased blood flow of hypoxia. Cerebellar engorgement (by increasing small venous vessel distension) could further compress the thin-walled transverse sinus, restricting outflow further and compounding the problem. It is interesting to note that palsies of the abducens nerve (which traverses the petro-clival venous confluence within Dorello's canal) are the palsies more often associated with cavernous sinus pathology than raised ICP (Keane, 2005). They are also the commonest cranial nerve palsy both in IIH (Krishna et al, 1998) and AMS (Basnyat et al, 2004). IIH and altitude illness are similarly associated with increased frequency of sleep apnea and sinus thrombosis (Mokri et al, 1993).
Increased central venous pressures
Hypoxic pulmonary vasoconstriction (HPV) is a normal response to divert blood from areas of poorly oxygenated lung, to areas with better oxygenation. Hypoxia due to reduced inspired partial pressures of oxygen can result in diffuse pulmonary vasoconstriction, the degree of which varies between individuals. High altitude pulmonary edema (HAPE) and high altitude cerebral edema (HACE) are closely intertwined, and treating HAPE usually improves HACE. Increased pulmonary artery pressures might well increase central pressures which, even if only slightly raised, will increase cerebral venous pressures. Acetazolomide prevents hypoxia induced pulmonary vasoconstriction (Hohne et al. 2004). Interestingly, in patients with heart failure, acetazolomide improves central sleep apnea (Javaheri, 2006). Cerebral NIRS correlates better than hemodynamic parameters to left ventricular dysfunction (Paquet et al, 2008) and hence this supports a cardiopulmonary failing mechanism. Elevated intrabdominal pressures (as in obesity, which is very common in both IIH and OSA) could also increase venous pressures. Like headaches associated with intracerebral space occupying lesions, the headache of AMS is often worse at night or on waking. The increased central venous pressures and reduced cerebral drainage resulting from lying down may account for this.
Estimates of internal jugular vein valve incompetence in normal subjects range from 20% (Doepp et al, 2008b) to 33% (Schreiber et al, 2005). In subjects with COPD or pulmonary hypertension (akin to pulmonary hypertension secondary to hypoxic pulmonary vasoconstriction) this increases to 60% and 100%, respectively (Doepp et al, 2008a).
Other factors could compound this venous mechanism. The increase in hematocrit with acclimatization could increase flow resistance (viscosity) and hence venous pressures. The increased incidence of cerebral sinus thrombosis may also be related (Torgovicky et al, 2005).
Hypoxia itself could have a direct (possibly nitric oxide-mediated) venous effect. Nitroglycerin is a prodrug for NO (Olesen, 2008) and is well known for causing a classic headache after its use in normoxia. It also exacerbates AMS (Mazzuero et al, 2008), hence it could be hypothesized that hypoxia-induced NO formation has a greater effect on cerebral venous vessels.
Cerebral edema
One of the main arguments for cerebral edema being a component of the pathogenesis of altitude headache is that dexamethasone helps prevent it. However, dexamethasone has many effects, including cardiac effects that could account for the benefit it brings (Xia et al, 2007). The recent study demonstrating its use in preventing and treating HAPE (albeit via mechanisms not well understood at this point) might speak of its potential for reducing central pressures and subsequent cerebral venous pressures, as well (Maggiorini et al, 2006). The headache of hypoxia also occurs rapidly after exposure, which suggests that edema is not, at least in the initial stages, the principal cause of the headache. Prolonged venous hypertension however, could be a contributor to cerebral edema later in the condition.
The term “high altitude cerebral edema” is clinically defined but has a pathological component to its nomenclature that may not be correct (the patient symptoms may relate to hypoxia rather than edema); hence we need to use this term with caution.
Venous Obstruction and an Exponential Rise in ICP with Traumatic Brain Injury
Further Investigation of the cerebral venous system and its involvement in a number of clinical conditions may be aided by using hypoxia. In addition to the conditions of IIH, OSA, posterior fossa syndromes, migraine, and possibly other forms of headache, a number of other conditions such as traumatic brain injury (TBI) (Matsushige et al, 2004) may be associated with venous obstruction. In TBI, venous congestion causes reversible changes on imaging and may well contribute more to a rise in ICP than the classic parenchymal swelling to which raised ICP is normally attributed (Andeweg, 1999). Once cisterns are effaced, the venous obstruction compounds the system and may account for the exponential increase in ICP at the limits of brain compliance more than a simple hydraulic effect.
A Brief Report of an MRI Study of Venous Changes with Hypoxia
We have recently reported a study using 3 Tesla MRI to demonstrate middle cerebral dilatation in hypoxia (Wilson et al, 2011). Full methodological and objective data can be found in this article. We performed a pilot study on all 7 subjects (5 males; mean age: 34.4 years, range: 22–48 years) utilizing susceptibility-weighted images to demonstrate the venous structures. Studies were performed in normoxia and after 3 hours of hypoxia (Fi
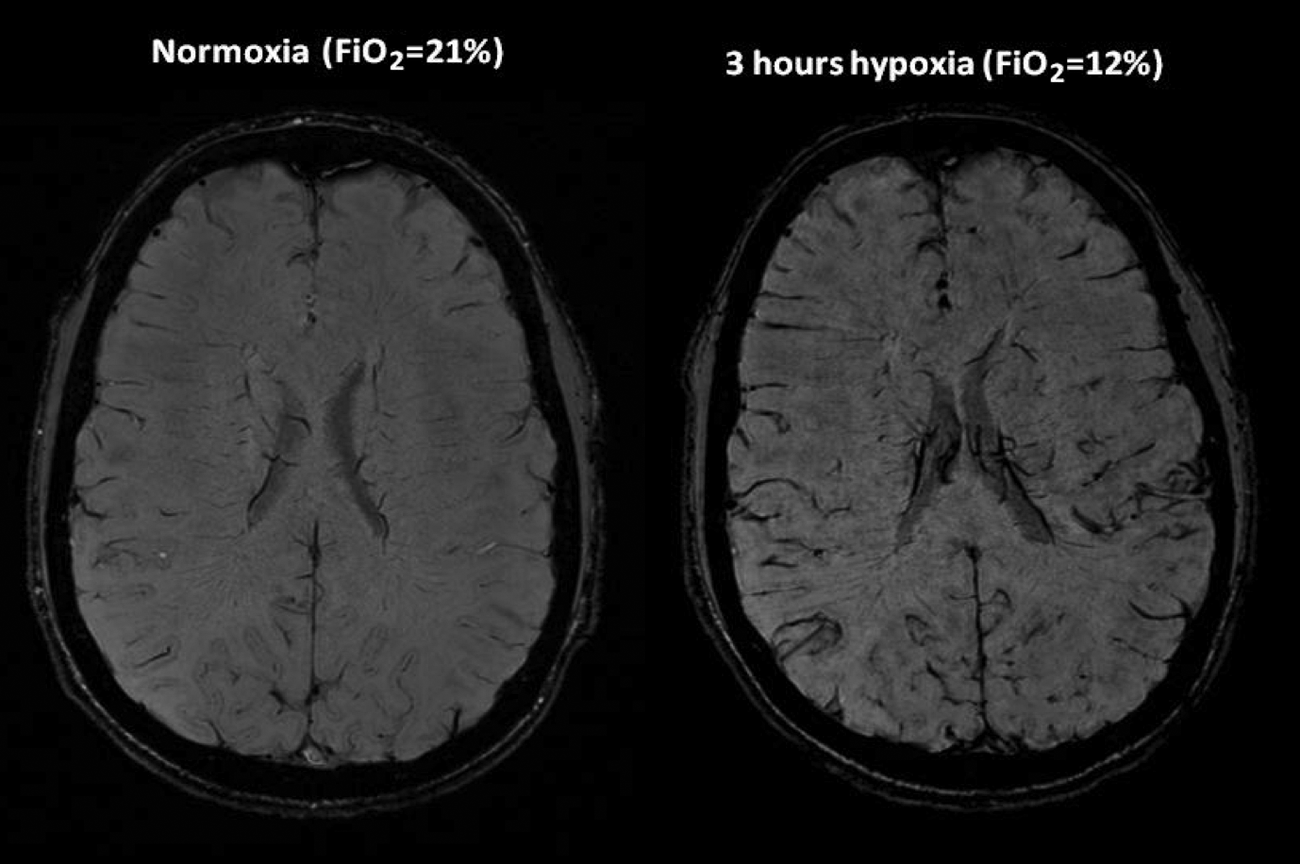
Susceptibility weighted axial 3 Tesla MRI images in normoxia and after 3 hours of hypoxia (Fi
Further Proposed Mechanisms of Study
The cerebral venous system can be investigated further in a number of ways. Invasive cerebral venous pressure monitoring, venous imaging (cerebral venography), and further retinal studies during hypoxia are required. An additional study assessing central venous pressures (CVP, invasively or inferred from echocardiography) may help elucidate if hypoxia induced right heart failure is the underlying mechanism. A further study should look for BTSS in those who suffer AMS using MRV, and another study could establish if hypoxia exacerbates IIH. Studies could also be performed assessing the competence of the internal jugular vein valve (looking for retrograde flow during valsalva) in subjects who are altitude headache prone and resistant.
When investigating migraine, it should be noted that simple T1 and T2 sequences do not demonstrate venous anatomy well and hence specific (e.g., susceptibility weighted studies) should be performed when investigating idiopathic headaches.
Space Adaptation Syndrome
Space motion sickness occurs in 67% of shuttle astronauts, and theories of its pathogenesis range from vestibular disturbance through to cranial fluid shifts occurring with the loss of gravity (Hargens, 1983; Iwase and Mano, 2000; Watenpaugh and Hargens, 1996). Microgravity-induced cranial fluid shifts would in itself be expected to cause cranial venous congestion. There have been a number of reports recently of astronauts suffering headache and loss of peripheral vision. Again, this is very similar to the symptomatology of IIH, the venous pathogenesis of which is described above. Stolz et al (2009) have shown that large changes in venous outflow velocities occur with orthostatic position and lower limb pressure. There is also some evidence that, in simulated microgravity, subjects can be divided into a group that has a net cerebral inflow and a group that has a net outflow. Those with net inflow get headaches, implying a relative venous insufficiency (Hu et al, 1999). Increased venous pressure across the capillary bed would shift the Starling-Landis Equilibrium to greater filtration into tissues (Norsk, 2005), which may well be the cause of both visible facial edema (commonly seen in astronauts) and assumed cerebral edema.
Conclusions
There is increasing interest in the role of the venous system in neurological disorders from headache to multiple sclerosis (Zamboni et al, 2010). There is little evidence that hypoxic headache is due to edema. A venous component to the pathological mechanism is implied when considering similar clinical syndromes. This may relate to a relative inability to drain the cerebral vasculature adequately in physiological extremis. Perhaps experimental treatments could be considered to improve venous drainage rather than reduce cerebral edema. Further studies into the cerebral venous system may suggest mechanisms both for clinical and altitude-related headaches, and hypoxia may be a repeatable trigger that can act as a model for further studies. Finally, investigations for venous pathology should be considered in those with otherwise idiopathic headaches.
Footnotes
Acknowledgments
The authors thank The Caudwell Xtreme Everest Group, Centre for Altitude, Space and Xtreme Environment Medicine: Professor Hugh Montgomery, Dr. Dan Martin, Dr. Mike Grocott, and Dr. Jeremy Windsor; The National Hospital for Neurology and Neurosurgery, London: Mr. Laurence Watkins, Dr. Stefan Brew, Dr. Fergus Robertson, Dr. Ian Calder, Dr. Sally Wilson, Mr. Neil Kitchen, Dr. Indran Davagnanam, and Ms. Huma Sethi. The concept for this article and the original manuscript was by Mark Wilson.
Author Disclosure Statement
No competing financial interests exist.
Christopher Imray and Alan Hargens are joint senior authors.
