Abstract
Background:
Pelvic exenteration can extend survival in patients with pelvic recurrence following radiotherapy for gynecological cancer, but there is a high risk of recurrence.
Objectives:
To describe patterns of relapse following pelvic exenteration for central recurrence and determine whether postoperative stereotactic radiotherapy is feasible to improve loco-regional control.
Methodology:
Outcomes were reviewed for all patients who underwent pelvic exenteration for central pelvic recurrence of gynecological cancer in a single center. The sites of subsequent post exenteration recurrences were mapped on a reference computed tomography scan, and the relationship to preoperative disease was recorded. A dosimetric study was then conducted to evaluate feasible target volumes for postoperative stereotactic radiotherapy.
Results:
Of 101 evaluated patients, 61 (60.4%) relapsed following pelvic exenteration. The site of recurrence was pelvic only in 42 patients (68.9%) of recurrences, distant only in 11 patients (18.0%), and both in 8 patients (13.1%). Five-year locoregional control was 66.7% with a clear resection margin compared with 35.0% with an involved or close margin (p = 0.03), and median progression-free and overall survival were 12.6 versus 7.4 months (p = 0.12) and 20.3 versus 19.7 months (p = 0.91), respectively. With a stereotactic radiotherapy planning study, it was feasible to deliver 30 Gray in 5 fractions while meeting normal tissue re-irradiation dose constraints. Target volumes included the site of the close margin and the region encompassing the lateral margin of all preoperative disease.
Conclusion:
Locoregional relapse close to the initial disease site is the dominant pattern of recurrence following pelvic exenteration for recurrent gynecological cancer. Stereotactic radiotherapy following pelvic exenteration is a feasible option to encompass the high-risk site and should be used to potentially improve outcomes.
Introduction
Despite recent improvements in the management of gynecological cancer, locoregional relapse after surgery and radiotherapy remains a major therapeutic challenge. Re-irradiation with conventional radiotherapy techniques causes significant morbidity, whereas chemotherapy has limited efficacy in a previously irradiated field. 1 Pelvic exenteration is the established potentially curative option for pelvic relapse in women previously treated with radiotherapy (with or without prior surgery), but this is associated with significant perioperative morbidity and mortality and requires careful case selection.2–6 Retrospective studies have identified several factors that affect outcomes for women undergoing pelvic exenteration. These include the site of recurrence (central versus pelvic sidewall), size of relapse, disease-free interval, lymph node involvement, lymphovascular space invasion, and surgical resection margins4,7–10 classified as R0 (histologically negative), R1 (macroscopically clear but histologically positive) and R2 (gross residual disease). Despite using modern imaging techniques, laparoscopy to exclude peritoneal disease and intraoperative histopathological margin evaluation, exenterative procedures with curative intent still have histologically involved margins in 7–35% of cases. Where R0 has not been achieved, survival can be reduced to as low as 0% at five years.3,4,11,12
There is currently a lack of data on postoperative therapeutic approaches to improve clinical outcomes for those women at the highest risk of locoregional relapse. There are no studies using postoperative external beam radiotherapy for positive or close margins. One approach to improve local control has been the addition of intraoperative radiotherapy, which has been reported to potentially improve outcomes following complete macroscopic resection. However, it has been associated with a higher risk of fistulae formation and significant gastrointestinal and sciatic nerve toxicity.13–15 Limitations to this technique include access to the operative site and the delivery of treatment without knowing the final histopathology results.
Postoperatively, stereotactic radiotherapy offers a treatment option following histopathological assessment of surgical margins. Stereotactic radiotherapy delivers highly conformal treatment, minimizing dose to adjacent normal structures, which therefore provides the option of re-irradiation.16–18 However, a major challenge is defining the target volume since the tumor removal and surgical excision of pelvic organs alters soft-tissue anatomy. Re-irradiation increases the risk of severe late toxicity, so it is essential to minimize dose to normal tissues. Therefore, there is a need to develop a reliable method for defining the target volume and normal tissue dose constraints following pelvic exenteration.
The objectives of this study were to:
Describe the patterns of relapse following exenterative surgery for recurrent gynecological cancer. Determine the feasibility of delivering stereotactic radiotherapy for close or positive surgical margins.
Methods
A retrospective analysis was carried out of patients who underwent exenterative surgery for central pelvic recurrence of gynecological cancer at the Royal Marsden Hospital between 1993 and 2018. Patients were identified using a prospectively recorded surgical database, while electronic patient records were used for demographic data and clinical outcomes. Only patients with central recurrent disease and patients with persistent disease were included in the study. Patients with pelvic side wall recurrence and patients who underwent pelvic exenteration for primary disease were excluded from the study. The local Ethics Committee at The Royal Marsden Hospital approved the study.
Pathological margins
For this study, a clear margin was defined as at least 5 mm histopathological clearance, a close margin was 0.1 mm to 5 mm, and an involved margin was a tumor extending to the resection margin (0 to <0.1 mm).
Patterns of recurrence and relationship to preoperative disease
Relapse following exenteration was classified as locoregional, distant, or both. In order to study the patterns of loco-regional relapse in more detail, the site of disease for each patient was reconstructed on a reference computerised tomography (CT) scan, creating a cumulative anatomical distribution map. Furthermore, the diagnostic imaging from 2006 onward (when the hospital picture archiving and communication system was introduced) was exported to the radiotherapy treatment planning system. Rigid registration with bone matching was performed using CT or magnetic resonance imaging (MRI) imaging prior to exenteration and the scans demonstrating subsequent pelvic relapse. Three-dimensional tumor volumes were contoured on each imaging dataset, and a 5 mm isometric expansion was added to the preoperative tumor volume to account for contouring uncertainties and subclinical microscopic disease. The volume of overlap of these two volumes was measured to assess the relationship between pre- and post-exenterative margins (Fig. 1).
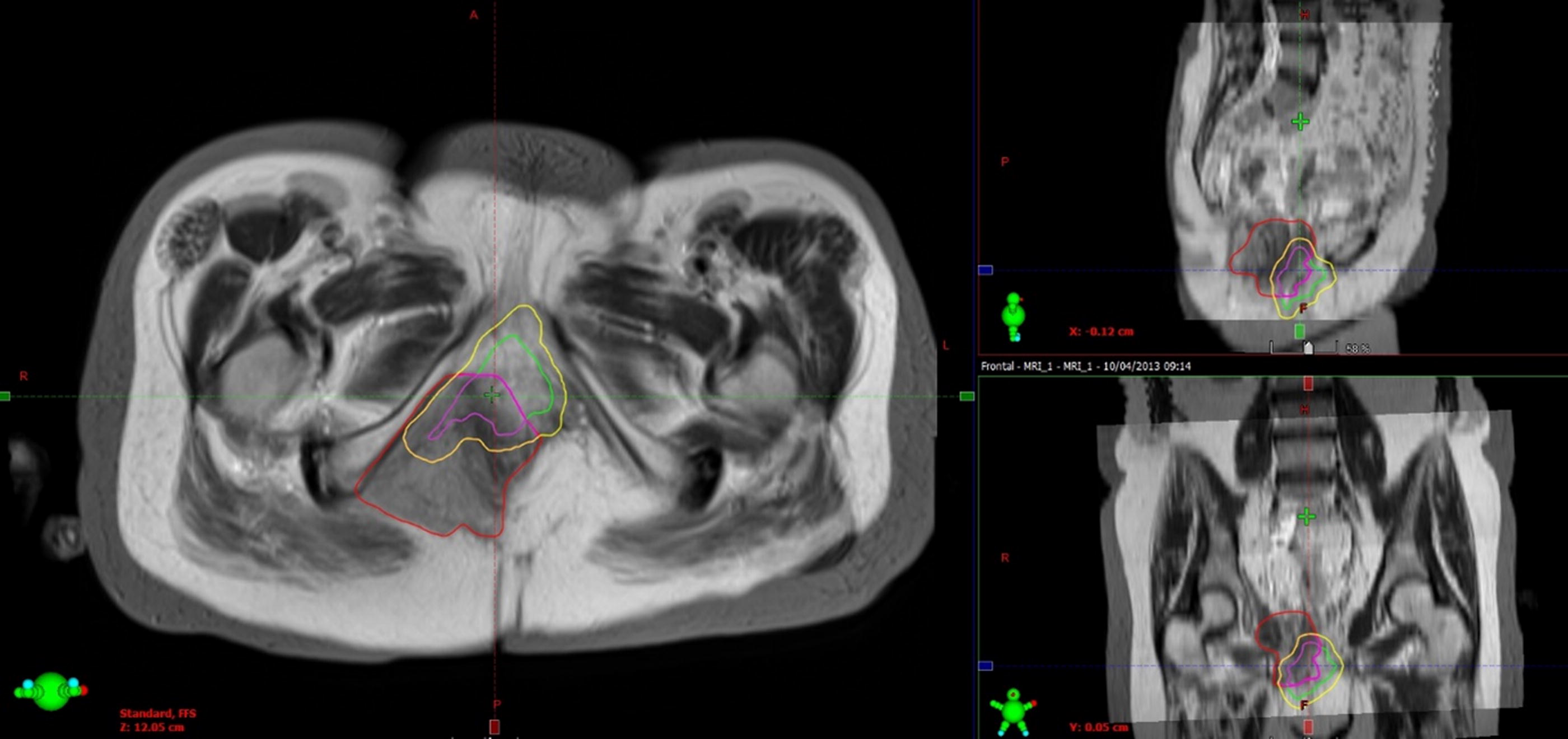
Preoperative tumor volume (green), postoperative relapsed tumor volume (red), 5 mm margins (yellow), overlap (purple).
Stereotactic radiotherapy planning study
A dosimetric planning study was conducted to assess the feasibility of re-irradiation with stereotactic radiotherapy. Postoperative CT scans were used from 10 patients who had a positive or close resection margin. Preoperative MRI scans were co-registered to enable the site of pre-exenteration disease to be localized.
Clinical target volumes
Target volume definition following exenteration can be challenging because of the changes in anatomy. In order to evaluate which clinical target volume could feasibly be treated while keeping within normal tissue dose constraints three radiotherapy clinical target volumes were contoured on each CT scan Target volume definition was aided by co-registered preoperative and postoperative MRI scans, histopathology results, and operation notes.
Clinical target volume (CTV)_margin was the site of likely positive or close margin.
CTV_highrisk included the lateral margin of all preoperative disease and the high-risk region bordering the positive/close margin, e.g., the entire muscle edge bordering the tumor.
CTV_total combined CTV_highrisk with the site of all preoperative disease.
Planning target volumes were created using a 5 mm isotropic expansion of each clinical target volume (Fig. 2).
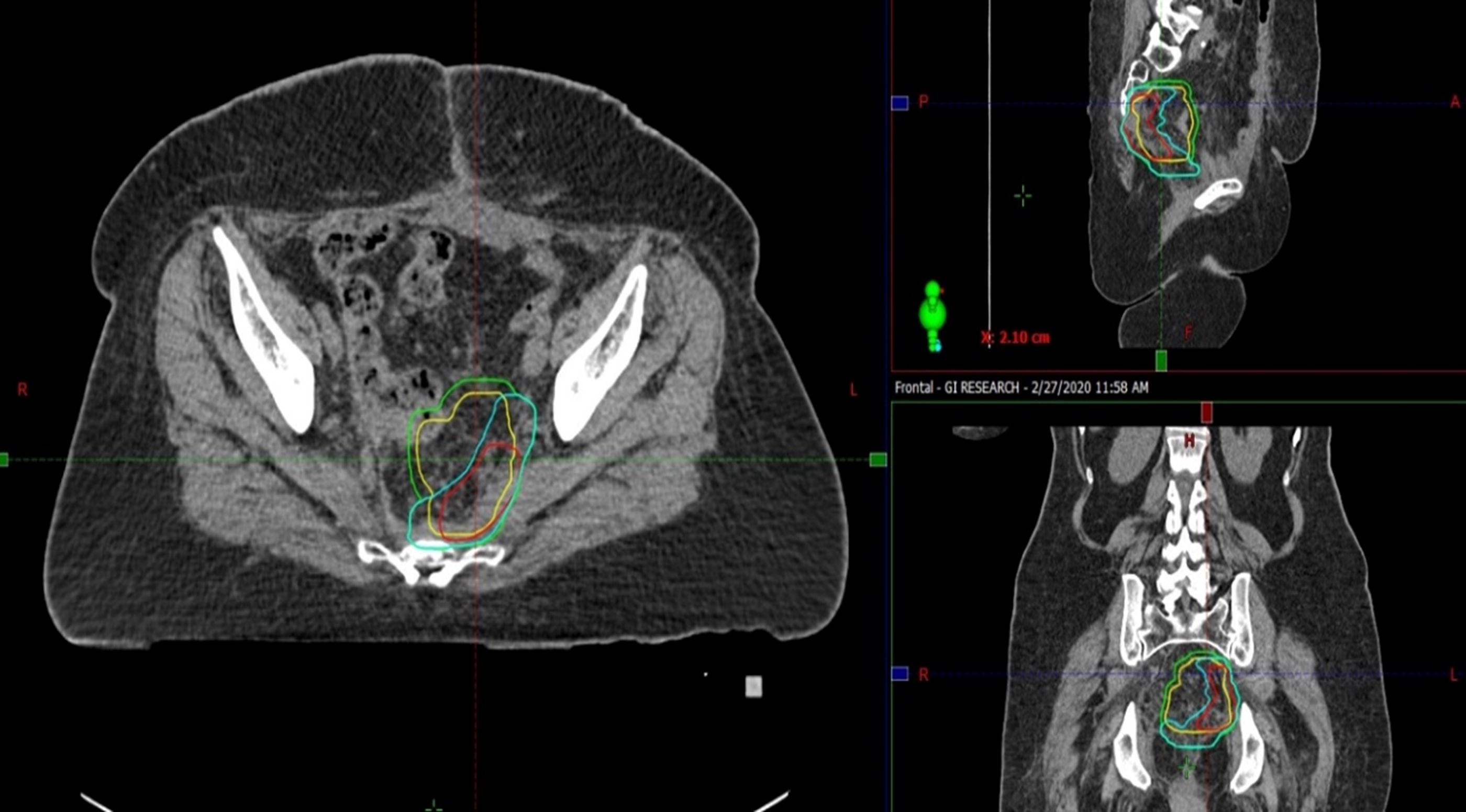
Planning target volumes demonstrating clinical tumor volume margin (red), clinical tumor volume high-risk (blue), clinical tumor volume total (green), and preoperative gross tumor volume (yellow).
Organ at-risk contouring
Organs at risk were contoured following the Radiation Therapy Oncology Group contouring guidelines, including bilateral femoral heads, bowel loops, bladder (following posterior pelvic exenteration), and rectum (following anterior pelvic exenteration). 19 The sciatic nerve was contoured according to Yi et al. sacral plexus contouring guidelines, and a 5 mm isotropic expansion was used to create the nerve planning risk volume. 20
Stereotactic radiotherapy planning
All stereotactic radiotherapy plans were generated using the Eclipse radiotherapy planning system to deliver 30 Gy in five fractions. Plans were normalized to achieve 95% target volume coverage by the prescription isodose and a maximum dose of 125%–140% prescribed dose while maximally sparing normal tissues. For the purposes of this study, planning target volume coverage was not compromised to meet any normal tissue tolerances.
There are limited data on the cumulative dose constraints for re-irradiation in the pelvis.21,22 A previous pelvic radiotherapy dose of 50 Gy Equivalent dose in 2Gy fractions (EQD2) was assumed with at least six months of recovery to calculate cumulative organ at-risk doses for this study. The optimal and mandatory dose constraints used in this study for five fraction plans are shown in Supplementary Table S1. These values were derived based on a literature review, the cumulative dose limits reported by Abusaris et al., and the clinical outcome data from re-irradiation with brachytherapy.23–25
In addition, constraints were also calculated by allowing 50% recovery from the previous treatment to the internationally established dose constraints for primary treatment of cervical cancer. 26 Both methods for calculating tolerances had very similar results, but when there was a difference, the lower value was used for this study.
Statistical analysis
Descriptive statistics were used for all study variables. Survival analysis was performed using the Kaplan–Meier method. Cox proportional hazards regression was used to model the effects of clinical variables on overall and disease-free survival. For the planning study, comparative analysis was carried out between each target volume group to compare target volume median dose, dose to organs at risk, and number of plans that met all dose constraints without compromising target volume coverage.
Results
A total of 101 patients were included in this study, with a median follow-up of 49.4 months (2.1–280 months). Median age at the time of pelvic exenteration was 56 years (range 24–83 years). The median time interval between the end of treatment of primary or previous recurrence and pelvic exenteration was 8.1 months (0.6–283.6 months). The disease-free interval was less than two years in 64.6%. The tumor characteristics are summarized in Table 1.
Summary of Tumor Characteristics and Treatment
Patient outcomes
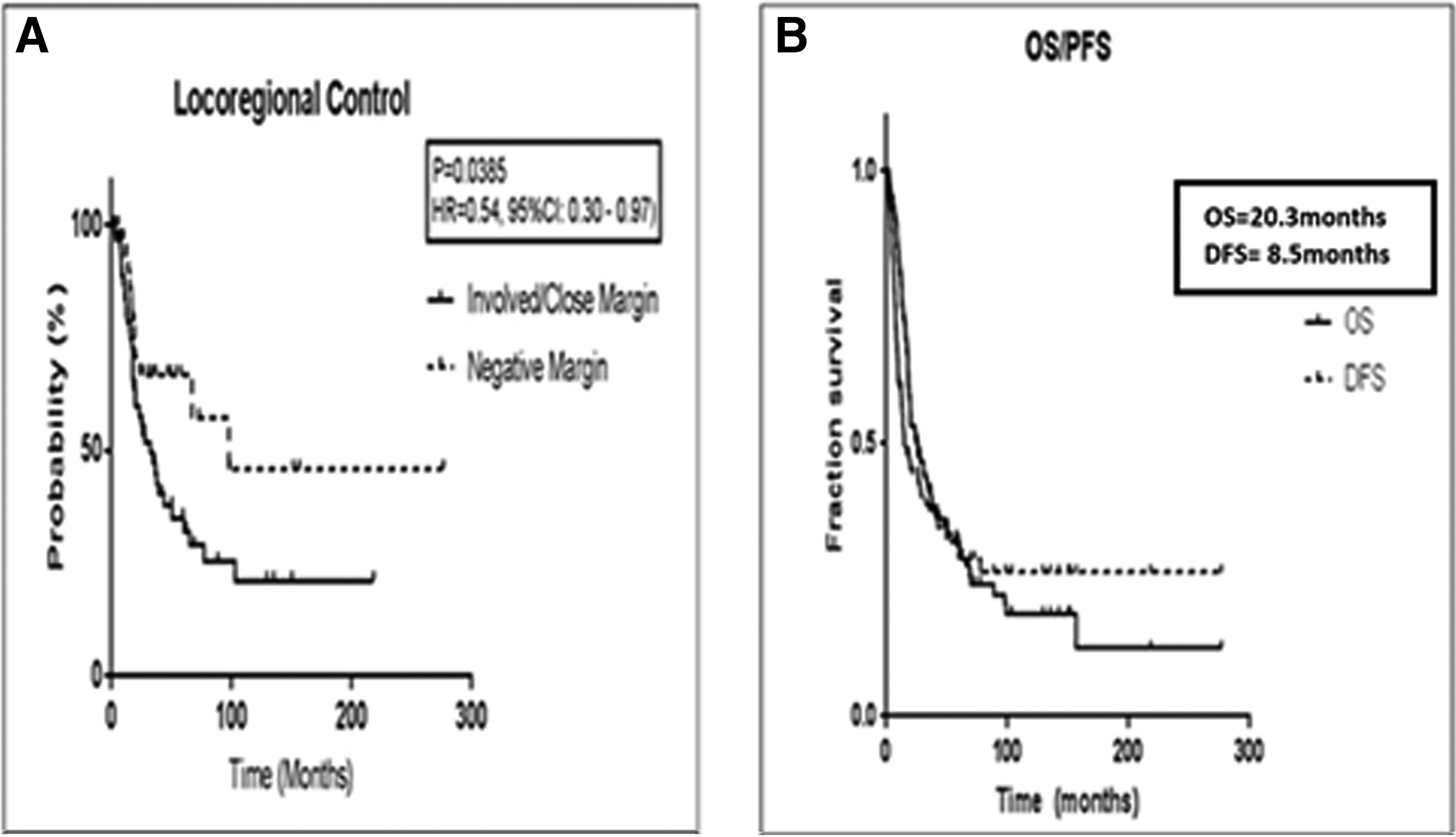
Median progression-free survival was 8.5 months, and median overall survival was 20.3, with 48.5% and 24.8% of patients alive at two and five years, respectively. There was a higher recurrence rate of 71.4% when lymphovascular space invasion was present compared with 44.4% when absent. Furthermore, 64.7% of patients with perineural invasion relapsed compared with 45.8% without perineural invasion (Table 2). The median progression-free and overall survival for involved/close versus negative margins were 7.4 versus 12.6 months and 19.7 months versus 20.3 months, respectively. Five-year locoregional control was 66.7% for the negative margin group compared with 35.0% for the involved/close margin group (p = 0.04; HR = 0.54, 95% CI: 0.30–0.97) (Fig. 3).
Relapse in Relation to Histological Characteristics and Margin Status
Cox proportional hazard regression did not show a significant association between the primary site of diseases or histological subtype and the rate or pattern of recurrence.
Site of locoregional relapse
Sixty-one patients (60.4%) had a recurrence, with 42 occurring loco-regionally (68.9% recurrences), 11 distant (18.0%), and eight patients had both (13.1%).
Out of the seven patients who received chemotherapy post-exenteration, five experienced recurrences. Two had local recurrence, two had distant recurrence, and one had both.
There were nine patients who had radiotherapy post-exenteration, and six subsequently relapsed. Four patients had local recurrence, one developed distant metastasis, and one had both types of recurrence.
The specific anatomical sites of relapse are described in Table 3, and Figure 4 illustrates this by surgical procedure. The most common location of locoregional recurrence out of 61 patients with recurrences was the pelvic side wall 29.5%, followed by central recurrence 24.6%, anterior 9.8%, inguinal 6.6%, and posterior pelvis 6.6%. After anterior exenteration, recurrence was predominantly central at 36%; however, after a posterior and after a total exenteration, it was predominantly pelvic side wall at 41.7% and 33.3%, respectively.
Anatomical Sites of Relapse Following Pelvic Exenteration
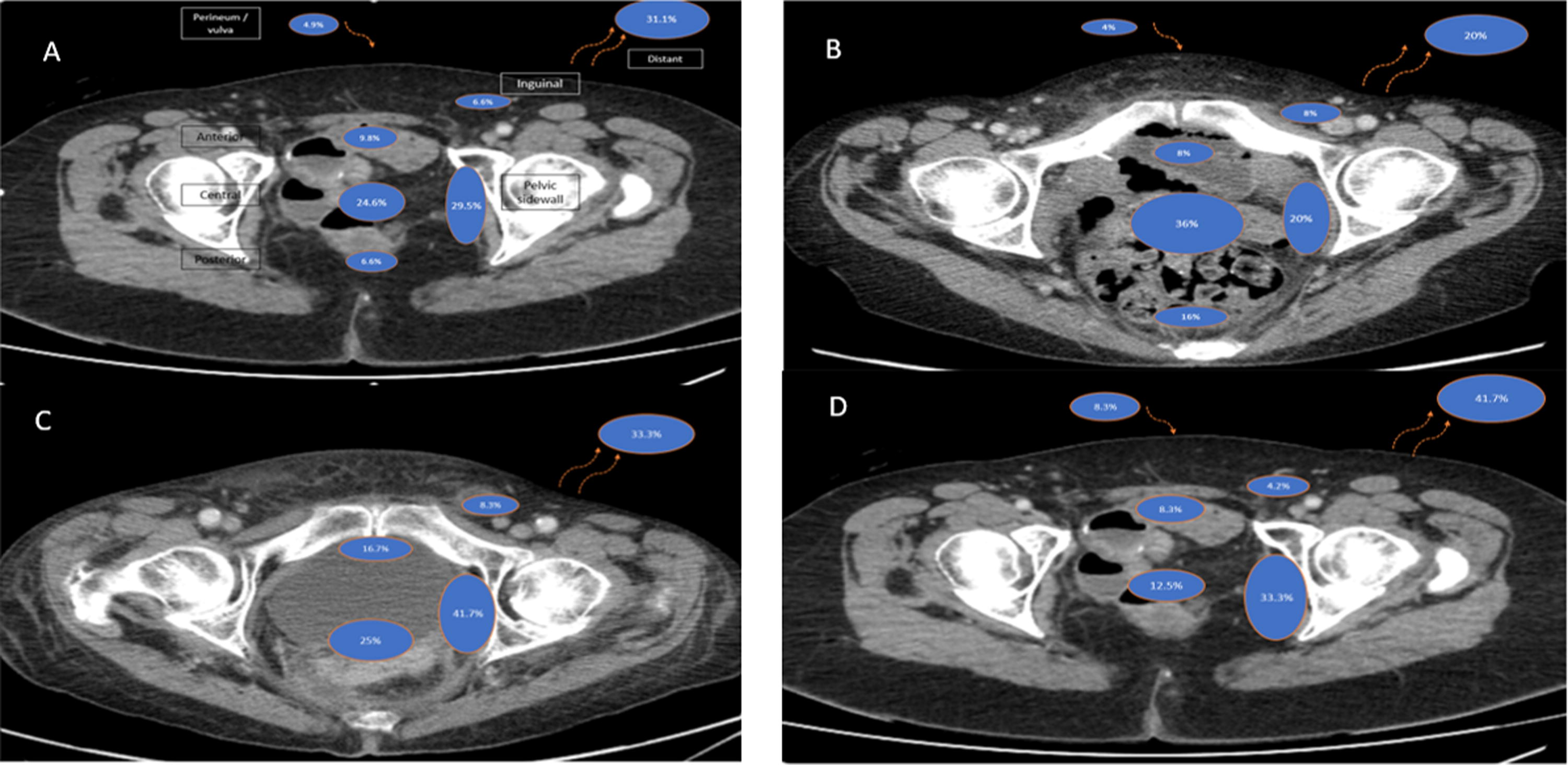
Patterns of relapse following pelvic exenteration for recurrent gynecological malignancy regardless of exenteration type
Surgical margin status
An involved margin was twice as common in the relapsed group than in those without relapse. Recurrence occurred in 52.6% of patients with a clear margin, 56.8% with a close margin, and 75% with an involved margin. Locoregional recurrence occurred in only 11 patients (28.9%) with a clear margin, 8 (21%) patients had distant disease and 5 (13%) had both. In contrast, of the 39 patients with involved or close margins who relapsed after pelvic exenteration, 31 (79.5%) had locoregional relapse only, 2 (5.1%) had distant relapse only, and 7 (17.9%) had both. Eleven patients developed distant metastases only, of which the margin was clear in 7 (63.6%), close in 3 (27.3%), and unrecorded in one case (9.1%).
Of 11 patients who experienced distant recurrence, 7 (63.6%) had lymphovascular space invasion.
Relationship of recurrence to pre-operative disease
Imaging of both pre-exenteration disease and postoperative relapse was available for 21 patients, 15 with close or involved margins and six with negative margins. Recurrence occurred at the same site, or within 5 mm, as the preoperative tumor in 65% of cases. The mean overlap volume was 14.3 cm3 (0–138.5 cm3), demonstrating that the majority of relapses occur within close proximity to the initial site of disease.
Stereotactic radiotherapy planning study
The scans from 10 consecutive patients with a close or positive margin were used to include a range of surgical procedures. Six patients had cervical cancer, three endometrial cancer, and one Bartholin’s gland cancer. Six patients had a total pelvic exenteration, and four had anterior pelvic exenteration. Twenty-nine plans were developed, with one patient having only two plans as the pre-exenteration tumor volume was too large for stereotactic planning of Planning target volume (PTV)_Total. Six cases had a planning target volume involving the sciatic nerve; the rectum was an organ at risk in the four cases with anterior exenteration.
Mean planning target volumes were 70.8 cm3 for PTV_margin, 155.8 cm3 PTV_high-risk, and 214.9 cm3 PTV_total. Target volume and stereotactic radiotherapy plan characteristics are summarized in Supplementary Table S2. All 29 plans met the planning objective to deliver the prescribed dose to at least 95% of the PTV.
All organs at risk mandatory dose constraints were met in 90% plans for PTV_margin, in 70% for PTV_Highrisk-risk and 50% for PTV_total. All optimal organs at risk dose constraints were met in 70% PTV_margin, 50% PTV_high risk, and 30% PTV_Total (Table 4).
Number of Plans Meeting the Mandatory and Optimal Organs at Risk Dose Constraints Without Any Compromise to PTV Coverage (CTV_Margin was the site of likely positive or close Margin. CTV_Highrisk included the lateral margin of all pre-operative diseases and the high-risk region bordering the positive/close margin.CTV_Total combined CTV_Highrisk with the site of all pre-perative disease)
The dosimetric study demonstrated that defining the target volume is key for delivering stereotactic radiotherapy post-pelvic exenteration. Using the smaller target volumes allows for better plan characteristics but with greater uncertainty of covering the true margin. Stereotactic radiotherapy was also feasible for the majority of cases using a larger target volume encompassing the high-risk area bordering the whole preoperative tumor margin. In clinical practice, a small compromise to target volume coverage would also be accepted to enable further compliance to organ at-risk doses.
Discussion
The first objective of our study was to describe the patterns of relapse following exenterative surgery for recurrent gynecological cancer. Our study showed that locoregional relapse was the dominant pattern of recurrence following pelvic exenteration for centrally recurrent gynecological cancers. It also showed a significantly higher 5-year locoregional control for those patients in the negative margin group compared with those patients in the involved/close margin group, thus highlighting the particular need for an effective postoperative therapeutic approach for patients with involved and close margins. Re-irradiation with stereotactic radiotherapy was dosimetrically feasible for the majority of patients. Importantly, it was also noted that despite negative or close margins (<5 mm), patients post-exenteration are at risk of loco-regional relapse; there is the need for regular post-exenteration surveillance and re-irradiation in these patients may need to be considered, although this is not evidence-based, we routinely scan every six months for the first three years post-exenteration.
Our study group had characteristics that are associated with an increased risk of relapse post-exenteration—in particular (1) short disease-free interval (median 8.1 months from completion of primary treatment or completion of radiotherapy for first relapse to the recurrence treated with pelvic exenteration), (2) large recurrence—31 tumors were larger than 5 cm in maximum diameter in the exenteration specimen (30.7% cases) and (3) close or involved margins in 60.4% of patients. We used a strict definition of close margin (up to 5 mm), in contrast to other reports. Up to 61 (60.4%) patients had at least one risk factor for recurrence, 24 (23.8%) patients had two, 13 (12.9%) had three risk factors, and 3 (2.9%) patients had more than three risk factors.
In comparison, the published 20-year experience of Sharma et al. of 48 women treated with exenteration for recurrent gynecological malignancy, where the majority received prior radiation therapy, also demonstrated a 60% post-exenteration recurrence rate. However, the median overall survival was 35 months, and progression-free survival was 32 months, which was longer than in our study group. This may reflect different patient characteristics and case selection compared with our study. In their report, eight patients received intraoperative radiation, although these patients had worse outcomes (median survival 11.3 vs. 35 months, p = 0.003). These high-risk patients who received intraoperative radiotherapy had outcomes similar to our study. 8
Other series have recognized the importance of R0 resection in achieving loco-regional control. In the PelvEx Collaborative study, where 1293 patients were identified who underwent exenterative surgery (in most cases for rectal cancer), multivariable analysis showed R0 resection was the main factor associated with long-term survival. The 3-year overall survival rate for R0 resection of the 525 patients with gynecological cancer was similar between cancer types: 48% for endometrial, 40.6% for ovarian, 49.4% cervical, and 43.8% vaginal cancer. 27
Most series describing relapse post-exenteration are small.28–30 For example, case series on post-pelvic exenteration recurrence in patients with cervical cancer treated with exenteration only for central pelvic disease (n = 14) and combined surgery with radiotherapy for pelvic side-wall recurrence (n = 18) have been described. The results showed that 5–58 months after treatment, seven patients (50%) with central recurrence and 11 patients (61%) with pelvic sidewall recurrences experienced further recurrences.
The site of recurrent tumor progression (local versus distant) was similar in both groups. Only tumor size more than 5 cm was identified as a key factor influencing survival. 29 Compared with these series, our data comprise a larger cohort focused only on central pelvic recurrence. Despite this, the similar outcomes reflect the influence of many variables such as patient and tumor heterogeneity, tumor biology, and different treatments.
The second objective of this study was to determine the feasibility of delivering stereotactic radiotherapy for close or positive surgical margins. There is currently no proven treatment strategy for patients with close and involved margins after pelvic exenteration following previous radiotherapy treatment. Our study has also shown that it is feasible to deliver stereotactic radiotherapy after pelvic exenteration. The dose to organs at risk was comparable when treating just the presumed site of a positive margin compared with a larger volume bordering the whole initial site of disease, which may be a more effective method for defining the at-risk region. We have previously reported our initial clinical experience using stereotactic radiotherapy for four patients with positive margins after pelvic exenteration in patients with previously treated radiotherapy. 31 There were no grade 3 or 4 toxicities related to treatment and no recurrence in the irradiated site. After a median follow-up of 14.3 months, one patient died from multifocal recurrence, one was alive with pelvic recurrence, and the other two were alive with no evidence of disease.
Andikyan et al. aimed to reduce post-exenteration recurrence with additional platinum-based doublet chemotherapy. 32 Of 42 patients treated with pelvic exenteration over a six-year period, chemotherapy was recommended to 11 (26%) based on surgeon’s discretion and/or presence of high-risk features: positive margins, positive lymph nodes, and/or lymphovascular space invasion of the 42 patients treated. With a median follow-up of 25 months (range 6–56 months), the 3-year progression-free survival and overall survival of the eight patients who received chemotherapy were 58% and 54%, respectively. Although small numbers, the data provide some evidence for considering additional treatment to improve survival.
Our study showed that 7 out of 11 (63.6%) patients with distant recurrence had lymphovascular space invasion. Therefore, post-exenteration chemotherapy might be beneficial in these patients, although this requires further investigation.
The limitations of our study include the retrospective cohort analyses on a heterogeneous group of patients who underwent exenteration in a single cancer center. These limitations are inherent in all reports on pelvic exenteration for recurrent gynecological and non-gynecological cancer because of the necessary high level of selection of cases. Another limitation of this study is the limited data on the cumulative optimal dose constraints for re-irradiation in the pelvis. For this feasibility study, prior treatment with 50 Gy (EQD2) was assumed since most patients received 45 Gy in 25 fractions to the pelvis, followed by brachytherapy, which contributes approximately 1–1.5 Gy per fraction to the pelvic sidewall. However, in clinical practice, it will be important to consider the previous treatment details as dose constraints may need to be adjusted accordingly. In this study, we did not differentiate between small and large bowels for the planning study. Following pelvic exenteration, it is usually a small bowel that moves into the pelvic space following surgery. The purpose of pelvic floor reconstruction is to keep the small bowel out of the pelvis (empty pelvis syndrome) and avoid enterocutaneous or other enteric fistulae. The colostomy is usually out of the pelvis, and the cecum is lateral and at or above the pelvic brim. There is insufficient information about the tolerances for different functional areas of the bowel with re-irradiation, and therefore, it is safest to use small bowel tolerances for all bowel in this setting. Future studies should collect the cumulative doses to normal tissues in order to be able to further refine these dose constraints.
The dosimetric study showed that the addition of stereotactic radiotherapy was feasible, but there is uncertainty about target volume definition requiring specific expertise in this rare scenario. A simple, reliable method to improve the localization of surgical resection beds considered at risk of a close margin would be the insertion of radio-opaque fiducial markers during surgery in a similar approach to that used for patients with breast cancer.33,34 Likewise, marking of the fresh specimen is useful for the pathologist to determine the location of positive margins.
Pelvic exenteration, which can be the only potentially curative option for patients with recurrent disease post-radiotherapy, requires multidisciplinary specialist expertise. A larger prospective multicenter collection of data is required. In the future, serum biomarkers and multimodality functional imaging may further aid treatment selection. The combination of stereotactic radiotherapy post-exenteration with systemic therapies, including immunotherapy, should be further evaluated.
Conclusion
In summary, our study showed that locoregional relapse was the most common recurrence pattern following pelvic exenteration for centrally recurrent gynecological cancer post-radiotherapy. There was a significantly higher risk of pelvic relapse with close or involved margins, and the relapse was frequently at the same site as the initial disease. Two-thirds of patients with a histological margin of 5 mm or more will live beyond five years, and most of these will be cured. Stereotactic radiotherapy offers a potentially effective option to improve outcomes by eradicating residual microscopic disease. Prospective clinical studies are needed to ascertain toxicity and determine efficacy.
Footnotes
Authors’ Contributions
N.G.—collecting and analyzing data and writing the original draft of the article. G.I.—collecting data, analyzing data, and writing the first draft. D.P.B. and A.T.—conceptualization, defining methodology, and reviewing and editing the final draft equal. N.S. and O.H.—review and editing equal. X.D.—assisting in radiotherapy planning. A.A. and K.V.—providing histopathologist input equally.
Author Disclosure Statement
No conflict of interest.
Funding Information
No funding was received for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
