Abstract
Abstract
Introduction
Case
This is the case of a 34-year-old gravida 1, para 0 with a CP, who was managed with intramuscular (IM) and intra-sac methotrexate administration followed by hysteroscopic removal. Prior to her presentation at New York Methodist Hospital, she underwent laparoscopic lysis of adhesions and fulguration of endometriotic implants. In January 2011, ∼4 months after her initial laparoscopic surgery, the patient was found to be pregnant. On day 27 of her menstrual cycle, beta human chorionic gonadotropin () was found to be 86. At 5 weeks of gestation, with a βhCG >1500 milli-international units (mIU)/mL, no intrauterine pregnancy was noted on ultrasound examination. At that time, the patient began to experience bleeding and cramping, and repeat ultrasound examination showed a gestational sac with a cervical implantation site with positive cardiac activity (Figs. 1 and 2). The patient elected to receive IM methotrexate at 6 and 7 weeks of gestation with continued increasing size of the sac noted in addition to continued cardiac activity. Intra-amniotic instillation of methotrexate was then initiated at 8 weeks of gestation with prompt cessation of cardiac activity. βhCG at this time was 3500 mIU/mL. The patient was then monitored without passage of the pregnancy tissue, and a βhCG value that plateaued at 2400 mIU/mL. At approximately 10 weeks of gestation (2 weeks after the intra-sac administration of methotrexate), the patient underwent hysteroscopic removal of the cervical ectopic pregnancy without complication. Standard Karl Storz hysteroscopic equipment was used, including Hamou Endomat Pump manager, bipolar resectoscope 26 Fr with high frequency AUTOCON® 400, and continuous flow examination sheath. On hysteroscopic examination, the products of conception (POCs) were identified within an enlarged area of concavity within the right portion of the cervical canal (2.48 cm from the ectocervix). The products of conception were removed with the resectoscope without complication (Fig. 3). All blood vessels were directly coagulated as the POC were removed. On completion of the surgery, examination of the uterine cavity revealed no further POCs (Fig. 4). Estimated blood loss for the procedure was 30–50 cc, and the patient was monitored for 4 hours in the recovery room and subsequently discharged home. βhCG levels normalized (<5 mIU/mL) within 1 week after resection. The patient returned 3 months postoperatively and underwent ovulation induction with clomiphene citrate followed by intrauterine insemination. She delivered that pregnancy at term after an uneventful prenatal course.
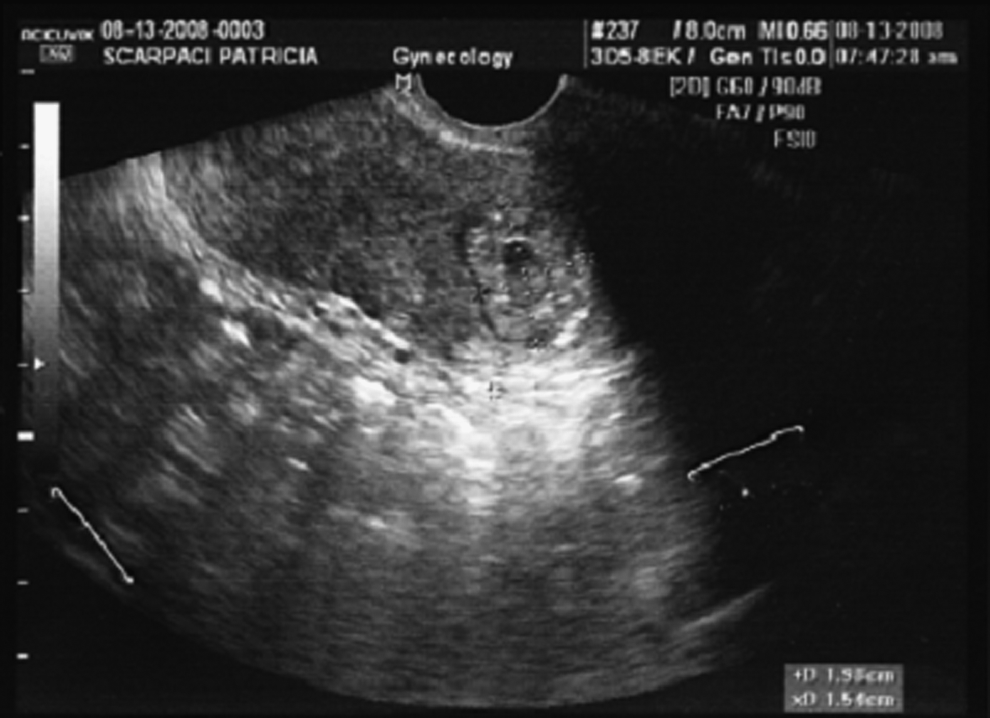
Sonographic evidence of cervical pregnancy within the cervical canal.
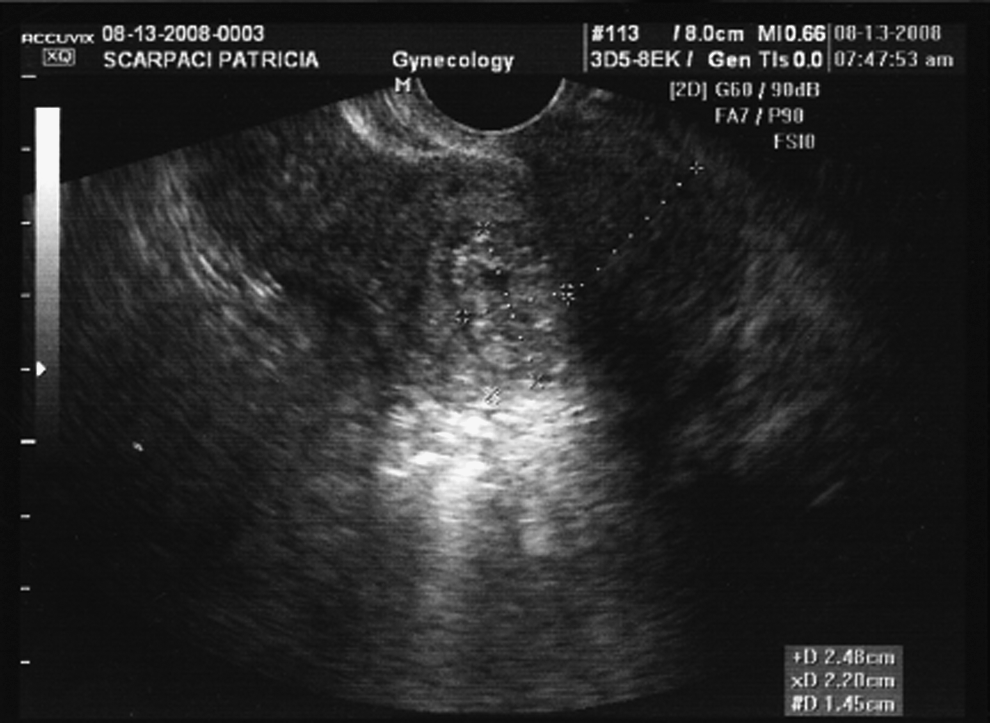
Sonographic image showing distance of cervical pregnancy from ectocervix.
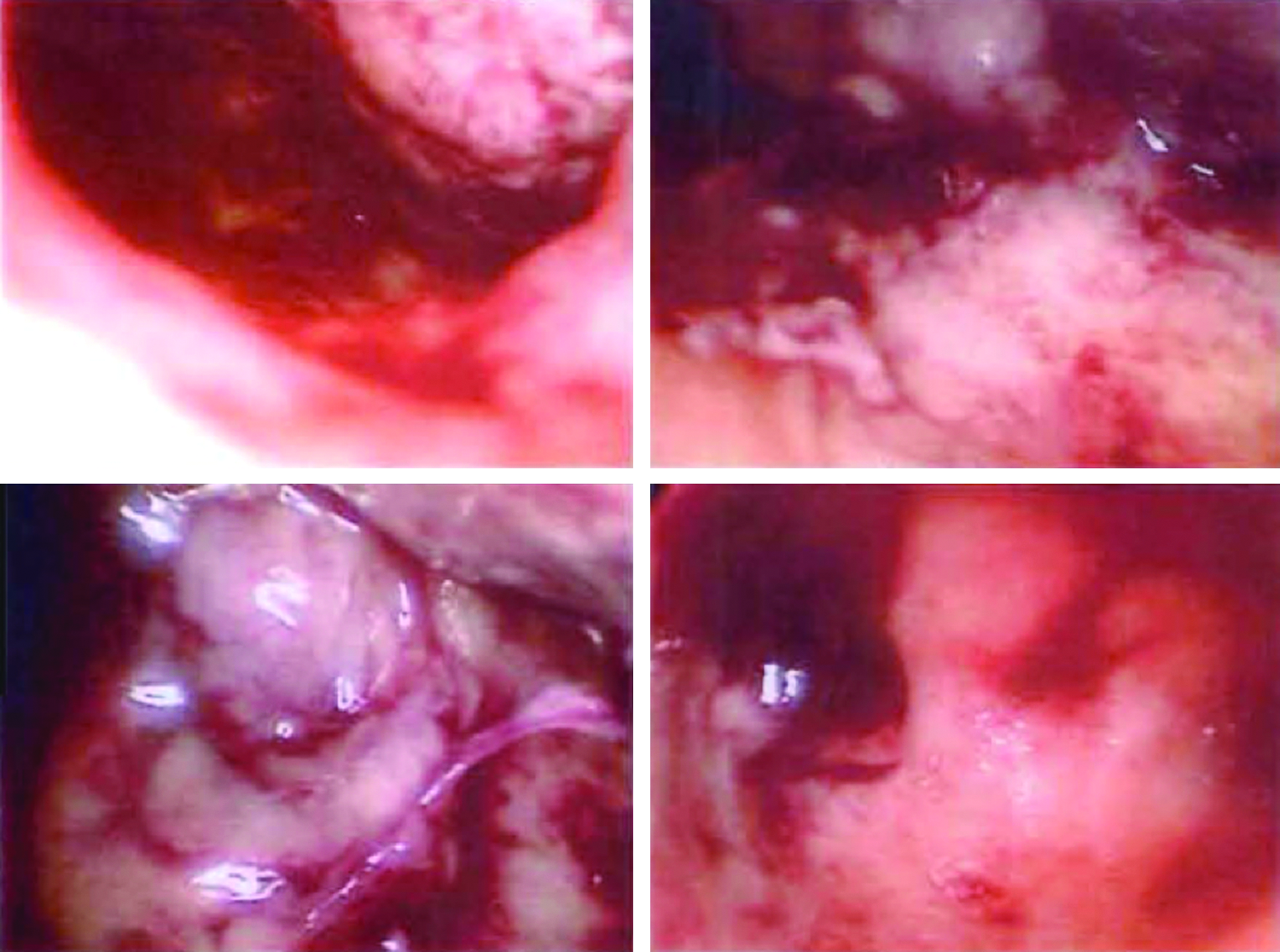
Cervical pregnancy within the right cervical canal.
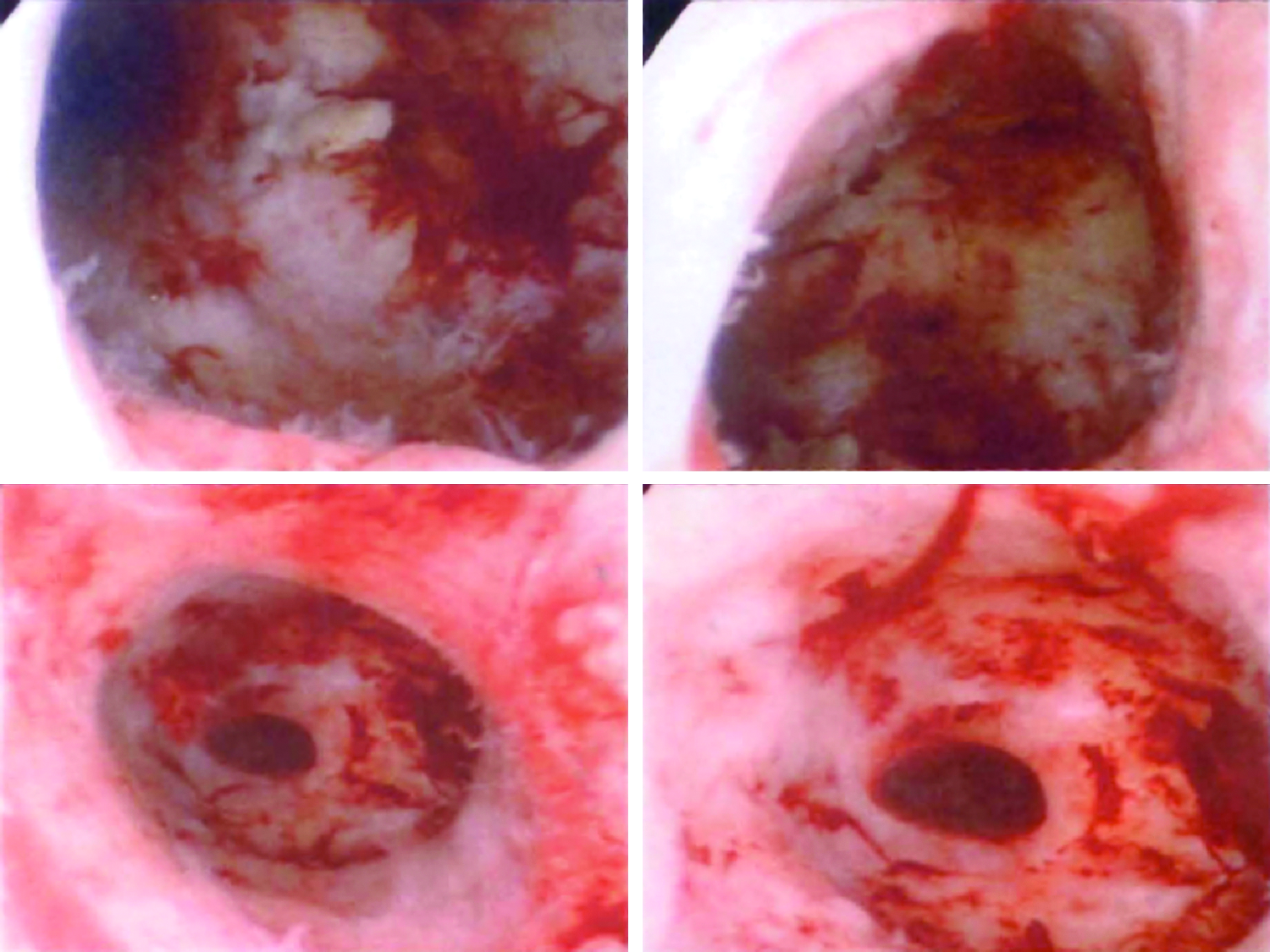
Empty uterine cavity after resection of cervical pregnancy.
Discussion
Cervical implantation is a rare event, typically accounting for <1% of all ectopic pregnancies; however, the consequences of a CP can be devastating. Historically, CPs were difficult to diagnose and were identified at later gestational ages than were tubal ectopic pregnancies. Because of the relatively large gestational sac and the highly vascular nature of the cervical tissue, treatment of CP was often associated with massive hemorrhage from the implantation site, frequently requiring hysterectomy.
More recently, with the advent of sensitive diagnostic methods, the use of conservative and fertility-sparing methods for the management of CP has been described. Data regarding the use of hysteroscopy in CP are limited, but there are case reports that describe this method. In 1992, Roussis et al. described the first case in which hysteroscopy was used to visualize a CP and guide removal by suction curettage after sonography revealed incomplete resolution despite multiple doses of systemic methotrexate. 1 Four years later, Ash and Farrell published the first case using operative hysteroscopy, without prior chemotherapy, to completely resect a viable CP. 2 In 2006, Matteo et al. also used hysteroscopy to successfully resect a CP – in this case, after two cycles of methotrexate treatment – and found that hemostasis could be achieved via direct hysteroscopic coagulation of bleeding vessels. 3 In both cases, operative time was ∼35 minutes, there was minimal bleeding, and there was rapid resolution of the βhCG post-resection.
In 2004, Kung et al. performed laparoscopy-assisted uterine artery ligation in conjunction with hysteroscopic endocervical resection of a cervical ectopic pregnancy in 6 patients, eliminating the need for adjuvant chemotherapy prior to hysteroscopy. 4 Normal menstruation resumed in a mean period of 2 months, and 1 patient achieved spontaneous pregnancy 14 months postoperatively. Alhough this technique effectively controlled bleeding and preserved the uterus, it has been criticized as being too invasive. 3
Conclusions
When managing any cervical ectopic pregnancy, the major goals are to minimize hemorrhage and preserve future fertility. Jozwiak et al. in their 2003 report demonstrated a successful live birth in a subsequent pregnancy after hysteroscopic removal of a CP. 5 In this case, it is also demonstrated that hysteroscopic resection is a potentially safe and effective option for fertility-sparing management after failure of more traditional measures such as IM methotrexate. Operative hysteroscopy allows direct visualization of a CP, thereby enabling the surgeon to resect the ectopic pregnancy and simultaneously ablate bleeding vessels if necessary. Importantly, hysteroscopic removal in this case preserved fertility and the integrity of the cervix such that, 3 months later, the patient was able to achieve a clinical pregnancy, which was carried to term without any complications or cervical insufficiency. This particular case also highlighted the potential benefit of intragestational sac methotrexate for cessation of growth of the gestational sac; after failure of IM methotrexate. The goal of treatment as highlighted in this case is safe removal of the abnormal pregnancy with preservation of fertility.
Footnotes
Disclosure Statement
No competing financial conflicts exist.
